Exploring the Potential Medicinal Benefits of Ganoderma lucidum: From Metabolic Disorders to Coronavirus Infections
Abstract
1. Introduction
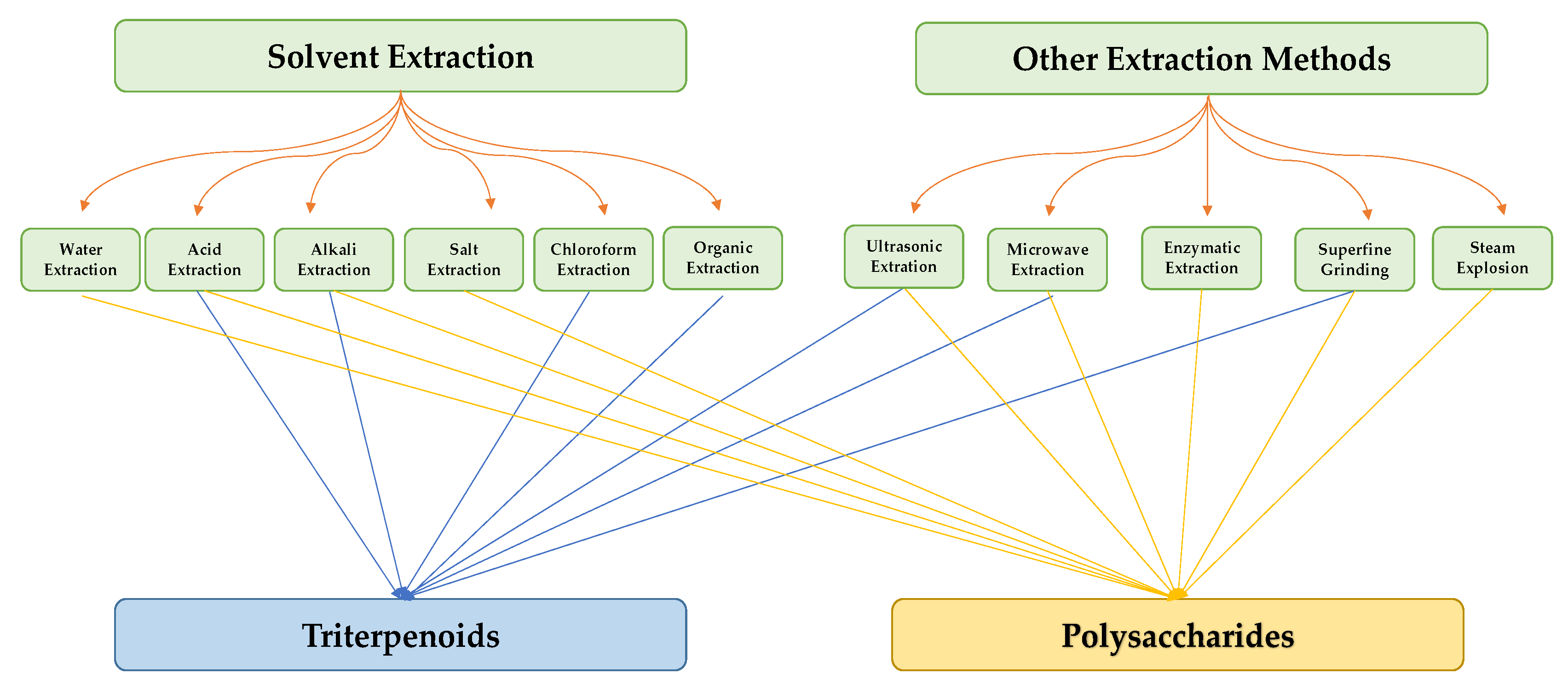
1.1. Ganoderma Lucidum Components
1.1.1. Triterpenes
1.1.2. Polysaccharides
1.1.3. Other Bioactive Components
1.2. Consumption of G. lucidum and Its Effects on Health
1.2.1. Mechanism of Action as Antioxidant
1.2.2. Mechanism of Action as Anticancer
1.2.3. Mechanism of Action against Cardiovascular Diseases
1.2.4. Antidiabetic Mechanism of Action
1.2.5. The Mechanism of Action against COVID-19
1.3. Potential Properties and Preparations of G. lucidum: A Need for Further Research and Quality Control
- Capsules or tablets containing concentrated extracts of the mushroom.
- Powdered forms of the mushroom, which can be added to food or beverages.
- Tinctures or liquid extracts, which can be added to water or other liquids.
- Tea bags or loose tea leaves, which can be brewed similarly to traditional tea.
1.4. Challenges and Limitations in the Clinical Application of G. lucidum Active Constituents against Metabolic Disorders and Coronavirus
2. Conclusions
3. Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sharma, C.; Bhardwaj, N.; Sharma, A.; Tuli, H.S.; Batra, P.; Beniwal, V.; Gupta, G.K.; Sharma, A.K. Bioactive metabolites of Ganoderma lucidum: Factors, mechanism and broad spectrum therapeutic potential. J. Herb. Med. 2019, 17, 100268. [Google Scholar] [CrossRef]
- Nguyen, A.N.; Johnson, T.E.; Jeffery, D.W.; Danner, L.; Bastian, S.E. A cross-cultural examination of Australian, Chinese and Vietnamese consumers’ attitudes toward a new Australian wine product containing Ganoderma lucidum extract. Food Res. Int. 2019, 115, 393–399. [Google Scholar] [CrossRef] [PubMed]
- Wasser, S.P. Reishi or ling zhi (Ganoderma lucidum). Encycl. Diet. Suppl. 2005, 1, 603–622. [Google Scholar]
- Ferreira, I.C.; Heleno, S.A.; Reis, F.S.; Stojkovic, D.; Queiroz, M.J.R.; Vasconcelos, M.H.; Sokovic, M. Chemical features of Ganoderma polysaccharides with antioxidant, antitumor and antimicrobial activities. Phytochem. 2015, 114, 38–55. [Google Scholar] [CrossRef]
- El Sheikha, A.F. Nutritional Profile and Health Benefits of Ganoderma lucidum “Lingzhi, Reishi, or Mannentake” as Functional Foods: Current Scenario and Future Perspectives. Foods 2022, 11, 1030. [Google Scholar] [CrossRef]
- Seng, L.S. Mushroom of Immortality Ganoderma lucidum; Gano Excel: Chino, CA, USA, 2014; pp. 1–40. [Google Scholar]
- Jiang, Y.; Chang, Y.; Liu, Y.; Zhang, M.; Luo, H.; Hao, C.; Zeng, P.; Sun, Y.; Wang, H.; Zhang, L. Overview of Ganoderma sinense polysaccharide—An adjunctive drug used during concurrent chemo/radiation therapy for cancer treatment in China. Biomed. Pharmacother. 2017, 96, 865–870. [Google Scholar] [CrossRef]
- Ahmad, M.F.; Wahab, S.; Ahmad, F.A.; Ashraf, S.A.; Abullais, S.S.; Saad, H.H. Ganoderma lucidum: A potential pleiotropic approach of ganoderic acids in health reinforcement and factors influencing their production. Fungal Biol. Rev. 2022, 39, 100–125. [Google Scholar] [CrossRef]
- Baby, S.; Johnson, A.J.; Govindan, B. Secondary metabolites from Ganoderma. Phytochemistry 2015, 114, 66–101. [Google Scholar] [CrossRef]
- Liang, C.; Tian, D.; Liu, Y.; Li, H.; Zhu, J.; Li, M.; Xin, M.; Xia, J. Review of the molecular mechanisms of Ganoderma lucidum triterpenoids: Ganoderic acids A, C2, D, F, DM, X and Y. Eur. J. Med. Chem. 2019, 174, 130–141. [Google Scholar] [CrossRef]
- Zhao, C.; Zhang, C.; Xing, Z.; Ahmad, Z.; Li, J.S.; Chang, M.W. Pharmacological effects of natural Ganoderma and its extracts on neurological diseases: A comprehensive review. Int. J. Biol. Macromol. 2019, 121, 1160–1178. [Google Scholar] [CrossRef]
- Dong, Z.; Dong, G.; Lai, F.; Wu, H.; Zhan, Q. Purification and comparative study of bioactivities of a natural selenized polysaccharide from Ganoderma lucidum mycelia. Int. J. Biol. Macromol. 2021, 190, 101–112. [Google Scholar] [CrossRef]
- Upton, R.; Petrone, C.; Graff, A.; Swisher, D.; McGuffin, M.; Pizzorno, J. Reishi mushroom: Ganoderma lucidum: Standards of analysis, quality control, and therapeutics. Am. Herb. Pharm. 2000, 11, 13–20. [Google Scholar]
- Vo, T.S.; Vo, T.T.B.C.; Vo, T.T.T.N. Available chemical constituents and activities of Ganoderma lucidum (Lingzhi or red Reishi) utilizing in disease treatment: A mini review. J. Res. Clin. Med. 2021, 9, 32. [Google Scholar] [CrossRef]
- Ahmad, M.F. Ganoderma lucidum: A rational pharmacological approach to surmount cancer. J. Ethnopharmacol. 2020, 260, 113047. [Google Scholar] [CrossRef] [PubMed]
- Sirisidthi, K.; Kosai, P.; Jiraungkoorskul, W. Antidiabetic activity of the lingzhi or reishi medicinal mushroom Ganoderma lucidum. SA Pharm. J. 2016, 83, 45–47. Available online: https://hdl.handle.net/10520/EJC196849 (accessed on 10 October 2022).
- Paterson, R.R.M. Ganoderma—A therapeutic fungal biofactory. Phytochemistry 2006, 67, 1985–2001. [Google Scholar] [CrossRef]
- Zhou, X.W.; Su, K.Q.; Zhang, Y.M. Applied modern biotechnology for cultivation of Ganoderma and development of their products. Appl. Microbiol. Biotechnol. 2012, 93, 941–963. [Google Scholar] [CrossRef]
- Guo, J.C.; Yang, L.; Ma, Q.Y.; Ge, Y.Z.; Kong, F.D.; Zhou, L.M.; Zhang, F.; Xie, Q.Y.; Yu, Z.F.; Dai, H.F.; et al. Triterpenoids and meroterpenoids with α-glucosidase inhibitory activities from the fruiting bodies of Ganoderma australe. Bioorg. Chem. 2021, 117, 105448. [Google Scholar] [CrossRef]
- Taofiq, O.; Heleno, S.A.; Calhelha, R.C.; Alves, M.J.; Barros, L.; González-Paramás, A.M.; Barreiro, M.F.; Ferreira, I.C. The potential of Ganoderma lucidum extracts as bioactive ingredients in topical formulations, beyond its nutritional benefits. Food Chem. Toxicol. 2017, 108, 139–147. [Google Scholar] [CrossRef]
- Zhang, Y.; Cai, H.; Tao, Z.; Yuan, C.; Jiang, Z.; Liu, J.; Kurihara, H.; Xu, W. Ganoderma lucidum spore oil (GLSO), a novel antioxidant, extends the average life span in Drosophila melanogaster. Food Sci. Hum. Wellness 2021, 10, 38–44. [Google Scholar] [CrossRef]
- Kou, R.W.; Xia, B.; Wang, Z.J.; Li, J.N.; Yang, J.R.; Gao, Y.Q.; Yin, X.; Gao, J.M. Triterpenoids and meroterpenoids from the edible Ganoderma resinaceum and their potential anti-inflammatory, antioxidant and anti-apoptosis activities. Bioorg. Chem. 2022, 121, 105689. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; He, R.; Sun, P.; Zhang, F.; Linhardt, R.J.; Zhang, A. Molecular mechanisms of bioactive polysaccharides from Ganoderma lucidum (Lingzhi), A review. Int. J. Biol. Macromol. 2020, 150, 765–774. [Google Scholar] [CrossRef] [PubMed]
- Adeyi, A.O.; Awosanya, S.A.; Adeyi, O.E.; James, A.S.; Adenipekun, C.O. Ganoderma lucidum ethanol extract abrogates metabolic syndrome in rats: In vivo evaluation of hypoglycemic, hypolipidemic, hypotensive and antioxidant properties. Obes. Med. 2021, 22, 100320. [Google Scholar] [CrossRef]
- Ayvaz, Z. Çin tıbbında 4000 yıldır ilaç olarak kullanılan Reishi mantarı. Ekoloji 2022, 2, 27–29. [Google Scholar]
- Boh, B.; Berovic, M.; Zhang, J.; Zhi-Bin, L. Ganoderma lucidum and its pharmaceutically active compounds. Biotechnol. Annu. Rev. 2007, 13, 265–301. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, M.F. Ganoderma lucidum: Persuasive biologically active constituents and their health endorsement. Biomed. Pharmacother. 2018, 107, 507–519. [Google Scholar] [CrossRef]
- Cör, D.; Knez, Ž.; Knez Hrnčič, M. Antitumor, antimicrobial, antioxidant and antiacetylcholinesterase effect of Ganoderma lucidum terpenoids and polysaccharides: A review. Molecules 2018, 23, 649. [Google Scholar] [CrossRef]
- Domínguez, R.; Pateiro, M.; Gagaoua, M.; Barba, F.J.; Zhang, W.; Lorenzo, J.M. A comprehensive review on lipid oxidation in meat and meat products. Antioxidants 2019, 8, 429. [Google Scholar] [CrossRef]
- Sharifi-Rad, M.; Anil Kumar, N.V.; Zucca, P.; Varoni, E.M.; Dini, L.; Panzarini, E.; Rajkovic, J.; Fokou, P.V.T.; Azzini, E.; Peluso, I.; et al. Lifestyle, oxidative stress, and antioxidants: Back and forth in the pathophysiology of chronic diseases. Front. Physiol. 2020, 11, 694. [Google Scholar] [CrossRef]
- Fleming, E.; Luo, Y. Codelivery of synergistic antioxidants from food sources for the prevention of oxidative stress. J. Agric. Food Res. 2021, 3, 100107. [Google Scholar] [CrossRef]
- Choe, E.; Min, D.B. Mechanisms of antioxidants in the oxidation of foods. Compr. Rev. Food Sci. Food Saf. 2009, 8, 345–358. [Google Scholar] [CrossRef]
- Ahmed, M.; Pickova, J.; Ahmad, T.; Liaquat, M.; Farid, A.; Jahangir, M. Oxidation of Lipids in Foods. Sarhad J. Agric. 2016, 32, 230–238. [Google Scholar] [CrossRef]
- Si, J.; Meng, G.; Wu, Y.; Ma, H.F.; Cui, B.K.; Dai, Y.C. Medium composition optimization, structural characterization, and antioxidant activity of exopolysaccharides from the medicinal mushroom Ganoderma lingzhi. Int. J. Biol. Macromol. 2019, 124, 1186–1196. [Google Scholar] [CrossRef] [PubMed]
- Mahendran, S.; Anandapandian, K.T.K.; Shankar, T.; Chellaram, C.; Vijayabaskar, P. Antioxidant properties of Ganoderma lucidum crude exopolysaccharide. Indian J. Innov. Dev. 2012, 1, 1–6. [Google Scholar]
- Veljović, S.; Veljović, M.; Nikićević, N.; Despotović, S.; Radulović, S.; Nikšić, M.; Filipović, L. Chemical composition, antiproliferative and antioxidant activity of differently processed Ganoderma lucidum ethanol extracts. J. Food Sci. Technol. 2017, 54, 1312–1320. [Google Scholar] [CrossRef]
- Ryu, D.H.; Cho, J.Y.; Sadiq, N.B.; Kim, J.C.; Lee, B.; Hamayun, M.; Lee, T.S.; Kim, H.S.; Park, H.S.; Nho, C.W.; et al. Optimization of antioxidant, anti-diabetic, and anti-inflammatory activities and ganoderic acid content of differentially dried Ganoderma lucidum using response surface methodology. Food Chem. 2021, 335, 127645. [Google Scholar] [CrossRef]
- Zhang, L.; Qiao, H.Y.; Liu, H.X.; Jiang, G.C.; Wang, L.Y.; Liu, X.J. Antioxidant, hypoglycemic and protection of acute liver injury activities of Ganoderma lucidum spore water extract. J. Funct. Foods 2022, 97, 105254. [Google Scholar] [CrossRef]
- Mathur, G.; Nain, S.; Sharma, P. Cancer: An overview. Acad. J. Cancer Res. 2015, 8, 1–9. [Google Scholar]
- Kumari, M. Cancer notes. Nutr. Cancer 2020, 1, 1–15. [Google Scholar]
- American Cancer Society, Cancer Facts & Figures. 2022. Available online: https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2022/2022-cancer-facts-and-figures.pdf (accessed on 13 September 2022).
- Donaldson, M.S. Nutrition and cancer: A review of the evidence for an anticancer diet. Nutr. J. 2004, 3, 19. [Google Scholar] [CrossRef]
- Kamal, N.; Ilowefah, M.A.; Hilles, A.R.; Anua, N.A.; Awin, T.; Alshwyeh, H.A.; Aldosary, S.K.; Jambocus, N.G.S.; Alosaimi, A.A.; Rahman, A.; et al. Genesis and Mechanism of Some Cancer Types and an Overview on the Role of Diet and Nutrition in Cancer Prevention. Molecules 2022, 27, 1794. [Google Scholar] [CrossRef]
- Stepien, M.; Chajes, V.; Romieu, I. The role of diet in cancer: The epidemiologic link. Salud Publica De Mex. 2016, 58, 261–273. [Google Scholar] [CrossRef] [PubMed]
- de la Fuente, B.; Berrada, H.; Barba, F.J. Marine resources and cancer therapy: From current evidence to challenges for functional foods development. Curr. Opin. Food Sci. 2022, 44, 100805. [Google Scholar] [CrossRef]
- Pathak, M.P.; Pathak, K.; Saikia, R.; Gogoi, U.; Ahmad, M.Z.; Patowary, P.; Das, A. Immunomodulatory effect of mushrooms and their bioactive compounds in cancer: A comprehensive review. Biomed. Pharmacother. 2022, 149, 112901. [Google Scholar] [CrossRef]
- Sliva, D. Ganoderma lucidum (Reishi) in cancer treatment. Integr. Cancer Ther. 2003, 2, 358–364. [Google Scholar] [CrossRef] [PubMed]
- Sui, X.C.; Guo, Q.B.; Xia, Y.M.; Cui, S.W.; Shen, J.; Zhang, J.; Ding, Z.Y. Structure features of the intracellular polysaccharide from Ganoderma lucidum and the irrelative immune-anticancer activities of GLPs. Bioact. Carbohydr. Diet. Fibre 2016, 8, 43–50. [Google Scholar] [CrossRef]
- Chen, S.; Li, X.; Yong, T.; Wang, Z.; Su, J.; Jiao, C.; Xie, Y.; Yang, B.B. Cytotoxic lanostane-type triterpenoids from the fruiting bodies of Ganoderma lucidum and their structure–activity relationships. Oncotarget 2017, 8, 10071. [Google Scholar] [CrossRef]
- Wang, G.; Zhao, J.; Liu, J.; Huang, Y.; Zhong, J.J.; Tang, W. Enhancement of IL-2 and IFN-γ expression and NK cells activity involved in the anti-tumor effect of ganoderic acid Me in vivo. Int. Immunopharmacol. 2007, 7, 864–870. [Google Scholar] [CrossRef]
- Weng, C.J.; Chau, C.F.; Hsieh, Y.S.; Yang, S.F.; Yen, G.C. Lucidenic acid inhibits PMA-induced invasion of human hepatoma cells through inactivating MAPK/ERK signal transduction pathway and reducing binding activities of NF-κB and AP-1. Carcinogenesis 2008, 29, 147–156. [Google Scholar] [CrossRef]
- Min, B.S.; Gao, J.J.; Nakamura, N.; Hattori, M. Triterpenes from the spores of Ganoderma lucidum and their cytotoxicity against meth-A and LLC tumor cells. Chem. Pharm. Bull. 2000, 48, 1026–1033. [Google Scholar] [CrossRef]
- Wattanapitayakul, S.K.; Bauer, J.A. Oxidative pathways in cardiovascular disease: Roles, mechanisms, and therapeutic implications. Pharmacol. Ther. 2001, 89, 187–206. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wu, W.; Liu, W.; Zhou, M. Roles and mechanisms of renalase in cardiovascular disease: A promising therapeutic target. Biomed. Pharmacother. 2020, 131, 110712. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.D.; Luo, M.; Shang, A.; Mao, Q.Q.; Li, B.Y.; Gan, R.Y.; Li, H.B. Antioxidant food components for the prevention and treatment of cardiovascular diseases: Effects, mechanisms, and clinical studies. Oxid. Med. Cell. Longev. 2021, 2021, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Xiong, Y.; Yang, T.; Wang, Y.; Zeng, J.; Zhou, S.; Luo, Y.; Li, L. Icariin and its metabolites as potential protective phytochemicals against cardiovascular disease: From effects to molecular mechanisms. Biomed. Pharmacother. 2022, 147, 112642. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Cardiovascular Diseases. 2021. Available online: https://www.who.int/health-topics/cardiovascular-diseases#tab=tab_1 (accessed on 15 August 2022).
- Juul, F.; Vaidean, G.; Parekh, N. Ultra-processed foods and cardiovascular diseases: Potential mechanisms of action. Adv. Nutr. 2021, 12, 1673–1680. [Google Scholar] [CrossRef]
- Griffiths, K.; Aggarwal, B.B.; Singh, R.B.; Buttar, H.S.; Wilson, D.; De Meester, F. Food antioxidants and their anti-inflammatory properties: A potential role in cardiovascular diseases and cancer prevention. Diseases 2016, 4, 28. [Google Scholar] [CrossRef]
- Asgary, S.; Rastqar, A.; Keshvari, M. Functional food and cardiovascular disease prevention and treatment: A review. J. Am. Coll. Nutr. 2018, 37, 429–455. [Google Scholar] [CrossRef]
- Meneses, M.E.; Martínez-Carrera, D.; Torres, N.; Sánchez-Tapia, M.; Aguilar-López, M.; Morales, P.; Sobal, M.; Bernabé, T.; Escudero, H.; Granados-Portillo, O.; et al. Hypocholesterolemic properties and prebiotic effects of Mexican Ganoderma lucidum in C57BL/6 mice. PLoS ONE 2016, 11, e0159631. [Google Scholar] [CrossRef]
- Wu, Q.; Zhang, H.; Wang, P.G.; Chen, M. Evaluation of the efficacy and safety of Ganoderma lucidum mycelium-fermented liquid on gut microbiota and its impact on cardiovascular risk factors in human. Rsc Adv. 2017, 7, 45093–45100. [Google Scholar] [CrossRef]
- He, J.; Sun, Y.; Jia, Y.; Geng, X.; Chen, R.; Zhou, H.; Yang, B. Ganoderma triterpenes protect against hyperhomocysteinemia induced endothelial-mesenchymal transition via TGF-β signaling inhibition. Front. Physiol. 2019, 10, 192. [Google Scholar] [CrossRef]
- Shaher, F.; Qiu, H.; Wang, S.; Hu, Y.; Wang, W.; Zhang, Y.; Wei, Y.; AL-ward, H.; Abdulghani, M.A.M.; Alenezi, S.K.; et al. Associated targets of the antioxidant cardioprotection of Ganoderma lucidum in diabetic cardiomyopathy by using open targets platform: A systematic review. BioMed Res. Int. 2020, 2020, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Ghista, D.N.; Acharya, U.R.; Desai, K.D.; Dittakavi, S.; Adeneye, A.A.; Meng, L.K. Diabetes Mechanisms, Detection and Complications Monitoring. In Biomedical Science, Engineering and Technology; Ghista, D.N., Ed.; IntechOpen: London, UK, 2012; pp. 419–446, Chapter 17. [Google Scholar] [CrossRef]
- Awuchi, C.G.; Echeta, C.K.; Igwe, V.S. Diabetes and the nutrition and diets for its prevention and treatment: A systematic review and dietetic perspective. Health Sci. Res. 2020, 6, 5–19. [Google Scholar]
- He, L.; Yang, F.Q.; Tang, P.; Gao, T.H.; Yang, C.X.; Tan, L.; Yue, P.; Hua, Y.N.; Liu, S.J.; Guo, J.L. Regulation of the intestinal flora: A potential mechanism of natural medicines in the treatment of type 2 diabetes mellitus. Biomed. Pharmacother. 2022, 151, 113091. [Google Scholar] [CrossRef]
- IDF Diabetes around the World in 2021. 2021. Available online: https://diabetesatlas.org (accessed on 18 September 2022).
- Li, X.; Bai, Y.; Jin, Z.; Svensson, B. Food-derived nonphenolic α-amylase and α-glucosidase inhibitors for controlling starch digestion rate and guiding diabetes-friendly recipes. LWT 2022, 153, 112455. [Google Scholar] [CrossRef]
- Ma, H.T.; Hsieh, J.F.; Chen, S.T. Anti-diabetic effects of Ganoderma lucidum. Phytochemistry 2015, 114, 109–113. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Diabetes. 2020. Available online: https://www.who.int/health-topics/diabetes#tab=tab_1 (accessed on 12 October 2022).
- Seweryn, E.; Ziała, A.; Gamian, A. Health-promoting of polysaccharides extracted from Ganoderma lucidum. Nutrients 2021, 13, 2725. [Google Scholar] [CrossRef]
- Tong, A.; Wu, W.; Chen, Z.; Wen, J.; Jia, R.; Liu, B.; Cao, H.; Zhao, C. Modulation of gut microbiota and lipid metabolism in rats fed high-fat diets by Ganoderma lucidum triterpenoids. Curr. Res. Food Sci. 2022, 6, 100427. [Google Scholar] [CrossRef]
- Chen, M.; Xiao, D.; Liu, W.; Song, Y.; Zou, B.; Li, L.; Li, P.; Cai, Y.; Liu, D.; Liao, Q.; et al. Intake of Ganoderma lucidum polysaccharides reverses the disturbed gut microbiota and metabolism in type 2 diabetic rats. Int. J. Biol. Macromol. 2020, 155, 890–902. [Google Scholar] [CrossRef]
- Yamamoto, V.; Bolanos, J.F.; Fiallos, J.; Strand, S.E.; Morris, K.; Shahrokhinia, S.; Cushing, T.R.; Hopp, L.; Tiwari, A.; Hariri, R.; et al. COVID-19: Review of a 21st century pandemic from etiology to neuro-psychiatric implications. J. Alzheimer’s Dis. 2020, 77, 459–504. [Google Scholar] [CrossRef]
- Yang, Y.; Islam, M.S.; Wang, J.; Li, Y.; Chen, X. Traditional Chinese medicine in the treatment of patients infected with 2019-new coronavirus (SARS-CoV-2): A review and perspective. Int. J. Biol. Sci. 2020, 16, 1708. [Google Scholar] [CrossRef]
- Zhou, C.M.; Qin, X.R.; Yan, L.N.; Jiang, Y.; Ke, H.N.; Yu, X.J. Global trends in COVID-19. Infect. Med. 2022, 1, 31–39. [Google Scholar] [CrossRef]
- Vishwakarma, S.; Panigrahi, C.; Barua, S.; Sahoo, M.; Mandliya, S. Food nutrients as inherent sources of immunomodulation during COVID-19 pandemic. LWT 2022, 158, 113154. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). WHO Coronavirus (COVID-19) Dashboard. 2022. Available online: https://covid19.who.int (accessed on 17 November 2022).
- Liu, K.; Chen, Y.; Lin, R.; Han, K. Clinical features of COVID-19 in elderly patients: A comparison with young and middle-aged patients. J. Infect. 2020, 80, e14–e18. [Google Scholar] [CrossRef] [PubMed]
- Žontar, T.P.; Vidrih, R. Nutrition and COVID-19 epidemic. Acta Agric. Slov. 2021, 117, 1–7. [Google Scholar] [CrossRef]
- Quiles, J.L.; Rivas-García, L.; Varela-López, A.; Llopis, J.; Battino, M.; Sánchez-González, C. Do nutrients and other bioactive molecules from foods have anything to say in the treatment against COVID-19? Environ. Res. 2020, 191, 110053. [Google Scholar] [CrossRef]
- Agarwala, R.; Maria, I.J.; Dewan, P.; Rahman, M.M.; Hosen, Z.; Adnan, M. Exploring the impact of daily food habit and modification of lifestyle for boosting immunity against COVID-19. Heliyon 2022, 8, e08983. [Google Scholar] [CrossRef] [PubMed]
- Wessels, I.; Maywald, M.; Rink, L. Zinc as a gatekeeper of immune function. Nutrients 2017, 9, 1286. [Google Scholar] [CrossRef]
- Huang, Z.; Liu, Y.; Qi, G.; Brand, D.; Zheng, S.G. Role of vitamin An in the immune system. J. Clin. Med. 2018, 7, 258. [Google Scholar] [CrossRef]
- Xu, J.; Gao, L.; Liang, H.; Chen, S.D. In silico screening of potential anti–COVID-19 bioactive natural constituents from food sources by molecular docking. Nutrition 2021, 82, 111049. [Google Scholar] [CrossRef]
- Rahman, M.A.; Rahman, M.S.; Bashir, N.M.B.; Mia, R.; Hossain, A.; Saha, S.K.; Kakon, A.J.; Sarker, N.C. Rationalization of mushroom-based preventive and therapeutic approaches to COVID-19. Int. J. Med. Mushrooms 2021, 23, 1–11. [Google Scholar] [CrossRef]
- El, O. Possible use of Ganoderma mushroom extracts for the treatment of some chronic risk factors associated with severe COVID-19. Virol. Immunol. J. 2020, 4, 000254. [Google Scholar] [CrossRef]
- Al-Jumaili, M.M.O.; Al-dulaimi, F.K.; Ajeel, M.A. The role of Ganoderma lucidum uptake on some hematological and immunological response in patients with coronavirus (COVID-19). Sys. Rev. Pharm. 2020, 11, 537–541. [Google Scholar]
- Watchel-Galor, S.; Yuen, J.; Buswell, J.A.; Benzie, I.F.F. Ganoderma lucidum (Lingzhi or Reishi). A medicinal mushroom. In Herbal Medicine: Biomolecular and Clinical Aspects, 2nd ed.; CRC Press Taylor & Francis: Boca Raton, FL, USA, 2011; Chapter 9. Available online: https://www.ncbi.nlm.nih.gov/books/NBK92757/ (accessed on 10 March 2023).

| Color | Taste | Japanese Name | Usage Area | Reference |
|---|---|---|---|---|
| Blue | Sour | Aoshiba | Improves nervous system, eyesight and liver functions | Wasser [3] |
| Red | Bitter | Akashiba | Helps internal organs, improves memory, increases vitality | |
| Yellow | Sweet | Kishiba | Strengthens spleen function, calms the soul | |
| White | Bitter | Shiroshiba | Improves lung function, gives courage and strong will | |
| Black | Salty | Kuroshiba | Protects the kidneys | |
| Purple | Sweet | Murasakishiba | Improves the function of ears, joints, muscles, helps skin |
| G. lucidum Compounds | Anticancer Mechanism of Action | Extract/Solvent/Extraction Method | Reference |
|---|---|---|---|
| Polysaccharides |
| Ethanol precipitation method/anion exchange chromatography | Lu et al. [23]; Sliva [47] |
Ganoderic acid T |
| Ethyl acetate/reversed phase high performance liquid chromatography (HPLC) | Ahmad [15]; Sliva [47] |
Ganoderic acid DM |
| Ethanol, methanol, ether, ethanol-water, and ethyl acetate | Ahmad [15] |
Ganoderic acid C |
| Ethanol extracts | Ahmad [15] |
Ganoderic acid H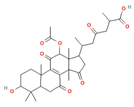 |
| Ahmad [15] | |
Ganoderic acid A |
| Ahmad [15] | |
Ganoderic acid y (GA-y)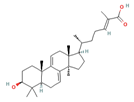 Ganoderic acid F (GA-F) 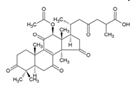 Ganoderic acid ε (GA-ε) 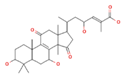 |
| Ahmad [15]; Sliva [47] | |
Ganoderic acid Me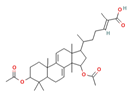 |
| Methanol extract | Ahmad [15] Wang et al. [50] |
Lucidenic acid A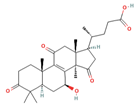 Lucidenic acid B  Lucidenic acid C  Lucidenic acid N  |
| Reversed phase high performance liquid chromatography (HPLC) | Ahmad [15]; Sliva [47] Weng et al. [51] |
Ganoderiol A Ganoderiol B  Ganoderiol F  Lucidumol B  Ganodermatriol 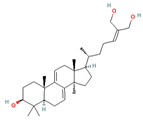 Ganodermanontriol (GDNT)  |
| Methanol/HPLC | Ahmad [15]; Sliva [47] Min et al. [52] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ekiz, E.; Oz, E.; Abd El-Aty, A.M.; Proestos, C.; Brennan, C.; Zeng, M.; Tomasevic, I.; Elobeid, T.; Çadırcı, K.; Bayrak, M.; et al. Exploring the Potential Medicinal Benefits of Ganoderma lucidum: From Metabolic Disorders to Coronavirus Infections. Foods 2023, 12, 1512. https://doi.org/10.3390/foods12071512
Ekiz E, Oz E, Abd El-Aty AM, Proestos C, Brennan C, Zeng M, Tomasevic I, Elobeid T, Çadırcı K, Bayrak M, et al. Exploring the Potential Medicinal Benefits of Ganoderma lucidum: From Metabolic Disorders to Coronavirus Infections. Foods. 2023; 12(7):1512. https://doi.org/10.3390/foods12071512
Chicago/Turabian StyleEkiz, Elif, Emel Oz, A. M. Abd El-Aty, Charalampos Proestos, Charles Brennan, Maomao Zeng, Igor Tomasevic, Tahra Elobeid, Kenan Çadırcı, Muharrem Bayrak, and et al. 2023. "Exploring the Potential Medicinal Benefits of Ganoderma lucidum: From Metabolic Disorders to Coronavirus Infections" Foods 12, no. 7: 1512. https://doi.org/10.3390/foods12071512
APA StyleEkiz, E., Oz, E., Abd El-Aty, A. M., Proestos, C., Brennan, C., Zeng, M., Tomasevic, I., Elobeid, T., Çadırcı, K., Bayrak, M., & Oz, F. (2023). Exploring the Potential Medicinal Benefits of Ganoderma lucidum: From Metabolic Disorders to Coronavirus Infections. Foods, 12(7), 1512. https://doi.org/10.3390/foods12071512