Discrimination Between Commercial Tomato Juices from Non-Concentrate and Concentrate Based on Their Volatile Profiles
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples
2.2. Reagents and Standards
2.3. Measurement of the Basic Properties of the Juices
2.4. Sensory Evaluation
2.5. VOC Extraction by Headspace Gas/Solid-Phase Microextraction (HS-SPME)
2.6. Confirmation of the Aroma Characteristics of VOCs by GC-MS/Olfactometry (GC-MS/O) Analysis
2.7. Preparation of Model Juice Samples by Concentration and Recovery
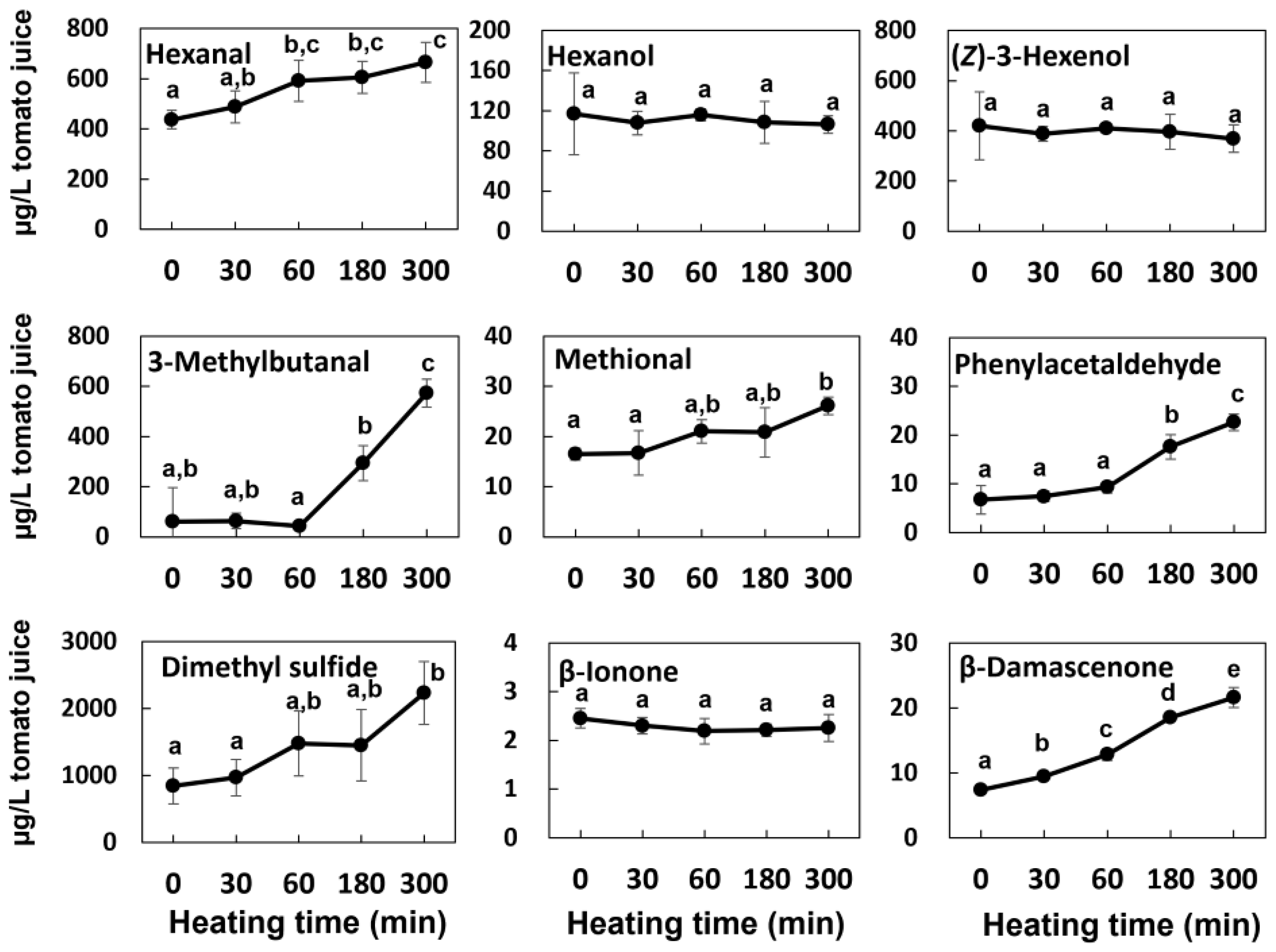
2.8. Stability of VOCs in the Sealed Tomato Juice While Heating
2.9. GC-MS Conditions
2.10. Compound Identification and Quantification
2.11. Data Processing, Multivariate Analysis, and Statistical Analysis
3. Results and Discussion
3.1. Differences in Flavor Characteristics Between JFC and JNFC, as Determined by Sensory Evaluation
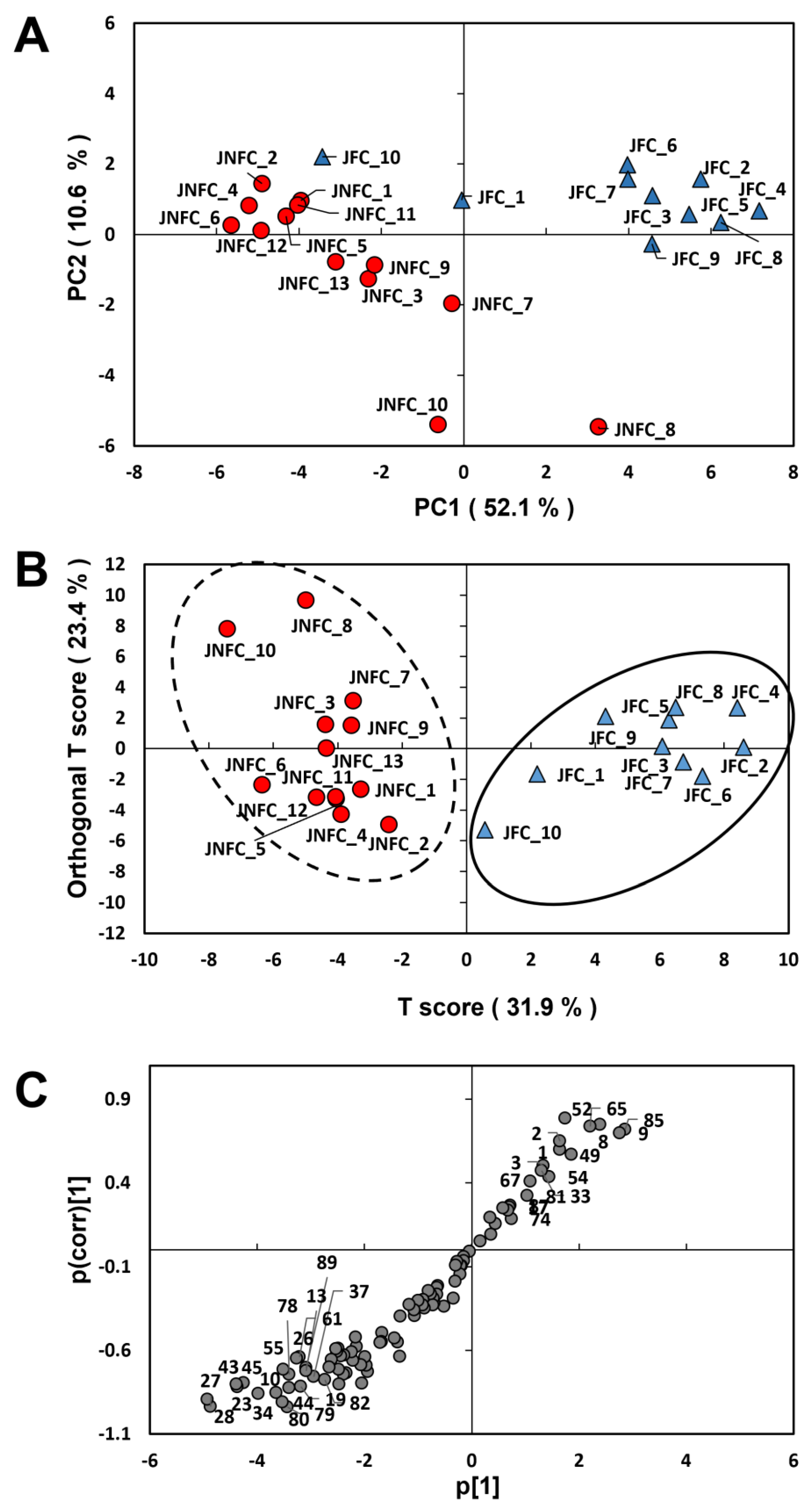
3.2. Differentiation of Tomato Juices Based on VOC Composition
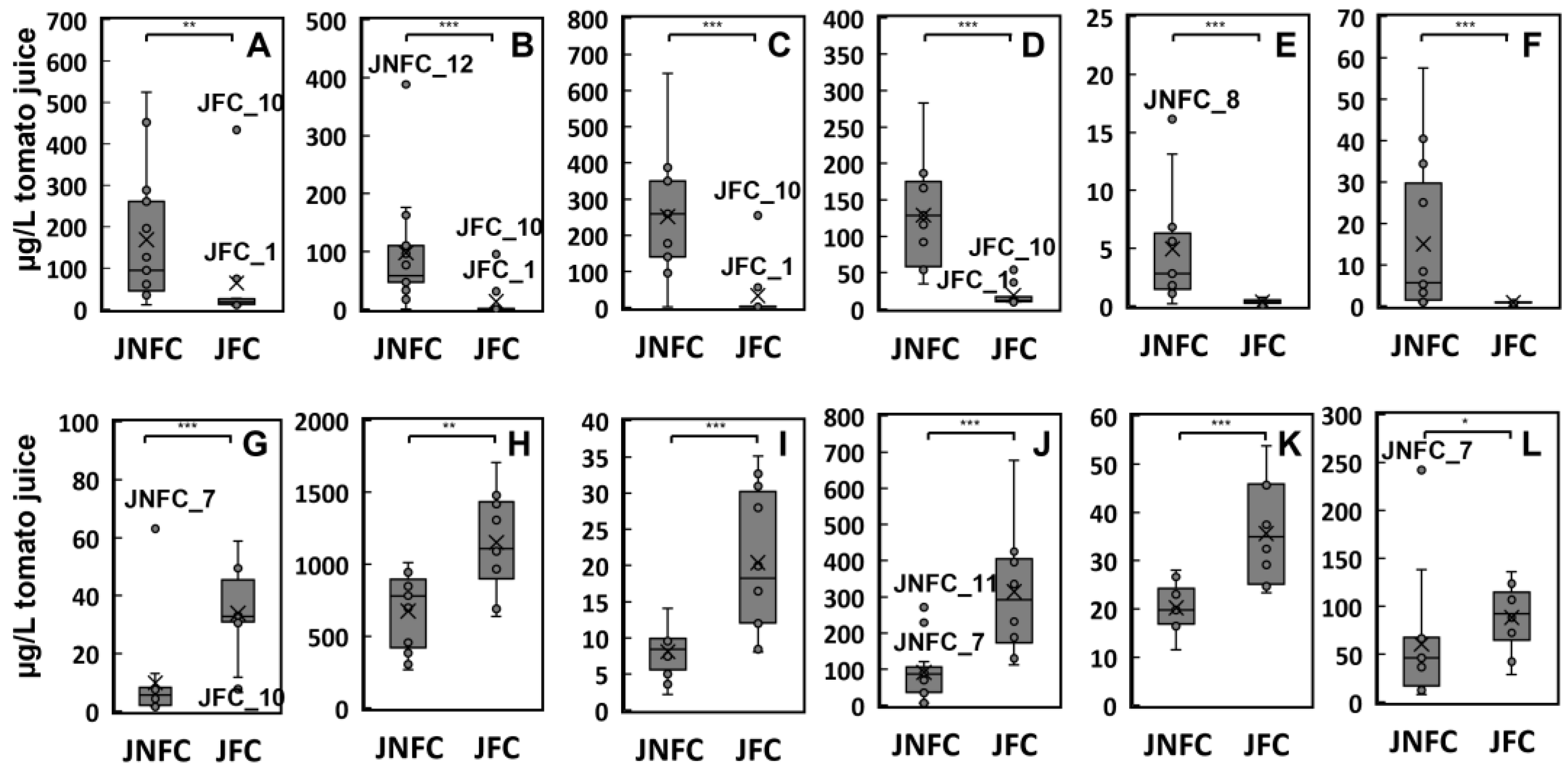
3.3. Odor Activity of Aroma Active Compounds
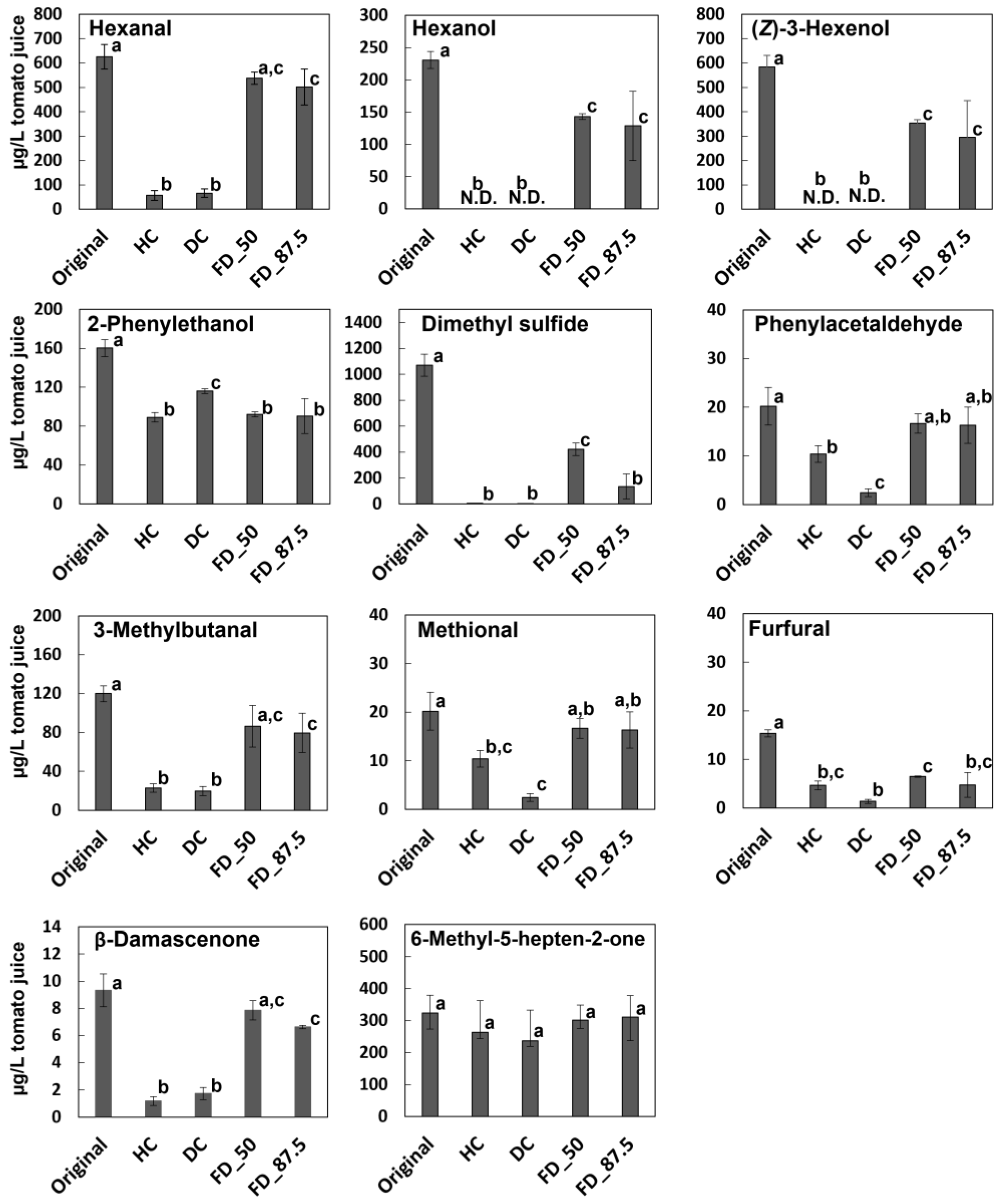
3.4. Effect of Concentration Condition on the Aroma Profiles of Reduced Tomato Juices
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| VOC | Volatile organic compounds |
| JNFC | Juice not from concentrate |
| JFC | Juice from concentrate |
| DVB/CAR/PDMS | Divinylbenzene/carboxen/polysimethylsiloxane |
| HS-SPME-GC-MS | Headspace solid phase microextraction -gas chromatography mass spectrometry |
| PCA | Principal component analysis |
| OPLS-DA | Orthogonal partial least squares discriminant analysis |
| GC-MS/O | GC-MS/olfactometry |
| SAFE | Solvent-assisted flavor evaporation |
References
- Petro-Turza, M. Flavor of tomato and tomato products. Food Rev. Int. 1986, 2, 309–351. [Google Scholar] [CrossRef]
- Nasir, M.U.; Hussain, S.; Jabbar, S. Tomato processing, lycopene and health benefits: A review. Sci. Lett. 2015, 3, 1–5. [Google Scholar]
- Zhu, Y.; Shen, M.; Sims, C.A.; Marshall, M.R.; House, L.A.; Sarnoski, P.J. Consumer preference and willingness to pay for tomato juice. Int. Food Agribus. Manag. 2018, 21, 1167–1184. [Google Scholar] [CrossRef]
- Mirondo, R.; Barringer, S. Improvement of flavor and viscosity in hot and cold break tomato juice and sauce by peel removal. J. Food Sci. 2015, 80, S171–S179. [Google Scholar] [CrossRef]
- Goodman, C.L.; Fawcett, S.; Barringer, S. Flavor, viscosity, and color analyses of hot and cold break tomato juices. J. Food Sci. 2002, 67, 404–408. [Google Scholar] [CrossRef]
- Iijima, Y.; Iwasaki, Y.; Otagiri, Y.; Tsugawa, H.; Sato, T.; Otomo, H.; Sekine, Y.; Obata, A. Flavor characteristics of the juices from fresh market tomatoes differentiated from those from processing tomatoes by combined analysis of volatile profiles with sensory evaluation. Biosci. Biotechnol. Biochem. 2016, 80, 2401–2411. [Google Scholar] [CrossRef][Green Version]
- Servili, M.; Selvaggini, R.; Taticchi, A.; Begliomini, A.L.; Montedoro, G. Relationships between the volatile compounds evaluated by solid phase microextraction and the thermal treatment of tomato juice: Optimization of the blanching parameters. Food Chem. 2000, 71, 407–415. [Google Scholar] [CrossRef]
- Koltun, S.J.; MacIntosh, A.J.; Goodrich-Schneider, R.M.; Klee, H.J.; Hutton, S.F.; Junoy, L.J.; Sarnoski, P.J. Effects of thermal processing on flavor and consumer perception using tomato juice produced from Florida grown fresh market cultivars. J. Food Process. Preserv. 2022, 46, e16164. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, C.; Wang, Q.; Zhang, J.; Zhang, L. Identification and confirmation of key compounds causing cooked off-flavor in heat-treated tomato juice. J. Food Sci. 2022, 87, 2515–2526. [Google Scholar] [CrossRef]
- Deshpande, S.; Cheryan, M.; Sathe, S.K.; Salunkhe, D.; Luh, B. Freeze concentration of fruit juices. Crit. Rev. Food Sci. Nutr. 1984, 20, 173–248. [Google Scholar] [CrossRef]
- Yoda, T.; Miyaki, H.; Saito, T. Freeze concentrated apple juice maintains its flavor. Sci. Rep. 2021, 11, 12679. [Google Scholar] [CrossRef]
- Jiao, B.; Cassano, A.; Drioli, E. Recent advances on membrane processes for the concentration of fruit juices: A review. J. Food Eng. 2004, 63, 303–324. [Google Scholar] [CrossRef]
- Sarbatly, R.; Sariau, J.; Krishnaiah, D. Recent developments of membrane technology in the clarification and concentration of fruit juices. Food Eng. Rev. 2023, 15, 420–437. [Google Scholar] [CrossRef]
- Bahçeci, K.S.; Akıllıoğlu, H.G.; Gökmen, V. Osmotic and membrane distillation for the concentration of tomato juice: Effects on quality and safety characteristics. Innov. Food Sci. Emerg. Technol. 2015, 31, 131–138. [Google Scholar] [CrossRef]
- Zhou, X.; Wang, W.; Ma, X.; Xu, E.; Liu, D. Ultrasonication of Thawed Huyou Juice: Effects on cloud stability, physicochemical properties and bioactive compounds. Foods 2021, 10, 1695. [Google Scholar] [CrossRef]
- Jitsuyama, Y.; Ichiki, A.; Ide, R.; Shimura, H.; Suzuki, T. The processing tomato cultivar ‘Natsunoshun’is susceptible to an excess or lack of soil moisture after the flowering stage. Hortic. J. 2019, 88, 232–244. [Google Scholar] [CrossRef]
- ISO 5495:2005; Sensory Analysis-Methodology-Paired Comparison Test. ISO: Geneva, Switzerland, 2005. Available online: https://www.iso.org/standard/31621.html (accessed on 10 August 2025).
- Baba, R.; Amano, Y.; Wada, Y.; Kumazawa, K. Characterization of the potent odorants contributing to the characteristic aroma of matcha by gas chromatography–olfactometry techniques. J. Agric. Food Chem. 2017, 65, 2984–2989. [Google Scholar] [CrossRef]
- Pang, Z.; Chong, J.; Zhou, G.; de Lima Morais, D.A.; Chang, L.; Barrette, M.; Gauthier, C.; Jacques, P.-É.; Li, S.; Xia, J. MetaboAnalyst 5.0: Narrowing the gap between raw spectra and functional insights. Nucleic Acids Res. 2021, 49, W388–W396. [Google Scholar] [CrossRef]
- Buttery, R.G.; Teranishi, R.; Ling, L.C. Fresh tomato aroma volatiles: A quantitative study. J. Agric. Food Chem. 1987, 35, 540–544. [Google Scholar] [CrossRef]
- Selli, S.; Kelebek, H.; Ayseli, M.T.; Tokbas, H. Characterization of the most aroma-active compounds in cherry tomato by application of the aroma extract dilution analysis. Food Chem. 2014, 165, 540–546. [Google Scholar] [CrossRef]
- Wang, L.; Baldwin, E.A.; Bai, J. Recent advance in aromatic volatile research in tomato fruit: The metabolisms and regulations. Food Bioprocess Technol. 2016, 9, 203–216. [Google Scholar] [CrossRef]
- Baenas, N.; Bravo, S.; García-Alonso, F.J.; Gil, J.V.; Periago, M.J. Changes in volatile compounds, flavour-related enzymes and lycopene in a refrigerated tomato juice during processing and storage. Eur. Food Res. Technol. 2021, 247, 975–984. [Google Scholar] [CrossRef]
- Jia, X.; Ren, J.; Fan, G.; Reineccius, G.A.; Li, X.; Zhang, N.; An, Q.; Wang, Q.; Pan, S. Citrus juice off-flavor during different processing and storage: Review of odorants, formation pathways, and analytical techniques. Crit. Rev. Food Sci. Nutr. 2024, 64, 3018–3043. [Google Scholar] [CrossRef]
- An, K.; Liu, H.; Fu, M.; Qian, M.C.; Yu, Y.; Wu, J.; Xiao, G.; Xu, Y. Identification of the cooked off-flavor in heat-sterilized lychee (Litchi chinensis Sonn.) juice by means of molecular sensory science. Food Chem. 2019, 301, 125282. [Google Scholar] [CrossRef]
- Dhuey, E.; Castada, H.Z.; Barringer, S.; Joseph, J.; Hadad, C.M.; Ruffley, K.; Pascall, M.A. Heat-induced compounds development in processed tomato and their influence on corrosion initiation in metal food cans. Food Sci. Nutr. 2021, 9, 4134–4145. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, T.; Münch, P.; Schieberle, P. Quantitative model studies on the formation of aroma-active aldehydes and acids by Strecker-type reactions. J. Agric. Food Chem. 2000, 48, 434–440. [Google Scholar] [CrossRef] [PubMed]
- Distefano, M.; Mauro, R.P.; Page, D.; Giuffrida, F.; Bertin, N.; Leonardi, C. Aroma volatiles in tomato fruits: The role of genetic, preharvest and postharvest factors. Agronomy 2022, 12, 376. [Google Scholar] [CrossRef]
- Wang, L.; Qian, C.; Bai, J.; Luo, W.; Jin, C.; Yu, Z. Difference in volatile composition between the pericarp tissue and inner tissue of tomato (Solanum lycopersicum) fruit. J. Food Process. Preserv. 2018, 42, e13387. [Google Scholar] [CrossRef]
- Cheng, G.; Chang, P.; Shen, Y.; Wu, L.; El-Sappah, A.H.; Zhang, F.; Liang, Y. Comparing the Flavor Characteristics of 71 Tomato (Solanum lycopersicum) Accessions in Central Shaanxi. Front. Plant Sci. 2020, 11, 586834. [Google Scholar] [CrossRef]
- Czerny, M.; Christlbauer, M.; Christlbauer, M.; Fischer, A.; Granvogl, M.; Hammer, M.; Hartl, C.; Hernandez, N.M.; Schieberle, P. Re-investigation on odour thresholds of key food aroma compounds and development of an aroma language based on odour qualities of defined aqueous odorant solutions. Eur. Food Res. Technol. 2008, 228, 265–273. [Google Scholar] [CrossRef]
- Kreissl, J.; Schieberle, P. Characterization of Aroma-Active Compounds in Italian Tomatoes with Emphasis on New Odorants. J. Agric. Food Chem. 2017, 65, 5198–5208. [Google Scholar] [CrossRef]
- Agcam, E. A kinetic approach to explain hydroxymethylfurfural and furfural formations induced by Maillard, caramelization, and ascorbic acid degradation reactions in fruit juice-based mediums. Food Anal. Methods 2022, 15, 1286–1299. [Google Scholar] [CrossRef]
- Jeyaprakash, S.; Heffernan, J.E.; Driscoll, R.H.; Frank, D.C. Impact of drying technologies on tomato flavor composition and sensory quality. LWT 2020, 120, 108888. [Google Scholar] [CrossRef]
- Ozkaya, O.; Sen, K.; Aubert, C.; Dundar, O.; Gunata, Z. Characterization of the free and glycosidically bound aroma potential of two important tomato cultivars grown in Turkey. J. Food Sci. Technol. 2018, 55, 4440–4449. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Quek, S.Y. Free and glycosidically bound aroma compounds in fruit: Biosynthesis, transformation, and practical control. Crit. Rev. Food Sci. Nutr. 2023, 63, 9052–9073. [Google Scholar] [CrossRef] [PubMed]
| Sample No. | Origin of Sample * | Brix (°) | Acidity (w/v %) | Viscosity (mPa/s) |
|---|---|---|---|---|
| JNFC_1 | JNFC | 4.9 | 0.52 | 73 |
| JNFC_2 | JNFC | 5.1 | 0.84 | 79 |
| JNFC_3 | JNFC | 4.6 | 0.56 | 84 |
| JNFC_4 | JNFC | 4.7 | 0.52 | 116 |
| JNFC_5 | JNFC | 5.5 | 0.71 | 145 |
| JNFC_6 | JNFC | 4.7 | 0.55 | 83 |
| JNFC_7 | JNFC | 8.0 | 0.75 | 172 |
| JNFC_8 | JNFC | 7.1 | 0.74 | 188 |
| JNFC_9 | JNFC | 5.6 | 0.62 | 72 |
| JNFC_10 | JNFC | 5.8 | 0.48 | 77 |
| JNFC_11 | JNFC | 8.0 | 0.81 | 136 |
| JNFC_12 | JNFC | 5.1 | 0.60 | 129 |
| JNFC_13 | JNFC | 4.6 | 0.48 | 89 |
| JFC_1 | JFC | 5.4 | 0.60 | 84 |
| JFC_2 | JFC | 4.4 | 0.51 | 84 |
| JFC_3 | JFC | 5.1 | 0.86 | 81 |
| JFC_4 | JFC | 5.8 | 0.58 | 133 |
| JFC_5 | JFC | 5.2 | 0.98 | 84 |
| JFC_6 | JFC | 4.6 | 0.56 | 70 |
| JFC_7 | JFC | 4.9 | 0.62 | 106 |
| JFC_8 | JFC | 5.2 | 0.60 | 112 |
| JFC_9 | JFC | 5.4 | 0.69 | 132 |
| JFC_10 | JFC | 5.2 | 0.62 | 73 |
| Sample Pair | A | B | C | D | |||||
|---|---|---|---|---|---|---|---|---|---|
| JNFC_1 | JFC_1 | JNFC_2 | JFC_2 | JNFC_3 | JFC_3 | JNFC_6 | JFC_4 | ||
| aroma | fresh | 14 a | 4 | 17 c | 1 | 12 | 6 | 15 b | 3 |
| cooked | 4 | 14 a | 1 | 17 c | 6 | 12 | 3 | 15 b | |
| flavor | fresh | 12 | 6 | 13 b | 5 | 15 b | 3 | 17 c | 1 |
| cooked | 6 | 12 | 5 | 13 b | 3 | 15 b | 1 | 17 c | |
| Peak No. | RI | Identified Compounds | VIPs for OPLS-DA | Fold Change (JNFC/JFC) | p-Value |
|---|---|---|---|---|---|
| 1 | 715 | acetaldehyde | 1.059 | 0.581 | 0.021 |
| 2 | 752 | dimethyl sulfide | 1.151 | 0.586 | 0.003 |
| 3 | 803 | furan | 0.887 | 0.734 | 0.049 |
| 4 | 817 | acetone | 0.168 | 1.134 | 0.620 |
| 5 | 870 | 3-methylfuran | 0.618 | 1.479 | 0.112 |
| 6 | 893 | ethyl acetate | 1.043 | 2.941 | 0.009 |
| 7 | 901 | 2-metnylfuran | 1.112 | 3.953 | 0.002 |
| 8 | 914 | 2-methylbutanal | 1.326 | 0.400 | 0.001 |
| 9 | 917 | 3-methylbutanal | 1.235 | 0.292 | 0.001 |
| 10 | 940 | ethanol | 1.510 | 9.231 | 0.000 |
| 11 | 952 | 2-ethylfuran | 1.263 | 3.739 | 0.000 |
| 12 | 979 | pentanal | 0.126 | 1.371 | 0.844 |
| 13 | 1036 | 2-butanol | 1.279 | 4.574 | 0.001 |
| 14 | 1038 | toluene | 0.116 | 1.288 | 0.507 |
| 15 | 1050 | 2-methyl-3-buten-2-ol | 1.132 | 2.340 | 0.002 |
| 16 | 1055 | ethyl 2-methylbutyrate | 0.164 | 0.456 | 0.842 |
| 17 | 1071 | dimethyl disulfide | 0.451 | 1.058 | 0.563 |
| 18 | 1085 | hexanal | 1.073 | 2.636 | 0.010 |
| 19 | 1094 | 2-methyl-2-butenal | 1.443 | 6.958 | 0.000 |
| 20 | 1113 | 2-methyl-1-propanol | 1.219 | 2.970 | 0.001 |
| 21 | 1126 | isovaleronitrile | 0.092 | 1.108 | 0.994 |
| 22 | 1131 | p-xylene | 0.442 | 0.952 | 0.402 |
| 23 | 1177 | 1-penten-3-ol | 1.404 | 6.968 | 0.000 |
| 24 | 1185 | limonene | 0.018 | 1.236 | 0.975 |
| 25 | 1202 | 3-methyl-2-butenal | 0.584 | 1.775 | 0.106 |
| 26 | 1218 | (E)-2-hexenal | 1.144 | 4.182 | 0.002 |
| 27 | 1222 | 2-methylbutanol | 1.657 | 10.857 | 0.000 |
| 28 | 1223 | 3-methylbutanol | 1.580 | 8.525 | 0.000 |
| 29 | 1227 | 2-pentylfuran | 0.635 | 1.780 | 0.110 |
| 30 | 1230 | (E)-β -ocimene | 0.970 | 2.459 | 0.008 |
| 31 | 1247 | 2, 4-dimethylphenol | 1.025 | 2.191 | 0.010 |
| 32 | 1257 | 2-methyl-1-penten-3-ol | 0.529 | 1.818 | 0.127 |
| 33 | 1261 | methyl propyl sulfide | 0.835 | 0.681 | 0.050 |
| 34 | 1265 | pentanol | 1.519 | 8.120 | 0.000 |
| 35 | 1274 | hexyl acetate | 0.435 | 2.946 | 0.391 |
| 36 | 1275 | 4-ethyltoluene * | 0.563 | 1.387 | 0.130 |
| 37 | 1292 | acetoin | 1.342 | 5.222 | 0.000 |
| 38 | 1301 | (E)-2-(2-pentenyl)furan * | 0.696 | 1.554 | 0.090 |
| 39 | 1313 | 2, 2, 6-trimethylcyclohexanone | 0.512 | 1.339 | 0.269 |
| 40 | 1316 | 3, 4-dimethylcyclohexanol * | 0.162 | 1.707 | 0.478 |
| 41 | 1320 | 6-methyl-6-hepten-2-one * | 0.702 | 1.805 | 0.098 |
| 42 | 1343 | 6-methyl-5-hepten-2-one | 0.466 | 1.489 | 0.274 |
| 43 | 1367 | hexanol | 1.422 | 7.285 | 0.000 |
| 44 | 1376 | (E)-3-hexen-1-ol | 1.458 | 5.771 | 0.000 |
| 45 | 1397 | (Z)-3-hexen-1-ol | 1.448 | 7.469 | 0.000 |
| 46 | 1410 | 2-isobutylthiazole | 1.049 | 2.436 | 0.012 |
| 47 | 1416 | 1-(1-methycyclopentyl)-ethanone * | 0.924 | 4.762 | 0.029 |
| 48 | 1421 | perillene * | 0.073 | 1.165 | 0.987 |
| 49 | 1427 | 1, 3-di-tert-butylbenzene | 1.006 | 0.485 | 0.007 |
| 50 | 1451 | linalool oxide | 0.169 | 1.132 | 0.565 |
| 51 | 1457 | acetic acid | 0.340 | 0.948 | 0.555 |
| 52 | 1457 | methional | 1.388 | 0.569 | 0.000 |
| 53 | 1462 | 1-octen-3-ol | 1.242 | 4.347 | 0.001 |
| 54 | 1467 | -furfural | 0.772 | 0.686 | 0.046 |
| 55 | 1476 | 6-methyl-5-hepten-2-ol | 1.266 | 2.096 | 0.002 |
| 56 | 1522 | benzaldehyde | 0.332 | 1.105 | 0.192 |
| 57 | 1538 | 2-(methylthio) ethanol | 1.303 | 3.587 | 0.000 |
| 58 | 1558 | linalool | 0.980 | 2.395 | 0.009 |
| 59 | 1571 | (Z,Z)-2,4-hexadiene * | 0.399 | 1.594 | 0.292 |
| 60 | 1597 | 6-methyl-3,5-heptadien-2-one | 1.170 | 2.734 | 0.001 |
| 61 | 1610 | terpinen-4-ol | 1.131 | 9.180 | 0.003 |
| 62 | 1615 | 1-p-menthen-9-al * | 1.125 | 1.547 | 0.002 |
| 63 | 1629 | γ-butyrolactone | 0.599 | 1.140 | 0.080 |
| 64 | 1637 | butanoic acid | 0.582 | 2.443 | 0.220 |
| 65 | 1642 | phenylacetaldehyde | 1.305 | 0.397 | 0.001 |
| 66 | 1653 | acetophenone | 0.511 | 1.078 | 0.118 |
| 67 | 1679 | 3-methylbutanoic acid | 0.726 | 0.671 | 0.083 |
| 68 | 1685 | neral | 0.419 | 0.936 | 0.346 |
| 69 | 1707 | α-terpineol | 1.412 | 2.242 | 0.000 |
| 70 | 1728 | methionol | 0.936 | 2.027 | 0.012 |
| 71 | 1736 | geranial | 0.275 | 0.999 | 0.542 |
| 72 | 1744 | 1, 2-dihydro-1,1,6-trimethylnaphthalene * | 0.381 | 2.257 | 0.249 |
| 73 | 1776 | methyl salicylate | 1.080 | 5.854 | 0.001 |
| 74 | 1811 | 2, 3-dimethylbenzaldehyde | 0.329 | 0.533 | 0.267 |
| 75 | 1826 | (E)-β-damascenone | 0.469 | 1.302 | 0.257 |
| 76 | 1855 | hexanoic acid | 1.419 | 3.187 | 0.000 |
| 77 | 1861 | (E)-geranylacetone | 1.123 | 3.081 | 0.004 |
| 78 | 1866 | 2-methoxyphenol | 1.317 | 13.191 | 0.000 |
| 79 | 1885 | benzyl alcohol | 1.663 | 5.799 | 0.000 |
| 80 | 1920 | phenylethyl alcohol | 1.610 | 6.862 | 0.000 |
| 81 | 1929 | benzonitrile | 0.572 | 0.626 | 0.328 |
| 82 | 1944 | β-ionone | 1.373 | 3.552 | 0.000 |
| 83 | 1961 | 2-ethylhexanoic acid | 0.583 | 1.477 | 0.107 |
| 84 | 1998 | 2H-pyran-2,6(3H)-dione * | 0.976 | 1.437 | 0.042 |
| 85 | 2030 | formylpyrrole | 1.271 | 0.290 | 0.000 |
| 86 | 2078 | octanoic acid | 0.539 | 0.688 | 0.129 |
| 87 | 2098 | ψ-ionone | 0.470 | 0.970 | 0.414 |
| 88 | 2142 | ψ-ionone isomer * | 0.877 | 2.619 | 0.017 |
| 89 | 2173 | eugenol | 1.247 | 16.981 | 0.000 |
| 90 | 2184 | 4-ethylphenol | 1.162 | 9.502 | 0.001 |
| 91 | 2201 | 2-methoxy-4-vinylphenol | 1.315 | 3.724 | 0.000 |
| 92 | 2343 | dihydroactinidiolide * | 1.223 | 2.510 | 0.001 |
| 93 | 2400 | 2, 3-dihydrobenzofuran | 1.287 | 2.406 | 0.000 |
| 94 | 2435 | benzoic acid | 0.259 | 1.054 | 0.436 |
| Peak No. | RI | Identified Compounds | Odor Description | Odor Threshold | OAVs of Tomato Juices * | ||
|---|---|---|---|---|---|---|---|
| μg/L(kg) in Water | Ref. | JNFC | JFC | ||||
| 2 | 752 | dimethyl sulfide | seaweed, cooked corn | 0.84 | [31] | 318–1202 (802) | 760–2030 (1370) |
| 9 | 917 | 3-methylbutanal | cooked, malty | 0.5 | [31] | 119–543 (183) | 225–1354 (629) |
| 18 | 1085 | hexanal | green, grassy | 4.5 | [8] | 2.43–117 (37.5) | 2.26–96.3 (14.2) |
| 29 | 1227 | 2-pentylfuran | fatty, metallic | 6 | [29] | 0.0320–0.659 (0.213) | 0.0298–0.444 (0.187) |
| 42 | 1343 | 6-methyl-5-hepten-2-one | fruity | 50 | [8] | 0.856–7.93 (4.57) | 1.37–7.37 (3.07) |
| 43 | 1367 | hexanol | green, leafy | 200 | [8] | 0.004–1.94 (0.488) | 0.003–0.476 (0.0670) |
| 45 | 1397 | (Z)-3-hexen-1-ol | green, leafy | 3.9 | [31] | 0.506–166 (64.5) | 0.732–65.4 (8.64) |
| 46 | 1410 | 2-isobutylthiazole | tomato vine | 3.5 | [8] | 0.337–16.7 (7.09) | 0.312–17.6 (2.91) |
| 52 | 1457 | methional | boiled potato | 0.43 | [32] | 26.8–65.3 (47.1) | 54.4–125 (82.8) |
| 53 | 1462 | 1-octen-3-ol | mushroom-like | 14 | [8] | 0.002–0.061 (0.0250) | 0.002–0021 (0.006) |
| 58 | 1558 | linalool | sweet, flower-like | 6 | [8] | 0.254–3.69 (1.35) | 0.245–1.14 (0.563) |
| 64 | 1637 | butyric acid | cheese-like | 50 | [30] | 0.134–5.31 (0.893) | 0.134–1.36 (0.366) |
| 65 | 1642 | phenylacetaldehyde | floral | 5.2 | [32] | 0.415–2.71 (1.56) | 1.54–6.75 (3.92) |
| 67 | 1679 | 3-methylbutanoic acid | cheese-like | 490 | [32] | 0.0156–0.127 (0.047) | 0.0234–0.128 (0.070) |
| 71 | 1736 | geranial | citrus-like | 32 | [8] | 0.0184–0.419 (0.119) | 0.0483–0.194 (0.119) |
| 73 | 1776 | methyl salicylate | medicinal | 40 | [8] | 0.005–0.158 (0.049) | 0.00607–0.0108 (0.008) |
| 75 | 1826 | (E)-β-damascenone | sweet, floral | 0.013 | [32] | 303–1334 (706) | 338–1183 (542) |
| 78 | 1866 | 2-methoxyphenol | medicinal | 0.84 | [32] | 0.258–19.2 (5.88) | 0.032–0.912 (0.446) |
| 80 | 1920 | 2-phenylethanol | floral | 140 | [31] | 0.249–2.02 (0.921) | 0.055–0.385 (0.134) |
| 82 | 1944 | β-ionone | fruity | 0.007 | [8] | 43.4–935 (367) | 60.6–485 (140) |
| 89 | 2173 | eugenol | spicy, woody | 6 | [8] | 0.155–9.58 (2.50) | 0.121–0.176 (0.147) |
| 91 | 2201 | 2-methoxy-4-vinylphenol | spicy | 5.1 | [31] | 0.774–9.31 (2.72) | 0.474–1.21 (0.731) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iijima, Y.; Saisho, K.; Maeoka, T. Discrimination Between Commercial Tomato Juices from Non-Concentrate and Concentrate Based on Their Volatile Profiles. Foods 2025, 14, 2993. https://doi.org/10.3390/foods14172993
Iijima Y, Saisho K, Maeoka T. Discrimination Between Commercial Tomato Juices from Non-Concentrate and Concentrate Based on Their Volatile Profiles. Foods. 2025; 14(17):2993. https://doi.org/10.3390/foods14172993
Chicago/Turabian StyleIijima, Yoko, Katsutoshi Saisho, and Taiki Maeoka. 2025. "Discrimination Between Commercial Tomato Juices from Non-Concentrate and Concentrate Based on Their Volatile Profiles" Foods 14, no. 17: 2993. https://doi.org/10.3390/foods14172993
APA StyleIijima, Y., Saisho, K., & Maeoka, T. (2025). Discrimination Between Commercial Tomato Juices from Non-Concentrate and Concentrate Based on Their Volatile Profiles. Foods, 14(17), 2993. https://doi.org/10.3390/foods14172993






