Nutrient and Total Polyphenol Contents of Dark Green Leafy Vegetables, and Estimation of Their Iron Bioaccessibility Using the In Vitro Digestion/Caco-2 Cell Model
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Cultivation and Collection
2.2. Sample Preparation
2.3. Compositional Analysis
2.3.1. Moisture and Protein
2.3.2. Mineral Analysis: Calcium, Iron, and Zinc
2.3.3. β-Carotene
2.3.4. Ascorbic Acid
2.3.5. Polyphenols
2.4. In Vitro Digestion/Caco-2 Cell Model for Iron Availability
2.5. Statistical Analysis
3. Results
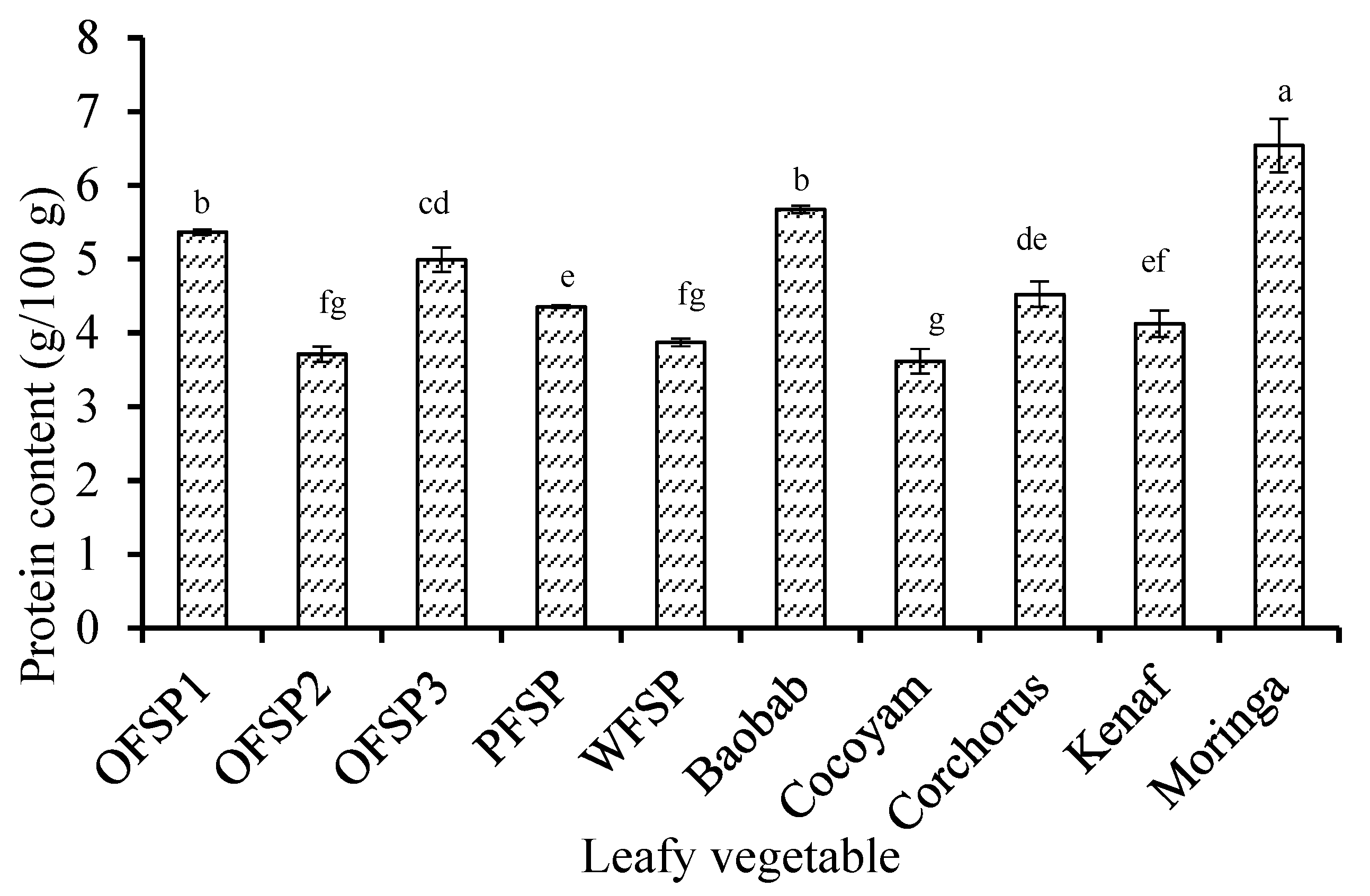
3.1. Compositional Profile
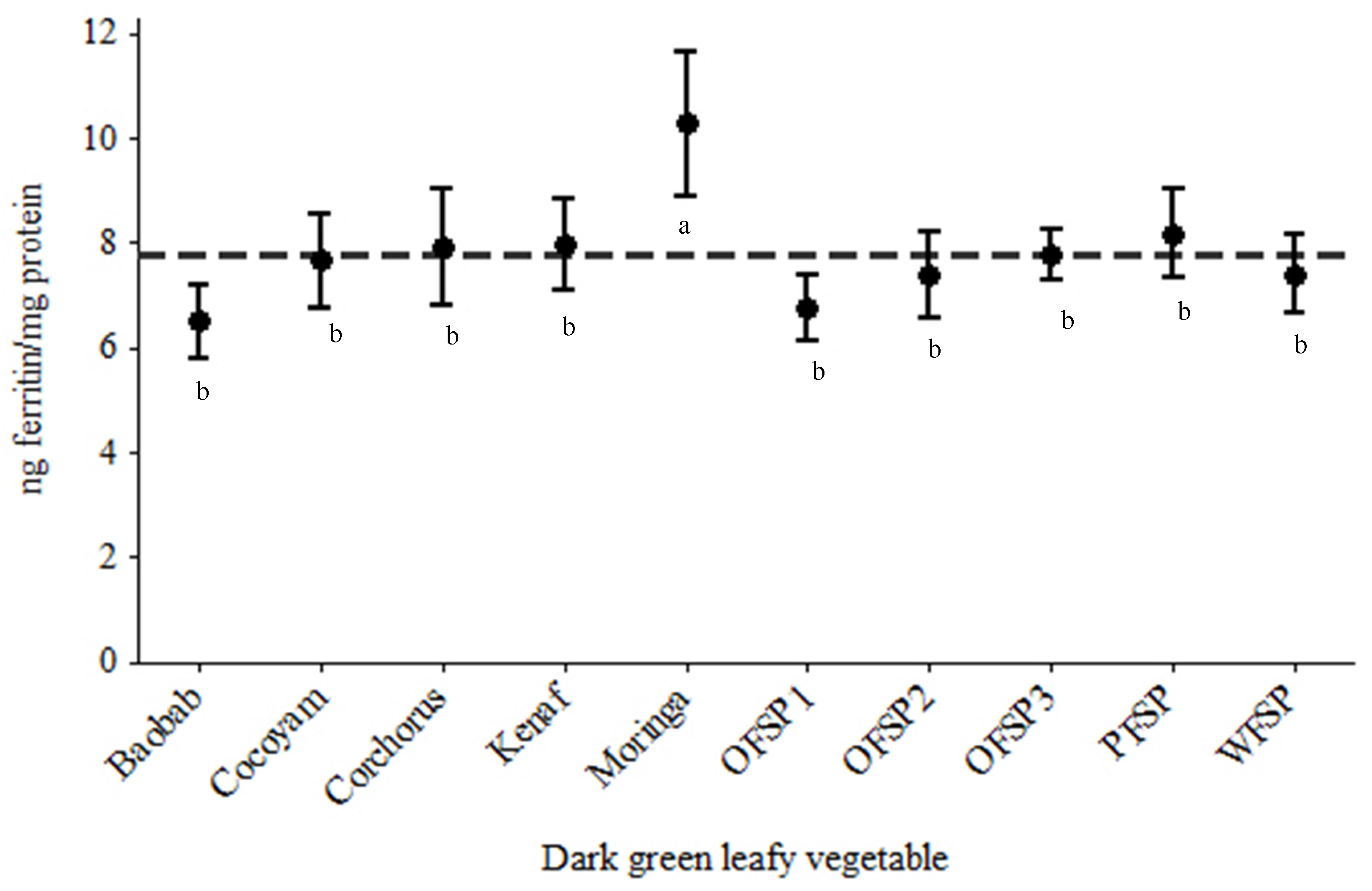
3.2. In Vitro Iron Bioaccessibility Using Caco-2 Cells as a Model
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Faber, M.; Van Jaarsveld, P.; Laubscher, R. The contribution of dark-green leafy vegetables to total micronutrient intake of two-to five-year-old children in a rural setting. Water SA 2007, 33, 407–412. [Google Scholar]
- De Benoist, B.; McLean, E.; Egli, I.; Cogswell, M. Worldwide Prevalence of Anaemia 1993–2005: WHO Global Database on Anaemia; WHO Press: Geneva, Switzerland, 2008. [Google Scholar]
- World Health Organization. Global Prevalence of Vitamin A Deficiency in Populations at Risk 1995–2005: WHO Global Database on Vitamin A Deficiency; WHO Press: Geneva, Switzerland, 2009. [Google Scholar]
- Cercamondi, C.I.; Icard-Vernière, C.; Egli, I.M.; Vernay, M.; Hama, F.; Brouwer, I.D.; Zeder, C.; Berger, J.; Hurrell, R.F.; Mouquet-Rivier, C. A higher proportion of iron-rich leafy vegetables in a typical Burkinabe maize meal does not increase the amount of iron absorbed in young women. J. Nutr. 2014, 144, 1394–1400. [Google Scholar] [PubMed]
- Davies, N.T.; Reid, H. An evaluation of the phytate, zinc, copper, iron and manganese contents of, and Zn availability from, soya-based textured-vegetable-protein meat-substitutes or meat-extenders. Br. J. Nutr. 1979, 41, 579–589. [Google Scholar] [PubMed]
- Gautam, S.; Platel, K.; Srinivasan, K. Promoting influence of combinations of amchur, β-carotene-rich vegetables and Allium spices on the bioaccessibility of zinc and iron from food grains. Int. J. Food Sci. Nutr. 2011, 62, 518–524. [Google Scholar] [PubMed]
- Garcia-Casal, M.N.; Layrisse, M.; Solano, L.; Baron, M.A.; Arguello, F.; Llovera, D.; Ramirez, J.; Leets, I.; Tropper, E. Vitamin A and beta-carotene can improve nonheme iron absorption from rice, wheat and corn by humans. J. Nutr. 1998, 128, 646–650. [Google Scholar] [PubMed]
- Garcia-Casal, M.N.; Leets, I. Carotenoids, but not vitamin A, improve iron uptake and ferritin synthesis by Caco-2 cells from ferrous fumarate and NaFe-EDTA. J. Food Sci. 2014, 79, H706–H712. [Google Scholar] [PubMed]
- Van Jaarsveld, P.; Faber, M.; van Heerden, I.; Wenhold, F.; van Rensburg, W.J.; van Averbeke, W. Nutrient content of eight African leafy vegetables and their potential contribution to dietary reference intakes. J. Food Compos. Anal. 2014, 33, 77–84. [Google Scholar]
- Takyi, E.E.K. Children’s consumption of dark green, leafy vegetables with added fat enhances serum retinol. J. Nutr. 1999, 129, 1549–1554. [Google Scholar] [PubMed]
- ORC Macro. Ghana Demographic and Health Survey 2003. Available online: http://www.dhsprogram.com/pubs/pdf/FR152/FR152.pdf (accessed on 7 July 2017).
- Ghana Statistical Service (GSS); Ghana Health Service (GHS); ICF International. Ghana Demographic and Health Survey 2014. Available online: https://dhsprogram.com/pubs/pdf/FR307/FR307.pdf (accessed on 7 July 2017).
- Ghana Statistical Service (GSS); Ghana Health Service (GHS); ICF Macro. Ghana Demographic and Health Survey 2008. Available online: http://www.dhsprogram.com/pubs/pdf/FR221/FR221[13Aug2012].pdf (accessed on 7 July 2017).
- World Health Organization. WHO Global Database on Vitamin A Deficiency. Available online: http://www.who.int/vmnis/vitamina/data/database/countries/gha_vita.pdf (accessed on 1 August 2016).
- Amagloh, F.K.; Nyarko, E.S. Mineral nutrient content of commonly consumed leafy vegetables in northern Ghana. Afr. J. Food Agric. Nutr. Dev. 2012, 12, 6397–6408. [Google Scholar]
- Dittoh, S. Improving availability of nutritionally adequate and affordable food supplies at community levels in West Africa. In International Workshop: Food-Based Approaches for a Healthy Nutrition in West Africa, Proceedings of the 2nd International Workshop, Ouagadougou, Burkina Faso, 23–28 November 2003; Presses Universitaires de Ouagadougou: Ouagadougou, Burkina Faso, 2004; pp. 23–28. [Google Scholar]
- Oduro, I.; Ellis, W.; Owusu, D. Nutritional potential of two leafy vegetables: Moringa oleifera and Ipomoea batatas leaves. Sci. Res. Essays 2008, 3, 57–60. [Google Scholar]
- Barrera, W.A.; Picha, D.H. Ascorbic acid, thiamin, riboflavin, and vitamin B6 contents vary between sweetpotato tissue types. HortScience 2014, 49, 1470–1475. [Google Scholar]
- Faber, M.; Laurie, S.M.; van Jaarsveld, P.J. Total β-carotene content of orange sweetpotato cultivated under optimal conditions and at a rural village. Afr. J. Biotechnol. 2013, 12, 3947–3951. [Google Scholar]
- Islam, S. Antimutagenicity and antioxidant activity in the Ipomoea batatas L. genotypes in relation to polyphenolics. In Proceedings of the International Conference on Advances in Agricultural, Biological & Environmental Sciences, London, UK, 22–23 July 2015; pp. 1–7. [Google Scholar]
- Tako, E.; Beebe, S.; Reed, S.; Hart, J.; Glahn, R. Polyphenolic compounds appear to limit the nutritional benefit of biofortified higher iron black bean (Phaseolus vulgaris L.). Nutr. J. 2014, 13, 28. [Google Scholar] [CrossRef] [PubMed]
- Tako, E.; Reed, S.M.; Budiman, J.; Hart, J.J.; Glahn, R.P. Higher iron pearl millet (Pennisetum glaucum L.) provides more absorbable iron that is limited by increased polyphenolic content. Nutr. J. 2015, 14. [Google Scholar] [CrossRef] [PubMed]
- Petry, N.; Egli, I.; Campion, B.; Nielsen, E.; Hurrell, R. Genetic reduction of phytate in common bean (Phaseolus vulgaris L.) seeds increases iron absorption in young women. J. Nutr. 2013, 143, 1219–1224. [Google Scholar] [CrossRef] [PubMed]
- Abizari, A.-R.; Moretti, D.; Schuth, S.; Zimmermann, M.B.; Armar-Klemesu, M.; Brouwer, I.D. Phytic acid-to-iron molar ratio rather than polyphenol concentration determines iron bioavailability in whole-cowpea meal among young women. J. Nutr. 2012, 142, 1950–1955. [Google Scholar] [CrossRef] [PubMed]
- Glahn, R.P.; Rassier, M.; Goldman, M.I.; Lee, O.A.; Cha, J. A comparison of iron availability from commercial iron preparations using an in vitro digestion/Caco-2 cell culture model. J. Nutr. Biochem. 2000, 11, 62–68. [Google Scholar] [CrossRef]
- Kamiloglu, S.; Capanoglu, E.; Grootaert, C.; Van Camp, J. Anthocyanin Absorption and Metabolism by Human Intestinal Caco-2 Cells—A Review. Int. J. Mol. Sci. 2015, 16, 21555–21574. [Google Scholar] [CrossRef] [PubMed]
- Fairweather-Tait, S.; Lynch, S.; Hotz, C.; Hurrell, R.F.; Abrahamse, L.; Beebe, S.; Bering, S.; Bukhave, K.; Glahn, R.; Hambidge, M.; et al. The usefulness of in vitro models to predict the bioavailability of iron and zinc: A consensus statement from the HarvestPlus expert consultation. Int. J. Vitam. Nutr. Res. 2005, 75, 371–374. [Google Scholar] [CrossRef] [PubMed]
- Tako, E.; Bar, H.; Glahn, R. The combined application of the Caco-2 cell bioassay coupled with in vivo (Gallus gallus) feeding trial represents an effective approach to predicting Fe bioavailability in humans. Nutrients 2016, 8, 732. [Google Scholar] [CrossRef] [PubMed]
- Association of Official Analytical Chemists (AOAC). Official Methods of Analysis of AOAC International, 18th ed.; AOAC International: Gaithersburg, MD, USA, 2005. [Google Scholar]
- Zand, N.; Chowdhry, B.Z.; Wray, D.S.; Pullen, F.S.; Snowden, M.J. Elemental content of commercial ‘ready to-feed’ poultry and fish based infant foods in the UK. Food Chem. 2012, 135, 2796–2801. [Google Scholar] [CrossRef] [PubMed]
- Bechoff, A.; Westby, A.; Owori, C.; Menya, G.; Dhuique-Mayer, C.; Dufour, D.; Tomlins, K. Effect of drying and storage on the degradation of total carotenoids in orange-fleshed sweetpotato cultivars. J. Sci. Food Agric. 2010, 90, 622–629. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.S.; Coates, G.A. Liquid chromatographic determination of vitamin C in commercial Florida citrus juices. J. Micronutr. Anal. 1987, 3, 199–209. [Google Scholar]
- Isabelle, M.; Lee, B.L.; Lim, M.T.; Koh, W.-P.; Huang, D.; Ong, C.N. Antioxidant activity and profiles of common vegetables in Singapore. Food Chem. 2010, 120, 993–1003. [Google Scholar] [CrossRef]
- Christides, T.; Amagloh, F.K.; Coad, J. Iron bioavailability and provitamin A from sweet potato- and cereal-based complementary foods. Foods 2015, 4, 463–476. [Google Scholar] [CrossRef] [PubMed]
- Glahn, R.P.; Lee, O.A.; Yeung, A.; Goldman, M.I.; Miller, D.D. Caco-2 cell ferritin formation predicts nonradiolabeled food iron availability in an in vitro digestion Caco-2 cell culture model. J. Nutr. 1998, 128, 1555–1561. [Google Scholar] [PubMed]
- Yun, S.M.; Habicht, J.P.; Miller, D.D.; Glahn, R.P. An in vitro digestion/Caco-2 cell culture system accurately predicts the effects of ascorbic acid and polyphenolic compounds on iron bioavailability in humans. J. Nutr. 2004, 134, 2717–2721. [Google Scholar] [PubMed]
- Sharp, P.; Tandy, S.; Yamaji, S.; Tennant, J.; Williams, M.; Singh Srai, S.K. Rapid regulation of divalent metal transporter (DMT1) protein but not mRNA expression by non-haem iron in human intestinal Caco-2 cells. FEBS Lett. 2002, 510, 71–76. [Google Scholar] [CrossRef]
- Sun, H.; Mu, T.; Xi, L.; Zhang, M.; Chen, J. Sweet potato (Ipomoea batatas L.) leaves as nutritional and functional foods. Food Chem. 2014, 156, 380–389. [Google Scholar] [CrossRef] [PubMed]
- Islam, S.N.; Nusrat, T.; Begum, P.; Ahsan, M. Carotenoids and β-Carotene in orange fleshed sweet Potato: A possible solution to vitamin A deficiency. Food Chem. 2016, 199, 628–631. [Google Scholar] [CrossRef] [PubMed]
- Van Jaarsveld, P.J.; Faber, M.; Tanumihardjo, S.A.; Nestel, P.; Lombard, C.J.; Benade, A.J.S. β–carotene-rich orange-fleshed sweet potato improves the vitamin A status of primary school children assessed with the modified-relative-dose-response test. Am. J. Clin. Nutr. 2005, 81, 1080–1087. [Google Scholar] [PubMed]
- Tumwegamire, S.; Mwanga, R.O.M.; Andrade, M.; Low, J.W.; Kapinga, R.E.; Ssemakula, G.N.; Laurie, S.M.; Chipungu, F.P.; Ndirigue, J.; Agili, S.; et al. Orange-Fleshed Sweetpotato for Africa: Catalogue 2014, 2nd ed.; International Potato Center (CIP): Lima, Peru, 2014. [Google Scholar]
- Lartey, A.; Manu, A.; Brown, K.H.; Peerson, J.M.; Dewey, K.G. A randomized, community-based trial of the effects of improved, centrally processed complementary foods on growth and micronutrient status of Ghanaian infants from 6 to 12 mo of age. Am. J. Clin. Nutr. 1999, 70, 391–404. [Google Scholar] [PubMed]
- Lartey, A.; Manu, A.; Brown, K.H.; Peerson, J.M.; Dewey, K.G. Predictors of growth from 1 to 18 months among breast-fed Ghanaian infants. Eur. J. Clin. Nutr. 2000, 54, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Teucher, B.; Olivares, M.; Cori, H. Enhancers of iron absorption: Ascorbic acid and other organic acids. Int. J. Vitam. Nutr. Res. 2004, 74, 403–419. [Google Scholar] [CrossRef] [PubMed]
- Hallberg, L.; Brune, M.; Erlandsson, M.; Sandberg, A.-S.; Rossander-Hulten, L. Calcium: Effect on different amounts on non heme-iron and heme-iron absorption in humans. Am. J. Clin. Nutr. 1991, 53, 112–119. [Google Scholar] [PubMed]
- Amagloh, F.K.; Brough, L.; Weber, J.L.; Mutukumira, A.N.; Hardacre, A.; Coad, J. Sweetpotato-based complementary food would be less inhibitory on mineral absorption than a maize-based infant food assessed by compositional analysis. Int. J. Food Sci. Nutr. 2012, 63, 957–963. [Google Scholar] [CrossRef] [PubMed]
- Amagloh, F.K.; Coad, J. Orange-fleshed sweet potato-based infant food is a better source of dietary vitamin A than a maize-legume blend as complementary food. Food Nutr. Bull. 2014, 35, 51–59. [Google Scholar] [CrossRef] [PubMed]

| DGLV | Moisture (g) ¥ | Calcium (mg) | Iron (mg) | Zinc (mg) | β-Carotene (μg) | Ascorbic Acid (mg) | Total Polyphenols (mg GAE) † |
|---|---|---|---|---|---|---|---|
| OFSP1 | 84.09 ± 0.34 c,d | 95.61 ± 8.01 c,d | 3.41 ± 0.36 a,b | 0.44 ± 0.01 b | 10,533 a,b | 0.74 ± 0.16 c | 506.93 ± 86.76 b |
| OFSP2 | 84.76 ± 0.75 b,c,d | 81.04 ± 3.24 d | 1.89 ± 0.29 b | 0.42 ± 0.02 b | 8280 a,b,c | 0.50 ± 0.15 c | 356.69 ± 79.60 c |
| OFSP3 | 87.24 ± 0.13 a | 103.25 ± 2.59 c,d | 2.58 ± 0.21 a,b | 0.36 ± 0.03 b | 7053 b,c | 0.45 ± 0.07 c | 336.38 ± 63.15 c,d,e |
| PFSP | 84.30 ± 0.26 c,d | 84.75 ± 8.83 c,d | 2.04 ± 0.36 b | 0.44 ± 0.04 b | 4472 b,c | 0.48 ± 0.03 c | 231.44 ± 49.77 c,d,e |
| WFSP | 83.91 ± 0.26 d | 87.02 ± 6.80 c,d | 3.27 ± 0.34 a,b | 0.40 ± 0.03 b | 9501 a,b,c | 0.34 ± 0.10 c | 234.86 ± 0.16 c,d,e |
| Baobab | 85.97 ± 0.53 b | 535.63 ± 22.93 a | 4.59 ± 1.28 a | 0.65 ± 0.03 b | 7166 b,c | 25.50 ± 0.01 b | 1646.75 ± 69.44 a |
| Cocoyam | 85.23 ± 0.64 b,c | 166.39 ± 15.13 b | 2.64 ± 0.16 a,b | 1.49 ± 0.47 a | 3911 c | 1.14 ± 0.01 c | 196.05 ± 10.96 e |
| Corchorus | 78.99 ± 0.38 f | 121.41 ± 3.61 c | 2.48 ± 0.23 a,b | 0.45 ± 0.02 b | 9298 a,b,c | 3.53 ± 0.58 c | 337.94 ± 16.44 c,d,e |
| Kenaf | 80.68 ± 0.18 e | 90.24 ± 17.76 c,d | 2.94 ± 0.25 a,b | 0.35 ± 0.05 b | 8959 a,b,c | 21.79 ± 1.54 b | 202.42 ± 9.29 d,e |
| Moringa | 78.81 ± 0.42 f | 186.22 ± 23.81 b | 4.55 ± 1.88 a | 0.77 ± 0.06 b | 14,169 a | 46.30 ± 4.78 a | 347.38 ± 14.59 c,d |
| p-Value | <0.001 | <0.001 | 0.002 | <0.001 | <0.001 | <0.001 | <0.001 |
| Variable # | Estimate (Standard Error) | p-Value |
|---|---|---|
| Intercept | 10.26 (3.24) | 0.09 |
| Calcium (mg/100 g) | −0.00 (0.03) | 0.99 |
| Iron (mg/100 g) | 0.13 (1.18) | 0.92 |
| Zinc (mg/100 g) | −0.87 (1.81) | 0.68 |
| β-carotene (μg/100 g) | −0.00 (0.00) | 0.35 |
| Ascorbic acid (mg/100 g) | 0.01 (0.08) | 0.93 |
| Total polyphenols (mg GAE/100 g) | −0.00 (0.01) | 0.91 |
| Protein (g/100 g) | 0.29 (0.78) | 0.74 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amagloh, F.K.; Atuna, R.A.; McBride, R.; Carey, E.E.; Christides, T. Nutrient and Total Polyphenol Contents of Dark Green Leafy Vegetables, and Estimation of Their Iron Bioaccessibility Using the In Vitro Digestion/Caco-2 Cell Model. Foods 2017, 6, 54. https://doi.org/10.3390/foods6070054
Amagloh FK, Atuna RA, McBride R, Carey EE, Christides T. Nutrient and Total Polyphenol Contents of Dark Green Leafy Vegetables, and Estimation of Their Iron Bioaccessibility Using the In Vitro Digestion/Caco-2 Cell Model. Foods. 2017; 6(7):54. https://doi.org/10.3390/foods6070054
Chicago/Turabian StyleAmagloh, Francis Kweku, Richard Atinpoore Atuna, Richard McBride, Edward Ewing Carey, and Tatiana Christides. 2017. "Nutrient and Total Polyphenol Contents of Dark Green Leafy Vegetables, and Estimation of Their Iron Bioaccessibility Using the In Vitro Digestion/Caco-2 Cell Model" Foods 6, no. 7: 54. https://doi.org/10.3390/foods6070054
APA StyleAmagloh, F. K., Atuna, R. A., McBride, R., Carey, E. E., & Christides, T. (2017). Nutrient and Total Polyphenol Contents of Dark Green Leafy Vegetables, and Estimation of Their Iron Bioaccessibility Using the In Vitro Digestion/Caco-2 Cell Model. Foods, 6(7), 54. https://doi.org/10.3390/foods6070054





