Influence of Food Characteristics and Food Additives on the Antimicrobial Effect of Garlic and Oregano Essential Oils
Abstract
:1. Introduction
2. Material and Methods
2.1. Gas Chromatography-Mass Spectrometry Analysis of Essential Oils
2.2. Microorganisms and Growth Conditions
2.3. Antimicrobial Effect on Disk Diffusion Assay
2.4. Determination of MIC and MBC
2.5. Preparation of Food Model Media
Influence of Fat, Protein, pH and aw
2.6. Influence of Food Additives (Sodium Nitrite, Commercial Phosphates and Sodium Lactate)
2.7. Microbial Preparation
2.8. Statistical Analysis
3. Results and Discussion
3.1. Chemical Composition and Antimicrobial Properties of EOs
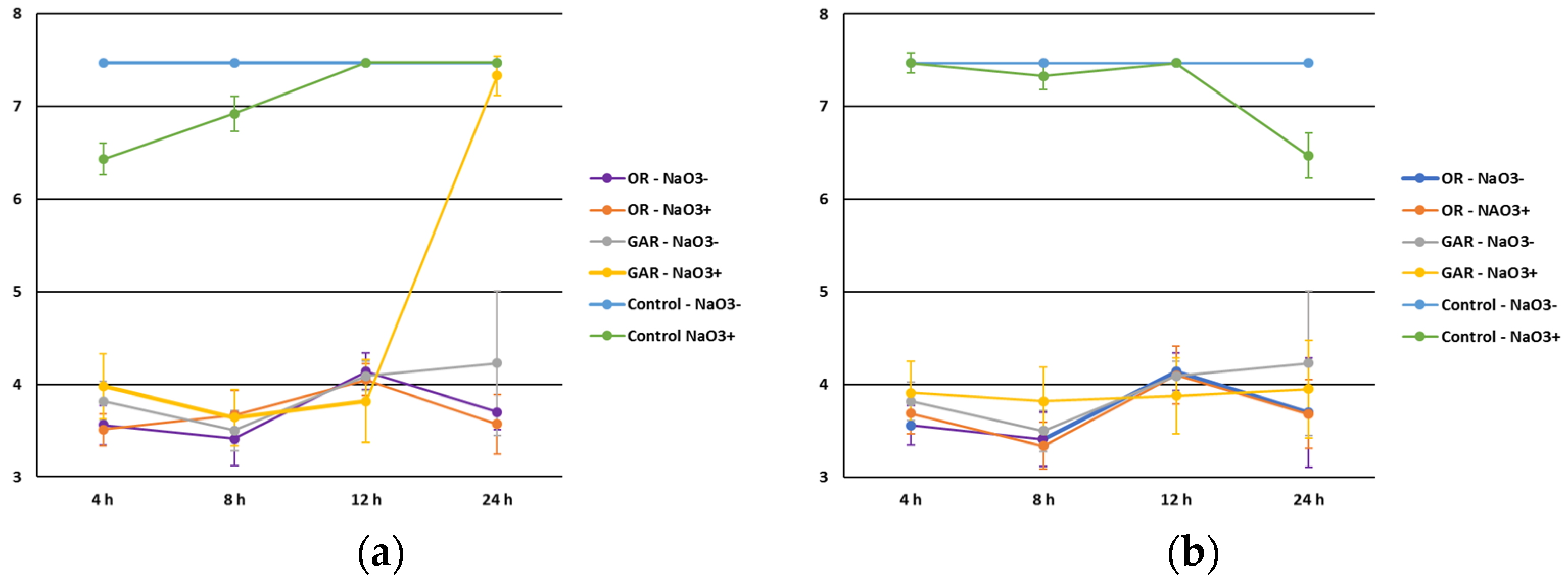
3.2. Effect of Fat, Protein, aw, pH and Food Additives (Sodium Nitrite, Sodium Lactate and Food Phosphates)
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hyldgaard, M.; Mygind, T.; Meyer, R.L. Essential oils in food preservation: Mode of action, synergies, and interactions with food matrix components. Front. Microbiol. 2012, 3, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, B.K.; Valdramidis, V.P.; O’Donnell, C.P.; Muthukumarappan, K.; Bourke, P.; Cullen, P.J. Application of natural antimicrobials for food preservation. J. Agric. Food Chem. 2009, 57, 5987–6000. [Google Scholar] [CrossRef] [PubMed]
- García-Díez, J.; Patarata, L. Behaviour of Salmonella spp., Listeria monocytogenes, and Staphylococcus aureus in Chouriço de Vinho, a dry fermented sausage made from wine-marinated meat. J. Food Prot. 2013, 76, 588–594. [Google Scholar] [CrossRef] [PubMed]
- Linares, M.B.; Garrido, M.D.; Martins, C.; Patarata, L. Efficacies of Garlic and L. Sakei in wine-based marinades for controlling Listeria monocytogenes and Salmonella spp. inchouriço de vinho, a dry sausage made from wine-marinated pork. J. Food Sci. 2013, 78, M719–M724. [Google Scholar] [CrossRef] [PubMed]
- Perricone, M.; Arace, E.; Corbo, M.R.; Sinigaglia, M.; Bevilacqua, A. Bioactivity of essential oils: A review on their interaction with food components. Front. Microbiol. 2015, 6, 76. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, J.; Barry-Ryan, C.; Bourke, P. Antimicrobial activity of plant essential oils using food model media: Efficacy, synergistic potential and interactions with food components. Food Microbiol. 2009, 26, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Hierro, E.; Fernandez, M.; de la Hoz, L. Mediterranean Products. In Handbook of Fermented Meat and Poultry; Toldrá, F., Hui, Y.H., Astiasaran, I., Sebranek, J., Talon, R., Eds.; Willey Blackwell: West Sussex, UK, 2015; pp. 301–312. [Google Scholar]
- García-Díez, J.; Alheiro, J.; Falco, V.; Fraqueza, M.J.; Patarata, L. Chemical characterization and antimicrobial properties of herbs and spices essential oils against pathogens and spoilage bacteria associated to dry-cured meat products. J. Essent. Oil Res. 2016, 29, 117–125. [Google Scholar] [CrossRef]
- Talon, R.; Lebert, I.; Lebert, A.; Leroy, S.; Garriga, M.; Aymerich, T.; Drosinos, E.H.; Zanardi, E.; Ianieri, A.; Fraqueza, M.J.; et al. Traditional dry fermented sausages produced in small-scale processing units in Mediterranean countries and Slovakia. 1. Microbial ecosystems of processing environments. Meat Sci. 2007, 77, 570–579. [Google Scholar] [CrossRef] [PubMed]
- Zaika, L.L. Spices and herbs: Their antimicrobial activity and its determination. J. Food Saf. 1987, 9, 97–118. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically. Approved Standard—Eighth Edition. Clin. Lab. Stand. Inst. 2009, 29, 2. [Google Scholar]
- Troller, J.A.; Stinson, J.V. Moisture requirements for growth and metabolite production by lactic acid bacteria. Appl. Environ. Microbiol. 1981, 42, 682–687. [Google Scholar] [PubMed]
- Fratianni, F.; De Martino, L.; Melone, A.; De Feo, V.; Coppola, R.; Nazzaro, F. Preservation of chicken breast meat treated with thyme and balm essential oils. J. Food Sci. 2010, 75, M528–M535. [Google Scholar] [CrossRef] [PubMed]
- Karabagias, I.; Badeka, A.; Kontominas, M.G. Shelf life extension of lamb meat using thyme or oregano essential oils and modified atmosphere packaging. Meat Sci. 2011, 88, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Soultos, N.; Tzikas, Z.; Christaki, E.; Papageorgiou, K.; Steris, V. The effect of dietary oregano essential oil on microbial growth of rabbit carcasses during refrigerated storage. Meat Sci. 2009, 81, 474–478. [Google Scholar] [CrossRef] [PubMed]
- Dussault, D.; Dang Vu, K.; Lacroix, M. In vitro evaluation of antimicrobial activities of various commercial essential oils, oleoresin and pure compounds against food pathogens and application in ham. Meat Sci. 2014, 96, 514–520. [Google Scholar] [CrossRef] [PubMed]
- Viuda-Martos, M.; Ruiz-Navajas, Y.; Fernández-López, J.; Pérez-Álvarez, J.A. Effect of added citrus fibreand spice essential oils on quality characteristics and shelf-life of mortadella. Meat Sci. 2010, 85, 568–576. [Google Scholar] [CrossRef] [PubMed]
- Benkeblia, N. Antimicrobial activity of essential oil extracts of various onions (Allium cepa) and garlic (Allium sativum). LWT Food Sci. Technol. 2004, 37, 263–268. [Google Scholar] [CrossRef]
- Smith-Palmer, A.; Stewart, J.; Fyfe, L. The potential application of plant essential oils as natural food preservatives in soft cheese. Food Microbiol. 2001, 18, 463–470. [Google Scholar] [CrossRef]
- Solórzano-Santos, F.; Miranda-Novales, M.G. Essential oils from aromatic herbs as antimicrobial agents. Curr. Opin. Biotechnol. 2012, 23, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Gil, A.O.; Delaquis, P.; Russo, P.; Holley, R.A. Evaluation of antilisterial action of cilantro oil on vacuum packed ham. Int. J. Food Microbiol. 2002, 73, 83–92. [Google Scholar] [CrossRef]
- Cava, R.; Nowak, E.; Taboada, A.; Marin-Iniesta, F. Antimicrobial activity of clove and cinnamon essential oils against Listeria monocytogenes in pasteurized milk. J. Food Protect. 2007, 70, 2757–2763. [Google Scholar] [CrossRef]
- Singh, A.; Singh, R.K.; Bhunia, A.K.; Singh, N. Efficacy of plant essential oils as antimicrobial agents against Listeria monocytogenes in hotdogs. LWT Food Sci. Technol. 2003, 36, 787–794. [Google Scholar] [CrossRef]
- Drumm, T.D.; Spanier, A.M. Changes in the content of lipid autoxidation and sulfur-containing compounds in cooked beef during storage. J. Agric. Food Chem. 1991, 39, 336–343. [Google Scholar] [CrossRef]
- Abdollahzadeh, E.; Rezaei, M.; Hosseini, H. Antibacterial activity of plant essential oils and extracts: The role of thyme essential oil, nisin, and their combination to control Listeria monocytogenes inoculated in minced fish meat. Food Control 2014, 35, 177–183. [Google Scholar] [CrossRef]
- Baranauskien, R.; Venskutonis, P.R.; Dewettinck, K.; Verhe, R. Properties of oregano (Origanum vulgare L.), citronella (Cymbopogonnardus G.) and marjoram (Majorana hortensis L.) flavors encapsulated into milk protein-based matrices. Food Res. Int. 2006, 39, 413–425. [Google Scholar] [CrossRef]
- Skandamis, P.; Tsigarida, E.; Nychas, G.J. Ecophysiological attributes of Salmonella typhimurium in liquid culture and within a gelatin gel with or without the addition of oregano essential oil. World J. Microbiol. Biotechnol. 2000, 16, 31–35. [Google Scholar] [CrossRef]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef] [PubMed]
- Mølbak, K.; Olsen, J.E.; Wegener, H.C. Salmonella infections. In Foodborne Infections and Intoxications; Cliver, D.O., Potter, M., Riemann, H.P., Eds.; Academic Press: Washington, DC, USA, 2011; pp. 57–136. [Google Scholar]
- Pagotto, F.; Corneau, N.; Farber, J. Listeria monocytogenes. In Foodborne Infections and Intoxications; Cliver, D.O., Potter, M., Riemann, R.P., Eds.; Academic Press: Washington, DC, USA, 2011; pp. 312–340. [Google Scholar]
- Gandhi, M.; Chikindas, M.L. Listeria: A foodborne pathogen that knows how to survive. Int. J. Food Microbiol. 2007, 113, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Skandamis, P.N.; Nychas, G.J.E. Development and evaluation of a model predicting the survival of Escherichia coli O157: H7 NCTC 12900 in homemade eggplant salad at various temperatures, pHs, and oregano essential oil concentrations. Appl. Environ. Microbiol. 2000, 66, 1646–1653. [Google Scholar] [CrossRef] [PubMed]
- Negi, P.S. Plant extracts for the control of bacterial growth: Efficacy, stability and safety issues for food application. Int. J. Food Microbiol. 2012, 156, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Cassens, R.G. Composition and safety of cured meats in the USA. Food Chem. 1997, 59, 561–566. [Google Scholar] [CrossRef]
- Gonzalez, B.; Díez, V. The effect of nitrite and starter culture on microbiological quality of “chorizo”—A Spanish dry cured sausage. Meat Sci. 2002, 60, 295–298. [Google Scholar] [CrossRef]
- Dickson, J.S.; Cutter, C.G.; Siragusa, G.R. Antimicrobial effects of trisodium phosphate against bacteria attached to beef tissue. J. Food Prot. 1994, 57, 952–955. [Google Scholar] [CrossRef]






| Microorganisms | Strain | Source 1 |
|---|---|---|
| Salmonella spp. | MPI-B-S07 | Chouriço batter |
| EDS-E-S02 | Environment of meat products preparation | |
| L. monocytogenes | EDS-B-LM02 | Chouriço batter |
| MPI-E-LM01 | Environment of meat products preparation |
| Garlic essential oil | Oregano essential oil | ||
|---|---|---|---|
| Compounds | % | Compounds | % |
| Diallyl sulfide | 8.36 | á-Pinene | 0.27 |
| Methyl allyl disulfide | 2.76 | á-Terpinolene | 0.27 |
| diallyl disulfide | 18.86 | p-cymene | 0.99 |
| Methyl allyltrisulfide | 9.04 | á-Terpinene | 1.29 |
| 1,3,5 trithiane | 0.75 | Linalool | 0.21 |
| 2-vinil-1,3-dithiane | 0.75 | Thymol | 93.34 |
| diallyltrisulfide | 33.82 | Trans-Caryophyllene | 0.72 |
| Hexamethylenesulfoxide | 0.24 | Germacrene | 0.13 |
| Methyl allyl disulfide | 2.75 | ||
| Diallyltetrasulfide | 10.97 | ||
| Essential Oil | Assay | Listeria monocytogenes | Salmonella spp. |
|---|---|---|---|
| garlic | DDA (mm) | 10.5 ± 1.6 | 15.03 ± 2.6 |
| MIC (%) | 2 | 0.0125 | |
| MBC (%) | 4 | 2 | |
| oregano | DDA (mm) | 46.5 ± 3.2 | 36.4 ± 1.3 |
| MIC (%) | 0.005 | 0.005 | |
| MBC (%) | >0.005 | >0.005 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
García-Díez, J.; Alheiro, J.; Pinto, A.L.; Soares, L.; Falco, V.; Fraqueza, M.J.; Patarata, L. Influence of Food Characteristics and Food Additives on the Antimicrobial Effect of Garlic and Oregano Essential Oils. Foods 2017, 6, 44. https://doi.org/10.3390/foods6060044
García-Díez J, Alheiro J, Pinto AL, Soares L, Falco V, Fraqueza MJ, Patarata L. Influence of Food Characteristics and Food Additives on the Antimicrobial Effect of Garlic and Oregano Essential Oils. Foods. 2017; 6(6):44. https://doi.org/10.3390/foods6060044
Chicago/Turabian StyleGarcía-Díez, Juan, Joana Alheiro, Ana Luisa Pinto, Luciana Soares, Virgilio Falco, Maria João Fraqueza, and Luis Patarata. 2017. "Influence of Food Characteristics and Food Additives on the Antimicrobial Effect of Garlic and Oregano Essential Oils" Foods 6, no. 6: 44. https://doi.org/10.3390/foods6060044
APA StyleGarcía-Díez, J., Alheiro, J., Pinto, A. L., Soares, L., Falco, V., Fraqueza, M. J., & Patarata, L. (2017). Influence of Food Characteristics and Food Additives on the Antimicrobial Effect of Garlic and Oregano Essential Oils. Foods, 6(6), 44. https://doi.org/10.3390/foods6060044






