Characterization of Genomic, Physiological, and Probiotic Features of Lactiplantibacillus plantarum JS21 Strain Isolated from Traditional Fermented Jiangshui
Abstract
1. Introduction
2. Materials and Methods
2.1. Isolation of Bacterial Strain and Growth Conditions
2.2. DNA Extraction, Identification, Whole Genome Sequencing, and Assembly
2.3. Bioinformatic Analysis
2.4. Carbohydrate Fermentation
2.5. Determination of Antibiotic Susceptibility
2.6. Determination of Probiotic Properties
3. Results and Discussion
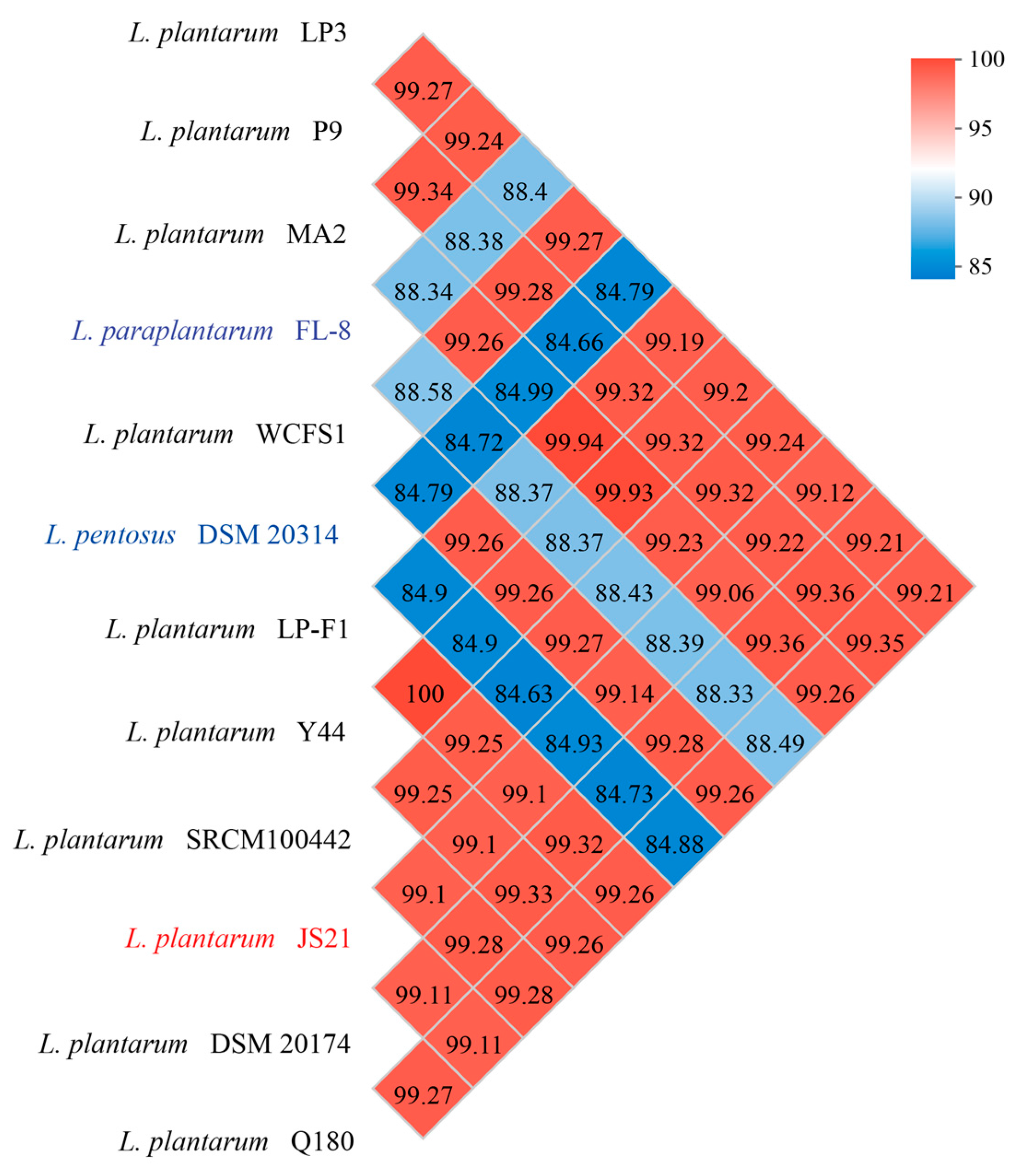
3.1. Functional Genomic Characterization
3.2. Prophages, Related Horizontal Gene Transfer, and Mobilome
3.3. Phenotypic Antibiotic Resistance and Safety-Related Gene Assessment
3.4. Carbohydrate Fermentation Patterns and Active Enzymes
3.5. Secondary Metabolite Biosynthetic Gene Clusters
3.6. Probiotic Properties
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Seddik, H.A.; Bendali, F.; Gancel, F.; Fliss, I.; Spano, G.; Drider, D. Lactobacillus plantarum and itsprobiotic and food potentialities. Probiotics Antimicrob. Proteins 2017, 9, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Ishii, M.; Nishida, S.; Kataoka, K.; Nishiyama, Y.; Abe, S.; Sekimizu, K. Lactic acid bacteria of the Leuconostoc genus with high innate immunity-stimulating activity. Drug Discov. Ther. 2017, 11, 25–29. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Jha, R.; Li, A.; Liu, H.; Zhang, Z.; Zhang, C.; Zhai, Q.; Zhang, J. Probiotics (Lactobacillus plantarum HNU082) supplementation relieves ulcerative colitis by affecting intestinal barrier functions, immunity-related gene expression, gut microbiota, and metabolic pathways in mice. Microbiol. Spectr. 2022, 10, e0165122. [Google Scholar] [CrossRef] [PubMed]
- Xing, J.H.; Shi, C.W.; Sun, M.J.; Gu, W.; Zhang, R.R.; Chen, H.L.; Li, Y.; Wang, D.; Li, J.; Niu, T.M.; et al. Lactiplantibacillus plantarum 0111 protects against influenza virus by modulating intestinal microbial-mediated immune responses. Front. Microbiol. 2022, 13, 820484. [Google Scholar] [CrossRef] [PubMed]
- Gizachew, S.; Van Beeck, W.; Spacova, I.; Dekeukeleire, M.; Alemu, A.; Woldemedhin, W.M.; Mariam, S.H.; Lebeer, S.; Engidawork, E. Antibacterial and immunostimulatory activity of potential probiotic lactic acid bacteria isolated from ethiopian fermented dairy products. Fermentation 2023, 9, 258. [Google Scholar] [CrossRef]
- Hudson, G.A.; Mitchell, D.A. RiPP antibiotics: Biosynthesis and engineering potential. Curr. Opin. Microbiol. 2018, 45, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Sanders, M.E.; Akkermans, L.M.; Haller, D.; Hammerman, C.; Heimbach, J.T.; Hörmannsperger, G.; Huys, G. Safety assessment of probiotics for human use. Gut Microbes 2010, 1, 164–185. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Kang, J.; Ma, Z.; Li, X.; Liu, L.; Hu, X. Microbial succession and metabolite changes during traditional serofluid dish fermentation. LWT 2017, 84, 771–779. [Google Scholar] [CrossRef]
- Chen, P.; Zhao, Y.; Wu, Z.; Liu, R.; Xu, R.; Yan, L.; Li, H. Metagenomic data of fungal internal transcribed spacer from serofluid dish, a traditional Chinese fermented food. Genom. Data 2016, 7, 134–136. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Wu, Z.; Zhao, Y.; Wei, Y.; Xu, R.; Yan, L.; Li, H. Cultivation-independent comprehensive investigations on bacterial communities in serofluid dish, a traditional Chinese fermented food. Genom. Data 2016, 7, 127–128. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Ju, N.; Zhang, S.; Wang, Y.; Luo, Y. Evaluation of microbial diversity of Jiangshui from the Ningxia Hui autonomous region in China. Food Biotechnol. 2022, 36, 173–190. [Google Scholar] [CrossRef]
- Liu, Z.; Li, J.; Wei, B.; Huang, T.; Xiao, Y.; Peng, Z.; Xie, M.; Xiong, T. Bacterial community and composition in Jiang-shui and Suan-cai revealed by high-throughput sequencing of 16S rRNA. Int. J. Food Microbiol. 2019, 306, 108271. [Google Scholar] [CrossRef] [PubMed]
- Elio, L.; Antonio, B.; Javier, R.; Victor, L.; Noé, A.F. In Silico evidence of the multifunctional features of Lactiplantibacillus pentosus LPG1, a natural fermenting agent isolated from table olive biofilms. Foods 2023, 12, 938. [Google Scholar] [CrossRef]
- Choi, A.R.; Patra, J.K.; Kim, W.J.; Kang, S.S. Antagonistic activities and probiotic potential of lactic acid bacteria derived from a Plant-Based fermented food. Front. Microbiol. 2018, 9, 1963. [Google Scholar] [CrossRef] [PubMed]
- Vera-Pingitore, E.; Jiménez, M.E.; Dallagnol, A.M.; Belfiore, C.; Fontana, C.A.; Fontana, P.D.; Vignolo, G.M.; Plumed-Ferrer, C. Screening and characterization of potential probiotic and starter bacteria for plant fermentations. LWT-Food Sci. Technol. 2016, 71, 288–294. [Google Scholar] [CrossRef]
- Thakur, N.; Rokana, N.; Panwar, H. Probiotics: Selection criteria, safety and role in health and disease. J. Innov. Biol. January 2016, 3, 259–270. [Google Scholar]
- Ou, D.; Ling, N.; Wang, X.; Zou, Y.; Dong, J.; Zhang, D.; Shen, Y.; Ye, Y. Safety assessment of one Lactiplantibacillus plantarum isolated from the traditional chinese fermented vegetables-Jiangshui. Foods 2022, 11, 2177. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Ye, Z.; Feng, P.; Li, R.; Chen, X.; Tian, X.; Han, R.; Kakade, A.; Liu, P.; Li, X. Limosilactobacillus fermentum JL-3 isolated from “Jiangshui” ameliorates hyperuricemia by degrading uric acid. Gut Microbes 2021, 13, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.H.; Pan, T.M. Characterization of an antimicrobial substance produced by Lactobacillus plantarum NTU 102. J. Microbiol. Immunol. Infect. 2019, 52, 409–417. [Google Scholar] [CrossRef]
- Jang, H.J.; Song, M.W.; Lee, N.K.; Paik, H.D. Antioxidant effects of live and heat-killed probiotic Lactobacillus plantarum Ln1 isolated from kimchi. J. Food Sci. Technol. 2018, 55, 3174–3180. [Google Scholar] [CrossRef]
- Zdziobek, P.; Jodłowski, G.S.; Strzelec, E.A. Biopreservation and bioactivation juice from waste broccoli with Lactiplantibacillus plantarum. Molecules 2023, 28, 4594. [Google Scholar] [CrossRef] [PubMed]
- Shi, R.; Yu, F.; Hu, X.; Liu, Y.; Jin, Y.; Ren, H.; Lu, S.; Guo, J.; Chang, J.; Li, Y.; et al. Protective effect of Lactiplantibacillus plantarum subsp. plantarum SC-5 on dextran sulfate sodium—induced colitis in mice. Foods 2023, 12, 897. [Google Scholar] [PubMed]
- Yu, X.; Wei, M.; Yang, D.; Wu, X.; Wei, H.; Xu, F. Lactiplantibacillus plantarum strain FLPL05 promotes longevity in mice by improving intestinal barrier. Probiotics Antimicrob. Proteins 2022, 15, 1193–1205. [Google Scholar] [CrossRef]
- Delcher, A.L.; Bratke, K.A.; Powers, E.C.; Salzberg, S.L. Identifying bacterial genes and endosymbiont DNA with Glimmer. Bioinformatics 2007, 23, 673–679. [Google Scholar] [CrossRef] [PubMed]
- Alikhan, N.F.; Petty, N.K.; Ben Zakour, N.L.; Beatson, S.A. BLAST Ring Image Generator (BRIG): Simple prokaryote genome comparisons. BMC Genom. 2011, 12, 402. [Google Scholar] [CrossRef] [PubMed]
- Arndt, D.; Grant, J.R.; Marcu, A.; Sajed, T.; Pon, A.; Liang, Y.; Wishart, D.S. PHASTER: A better, faster version of the PHAST phage search tool. Nucleic Acids Res. 2016, 44, W16–W21. [Google Scholar] [CrossRef] [PubMed]
- Yetiman, A.E.; Keskin, A.; Darendeli, B.N.; Kotil, S.E.; Ortakci, F.; Dogan, M. Characterization of genomic, physiological, and probiotic features Lactiplantibacillus plantarum DY46 strain isolated from traditional lactic acid fermented shalgam beverage. Food Biosci. 2022, 46, 101499. [Google Scholar] [CrossRef]
- Kanehisa, M.; Sato, Y.; Morishima, K. BlastKOALA and GhostKOALA: KEGG tools for functional characterization of genome and metagenome sequences. J. Mol. Biol. 2016, 428, 726–731. [Google Scholar] [CrossRef] [PubMed]
- Bortolaia, V.; Kaas, R.S.; Ruppe, E.; Roberts, M.C.; Schwarz, S.; Cattoir, V.; Philippon, A.; Allesoe, R.L.; Rebelo, A.R.; Florensa, A.F.; et al. ResFinder 4.0 for predictions of phenotypes from genotypes. J. Antimicrob. Chemother. 2020, 75, 3491–3500. [Google Scholar] [CrossRef] [PubMed]
- Alcock, B.P.; Raphenya, A.R.; Lau, T.T.Y.; Tsang, K.K.; Bouchard, M.; Edalatmand, A.; Huynh, W.; Nguyen, A.V.; Cheng, A.A.; Liu, S.; et al. CARD 2020: Antibiotic resistome surveillance with the comprehensive antibiotic resistance database. Nucleic Acids Res 2020, 48, D517–D525. [Google Scholar] [CrossRef] [PubMed]
- Blin, K.; Shaw, S.; Augustijn, H.E.; Reitz, Z.L.; Biermann, F.; Alanjary, M.; Fetter, A.; Terlouw, B.R.; Metcalf, W.W.; Helfrich, E.J.N.; et al. antiSMASH 7.0: New and improved predictions for detection, regulation, chemical structures and visualisation. Nucleic Acids Res. 2023, 51, W46–W50. [Google Scholar] [CrossRef] [PubMed]
- Khan, Z.A.; Siddiqui, M.F.; Park, S. Current and emerging methods of antibiotic susceptibility testing. Diagnostics 2019, 9, 49. [Google Scholar] [CrossRef] [PubMed]
- Patel, J.B.; Weinstein, M.P.; Eliopoulos, G.M.; Jenkins, S.G.; Lewis, J.S.; Limbago, B.M.; Mathers, A.J.; Mazzulli, T.; Patel, R.; Richter, S.S.; et al. Performance Standards for Antimicrobial Susceptibility Testing, 32nd ed.; CLSI: Wayne, PA, USA, 2022. [Google Scholar]
- Charteris, W.P.; Kelly, P.M.; Morelli, L.; Collins, J.K. Antibiotic susceptibility of potentially probiotic Lactobacillus species. J. Food Prot. 1998, 61, 1636–1643. [Google Scholar] [CrossRef] [PubMed]
- Angmo, K.; Kumari, A.; Savitri; Bhalla, T.C. Probiotic characterization of lactic acid bacteria isolated from fermented foods and beverage of Ladakh. LWT-Food Sci. Technol. 2016, 66, 428–435. [Google Scholar] [CrossRef]
- Chinese Pharmacopoeia Commission. Pharmacopoeia of the People’s Republic of China 2010 Edition (Part Two); Appendix S; China Medical Science Press: Beijing, China, 2010. [Google Scholar]
- Chinese Pharmacopoeia Commission. Pharmacopoeia of the People’s Republic of China (Part One); China Medical Science Press: Beijing, China, 2015. [Google Scholar]
- Liu, C.; Xue, W.J.; Ding, H.; An, C.; Ma, S.J.; Liu, Y. Probiotic Potential of lactobacillus strains isolated from fermented vegetables in Shaanxi, China. Front. Microbiol. 2021, 12, 774903. [Google Scholar] [CrossRef] [PubMed]
- Behbahani, A.B.; Noshad, M.; Falah, F. Inhibition of Escherichia coli adhesion to human intestinal Caco-2 cells by probiotic candidate Lactobacillus plantarum strain L15. Microb. Pathog. 2019, 136, 103677. [Google Scholar] [CrossRef] [PubMed]
- Juhas, M.; van der Meer, J.R.; Gaillard, M.; Harding, R.M.; Hood, D.W.; Crook, D.W. Genomic islands: Tools of bacterial horizontal gene transfer and evolution. FEMS Microbiol. Rev. 2009, 33, 376–393. [Google Scholar] [CrossRef] [PubMed]
- Yetiman, A.; Horzum, M.; Bahar, D.; Akbulut, M. Assessment of genomic and metabolic characteristics of cholesterol-reducing and GABA producer Limosilactobacillus fermentum AGA52 isolated from lactic acid fermented Shalgam based on “in silico” and “in vitro” approaches. Probiotics Antimicrob. Proteins 2023. [Google Scholar] [CrossRef]
- Ma, T.; Yang, N.; Xie, Y.; Li, Y.; Xiao, Q.; Li, Q.; Jin, H.; Zheng, L.; Sun, Z.; Zuo, K.; et al. Effect of the probiotic strain, Lactiplantibacillus plantarum P9, on chronic constipation: A randomized, double-blind, placebo-controlled study. Pharmacol. Res. 2023, 191, 106755. [Google Scholar] [CrossRef] [PubMed]
- Kleerebezem, M.; Boekhorst, J.; van Kranenburg, R.; Molenaar, D.; Kuipers, O.P.; Leer, R.; Tarchini, R.; Peters, S.A.; Sandbrink, H.M.; Fiers, M.W.; et al. Complete genome sequence of Lactobacillus plantarum WCFS1. Proc. Natl. Acad. Sci. USA 2003, 100, 1990–1995. [Google Scholar] [CrossRef] [PubMed]
- van den Nieuwboer, M.; van Hemert, S.; Claassen, E.; de Vos, W.M. Lactobacillus plantarum WCFS1 and its host interaction: A dozen years after the genome. Microb. Biotechnol. 2016, 9, 452–465. [Google Scholar] [CrossRef] [PubMed]
- Starzyńska-Janiszewska, A.; Stodolak, B.; Mickowska, B. Effect of controlled lactic acid fermentation on selected bioactive and nutritional parameters of tempeh obtained from unhulled common bean (Phaseolus vulgaris) seeds. J. Sci. Food Agric. 2014, 94, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Wang, M.; Zheng, Y.; Miao, K.; Qu, X. The carbohydrate metabolism of Lactiplantibacillus plantarum. Int. J. Mol. Sci. 2021, 22, 13452. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Liu, Y.; Sun, M.; Zhang, H.; Mu, G.; Tuo, Y. Physiological function analysis of Lactobacillus plantarum Y44 based on genotypic and phenotypic characteristics. J. Dairy. Sci. 2020, 103, 5916–5930. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.J.; Chun, B.H.; Jung, H.S.; Chu, J.; Joung, H.; Park, S.Y.; Kim, B.K.; Jeon, C.O. Safety assessment of Lactiplantibacillus (formerly Lactobacillus) plantarum Q180. J. Microbiol. Biotechnol. 2021, 31, 1420–1429. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Cho, S.A.; Kim, S.H.; Lim, S.D. Physiological characteristics and anti-obesity effect of Lactobacillus plantarum Q180 isolated from feces. Korean J. Food Sci. Anim. Resour. 2014, 34, 647–655. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.; Li, C.; He, Z.; Pan, F.; Pan, S.; Wang, Y. Probiotic properties and cellular antioxidant activity of Lactobacillus plantarum MA2 isolated from tibetan kefir grains. Probiotics Antimicrob. Proteins 2018, 10, 523–533. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.; Ouk Kim, Y.; Park, S.C.; Chun, J. OrthoANI: An improved algorithm and software for calculating average nucleotide identity. Int. J. Syst. Evol. Microbiol. 2016, 66, 1100–1103. [Google Scholar] [CrossRef] [PubMed]
- Page, C.A.; Pérez-Díaz, I.M.; Pan, M.; Barrangou, R. Genome-wide comparative analysis of Lactiplantibacillus pentosus isolates autochthonous to cucumber fermentation reveals subclades of divergent ancestry. Foods 2023, 12, 2455. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Bowers, J.E.; Wang, X.; Ming, R.; Alam, M.; Paterson, A.H. Synteny and collinearity in plant genomes. Science 2008, 320, 486–488. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chen, L.L.; Xing, F.; Kudrna, D.A.; Yao, W.; Copetti, D.; Mu, T.; Li, W.; Song, J.M.; Xie, W.; et al. Extensive sequence divergence between the reference genomes of two elite indica rice varieties Zhenshan 97 and Minghui 63. Proc. Natl. Acad. Sci. USA 2016, 113, E5163–E5171. [Google Scholar] [CrossRef] [PubMed]
- Kandasamy, S.; Yoo, J.; Yun, J.; Lee, K.H.; Kang, H.B.; Kim, J.E.; Oh, M.H.; Ham, J.S. Probiogenomic in-silico analysis and safety assessment of Lactiplantibacillus plantarum DJF10 strain isolated from korean raw milk. Int. J. Mol. Sci. 2022, 23, 14494. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.J.; Wang, R.; Gong, F.M.; Liu, X.F.; Zheng, H.J.; Luo, Y.Y.; Li, X.R. Complete genome sequences and comparative genome analysis of Lactobacillus plantarum strain 5-2 isolated from fermented soybean. Genomics 2015, 106, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Hacker, J.; Kaper, J.B. Pathogenicity islands and the evolution of microbes. Annu. Rev. Microbiol. 2000, 54, 641–679. [Google Scholar] [CrossRef] [PubMed]
- Cahill, J.; Young, R. Phage lysis: Multiple genes for multiple barriers. Adv. Virus Res. 2019, 103, 33–70. [Google Scholar] [CrossRef] [PubMed]
- Pöntinen, A.; Aalto-Araneda, M.; Lindström, M.; Korkeala, H. Heat resistance mediated by pLM58 plasmid-borne ClpL in Listeria monocytogenes. mSphere 2017, 2, e00364-17. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.S.; Baek, K.H. Protective roles of cytosolic and plastidal proteasomes on abiotic stress and pathogen invasion. Plants 2020, 9, 832. [Google Scholar] [CrossRef] [PubMed]
- Tong, D. Antibiotic Resistance in Lactic Acid Bacteria. J. Probiotics Health 2021, 9, 1–4. [Google Scholar]
- Campedelli, I.; Mathur, H.; Salvetti, E.; Clarke, S.; Rea, M.C.; Torriani, S.; Ross, R.P.; Hill, C.; O’Toole, P.W. Genus-wide assessment of antibiotic resistance in Lactobacillus spp. Appl. Environ. Microbiol. 2019, 85, e01738-18. [Google Scholar] [CrossRef] [PubMed]
- Deghorain, M.; Goffin, P.; Fontaine, L.; Mainardi, J.L.; Daniel, R.; Errington, J.; Hallet, B.; Hols, P. Selectivity for D-lactate incorporation into the peptidoglycan precursors of Lactobacillus plantarum: Role of aad, a VanX-like D-alanyl-D-alanine dipeptidase. J. Bacteriol. 2007, 189, 4332–4337. [Google Scholar] [CrossRef] [PubMed]
- Gueimonde, M.; Sánchez, B.; de Los Reyes-Gavilán, C.G.; Margolles, A. Antibiotic resistance in probiotic bacteria. Front. Microbiol. 2013, 4, 202. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.Y.; Liu, C.; Zhu, Y.Z.; Wei, Y.X.; Tian, F.; Zhao, G.P.; Guo, X.K. Safety assessment of Lactobacillus plantarum JDM1 based on the complete genome. Int. J. Food Microbiol. 2012, 153, 166–170. [Google Scholar] [CrossRef] [PubMed]
- Adekunle, O. Effect of Probiotics in the Mitigation of Clostridium Difficile Associated Disease. Master’s Thesis, Georgia State University, Atlanta, GA, USA, 2015. [Google Scholar]
- Egervärn, M.; Roos, S.; Lindmark, H. Identification and characterization of antibiotic resistance genes in Lactobacillus reuteri and Lactobacillus plantarum. J. Appl. Microbiol. 2009, 107, 1658–1668. [Google Scholar] [CrossRef] [PubMed]
- Teuber, M.; Meile, L.; Schwarz, F. Acquired antibiotic resistance in lactic acid bacteria from food. Antonie van Leeuwenhoek 1999, 76, 115–137. [Google Scholar] [CrossRef] [PubMed]
- Oberg, C.; Sorensen, K.; Oberg, T.; Young, S.; Domek, M.; Culumber, M.; McMahon, D. Gluconate metabolism and gas production by Paucilactobacillus wasatchensis WDC04. J. Dairy. Sci. 2021, 104, 10586–10593. [Google Scholar] [CrossRef] [PubMed]
- Marco, M.L.; Peters, T.H.; Bongers, R.S.; Molenaar, D.; van Hemert, S.; Sonnenburg, J.L.; Gordon, J.I.; Kleerebezem, M. Lifestyle of Lactobacillus plantarum in the mouse caecum. Environ. Microbiol. 2009, 11, 2747–2757. [Google Scholar] [CrossRef] [PubMed]
- Gänzle, M.G.; Follador, R. Metabolism of oligosaccharides and starch in lactobacilli: A review. Front. Microbiol. 2012, 3, 340. [Google Scholar] [CrossRef] [PubMed]
- Krispin, O.; Allmansberger, R. The Bacillus subtilis AraE protein displays a broad substrate specificity for several different sugars. J. Bacteriol. 1998, 180, 3250–3252. [Google Scholar] [CrossRef] [PubMed]
- Eiteman, M.A.; Ramalingam, S. Microbial production of lactic acid. Biotechnol. Lett. 2015, 37, 955–972. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Ruan, L.; Sun, M.; Gänzle, M. A genomic view of Lactobacilli and Pediococci demonstrates that phylogeny matches ecology and physiology. Appl. Environ. Microbiol. 2015, 81, 7233–7243. [Google Scholar] [CrossRef] [PubMed]
- Buron-Moles, G.; Chailyan, A.; Dolejs, I.; Forster, J.; Mikš, M.H. Uncovering carbohydrate metabolism through a genotype-phenotype association study of 56 lactic acid bacteria genomes. Appl. Microbiol. Biotechnol. 2019, 103, 3135–3152. [Google Scholar] [CrossRef] [PubMed]
- Thomas, L.M.; Harper, A.R.; Miner, W.A.; Ajufo, H.O.; Branscum, K.M.; Kao, L.; Sims, P.A. Structure of Escherichia coli AdhP (ethanol-inducible dehydrogenase) with bound NAD. Acta Crystallogr. Sect. F Struct. Biol. Cryst. Commun. 2013, 69, 730–732. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Hu, J.; Chen, W. Analysis of the relationship between microorganisms and flavour development in dry-cured grass carp by high-throughput sequencing, volatile flavour analysis and metabolomics. Food Chem. 2022, 368, 130889. [Google Scholar] [CrossRef] [PubMed]
- Abegaz, B.M.; Kinfe, H.H. Secondary metabolites, their structural diversity, bioactivity, and ecological functions: An overview. Phys. Sci. Rev. 2019, 4, 20180100. [Google Scholar] [CrossRef]
- Choi, S.; Baek, M.G.; Chung, M.J.; Lim, S.; Yi, H. Distribution of bacteriocin genes in the lineages of Lactiplantibacillus plantarum. Sci. Rep. 2021, 11, 20063. [Google Scholar] [CrossRef] [PubMed]
- Moll, G.N.; van den Akker, E.; Hauge, H.H.; Nissen-Meyer, J.; Nes, I.F.; Konings, W.N.; Driessen, A.J. Complementary and overlapping selectivity of the two-peptide bacteriocins plantaricin EF and JK. J. Bacteriol. 1999, 181, 4848–4852. [Google Scholar] [CrossRef] [PubMed]
- Britton, A.P.; van der Ende, S.R.; van Belkum, M.J.; Martin-Visscher, L.A. The membrane topology of immunity proteins for the two-peptide bacteriocins carnobacteriocin XY, lactococcin G, and lactococcin MN shows structural diversity. Microbiologyopen 2020, 9, e00957. [Google Scholar] [CrossRef] [PubMed]
- Fimland, N.; Rogne, P.; Fimland, G.; Nissen-Meyer, J.; Kristiansen, P.E. Three-dimensional structure of the two peptides that constitute the two-peptide bacteriocin plantaricin EF. Biochim. Biophys. Acta 2008, 1784, 1711–1719. [Google Scholar] [CrossRef] [PubMed]
- Ekblad, B.; Kyriakou, P.K.; Oppegård, C.; Nissen-Meyer, J.; Kaznessis, Y.N.; Kristiansen, P.E. Structure-function analysis of the two-peptide bacteriocin plantaricin EF. Biochemistry 2016, 55, 5106–5116. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, J.C.; Buchanan, J.H. The Basis of Protein Chemistry. In Chemistry of Natural Protein Fibers; Springer: Boston, MA, USA, 1977. [Google Scholar]
- Lim, Y.P.; Go, M.K.; Yew, W.S. Exploiting the biosynthetic potential of type III polyketide synthases. Molecules 2016, 21, 806. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Sun, F.; Bartlam, M.; Li, X.; Li, R.; Rao, Z. The crystal structure of human isopentenyl diphosphate isomerase at 1.7 A resolution reveals its catalytic mechanism in isoprenoid biosynthesis. J. Mol. Biol. 2007, 366, 1447–1458. [Google Scholar] [CrossRef] [PubMed]
- Mull, R.W.; Harrington, A.; Sanchez, L.A.; Tal-Gan, Y. Cyclic peptides that govern signal transduction pathways: From prokaryotes to multi-cellular organisms. Curr. Top. Med. Chem. 2018, 18, 625–644. [Google Scholar] [CrossRef] [PubMed]
- Nami, Y.; Vaseghi Bakhshayesh, R.; Mohammadzadeh Jalaly, H.; Lotfi, H.; Eslami, S.; Hejazi, M.A. Probiotic properties of Enterococcus isolated from artisanal dairy products. Front. Microbiol. 2019, 10, 300. [Google Scholar] [CrossRef] [PubMed]
- de Melo Pereira, G.V.; de Oliveira Coelho, B.; Magalhães Júnior, A.I.; Thomaz-Soccol, V.; Soccol, C.R. How to select a probiotic? A review and update of methods and criteria. Biotechnol. Adv. 2018, 36, 2060–2076. [Google Scholar] [CrossRef] [PubMed]
- Rijnaarts, H.H.; Norde, W.; Bouwer, E.J.; Lyklema, J.; Zehnder, A.J. Bacterial adhesion under static and dynamic conditions. Appl. Environ. Microbiol. 1993, 59, 3255–3265. [Google Scholar] [CrossRef] [PubMed]
- Schillinger, U.; Guigas, C.; Holzapfel, W.H. In vitro adherence and other properties of lactobacilli used in probiotic yoghurt-like products. Int. Dairy. Int. 2005, 15, 1289–1297. [Google Scholar] [CrossRef]
- Butorac, K.; Banić, M.; Novak, J.; Leboš Pavunc, A.; Uroić, K.; Durgo, K.; Oršolić, N.; Kukolj, M.; Radović, S.; Scalabrin, S.; et al. The functional capacity of plantaricin-producing Lactobacillus plantarum SF9C and S-layer-carrying Lactobacillus brevis SF9B to withstand gastrointestinal transit. Microb. Cell Fact. 2020, 19, 106. [Google Scholar] [CrossRef] [PubMed]
- Qin, W.; Xia, Y.; Xiong, Z.; Song, X.; Ai, L.; Wang, G. The intestinal colonization of Lactiplantibacillus plantarum AR113 is influenced by its mucins and intestinal environment. Food Res. Int. 2022, 157, 111382. [Google Scholar] [CrossRef] [PubMed]
- Chaffanel, F.; Charron-Bourgoin, F.; Soligot, C.; Kebouchi, M.; Bertin, S.; Payot, S.; Le Roux, Y.; Leblond-Bourget, N. Surface proteins involved in the adhesion of Streptococcus salivarius to human intestinal epithelial cells. Appl. Microbiol. Biotechnol. 2018, 102, 2851–2865. [Google Scholar] [CrossRef] [PubMed]
- Chávarri, M.; Marañón, I.; Ares, R.; Ibáñez, F.C.; Marzo, F.; Villarán Mdel, C. Microencapsulation of a probiotic and prebiotic in alginate-chitosan capsules improves survival in simulated gastro-intestinal conditions. Int. J. Food Microbiol. 2010, 142, 185–189. [Google Scholar] [CrossRef] [PubMed]
- Solieri, L.; Bianchi, A.; Mottolese, G.; Lemmetti, F.; Giudici, P. Tailoring the probiotic potential of non-starter Lactobacillus strains from ripened Parmigiano Reggiano cheese by in vitro screening and principal component analysis. Food Microbiol. 2014, 38, 240–249. [Google Scholar] [CrossRef] [PubMed]
- Tulumoglu, S.; Yuksekdag, Z.N.; Beyatli, Y.; Simsek, O.; Cinar, B.; Yaşar, E. Probiotic properties of lactobacilli species isolated from children’s feces. Anaerobe 2013, 24, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Sarao, L.K.; Arora, M. Probiotics, prebiotics, and microencapsulation: A review. Crit. Rev. Food Sci. Nutr. 2017, 57, 344–371. [Google Scholar] [CrossRef] [PubMed]
- Kumari, M.; Kumar, R.; Singh, D.; Bhatt, S.; Gupta, M. Physiological and genomic characterization of an exopolysaccharide-producing Weissella cibaria CH2 from cheese of the western Himalayas. Food Biosci. 2020, 35, 100570. [Google Scholar] [CrossRef]
- Zhang, L.; Ma, H.; Kulyar, M.F.; Pan, H.; Li, K.; Li, A.; Mo, Q.; Wang, Y.; Dong, H.; Bao, Y.; et al. Complete genome analysis of Lactobacillus fermentum YLF016 and its probiotic characteristics. Microb. Pathog. 2022, 162, 105212. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Bajinka, O.; Jarju, P.O.; Tan, Y.; Taal, A.M.; Ozdemir, G. The varying effects of antibiotics on gut microbiota. AMB Express 2021, 11, 116. [Google Scholar] [CrossRef] [PubMed]
- Islam, R.; Hossain, M.N.; Alam, M.K.; Uddin, M.E.; Rony, M.H.; Imran, M.A.S.; Alam, M.F. Antibacterial activity of lactic acid bacteria and extraction of bacteriocin protein. Adv. Biosci. Biotechnol. 2020, 11, 49–59. [Google Scholar] [CrossRef]
- Hussain, N.; Tariq, M.; Saris, P.E.J.; Zaidi, A. Evaluation of the probiotic and postbiotic potential of lactic acid bacteria from artisanal dairy products against pathogens. J. Infect. Dev. Ctries. 2021, 15, 102–112. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Sun, M.; Mu, G.; Tuo, Y. Exopolysaccharide produced by Lactiplantibacillus plantarum Y12 exhibits inhibitory effect on the Shigella flexneri genes expression related to biofilm formation. Int. J. Biol. Macromol. 2023, 253, 127048. [Google Scholar] [CrossRef] [PubMed]
- Mahdhi, A.; Leban, N.; Chakroun, I.; Chaouch, M.A.; Hafsa, J.; Fdhila, K.; Mahdouani, K.; Majdoub, H. Extracellular polysaccharide derived from potential probiotic strain with antioxidant and antibacterial activities as a prebiotic agent to control pathogenic bacterial biofilm formation. Microb. Pathog. 2017, 109, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Salimi, F.; Farrokh, P. Recent advances in the biological activities of microbial exopolysaccharides. World J. Microbiol. Biotechnol. 2023, 39, 213. [Google Scholar] [CrossRef] [PubMed]
- Madhu, A.N.; Prapulla, S.G. Evaluation and functional characterization of a biosurfactant produced by Lactobacillus plantarum CFR 2194. Appl. Biochem. Biotechnol. 2014, 172, 1777–1789. [Google Scholar] [CrossRef] [PubMed]
- Abdalla, A.K.; Ayyash, M.M.; Olaimat, A.N.; Osaili, T.M.; Al-Nabulsi, A.A.; Shah, N.P.; Holley, R. Exopolysaccharides as antimicrobial agents: Mechanism and spectrum of activity. Front. Microbiol. 2021, 12, 664395. [Google Scholar] [CrossRef] [PubMed]
- Whiteley, M.; Diggle, S.P.; Greenberg, E.P. Progress in and promise of bacterial quorum sensing research. Nature 2017, 551, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Shin, H.; Lee, K.; Holzapfel, W. Autoinducer-2 properties of kimchi are associated with lactic acid bacteria involved in its fermentation. Int. J. Food Microbiol. 2016, 225, 38–42. [Google Scholar] [CrossRef] [PubMed]
- Man, L.L.; Meng, X.C.; Zhao, R.H. Induction of plantaricin MG under co-culture with certain lactic acid bacterial strains and identification of LuxS mediated quorum sensing system in Lactobacillus plantarum KLDS1.0391. Food Control 2012, 23, 462–469. [Google Scholar] [CrossRef]
- Costerton, W.J. Bacterial biofilms in nature and disease. Annu. Rev. Microbiol. 1987, 41, 435–464. [Google Scholar] [CrossRef] [PubMed]
- Vert, M.; Doi, Y.; Hellwich, K.-H.; Hess, M.J.; Hodge, P.; Kubisa, P.; Rinaudo, M.; Schué, F. Terminology for biorelated polymers and applications (IUPAC Recommendations 2012). Pure Appl. Chem. 2012, 84, 377–410. [Google Scholar] [CrossRef]
- Flemming, H.C.; Wuertz, S. Bacteria and archaea on Earth and their abundance in biofilms. Nat. Rev. Microbiol. 2019, 17, 247–260. [Google Scholar] [CrossRef] [PubMed]





| Type | Name | Size (bp) | G+C (%) | Genes | Pseudogenes (Total) | Protein | rRNA | tRNA | Non-Coding RNA | CRISPR_Array | CRISPR_Repeat | CRISPR_Spacer |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Chromosome | - | 3.126.700 | 44.79 | 3090 | 223 | 2747 | 64 | 16 | 40 | 6 | 85 | 79 |
| Plasmid | pWS01 | 72.513 | 42.33 | 79 | 12 | 67 | - | - | - | - | - | - |
| Plasmid | pWS02 | 71.633 | 40.97 | 71 | 6 | 65 | - | - | - | - | - | - |
| Plasmid | pWS03 | 54.200 | 40.14 | 55 | 10 | 45 | - | - | - | - | - | - |
| Plasmid | pWS04 | 51.130 | 41.74 | 70 | 5 | 65 | - | - | - | - | - | - |
| Plasmid | pWS05 | 47.792 | 43.9 | 42 | 8 | 34 | - | - | - | - | - | - |
| Sugars | JS21 |
|---|---|
| Control | − |
| Glycerol | + |
| Erythritol | − |
| D-Arabinose | − |
| L-Arabinose | − |
| D-Ribose | + |
| D-Xylose | − |
| L-Xylose | − |
| Adonitol | − |
| Methyl-β-D-xylopyranoside | − |
| D-Galactose | + |
| D-Glucose | + |
| D-Fructose | + |
| D-Mannose | + |
| D-Sorbose | − |
| D-Rhamnose | − |
| Dulcitol | − |
| Inositol | + |
| D-Mannitol | + |
| D-Sorbitol | + |
| Methyl-α-D-mannopyranoside | + |
| Methyl-α-D-glucopyranoside | − |
| N-Acetylglucosamine | + |
| Amygdalin | + |
| Arbutin | + |
| Esculin ferric citrate | + |
| Salicin | + |
| D-Cellobiose | + |
| D-Maltose | + |
| D-Lactose | + |
| D-Melibiose | + |
| D-Sucrose | + |
| D-Trehalose | + |
| Inulin | − |
| D-Melezitose | + |
| D-Raffinose | − |
| Amidon (Starch) | − |
| Glycogen | − |
| Xylitol | − |
| Gentiobiose | + |
| D-Turanose | − |
| D-Lyxose | − |
| D-Tagatose | − |
| D-Fucose | − |
| L-Fucose | − |
| D-Arabitol | − |
| L-Arabitol | − |
| Gluconate | − |
| 2-Keto-gluconate | − |
| 5-Keto-gluconate | − |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.; Wang, S.; Wang, L.; Lu, H.; Zhang, T.; Zeng, W. Characterization of Genomic, Physiological, and Probiotic Features of Lactiplantibacillus plantarum JS21 Strain Isolated from Traditional Fermented Jiangshui. Foods 2024, 13, 1082. https://doi.org/10.3390/foods13071082
Liu Y, Wang S, Wang L, Lu H, Zhang T, Zeng W. Characterization of Genomic, Physiological, and Probiotic Features of Lactiplantibacillus plantarum JS21 Strain Isolated from Traditional Fermented Jiangshui. Foods. 2024; 13(7):1082. https://doi.org/10.3390/foods13071082
Chicago/Turabian StyleLiu, Yang, Shanshan Wang, Ling Wang, Hongzhao Lu, Tao Zhang, and Wenxian Zeng. 2024. "Characterization of Genomic, Physiological, and Probiotic Features of Lactiplantibacillus plantarum JS21 Strain Isolated from Traditional Fermented Jiangshui" Foods 13, no. 7: 1082. https://doi.org/10.3390/foods13071082
APA StyleLiu, Y., Wang, S., Wang, L., Lu, H., Zhang, T., & Zeng, W. (2024). Characterization of Genomic, Physiological, and Probiotic Features of Lactiplantibacillus plantarum JS21 Strain Isolated from Traditional Fermented Jiangshui. Foods, 13(7), 1082. https://doi.org/10.3390/foods13071082




