Sustainable Precursor-Based Titanium Dioxide–Graphene Nanocomposite Electrochemical Sensor for Sensitive Detection of Diuron in Vegetables
Abstract
1. Introduction
2. Experimental Section
2.1. Reagents
2.2. Instruments
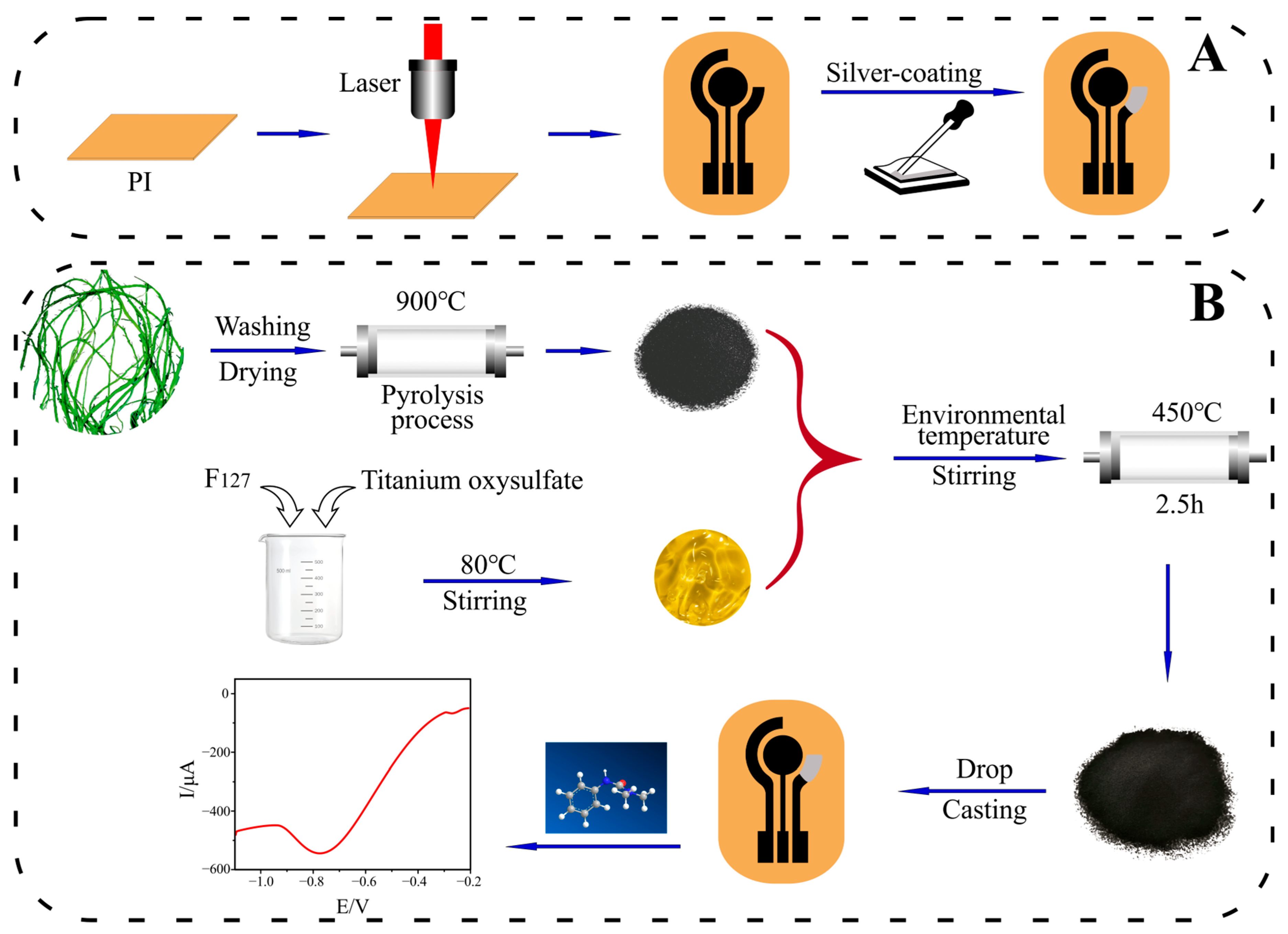
2.3. Design and Fabrication of LIGE
2.4. Preparation of TiO2@EDC/LIGE
2.5. Electrochemical Detection of Diuron
2.6. Real Sample Analysis
3. Results and Discussion
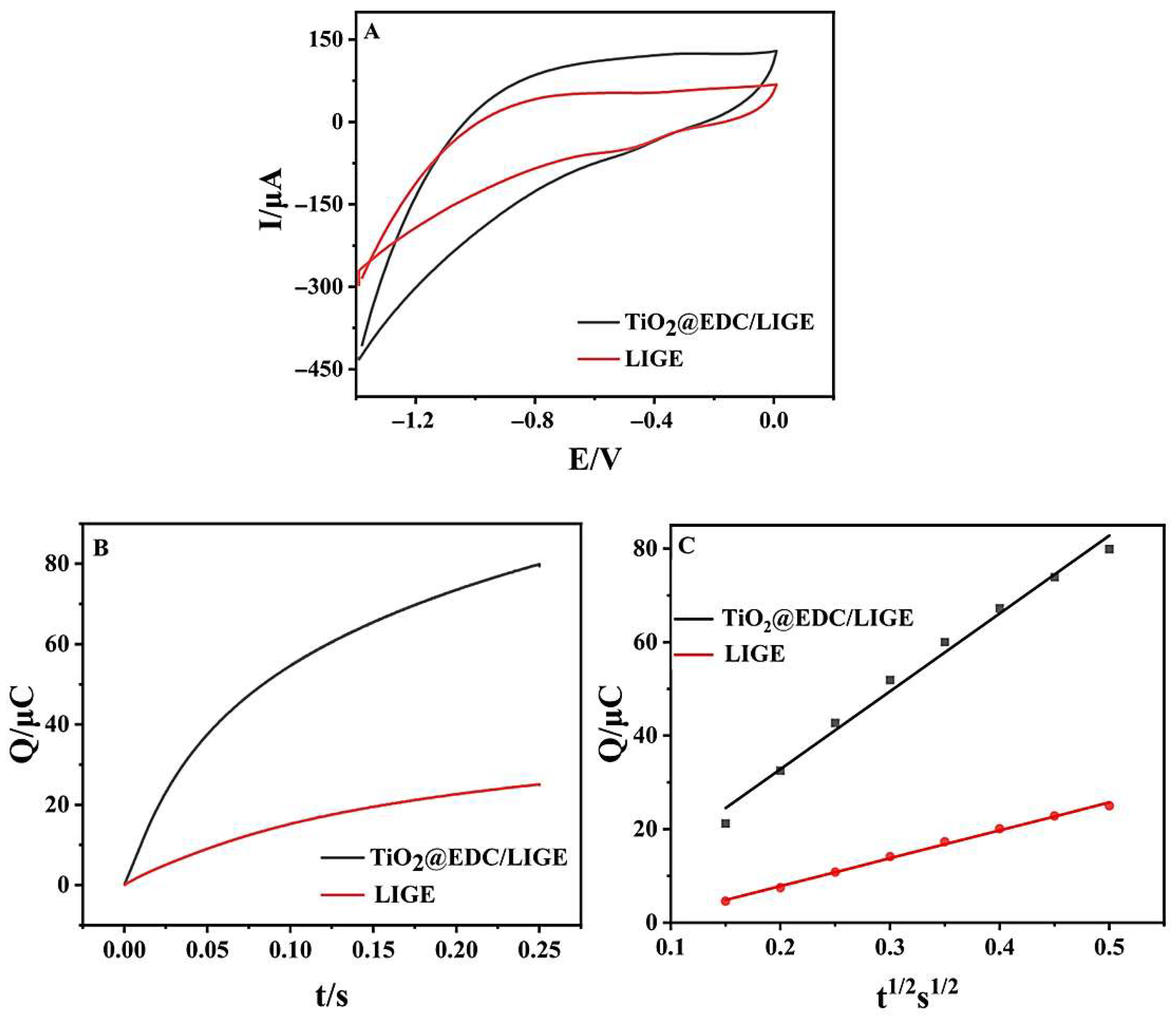
3.1. Characterization
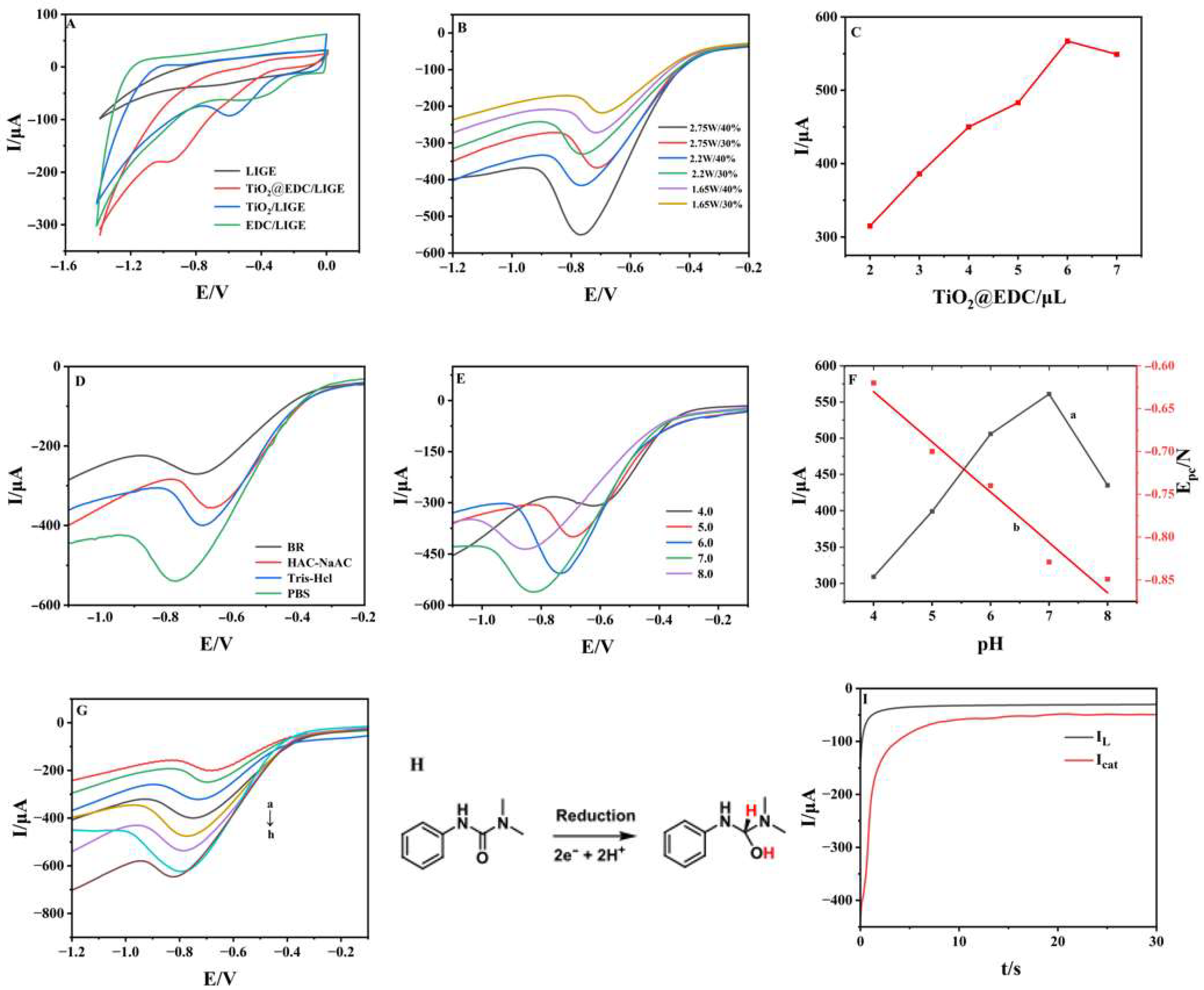
3.2. Electrochemical Behaviors of Different Electrodes
3.3. Electrochemical Performance and Kinetics for Diuron Detection
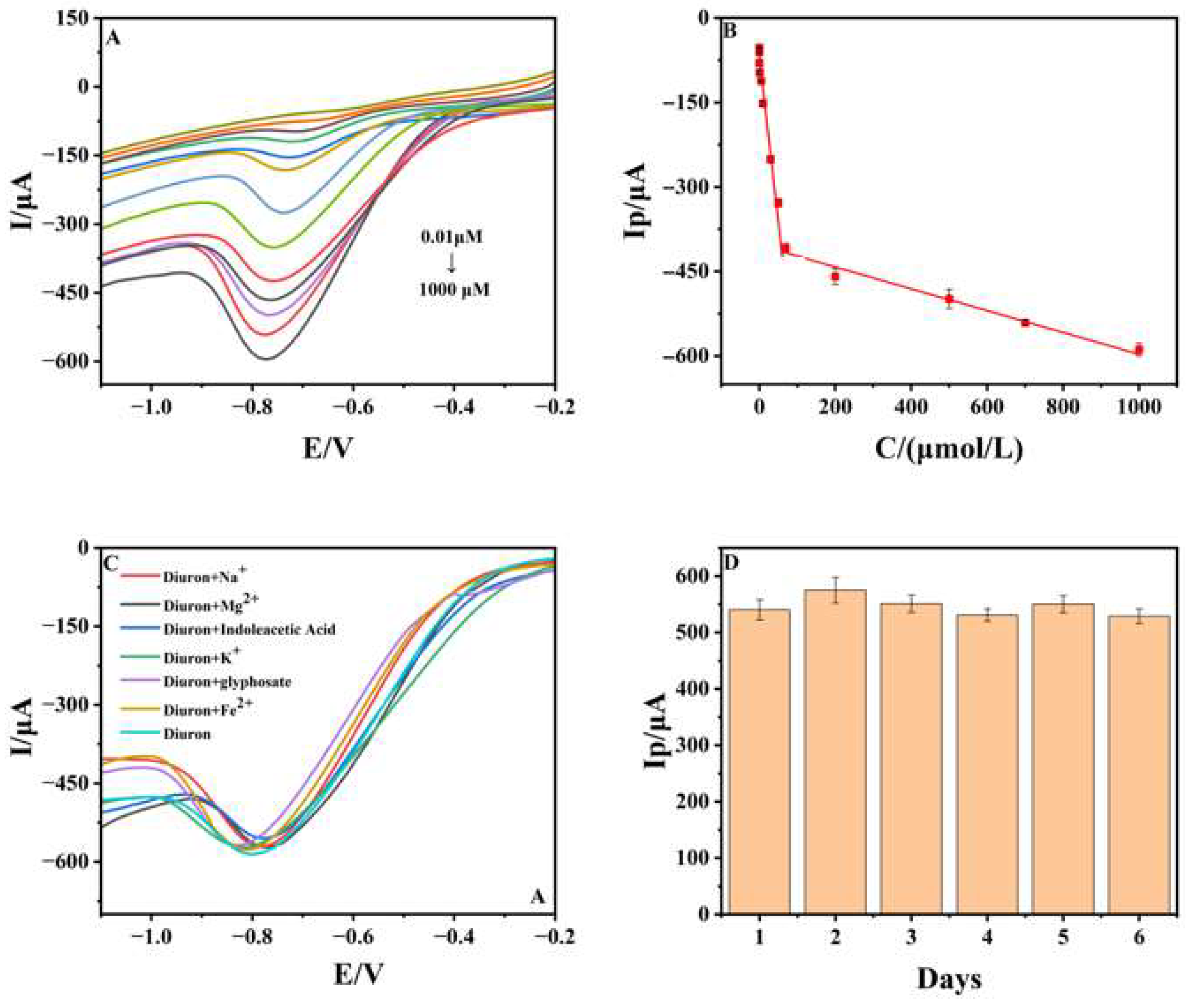
3.4. Analytical Performances
3.5. Real Sample Detection
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wong, A.; de Vasconcelos Lanza, M.R.; Sotomayor, M.D.P.T. Sensor for Diuron Quantitation Based on the P450 Biomimetic Catalyst Nickel(II) 1,4,8,11,15,18,22,25-Octabutoxy-29H,31H-Phthalocyanine. J. Electroanal. Chem. 2013, 690, 83–88. [Google Scholar] [CrossRef]
- Alves, G.F.; de Faria, L.V.; Lisboa, T.P.; Matos, M.A.C.; Muñoz, R.A.A.; Matos, R.C. Simple and Fast Batch Injection Analysis Method for Monitoring Diuron Herbicide Residues in Juice and Tap Water Samples Using Reduced Graphene Oxide Sensor. J. Food Compos. Anal. 2022, 106, 104284. [Google Scholar] [CrossRef]
- Giacomazzi, S.; Cochet, N. Environmental Impact of Diuron Transformation: A Review. Chemosphere 2004, 56, 1021–1032. [Google Scholar] [CrossRef] [PubMed]
- Annu; Raja, A.N.; Singh, K.; Halve, A.K.; Jain, R. Fabrication of Bismuth Oxide-Modified Pencil Graphite Sensors for Monitoring the Hazardous Herbicide Diuron. Nanoscale Adv. 2020, 2, 3404–3410. [Google Scholar] [CrossRef]
- Du, G.; Zhong, X.; Dupuy, C.; Che, S.; Lavaud, J. Diuron Effects on Photosynthesis and Vertical Migration of Microphytobenthos: Potential Rapid Bioassessment of Herbicide Toxicity in Coastal Sediments. Mar. Pollut. Bull. 2021, 170, 112619. [Google Scholar] [CrossRef] [PubMed]
- Skerratt, J.; Baird, M.E.; Mongin, M.; Ellis, R.; Smith, R.A.; Shaw, M.; Steven, A.D.L. Dispersal of the Pesticide Diuron in the Great Barrier Reef. Sci. Total Environ. 2023, 879, 163041. [Google Scholar] [CrossRef]
- Tlemsani, S.; Taleb, Z.; Pirault-Roy, L.; Taleb, S. Heterogeneous Catalytic Degradation of Diuron Using Algerian Sodium Montmorillonite. CLEAN—Soil Air Water 2022, 50, 2000468. [Google Scholar] [CrossRef]
- Medeiros, R.d.C.A.d.; Silva, T.S.; Teófilo, T.M.d.S.; da Silva, F.D.; Souza, M.d.F.; Passos, A.B.R.d.J.; Fernandes, B.C.C.; Lins, H.A.; das Chagas, P.S.F.; Souza, C.M.M.; et al. Herbicide Leaching in Soil with Different Properties: Perspectives from Commercial Formulations and Analytical Standards. Toxics 2024, 12, 199. [Google Scholar] [CrossRef]
- Araujo, G.R.; da Silva, L.B.X.; Vaz, V.; Borges, M.P.d.S.; Spolidorio, E.S.; Mendes, K.F. Cucumber Bioassay and HPLC Analysis to Detect Diuron Residues in Remineralized Soils Following Canavalia Ensiformis Cultivation as a Phytoremediator. Horticulturae 2023, 9, 1251. [Google Scholar] [CrossRef]
- Chen, B.; Wang, X. Combined Approach for Determining Diuron in Sugarcane and Soil: Ultrasound-Assisted Extraction, Carbon Nanotube-Mediated Purification, and Gas Chromatography–Electron Capture Detection. J. Food Sci. 2019, 84, 2402–2411. [Google Scholar] [CrossRef]
- Deveci, B.; Golge, O.; Kabak, B. Quantification of 363 Pesticides in Leafy Vegetables (Dill, Rocket and Parsley) in the Turkey Market by Using QuEChERS with LC-MS/MS and GC-MS/MS. Foods 2023, 12, 1034. [Google Scholar] [CrossRef] [PubMed]
- Soares, F.S.; Ramalho, R.T.E.; Silva, A.C.; Araujo, M.C.U. A Voltammetric Method for Diuron Determination in River Water. Electroanalysis 2023, 35, e202200486. [Google Scholar] [CrossRef]
- Liu, Y.; Xue, Q.; Chang, C.; Wang, R.; Liu, Z.; He, L. Recent Progress Regarding Electrochemical Sensors for the Detection of Typical Pollutants in Water Environments. Anal. Sci. 2022, 38, 55–70. [Google Scholar] [CrossRef] [PubMed]
- Kadu, R.D.; Keri, R.S.; H, D.N.; Budagumpi, S. State-of-the-Art Electrochemical Sensors for Quantitative Detection of Pesticides. Appl. Organomet. Chem. 2023, 37, e7097. [Google Scholar] [CrossRef]
- Madagalam, M.; Bartoli, M.; Tagliaferro, A. A Short Overview on Graphene and Graphene-Related Materials for Electrochemical Gas Sensing. Materials 2024, 17, 303. [Google Scholar] [CrossRef]
- Ren, H. Graphene and Its Derivatives for Electrochemical Sensing. Sensors 2025, 25, 1993. [Google Scholar] [CrossRef]
- Bressi, A.C.; Dallinger, A.; Steksova, Y.; Greco, F. Bioderived Laser-Induced Graphene for Sensors and Supercapacitors. ACS Appl. Mater. Interfaces 2023, 15, 35788–35814. [Google Scholar] [CrossRef]
- Movaghgharnezhad, S.; Kang, P. Laser-Induced Graphene: Synthesis Advances, Structural Tailoring, Enhanced Properties, and Sensing Applications. J. Mater. Chem. C 2024, 12, 6718–6742. [Google Scholar] [CrossRef]
- Aftab, S.; Koyyada, G.; Mukhtar, M.; Kabir, F.; Nazir, G.; Memon, S.A.; Aslam, M.; Assiri, M.A.; Kim, J.H. Laser-Induced Graphene for Advanced Sensing: Comprehensive Review of Applications. ACS Sens. 2024, 9, 4536–4554. [Google Scholar] [CrossRef]
- Ye, R.; James, D.K.; Tour, J.M. Laser-Induced Graphene. Acc. Chem. Res. 2018, 51, 1609–1620. [Google Scholar] [CrossRef]
- Han, X.; Ai, Y.; Wang, L.; Liu, T.; Badshah, A.; Hu, X.; Huang, Z.; Mansoor, A.; Sun, W. Flexible Electrochemical Sensing: Compact and Efficient Detection of Bisphenol A Using Copper Nanoparticle Decorated Laser-Induced Graphene-Based Electrode. Electroanalysis 2025, 37, e12025. [Google Scholar] [CrossRef]
- Wanjari, V.P.; Duttagupta, S.P.; Singh, S.P. Dual Linear Range Laser-Induced Graphene-Based Sensor for 4-Nitrophenol Detection in Water. ACS Appl. Nano Mater. 2023, 6, 11351–11360. [Google Scholar] [CrossRef]
- Ai, Y.; Ara, B.; Wang, B.; Ahmad, M.; Han, X.; Wang, L.; He, S.; Li, R.; Sun, W. One Step Laser-Induced Fe3O4 Loaded 3D Graphene Electrode for Electrochemical Sensing and Supercapacitor Applications. Microchem. J. 2025, 209, 112776. [Google Scholar] [CrossRef]
- Ferreira, B.; Crapnell, R.D.; Bernalte, E.; Paixão, T.R.L.C.; Banks, C.E. Low-Cost Conductive Polypropylene for Electroanalysis in Organic Solvents Using Additively Manufactured Electrodes. Electrochim. Acta 2025, 515, 145680. [Google Scholar] [CrossRef]
- Zhang, Z.; Cai, X.; Lv, Y.; Tang, X.; Shi, N.; Zhou, J.; Yan, M.; Li, Y. Self-Healing, Ultra-Stretchable, and Highly Sensitive Conductive Hydrogel Reinforced by Sulfate Polysaccharide from Enteromorpha Prolifera for Human Motion Sensing. Int. J. Biol. Macromol. 2023, 253, 126847. [Google Scholar] [CrossRef]
- Tembo, P.M.; Dhabarde, N.; Subramanian, V. TiO2 Nanotube-Based Sensor for the Detection of Cyanide in Water. J. Electrochem. Soc. 2021, 168, 057527. [Google Scholar] [CrossRef]
- Killedar, L.; Ilager, D.; Malode, S.J.; Shetti, N.P. Fast and Facile Electrochemical Detection and Determination of Fungicide Carbendazim at Titanium Dioxide Designed Carbon-Based Sensor. Mater. Chem. Phys. 2022, 285, 126131. [Google Scholar] [CrossRef]
- Pai, A.M.; Shanbhag, M.M.; Maiyalagan, T.; Alqarni, S.A.; Shetti, N.P. Activated Carbon Synthesized from Arecanut catechu L. as a Sustainable Precursor Intercalated TiO2 Modified Electrode for the Detection of Fungicide Dichlorophen. Diam. Relat. Mater. 2023, 140, 110561. [Google Scholar] [CrossRef]
- Wu, Y.; Pei, F.; Feng, S.; Wang, Z.; Lv, X.; Chen, S.-M.; Hao, Q.; Lei, W. Potentiostatic Oxidation of N-Doped Algae-Derived Carbon for P-Nitrophenol Sensitive Determination. J. Electroanal. Chem. 2020, 876, 114736. [Google Scholar] [CrossRef]
- Tang, H.; Zhong, Y.; Zeng, X.; Sang, Y.; Lin, F.; Zhu, Y.; Chen, Z.; Xu, L.; Huang, Z.; Zhou, P. Preparation of Platinum Decorated Laser-Induced Graphene Flexible Electrode and Its Application for Clenbuterol Detection. Int. J. Electrochem. Sci. 2022, 17, 220241. [Google Scholar] [CrossRef]
- Tang, X.; Yu, A.; Yang, Q.; Yuan, H.; Wang, Z.; Xie, J.; Zhou, L.; Guo, Y.; Ma, D.; Dai, S. Significance of Epitaxial Growth of PtO2 on Rutile TiO2 for Pt/TiO2 Catalysts. J. Am. Chem. Soc. 2024, 146, 3764–3772. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, W.; Zhang, K.; Liu, C.; Yu, A.; Peng, M.; Zhai, J. Raman Study of 2D Anatase TiO2 Nanosheets. Phys. Chem. Chem. Phys. 2016, 18, 32178–32184. [Google Scholar] [CrossRef] [PubMed]
- Anson, F.C. Application of Potentiostatic Current Integration to the Study of the Adsorption of Cobalt(III)-(Ethylenedinitrilo(Tetraacetate) on Mercury Electrodes. Anal. Chem. 1964, 36, 932–934. [Google Scholar] [CrossRef]
- Johnson, S.L.; Rumon, K.A. Ring-Opening Reaction of 1-(N,N-Dimethylcarbamoyl)Pyridinium Chloride with Hydroxide. A Model for the Alkaline Diphosphopyridine Nucleotide Reaction. Biochemistry 1970, 9, 847–857. [Google Scholar] [CrossRef] [PubMed]
- Shams, N.; Lim, H.N.; Hajian, R.; Yusof, N.A.; Abdullah, J.; Sulaiman, Y.; Ibrahim, I.; Huang, N.M.; Pandikumar, A. A Promising Electrochemical Sensor Based on Au Nanoparticles Decorated Reduced Graphene Oxide for Selective Detection of Herbicide Diuron in Natural Waters. J. Appl. Electrochem. 2016, 46, 655–666. [Google Scholar] [CrossRef]
- Laviron, E. General Expression of the Linear Potential Sweep Voltammogram in the Case of Diffusionless Electrochemical Systems. J. Electroanal. Chem. Interfacial Electrochem. 1979, 101, 19–28. [Google Scholar] [CrossRef]
- Polcaro, A.M.; Palmas, S.; Dernini, S. Electrochemical Reduction of Carbonyl Compounds at Modified Carbon Felt Electrodes. Electrochim. Acta 1993, 38, 199–203. [Google Scholar] [CrossRef]
- Galus, Z. Fundamentals of Electrochemical Analysis; Ellis Horwood series in analytical chemistry; Ellis Horwood: Chichester, UK, 1976; ISBN 978-0-470-15080-1. [Google Scholar]
- Morita, I.M.; Araújo, G.M.; Codognoto, L.; Simões, F.R. Functionalised Multi-Walled Carbon Nanotubes-Modified Electrode for Sensitive Determination of Diuron in Seawater Samples. Int. J. Environ. Anal. Chem. 2019, 99, 1565–1574. [Google Scholar] [CrossRef]
- Wong, A.; Foguel, M.V.; Khan, S.; de Oliveira, F.M.; Tarley, C.R.T.; Sotomayor, M.D.P.T. Development of an Electrochemical Sensor Modified with MWCNT-COOH and MIP for Detection of Diuron. Electrochim. Acta 2015, 182, 122–130. [Google Scholar] [CrossRef]
- Mbokou Foukmeniok, S.; Bako, R.F.Y.; Ilboudo, O.; Karanga, Y.; Njanja, E.; Pontie, M.; Tapsoba, I.; Tonle Kenfack, I.; Djouaka, R. Sensitive Carbon Fiber Microelectrode for the Quantification of Diuron in Quality Control of a Commercialized Formulation. Int. J. Anal. Chem. 2022, 2022, 9994639. [Google Scholar] [CrossRef]
- Wong, A.; Sotomayor, M.D.P.T. Determination of Carbofuran and Diuron in FIA System Using Electrochemical Sensor Modified with Organometallic Complexes and Graphene Oxide. J. Electroanal. Chem. 2014, 731, 163–171. [Google Scholar] [CrossRef]
- Manasa, G.; Bhakta, A.K.; Mascarenhas, R.J.; Shetti, N.P. BaO-MWCNT Composite Material-Based Electrocatalytic Amperometric Sensor for the Detection of Environmentally Hazardous Diuron. Microchem. J. 2023, 191, 108778. [Google Scholar] [CrossRef]
- Ouedraogo, B.; Tall, A.; Bako, Y.F.R.; Tapsoba, I. Sensitive Determination of Diuron on Zinc Oxide Nanoparticles Modified Carbon Paste Electrode in Soil and Water Samples. Electroanalysis 2023, 35, e202300101. [Google Scholar] [CrossRef]

| Modified Electrodes | Method | Sample | Linear Range (μmol/L) | LOD (μmol/L) | Ref. |
|---|---|---|---|---|---|
| rGO/AuNPs/ SPE | LSV | Lake water and sea water | 2.15–128.7 | 0.54 | [35] |
| MWCNT- COOH/GCE | DPV | Sea water | 0.215–2.15 | 0.068 | [39] |
| MWCNT- COOH-MIP/ CPE | SWV | River water | 0.05–1.25 | 0.009 | [40] |
| p-NiTSPc/ CFME | SWV | A commercialized formulation of diuron | 21.45–150 | 8.03 | [41] |
| NiOPc/hemin/GO/CPE | FIA/Amperometry | Soil and carrot | 5.00–140 | 1.72 | [42] |
| BaO-MWCNT/ MCPE | AMP i-t curve | Soil and water | 1–100 | 0.310 | [43] |
| ZnONPs/CPE | CV | Soil and water | 1.3–7.7, 8.6–30 | 0.223 | [44] |
| TiO2@EDC/ LIGE | LSV | Lettuce | 0.01–1000 | 2.99 × 10−3 | This work |
| Sample | Added (µM) | Found (µM) | RSD (%) | Recovery (%) |
|---|---|---|---|---|
| Lettuce | 0 | 0 | - | - |
| 6.50 | 6.61 | 4.96 | 101.69 | |
| 10.50 | 10.53 | 3.52 | 100.29 | |
| 14.50 | 14.23 | 1.11 | 98.14 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, L.; Li, X.; Ai, Y.; Mohan, B.; Li, H.; Lu, Z.; Wang, B.; Sun, W. Sustainable Precursor-Based Titanium Dioxide–Graphene Nanocomposite Electrochemical Sensor for Sensitive Detection of Diuron in Vegetables. Foods 2025, 14, 2946. https://doi.org/10.3390/foods14172946
Wang L, Li X, Ai Y, Mohan B, Li H, Lu Z, Wang B, Sun W. Sustainable Precursor-Based Titanium Dioxide–Graphene Nanocomposite Electrochemical Sensor for Sensitive Detection of Diuron in Vegetables. Foods. 2025; 14(17):2946. https://doi.org/10.3390/foods14172946
Chicago/Turabian StyleWang, Lisi, Xiaoqing Li, Yijing Ai, Brij Mohan, Hongji Li, Zhisong Lu, Baoli Wang, and Wei Sun. 2025. "Sustainable Precursor-Based Titanium Dioxide–Graphene Nanocomposite Electrochemical Sensor for Sensitive Detection of Diuron in Vegetables" Foods 14, no. 17: 2946. https://doi.org/10.3390/foods14172946
APA StyleWang, L., Li, X., Ai, Y., Mohan, B., Li, H., Lu, Z., Wang, B., & Sun, W. (2025). Sustainable Precursor-Based Titanium Dioxide–Graphene Nanocomposite Electrochemical Sensor for Sensitive Detection of Diuron in Vegetables. Foods, 14(17), 2946. https://doi.org/10.3390/foods14172946








