Exploring Intervention Strategies for Microbial Biofilms in the Food Industry Based on a Biomolecular Mechanism Perspective: Recent Advances and Emerging Trends
Abstract
1. Introduction
2. Initial Adhesion-Targeting Strategies in Food Chain
2.1. Physicochemical Forces Governing Initial Adhesion
2.2. Strategies to Prevent Initial Adhesion
2.2.1. Surface Engineering and Material Modification
2.2.2. Natural Anti-Adhesion Compounds
2.2.3. Competitive Exclusion by Probiotic or Benign Microbial Films
3. Targeting Biofilm Formation and Maturation in Food Industry Settings
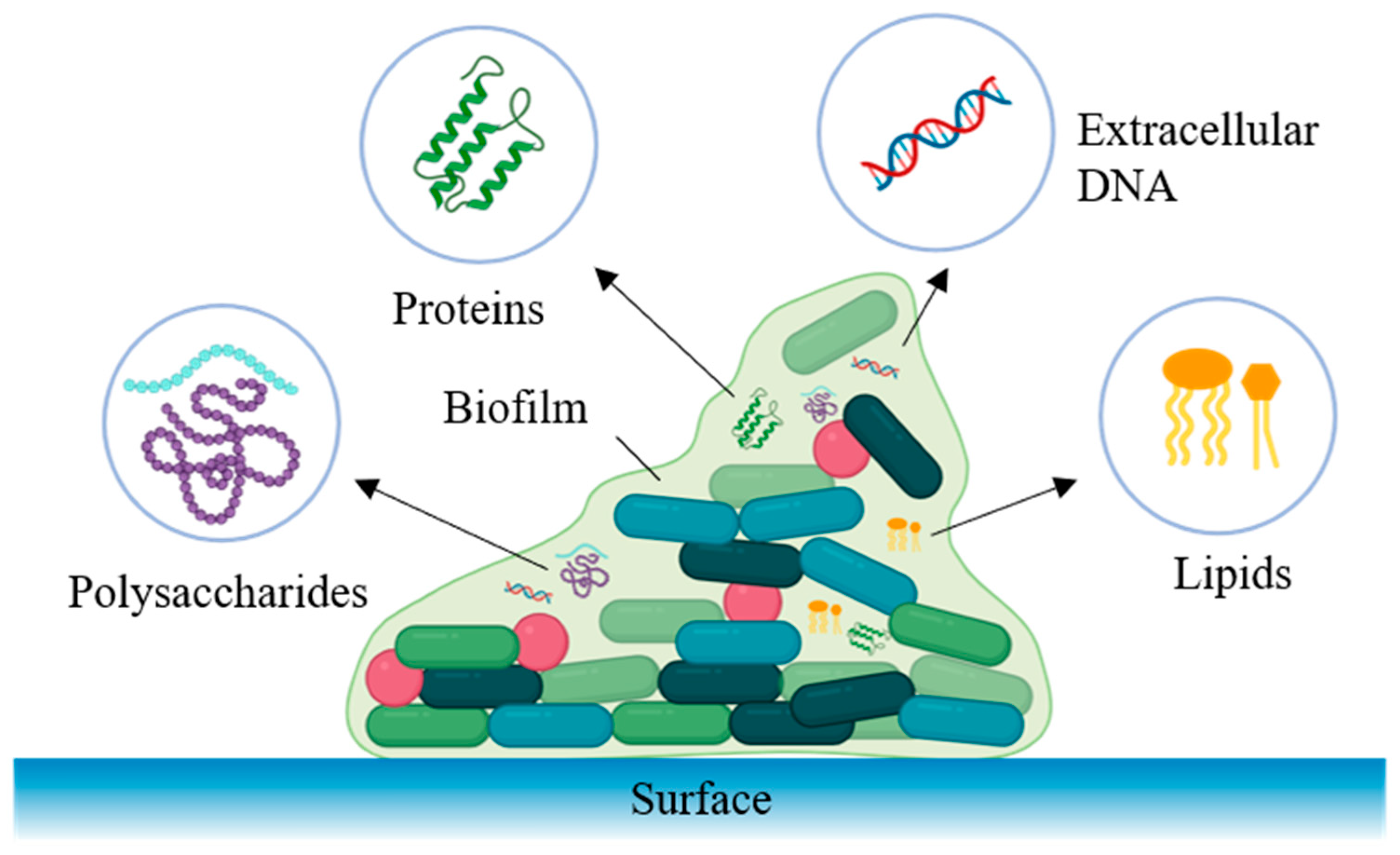
3.1. Composition and Function of the Biofilm EPS Matrix
3.2. Intervention Strategies Targeting EPS
3.3. Microenvironmental Effects on Biofilm Development and Stability
3.4. Intervention via Microenvironment Manipulation
3.5. Population Behavior and Phenotypic Changes During Biofilm Maturation
3.6. Intervention Targeting Persisters and QS
4. Biofilm Dispersion and Control Strategies
4.1. Mechanisms Triggering Biofilm Dispersion
4.2. Risks and Microbial Spread Associated with Dispersion
4.3. Intervention and Control Strategies for Biofilm Dispersion
5. Future Directions and Emerging Technologies
5.1. Data Integration and Predictive Modeling
| Intervention Strategies | Molecular Targets/Mechanism | Advantages | Limitations | Industry Applicability | References |
|---|---|---|---|---|---|
| Smart Antimicrobial Coatings (e.g., pH-responsive phage release) | Bacterial metabolism triggers antimicrobial release | On-demand action, extended protection | Material stability and food compatibility need validation | High-risk niches (drains, filler heads) | [85,86,87] |
| Real-time Biosensors (e.g., electrochemical, optical) | Detects early attachment and EPS changes | Early warning, real-time monitoring | High cost, requires calibration and maintenance | Smart food factories | [88,89,90] |
| CRISPR-based Antimicrobials | Precise cleavage of pathogen genes | High specificity, low resistance development | Not yet regulated, still in experimental stages | Future precision biocontrol | [91] |
5.2. Smart Antimicrobial Surfaces and Responsive Coatings
5.3. Biosensors and Monitoring
5.4. CRISPR and Ecological Microbial Control
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| EPS | Extracellular polymeric substance |
| QS | Quorum sensing |
| eDNA | Extracellular DNA |
| c-di-GMP | cyclic-di-GMP |
| QSIs | Quorum sensing inhibitors |
| T4P | Type IV pili |
| PIA/PNAG | Poly-β-1,6-N-acetylglucosamine |
| SCVs | Small colony variants |
| PDEs | Phosphodiesterases |
| HGT | Horizontal gene transfer |
| CIP | Cleaning-in-place |
| IoT | Internet of Things |
References
- Pratt, L.A.; Kolter, R. Genetic analysis of Escherichia coli biofilm formation: Roles of flagella, motility, chemotaxis and type I pili. Mol. Microbiol. 1998, 30, 285–293. [Google Scholar] [CrossRef]
- Costerton, J.W.; Stewart, P.S.; Greenberg, E.P. Bacterial biofilms: A common cause of persistent infections. Science 1999, 284, 1318–1322. [Google Scholar] [CrossRef] [PubMed]
- David, A.; Tahrioui, A.; Tareau, A.-S.; Forge, A.; Gonzalez, M.; Bouffartigues, E.; Lesouhaitier, O.; Chevalier, S. Pseudomonas aeruginosa Biofilm Lifecycle: Involvement of Mechanical Constraints and Timeline of Matrix Production. Antibiotics 2024, 13, 688. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, D.; Waters, C.M. A Tangled Web: Regulatory Connections between Quorum Sensing and Cyclic Di-GMP. J. Bacteriol. 2012, 194, 4485–4493. [Google Scholar] [CrossRef]
- Ramírez-Mata, A.; Fernández-Domínguez, I.J.; Nuñez-Reza, K.J.; Xiqui-Vázquez, M.L.; Baca, B.E. Redes de señalización en la producción de biopelículas en bacterias: Quorum sensing, di-GMPc y óxido nítrico. Rev. Argent. Microbiol. 2014, 46, 242–255. [Google Scholar] [CrossRef]
- Carrascosa, C.; Raheem, D.; Ramos, F.; Saraiva, A.; Raposo, A. Microbial Biofilms in the Food Industry—A Comprehensive Review. Int. J. Environ. Res. Public Health 2021, 18, 2014. [Google Scholar] [CrossRef]
- Guzmán-Soto, I.; McTiernan, C.; Gonzalez-Gomez, M.; Ross, A.; Gupta, K.; Suuronen, E.J.; Mah, T.-F.; Griffith, M.; Alarcon, E.I. Mimicking biofilm formation and development: Recent progress in in vitro and in vivo biofilm models. iScience 2021, 24, 102443. [Google Scholar] [CrossRef]
- Shrestha, L.; Fan, H.-M.; Tao, H.-R.; Huang, J.-D. Recent Strategies to Combat Biofilms Using Antimicrobial Agents and Therapeutic Approaches. Pathogens 2022, 11, 292. [Google Scholar] [CrossRef]
- Mgomi, F.C.; Yang, Y.-r.; Cheng, G.; Yang, Z.-q. Lactic acid bacteria biofilms and their antimicrobial potential against pathogenic microorganisms. Biofilm 2023, 5, 100118. [Google Scholar] [CrossRef]
- Jiang, X.; Whitehead, K.A.; Arneborg, N.; Fang, Y.; Risbo, J. Understanding bacterial surface and adhesion properties and the implications for Pickering stabilization of colloidal structures. Curr. Opin. Colloid Interface Sci. 2024, 69, 101767. [Google Scholar] [CrossRef]
- Kimkes, T.E.P.; Heinemann, M. How bacteria recognise and respond to surface contact. FEMS Microbiol. Rev. 2019, 44, 106–122. [Google Scholar] [CrossRef]
- Zheng, S.; Bawazir, M.; Dhall, A.; Kim, H.-E.; He, L.; Heo, J.; Hwang, G. Implication of Surface Properties, Bacterial Motility, and Hydrodynamic Conditions on Bacterial Surface Sensing and Their Initial Adhesion. Front. Bioeng. Biotechnol. 2021, 9, 643722. [Google Scholar] [CrossRef]
- Ruan, B.; Wu, P.; Liu, J.; Jiang, L.; Wang, H.; Qiao, J.; Zhu, N.; Dang, Z.; Luo, H.; Yi, X. Adhesion of Sphingomonas sp. GY2B onto montmorillonite: A combination study by thermodynamics and the extended DLVO theory. Colloids Surf. B Biointerfaces 2020, 192, 111085. [Google Scholar] [CrossRef]
- Zhang, M.; He, L.; Qin, J.; Wang, S.; Tong, M. Influence of flagella and their property on the initial attachment behaviors of bacteria onto plastics. Water Res. 2023, 231, 119656. [Google Scholar] [CrossRef] [PubMed]
- Krsmanovic, M.; Biswas, D.; Ali, H.; Kumar, A.; Ghosh, R.; Dickerson, A.K. Hydrodynamics and surface properties influence biofilm proliferation. Adv. Colloid Interface Sci. 2021, 288, 102336. [Google Scholar] [CrossRef] [PubMed]
- Hillig, N.; Schumann-Muck, F.; Hamedy, A.; Braun, P.G.; Koethe, M. Impact of nanoscale silicon dioxide coating of stainless-steel surfaces on Listeria monocytogenes. Folia Microbiol. 2024, 69, 173–180. [Google Scholar] [CrossRef]
- Mishra, R.; Panda, A.K.; De Mandal, S.; Shakeel, M.; Bisht, S.S.; Khan, J. Natural Anti-biofilm Agents: Strategies to Control Biofilm-Forming Pathogens. Front. Microbiol. 2020, 11, 566325. [Google Scholar] [CrossRef] [PubMed]
- Ammar, H.A.; Samy, R.; Reda, F.M.; Hassanein, W.A. Essential oils and Lactobacillus metabolites as alternative antibiofilm agents against foodborne bacteria and molecular analysis of biofilm regulatory genes. Sci. Rep. 2025, 15, 7576. [Google Scholar] [CrossRef]
- El-Tarabily, K.A.; El-Saadony, M.T.; Alagawany, M.; Arif, M.; Batiha, G.E.; Khafaga, A.F.; Elwan, H.A.M.; Elnesr, S.S.; E Abd El-Hack, M. Using essential oils to overcome bacterial biofilm formation and their antimicrobial resistance. Saudi J. Biol. Sci. 2021, 28, 5145–5156. [Google Scholar] [CrossRef]
- Tomé, A.R.; Carvalho, F.M.; Teixeira-Santos, R.; Burmølle, M.; Mergulhão, F.J.M.; Gomes, L.C. Use of Probiotics to Control Biofilm Formation in Food Industries. Antibiotics 2023, 12, 754. [Google Scholar] [CrossRef]
- Zhao, A.; Sun, J.; Liu, Y. Understanding bacterial biofilms: From definition to treatment strategies. Front. Cell. Infect. Microbiol. 2023, 13, 1137947. [Google Scholar] [CrossRef] [PubMed]
- Patrick Di, M. Extracellular polymeric substances, a key element in understanding biofilm phenotype. AIMS Microbiol. 2018, 4, 274–288. [Google Scholar] [CrossRef]
- Tursi, S.A.; Tükel, Ç. Curli-Containing Enteric Biofilms Inside and Out: Matrix Composition, Immune Recognition, and Disease Implications. Microbiol. Mol. Biol. Rev. 2018, 82, 4. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.T.T.; Nguyen, T.H.; Otto, M. The staphylococcal exopolysaccharide PIA–Biosynthesis and role in biofilm formation, colonization, and infection. Comput. Struct. Biotechnol. J. 2020, 18, 3324–3334. [Google Scholar] [CrossRef]
- GBD 2023 Cancer Collaborators. The global, regional, and national burden of cancer, 1990–2023, with forecasts to 2050: A systematic analysis for the Global Burden of Disease Study 2023. Lancet 2025, 406, 1565–1586. [Google Scholar] [CrossRef] [PubMed]
- Das, T.; Sharma, P.K.; Busscher, H.J.; Mei, H.C.v.d.; Krom, B.P. Role of Extracellular DNA in Initial Bacterial Adhesion and Surface Aggregation. Appl. Environ. Microbiol. 2010, 76, 3405–3408. [Google Scholar] [CrossRef]
- Kakar, M.; Berezovska, M.M.; Broks, R.; Asare, L.; Delorme, M.; Crouzen, E.; Zviedre, A.; Reinis, A.; Engelis, A.; Kroica, J.; et al. Serum and Urine Biomarker Leucine-Rich Alpha-2 Glycoprotein 1 Differentiates Pediatric Acute Complicated and Uncomplicated Appendicitis. Diagnostics 2021, 11, 860. [Google Scholar] [CrossRef]
- Poulin, M.B.; Kuperman, L.L. Regulation of Biofilm Exopolysaccharide Production by Cyclic Di-Guanosine Monophosphate. Front. Microbiol. 2021, 12, 730980. [Google Scholar] [CrossRef]
- Lozano Fernandez, M.E.; Miskolczi, N. Production of Cellulose Nano-Fibers and Its Application in Poly-Lactic-Acid: Property Improvement by New Types of Coupling Agents. Polymers 2022, 14, 1887. [Google Scholar] [CrossRef]
- Miranda, S.W.; Asfahl, K.L.; Dandekar, A.A.; Greenberg, E.P. Pseudomonas aeruginosa Quorum Sensing. In Pseudomonas aeruginosa: Biology, Pathogenesis and Control Strategies; Filloux, A., Ramos, J.-L., Eds.; Springer International Publishing: Cham, Switzerland, 2022; pp. 95–115. [Google Scholar] [CrossRef]
- Waters, C.M.; Lu, W.; Rabinowitz, J.D.; Bassler, B.L. Quorum Sensing Controls Biofilm Formation in Vibrio cholerae through Modulation of Cyclic Di-GMP Levels and Repression of vpsT. J. Bacteriol. 2008, 190, 2527–2536. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishnan, R.; Singh, A.K.; Singh, S.; Chakravortty, D.; Das, D. Enzymatic dispersion of biofilms: An emerging biocatalytic avenue to combat biofilm-mediated microbial infections. J. Biol. Chem. 2022, 298, 102352. [Google Scholar] [CrossRef]
- Harmsen, M.; Lappann, M.; Knøchel, S.; Molin, S. Role of Extracellular DNA during Biofilm Formation by Listeria monocytogenes. Appl. Environ. Microbiol. 2010, 76, 2271–2279. [Google Scholar] [CrossRef] [PubMed]
- Eddenden, A.; Nitz, M. Chapter Nine-Applications of an inactive Dispersin B probe to monitor biofilm polysaccharide production. In Methods in Enzymology; Carlson, E.E., Ed.; Academic Press: Cambridge, MA, USA, 2022; Volume 665, pp. 209–231. [Google Scholar] [CrossRef]
- Ramírez-Larrota, J.S.; Eckhard, U. An Introduction to Bacterial Biofilms and Their Proteases, and Their Roles in Host Infection and Immune Evasion. Biomolecules 2022, 12, 306. [Google Scholar] [CrossRef]
- Esposito, M.M.; Turku, S. The Use of Natural Methods to Control Foodborne Biofilms. Pathogens 2023, 12, 45. [Google Scholar] [CrossRef]
- Lu, L.; Zhao, Y.; Li, M.; Wang, X.; Zhu, J.; Liao, L.; Wang, J. Contemporary strategies and approaches for characterizing composition and enhancing biofilm penetration targeting bacterial extracellular polymeric substances. J. Pharm. Anal. 2024, 14, 100906. [Google Scholar] [CrossRef]
- Roy, R.; Monalisa, T.; Gianfranco, D.; Tiwari, V. Strategies for combating bacterial biofilms: A focus on anti-biofilm agents and their mechanisms of action. Virulence 2018, 9, 522–554. [Google Scholar] [CrossRef]
- Li, Y.; Qu, Y.; Yang, H.; Zhou, X.; Xiao, P.; Shao, T. Combatting biofilms in potable water systems: A comprehensive overview to ensuring industrial water safety. Environ. Microbiol. Rep. 2023, 15, 445–454. [Google Scholar] [CrossRef]
- Kashi, M.; Noei, M.; Chegini, Z.; Shariati, A. Natural compounds in the fight against Staphylococcus aureus biofilms: A review of antibiofilm strategies. Front. Pharmacol. 2024, 15, 1491363. [Google Scholar] [CrossRef] [PubMed]
- Naga, N.G.; Shaaban, M.I.; El-Metwally, M.M. An insight on the powerful of bacterial quorum sensing inhibition. Eur. J. Clin. Microbiol. Infect. Dis. 2024, 43, 2071–2081. [Google Scholar] [CrossRef]
- Sauer, K.; Stoodley, P.; Goeres, D.M.; Hall-Stoodley, L.; Burmølle, M.; Stewart, P.S.; Bjarnsholt, T. The biofilm life cycle: Expanding the conceptual model of biofilm formation. Nat. Rev. Microbiol. 2022, 20, 608–620. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, H.A.H.; Abou Elhassayeb, H.E.; El-Sayed, W.M.M. Potential functions and applications of diverse microbial exopolysaccharides in marine environments. J. Genet. Eng. Biotechnol. 2022, 20, 151. [Google Scholar] [CrossRef] [PubMed]
- Araújo, D.; Silva, A.R.; Fernandes, R.; Serra, P.; Barros, M.M.; Campos, A.M.; Oliveira, R.; Silva, S.; Almeida, C.; Castro, J. Emerging Approaches for Mitigating Biofilm-Formation-Associated Infections in Farm, Wild, and Companion Animals. Pathogens 2024, 13, 320. [Google Scholar] [CrossRef]
- Solano, C.; Echeverz, M.; Lasa, I. Biofilm dispersion and quorum sensing. Curr. Opin. Microbiol. 2014, 18, 96–104. [Google Scholar] [CrossRef]
- Koo, H.; Allan, R.N.; Howlin, R.P.; Stoodley, P.; Hall-Stoodley, L. Targeting microbial biofilms: Current and prospective therapeutic strategies. Nat. Rev. Microbiol. 2017, 15, 740–755. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, S.; Gomes, I.B.; Simões, M.; Simões, L.C. Novel chemical-based approaches for biofilm cleaning and disinfection. Curr. Opin. Food Sci. 2024, 55, 101124. [Google Scholar] [CrossRef]
- Klopper, K.B.; Bester, E.; Wolfaardt, G.M. Listeria monocytogenes Biofilms Are Planktonic Cell Factories despite Peracetic Acid Exposure under Continuous Flow Conditions. Antibiotics 2023, 12, 209. [Google Scholar] [CrossRef]
- Almatroudi, A. Biofilm Resilience: Molecular Mechanisms Driving Antibiotic Resistance in Clinical Contexts. Biology 2025, 14, 165. [Google Scholar] [CrossRef]
- Lu, J.; Hu, X.; Ren, L. Biofilm control strategies in food industry: Inhibition and utilization. Trends Food Sci. Technol. 2022, 123, 103–113. [Google Scholar] [CrossRef]
- Yin, R.; Cheng, J.; Wang, J.; Li, P.; Lin, J. Treatment of Pseudomonas aeruginosa infectious biofilms: Challenges and strategies. Front. Microbiol. 2022, 13, 955286. [Google Scholar] [CrossRef] [PubMed]
- Ahn, S.-J.; Burne, R.A. Effects of Oxygen on Biofilm Formation and the AtlA Autolysin of Streptococcus mutans. J. Bacteriol. 2007, 189, 6293–6302. [Google Scholar] [CrossRef]
- Coughlan, L.M.; Cotter, P.D.; Hill, C.; Alvarez-Ordóñez, A. New Weapons to Fight Old Enemies: Novel Strategies for the (Bio)control of Bacterial Biofilms in the Food Industry. Front. Microbiol. 2016, 7, 1641. [Google Scholar] [CrossRef]
- Uruén, C.; Chopo-Escuin, G.; Tommassen, J.; Mainar-Jaime, R.C.; Arenas, J. Biofilms as Promoters of Bacterial Antibiotic Resistance and Tolerance. Antibiotics 2021, 10, 3. [Google Scholar] [CrossRef]
- Kunnath, A.P.; Suodha Suoodh, M.; Chellappan, D.K.; Chellian, J.; Palaniveloo, K. Bacterial Persister Cells and Development of Antibiotic Resistance in Chronic Infections: An Update. Br. J. Biomed. Sci. 2024, 81, 12958. [Google Scholar] [CrossRef] [PubMed]
- Wood, T.K.; Knabel, S.J.; Kwan, B.W. Bacterial Persister Cell Formation and Dormancy. Appl. Environ. Microbiol. 2013, 79, 7116–7121. [Google Scholar] [CrossRef] [PubMed]
- Grassi, L.; Di Luca, M.; Maisetta, G.; Rinaldi, A.C.; Esin, S.; Trampuz, A.; Batoni, G. Generation of Persister Cells of Pseudomonas aeruginosa and Staphylococcus aureus by Chemical Treatment and Evaluation of Their Susceptibility to Membrane-Targeting Agents. Front. Microbiol. 2017, 8, 1917. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Tong, Y.; Cheng, J.; Abbas, Z.; Li, Z.; Wang, J.; Zhou, Y.; Si, D.; Zhang, R. Biofilm and Small Colony Variants—An Update on Staphylococcus aureus Strategies toward Drug Resistance. Int. J. Mol. Sci. 2022, 23, 1241. [Google Scholar] [CrossRef]
- Dusane, D.H.; Zinjarde, S.S.; Venugopalan, V.P.; Mclean, R.J.; Weber, M.M.; Rahman, P.K. Quorum sensing: Implications on Rhamnolipid biosurfactant production. Biotechnol. Genet. Eng. Rev. 2010, 27, 159–184. [Google Scholar] [CrossRef]
- D’Aquila, P.; De Rose, E.; Sena, G.; Scorza, A.; Cretella, B.; Passarino, G.; Bellizzi, D. Quorum Quenching Approaches against Bacterial-Biofilm-Induced Antibiotic Resistance. Antibiotics 2024, 13, 619. [Google Scholar] [CrossRef]
- Ng, W.-L.; Perez, L.; Cong, J.; Semmelhack, M.F.; Bassler, B.L. Broad Spectrum Pro-Quorum-Sensing Molecules as Inhibitors of Virulence in Vibrios. PLoS Pathog. 2012, 8, e1002767. [Google Scholar] [CrossRef]
- Anju, V.T.; Busi, S.; Imchen, M.; Kumavath, R.; Mohan, M.S.; Salim, S.A.; Subhaswaraj, P.; Dyavaiah, M. Polymicrobial Infections and Biofilms: Clinical Significance and Eradication Strategies. Antibiotics 2022, 11, 1731. [Google Scholar] [CrossRef]
- Barraud, N.; Buson, A.; Jarolimek, W.; Rice, S.A. Mannitol Enhances Antibiotic Sensitivity of Persister Bacteria in Pseudomonas aeruginosa Biofilms. PLoS ONE 2013, 8, e84220. [Google Scholar] [CrossRef]
- Mazaheri, T.; Cervantes-Huamán, B.R.H.; Bermúdez-Capdevila, M.; Ripolles-Avila, C.; Rodríguez-Jerez, J.J. Listeria monocytogenes Biofilms in the Food Industry: Is the Current Hygiene Program Sufficient to Combat the Persistence of the Pathogen? Microorganisms 2021, 9, 181. [Google Scholar] [CrossRef]
- Muñoz-Cázares, N.; Castillo-Juárez, I.; García-Contreras, R.; Castro-Torres, V.A.; Díaz-Guerrero, M.; Rodríguez-Zavala, J.S.; Quezada, H.; González-Pedrajo, B.; Martínez-Vázquez, M. A Brominated Furanone Inhibits Pseudomonas aeruginosa Quorum Sensing and Type III Secretion, Attenuating Its Virulence in a Murine Cutaneous Abscess Model. Biomedicines 2022, 10, 1847. [Google Scholar] [CrossRef]
- Brackman, G.; Cos, P.; Maes, L.; Nelis, H.J.; Coenye, T. Quorum Sensing Inhibitors Increase the Susceptibility of Bacterial Biofilms to Antibiotics In Vitro and In Vivo. Antimicrob. Agents Chemother. 2011, 55, 2655–2661. [Google Scholar] [CrossRef]
- Dawan, J.; Zhang, S.; Ahn, J. Recent Advances in Biofilm Control Technologies for the Food Industry. Antibiotics 2025, 14, 254. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Sauer, K. Controlling Biofilm Development Through Cyclic di-GMP Signaling. In Pseudomonas aeruginosa: Biology, Pathogenesis and Control Strategies; Filloux, A., Ramos, J.-L., Eds.; Springer International Publishing: Cham, Switzerland, 2022; pp. 69–94. [Google Scholar] [CrossRef]
- Martínez-Méndez, R.; Camacho-Hernández, D.A.; Sulvarán-Guel, E.; Zamorano-Sánchez, D. A Trigger Phosphodiesterase Modulates the Global c-di-GMP Pool, Motility, and Biofilm Formation in Vibrio parahaemolyticus. J. Bacteriol. 2021, 203, 13. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.D. Breathing new life into nitric oxide signaling: A brief overview of the interplay between oxygen and nitric oxide. Redox Biol. 2015, 5, 225–233. [Google Scholar] [CrossRef]
- Qin, S.; Xiao, W.; Zhou, C.; Pu, Q.; Deng, X.; Lan, L.; Liang, H.; Song, X.; Wu, M. Pseudomonas aeruginosa: Pathogenesis, virulence factors, antibiotic resistance, interaction with host, technology advances and emerging therapeutics. Signal Transduct. Target. Ther. 2022, 7, 199. [Google Scholar] [CrossRef]
- Mirghani, R.; Saba, T.; Khaliq, H.; Mitchell, J.; Do, L.; Chambi, L.; Diaz, K.; Kennedy, T.; Alkassab, K.; Huynh, T.; et al. Biofilms: Formation, drug resistance and alternatives to conventional approaches. AIMS Microbiol. 2022, 8, 239–277. [Google Scholar] [CrossRef] [PubMed]
- Lebeaux, D.; Ghigo, J.-M.; Beloin, C. Biofilm-Related Infections: Bridging the Gap between Clinical Management and Fundamental Aspects of Recalcitrance toward Antibiotics. Microbiol. Mol. Biol. Rev. 2014, 78, 510–543. [Google Scholar] [CrossRef]
- Kaplan, J.B.; Sukhishvili, S.A.; Sailer, M.; Kridin, K.; Ramasubbu, N. Aggregatibacter actinomycetemcomitans Dispersin B: The Quintessential Antibiofilm Enzyme. Pathogens 2024, 13, 668. [Google Scholar] [CrossRef]
- Karatan, E.; Watnick, P. Signals, Regulatory Networks, and Materials That Build and Break Bacterial Biofilms. Microbiol. Mol. Biol. Rev. 2009, 73, 310–347. [Google Scholar] [CrossRef]
- Poimenidou, S.V.; Manios, S.G.; Skandamis, P.N. Control of Listeria monocytogenes Biofilms in a Simulated Food-Processing Environment. In Listeria Monocytogenes: Methods and Protocols; Fox, E.M., Bierne, H., Stessl, B., Eds.; Springer: New York, NY, USA, 2021; pp. 219–231. [Google Scholar] [CrossRef]
- Maillard, J.-Y.; Centeleghe, I. How biofilm changes our understanding of cleaning and disinfection. Antimicrob. Resist. Infect. Control 2023, 12, 95. [Google Scholar] [CrossRef]
- Michaelis, C.; Grohmann, E. Horizontal Gene Transfer of Antibiotic Resistance Genes in Biofilms. Antibiotics 2023, 12, 328. [Google Scholar] [CrossRef]
- Howlin, R.P.; Cathie, K.; Hall-Stoodley, L.; Cornelius, V.; Duignan, C.; Allan, R.N.; Fernandez, B.O.; Barraud, N.; Bruce, K.D.; Jefferies, J.; et al. Low-Dose Nitric Oxide as Targeted Anti-biofilm Adjunctive Therapy to Treat Chronic Pseudomonas aeruginosa Infection in Cystic Fibrosis. Mol. Ther. 2017, 25, 2104–2116. [Google Scholar] [CrossRef]
- Hentzer, M.; Riedel, K.; Rasmussen, T.B.; Heydorn, A.; Andersen, J.B.; Parsek, M.R.; Rice, S.A.; Eberl, L.; Molin, S.; Høiby, N.; et al. Inhibition of quorum sensing in Pseudomonas aeruginosa biofilm bacteria by a halogenated furanone compound. Microbiology 2002, 148, 87–102. [Google Scholar] [CrossRef] [PubMed]
- Arthur, M.; Afari, E.L.; Alexa, E.-A.; Zhu, M.-J.; Gaffney, M.T.; Celayeta, J.M.F.; Burgess, C.M. Recent advances in examining the factors influencing the efficacy of biocides against Listeria monocytogenes biofilms in the food industry: A systematic review. Compr. Rev. Food Sci. Food Saf. 2025, 24, e70083. [Google Scholar] [CrossRef] [PubMed]
- Taiwo, O.R.; Onyeaka, H.; Oladipo, E.K.; Oloke, J.K.; Chukwugozie, D.C. Advancements in Predictive Microbiology: Integrating New Technologies for Efficient Food Safety Models. Int. J. Microbiol. 2024, 2024, 6612162. [Google Scholar] [CrossRef]
- Gemba, M.; Rosiak, E.; Kołożyn-Krajewska, D. Development of predictive models of biofilm formation by C. sakazakii, E. cloacae on surfaces used in the food industry and medicine. Int. J. Food Microbiol. 2025, 434, 111131. [Google Scholar] [CrossRef] [PubMed]
- Simeone, A.; Woolley, E.; Escrig, J.; Watson, N.J. Intelligent Industrial Cleaning: A Multi-Sensor Approach Utilising Machine Learning-Based Regression. Sensors 2020, 20, 3642. [Google Scholar] [CrossRef]
- Zuo, P.; Metz, J.; Yu, P.; Alvarez, P.J.J. Biofilm-responsive encapsulated-phage coating for autonomous biofouling mitigation in water storage systems. Water Res. 2022, 224, 119070. [Google Scholar] [CrossRef]
- Choi, H.S.; Yun, J.; Jeong, Y.; Jo, Y.K.; Cha, H.J. Self-controllable proteinic antibacterial coating with bacteria-triggered antibiotic release for prevention of periprosthetic infection. Biomaterials 2024, 305, 122457. [Google Scholar] [CrossRef]
- Cassa, M.A.; Gentile, P.; Girón-Hernández, J.; Ciardelli, G.; Carmagnola, I. Smart self-defensive coatings with bacteria-triggered antimicrobial response for medical devices. Biomater. Sci. 2024, 12, 5433–5449. [Google Scholar] [CrossRef]
- Kumar, S.; Nguyen, A.T.; Goswami, S.; Ferracane, J.; Koley, D. Real-time monitoring of biofilm formation using a noninvasive impedance-based method. Sens. Actuators B Chem. 2023, 376, 133034. [Google Scholar] [CrossRef]
- Rakhimbekova, A.; Kudaibergenov, B.; Moldabay, D.; Zharylgap, A.; Ajunwa, O.M.; Marsili, E.; Tosi, D. Biofilm Detection by a Fiber-Tip Ball Resonator Optical Fiber Sensor. Biosensors 2022, 12, 481. [Google Scholar] [CrossRef]
- Shafaat, A.; Gonzalez-Martinez, J.F.; Silva, W.O.; Lesch, A.; Nagar, B.; Lopes da Silva, Z.; Neilands, J.; Sotres, J.; Björklund, S.; Girault, H.; et al. A Rapidly Responsive Sensor for Wireless Detection of Early and Mature Microbial Biofilms. Angew. Chem. Int. Ed. 2023, 62, e202308181. [Google Scholar] [CrossRef] [PubMed]
- Gencay, Y.E.; Jasinskytė, D.; Robert, C.; Semsey, S.; Martínez, V.; Petersen, A.Ø.; Brunner, K.; de Santiago Torio, A.; Salazar, A.; Turcu, I.C.; et al. Engineered phage with antibacterial CRISPR–Cas selectively reduce E. coli burden in mice. Nat. Biotechnol. 2024, 42, 265–274. [Google Scholar] [CrossRef] [PubMed]
- Bonneville, L.; Maia, V.; Barroso, I.; Martínez-Suárez, J.V.; Brito, L. Lactobacillus plantarum in Dual-Species Biofilms With Listeria monocytogenes Enhanced the Anti-Listeria Activity of a Commercial Disinfectant Based on Hydrogen Peroxide and Peracetic Acid. Front. Microbiol. 2021, 12, 631627. [Google Scholar] [CrossRef]
- Geurtsen, J.; de Been, M.; Weerdenburg, E.; Zomer, A.; McNally, A.; Poolman, J. Genomics and pathotypes of the many faces of Escherichia coli. FEMS Microbiol. Rev. 2022, 46, fuac031. [Google Scholar] [CrossRef]
- Jin, X.; Sun, X.; Lu, Q.; Wang, Z.; Zhang, Z.; Ling, X.; Xu, Y.; Liang, R.; Yang, J.; Li, L.; et al. Salmonella Phage vB_SpuM_X5: A Novel Approach to Reducing Salmonella Biofilms with Implications for Food Safety. Microorganisms 2024, 12, 2400. [Google Scholar] [CrossRef] [PubMed]


| Biofilm Stage | Intervention Strategies | Molecular Targets/Mechanisms | Advantages | Limitations | Industrial Applicability | References |
|---|---|---|---|---|---|---|
| Initial Adhesion | Surface Engineering and Material Modification (e.g., nano-silica coating) | Alters surface hydrophobicity, charge, roughness | Non-biocidal, reduces bacterial attachment, easier cleaning | Limited coating durability, higher cost | Food-contact surfaces (stainless steel, conveyor belts) | [16] |
| Natural Anti-adhesion Compounds (e.g., polyphenols, essential oils) | Alters bacterial surface hydrophobicity, blocks adhesins | Green, safe, food-compatible | Efficacy is concentration-dependent, volatile | Treatment of food processing equipment surfaces | [17,18,19] | |
| Competitive Exclusion by Probiotics | Occupies binding sites, secretes antimicrobials (e.g., bacteriocins) | Self-sustaining, no chemical residues | Requires robust, cleaning-tolerant strains | Dairy, meat processing equipment | [20] | |
| EPS Formation and Maturation | Enzymatic Degradation (e.g., DNase, Dispersin B, proteases) | Degrades EPS components (eDNA, polysaccharides, proteins) | High specificity, enhances sanitizer penetration | Enzyme stability issues, high cost | Dairy industry, pipeline systems | [32,33,34,35] |
| EPS-disrupting Surfactants (e.g., rhamnolipids) | Disrupts polysaccharide-protein interactions | Enhances antimicrobial penetration, biodegradable | Concentration-dependent, may affect product sensory | Food processing surfaces in combination with cleaners | [8,36,37] | |
| Microenvironment Manipulation (e.g., nutrient pulses, temperature, pH shifts) | Activates dormant cells, disrupts internal homeostasis | Increases antimicrobial susceptibility, no chemical residues | Difficult to control, requires precise operation | Liquid food processing systems | [48,49,50,51,52,53] | |
| Maturation: Persisters and QS | “Wake-and-Kill” Strategy (e.g., mannitol, glucose) | Metabolic reactivation of persister cells | Effective against dormant cells, enhances eradication | Requires precise control of concentration and timing | High-risk areas (filler heads, pipelines) | [63,64] |
| Quorum Sensing Inhibitors (QSIs, e.g., halogenated furanones, flavonoids) | Blocks QS signaling, inhibits EPS synthesis and persister formation | Low resistance risk, synergistic with sanitizers | Natural QSIs have low stability, synthetic ones are costly | Broadly applicable in various food processing environments | [40,41,65,66,67] | |
| Dispersion | Induced Controlled Dispersion (e.g., NO-donors, QSIs) | Lowers c-di-GMP levels, activates dispersal mechanisms | Synchronized dispersal allows centralized removal | Timing is hard to control, risk of secondary contamination | Pipes, wet processing areas | [79,80] |
| Physical Removal (e.g., high-pressure water, ultrasonication) | Mechanical disruption of EPS structure | Fast, effective, no chemical residues | May damage equipment, generates aerosols | Large equipment, hard-to-reach surfaces | [53] | |
| Bacteriophage Treatment | Specific lysis of dispersed cells | Self-amplifying, no residues, highly targeted | Narrow host range, potential for resistance development | Targeted pathogen control (e.g., Listeria) | [81,94] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, L.; Xu, B.; Tao, Y.; Liang, Y.; Chen, X. Exploring Intervention Strategies for Microbial Biofilms in the Food Industry Based on a Biomolecular Mechanism Perspective: Recent Advances and Emerging Trends. Foods 2025, 14, 4192. https://doi.org/10.3390/foods14244192
Sun L, Xu B, Tao Y, Liang Y, Chen X. Exploring Intervention Strategies for Microbial Biofilms in the Food Industry Based on a Biomolecular Mechanism Perspective: Recent Advances and Emerging Trends. Foods. 2025; 14(24):4192. https://doi.org/10.3390/foods14244192
Chicago/Turabian StyleSun, Luchuanyang, Bingbing Xu, Ye Tao, Yan Liang, and Xianggui Chen. 2025. "Exploring Intervention Strategies for Microbial Biofilms in the Food Industry Based on a Biomolecular Mechanism Perspective: Recent Advances and Emerging Trends" Foods 14, no. 24: 4192. https://doi.org/10.3390/foods14244192
APA StyleSun, L., Xu, B., Tao, Y., Liang, Y., & Chen, X. (2025). Exploring Intervention Strategies for Microbial Biofilms in the Food Industry Based on a Biomolecular Mechanism Perspective: Recent Advances and Emerging Trends. Foods, 14(24), 4192. https://doi.org/10.3390/foods14244192







