Algae-Boosted Chickpea Hummus: Improving Nutrition and Texture with Seaweeds and Microalgae
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Hummus Preparation
2.2.2. Hummus Nutritional Composition
2.2.3. Hummus Bioactivity Assays
2.2.4. Hummus pH, aw, and Color Measurements
2.2.5. Hummus Rheology Evaluation
2.2.6. Hummus Texture Analysis
2.2.7. Hummus Microscopy Analysis
2.2.8. Statistical Analysis
3. Results and Discussion
3.1. Nutritional Characterization and Mineral Profile
3.1.1. Nutritional Composition
3.1.2. Mineral Profile
3.2. Phenolic Compounds and Antioxidant Capacity
3.3. pH, aw, and Color Interpretations
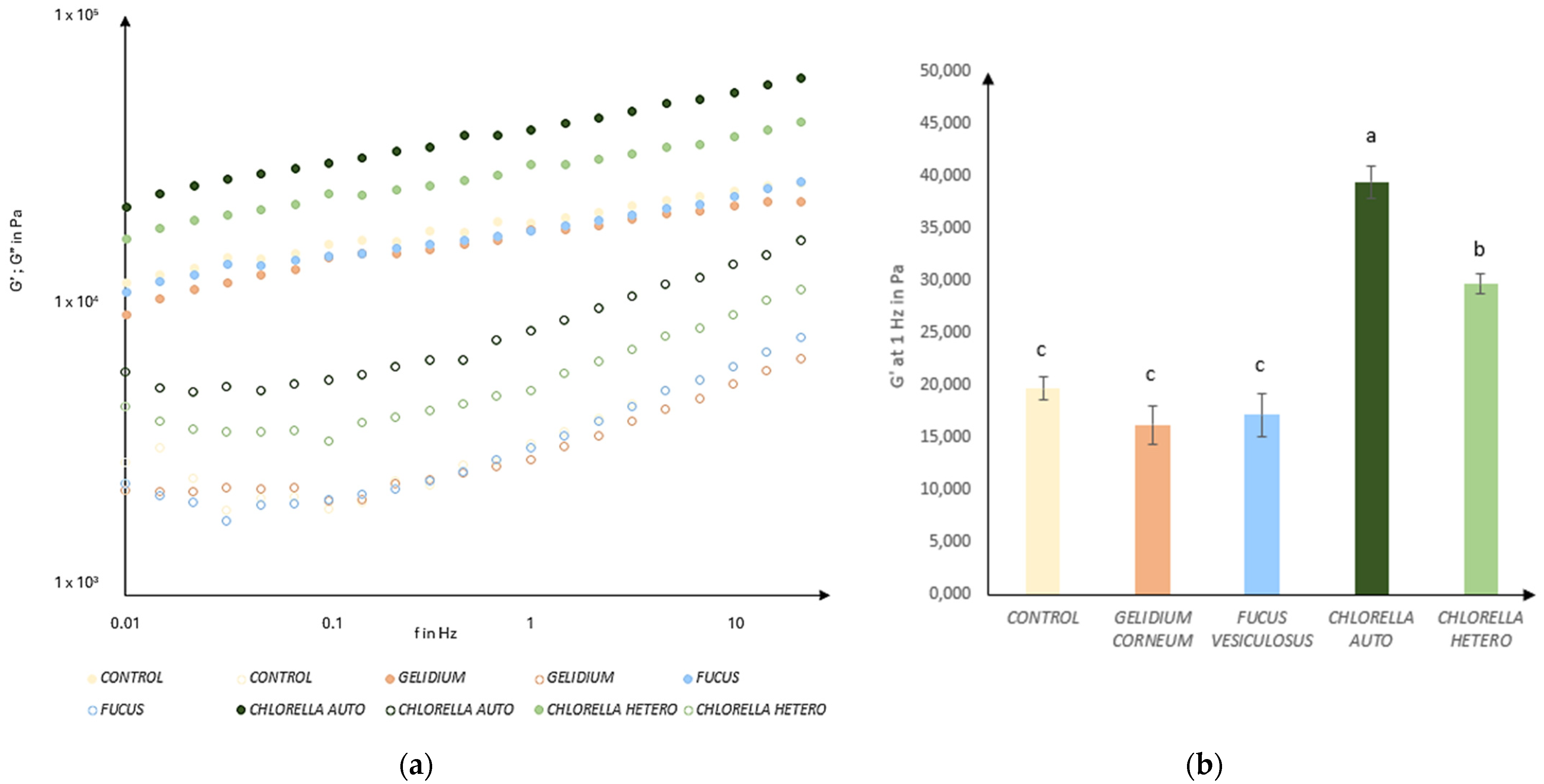
3.4. Rheology Behavior
3.5. Texture Results
3.6. Microscopy
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Reister, E.J.; Belote, L.N.; Leidy, H.J. The Benefits of Including Hummus and Hummus Ingredients into the American Diet to Promote Diet Quality and Health: A Comprehensive Review. Nutrients 2020, 12, 3678. [Google Scholar] [CrossRef] [PubMed]
- International Center for Advanced Mediterranean Agronomic Studies (CIHEAM). The Mediterranean Diet for Sustainable Regional Development. In MediTERRA; Annual Editor; Presses de Sciences Po: France, Paris, 2012. [Google Scholar] [CrossRef]
- Andersen, N.R.; van Deurs Petersen, R.; Frøst, M.B. Consumer interest in hummus made from different pulses: Effects of information about origin and variety seeking tendency. Int. J. Gastron. Food Sci. 2022, 29, 100572. [Google Scholar] [CrossRef]
- Malcolmson, L.; Sissons, M. Grains and Pulses Fuel Consumer Trends. Cereal Foods World 2017, 62, 195. [Google Scholar] [CrossRef][Green Version]
- Wallace, T.C.; Murray, R.; Zelman, K.M. The Nutritional Value and Health Benefits of Chickpeas and Hummus. Nutrients 2016, 8, 766. [Google Scholar] [CrossRef] [PubMed]
- Tosti, V.; Bertozzi, B.; Fontana, L.V. Health Benefits of the Mediterranean Diet: Metabolic and Molecular Mechanisms. J. Gerontol. Ser. A 2018, 73, 318–326. [Google Scholar] [CrossRef] [PubMed]
- Shanthakumar, P.; Klepacka, J.; Bains, A.; Chawla, P.; Dhull, S.B.; Najda, A. The Current Situation of Pea Protein and Its Application in the Food Industry. Molecules 2022, 27, 5354. [Google Scholar] [CrossRef] [PubMed]
- United Nations Department of Economic and Social Affairs, Population Division. World Population Prospects 2022: Summary of Results; UN DESA/POP/2022/TR/NO. 3; United Nations Department of Economic and Social Affairs, Population Division: New York City, NY, USA, 2022. [Google Scholar]
- Jehn, F.U.; Dingal, F.J.; Mill, A.; Harrison, C.; Ilin, E.; Roleda, M.Y.; James, S.C.; Denkenberger, D. Seaweed as a Resilient Food Solution After a Nuclear War. Earth’s Future 2024, 12, e2023EF003710. [Google Scholar] [CrossRef]
- Zhao, W.; Subbiah, V.; Xie, C.; Yang, Z.; Shi, L.; Barrow, C. Bioaccessibility and Bioavailability of Phenolic Compounds in Seaweed. Food Rev. Int. 2023, 39, 5729–5760. [Google Scholar] [CrossRef]
- Mouritsen, O.G.; Cornish, M.L.; Critchley, A.T.; Pérez-Lloréns, J.L. Chapter 1—History of seaweeds as a food. In Applications of Seaweeds in Food and Nutrition; Elsevier: Amsterdam, The Netherlands, 2023; pp. 1–17. [Google Scholar] [CrossRef]
- Ebrahimzadeh, S.; Biswas, D.; Roy, S.; McClements, D.J. Incorporation of essential oils in edible seaweed-based films: A comprehensive review. Trends Food Sci. Technol. 2023, 135, 43–56. [Google Scholar] [CrossRef]
- Mouritsen, O.G.; Rhatigan, P.; Pérez-Lloréns, J.L. World cuisine of seaweeds: Science meets gastronomy. Int. J. Gastron. Food Sci. 2018, 14, 55–65. [Google Scholar] [CrossRef]
- Batista, A.P.; Gouveia, L.; Bandarra, N.M.; Franco, J.M.; Raymundo, A. Comparison of microalgal biomass profiles as novel functional ingredient for food products. Algal Res. 2013, 2, 164–173. [Google Scholar] [CrossRef]
- Sun, H.; Wang, Y.; He, Y.; Liu, B.; Mou, H.; Chen, F.; Yang, S. Microalgae-Derived Pigments for the Food Industry. Mar. Drugs 2023, 21, 82. [Google Scholar] [CrossRef] [PubMed]
- Naik, B.; Mishra, R.; Kumar, V.; Mishra, S.; Gupta, U.; Rustagi, S.; Gupta, A.K.; Preet, M.S.; Bhatt, S.C.; Rizwanuddin, S. Micro-algae: Revolutionizing food production for a healthy and sustainable future. J. Agric. Food Res. 2024, 15, 100939. [Google Scholar] [CrossRef]
- Nunes, M.C.; Ferreira, J.; Raymundo, A. Volatile fingerprint impact on the sensory properties of microalgae and development of mitigation strategies. Curr. Opin. Food Sci. 2023, 51, 101040. [Google Scholar] [CrossRef]
- United Nations Department of Economic and Social Affairs, Sustainable Division, THE 17 GOALS. Available online: https://sdgs.un.org/goals (accessed on 12 January 2024).
- Schirmacher, H.; Elshiewy, O.; Boztug, Y. That’s not natural! Consumer response to disconfirmed expectations about ‘natural’ food. Appetite 2023, 180, 106270. [Google Scholar] [CrossRef] [PubMed]
- SUMOL + COMPAL, 75 Anos de História. Available online: https://sumolcompal.pt/ (accessed on 12 January 2024).
- Iberagar, Especialista em Macroalgas Para o Setor dos Hidrocolóides. Available online: https://iberagar.com/?lang=pt-pt (accessed on 20 January 2024).
- ALGA+, I&D em Consórcio. Available online: https://www.algaplus.pt/investigacao/ (accessed on 3 January 2024).
- Allmicroalgae, Cultivando Soluções Sustentáveis de Microalgas—Allmicralgae. Available online: https://www.allmicroalgae.com/pt-pt/ (accessed on 31 January 2024).
- Cebrián-Lloret, V.; Martínez-Abad, A.; López-Rubio, A.; Martínez-Sanz, M. Sustainable Bio-Based Materials from Minimally Processed Red Seaweeds: Effect of Composition and Cell Wall Structure. J. Polym. Environ. 2023, 31, 886–899. [Google Scholar] [CrossRef]
- Morillas-Ruiz, J.M.; Delgado-Alarcón, J.M. Nutritional analysis of vegetable food with different origins: Evaluation of antioxidant capacity and phenolic total compounds. Nutr. Clínica Dietética Hosp. 2012, 32, 8–20. [Google Scholar]
- Aybar, M.; Simões, S.; Sales, J.R.; Santos, J.; Figueira, D.; Raymundo, A. Tenebrio molitor as a Clean Label Ingredient to Produce Nutritionally Enriched Food Emulsions. Insects 2023, 14, 147. [Google Scholar] [CrossRef] [PubMed]
- AACC International, Ash—Basic Method. 1999. Available online: https://www.cerealsgrains.org/resources/Methods/Pages/08TotalAsh.aspx (accessed on 1 February 2024).
- Oliveira, S.; Torres Pérez, M.D.; Sousa, I.; Raymundo, A. 3D-printed Chlorella vulgaris snacks: A contribution to a healthy diet. Front. Food Sci. Technol. 2023, 3, 1265828. [Google Scholar] [CrossRef]
- Mota, J.; Lima, A.; Ferreira, R.B.; Raymundo, A. Technological Potential of a Lupin Protein Concentrate as a Nutraceutical Delivery System in Baked Cookies. Foods 2021, 10, 1929. [Google Scholar] [CrossRef]
- Beltrão, M.R.; Gouvinhas, I.; Nunes, M.C.; Peres, J.A.; Raymundo, A.; Barros, A.I.R.N.A. Acorn Flour as a Source of Bioactive Compounds in Gluten-Free Bread. Molecules 2020, 25, 3568. [Google Scholar] [CrossRef]
- BMG LABTECH, CLARIOstar Plus Microplate Reader—Most flexible. Available online: https://www.bmglabtech.com/en/clariostar-plus/ (accessed on 15 January 2024).
- Zhang, Y.; Li, Y.; Ren, X.; Zhang, X.; Wu, Z.; Liu, L. The positive correlation of antioxidant activity and prebiotic effect about oat phenolic compounds. Food Chem. 2023, 402, 134231. [Google Scholar] [CrossRef] [PubMed]
- Reis, F.S.; Martins, A.; Barros, L.; Ferreira, I.C.F.R. Antioxidant properties and phenolic profile of the most widely appreciated cultivated mushrooms: A comparative study between in vxivo and in vitro samples. Food Chem. Toxicol. 2012, 50, 1201–1207. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-G.; Nagahawatta, D.P.; Amarasiri, R.P.G.S.K.; Jeon, Y.-J.; Kang, M.-C. Physico-chemical and DPPH-hydroxyl radical scavenging characteristics of crude polysaccharides from Sargassum thunbergia. Algal Res. 2023, 73, 103152. [Google Scholar] [CrossRef]
- Gulcin, İ.; Alwasel, S.H. DPPH Radical Scavenging Assay. Processes 2023, 11, 2248. [Google Scholar] [CrossRef]
- Zhang, M.-Y.; Chen, S.-L.; Lin, C.-Y.; Zhang, H.-X.; Zhang, T.; Zou, Z.-M. New caffeoyl derivatives with potent DPPH radical scavenging activity from Elephantopus tomentosus. J. Asian Nat. Prod. Res. 2024, 26, 26–37. [Google Scholar] [CrossRef] [PubMed]
- Olufemi, A.; Rabiat, A.S.; Raphael, D.O.; Mustapha, B.O.; Salau, S. Phytochemical Profiling and Molecular Docking Investigation of Avocado (Persea Americana Mill. Cultivar Hass) Leaves and Seeds: Implications for Antioxidant Activity and Health Benefits. Res. Sq. 2024; Preprint. [Google Scholar] [CrossRef]
- Bulut, O.; Sönmez, Ç.; Öktem, H.A. Hindakia tetrachotoma ME03 (Chlorophyta) has high phenolic content, antioxidant capacity, and attenuates H2O2-induced oxidative stress and apoptosis in human cells. Phycologia 2023, 62, 293–302. [Google Scholar] [CrossRef]
- Neutec Group, LabMaster Neo-Water Activity Meter. Available online: https://www.neutecgroup.com/water-activity-measurement/water-activity-labmaster-neo-detail (accessed on 20 January 2024).
- Vieira, M.R.; Simões, S.; Carrera-Sánchez, C.; Raymundo, A. Development of a Clean Label Mayonnaise Using Fruit Flour. Foods 2023, 12, 2111. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Castillo, E.; Oliveira, S.; Bengoechea, C.; Sousa, I.; Raymundo, A.; Guerrero, A. A rheological approach to 3D printing of plasma protein based doughs. J. Food Eng. 2021, 288, 110255. [Google Scholar] [CrossRef]
- Mezger, T.G. Applied Rheology: With Joe Flow on Rheology Road, 9th ed.; Antoon Paar GmbH: Graz, Austria, 2014. [Google Scholar]
- Simões, S.; Carrera Sanchez, C.; Santos, A.J.; Figueira, D.; Prista, C.; Raymundo, A. Impact of Grass Pea Sweet Miso Incorporation in Vegan Emulsions: Rheological, Nutritional and Bioactive Properties. Foods 2023, 12, 1362. [Google Scholar] [CrossRef]
- Silva, F.G.; Passerini, A.B.S.; Ozorio, L.; Picone, C.S.F.; Perrechil, F.A. Interactions between pea protein and gellan gum for the development of plant-based structures. Int. J. Biol. Macromol. 2024, 255, 128113. [Google Scholar] [CrossRef] [PubMed]
- Graça, C.; Raymundo, A.; de Sousa, I. Yoghurt and curd cheese addition to wheat bread dough: Impact on in vitro starch digestibility and estimated glycemic index. Food Chem. 2021, 339, 127887. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Wang, L.; Yu, X.; Wang, X.; Zheng, Y.; Hu, X.; Zhang, P.; Sun, Q.; Wang, Q.; Li, N. Effect of polysaccharide addition on food physical properties: A review. Food Chem. 2024, 431, 137099. [Google Scholar] [CrossRef] [PubMed]
- Regulamento 1924/2006 do Parlamento Europeu e do Conselho (20/12/2006) Relativo às Alegações Nutricionais e de Saúde Sobre os Alimentos. Available online: https://eur-lex.europa.eu/legal-content/PT/TXT/PDF/?uri=CELEX:32006R1924 (accessed on 30 January 2024).
- Safi, C.; Zebib, B.; Merah, O.; Pontalier, P.-Y.; Vaca-Garcia, C. Morphology, composition, production, processing and applications of Chlorella vulgaris: A review. Renew. Sustain. Energy Rev. 2014, 35, 265–278. [Google Scholar] [CrossRef]
- Nova, P.; Pimenta-Martins, A.; Maricato, É.; Nunes, C.; Abreu, H.; Coimbra, M.A.; Freitas, A.C.; Gomes, A.M. Chemical Composition and Antioxidant Potential of Five Algae Cultivated in Fully Controlled Closed Systems. Molecules 2023, 28, 4588. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Ardehali, H.; Min, J.; Wang, F. The molecular and metabolic landscape of iron and ferroptosis in cardiovascular disease. Nat. Rev. Cardiol. 2023, 20, 7–23. [Google Scholar] [CrossRef] [PubMed]
- Baj, J.; Flieger, W.; Barbachowska, A.; Kowalska, B.; Flieger, M.; Forma, A.; Teresiński, G.; Portincasa, P.; Buszewicz, G.; Radzikowska-Büchner, E.; et al. Consequences of Disturbing Manganese Homeostasis. Int. J. Mol. Sci. 2023, 24, 14959. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz-Ersan, L.; Suna, G. Comparison of the targeted metabolomics and nutritional quality indices of the probiotic cheese enriched with microalgae. Talanta 2024, 272, 125801. [Google Scholar] [CrossRef] [PubMed]
- Loughrill, E.; Wray, D.; Christides, T.; Zand, N. Calcium to phosphorus ratio, essential elements and vitamin D content of infant foods in the UK: Possible implications for bone health. Matern. Child Nutr. 2017, 13, e12368. [Google Scholar] [CrossRef]
- Guasti, L.; Cianferotti, L.; Pampaloni, B.; Tonelli, T.; Mertelli, F.; Iantomasi, T.; Brandi, M.L. Evaluation of food and nutrient intake in a population of subjects affected by periodontal disease with different levels of bone mineral density. Front. Endocrinol. 2023, 14, 1098366. [Google Scholar] [CrossRef]
- Tang, P.; Giusti, M.M. Metal Chelates of Petunidin Derivatives Exhibit Enhanced Color and Stability. Foods 2020, 9, 1426. [Google Scholar] [CrossRef] [PubMed]
- Belattmania, Z.; Bhaby, S.; Nadri, A.; Khaya, K.; Bentiss, F.; Jama, C.; Reani, A.; Vasconcelos, V.; Sabour, B. Gracilaria gracilis (Gracilariales, Rhodophyta) from Dakhla (Southern Moroccan Atlantic Coast) as Source of Agar: Content, Chemical Characteristics, and Gelling Properties. Mar. Drugs 2021, 19, 672. [Google Scholar] [CrossRef] [PubMed]
- Shewan, H.M.; Stokes, J.R.; Smyth, H.E. Influence of particle modulus (softness) and matrix rheology on the sensory experience of ‘grittiness’ and ‘smoothness’. Food Hydrocoll. 2020, 103, 105662. [Google Scholar] [CrossRef]
- Zhang, Z.-H.; Wang, L.-H.; Zeng, X.-A.; Han, Z.; Brennan, C.S. Non-thermal technologies and its current and future application in the food industry: A review. Int. J. Food Sci. Technol. 2019, 54, 1–13. [Google Scholar] [CrossRef]
- Batista, A.P.; Nunes, M.C.; Raymundo, A.; Gouveia, L.; Sousa, I.; Cordobés, F.; Guerrero, A.; Franco, J.M. Microalgae biomass interaction in biopolymer gelled systems. Food Hydrocoll. 2011, 25, 817–825. [Google Scholar] [CrossRef]
- Kaur, M.; Singh, N. Studies on functional, thermal and pasting properties of flours from different chickpea (Cicer arietinum L.) cultivars. Food Chem. 2005, 91, 403–411. [Google Scholar] [CrossRef]
- Bertsch, P.; Böcker, L.; Mathys, A.; Fischer, P. Proteins from microalgae for the stabilization of fluid interfaces, emulsions, and foams. Trends Food Sci. Technol. 2021, 108, 326–342. [Google Scholar] [CrossRef]
- Paul, A.A.; Kumar, S.; Kumar, V.; Sharma, R. Milk Analog: Plant based alternatives to conventional milk, production, potential and health concerns. Crit. Rev. Food Sci. Nutr. 2020, 60, 3005–3023. [Google Scholar] [CrossRef] [PubMed]
- Grossmann, L.; Hinrichs, J.; Goff, H.D.; Weiss, J. Heat-induced gel formation of a protein-rich extract from the microalga Chlorella sorokiniana. Innov. Food Sci. Emerg. Technol. 2019, 56, 102176. [Google Scholar] [CrossRef]
- Bernaerts, T.M.M.; Gheysen, L.; Foubert, I.; Hendrickx, M.E.; Van Loey, A.M. The potential of microalgae and their biopolymers as structuring ingredients in food: A review. Biotechnol. Adv. 2019, 37, 107419. [Google Scholar] [CrossRef]
- Braga, A.R.C.; Nunes, M.C.; Raymundo, A. The Experimental Development of Emulsions Enriched and Stabilized by Recovering Matter from Spirulina Biomass: Valorization of Residue into a Sustainable Protein Source. Molecules 2023, 28, 6179. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Yin, Z.; Sun, W.; Zhong, Q.; Zhang, Y.; Zeng, M. Microalgae play a structuring role in food: Effect of spirulina platensis on the rheological, gelling characteristics, and mechanical properties of soy protein isolate hydrogel. Food Hydrocoll. 2023, 136, 108244. [Google Scholar] [CrossRef]
- Cebrián-Lloret, V.; Martínez-Abad, A.; López-Rubio, A.; Martínez-Sanz, M. Exploring alternative red seaweed species for the production of agar-based hydrogels for food applications. Food Hydrocoll. 2024, 146, 109177. [Google Scholar] [CrossRef]
- Oprea, O.B.; Tolstorebrov, I.; Claussen, I.C.; Sannan, S.; Apostol, L.; Moșoiu, C.; Gaceu, L. Potential for Saccharina latissima Flour as a Functional Ingredient in the Baking Sector. Foods 2023, 12, 4498. [Google Scholar] [CrossRef]
- Bose, I.; Nousheen; Roy, S.; Yaduvanshi, P.; Sharma, S.; Chandel, V.; Biswas, D. Unveiling the Potential of Marine Biopolymers: Sources, Classification, and Diverse Food Applications. Materials 2023, 16, 4840. [Google Scholar] [CrossRef]




| Algae | Protein (g/100 g) | Fat (g/100 g) | Carbohydrates (g/100 g) | Dietary Fiber (g/100 g) |
|---|---|---|---|---|
| Gelidium corneum | 19.4 | 4.7 | 35.2 | nd 1 |
| Fucus vesiculosus | 14.7 | 2.2 | 10.0 | 41.5 |
| Chlorella vulgaris (heterotrophic) | 50.0 | 9.5 | 14.2 | 15.2 |
| Chlorella vulgaris (autotrophic) | 38.3 | 14.2 | 21.8 | 17.7 |
| Ingredients | Amount (in g) |
|---|---|
| Tahini | 150 (70 of Oo and 80 of Ss) 1 |
| Canned chickpeas | 400 |
| Lemon juice | 50 |
| Garlic | 5 |
| Balsamic vinegar | 5 |
| Salt | 6 |
| Aquafaba (from the canned chickpeas) | 50 |
| Hummus | Humidity (g/100 g) | Ash (g/100 g) | Protein (g/100 g) | Fat (g/100 g) | Carbohydrates (g/100 g) |
|---|---|---|---|---|---|
| Control | 62.94 ± 0.79 a | 2.19 ± 0.26 a | 7.23 ± 0.04 c | 10.05 ± 0.98 a | 17.59 ± 0.12 c |
| Gelidium corneum | 61.31 ± 0.09 a | 2.27 ± 0.13 a | 7.13 ± 0.10 cd | 8.18 ± 1.26 b | 21.11 ± 0.51 b |
| Fucus vesiculosus | 55.81 ± 1.88 b | 2.30 ± 0.36 a | 6.94 ± 0.02 d | 9.03 ± 1.15 b | 25.92 ± 1.47 a |
| Chlorella vulgaris (heterotrophic) | 51.86 ± 0.77 c | 2.68 ± 0.05 a | 9.70 ± 0.14 a | 13.97 ± 0.72 a | 21.78 ± 0.15 b |
| Chlorella vulgaris (autotrophic) | 51.77 ± 0.36 c | 2.39 ± 0.05 a | 8.65 ± 0.02 b | 13.77 ± 0.13 a | 23.42 ± 0.29 b |
| Minerals | Recommended Values in mg/100 g Source of/Rich in | Control | Gelidium corneum | Fucus vesiculosus | Chlorella Vulgaris Autotrophic | Chlorella Vulgaris Heterotrophic |
|---|---|---|---|---|---|---|
| Na | - | 555.32 ± 3.29 b | 521.13 ± 3.36 c | 631.89 ± 4.70 a | 532.55 ± 13.21 bc | 524.34 ± 13.92 c |
| K | 300/600 | 211.32 ± 1.30 d | 317.75 ± 0.87 b | 361.45 ± 7.31 a | 249.04 ± 7.94 c | 319.69 ± 7.16 b |
| Ca | 120/240 | 132.50 ± 5.44 c | 112.89 ± 2.36 d | 162.15 ± 0.84 b | 197.02 ± 6.58 a | 113.58 ± 3.95 d |
| Mg | 57/114 | 55.98 ± 0.35 d | 72.70 ± 0.55 b | 84.93 ± 1.26 a | 66.87 ± 1.83 c | 73.15 ± 2.00 b |
| P | 105/210 | 124.01 ± 2.12 b | 105.58 ± 1.67 c | 106.44 ± 0.45 c | 217.14 ± 6.14 a | 106.24 ± 3.63 c |
| S | - | 96.80 ± 1.11 d | 185.86 ± 4.20 b | 216.35 ± 8.98 a | 126.05 ± 2.68 c | 187.04 ± 8.00 b |
| Fe | 2.1/4.2 | 2.49 ± 0.04 c | 2.62 ± 0.09 c | 6.49 ± 0.23 b | 8.53 ± 0.25 a | 2.64 ± 0.14 c |
| Cu | 0.15/0.3 | 0.36 ± 0.01 b | 0.28 ± 0.01 c | 0.33 ± 0.01 b | 0.43 ± 0.02 a | 0.29 ± 0.02 c |
| Zn | 1.5/3 | 1.10 ± 0.02 bc | 1.05 ± 0.02 c | 1.19 ± 0.01 b | 2.66 ± 0.08 a | 1.06 ± 0.04 c |
| Mn | 0.3/0.6 | 0.63 ± 0.00 d | 0.71 ± 0.01 c | 2.54 ± 0.01 a | 1.19 ± 0.04 b | 0.72 ± 0.02 c |
| Samples | L* | a* | b* | ΔE |
|---|---|---|---|---|
| Control | 63.41 ± 0.33 a | 3.69 ± 0.13 a | 25.50 ± 0.35 b | - |
| Gelidium corneum | 56.31 ± 0.57 b | 3.17 ± 0.83 a | 14.80 ± 0.18 e | 12.85 |
| Fucus vesiculosus | 62.41 ± 0.63 a | 0.58 ± 0.11 b | 21.62 ± 0.38 c | 5.07 |
| Chlorella vulgaris (heterotrophic) | 34.55 ± 0.87 d | −3.44 ± 0.20 c | 16.31 ± 0.36 d | 31.11 |
| Chlorella vulgaris (autotrophic) | 47.94 ± 0.91 c | −4.51 ± 0.08 c | 29.45 ± 0.55 a | 17.95 |
| Samples | Firmness (N) | Adhesiveness (−N.s) | Cohesiveness |
|---|---|---|---|
| Control | 0.32 ± 0.02 c | 1.57 ± 0.17 c | 0.80 ± 0.02 a |
| Gelidium corneum | 0.22 ± 0.01 c | 0.80 ± 0.04 d | 0.60 ± 0.03 c |
| Fucus vesiculosus | 0.28 ± 0.03 c | 1.39 ± 0.18 c | 0.81 ± 0.03 a |
| Chlorella vulgaris (heterotrophic) | 0.82 ± 0.07 a | 3.91 ± 0.22 a | 0.73 ± 0.02 b |
| Chlorella vulgaris (autotrophic) | 0.46 ± 0.01 b | 2.46 ± 0.12 b | 0.78 ± 0.03 ab |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matheus, J.; Alegria, M.J.; Nunes, M.C.; Raymundo, A. Algae-Boosted Chickpea Hummus: Improving Nutrition and Texture with Seaweeds and Microalgae. Foods 2024, 13, 2178. https://doi.org/10.3390/foods13142178
Matheus J, Alegria MJ, Nunes MC, Raymundo A. Algae-Boosted Chickpea Hummus: Improving Nutrition and Texture with Seaweeds and Microalgae. Foods. 2024; 13(14):2178. https://doi.org/10.3390/foods13142178
Chicago/Turabian StyleMatheus, José, Maria João Alegria, Maria Cristiana Nunes, and Anabela Raymundo. 2024. "Algae-Boosted Chickpea Hummus: Improving Nutrition and Texture with Seaweeds and Microalgae" Foods 13, no. 14: 2178. https://doi.org/10.3390/foods13142178
APA StyleMatheus, J., Alegria, M. J., Nunes, M. C., & Raymundo, A. (2024). Algae-Boosted Chickpea Hummus: Improving Nutrition and Texture with Seaweeds and Microalgae. Foods, 13(14), 2178. https://doi.org/10.3390/foods13142178









