Molecular Insights into Bioactive Interactions Within Protein- and Polysaccharide-Based Colloids: Implications for Stability, Functionality, and Bioavailability
Abstract
1. Introduction
2. Fundamentals of Colloid-Bioactive Compound Interactions
3. Protein-Polyphenol Interactions
3.1. Impacts of Polyphenol Interactions on the Structure and Functionality of Proteins
3.1.1. Conformational Alterations
3.1.2. Functional Properties
3.2. Changes in Biological Properties of Polyphenols
3.2.1. Bioaccessibility During Gastrointestinal Digestion
3.2.2. Antioxidant Activity
4. Polysaccharide–Carotenoid Interactions
5. Protein–Polysaccharide-Based Hybrid Colloidal Systems
6. Advanced Characterization Techniques
6.1. Spectroscopic Analysis
6.2. Microscopic and Scattering Methods
6.3. Computational and Integrative Approaches to Colloid-Bioactive Interactions
7. Effects on Nutrient Delivery and Bioactivity
8. Challenges and Future Perspectives
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bazana, M.T.; Codevilla, C.F.; de Menezes, C.R. Nanoencapsulation of Bioactive Compounds: Challenges and Perspectives. Curr. Opin. Food Sci. 2019, 26, 47–56. [Google Scholar] [CrossRef]
- Singh, A.; Prakash, A.; Choudhary, R. Bioactive Components Having Antimicrobial and Anticancerous Properties: A Review. In Bioactive Components; Springer: Singapore, 2023; pp. 271–299. [Google Scholar] [CrossRef]
- Liu, F.; McClements, D.J.; Ma, C.; Liu, X. Novel Colloidal Food Ingredients: Protein Complexes and Conjugates. Annu. Rev. Food Sci. Technol. 2023, 14, 35–61. [Google Scholar] [CrossRef]
- Cheng, F.; Chen, M.; Duan, Z.; Zou, Y.; He, Y.; Zeng, F.; Yuan, Y.; Fu, T.; Tu, H.; Li, R.; et al. Fabrication, Characterization, and Bioactivity of Self-Assembled Carrier-Free Colloidal Dispersions from Citrus × Limon ‘Rosso’ Essential Oil and Tea Polyphenols. Food Chem. 2024, 457, 140058. [Google Scholar] [CrossRef]
- Chen, Y.; Yao, M.; Peng, S.; Fang, Y.; Wan, L.; Shang, W.; Xiang, D.; Zhang, W. Development of Protein-Polyphenol Particles to Stabilize High Internal Phase Pickering Emulsions by Polyphenols’ Structure. Food Chem. 2023, 428, 136773. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Zhang, P.; Yan, W.; Wu, Y.; Jin, G.; Song, H.; An, F.; Wu, Y.; Li, X.; Luo, P.; et al. Mechanisms of Ultrasound-Synergized Polyphenol Enhancing the Foaming Properties of Ovalbumin: Focus on Interfacial Behavior and Microstructure. Ultrason. Sonochem. 2025, 122, 107605. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; He, D.; Li, B.; Lund, M.N.; Xing, Y.; Wang, Y.; Li, F.; Cao, X.; Liu, Y.; Chen, X.; et al. Engineering Polyphenols with Biological Functions via Polyphenol-Protein Interactions as Additives for Functional Foods. Trends Food Sci. Technol. 2021, 110, 470–482. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, X.; Sang, S.; McClements, D.J.; Chen, L.; Long, J.; Jiao, A.; Jin, Z.; Qiu, C. Polyphenols as Plant-Based Nutraceuticals: Health Effects, Encapsulation, Nano-Delivery, and Application. Foods 2022, 11, 2189. [Google Scholar] [CrossRef]
- Antony, A.; Farid, M. Effect of Temperatures on Polyphenols during Extraction. Appl. Sci. 2022, 12, 2107. [Google Scholar] [CrossRef]
- Wang, Y.; Shen, J.; Zou, B.; Zhang, L.; Xu, X.; Wu, C. Determination of the Critical pH for Unfolding Water-Soluble Cod Protein and Its Effect on Encapsulation Capacities. Food Res. Int. 2023, 174, 113621. [Google Scholar] [CrossRef]
- Sun, J.; Liu, T.; Zhang, F.; Huang, Y.; Zhang, Y.; Xu, B. Tea Polyphenols on Emulsifying and Antioxidant Properties of Egg White Protein at Acidic and Neutral pH Conditions. LWT 2022, 153, 112537. [Google Scholar] [CrossRef]
- Cuomo, F.; Iacovino, S.; Sacco, P.; De Leonardis, A.; Ceglie, A.; Lopez, F. Progress in Colloid Delivery Systems for Protection and Delivery of Phenolic Bioactive Compounds: Two Study Cases—Hydroxytyrosol and Curcumin. Molecules 2022, 27, 921. [Google Scholar] [CrossRef]
- Wang, B.; Pham, L.B.; Adhikari, B. Complexation and Conjugation between Phenolic Compounds and Proteins: Mechanisms, Characterisation and Applications as Novel Encapsulants. Sustain. Food Technol. 2024, 2, 1206–1227. [Google Scholar] [CrossRef]
- Yan, S.; Regenstein, J.M.; Qi, B.; Li, Y. Construction of Protein-, Polysaccharide- and Polyphenol-Based Conjugates as Delivery Systems. Crit. Rev. Food Sci. Nutr. 2025, 65, 1363–1381. [Google Scholar] [CrossRef]
- Ran, Y.; Li, F.; Xu, Z.; Zeng, K.; Ming, J. Recent Advances in Dietary Polyphenols (DPs): Antioxidant Activities, Nutrient Interactions, Delivery Systems, and Potential Applications. Food Funct. 2024, 15, 10213–10232. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, H.; Wang, Y.; Zhang, Z.; Wang, Q. Preparation, Characterization and Release Kinetics of a Multilayer Encapsulated Perilla frutescens L. Essential Oil Hydrogel Bead. Int. J. Biol. Macromol. 2023, 249, 124776. [Google Scholar] [CrossRef]
- Cheng, Y.; Chen, X.; Lin, Q.; Wang, J.; Chen, Y.; Mo, H.; Chen, M.; Xu, Z.; Wen, L.; Jiao, Y. Improvement Effect of Different Polysaccharides on the Functional Properties of Curcumin-Loaded Rice Protein Hydrolysate Nanoparticles. LWT 2024, 204, 116471. [Google Scholar] [CrossRef]
- Umar, M.; Zafar, S.; Fikry, M.; Medhe, S.V.; Rungraeng, N. Non-Covalent Complexes of Plant-Based Proteins-Polysaccharides and Their Applications to Stabilize the Delivery Systems for Bioactive Compounds. Food Rev. Int. 2025, 18, 1–31. [Google Scholar] [CrossRef]
- Manzoor, M.; Tchameni, Z.F.N.; Bhat, Z.F.; Jaiswal, A.K.; Jaglan, S. Recent Insights on the Conformational Changes, Functionality, and Physiological Properties of Plant-Based Protein-Polyphenol Conjugates. Food Bioprocess Technol. 2024, 17, 2131–2154. [Google Scholar] [CrossRef]
- Ke, Y.; Deng, L.; Dai, T.; Xiao, M.; Chen, M.; Liang, R.; Liu, W.; Liu, C.; Chen, J. Effects of Cell Wall Polysaccharides on the Bioaccessibility of Carotenoids, Polyphenols, and Minerals: An Overview. Crit. Rev. Food Sci. Nutr. 2023, 63, 11385–11398. [Google Scholar] [CrossRef] [PubMed]
- Meng, Y.; Qiu, C.; Li, X.; McClements, D.J.; Sang, S.; Jiao, A.; Jin, Z. Polysaccharide-Based Nano-Delivery Systems for Encapsulation, Delivery, and pH-Responsive Release of Bioactive Ingredients. Crit. Rev. Food Sci. Nutr. 2024, 64, 187–201. [Google Scholar] [CrossRef]
- Poluri, K.M.; Gulati, K.; Sarkar, S. Structural and Functional Properties of Proteins. In Protein-Protein Interactions: Principles and Techniques: Volume I; Springer: Singapore, 2021; pp. 1–60. ISBN 978-981-16-1594-8. [Google Scholar]
- Horita, K.; Kameda, T.; Suga, H.; Hirano, A. Molecular Mechanism of the Interactions between Coffee Polyphenols and Milk Proteins. Food Res. Int. 2025, 202, 115573. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhou, H.; Tan, Y.; Muriel Mundo, J.L.; McClements, D.J. Comparison of Plant-Based Emulsifier Performance in Water-in-Oil-in-Water Emulsions: Soy Protein Isolate, Pectin and Gum Arabic. J. Food Eng. 2021, 307, 110625. [Google Scholar] [CrossRef]
- Estupiñan-Amaya, M.; Fuenmayor, C.A.; López-Córdoba, A. Evaluation of Mixtures of Maltodextrin and Gum Arabic for the Encapsulation of Andean Blueberry (Vaccinium Meridionale) Juice by Freeze–Drying. Int. J. Food Sci. Technol. 2022, 57, 7379–7390. [Google Scholar] [CrossRef]
- Hashemzadeh, H.; Hanafi-Bojd, M.Y.; Iranshahy, M.; Zarban, A.; Raissi, H. The Combination of Polyphenols and Phospholipids as an Efficient Platform for Delivery of Natural Products. Sci. Rep. 2023, 13, 2501. [Google Scholar] [CrossRef]
- Abdullah; Liu, L.; Javed, H.U.; Xiao, J. Engineering Emulsion Gels as Functional Colloids Emphasizing Food Applications: A Review. Front. Nutr. 2022, 9, 890188. [Google Scholar] [CrossRef]
- Ashfaq, A.; Jahan, K.; Islam, R.U.; Younis, K. Protein-Based Functional Colloids and Their Potential Applications in Food: A Review. LWT 2022, 154, 112667. [Google Scholar] [CrossRef]
- Bonechi, C.; Donati, A.; Tamasi, G.; Pardini, A.; Rostom, H.; Leone, G.; Lamponi, S.; Consumi, M.; Magnani, A.; Rossi, C. Chemical Characterization of Liposomes Containing Nutraceutical Compounds: Tyrosol, Hydroxytyrosol and Oleuropein. Biophys. Chem. 2019, 246, 25–34. [Google Scholar] [CrossRef]
- Kalaitzaki, A.; Emo, M.; Stébé, M.J.; Xenakis, A.; Papadimitriou, V. Biocompatible Nanodispersions as Delivery Systems of Food Additives: A Structural Study. Food Res. Int. 2013, 54, 1448–1454. [Google Scholar] [CrossRef]
- Esperón-Rojas, A.A.; Baeza-Jiménez, R.; Santos-Luna, D.; Velasco-Rodríguez, L.D.C.; Ochoa-Rodríguez, L.R.; García, H.S. Bioavailability of Curcumin in Nanoemulsions Stabilized with Mono- and Diacylglycerols Structured with Conjugated Linoleic Acid and n-3 Fatty Acids. Biocatal. Agric. Biotechnol. 2020, 26, 101638. [Google Scholar] [CrossRef]
- Pool, H.; Mendoza, S.; Xiao, H.; Julian McClements, D. Encapsulation and Release of Hydrophobic Bioactive Components in Nanoemulsion-Based Delivery Systems: Impact of Physical Form on Quercetin Bioaccessibility. Food Func. 2012, 4, 162–174. [Google Scholar] [CrossRef]
- Han, G.; Duan, X.; Jiang, B.; Li, Y.; Li, B.; Yang, J.; Pan, S.; Liu, F. Emulsifying Properties, in Vitro Digestive Characteristics, and β-Carotene Bioaccessibility of Mandarin Peel Pectin Emulsions Prepared with Different Carrier Oil Phases. Int. J. Biol. Macromol. 2023, 242, 124961. [Google Scholar] [CrossRef]
- Mehnert, W.; Mäder, K. Solid Lipid Nanoparticles: Production, Characterization and Applications. Adv. Drug Deliv. Rev. 2012, 64, 83–101. [Google Scholar] [CrossRef]
- Andreazza, R.; Morales, A.; Pieniz, S.; Labidi, J. Gelatin-Based Hydrogels: Potential Biomaterials for Remediation. Polymers 2023, 15, 1026. [Google Scholar] [CrossRef] [PubMed]
- Ashwar, B.A.; Gani, A. Noncovalent Interactions of Sea Buckthorn Polyphenols with Casein and Whey Proteins: Effect on the Stability, Antioxidant Potential, and Bioaccessibility of Polyphenols. ACS Food Sci. Technol. 2021, 1, 1206–1214. [Google Scholar] [CrossRef]
- Inbaraj, S.; Sridhar, K.; Sharma, M.; Said, N.S.; Fola Olawuyi, I.; Lee, W.Y. Pectin Hydrogels: Gel-Forming Behaviors, Mechanisms, and Food Applications. Gels 2023, 9, 732. [Google Scholar] [CrossRef]
- Alkekhia, D.; Hammond, P.T.; Shukla, A. Layer-by-Layer Biomaterials for Drug Delivery. Annu. Rev. Biomed. Eng. 2020, 22, 1–24. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, Y.; Gao, X.; Fu, J.; Hu, L. Application of Cyclodextrin in Food Industry. Crit. Rev. Food Sci. Nutr. 2022, 62, 2627–2640. [Google Scholar] [CrossRef] [PubMed]
- Ozdal, T.; Capanoglu, E.; Altay, F. A Review on Protein–Phenolic Interactions and Associated Changes. Food Res. Int. 2013, 51, 954–970. [Google Scholar] [CrossRef]
- Zhang, H.; Yu, D.; Sun, J.; Liu, X.; Jiang, L.; Guo, H.; Ren, F. Interaction of Plant Phenols with Food Macronutrients: Characterisation and Nutritional–Physiological Consequences. Nutr. Res. Rev. 2014, 27, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Bohin, M.C.; Vincken, J.P.; Westphal, A.H.; Tripp, A.M.; Dekker, P.; Van Der Hijden, H.T.W.M.; Gruppen, H. Interaction of Flavan-3-Ol Derivatives and Different Caseins Is Determined by More than Proline Content and Number of Proline Repeats. Food Chem. 2014, 158, 408–416. [Google Scholar] [CrossRef]
- Yildirim-Elikoglu, S.; Erdem, Y.K. Interactions between Milk Proteins and Polyphenols: Binding Mechanisms, Related Changes, and the Future Trends in the Dairy Industry. Food Rev. Int. 2018, 34, 665–697. [Google Scholar] [CrossRef]
- Hu, Y.; Zhou, C.; Du, L.; Zhan, F.; Sun, Y.; Wu, Z.; Pan, D. Phenolic Structure Dependent Interaction onto Modified Goose Liver Protein Enhanced by pH Shifting: Modulations on Protein Interfacial and Emulsifying Properties. Int. J. Biol. Macromol. 2023, 253, 126810. [Google Scholar] [CrossRef]
- Kaspchak, E.; Goedert, A.C.; Igarashi-Mafra, L.; Mafra, M.R. Effect of Divalent Cations on Bovine Serum Albumin (BSA) and Tannic Acid Interaction and Its Influence on Turbidity and in Vitro Protein Digestibility. Int. J. Biol. Macromol. 2019, 136, 486–492. [Google Scholar] [CrossRef]
- Jakobek, L. Interactions of Polyphenols with Carbohydrates, Lipids and Proteins. Food Chem. 2015, 175, 556–567. [Google Scholar] [CrossRef]
- Quan, T.H.; Benjakul, S.; Sae-leaw, T.; Balange, A.K.; Maqsood, S. Protein-Polyphenol Conjugates: Antioxidant Property, Functionalities and Their Applications. Trends Food Sci. Technol. 2019, 91, 507–517. [Google Scholar] [CrossRef]
- Le Bourvellec, C.; Renard, C.M.G.C. Interactions between Polyphenols and Macromolecules: Quantification Methods and Mechanisms. Crit. Rev. Food Sci. Nutr. 2012, 52, 213–248. [Google Scholar] [CrossRef]
- Mao, T.; Akshit, F.N.U.; Matiwalage, I.; Sasidharan, S.; Alvarez, C.M.; Wescombe, P.; Mohan, M.S. Preferential Binding of Polyphenols in Blackcurrant Extracts with Milk Proteins and the Effects on the Bioaccessibility and Antioxidant Activity of Polyphenols. Foods 2024, 13, 515. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Ma, C.; Gao, Y.; McClements, D.J. Food-Grade Covalent Complexes and Their Application as Nutraceutical Delivery Systems: A Review. Compr. Rev. Food Sci. Food Saf. 2017, 16, 76–95. [Google Scholar] [CrossRef] [PubMed]
- Sui, X.; Sun, H.; Qi, B.; Zhang, M.; Li, Y.; Jiang, L. Functional and Conformational Changes to Soy Proteins Accompanying Anthocyanins: Focus on Covalent and Non-Covalent Interactions. Food Chem. 2018, 245, 871–878. [Google Scholar] [CrossRef]
- Chao Song, Z.; Zhang, H.; Fei Niu, P.; Shi, L.S.; Yan Yang, X.; Hong Meng, Y.; Yu Wang, X.; Gong, T.; Rong Guo, Y. Fabrication of a Novel Antioxidant Emulsifier through Tuning the Molecular Interaction between Soy Protein Isolates and Young Apple Polyphenols. Food Chem. 2023, 420, 136110. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Li, Y.; Liu, H.; Li, P.; Du, B.; Xie, X.A.; Li, L. Covalent Conjugation of Inca Peanut Albumin and Polyphenols with Different Phenolic Hydroxyl Numbers through Laccase Catalysis to Improve Functional Properties. J. Sci. Food Agric. 2024, 104, 4028–4038. [Google Scholar] [CrossRef]
- Shahidi, F.; Dissanayaka, C.S. Phenolic-Protein Interactions: Insight from in-Silico Analyses—A Review. Food Prod. Process. Nutr. 2023, 5, 2. [Google Scholar] [CrossRef]
- Poklar Ulrih, N. Analytical Techniques for the Study of Polyphenol–Protein Interactions. Crit. Rev. Food Sci. Nutr. 2017, 57, 2144–2161. [Google Scholar] [CrossRef]
- Ma, D.; Yang, B.; Zhao, J.; Yuan, D.; Li, Q. Advances in Protein-Based Microcapsules and Their Applications: A Review. Int. J. Biol. Macromol. 2024, 263, 129742. [Google Scholar] [CrossRef]
- Zhou, F.; Lu, B.; Chen, X.; Jia, Z.; Tao, F.; Peng, J. Interaction of Major Tea Polyphenols with Bovine Milk Proteins and Its Effect on in Vitro Bioaccessibility of Tea Polyphenols. Food Chem. 2025, 475, 143341. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, C.; Shen, X.; McClements, D.J.; Liu, X.; Liu, F. Effects of Combined Hot Alkaline and pH-Shift Treatments on Structure and Functionality of Legume Protein-EGCG Conjugates: Soybean-, Pea-, and Chickpea Protein-EGCG Systems. Food Hydrocoll. 2025, 158, 110424. [Google Scholar] [CrossRef]
- Guo, X.; Wei, Y.; Liu, P.; Deng, X.; Zhu, X.; Wang, Z.; Zhang, J. Study of Four Polyphenol-Coregonus Peled (C. Peled) Myofibrillar Protein Interactions on Protein Structure and Gel Properties. Food Chem. X 2024, 21, 101063. [Google Scholar] [CrossRef]
- Shi, J.; Cui, Y.F.; Zhou, G.; Li, N.; Sun, X.; Wang, X.; Xu, N. Covalent Interaction of Soy Protein Isolate and Chlorogenic Acid: Effect on Protein Structure and Functional Properties. LWT 2022, 170, 114081. [Google Scholar] [CrossRef]
- Chima, B.; Mathews, P.; Morgan, S.; Johnson, S.A.; Van Buiten, C.B. Physicochemical Characterization of Interactions between Blueberry Polyphenols and Food Proteins from Dairy and Plant Sources. Foods 2022, 11, 2846. [Google Scholar] [CrossRef]
- Meng, Y.; Wei, Z.; Xue, C. Deciphering the Interaction Mechanism and Binding Mode between Chickpea Protein Isolate and Flavonoids Based on Experimental Studies and Molecular Simulation. Food Chem. 2023, 429, 136848. [Google Scholar] [CrossRef]
- Gu, M.; Shi, J.; Zhang, B.; Wang, X.; Wang, X. Covalent Modification of Soy Protein Isolates by Hydroxytyrosol: Effects on Structural and Functional Properties of Adducts. LWT 2024, 198, 116041. [Google Scholar] [CrossRef]
- GopikaJayaprakash; Chawla, P.; Sridhar, K.; Bains, A. Interactions of Legume Phenols-Rice Protein Concentrate towards Improving Vegan Food Quality: Development of a Protein-Phenols Enriched Fruit Smoothie. Food Res. Int. 2023, 171, 113075. [Google Scholar] [CrossRef] [PubMed]
- He, W.; He, K.; Liu, X.; Ye, L.; Lin, X.; Ma, L.; Yang, P.; Wu, X. Modulating the Allergenicity and Functional Properties of Peanut Protein by Covalent Conjugation with Polyphenols. Food Chem. 2023, 415, 135733. [Google Scholar] [CrossRef]
- Man, H.; Jia, Y.; Song, H.; Yan, X.; Zhang, D.; Huang, Y.; Qi, B.; Zhang, S.; Li, Y. Effects of κ-Carrageenan Addition and Chlorogenic Acid Covalent Crosslinking on Protein Conformation and Gelling Properties of Soy Protein Hydrogels. LWT 2023, 174, 114434. [Google Scholar] [CrossRef]
- Guo, K.; Zhou, G.; Lok, U.S.; Wang, X.; Jiang, L. Improving Interface-Related Functions and Antioxidant Activities of Soy Protein Isolate by Covalent Conjugation with Chlorogenic Acid. J. Food Meas. Charact. 2022, 16, 202–213. [Google Scholar] [CrossRef]
- Han, S.; Cui, F.; McClements, D.J.; Xu, X.; Ma, C.; Wang, Y.; Liu, X.; Liu, F. Structural Characterization and Evaluation of Interfacial Properties of Pea Protein Isolate–EGCG Molecular Complexes. Foods 2022, 11, 2895. [Google Scholar] [CrossRef]
- Hao, L.; Sun, J.; Pei, M.; Zhang, G.; Li, C.; Li, C.; Ma, X.; He, S.; Liu, L. Impact of Non-Covalent Bound Polyphenols on Conformational, Functional Properties and in Vitro Digestibility of Pea Protein. Food Chem. 2022, 383, 132623. [Google Scholar] [CrossRef]
- Geng, M.; Feng, X.; Yang, H.; Wu, X.; Li, L.; Li, Y.; Teng, F. Comparison of Soy Protein Isolate-(–)-Epigallocatechin Gallate Complexes Prepared by Mixing, Chemical Polymerization, and Ultrasound Treatment. Ultrason. Sonochem. 2022, 90, 106172. [Google Scholar] [CrossRef]
- Guo, Y.; Bao, Y.H.; Sun, K.F.; Chang, C.; Liu, W.F. Effects of Covalent Interactions and Gel Characteristics on Soy Protein-Tannic Acid Conjugates Prepared under Alkaline Conditions. Food Hydrocoll. 2021, 112, 106293. [Google Scholar] [CrossRef]
- Li, C.; Dai, T.; Chen, J.; Li, X.; Li, T.; Liu, C.; McClements, D.J. Protein-Polyphenol Functional Ingredients: The Foaming Properties of Lactoferrin Are Enhanced by Forming Complexes with Procyanidin. Food Chem. 2021, 339, 128145. [Google Scholar] [CrossRef]
- de Morais, F.P.R.; Pessato, T.B.; Rodrigues, E.; Peixoto Mallmann, L.; Mariutti, L.R.B.; Netto, F.M. Whey Protein and Phenolic Compound Complexation: Effects on Antioxidant Capacity before and after in Vitro Digestion. Food Res. Int. 2020, 133, 109104. [Google Scholar] [CrossRef]
- Shi, W.; Xie, H.; Ouyang, K.; Wang, S.; Xiong, H.; Woo, M.W.; Zhao, Q. The Effect of Rice Protein-Polyphenols Covalent and Non-Covalent Interactions on the Structure, Functionality and in Vitro Digestion Properties of Rice Protein. Food Chem. 2024, 450, 139241. [Google Scholar] [CrossRef]
- Huang, X.; Yan, C.; Lin, M.; He, C.; Xu, Y.; Huang, Y.; Zhou, Z. The Effects of Conjugation of Walnut Protein Isolate with Polyphenols on Protein Solubility, Antioxidant Activity, and Emulsifying Properties. Food Res. Int. 2022, 161, 111910. [Google Scholar] [CrossRef]
- Han, J.; Du, Y.; Yan, J.; Jiang, X.; Wu, H.; Zhu, B. Effect of Non-Covalent Binding of Phenolic Derivatives with Scallop (Patinopecten yessoensis) Gonad Protein Isolates on Protein Structure and in Vitro Digestion Characteristics. Food Chem. 2021, 357, 129690. [Google Scholar] [CrossRef]
- Faber, I.; Pouvreau, L.; Jan van der Goot, A.; Keppler, J. Modulating Commercial Pea Protein Gel Properties through the Addition of Phenolic Compounds. Food Hydrocoll. 2024, 154, 110123. [Google Scholar] [CrossRef]
- Milinčić, D.D.; Salević-Jelić, A.S.; Kostić, A.Ž.; Stanojević, S.P.; Nedović, V.; Pešić, M.B. Food Nanoemulsions: How Simulated Gastrointestinal Digestion Models, Nanoemulsion, and Food Matrix Properties Affect Bioaccessibility of Encapsulated Bioactive Compounds. Crit. Rev. Food Sci. Nutr. 2024, 64, 8091–8113. [Google Scholar] [CrossRef]
- Sęczyk, Ł.; Jariene, E.; Sugier, D.; Kołodziej, B. Effects of the Dose of Administration, Co-Antioxidants, Food Matrix, and Digestion-Related Factors on the in Vitro Bioaccessibility of Rosmarinic Acid—A Model Study. Food Chem. 2024, 449, 139201. [Google Scholar] [CrossRef] [PubMed]
- Sęczyk, Ł.; Sugier, D.; Świeca, M.; Gawlik-Dziki, U. The Effect of in Vitro Digestion, Food Matrix, and Hydrothermal Treatment on the Potential Bioaccessibility of Selected Phenolic Compounds. Food Chem. 2021, 344, 128581. [Google Scholar] [CrossRef] [PubMed]
- Lippolis, T.; Cofano, M.; Caponio, G.R.; De Nunzio, V.; Notarnicola, M. Bioaccessibility and Bioavailability of Diet Polyphenols and Their Modulation of Gut Microbiota. Int. J. Mol. Sci. 2023, 24, 3813. [Google Scholar] [CrossRef]
- Guo, F.; Peng, L.; Xiong, H.; Tsao, R.; Zhang, H.; Jiang, L.; Sun, Y. Bioaccessibility and Transport of Lentil Hull Polyphenols in Vitro, and Their Bioavailability and Metabolism in Rats. Food Res. Int. 2023, 167, 112634. [Google Scholar] [CrossRef]
- Reis, A.; Perez-Gregorio, R.; Mateus, N.; de Freitas, V. Interactions of Dietary Polyphenols with Epithelial Lipids: Advances from Membrane and Cell Models in the Study of Polyphenol Absorption, Transport and Delivery to the Epithelium. Crit. Rev. Food Sci. Nutr. 2021, 61, 3007–3030. [Google Scholar] [CrossRef]
- Bhuyan, U.; Handique, J.G. Plant Polyphenols as Potent Antioxidants: Highlighting the Mechanism of Antioxidant Activity and Synthesis/Development of Some Polyphenol Conjugates. Stud. Nat. Prod. Chem. 2022, 75, 243–266. [Google Scholar] [CrossRef]
- Gebregziabher, B.S.; Gebremeskel, H.; Debesa, B.; Ayalneh, D.; Mitiku, T.; Wendwessen, T.; Habtemariam, E.; Nur, S.; Getachew, T. Carotenoids: Dietary Sources, Health Functions, Biofortification, Marketing Trend and Affecting Factors—A Review. J. Agric. Food Res. 2023, 14, 100834. [Google Scholar] [CrossRef]
- Gao, J.; Qiu, Y.; Chen, F.; Zhang, L.; Wei, W.; An, X.; Zhu, Q. Pomelo Peel Derived Nanocellulose as Pickering Stabilizers: Fabrication of Pickering Emulsions and Their Potential as Sustained-Release Delivery Systems for Lycopene. Food Chem. 2023, 415, 135742. [Google Scholar] [CrossRef]
- Zhao, J.; Liu, M.; Li, S.; Gu, L. Optimization of Lycopene Spray Drying Encapsulation in Basil Seed Gum: Boosting Bioavailability and Mayonnaise Stability. Int. J. Biol. Macromol. 2024, 282, 136572. [Google Scholar] [CrossRef]
- Wu, Z.; Tang, X.; Liu, S.; Li, S.; Zhao, X.; Wang, Y.; Wang, X.; Li, H. Mechanism Underlying Joint Loading and Controlled Release of β-Carotene and Curcumin by Octenylsuccinated Gastrodia Elata Starch Aggregates. Food Res. Int. 2023, 172, 113136. [Google Scholar] [CrossRef] [PubMed]
- Bahrololoumi, S.; Khosroshahi, E.D.; Razavi, S.H.; Kiani, H. A Novel Approach for Solubility and Bioavailability Enhancement of Canthaxanthin Obtained from Dietzia Natronolimnaea HS-1 by Canthaxanthin-V-Amylose Complex. Food Bioprocess Technol. 2025, 18, 2325–2339. [Google Scholar] [CrossRef]
- Liu, M.; Liu, X.; Jiang, X.; Chen, J.; Bi, J. Exploring the Potential of Peach Gum Polysaccharides in β-Carotene Emulsions Encapsulation during Storage. Bioact. Carbohydr. Diet. Fibre 2024, 32, 100461. [Google Scholar] [CrossRef]
- Li, Y.; Fei, S.; Yu, D.; Zhang, L.; Li, J.; Liu, R.; Tan, M. Preparation and Evaluation of Undaria Pinnatifida Nanocellulose in Fabricating Pickering Emulsions for Protection of Astaxanthin. Foods 2022, 11, 876. [Google Scholar] [CrossRef]
- Zhang, L.; Liao, W.; Tong, Z.; Wang, Y.; Liu, J.; Mao, L.; Yuan, F.; Gao, Y. Impact of Biopolymer-Surfactant Interactions on the Particle Aggregation Inhibition of β-Carotene in High Loaded Microcapsules: Spontaneous Dispersibility and in Vitro Digestion. Food Hydrocoll. 2023, 134, 108043. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhang, F.; Chen, M.; Liu, F.; Zheng, B.; Miao, W.; Gao, H.; Zhou, R. Cellulose Nanofibrils-Stabilized Food-Grade Pickering Emulsions: Clarifying Surface Charge’s Contribution and Advancing Stabilization Mechanism Understanding. Food Hydrocoll. 2024, 152, 109920. [Google Scholar] [CrossRef]
- Wang, X.; Huang, L.; Zhang, C.; Deng, Y.; Xie, P.; Liu, L.; Cheng, J. Research Advances in Chemical Modifications of Starch for Hydrophobicity and Its Applications: A Review. Carbohydr. Polym. 2020, 240, 116292. [Google Scholar] [CrossRef] [PubMed]
- Mohan, K.; Makebe, C.W.; Jayamurthy, P.; Nisha, P. Red Palm Oil Pickering Emulsion with Pectin Yields Improved in Vitro Beta Carotene Bioaccessibility and Oil Stability: Physicochemical Characterization and Shelf Stability Studies. J. Food Process. Preserv. 2022, 46, e16930. [Google Scholar] [CrossRef]
- Gajic, I.M.S.; Savic, I.M.; Gajic, D.G.; Dosic, A. Ultrasound-Assisted Extraction of Carotenoids from Orange Peel Using Olive Oil and Its Encapsulation in Ca-Alginate Beads. Biomolecules 2021, 11, 225. [Google Scholar] [CrossRef]
- Gherasim, C.E.; Focşan, M.; Ciont, C.; Bunea, A.; Rugină, D.; Pintea, A. Stability and Bioaccessibility of Carotenoids from Sea Buckthorn Pomace Encapsulated in Alginate Hydrogel Beads. Nutrients 2024, 16, 2726. [Google Scholar] [CrossRef]
- Kyriakoudi, A.; Klimantakis, K.; Kalaitzis, P.; Biliaderis, C.G.; Mourtzinos, I. Enrichment of Sunflower Oil with Tomato Carotenoids and Its Encapsulation in Ca-Alginate Beads: Preparation, Characterization and Chemical Stability upon in Vitro Digestion. Food Hydrocoll. 2024, 151, 109855. [Google Scholar] [CrossRef]
- Xu, P.; Song, J.; Dai, Z.; Xu, Y.; Li, D.; Wu, C. Effect of Ca2+ Cross-Linking on the Properties and Structure of Lutein-Loaded Sodium Alginate Hydrogels. Int. J. Biol. Macromol. 2021, 193, 53–63. [Google Scholar] [CrossRef]
- Wang, Y.; Shen, Z.; Wang, H.; Song, Z.; Yu, D.; Li, G.; Liu, X.; Liu, W. Progress in Research on Metal Ion Crosslinking Alginate-Based Gels. Gels 2024, 11, 16. [Google Scholar] [CrossRef]
- de Andrades, E.O.; da Costa, J.M.A.R.; de Lima Neto, F.E.M.; de Araujo, A.R.; de Oliveira Silva Ribeiro, F.; Vasconcelos, A.G.; de Jesus Oliveira, A.C.; Sobrinho, J.L.S.; de Almeida, M.P.; Carvalho, A.P.; et al. Acetylated Cashew Gum and Fucan for Incorporation of Lycopene Rich Extract from Red Guava (Psidium guajava L.) in Nanostructured Systems: Antioxidant and Antitumor Capacity. Int. J. Biol. Macromol. 2021, 191, 1026–1037. [Google Scholar] [CrossRef]
- González-Peña, M.A.; Lozada-Ramírez, J.D.; Ortega-Regules, A.E. Antioxidant Activities of Spray-Dried Carotenoids Using Maltodextrin-Arabic Gum as Wall Materials. Bull. Natl. Res. Cent. 2021, 45, 58. [Google Scholar] [CrossRef]
- Santos, J.; Trujillo-Cayado, L.A.; Barquero, M.; Calero, N. Influence of Type and Concentration of Biopolymer on β-Carotene Encapsulation Efficiency in Nanoemulsions Based on Linseed Oil. Polymers 2022, 14, 4640. [Google Scholar] [CrossRef]
- Meng, Y.; Hang, L.; Fang, S.; Li, Y.; Xu, X.; Zhang, F.; Chen, J. Fabrication of High-Acyl Gellan-Gum-Stabilized β-Carotene Emulsion: Physicochemical Properties and In Vitro Digestion Simulation. Foods 2022, 11, 1742. [Google Scholar] [CrossRef]
- Guo, Y.; Li, G.; Hao, J.; Li, J. Design of Astaxanthin Cold Application Gel Using Hydroxy Propyl Methyl Cellulose with Superior Release and Antioxidant Properties. Mater. Sci. Eng. B 2024, 299, 116982. [Google Scholar] [CrossRef]
- Fitri, I.A.; Mitbumrung, W.; Akanitkul, P.; Rungraung, N.; Kemsawasd, V.; Jain, S.; Winuprasith, T. Encapsulation of β-Carotene in Oil-in-Water Emulsions Containing Nanocellulose: Impact on Emulsion Properties, In Vitro Digestion, and Bioaccessibility. Polymers 2022, 14, 1414. [Google Scholar] [CrossRef]
- Bernice, Q.Q.L.; Chong, W.T.; Thilakarathna, R.C.N.; Tong, S.C.; Tang, T.K.; Phuah, E.T.; Lee, Y.Y. Palm-Based Nanofibrillated Cellulose (NFC) in Carotenoid Encapsulation and Its Incorporation into Margarine-like Reduced Fat Spread as Fat Replacer. J. Food Sci. 2024, 89, 5031–5046. [Google Scholar] [CrossRef]
- Basumatary, B.; Mahanta, C.L. Banana Rachis Nanocellulose: A Natural Stabilizer for Pickering Nanoemulsion and Its Potential as a Carrier of β-Carotene in Food Systems. J. Sci. Food Agric. 2025, 105, 3730–3741. [Google Scholar] [CrossRef]
- Xiao, S.; Ahn, D.U. Enhanced Lutein Stability under UV-Light and High Temperature by Loading It into Alginate-Chitosan Complex. LWT 2022, 164, 113663. [Google Scholar] [CrossRef]
- Yang, L.; Cao, X.; Gai, A.; Qiao, X.; Wei, Z.; Li, J.; Xu, J.; Xue, C. Chitosan/Guar Gum Nanoparticles to Stabilize Pickering Emulsion for Astaxanthin Encapsulation. LWT 2022, 165, 113727. [Google Scholar] [CrossRef]
- Wang, Y.; Zhou, Q.; Zheng, J.; Xiong, H.; Zhao, L.; Xu, Y.; Bai, C. Fabricating Pectin and Chitosan Double Layer Coated Liposomes to Improve Physicochemical Stability of Beta-Carotene and Alter Its Gastrointestinal Fate. Int. J. Biol. Macromol. 2023, 247, 125780. [Google Scholar] [CrossRef]
- Toragall, V.; Baskaran, V. Chitosan-Sodium Alginate-Fatty Acid Nanocarrier System: Lutein Bioavailability, Absorption Pharmacokinetics in Diabetic Rat and Protection of Retinal Cells against H2O2 Induced Oxidative Stress in Vitro. Carbohydr. Polym. 2021, 254, 117409. [Google Scholar] [CrossRef]
- Vakarelova, M.; Zanoni, F.; Donà, G.; Fierri, I.; Chignola, R.; Gorrieri, S.; Zoccatelli, G. Microencapsulation of Astaxanthin by Ionic Gelation: Effect of Different Gelling Polymers on the Carotenoid Load, Stability and Bioaccessibility. Int. J. Food Sci. Technol. 2023, 58, 2489–2497. [Google Scholar] [CrossRef]
- Wang, W.; Qin, Y.; Chen, K.; Gong, G.; Hou, Z.; Liu, L.; Li, C.; Zhang, G.; Yan, H.; Hu, Z.; et al. Lutein Encapsulated in Curdlan and Sodium Alginate by Emulsification/Internal Gelation: Construction, Characterization and Stability of Microcapsules. Int. J. Biol. Macromol. 2025, 318, 145313. [Google Scholar] [CrossRef]
- Celitan, E.; Gruskiene, R.; Kavleiskaja, T.; Sereikaite, J. β-Carotene—2-Hydroxypropyl-β-Cyclodextrin Complexes Coated with Pectin. Food Hydrocoll. 2022, 133, 107990. [Google Scholar] [CrossRef]
- Huang, J.; Zhang, S.; Liu, D.; Feng, X.; Wang, Q.; An, S.; Xu, M.; Chu, L. Preparation and Characterization of Astaxanthin-Loaded Microcapsules Stabilized by Lecithin-Chitosan-Alginate Interfaces with Layer-by-Layer Assembly Method. Int. J. Biol. Macromol. 2024, 268, 131909. [Google Scholar] [CrossRef]
- Feng, L.; Wu, J.; Cai, L.; Li, M.; Dai, Z.; Li, D.; Liu, C.; Zhang, M. Effects of Different Hydrocolloids on the Water Migration, Rheological and 3D Printing Characteristics of β-Carotene Loaded Yam Starch-Based Hydrogel. Food Chem. 2022, 393, 133422. [Google Scholar] [CrossRef]
- de Guzman, J.J.; Monserate, J.J.; Panajon, N.M.; Salazar, J.R.; Eugenio, P.J.G.; Sarong, M.M. Nanoencapsulation of Lycopene from Tomato Waste Using Chitosan and Alginate for Enhanced Stability and Antioxidant Activity. Int. J. Des. Nat. Ecodynamics 2024, 19, 1261–1267. [Google Scholar] [CrossRef]
- Chang, C.; Li, J.; Su, Y.; Gu, L.; Yang, Y.; Zhai, J. Protein Particle-Based Vehicles for Encapsulation and Delivery of Nutrients: Fabrication, Digestion, and Release Properties. Food Hydrocoll. 2022, 123, 106963. [Google Scholar] [CrossRef]
- Wang, Q.; Xiang, X.; Chen, B. Food Protein-Based Nanotechnology: From Delivery to Sensing Systems. Curr. Opin. Food Sci. 2024, 56, 101134. [Google Scholar] [CrossRef]
- Cao, T.; Wei, Z.; Xue, C. Recent Advances in Nutraceutical Delivery Systems Constructed by Protein–Polysaccharide Complexes: A Systematic Review. Compr. Rev. Food Sci. Food Saf. 2025, 24, e70115. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Zhou, Y.; Yue, W.; Qin, W.; Dong, H.; Vasanthan, T. Nanostructures of Protein-Polysaccharide Complexes or Conjugates for Encapsulation of Bioactive Compounds. Trends Food Sci. Technol. 2021, 109, 169–196. [Google Scholar] [CrossRef]
- Guo, Y.; Li, L.; Yang, X. Fabrication of Mung Bean Protein-Sugar Beet Pectin Hydrogels by Duo-Induction of Heating and Laccase: Gelling Properties and Delivery of Riboflavin. Food Hydrocoll. 2025, 159, 110690. [Google Scholar] [CrossRef]
- Hussain, A.; Hussain, M.; Ashraf, W.; Ali, K.; Hussain, A.; Phyo, H.M.; Khalid, M.U.; Hussain, M.; Lianfu, Z. Encapsulation, Optimization, and Characterization of Lycopene Using Soy Protein Isolate-Peach Gum Conjugate as Wall Material. Food Biosci. 2024, 62, 105548. [Google Scholar] [CrossRef]
- Liu, Y.; Fan, X.; Yu, X.; Liu, T.; Guo, X.; Zhang, J. Enhancing Curcumin Stability and Bioavailability through Chickpea Protein Isolate–Citrus Pectin Conjugate Emulsions: Targeted Delivery and Gut Microecology Modulation. Int. J. Biol. Macromol. 2025, 300, 140295. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, S.; Shi, S.; Wang, R.; Liu, H. Dual-Crosslinked Soy Protein–Soy Hull Polysaccharide Liposomal Gels for Targeted Lutein Delivery and Controlled Release in Vitro and in Vivo: Impact of Soy Hull Polysaccharide Concentration. Food Chem. 2025, 493, 145986. [Google Scholar] [CrossRef] [PubMed]
- Zhao, K.; Hao, Y.; Guo, X.; Chang, Y.; Shen, X. Development, Characterization and Underling Mechanism of 3D Printable Quinoa Protein Emulsion Gels by Incorporating of Different Polysaccharides for Curcumin Delivery. Int. J. Biol. Macromol. 2024, 280, 135648. [Google Scholar] [CrossRef]
- Feng, J.; Li, Z.; Huang, J.; Zhan, Z.; Feng, J.; Wu, Y.; Wei, X.; Wang, Z.; Wang, S.; Chen, Y.; et al. Quinoa Protein-Dextran Conjugates as Functional Stabilizers for Curcumin-Loaded Nanoemulsions. Food Res. Int. 2025, 219, 117107. [Google Scholar] [CrossRef]
- Sun, C.; Wang, S.; Wang, S.; Wang, P.; Zhang, G.; Liu, H.; Zhu, D. Characterization of High-Internal-Phase Emulsions Based on Soy Protein Isolate with Varying Concentrations of Soy Hull Polysaccharide and Their Capabilities for Probiotic Delivery: In Vivo and in Vitro Release and Thermal Stability. Food Res. Int. 2024, 186, 114371. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Wang, W.; Ji, S.; Xia, Q. Soy Protein Isolate-Xanthan Gum Complexes to Stabilize Pickering Emulsions for Quercetin Delivery. Food Chem. 2024, 461, 140794. [Google Scholar] [CrossRef] [PubMed]
- Xiao, S.; Ahn, D.U. Formulation and Characterization of Protein-Based Complexes for Nutrient Delivery: Impact of Polysaccharides on the Encapsulation of Curcumin with Ovalbumin. Food Chem. 2025, 486, 144617. [Google Scholar] [CrossRef]
- Xue, H.; Feng, J.; Tang, Y.; Wang, X.; Tang, J.; Cai, X.; Zhong, H. Research Progress on the Interaction of the Polyphenol–Protein–Polysaccharide Ternary Systems. Chem. Biol. Technol. Agric. 2024, 11, 95. [Google Scholar] [CrossRef]
- Ge, W.; Chen, J.; Wang, Y.; Hu, Z.; Wang, H. Insight on the Functionalization of Hesperidin: Encapsulated by the Complex of Soybean Protein Isolate and Polysaccharides. Food Chem. 2025, 488, 144902. [Google Scholar] [CrossRef]
- Tavares, L.; Santos, L.; Zapata Noreña, C.P. Bioactive Compounds of Garlic: A Comprehensive Review of Encapsulation Technologies, Characterization of the Encapsulated Garlic Compounds and Their Industrial Applicability. Trends Food Sci. Technol. 2021, 114, 232–244. [Google Scholar] [CrossRef]
- Ji, F.; Liu, H.; Wang, C.; Guo, N.; Shen, Y.; Luo, S.; Jiang, S.; Zheng, Z. Remodeling the Structure of Soy Protein Fibrils to Hydrogels for Co-Encapsulation of (−)-Epigallocatechin Gallate (EGCG) and Curcumin: Role of EGCG. Food Hydrocoll. 2024, 147, 109439. [Google Scholar] [CrossRef]
- Falsafi, S.R.; Rostamabadi, H.; Nishinari, K.; Amani, R.; Jafari, S.M. The Role of Emulsification Strategy on the Electrospinning of β-Carotene-Loaded Emulsions Stabilized by Gum Arabic and Whey Protein Isolate. Food Chem. 2022, 374, 131826. [Google Scholar] [CrossRef]
- Fan, C.; Feng, T.; Wang, X.; Xia, S.; John Swing, C. Liposomes for Encapsulation of Liposoluble Vitamins (A, D, E and K): Comparation of Loading Ability, Storage Stability and Bilayer Dynamics. Food Res. Int. 2023, 163, 112264. [Google Scholar] [CrossRef]
- Wang, X.; Chen, C.; Bao, Y.; Wang, Y.; Leonidovna Strakh, Y. Encapsulation of Three Different Types of Polyphenols in Casein Using a Customized pH-Driven Method: Preparation and Characterization. Food Res. Int. 2024, 189, 114547. [Google Scholar] [CrossRef]
- Shakoury, N.; Aliyari, M.A.; Salami, M.; Emam-Djomeh, Z.; Vardhanabhuti, B.; Moosavi-Movahedi, A.A. Encapsulation of Propolis Extract in Whey Protein Nanoparticles. LWT 2022, 158, 113138. [Google Scholar] [CrossRef]
- Geng, M.; Feng, X.; Wu, X.; Tan, X.; Liu, Z.; Li, L.; Huang, Y.; Teng, F.; Li, Y. Encapsulating Vitamins C and E Using Food-Grade Soy Protein Isolate and Pectin Particles as Carrier: Insights on the Vitamin Additive Antioxidant Effects. Food Chem. 2023, 418, 135955. [Google Scholar] [CrossRef]
- Zhu, Q.; Qiu, Y.; Zhang, L.; Lu, W.; Pan, Y.; Liu, X.; Li, Z.; Yang, H. Encapsulation of Lycopene in Pickering Emulsion Stabilized by Complexes of Whey Protein Isolate Fibrils and Sodium Alginate: Physicochemical Property, Structural Characterization and in Vitro Digestion Property. Food Res. Int. 2024, 191, 114675. [Google Scholar] [CrossRef]
- Bao, X.; Rong, S.; Fu, Q.; Liu, H.; Han, Y.; Liu, F.; Ye, Z.; Chen, S. Zein-Yeast Carboxymethyl Glucan Particles Formed by Anti-Solvent Precipitation for Encapsulating Resveratrol. Int. J. Biol. Macromol. 2023, 253, 127557. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Wang, Y.; Zhao, D. Chapter 3—Structural Analysis of Biomacromolecules Using Circular Dichroism Spectroscopy. In Advanced Spectroscopic Methods to Study Biomolecular Structure and Dynamics; Saudagar, P., Tripathi, T., Eds.; Academic Press: Cambridge, MA, USA, 2023; pp. 77–103. ISBN 978-0-323-99127-8. [Google Scholar]
- Yan, Y.; Tang, R.; Li, F.; Huang, L.; Chen, Y.; He, R. Spectral Analysis of the Impact of Various Polysaccharides on the Entrapment of Curcumin by Whey Protein Isolate. Food Chem. 2025, 468, 142441. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Li, Y.; Jin, Z.; Xue, C.; Wei, Z. Effect of Ultrasonication and Homogenization on Electrostatically Self-Assembled Whey Protein Isolate/Pectin Particles: EGCG Encapsulation, Stability, and Controlled Release Properties. Food Hydrocoll. 2025, 166, 111264. [Google Scholar] [CrossRef]
- Koshani, R.; Jafari, S.M.; van de Ven, T.G.M. Going Deep inside Bioactive-Loaded Nanocarriers through Nuclear Magnetic Resonance (NMR) Spectroscopy. Trends Food Sci. Technol. 2020, 101, 198–212. [Google Scholar] [CrossRef]
- El Nokab, M.E.H.; Alassmy, Y.A.; Abduljawad, M.M.; Al-shamrani, K.M.; Alnafisah, M.S.; Asgar Pour, Z.; Tucker, C.L.; Sebakhy, K.O. Solid-State NMR Spectroscopy: Towards Structural Insights into Starch-Based Materials in the Food Industry. Polymers 2022, 14, 4686. [Google Scholar] [CrossRef]
- Du, Z.; Lv, H.; Wang, C.; He, D.; Xu, E.; Jin, Z.; Yuan, C.; Guo, L.; Wu, Z.; Liu, P.; et al. Organic Solvent-Free Starch-Based Green Electrospun Nanofiber Mats for Curcumin Encapsulation and Delivery. Int. J. Biol. Macromol. 2023, 232, 123497. [Google Scholar] [CrossRef]
- Wang, C.; Wang, H.; Abd El-Aty, A.M.; Su, W.; Tan, M. Preparation of Bilayer Nano-Oleogel by Whey Protein Isolate and Soy Lecithin for Fish Oil Encapsulation and Its Application in Cookies. Food Hydrocoll. 2024, 146, 109280. [Google Scholar] [CrossRef]
- Rizvi, A.; Mulvey, J.T.; Carpenter, B.P.; Talosig, R.; Patterson, J.P. A Close Look at Molecular Self-Assembly with the Transmission Electron Microscope. Chem. Rev. 2021, 121, 14232–14280. [Google Scholar] [CrossRef]
- Chen, J.; Huang, S.; Guo, J.; Wang, J.; Yang, X. Fabrication of Stable Hydrogel Microspheres with Hydrophobic Shell Using Water-in-Water (W/W) Pickering Emulsion Template. Food Res. Int. 2024, 197, 115254. [Google Scholar] [CrossRef]
- Ruan, C.; Nian, Y.; Chen, Q.; Li, N.; He, X.; Li, C.; Hu, B. Higher Affinity of Polyphenol to Zein than to Amyloid Fibrils Leading to Nanoparticle-Embed Network Wall Scaffold to Construct Amyloid Fibril-Zein-EGCG Hydrogels for Coating of Beef. Food Res. Int. 2022, 156, 111187. [Google Scholar] [CrossRef] [PubMed]
- Ho, T.M.; Abik, F.; Mikkonen, K.S. An Overview of Nanoemulsion Characterization via Atomic Force Microscopy. Crit. Rev. Food Sci. Nutr. 2022, 62, 4908–4928. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liang, Q.; Liu, X.; Raza, H.; Ma, H.; Ren, X. Treatment with Ultrasound Improves the Encapsulation Efficiency of Resveratrol in Zein-Gum Arabic Complex Coacervates. LWT 2022, 153, 112331. [Google Scholar] [CrossRef]
- Tosi, M.M.; Ramos, A.P.; Esposto, B.S.; Jafari, S.M. Chapter Six—Dynamic Light Scattering (DLS) of Nanoencapsulated Food Ingredients. In Characterization of Nanoencapsulated Food Ingredients; Jafari, S.M., Ed.; Nanoencapsulation in the Food Industry; Academic Press: Cambridge, MA, USA, 2020; Volume 4, pp. 191–211. ISBN 978-0-12-815667-4. [Google Scholar]
- Malekjani, N.; Jafari, S.M. Modeling the Release of Food Bioactive Ingredients from Carriers/Nanocarriers by the Empirical, Semiempirical, and Mechanistic Models. Compr. Rev. Food Sci. Food Saf. 2021, 20, 3–47. [Google Scholar] [CrossRef] [PubMed]
- Tao, X.; Huang, Y.; Wang, C.; Chen, F.; Yang, L.; Ling, L.; Che, Z.; Chen, X. Recent Developments in Molecular Docking Technology Applied in Food Science: A Review. Int. J. Food Sci. Technol. 2020, 55, 33–45. [Google Scholar] [CrossRef]
- Wu, K.; Zhang, T.; Chai, X.; Duan, X.; He, D.; Yu, H.; Liu, X.; Tao, Z. Encapsulation Efficiency and Functional Stability of Cinnamon Essential Oil in Modified β-Cyclodextrins: In Vitro and In Silico Evidence. Foods 2023, 12, 45. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, C.; Qi, Z.; Zhao, R.; Wang, C.; Zhang, T. Pea Protein Based Nanocarriers for Lipophilic Polyphenols: Spectroscopic Analysis, Characterization, Chemical Stability, Antioxidant and Molecular Docking. Food Res. Int. 2022, 160, 111713. [Google Scholar] [CrossRef]
- Liu, Y.; Tan, X.; Li, L.; Xie, T.; Teng, F. Co-Encapsulation of Vitamin E and Quercetin by Soybean Lipophilic Proteins Based on pH-Shifting and Ultrasonication: Focus on Interaction Mechanisms, Structural and Physicochemical Properties. Food Chem. 2024, 460, 140608. [Google Scholar] [CrossRef]
- Xu, P.-W.; Yue, X.-J.; Yuan, X.-F.; Zhao, B. Non-Covalent Interaction between Hemp Seed Globulin and Two Hemp Seed Phenolic Compounds: Mechanism and Effects on Protein Structure, Bioactivity, and in Vitro Simulated Digestion. Int. J. Biol. Macromol. 2024, 255, 128077. [Google Scholar] [CrossRef] [PubMed]
- Chamizo-González, F.; Gordillo, B.; Heredia, F.J. Elucidation of the 3D Structure of Grape Seed 7S Globulin and Its Interaction with Malvidin 3-Glucoside: A Molecular Modeling Approach. Food Chem. 2021, 347, 129014. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Zhu, L.; Wu, G.; Zhang, H. The Interaction Mechanisms, Biological Activities and Digestive Properties between Tartary Buckwheat Protein and Phenolic Extract under pH-Driven Methods. Food Chem. 2023, 419, 135758. [Google Scholar] [CrossRef]
- Ali Redha, A.; Kodikara, C.; Cozzolino, D. Does Encapsulation Improve the Bioavailability of Polyphenols in Humans? A Concise Review Based on In Vivo Human Studies. Nutrients 2024, 16, 3625. [Google Scholar] [CrossRef]
- Rashwan, A.K.; Karim, N.; Xu, Y.; Xie, J.; Cui, H.; Mozafari, M.R.; Chen, W. Potential Micro-/Nano-Encapsulation Systems for Improving Stability and Bioavailability of Anthocyanins: An Updated Review. Crit. Rev. Food Sci. Nutr. 2023, 63, 3362–3385. [Google Scholar] [CrossRef]
- Garzón, A.G.; Cian, R.E.; Drago, S.R. Effects of Agar-Carrageenan Wall Materials and Core-to-Wall Material Ratio on Physicochemical Properties and in Vitro Bioaccessibility of Microencapsulated Bioactive Peptides. Food Hydrocoll. 2023, 139, 108570. [Google Scholar] [CrossRef]
- Lv, H.; Wang, C.; Xu, E.; Jin, Z.; Zhao, H.; Yuan, C.; Zhao, M.; Yu, B.; Wu, Z.; He, D.; et al. Preparation of Starch-Based Oral Fast-Disintegrating Nanofiber Mats for Astaxanthin Encapsulation and Delivery via Emulsion Electrospinning. Int. J. Biol. Macromol. 2025, 289, 136466. [Google Scholar] [CrossRef]
- Huang, R.; Song, H.; Wang, X.; Shen, H.; Li, S.; Guan, X. Fatty Acids-Modified Liposomes for Encapsulation of Bioactive Peptides: Fabrication, Characterization, Storage Stability and in Vitro Release. Food Chem. 2024, 440, 138139. [Google Scholar] [CrossRef]
- da Silva, L.C.; Castelo, R.M.; Magalhães, H.C.R.; Furtado, R.F.; Cheng, H.N.; Biswas, A.; Alves, C.R. Characterization and Controlled Release of Pequi Oil Microcapsules for Yogurt Application. LWT 2022, 157, 113105. [Google Scholar] [CrossRef]
- Su, X.; Xu, Y.; Xu, Z.; Hurley, K.; Feng, Y.; Yin, Y. Encapsulation of Hop (Humulus lupulus L.) Essential Oil for Controlled Release in the Non-Alcoholic Beverage Application. Food Hydrocoll. 2023, 134, 108039. [Google Scholar] [CrossRef]
- Lin, R.; Wang, Y.; Cheng, H.; Ye, X.; Chen, S.; Pan, H. Epigallocatechin-3-Gallate Stabilizes Aqueous Curcumin by Generating Nanoparticles and Its Application in Beverages. Food Chem. 2024, 444, 138655. [Google Scholar] [CrossRef]
- Shaddel, R.; Rajabi-Moghaddam, S. Encapsulation of Caffeine in Chitosan-Coated Nanoliposomes and Its Application in Drink Formulation. Food Hydrocoll. 2024, 149, 109598. [Google Scholar] [CrossRef]
- Jia, L.; Wang, X.; He, T.; Wang, S.; Zhang, X.; Hui, A.; Xie, F. Interaction Mechanism of Soy Protein Isolate with Aldehyde Flavor Compounds: Differences in Carbon Chain Length and Unsaturation. J. Mol. Liq. 2024, 412, 125876. [Google Scholar] [CrossRef]
- Ince, C.; Condict, L.; Stockmann, R.; Ashton, J.; Kasapis, S. Molecular Characterisation of Interactions between 11S Glycinin and Hexanal—An off-flavour Compound. Food Hydrocoll. 2025, 158, 110543. [Google Scholar] [CrossRef]
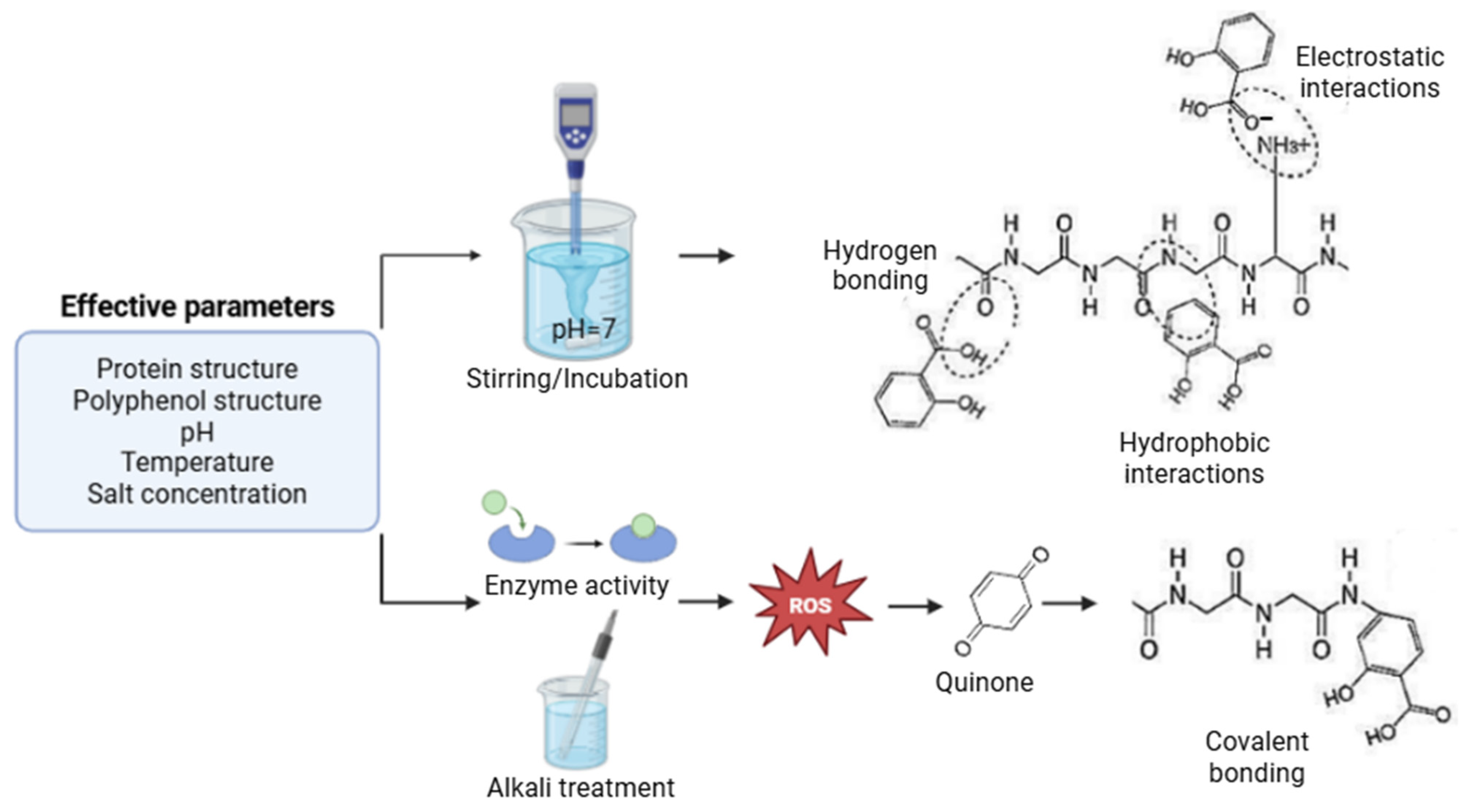
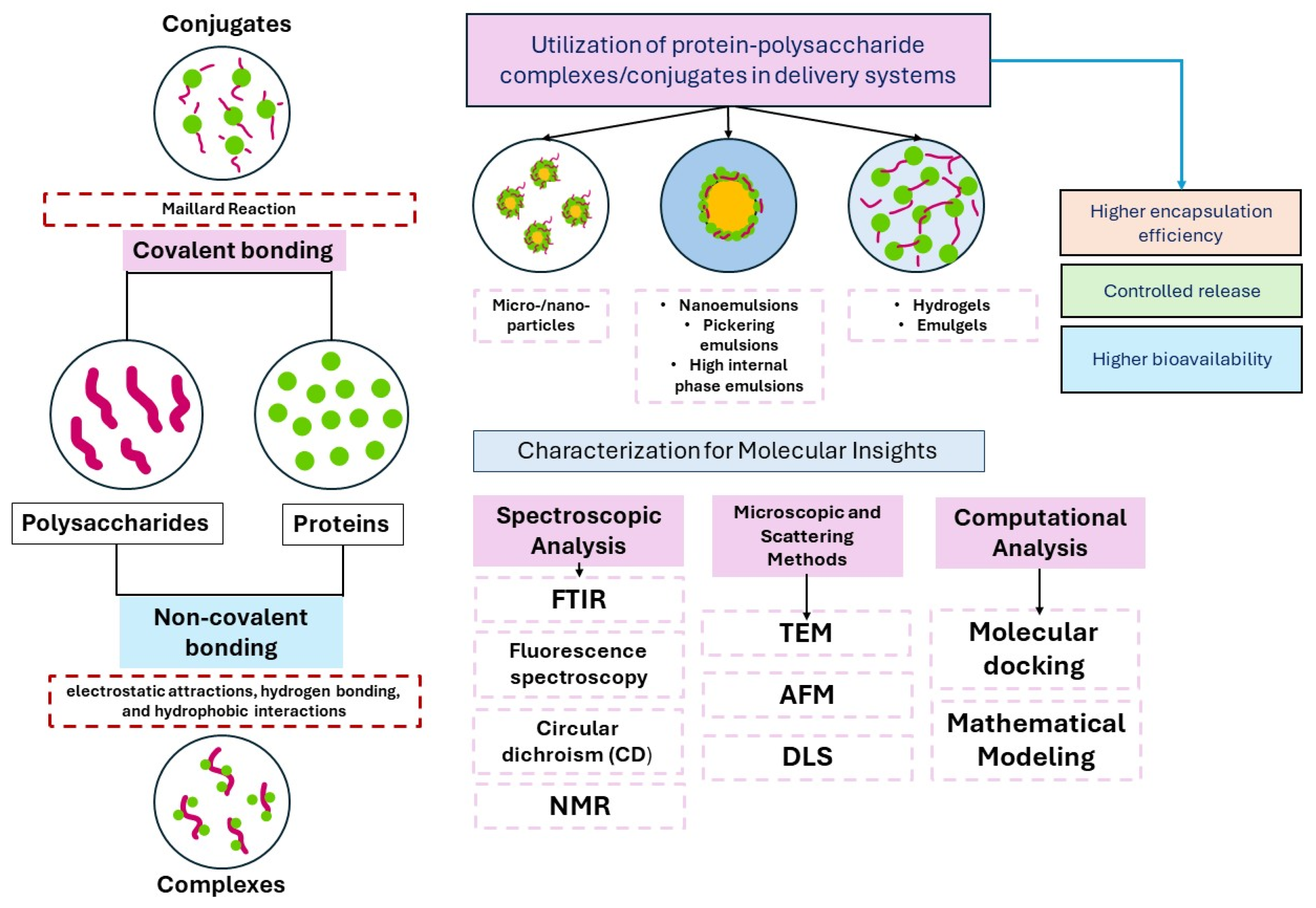
| Protein-Polyphenol Interactions | Interaction Type and Method | Characterization Techniques for Interactions | Structural Modification of Protein | Alterations in Functional Properties | Changes in Bioaccessibility and Antioxidant Activity of Polyphenols | References |
|---|---|---|---|---|---|---|
| Milk proteins-coffee polyphenols | Non-covalent interactions through mixing at pH 7 |
|
|
| n.i. | [23] |
| Chickpea protein isolate-epigallocatechin gallate | Covalent interactions through mixing at pH 9, 26 °C for 24 h, dialyzing, and lyophilization |
|
|
| n.i. | [58] |
| Milk proteins-blackcurrant polyphenols | Non-covalent interactions through mixing at a pH of 6.5, 28 °C for one h and lyophilization |
|
| n.i. |
| [49] |
| Chickpea protein isolate-naringenin | Non-covalent interactions through mixing at pH 7 |
|
|
| n.i. | [62] |
| Soy protein-hydroxytyrosol (HT) | Mixing SPI and HT solutions and freeze-drying |
|
|
| n.i. | [63] |
| Rice protein-legume polyphenols | Covalent interactions through mixing at 40 °C, and the pH of 8.0 |
|
|
| n.i. | [64] |
| Peanut protein extract-epigallocatechin gallate, chlorogenic acid | Alkali treatment method at pH 9 and lyophilization |
|
|
| n.i. | [65] |
| Goose liver protein- catechin, quercetin, and rutin | Non-covalent interactions through pH-shifting (from 11.5 to 7.0) |
|
|
| n.i. | [44] |
| Soy protein isolate-chlorogenic acid | Covalent interactions through mixing at pH 9, 25 °C for 7 h |
|
|
| n.i. | [66] |
| Soy protein-chlorogenic acid | Mixing of SPI and CA solutions at pH = 7 |
|
|
| n.i. | [67] |
| Soybean protein isolate- apple polyphenols | Non-covalent interactions through mixing at a pH of 7 |
|
|
| n.i. | [52] |
| Peanut protein-epigallocatechin gallate | Covalent interactions through mixing at 25 °C and freeze-drying |
|
|
| n.i. | [68] |
| Pea protein isolate-chlorogenic acid, epigallocatechin gallate, resveratrol | Non-covalent interactions through mixing overnight at 4 °C, pH of 7 |
|
|
| n.i. | [69] |
| Egg white protein-tea polyphenols | Non-covalent interactions through mixing at pH 7 °C, for two h, and lyophilization |
|
|
|
| [11] |
| Casein, whey proteins, sea buckthorn polyphenols | Non-covalent interactions through incubation at pH 2.5 °C, for one h under pH 7 |
|
| n.i. |
| [36] |
| Soy protein isolate- epigallocatechin gallate | Covalent interactions through ultrasonication (at 300 W for 10 min, 2 s on/off) |
|
| n.i. |
| [70] |
| Soy protein isolate-tannic acid | Mixing SPI and TA solutions under alkali pH (9, 10, 11) and aerobic conditions |
|
|
| n.i. | [71] |
| Bovine lactoferrin-procyanidin | Non-covalent interactions through mixing at pH 7 |
|
|
| n.i. | [72] |
| Whey protein- epigallocatechin gallate, quercetin, apigenin, naringenin | Covalent interactions through free radical grafting at pH 7.2 |
|
| n.i. |
| [6] |
| Whey protein-cholorogenic acid, epigallocatechin gallate | Non-covalent interactions through mixing in the dark at 25 °C for 60 min at pH 3.5 and 7 |
|
| n.i. |
| [73] |
| Carotene | Encapsulation Method | Carrier | Encapsulation/Loading Capacity | Physicochemical Properties | Release Kinetics/Bioaccessibility | Reference |
|---|---|---|---|---|---|---|
| β-carotene | Aggregation: High-speed homogenization and stirring | Octenylsuccinated Gastrodia elata starch | ↑ Aqueous solubility of β-carotene from 1.5 × 10−6 to 58.96 | NMR: β-carotene located in inner domains, proton peaks disappear UV-Vis (λmax): red shift, H/J-type aggregate structure FTIR: all β-carotene peaks disappeared → fully encapsulated, stabilized via hydrogen bonding XRD: Amorphous → stable aggregates ↓ Contact angle&Surface tension: Tight hydrophobic domain |
| [88] |
| β-carotene | Spray-drying: high-speed shear and homogenization | Octenyl succinic anhydride modified starch with tea saponins | ↑ EE and LC ↓ Particle size and ζ-potential via formation of OSA-starch-tea saponin complexes | ↓ Surface tension via tea saponin level FTIR: most β-carotene peaks disappeared → fully encapsulated, stabilized via hydrogen bonding XRD: Amorphous structure (low relative crystallinity 2.61%) with high tea saponin level |
| [92] |
| Canthaxanthin | Freeze-drying, after ultrasonication (2–10 min, 200 W, 20 kHz) | V amylose (DP 311) + maltodextrin | Optimum: 20 mg canthaxanthin + 400 mg V-amylose → Canthaxanthin recovery ≈ 90% | TEM: Resemble spherical or oval shape after ultrasonication XRD: Fully amorphous → canthaxanthin loses crystallinity DSC: Canthaxanthin peaks disappeared → fully encapsulated ↑ Antioxidant activity: DPPH and ABTS | n.i. | [89] |
| β-carotene | Emulsification with ultrasonication | Mandarin peel pectin | EE: 78.7–87.4% ζ-potential: −25.7 to −27.1 mV after β-carotene loading | ↑ Viscosity → higher emulsion stability and reduced β-carotene degradation |
| [33] |
| β-Carotene | Pickering emulsion | Pectin | ↑ Pectin concentration (0.5–3%): ↓ Creaming index (more stable), ↓ Droplet size |
|
| [95] |
| Carotenoids from orange peel | Emulsion and gelation | Sodium alginate + CaCl2 | Encapsulation yield: 92.3% EE: 89.5% |
| n.i. | [96] |
| Sea buckthorn pomace carotene extract: | Emulsification and ionic gelation | Sodium alginate | EE: 98.4% |
|
| [97] |
| Lycopene and β-carotene | Ionotropic gelation | Sodium alginate + CaCl2 | Encapsulation yield: 86.3% EE: 82.6% |
|
| [98] |
| Lutein | Hydrogelation | Sodium alginate + Ca2+-EGTA and D-gluconolactone | LC: 770.88 μg/g EE: 90% for nanoparticles, 99.39% for hydrogel ↑ Ca2+ concentration: ↑ Crosslinking density → better nanoparticle diffusion and higher EE |
|
| [99] |
| Carotenoid | Encapsulation Method | Carrier | Encapsulation Outcomes | Physical and Morphological Characterization | Release Kinetics/Bioaccessibility of Carotenoid | Reference |
|---|---|---|---|---|---|---|
| Lycopene-rich extract from red guava | Nanoprecipitation/high-shear homogenization | Cetylated cashew gum ± fucan | Particle Size: 189 → 251 nm EE: 8–13% for Cetylated cashew gum to ~60% with fucan |
| n.i. | [101] |
| β-Carotene | Spray-drying | Maltodextrin + acacia gum or mesquite gum | Encapsulation yield: Acacia gum → up to 85%; mesquite gum → 69% |
| n.i. | [102] |
| Lycopene | Emulsion: high-pressure homogenization and spray drying | Basil seed gum | Optimized Encapsulation Conditions: ≈19.5% gum level → EE: 86.78%, Encapsulation yield: 54.94% PDI < 0.30 (uniform dispersion); ζ-potential: −21.37 mV |
| n.i. | [87] |
| β-Carotene | Emulsification: | Xanthan gum, guar gum | EE: 87.2% |
| n.i. | [103] |
| β-carotene | Emulsification: high-speed shear and ultrasonic emulsification | High acyl gellan gum | Emulsification yield: gradual increase via gum concentration (0.05–0.2%), highest 84.4% at 0.175% Mean particle size: 4–7.2 µm ζ-potential: between −59.0 and −52.2 mV |
|
| [104] |
| β-carotene | Pickering emulsion: high-shear homogenization + high-pressure homogenization | Peach gum polysaccharide: MC, YQG, ZP11, TW-20 | EE: 71.92–89.08% |
|
| [90] |
| Natural palm mixed-carotene complex | Pickering emulsion | Nanofibrillated cellulose | EE: 87–89.80% ↑ Particle size: 73.67 → 94.73 via cellulose concentration (0.2–1%) ζ-potential: <−30 mV | n.i. | n.i. | [95] |
| Lycopene | Pickering emulsion | Cellulose nanofiber from papaya peel | n.i. | n.i. |
| [86] |
| β-carotene | Pickering emulsion | Cellulose nanofibers | EE: decreased from 94.24% at 25 °C to 57.23% at 75 °C | n.i. |
| [91] |
| Astaxanthin | Cold application gel | Hydroxypropyl methylcellulose, types: K1500, K250, K4M, E5 | Optimal HPMC-K4M dosage: 0.2 g or 107.50% |
|
| [105] |
| Carotenoid | Encapsulation Method | Carrier | Encapsulation Outcomes | Physical and Morphological Characterization | Release Kinetics/Bioaccessibility of Carotenoid | Reference |
|---|---|---|---|---|---|---|
| Lutein | Emulsion: Gelation and centrifugal washing | Curdlan ± Sodium Alginate |
|
|
| [114] |
| β-Carotene: | Liposome | Egg yolk phosphatidylcholine + cholesterol + chitosan ± pectin |
|
|
| [111] |
| Astaxanthin | Ionotropic gelation | Pectin + sodium alginate + chitosan in oleoresin |
|
|
| [113] |
| Lutein | Ionic gelation | Chitosan (1 mg/mL) and sodium alginate (0.5 mg/mL) |
|
|
| [112] |
| Lutein | Complex coacervation | Alginate + low-molecular-weight chitosan |
|
| n.i. | [109] |
| Astaxanthin | Pickering emulsion | Chitosan + Guar gum |
|
|
| [110] |
| β-Carotene: | Host–guest inclusion + coating | 2-Hydroxypropyl-β-cyclodextrin + High- or low-metoxyl pectin or pectic acid |
|
| n.i. | [115] |
| β-carotene: (20% w/w) | Gelation | Yam starch + guar gum, xanthan gum, carrageenan gum, or their combinations | n.i. |
| n.i. | [117] |
| Lycopene: 0.5, 1.0, and 1.5 | Emulsion-based polyelectrolyte complex | Sodium alginate + CaCl2 + chitosan |
|
| n.i. | [118] |
| Astaxanthin: 0.5 g | Tertiary emulsion: layer-by-layer self-assembly | Soy lecithin + chitosan + sodium alginate | LC: ≈ 0.56% EE > 90% ζ-potential: primary: −11.6 → −8.25 mV; secondary: 29.5 → 24.6 mV; tertiary: −22.7 → −18.4 mV | n.i. |
| [116] |
| Wall Material | Core Material | Encapsulation Method | Key Findings | Reference |
|---|---|---|---|---|
| Chickpea protein isolate (CPI) Citrus pectin (CP) | Curcumin | Emulsion formation by high-pressure homogenization with CPI–CP conjugates |
| [125] |
| Quinoa protein (QP) Inulin (INU), sodium alginate (SA), fucoidan (FU), and dextran (DX) | Curcumin | Emulsion gel formation by glucono delta-lactone induced gelation using QP-polysaccharide mixtures |
| [127] |
| QP)-DX conjugates | Curcumin | Nanoemulsion formation by high-pressure microfluidization using QP–DX conjugates as stabilizers |
| [128] |
| Mung bean protein (MBP) Sugar beet pectin (SBP) | Riboflavin | MBP–SBP hydrogel formation via laccase-induced crosslinking |
| [123] |
| SPI Soy hull polysaccharide | Lactobacillus plantarum | HIPE formation by homogenization of SPI–SHP mixtures with soybean oil |
| [129] |
| SPI Xanthan gum (XG) | Quercetin | Pickering emulsion formation by homogenization of SPI–XG mixtures with corn oil |
| [130] |
| SPI-peach gum conjugate | Lycopene | Microparticle formation by spray drying using SPI–PG Maillard conjugates |
| [124] |
| PPI-DX conjugates | Astaxanthin | Emulsion formation by high-pressure homogenization using PPI–DX complexes and conjugates |
| [105] |
| SPI Soy hull polysaccharide (SHP) | Lutein | Liposome formation by thin-film evaporation–sonication and incorporation into SPI–SHP hydrogel matrix |
| [126] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Cavdar Dincturk, H.; Akkuzu, N.; Günal-Köroğlu, D.; Can Karaca, A.; Capanoglu, E. Molecular Insights into Bioactive Interactions Within Protein- and Polysaccharide-Based Colloids: Implications for Stability, Functionality, and Bioavailability. Foods 2026, 15, 112. https://doi.org/10.3390/foods15010112
Cavdar Dincturk H, Akkuzu N, Günal-Köroğlu D, Can Karaca A, Capanoglu E. Molecular Insights into Bioactive Interactions Within Protein- and Polysaccharide-Based Colloids: Implications for Stability, Functionality, and Bioavailability. Foods. 2026; 15(1):112. https://doi.org/10.3390/foods15010112
Chicago/Turabian StyleCavdar Dincturk, Humeyra, Nisa Akkuzu, Deniz Günal-Köroğlu, Asli Can Karaca, and Esra Capanoglu. 2026. "Molecular Insights into Bioactive Interactions Within Protein- and Polysaccharide-Based Colloids: Implications for Stability, Functionality, and Bioavailability" Foods 15, no. 1: 112. https://doi.org/10.3390/foods15010112
APA StyleCavdar Dincturk, H., Akkuzu, N., Günal-Köroğlu, D., Can Karaca, A., & Capanoglu, E. (2026). Molecular Insights into Bioactive Interactions Within Protein- and Polysaccharide-Based Colloids: Implications for Stability, Functionality, and Bioavailability. Foods, 15(1), 112. https://doi.org/10.3390/foods15010112







