Simultaneous Determination of 18 Polycyclic Aromatic Hydrocarbons in Daily Foods (Hanoi Metropolitan Area) by Gas Chromatography–Tandem Mass Spectrometry
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Equipment
2.3. Sample Collection
2.4. Sample Preparation
2.5. Optimizing Solvent Evaporating Process
2.6. Method Validation
3. Results and Discussion
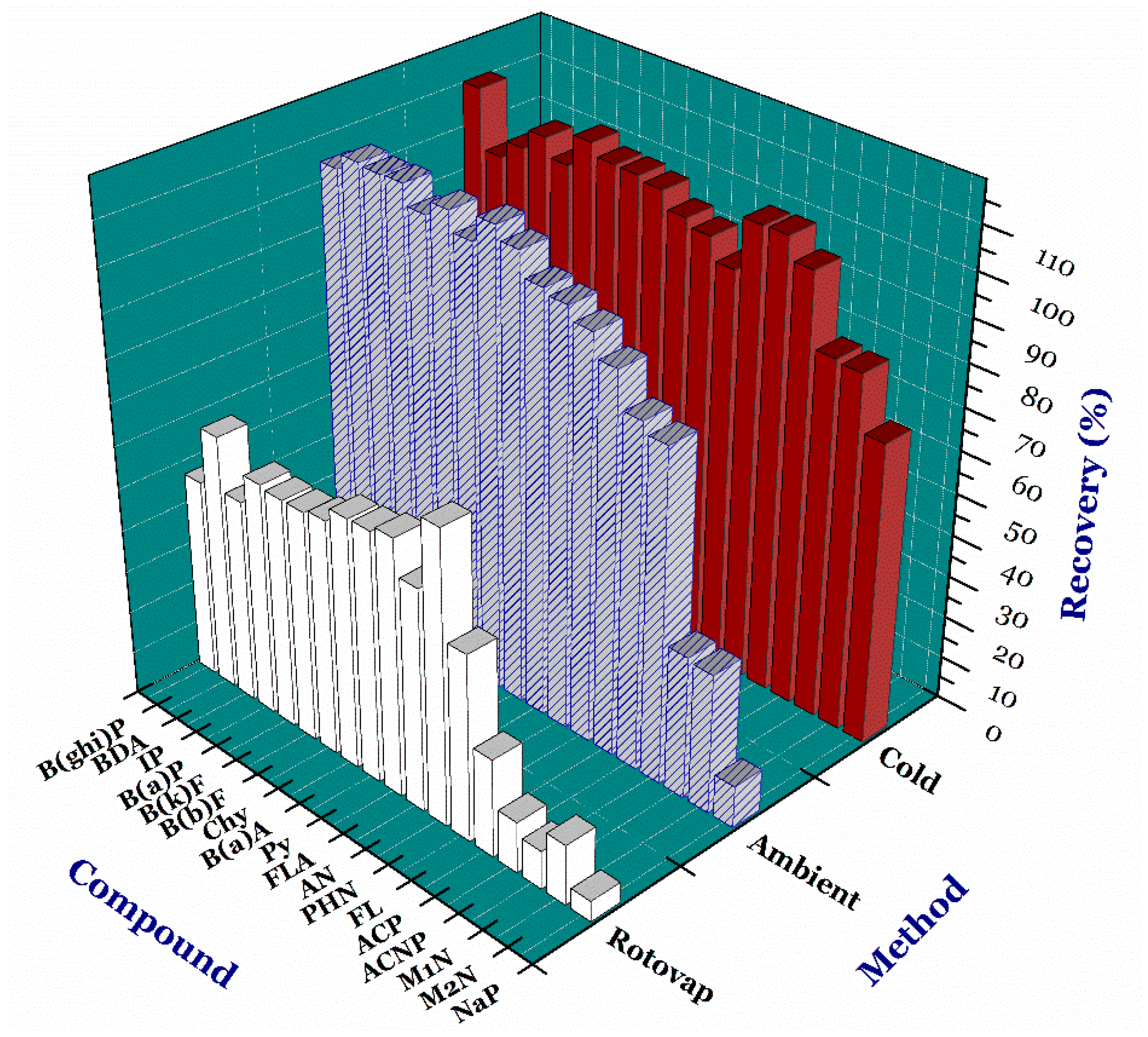
3.1. Optimizing Solvent Evaporating Process
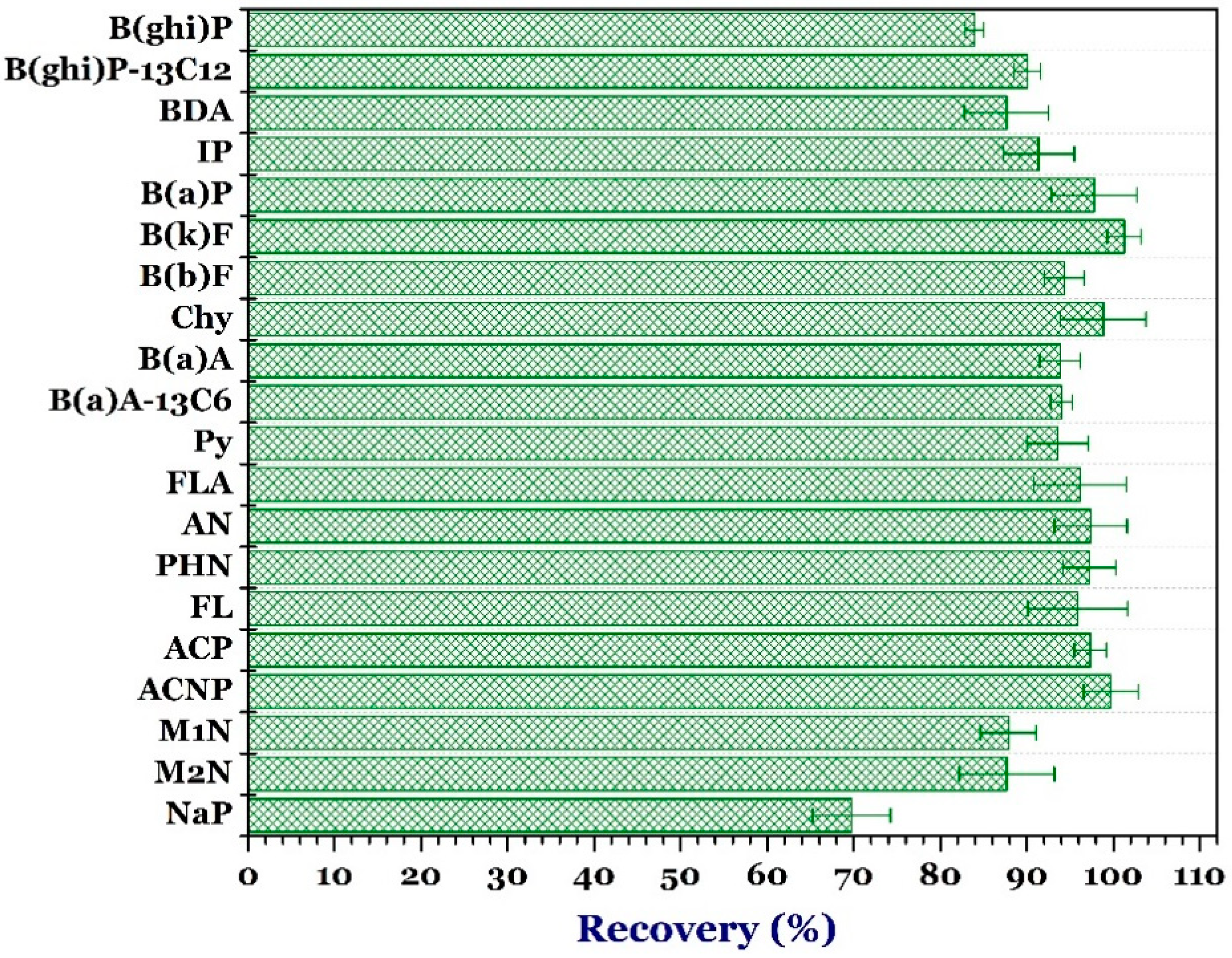
3.2. Method Validation
3.3. Recovery
3.4. Levels of PAHs in the Target Samples
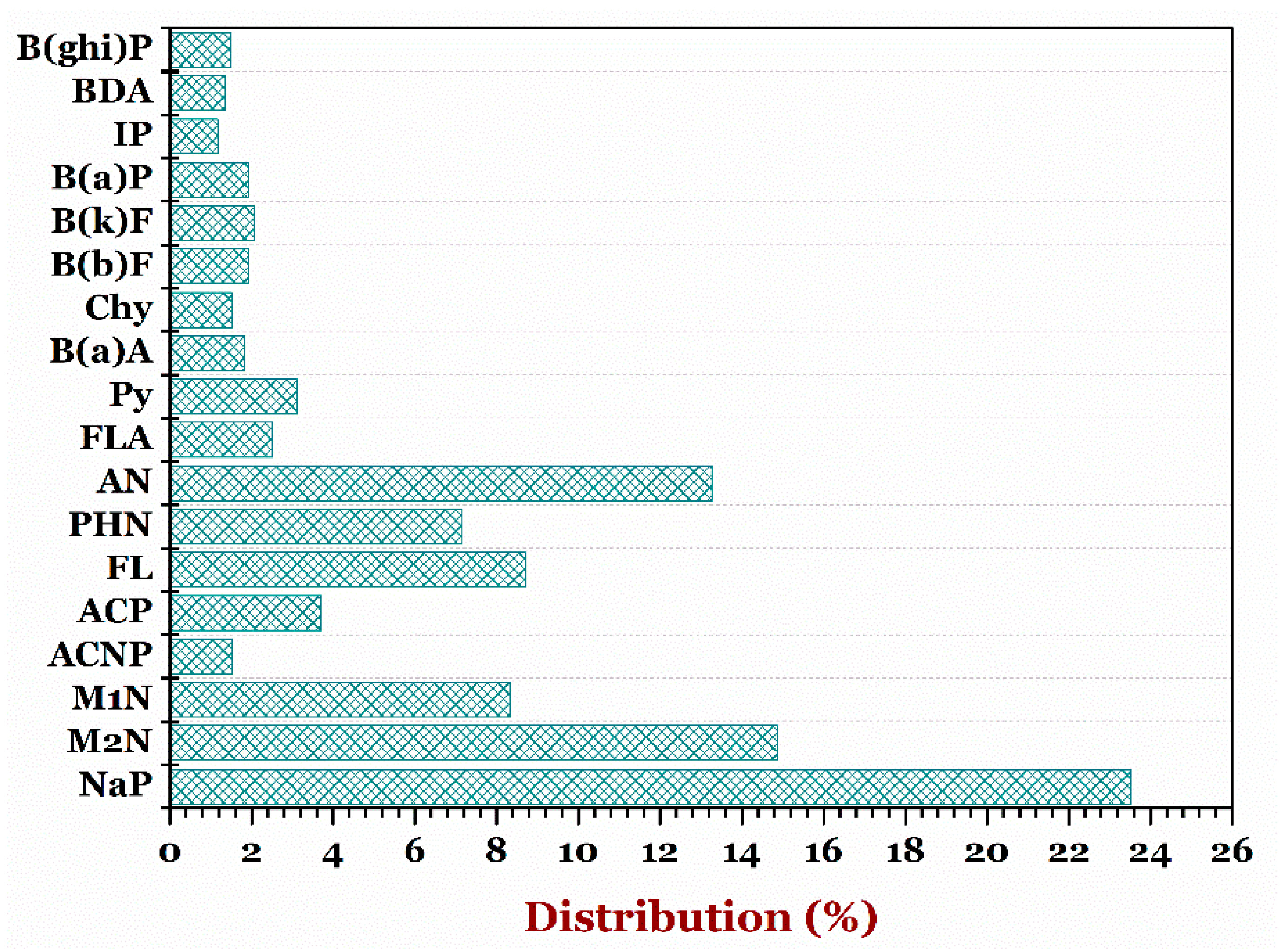
3.4.1. Instant Noodle
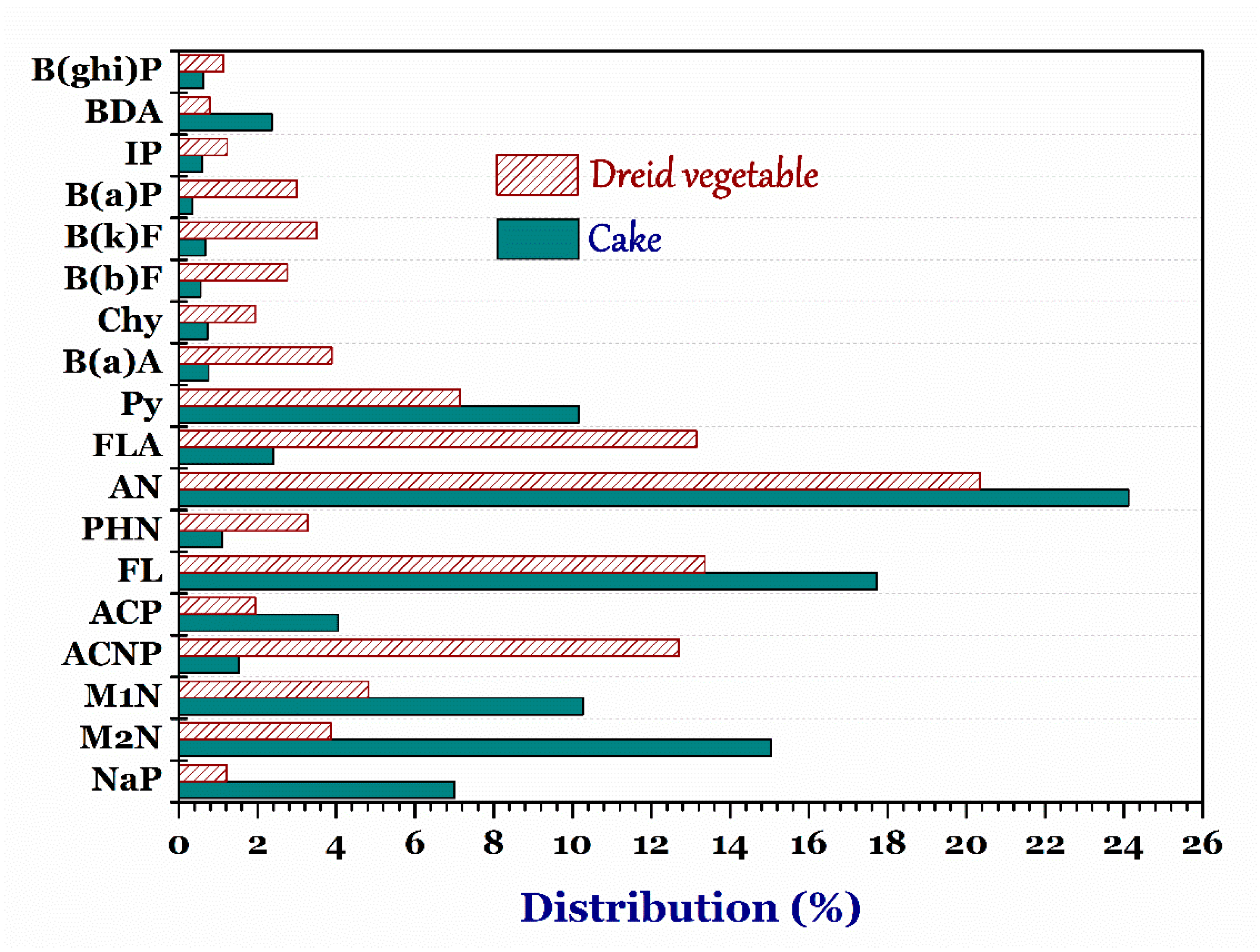
3.4.2. Cake and Dried Vegetable
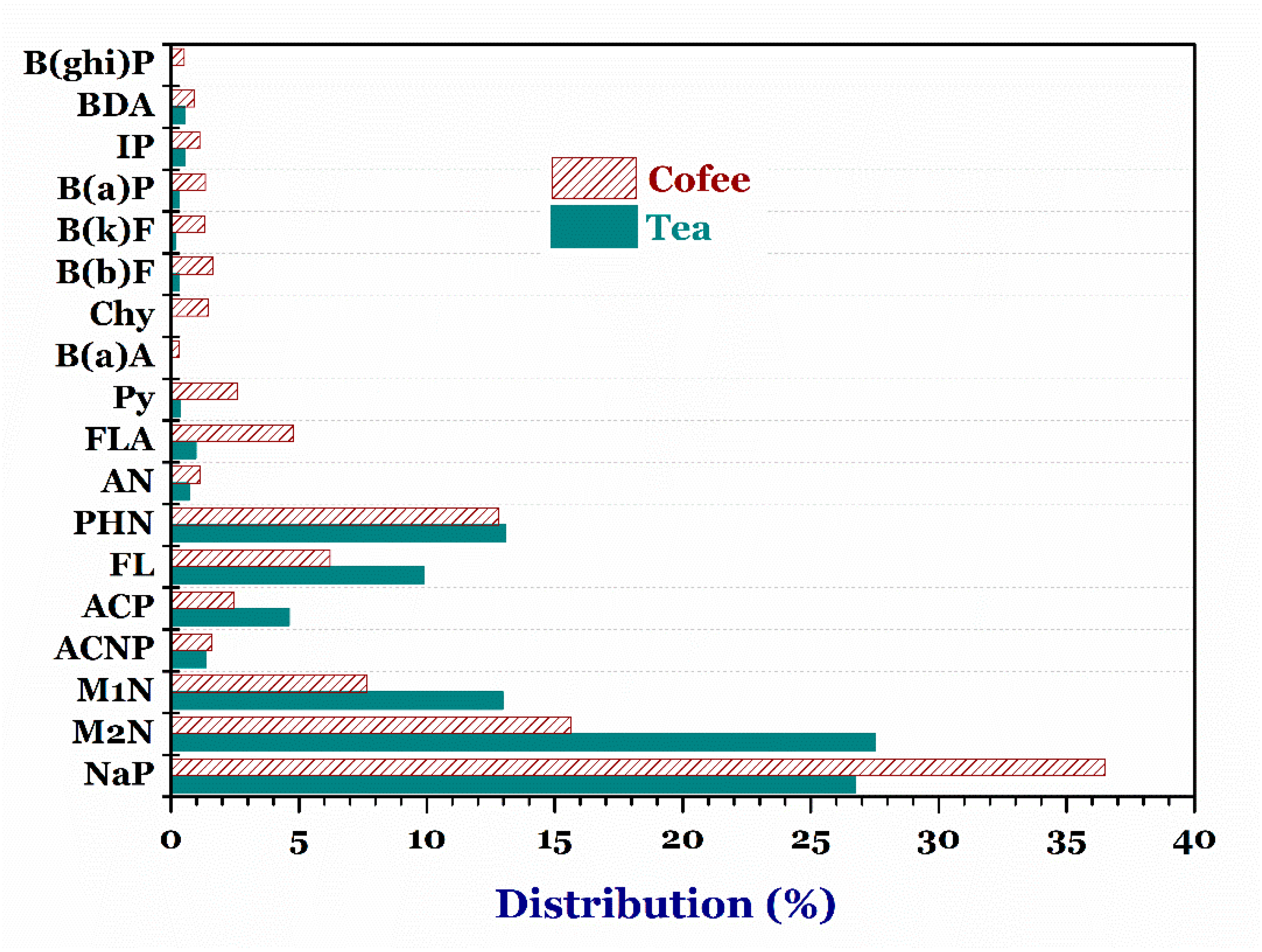
3.4.3. Tea and Coffee
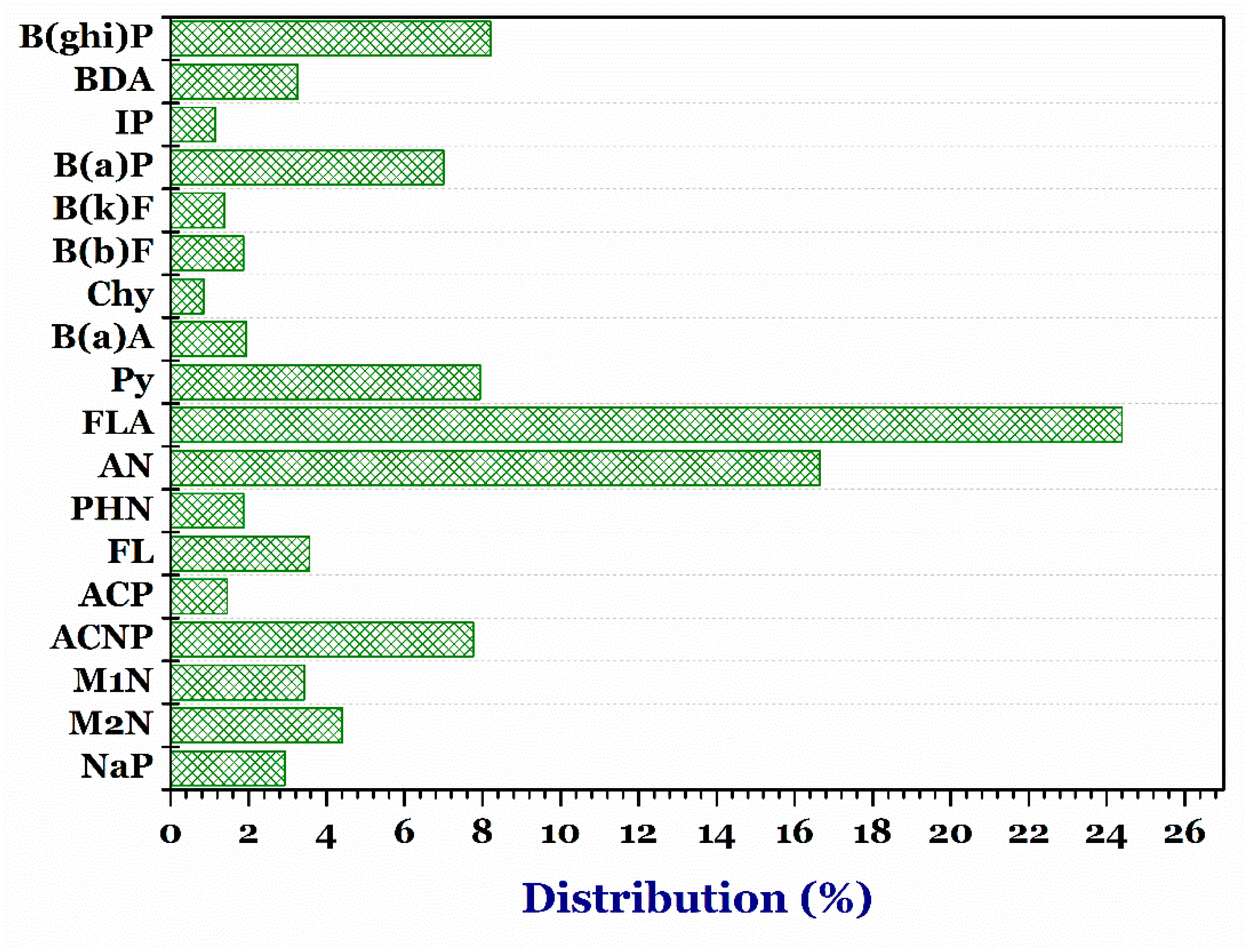
3.4.4. Grilled Meat
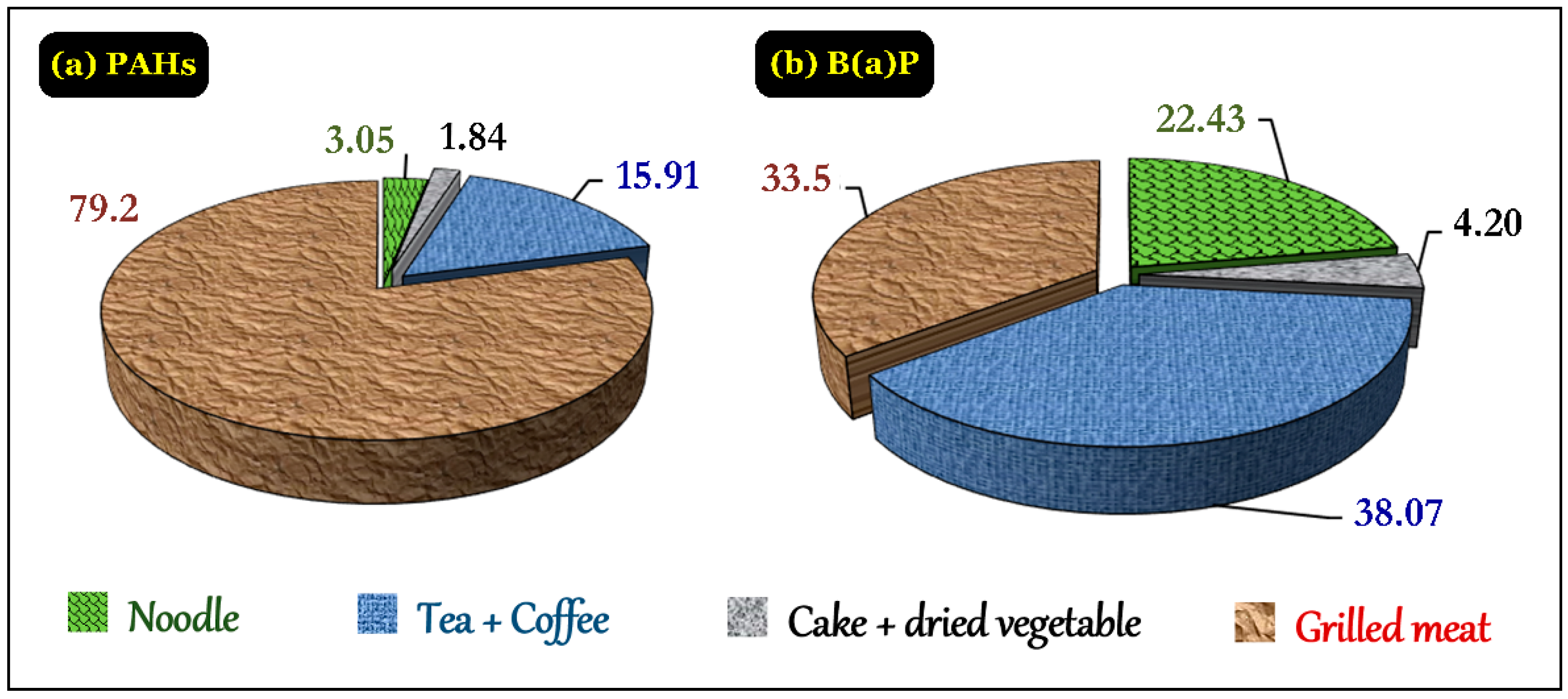
3.4.5. PAHs in Daily Foods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jägerstad, M.; Skog, K. Genotoxicity of heat-processed foods. Mutat. Res. 2005, 574, 156–172. [Google Scholar] [CrossRef] [PubMed]
- Sugimura, T.; Wakabayashi, K.; Nakagama, H.; Nagao, M. Heterocyclic amines: Mutagens/carcinogens produced during cooking of meat and fish. Cancer Sci. 2004, 95, 290–299. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Phillips, D.H. Polycyclic aromatic hydrocarbons in the diet. Mutat. Res. 1999, 443, 139–147. [Google Scholar] [CrossRef]
- Kim, K.-H.; Jahan, S.A.; Kabir, E.; Brown, R.J.C. A review of airborne polycyclic aromatic hydrocarbons (PAHs) and their human health effects. Environ. Int. 2013, 60, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Senthilkumar, K.; Alomirah, H.; Moon, H.-B.; Minh, T.B.; Mohd, M.A.; Nakata, H.; Kannan, K. Concentrations and Profiles of Urinary Polycyclic Aromatic Hydrocarbon Metabolites (OH-PAHs) in Several Asian Countries. Environ. Sci. Technol. 2013, 47, 2932–2938. [Google Scholar] [CrossRef] [PubMed]
- Jánská, M.; Hajšlová, J.; Tomaniová, M.; Kocourek, V.; Vávrová, M. Polycyclic Aromatic Hydrocarbons in Fruits and Vegetables Grown in the Czech Republic. Bull. Environ. Contam. Toxicol. 2006, 77, 492–499. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.; Tu, Y.; Zhu, L. Concentrations and health risk of polycyclic aromatic hydrocarbons in tea. Food Chem. Toxicol. 2005, 43, 41–48. [Google Scholar] [CrossRef]
- IARC. Certain Polycyclic Aromatic Hydrocarbons and Heterocyclic Compounds; IARC Monographs on the Evaluation of the Carcinogenic Risks to Humans (Book 3); World Health Organization: Geneva, Switzerland, 1973; Volume 3, p. 271. [Google Scholar]
- Schoket, B. DNA damage in humans exposed to environmental and dietary polycyclic aromatic hydrocarbons. Mutat. Res. 1999, 424, 143–153. [Google Scholar] [CrossRef]
- Lightfoot, T.J.; Coxhead, J.M.; Cupid, B.C.; Nicholson, S.; Garner, R.C. Analysis of DNA adducts by accelerator mass spectrometry in human breast tissue after administration of 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine and benzo[a]pyrene. Mutat. Res. 2000, 472, 119–127. [Google Scholar] [CrossRef]
- Rodriguez, L.V.; Dunsford, H.A.; Steinberg, M.; Chaloupka, K.K.; Zhu, L.; Safe, S.; Womack, J.E.; Goldstein, L.S. Carcinogenicity of benzo[a]pyrene and manufactured gas plant residues in infant mice. Carcinogenesis 1997, 18, 127–135. [Google Scholar] [CrossRef] [Green Version]
- Reinik, M.; Tamme, T.; Roasto, M.; Juhkam, K.; Tenno, T.; Kiis, A. Polycyclic aromatic hydrocarbons (PAHs) in meat products and estimated PAH intake by children and the general population in Estonia. Food Addit. Contam. 2007, 24, 429–437. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ravindra, K.; Sokhi, R.; Van Grieken, R. Atmospheric polycyclic aromatic hydrocarbons: Source attribution, emission factors and regulation. Atmos. Environ. 2008, 42, 2895–2921. [Google Scholar] [CrossRef] [Green Version]
- Raiyani, C.V.; Jani, J.P.; Desai, N.M.; Shah, S.H.; Shah, P.G.; Kashyap, S.K. Assessment of indoor exposure to polycyclic aromatic hydrocarbons for urban poor using various types of cooking fuels. Bull. Environ. Contam. Toxicol. 1993, 50, 757–763. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.S.; Ashley, D.L.; Watson, C.H. Determination of 10 carcinogenic polycyclic aromatic hydrocarbons in mainstream cigarette smoke. J. Agric. Food Chem. 2007, 55, 5966–5973. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.W. Determination of polycyclic aromatic hydrocarbons in water by solid-phase microextraction and liquid chromatography. Anal. Sci. 2004, 20, 1383–1388. [Google Scholar] [CrossRef] [PubMed]
- King, A.J.; Readman, J.W.; Zhou, J.L. Determination of polycyclic aromatic hydrocarbons in water by solid-phase microextraction–gas chromatography–mass spectrometry. Anal. Chim. Acta 2004, 523, 259–267. [Google Scholar] [CrossRef]
- Tao, S.; Jiao, X.C.; Chen, S.H.; Liu, W.X.; Coveney, R.M.; Zhu, L.Z.; Luo, Y.M. Accumulation and distribution of polycyclic aromatic hydrocarbons in rice (Oryza sativa). Environ. Pollut. 2006, 140, 406–415. [Google Scholar] [CrossRef]
- Dennis, M.J.; Massey, R.C.; Cripps, G.; Venn, I.; Howarth, N.; Lee, G. Factors affecting the polycyclic aromatic hydrocarbon content of cereals, fats and other food products. Food Addit. Contam. 1991, 8, 517–530. [Google Scholar] [CrossRef]
- Voutsa, D.; Samara, C. Dietary intake of trace elements and polycyclic aromatic hydrocarbons via vegetables grown in an industrial Greek area. Sci. Total Environ. 1998, 218, 203–216. [Google Scholar] [CrossRef]
- Kazerouni, N.; Sinha, R.; Hsu, C.-H.; Greenberg, A.; Rothman, N. Analysis of 200 food items for benzo[a]pyrene and estimation of its intake in an epidemiologic study. Food Chem. Toxicol. 2001, 39, 423–436. [Google Scholar] [CrossRef]
- Lin, D.; Zhu, L.; He, W.; Tu, Y. Tea Plant Uptake and Translocation of Polycyclic Aromatic Hydrocarbons from Water and around Air. J. Agric. Food Chem. 2006, 54, 3658–3662. [Google Scholar] [CrossRef] [PubMed]
- Houessou, J.K.; Maloug, S.; Leveque, A.-S.; Delteil, C.; Heyd, B.; Camel, V. Effect of Roasting Conditions on the Polycyclic Aromatic Hydrocarbon Content in Ground Arabica Coffee and Coffee Brew. J. Agric. Food Chem. 2007, 55, 9719–9726. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Shin, H.-S. Determination of polycyclic aromatic hydrocarbons in commercial roasted coffee beans. Food Sci. Biotechnol. 2010, 19, 1435–1440. [Google Scholar] [CrossRef]
- Lin, D.; Zhu, L. Polycyclic Aromatic Hydrocarbons: Pollution and Source Analysis of a Black Tea. J. Agric. Food Chem. 2004, 52, 8268–8271. [Google Scholar] [CrossRef] [PubMed]
- Grover, I.S.; Singh, S.; Pal, B. Priority PAHs in orthodox black tea during manufacturing process. Environ. Monit. Assess. 2013, 185, 6291–6294. [Google Scholar] [CrossRef] [PubMed]
- Charles, I.A.; Ogbolosingha, A.J.; Afia, I.U. Health risk assessment of instant noodles commonly consumed in Port Harcourt, Nigeria. Environ. Sci. Pollut. Res. 2018, 25, 2580–2587. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Shafy, H.I.; Mansour, M.S.M. A review on polycyclic aromatic hydrocarbons: Source, environmental impact, effect on human health and remediation. Egypt. J. Pet. 2016, 25, 107–123. [Google Scholar] [CrossRef]
- European Commission. Commission Regulation No. 208/2005 of 4 February 2005 amending Regulation (EC) no 466/2001 as regards polycylic aromatic hydrocarbons. Off. J. Eur. Union 2005, 34, 3–5. [Google Scholar]
- Sanz-Landaluze, J.; Bartolome, L.; Zuloaga, O.; González, L.; Dietz, C.; Cámara, C. Accelerated extraction for determination of polycyclic aromatic hydrocarbons in marine biota. Anal. Bioanal. Chem. 2006, 384, 1331–1340. [Google Scholar] [CrossRef]
- Ahmed, M.T.; Malhat, F.; Loutfy, N. Residue Levels, Profiles, Emission Source and Daily Intake of Polycyclic Aromatic Hydrocarbons Based on Smoked Fish Consumption, An Egyptian Pilot Study. Polycycl. Aromat. Compd. 2016, 36, 183–196. [Google Scholar] [CrossRef]
- Yoo, M.; Lee, S.; Kim, S.; Kim, S.-j.; Seo, H.-y.; Shin, D. A comparative study of the analytical methods for the determination of polycyclic aromatic hydrocarbons in seafood by high-performance liquid chromatography with fluorescence detection. J. Food Sci. Technol. 2014, 49, 1480–1489. [Google Scholar] [CrossRef]
- Chen, S.; Kao, T.H.; Chen, C.J.; Huang, C.W.; Chen, B.H. Reduction of carcinogenic polycyclic aromatic hydrocarbons in meat by sugar-smoking and dietary exposure assessment in taiwan. J. Agric. Food Chem. 2013, 61, 7645–7653. [Google Scholar] [CrossRef] [PubMed]
- Knobel, G.; Campiglia, A.D. Determination of polycyclic aromatic hydrocarbon metabolites in milk by a quick, easy, cheap, effective, rugged and safe extraction and capillary electrophoresis. J. Sep. Sci. 2013, 36, 2291–2298. [Google Scholar] [CrossRef] [PubMed]
- Forsberg, N.D.; Wilson, G.R.; Anderson, K.A. Determination of Parent and Substituted Polycyclic Aromatic Hydrocarbons in High-Fat Salmon Using a Modified QuEChERS Extraction, Dispersive SPE and GC–MS. J. Agric. Food Chem. 2011, 59, 8108–8116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smoker, M.; Tran, K.; Smith, R.E. Determination of Polycyclic Aromatic Hydrocarbons (PAHs) in Shrimp. J. Agric. Food Chem. 2010, 58, 12101–12104. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Wu, K.; Huo, X.; Xu, X. Sources, distribution, and toxicity of polycyclic aromatic hydrocarbons. J. Environ. Health 2011, 73, 22–25. [Google Scholar] [CrossRef] [PubMed]
- Larsen, J.C. Scientific opinion of the panel on contaminants in the food chain on a request from the European commission on polycyclic aromatic hydrocarbons in foods. EFSA J. 2008, 724, 1–114. [Google Scholar]
- Iwegbue, C.M.A.; Onyonyewoma, U.A.; Bassey, F.I.; Nwajei, G.E.; Martincigh, B.S. Concentrations and Health Risk of Polycyclic Aromatic Hydrocarbons in Some Brands of Biscuits in the Nigerian Market. Hum. Ecol. Risk Assess. 2015, 21, 338–357. [Google Scholar] [CrossRef]
- Lodovici, M.; Dolara, P.; Casalini, C.; Ciappellano, S.; Testolin, G. Polycyclic aromatic hydrocarbon contamination in the Italian diet. Food Addit. Contam. 1995, 12, 703–713. [Google Scholar] [CrossRef]
- Greenberg, A.; Luo, S.; Hsu, C.-H.; Creighton, P.; Waldman, J.; Lioy, P.J. Benzo[a]pyrene in Composite Prepared Meals: Results from the THEES (Total Human Exposure to Environmental Substances) Study. Polycycl. Aromat. Compd. 1990, 1, 221–231. [Google Scholar] [CrossRef]
- Masih, J.; Singhvi, R.; Taneja, A.; Kumar, K.; Masih, H. Gaseous/particulate bound polycyclic aromatic hydrocarbons (PAHs), seasonal variation in North central part of rural India. Sustain. Cities Soc. 2012, 3, 30–36. [Google Scholar] [CrossRef]
- Wickramasinghe, A.P.; Karunaratne, D.G.G.P.; Sivakanesan, R. PM10-bound polycyclic aromatic hydrocarbons: Concentrations, source characterization and estimating their risk in urban, suburban and rural areas in Kandy, Sri Lanka. Atmos. Environ. 2011, 45, 2642–2650. [Google Scholar] [CrossRef]
- Bi, X.; Sheng, G.; Peng, P.A.; Chen, Y.; Fu, J. Size distribution of n-alkanes and polycyclic aromatic hydrocarbons (PAHs) in urban and rural atmospheres of Guangzhou, China. Atmos. Environ. 2005, 39, 477–487. [Google Scholar] [CrossRef]
- Liu, J.; Li, J.; Lin, T.; Liu, D.; Xu, Y.; Chaemfa, C.; Qi, S.; Liu, F.; Zhang, G. Diurnal and nocturnal variations of PAHs in the Lhasa atmosphere, Tibetan Plateau: Implication for local sources and the impact of atmospheric degradation processing. Atmos. Res. 2013, 124, 34–43. [Google Scholar] [CrossRef] [Green Version]
- Schlemitz, S.; Pfannhauser, W. Supercritical fluid extraction of mononitrated polycyclic aromatic hydrocarbons from tea—Correlation with the PAH concentration. Z. Lebensm. Unters. Forsch. A 1997, 205, 305–310. [Google Scholar] [CrossRef]
- Duedahl-Olesen, L.; Navaratnam, M.A.; Jewula, J.; Jensen, A.H. PAH in Some Brands of Tea and Coffee. Polycycl. Aromat. Compd. 2015, 35, 74–90. [Google Scholar] [CrossRef]
- Drabova, L.; Pulkrabova, J.; Kalachova, K.; Tomaniova, M.; Kocourek, V.; Hajslova, J. Rapid determination of polycyclic aromatic hydrocarbons (PAHs) in tea using two-dimensional gas chromatography coupled with time of flight mass spectrometry. Talanta 2012, 100, 207–216. [Google Scholar] [CrossRef]
- Ishizaki, A.; Saito, K.; Hanioka, N.; Narimatsu, S.; Kataoka, H. Determination of polycyclic aromatic hydrocarbons in food samples by automated on-line in-tube solid-phase microextraction coupled with high-performance liquid chromatography-fluorescence detection. J. Chromatogr. A 2010, 1217, 5555–5563. [Google Scholar] [CrossRef]
- García-Falcón, M.S.; Cancho-Grande, B.; Simal-Gándara, J. Minimal clean-up and rapid determination of polycyclic aromatic hydrocarbons in instant coffee. Food Chem. 2005, 90, 643–647. [Google Scholar] [CrossRef]
- De Kruijf, N.; Schouten, T.; Van der Stegen, G.H.D. Rapid determination of benzo[a]pyrene in roasted coffee and coffee brew by high-performance liquid chromatography with fluorescence detection. J. Agric. Food Chem. 1987, 35, 545–549. [Google Scholar] [CrossRef]
- Chen, B.H.; Lin, Y.S. Formation of Polycyclic Aromatic Hydrocarbons during Processing of Duck Meat. J. Agric. Food Chem. 1997, 45, 1394–1403. [Google Scholar] [CrossRef]
- Masuda, Y.; Mori, K.; Kuratsune, M. Polycyclic aromatic hydrocarbons in common Japanese foods. I. Broiled fish, roasted barley, shoyu, and caramel. GANN Jpn. J. Cancer Res. 1966, 57, 133–142. [Google Scholar] [CrossRef]
- Lijinsky, W.; Shubik, P. Benzo(a)pyrene and other polynuclear hydrocarbons in charcoal-broiled meat. Science 1964, 145, 53–55. [Google Scholar] [CrossRef] [PubMed]
- Rey-Salgueiro, L.; Garcia-Falcon, M.S.; Martinez-Carballo, E.; Simal-Gandara, J. Effects of toasting procedures on the levels of polycyclic aromatic hydrocarbons in toasted bread. Food Chem. 2008, 108, 607–615. [Google Scholar] [CrossRef] [PubMed]
- Šimko, P. Determination of polycyclic aromatic hydrocarbons in smoked meat products and smoke flavouring food additives. J. Chromatogr. B 2002, 770, 3–18. [Google Scholar] [CrossRef]
- Janoszka, B.; Warzecha, L.; Blasz czyk, U.; Bodzek, D. Organic compounds formed in the thermally treated high-protein food. Part 1: Polycyclic aromatic hydrocarbons. Acta Chromatogr. 2004, 14, 115–128. [Google Scholar]
| Abbreviations | Compound | tR (min) | Prec. (m/z) | Frag. (m/z) | CE (eV) | Remark |
|---|---|---|---|---|---|---|
| NaP | Naphthalene | 7.1 | 128.2 | 127.2 (102.1) | 15 (20) | Q (C) |
| M2N | 2-Methylnaphthalene | 8.63 | 141.1 | 89.1 (115.1) | 16 (14) | Q (C) |
| M1N | 1-Methylnapththalene | 8.87 | 141.1 | 89.1 (115.1) | 32 (15) | Q (C) |
| ACNP | Acenaphthylene | 10.69 | 154.1 | 153.0 (152.0) | 15 (20) | Q (C) |
| ACP | Acenaphthene | 11.14 | 152.1 | 151.0 (150.0) | 15 (20) | Q (C) |
| FL | Fluorene | 12.32 | 166.1 | 154.0 (164.0) | 15 (20) | Q (C) |
| PHN | Phenalthrene | 14.55 | 178.2 | 176.0 (172.0) | 15 (20) | Q (C) |
| AN | Anthracene | 14.65 | 178.1 | 176.1 (152.1) | 15 (20) | Q (C) |
| FLA | Floranthene | 17.38 | 202.1 | 200.1 (152.1) | 15 (20) | Q (C) |
| Py | Pyrene | 17.88 | 202.1 | 200.2 (152.1) | 15 (20) | Q (C) |
| B(a)A | Benzo(a)athracene | 20.76 | 228.1 | 226.1 (202.2) | 30 (35) | Q (C) |
| Chy | Chrysene | 20.84 | 228.1 | 226.2 (202.1) | 30 (35) | Q (C) |
| B(b)F | Benzo(b)fluoranthene | 23.6 | 252.2 | 250.0 (226.0) | 25 (30) | Q (C) |
| B(k)F | Benzo(k)fluoranthene | 23.6 | 252.2 | 250.1 (226.1) | 25 (30) | Q (C) |
| B(a)P | Benzo(a)pyrene | 24.41 | 252.0 | 250.1 (226.1) | 25 (30) | Q (C) |
| IP | Indeno(1,2,3-cd)pyrene | 28.77 | 276.0 | 274.1 (250.0) | 30 (40) | Q (C) |
| BDA | Dibenz(a,h)anthracene | 28.99 | 278.2 | 276.1 (252.1) | 30 (40) | Q (C) |
| B(g,h,i)P | Benzo(g,h,i)perylence | 29.91 | 276.1 | 274.1 (250.1) | 30 (40) | Q (C) |
| B(a)A-13C6 | Benzo(a)anthracence-13C6 | 20.76 | 234.1 | 232.1 (208.1) | 30 (35) | Q (C) |
| B(ghi)P-13C12 | Benzo(g,h,i)pyrylene-13C12 | 29.91 | 288.2 | 286.2 (261.2) | 30 (40) | Q (C) |
| Food Types | n | B(a)P | Chrysene | Non-Carcinogenic PAHs | 18 PAHs |
|---|---|---|---|---|---|
| 1. Instant noodles | |||||
| Fried noodles | 45 | ND–11.9 (3.6) | ND–11.0 (2.9) | ND–182.8 (14.8) | ND–182.8 (9.6) |
| Non-fried noodles | 20 | ND–6.6 (2.7) | ND–5.9 (2.0) | ND–57.2 (15.1) | ND–57.2 (9.3) |
| 2. Cakes | |||||
| Snack (fried) | 7 | ND–2.9 (0.93) | 0.13–0.92 (0.64) | ND–11.99 (2.78) | ND–11.99 (1.74) |
| Snack (non-fried) | 4 | ND–0.65 (0.25) | 0.04–0.30 (0.16) | ND–1.99 (0.21) | ND–2.32 (0.22) |
| Cookie (fried) | 6 | 0.10–1.33 (0.71) | 1.09–3.11 (2.12) | ND–26.92 (3.33) | ND–26.92 (2.21) |
| Cookie (non-fried) | 5 | ND–0.56 (0.34) | ND–1.49 (0.64) | ND–23.94 (2.83) | ND–23.94 (1.72) |
| Fritter | 4 | ND–0.83 (0.34) | ND–0.19 (0.12) | ND–13.30 (3.98) | ND–13.30 (2.48) |
| 3. Dried vegetables | |||||
| Dried potato | 5 | ND–2.39 (1.20) | 0.11–0.56 (0.22) | ND–8.22 (2.12) | ND–8.22 (1.93) |
| Dried carrot | 4 | ND–2.11 (0.82) | 0.15–3.21 (1.07) | ND–8.61 (1.92) | ND–8.61 (1.72) |
| Dried sweet potato | 5 | ND–1.92 (0.81) | ND–1.81 (0.81) | ND–13.30 (3.12) | ND–13.3 (2.93) |
| Dried vegetable | 5 | ND–1.22 (0.75) | ND–1.52 (0.75) | ND–10.50 (5.06) | ND–10.5 (4.83) |
| Dried jackfruit | 6 | ND–1.04 (0.52) | ND–1.17 (0.56) | ND–5.09 (1.03) | ND–5.09 (0.91) |
| 4. Teas | |||||
| Green tea (TN) | 5 | 0.21–2.2 (1.03) | 1.09–2.71 (1.43) | ND–71.62 (9.24) | ND–71.6 (8.12) |
| Green tea (LD) | 5 | 0.14–1.82 (0.91) | 0.72–3.72 (1.12) | ND–81.2 (8.79) | ND–81.2 (5.14) |
| Oolong tea (TN) | 7 | 2.12–4.23 (3.21) | 3.21–8.32 (5.32) | 2.08–144.3 (38.12) | 2.08–144.3 (23.08) |
| Oolong tea (LD) | 5 | 1.01–2.95 (1.32) | ND–6.63 (2.42) | ND–90.2 (21.13) | ND–90.2 (12.45) |
| Black tea | 7 | 4.89–19.82 (9.42) | 7.64–17.2 (9.21) | 1.12–199 (37.91) | 0.912–318 (23.32) |
| 5. Coffees | |||||
| Arabica | 5 | ND–1.51 (1.21) | ND–2.13 (1.12) | ND–37.23 (6.18) | ND–37.23 (5.24) |
| Robusta | 7 | ND–1.34 (0.31) | ND | ND–30.32 (5.21) | ND–30.32 (4.82) |
| Cherry | 3 | 2.31–6.72 (4.52) | 1.43–3.92 (2.59) | ND–72.52 (16.32) | ND–72.52 (15.11) |
| Instant coffee | 9 | 4.52–21.35 (9.16) | 2.89–10.12 (7.26) | ND–119.10 (26.32) | ND–119.1 (24.35) |
| 6. Grilled meats | |||||
| Coal-grilled | 7 | 23.12–65.3 (49.15) | 2.13–5.62 (3.31) | 4.84–160.03 (30.41) | 2.13–160.03 (25.2) |
| Wrapped coal-grilled | 3 | 5.82–15.62 (10.41) | 1.23–3.01 (2.41) | 1.72–59.3 (13.12) | 1.23–59.3 (12.42) |
| Charcoal 1, grilled | 7 | 19.24–30.25 (26.1) | 3.65–12.42 (6.02) | 4.52–112.6 (24.85) | 3.65–112.6 (23.83) |
| Charcoal 2, grilled | 7 | 13.19–28.45 (23.4) | 4.45–10.72 (8.57) | 0.31–116.21 (18.74) | 0.31–116.2 (18.45) |
| Stove-grilled | 5 | ND–4.23 (1.08) | ND–0.23 (0.14) | ND–3.61 (1.52) | ND–3.61 (1.43) |
| Abbr. | Compounds | LOD (µg/kg) | LOQ (µg/kg) | RSDr (%) | RSDR (%) |
|---|---|---|---|---|---|
| NaP | Naphthalene | 0.10 | 0.30 | 20 | 17, 10, 20 |
| M2N | 2-Methylnaphthalene | 0.20 | 0.60 | 18 | 16, 9, 11 |
| M1N | 1-Methylnapththalene | 0.05 | 0.15 | 16 | 19, 13, 18 |
| ACNP | Acenaphthylene | 0.05 | 0.15 | 12 | 18, 6, 14 |
| ACP | Acenaphthene | 0.05 | 0.15 | 10 | 16, 5, 13 |
| FL | Fluorene | 0.01 | 0.03 | 14 | 8, 9, 15 |
| PHN | Phenalthrene | 0.05 | 0.15 | 8 | 13, 9, 18 |
| AN | Anthracene | 0.05 | 0.15 | 13 | 16, 7, 17 |
| FLA | Floranthene | 0.01 | 0.03 | 18 | 10, 6, 12 |
| Py | Pyrene | 0.05 | 0.15 | 7 | 11, 6, 15 |
| B(a)A | Benzo(a)athracene | 0.05 | 0.15 | 9 | 6, 8, 13 |
| Chy | Chrysene | 0.05 | 0.15 | 10 | 12, 7, 16 |
| B(b)F | Benzo(b)fluoranthene | 0.05 | 0.15 | 12 | 15, 5, 20 |
| B(k)F | Benzo(k)fluoranthene | 0.05 | 0.15 | 19 | 11, 8, 18 |
| B(a)P | Benzo(a)pyrene | 0.05 | 0.15 | 11 | 11, 5, 16 |
| IP | Indeno(1,2,3-cd)pyrene | 0.05 | 0.15 | 20 | 13, 8, 18 |
| BDA | Dibenz(a,h)anthracene | 0.10 | 0.30 | 15 | 10, 6, 15 |
| B(ghi)P | Benzo(g,h,i)perylence | 0.10 | 0.30 | 8 | 9, 8, 14 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tran-Lam, T.-T.; Hai Dao, Y.; Kim Thi Nguyen, L.; Kim Ma, H.; Nguyen Tran, H.; Truong Le, G. Simultaneous Determination of 18 Polycyclic Aromatic Hydrocarbons in Daily Foods (Hanoi Metropolitan Area) by Gas Chromatography–Tandem Mass Spectrometry. Foods 2018, 7, 201. https://doi.org/10.3390/foods7120201
Tran-Lam T-T, Hai Dao Y, Kim Thi Nguyen L, Kim Ma H, Nguyen Tran H, Truong Le G. Simultaneous Determination of 18 Polycyclic Aromatic Hydrocarbons in Daily Foods (Hanoi Metropolitan Area) by Gas Chromatography–Tandem Mass Spectrometry. Foods. 2018; 7(12):201. https://doi.org/10.3390/foods7120201
Chicago/Turabian StyleTran-Lam, Thanh-Thien, Yen Hai Dao, Lien Kim Thi Nguyen, Hoi Kim Ma, Hai Nguyen Tran, and Giang Truong Le. 2018. "Simultaneous Determination of 18 Polycyclic Aromatic Hydrocarbons in Daily Foods (Hanoi Metropolitan Area) by Gas Chromatography–Tandem Mass Spectrometry" Foods 7, no. 12: 201. https://doi.org/10.3390/foods7120201
APA StyleTran-Lam, T.-T., Hai Dao, Y., Kim Thi Nguyen, L., Kim Ma, H., Nguyen Tran, H., & Truong Le, G. (2018). Simultaneous Determination of 18 Polycyclic Aromatic Hydrocarbons in Daily Foods (Hanoi Metropolitan Area) by Gas Chromatography–Tandem Mass Spectrometry. Foods, 7(12), 201. https://doi.org/10.3390/foods7120201






