Carotenoid and Tocopherol Profiling in 18 Korean Traditional Green Leafy Vegetables by LC-SIM-MS
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents, Standards, and Plant Materials
2.2. Extraction of Carotenoids and Tocols
2.3. LC-MS Analysis
2.4. Calculation of Vitamin A Activity
2.5. Statistical Evaluation and Quality Assurance
3. Results and Discussion
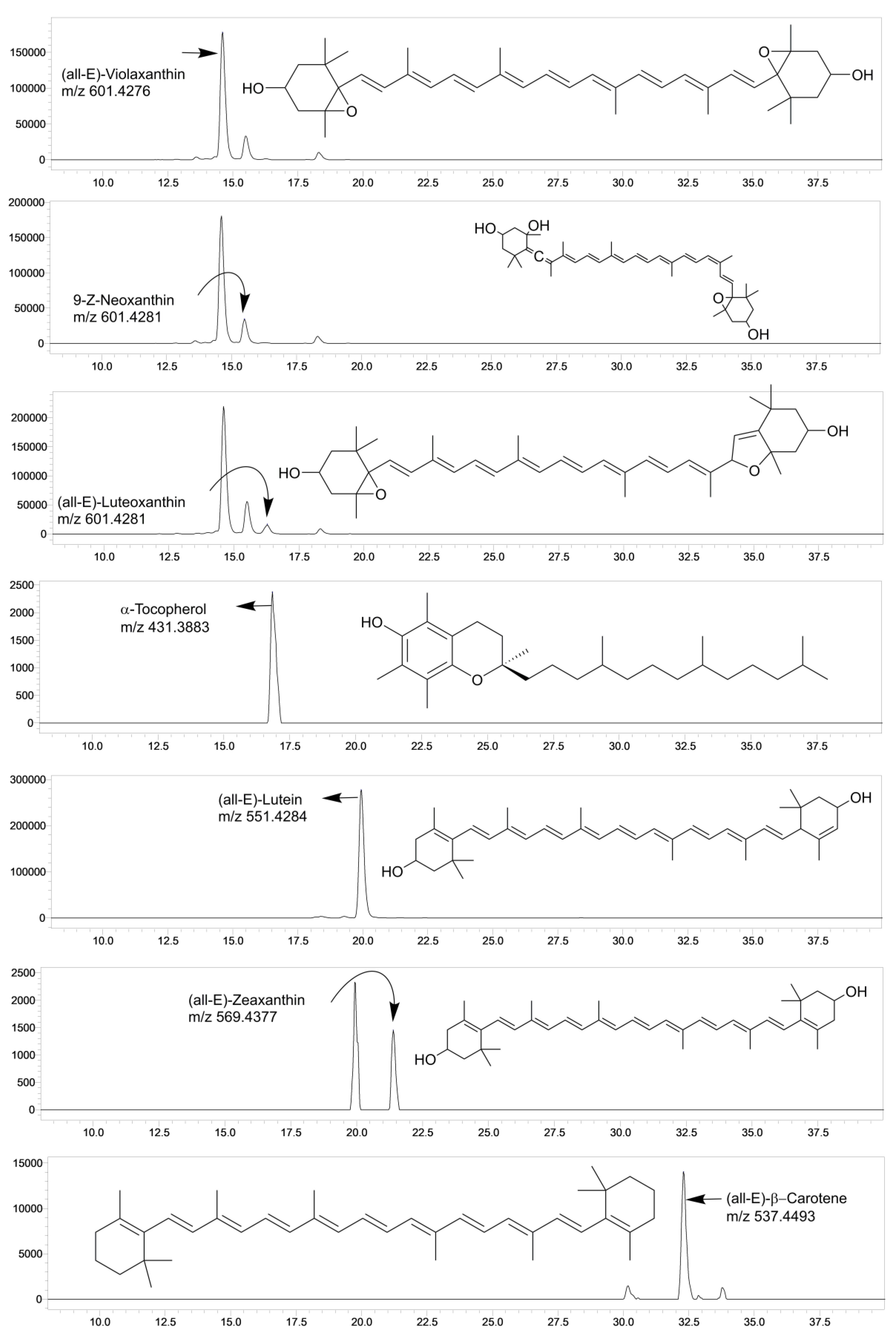
3.1. Validation of LC-MS/SIM Methodology
3.2. Carotenoid Composition
3.3. Tocols Composition
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Compounds | Working Range (μg/mL) | Area Counts Precision | Retention Time Precision | Limits | Correlation Coefficient (R2) | |||
|---|---|---|---|---|---|---|---|---|
| Compounds | Intra-Day CV (%, n = 6) | Inter-Day CV (%, n = 6 × 2) | Intra-Day CV (%, n = 6) | Inter-Day CV (%, n = 6 × 2) | LOQ (μg/g) | LOD (μg/g) | ||
| (all-E)-violaxanthin | 5–50 | 4.11 | 5.30 | 0.07 | 0.15 | 0.32 | 0.11 | 0.999 |
| 9-Z-neoxanthin | 5–50 | 7.01 | 7.42 | 0.09 | 0.17 | 0.84 | 0.28 | 0.999 |
| (all-E)-lactucaxanthin | 5–50 | 4.68 | 5.43 | 0.08 | 0.18 | 0.87 | 0.29 | 1.000 |
| (all-E)-lutein | 5–50 | 2.53 | 4.98 | 0.07 | 0.16 | 0.15 | 0.05 | 0.999 |
| (all-E)-zeaxanthin | 5–50 | 5.12 | 7.06 | 0.09 | 0.14 | 0.46 | 0.15 | 1.000 |
| (all-E)-β-carotene | 5–50 | 4.91 | 6.05 | 0.08 | 0.17 | 0.68 | 0.23 | 1.000 |
| α-tocopherol | 10–100 | 8.82 | 9.23 | 0.23 | 0.35 | 2.94 | 0.97 | 0.999 |
References
- World Health Organization. Diet, Nutrition, and the Prevention of Chronic Diseases: Report of a Joint WHO/FAO Expert Consultation; World Health Organization: Geneva, Switzerland, 2003; Volume 916. [Google Scholar]
- Wang, P.Y.; Fang, J.C.; Gao, Z.H.; Zhang, C.; Xie, S.Y. Higher intake of fruits, vegetables or their fiber reduces the risk of type 2 diabetes: A meta-analysis. J. Diabetes Investig. 2016, 7, 56–69. [Google Scholar] [CrossRef]
- Aune, D.; Giovannucci, E.; Boffetta, P.; Fadnes, L.T.; Keum, N.; Norat, T.; Greenwood, D.C.; Riboli, E.; Vatten, L.J.; Tonstad, S. Fruit and vegetable intake and the risk of cardiovascular disease, total cancer and all-cause mortality-a systematic review and dose-response meta-analysis of prospective studies. Int. J. Epidemiol. 2017, 46, 1029–1056. [Google Scholar] [CrossRef]
- Medina-Remón, A.; Kirwan, R.; Lamuela-Raventós, R.M.; Estruch, R. Dietary patterns and the risk of obesity, type 2 diabetes mellitus, cardiovascular diseases, asthma, and neurodegenerative diseases. Crit. Rev. Food Sci. Nutr. 2018, 58, 262–296. [Google Scholar] [CrossRef] [PubMed]
- Kaulmann, A.; Bohn, T. Carotenoids, inflammation, and oxidative stress—Implications of cellular signaling pathways and relation to chronic disease prevention. Nutr. Res. 2014, 34, 907–929. [Google Scholar] [CrossRef] [PubMed]
- Hajizadeh-Sharafabad, F.; Ghoreishi, Z.; Maleki, V.; Tarighat-Esfanjani, A. Mechanistic insights into the effect of lutein on atherosclerosis, vascular dysfunction, and related risk factors: A systematic review of in vivo, ex vivo and in vitro studies. Pharmacol. Res. 2019, 149, 104477. [Google Scholar] [CrossRef]
- Saini, R.K.; Rengasamy, K.R.R.; Mahomoodally, F.M.; Keum, Y.S. Protective effects of lycopene in cancer, cardiovascular, and neurodegenerative diseases: An update on epidemiological and mechanistic perspectives. Pharmacol. Res. 2020, 155, 104730. [Google Scholar] [CrossRef]
- Elvira-Torales, L.I.; Garcia-Alonso, J.; Periago-Caston, M.J. Nutritional Importance of Carotenoids and Their Effect on Liver Health: A Review. Antioxidants 2019, 8, 229. [Google Scholar] [CrossRef]
- Saini, R.K.; Keum, Y.S. Tocopherols and tocotrienols in plants and their products: A review on methods of extraction, chromatographic separation, and detection. Food Res. Int. 2016, 82, 59–70. [Google Scholar] [CrossRef]
- Marti, R.; Rosello, S.; Cebolla-Cornejo, J. Tomato as a Source of Carotenoids and Polyphenols Targeted to Cancer Prevention. Cancers 2016, 8, 58. [Google Scholar] [CrossRef]
- Koklesova, L.; Liskova, A.; Samec, M.; Buhrmann, C.; Samuel, S.M.; Varghese, E.; Ashrafizadeh, M.; Najafi, M.; Shakibaei, M.; Busselberg, D.; et al. Carotenoids in Cancer Apoptosis-The Road from Bench to Bedside and Back. Cancers 2020, 12, 2425. [Google Scholar] [CrossRef] [PubMed]
- Saini, R.K.; Prasad, P.; Lokesh, V.; Shang, X.M.; Shin, J.; Keum, Y.S.; Lee, J.H. Carotenoids: Dietary Sources, Extraction, Encapsulation, Bioavailability, and Health Benefits-A Review of Recent Advancements. Antioxidants 2022, 11, 795. [Google Scholar] [CrossRef]
- Monsen, E.R. Dietary reference intakes for the antioxidant nutrients: Vitamin C, vitamin E, selenium, and carotenoids. J. Acad. Nutr. Diet. 2000, 100, 637. [Google Scholar]
- Saini, R.K.; Keum, Y.S.; Daglia, M.; Rengasamy, K.R. Dietary carotenoids in cancer chemoprevention and chemotherapy: A review of emerging evidence. Pharmacol. Res. 2020, 157, 104830. [Google Scholar] [CrossRef] [PubMed]
- Chae, H.S.; Lee, S.H.; Jeong, H.S.; Kim, W.J. Antioxidant activity and physicochemical characteristics of Pimpinella brachycarpa Nakai with treatments methods. Korean J. Food Nutr. 2013, 26, 125–131. [Google Scholar] [CrossRef]
- Cho, Y.-O. Antioxidative Activity of the Korean Wild Leafy Vegetables: Aster scarer mild Ligularia fischeri. Prev. Nutr. Food Sci. 2002, 7, 146–150. [Google Scholar] [CrossRef]
- Duan, L.; Zhang, C.; Zhao, Y.; Chang, Y.; Guo, L. Comparison of Bioactive Phenolic Compounds and Antioxidant Activities of Different Parts of Taraxacum mongolicum. Molecules 2020, 25, 3260. [Google Scholar] [CrossRef]
- Hiemori-Kondo, M. Antioxidant compounds of Petasites japonicus and their preventive effects in chronic diseases: A review. J. Clin. Biochem. Nutr. 2020, 67, 10–18. [Google Scholar] [CrossRef]
- Jang, S.J.; Park, H.H.; Kuk, Y.I. Growth Promotion, Nutrition Levels, and Antioxidant Activity in Peucedanum japonicum Thunb. under Various Plant Extracts. Agronomy 2020, 10, 1494. [Google Scholar] [CrossRef]
- Kim, T.H.; Truong, V.L.; Jeong, W.S. Phytochemical Composition and Antioxidant and Anti-Inflammatory Activities of Ligularia fischeri Turcz: A Comparison between Leaf and Root Extracts. Plants 2022, 11, 3005. [Google Scholar] [CrossRef]
- Ng, X.; Chye, F.; Ismail, M.A. Nutritional profile and antioxidative properties of selected tropical wild vegetables. Int. Food Res. J. 2012, 19, 1487. [Google Scholar]
- Ozsoy, N.; Yilmaz, T.; Kurt, O.; Can, A.; Yanardag, R. In vitro antioxidant activity of Amaranthus lividus L. Food Chem. 2009, 116, 867–872. [Google Scholar] [CrossRef]
- Park, C.; Jeong, J.S.; Jeong, J.W.; Kim, S.O.; Kim, Y.J.; Kim, G.Y.; Hong, S.H.; Choi, Y.H. Ethanol extract of Kalopanax septemlobus leaf inhibits HepG2 human hepatocellular carcinoma cell proliferation via inducing cell cycle arrest at G1 phase. Asian Pac. J. Trop. Med. 2016, 9, 344–350. [Google Scholar] [CrossRef] [PubMed]
- Park, H.J.; Park, S.H. Hexane fraction of Adenophora triphylla var. japonica root extract induces apoptosis of human lung cancer cells by inactivating Src/STAT3 pathway. Nat. Prod. Res. 2022, 1–5. [Google Scholar] [CrossRef]
- Zhang, J.; Li, L.; Jiang, C.; Xing, C.; Kim, S.H.; Lu, J. Anti-cancer and other bioactivities of Korean Angelica gigas Nakai (AGN) and its major pyranocoumarin compounds. Anti-Cancer. Agents Med. Chem. 2012, 12, 1239–1254. [Google Scholar] [CrossRef]
- Zhao, Q.; Zhong, X.L.; Zhu, S.H.; Wang, K.; Tan, G.F.; Meng, P.H.; Zhang, J. Research Advances in Toona sinensis, a Traditional Chinese Medicinal Plant and Popular Vegetable in China. Diversity 2022, 14, 572. [Google Scholar] [CrossRef]
- Huang, G.J.; Deng, J.S.; Liao, J.C.; Hou, W.C.; Wang, S.Y.; Sung, P.J.; Kuo, Y.H. Inducible nitric oxide synthase and cyclooxygenase-2 participate in anti-inflammatory activity of imperatorin from Glehnia littoralis. J. Agric. Food Chem. 2012, 60, 1673–1681. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.; Jung, H.A.; Choi, J.S.; Kim, Y.S.; Kang, S.S.; Kim, H.P. Anti-inflammatory activity of the constituents of the roots of Aralia continentalis. Arch. Pharm. Res. 2009, 32, 1237–1243. [Google Scholar] [CrossRef]
- Alam, M.B.; Seo, B.J.; Zhao, P.; Lee, S.H. Anti-Melanogenic Activities of Heracleum moellendorffii via ERK1/2-Mediated MITF Downregulation. Int. J. Mol. Sci. 2016, 17, 1844. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.-H.; An, B.-J.; Kim, S.-G.; Park, T.-S.; Park, G.-H.; Son, J.-H. Antimelanogenic effect of Ligularia fischeri, Solidago virga-aurea, Aruncus dioicus extracts from Ullung island in murine melanoma cells. J. Life Sci. 2011, 21, 279–285. [Google Scholar] [CrossRef]
- Yang, D.K.; Lee, S.J.; Adam, G.O.; Kim, S.J. Aralia continentalis kitagawa Extract Attenuates the Fatigue Induced by Exhaustive Exercise through Inhibition of Oxidative Stress. Antioxidants 2020, 9, 379. [Google Scholar] [CrossRef]
- Oh, J.H.; Karadeniz, F.; Lee, J.I.; Seo, Y.; Kong, C.S. Artemisia princeps Inhibits Adipogenic Differentiation of 3T3-L1 Pre-Adipocytes via Downregulation of PPARgamma and MAPK Pathways. Prev. Nutr. Food Sci. 2019, 24, 299–307. [Google Scholar] [CrossRef] [PubMed]
- Nam, G.H.; Wee, J.-H.; Kim, S.Y.; Baek, J.-Y.; Kim, Y.M. Inhibitory Effect of the Ethanol Extract of Rudbeckia laciniata var. hortensis Bailey on Adipocyte Differentiation in 3T3-L1 Cells. J. Life Sci. 2019, 29, 1152–1158. [Google Scholar]
- Shin, S.; Saravanakumar, K.; Sathiyaseelan, A.; Mariadoss, A.V.A.; Park, S.; Park, S.; Han, K.; Wang, M.H. Phytochemical profile and antidiabetic effect of the bioactive fraction of Cirsium setidens in streptozotocin-induced type 2 diabetic mice. Process Biochem. 2022, 116, 60–71. [Google Scholar] [CrossRef]
- Son, H.J.; Eo, H.J.; Park, G.H.; Jeong, J.B. Heracleum moellendorffii root extracts exert immunostimulatory activity through TLR2/4-dependent MAPK activation in mouse macrophages, RAW264.7 cells. Food Sci. Nutr. 2021, 9, 514–521. [Google Scholar] [CrossRef]
- Cheng, K.W.; Yang, R.Y.; Tsou, S.C.S.; Lo, C.S.C.; Ho, C.T.; Lee, T.C.; Wang, M.F. Analysis of antioxidant activity and antioxidant constituents of Chinese toon. J. Funct. Foods 2009, 1, 253–259. [Google Scholar] [CrossRef]
- Han, G.-J.; Shin, D.-S.; Jang, M.-S. A study of the nutritional composition of Aralia continentalis Kitagawa and Aralia continentalis Kitagawa leaf. Korean J. Food Sci. Technol. 2008, 40, 680–685. [Google Scholar]
- Jin, Y.-X.; Cho, Y.-S.; Choi, Y. Nutritional quality of Peucedanum japonicum Thunb. leaves in relation to ripening time, growing condition and blanching. Korean J. Food Preserv. 2014, 21, 784–789. [Google Scholar] [CrossRef]
- Raju, M.; Varakumar, S.; Lakshminarayana, R.; Krishnakantha, T.P.; Baskaran, V. Carotenoid composition and vitamin A activity of medicinally important green leafy vegetables. Food Chem. 2007, 101, 1598–1605. [Google Scholar] [CrossRef]
- Sathasivam, R.; Kim, N.S.; Choi, M.; Kwon, H.; Nguyen, B.V.; Kim, J.K.; Jeong, D.H.; Park, E.J.; Park, H.W.; Park, S.U. Identification, In Silico Characterization, and Differential Expression Profiles of Carotenoid, Xanthophyll, Apocarotenoid Biosynthetic Pathways Genes, and Analysis of Carotenoid and Xanthophyll Accumulation in Heracleum moellendorffii Hance. Int. J. Mol. Sci. 2022, 23, 4845. [Google Scholar] [CrossRef]
- Saini, R.K.; Moon, S.H.; Gansukh, E.; Keum, Y.S. An efficient one-step scheme for the purification of major xanthophyll carotenoids from lettuce, and assessment of their comparative anticancer potential. Food Chem. 2018, 266, 56–65. [Google Scholar] [CrossRef]
- Crupi, P.; Milella, R.A.; Antonacci, D. Simultaneous HPLC-DAD-MS (ESI+) determination of structural and geometrical isomers of carotenoids in mature grapes. J. Mass Spectrom. 2010, 45, 971–980. [Google Scholar] [CrossRef]
- Saini, R.K.; Song, M.H.; Yu, J.W.; Lee, J.H.; Ahn, H.Y.; Keum, Y.S.; Lee, J.H. Profiling of Nutritionally Vital Bioactive Compounds in Emerging Green Leafy Vegetables: A Comparative Study. Foods 2022, 11, 3867. [Google Scholar] [CrossRef]
- Saini, R.K.; Keum, Y.-S. Carotenoid extraction methods: A review of recent developments. Food Chem. 2018, 240, 90–103. [Google Scholar] [CrossRef]
- Cruz, R.; Casal, S.; Mendes, E.; Costa, A.; Santos, C.; Morais, S. Validation of a Single-Extraction Procedure for Sequential Analysis of Vitamin E, Cholesterol, Fatty Acids, and Total Fat in Seafood. Food Anal. Methods 2013, 6, 1196–1204. [Google Scholar] [CrossRef]
- Institute of Medicine. Panel on, M. In Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc; National Academies Press: Washington, DC, USA, 2001. [Google Scholar] [CrossRef]
- De Bièvre, P.; Günzler, H. Validation in Chemical Measurement; Springer: Berlin, Germany, 2005. [Google Scholar]
- ICH. Q2 (R1): Validation of analytical procedures: Text and methodology. In Proceedings of the International Conference on Harmonization, Geneva, Switzerland, 2 June 2014. [Google Scholar]
- Taverniers, I.; De Loose, M.; Van Bockstaele, E. Trends in quality in the analytical laboratory. II. Analytical method validation and quality assurance. TrAC Trends Anal. Chem. 2004, 23, 535–552. [Google Scholar] [CrossRef]
- Saini, R.K.; Nile, S.H.; Park, S.W. Carotenoids from fruits and vegetables: Chemistry, analysis, occurrence, bioavailability and biological activities. Food Res. Int. 2015, 76, 735–750. [Google Scholar] [CrossRef] [PubMed]
- Yoon, G.A.; Yeum, K.J.; Cho, Y.S.; Chen, C.Y.; Tang, G.; Blumberg, J.B.; Russell, R.M.; Yoon, S.; Lee-Kim, Y.C. Carotenoids and total phenolic contents in plant foods commonly consumed in Korea. Nutr. Res. Pract. 2012, 6, 481–490. [Google Scholar] [CrossRef] [PubMed]
- Kao, T.H.; Loh, C.H.; Inbaraj, B.S.; Chen, B.H. Determination of carotenoids in Taraxacum formosanum by HPLC-DAD-APCI-MS and preparation by column chromatography. J. Pharm. Biomed. Anal. 2012, 66, 144–153. [Google Scholar] [CrossRef]
- Hu, C. Taraxacum: Phytochemistry and health benefits. Chin. Herb. Med. 2018, 10, 353–361. [Google Scholar] [CrossRef]
- Zhang, Y.; Hu, Y.F.; Li, W.; Xu, G.Y.; Wang, K.R.; Li, L.; Luo, H.; Zou, L.; Wu, J.S. Updates and advances on pharmacological properties of Taraxacum mongolicum Hand.-Mazz and its potential applications. Food Chem. 2022, 373, 131380. [Google Scholar] [CrossRef]
- Wilson, L.M.; Tharmarajah, S.; Jia, Y.; Semba, R.D.; Schaumberg, D.A.; Robinson, K.A. The Effect of Lutein/Zeaxanthin Intake on Human Macular Pigment Optical Density: A Systematic Review and Meta-Analysis. Adv. Nutr. 2021, 12, 2244–2254. [Google Scholar] [CrossRef] [PubMed]
- Saini, R.K.; Shang, X.M.; Ko, E.Y.; Choi, J.H.; Kim, D.; Keum, Y.S. Characterization of nutritionally important phytoconstituents in minimally processed ready-to-eat baby-leaf vegetables using HPLC-DAD and GC-MS. J. Food Meas. Charact. 2016, 10, 341–349. [Google Scholar] [CrossRef]
- Saini, R.K.; Keum, Y.S.; Rengasamy, K.R.R. Profiling of nutritionally important metabolites in green/red and green perilla (Perilla frutescens Britt.) cultivars: A comparative study. Ind. Crop. Prod. 2020, 151, 112441. [Google Scholar] [CrossRef]
- Kim, D.E.; Shang, X.; Assefa, A.D.; Keum, Y.S.; Saini, R.K. Metabolite profiling of green, green/red, and red lettuce cultivars: Variation in health beneficial compounds and antioxidant potential. Food Res. Int. 2018, 105, 361–370. [Google Scholar] [CrossRef]
- Wen Lee, H.; Bi, X.; Jeyakumar Henry, C. Carotenoids, tocopherols and phylloquinone content of 26 green leafy vegetables commonly consumed in Southeast Asia. Food Chem. 2022, 385, 132729. [Google Scholar] [CrossRef]
- Comitato, R.; Ambra, R.; Virgili, F. Tocotrienols: A Family of Molecules with Specific Biological Activities. Antioxidants 2017, 6, 93. [Google Scholar] [CrossRef]
- Ching, L.S.; Mohamed, S. Alpha-tocopherol content in 62 edible tropical plants. J. Agric. Food Chem. 2001, 49, 3101–3105. [Google Scholar] [CrossRef]
- Saini, R.K.; Yu, J.-W.; Song, M.-H.; Ahn, H.-Y.; Lee, J.-H.; Keum, Y.-S.; Lee, J.-H. Profiling of Redox-Active Lipophilic Constituents in Leaf Mustard (Brassica juncea (L.) Czern.) Cultivars Using LC–MS and GC–MS. Antioxidants 2022, 11, 2464. [Google Scholar] [CrossRef]
- Lewis, E.D.; Meydani, S.N.; Wu, D. Regulatory role of vitamin E in the immune system and inflammation. IUBMB Life 2019, 71, 487–494. [Google Scholar] [CrossRef]

| S/No. | Family | Scientific Name | Local Name | Place of Collection |
|---|---|---|---|---|
| 1 | Amaranthaceae | Amaranthus lividus L. | Chambireum | a |
| 2 | Apiaceae | Angelica gigas Nakai | Chamdangwi | a |
| 3 | Glehnia littoralis F. Schmidt ex Miq. | Haedangpung | a | |
| 4 | Heracleum moellendorffii Hance | Uhsuri | a | |
| 5 | Peucedanum japonicum Thunb. | Gatgireum | a | |
| 6 | Pimpinella brachycarpa (Kom.) Nakai | Chamnamul | a | |
| 7 | Araliaceae | Aralia continentalis Kitag. | Ttangdureup | a |
| 8 | Kalopanax septemlobus (Thunb. ex A.Murr.) Koidz. | Eomnamu | c | |
| 9 | Asteraceae | Artemisia princeps Pamp. | Suk | a |
| 10 | Cirsium setidens Nakai | Gondre | b | |
| 11 | Ligularia fischeri (Ledeb.) Turcz. | Gomchwi | a | |
| 12 | Petasites japonicus (Siebold & Zucc.) Maxim. | Mowi | b | |
| 13 | Rudbeckia laciniata L. | Samipgukhwa | b | |
| 14 | Solidago virga-aurea L. var. asictica Nakai | Miyeokchwi | a | |
| 15 | Taraxacum mongolicum Hand.-Mazz. | Mindle | a | |
| 16 | Campanulaceae | Adenophora triphylla (Thunb.) A.DC. var. japonica (Regel) H. Hara | Jandae | a |
| 17 | Liliaceae | Allium victorialis var. platyphyllum Makino | Sanmaneul | d |
| 18 | Meliaceae | Toona sinensis (A.Juss.) M.Roem. | Chamjuknamu | a |
| Class of Compounds | Compound | Transition (m/z) * |
|---|---|---|
| Carotenoids | (all-E)-β-carotene; β,β-carotene | 537.4493 |
| (all-E)-zeaxanthin; β,β-carotene-3,3′-diol | 569.4377 | |
| (all-E)-lutein; β,ε-Carotene-3,3′-diol | 551.4284 | |
| (all-E)-luteoxanthin; β-carotene-3,3′-diol, 5,6:5′,8′-diepoxy-5,5′,6,8′-tetrahydro- | 601.4281 | |
| 9-Z-neoxanthin; 5′,6′-epoxy-6,7-didehydro-5,6,5′,6′-tetrahydro-β,β-carotene-3,5,3′-triol | 601.4281 | |
| (all-E)-violaxanthin; 5,6,5′,6′-diepoxy-5,6,5′,6′-tetrahydro-β,β-carotene-3,3′-diol) | 601.4276 | |
Tocopherols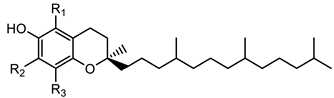 | α- tocopherol (R1 = R2 = R3 = CH3) | 431.3883 |
| β-tocopherol (R1 = R3 = CH3; R2 = H) | 416.3669 | |
| γ-tocopherol (R1 = H; R2 = R3 = CH3) | 416.3669 | |
| δ-tocopherol (R1 = R2 = H; R3 = CH3) | 402.3488 | |
Tocotrienols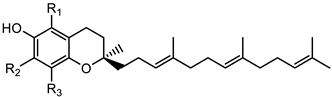 | α- tocotrienol (R1 = R2 = R3 = CH3) | 425.3423 |
| β-tocotrienol (R1 = R3 = CH3; R2 = H) | 411.3268 | |
| γ-tocotrienol (R1 = H; R2 = R3 = CH3) | 411.3268 | |
| δ-tocotrienol (R1 = R2 = H; R3 = CH3) | 397.3113 |
| S/No. | (all-E)-Violaxanthin | 9-Z-Neoxanthin | (all-E)-Luteoxanthin | (all-E)-Lutein | (all-E)-Zeaxanthin | Total Xanthophylls |
|---|---|---|---|---|---|---|
| 1 | 47.7 ± 2.11 | 27.0 ± 3.38 | 1.17 ± 0.11 | 113.5 ± 5.08 | 0.49 ± 0.01 | 189.9 ± 10.4 |
| 2 | 64.3 ± 5.30 | 29.0 ± 1.51 | 0.74 ± 0.08 | 138.7 ± 5.78 | 1.25 ± 0.22 | 233.9 ± 12.89 |
| 3 | 67.2 ± 1.11 | 30.1 ± 0.79 | 2.66 ± 0.54 | 130.9 ± 0.63 | 2.27 ± 0.50 | 233.0 ± 1.23 |
| 4 | 35.8 ± 4.4 | 22.1 ± 1.75 | 2.10 ± 0.56 | 111.6 ± 6.0 | 0.35 ± 0.03 | 171.9 ± 12.7 |
| 5 | 43.9 ± 0.35 | 23.2 ± 0.62 | 2.53 ± 0.86 | 100.3 ± 0.73 | 0.39 ± 0.05 | 170.3 ± 2.61 |
| 6 | 75.6 ± 5.95 a | 48.4 ± 2.37 a | 10.8 ± 1.14 a | 174.1 ± 6.51 a | 1.64 ± 0.12 | 310.5 ± 13.57 a |
| 7 | 43.9 ± 4.33 | 16.5 ± 2.14 | 1.85 ± 0.20 | 96.5 ± 3.34 | 1.02 ± 0.07 | 159.8 ± 10.1 |
| 8 | 54.2 ± 5.64 | 17.3 ± 2.58 | 1.33 ± 0.03 | 97.9 ± 3.83 | 0.39 ± 0.12 | 171.1 ± 12.2 |
| 9 | 66.3 ± 1.05 | 31.6 ± 1.47 | 1.89 ± 0.34 | 128.6 ± 4.38 | 1.85 ± 0.13 | 230.3 ± 6.43 |
| 10 | 49.2 ± 4.41 | 23.2 ± 0.61 | 3.74 ± 1.64 | 112.2 ± 5.76 | 2.17 ± 0.35 | 190.5 ± 12.1 |
| 11 | 67.2 ± 6.14 | 29.6 ± 2.43 | 1.31 ± 0.17 | 130.0 ± 9.38 | 1.12 ± 0.06 | 229.3 ± 17.9 |
| 12 | 48.8 ± 0.99 | 24.5 ± 0.49 | 2.624 ± 1.17 | 110.8 ± 0.50 | 1.02 ± 0.28 | 187.8 ± 1.45 |
| 13 | 70.4 ± 2.04 | 27.8 ± 2.27 | 1.68 ± 0.19 | 139.3 ± 10.5 | 1.76 ± 0.10 | 240.9 ± 14.7 |
| 14 | 61.8 ± 4.02 | 21.5 ± 2.17 | 1.23 ± 0.07 | 120.6 ± 4.54 | 14.44 ± 0.07 a | 219.6 ± 10.9 |
| 15 | 75.1 ± 2.44 a | 41.2 ± 0.94 | 2.04 ± 0.31 | 163.7 ± 4.95 a | 0.94 ± 0.28 | 283.0 ± 7.05 |
| 16 | 75.3 ± 1.47 a | 22.9 ± 1.37 | 1.19 ± 1.18 | 126.4 ± 3.81 | 3.51 ± 0.13 | 229.3 ± 7.70 |
| 17 | 49.0 ± 7.07 | 22.5 ± 1.13 | 1.39 ± 0.49 | 100.5 ± 4.77 | 0.63 ± 0.18 | 174.1 ± 12.7 |
| 18 | 59.1 ± 0.77 | 18.1 ± 0.09 | 7.97 ± 3.21 | 107.9 ± 5.95 | 2.53 ± 0.21 | 195.6 ± 10.2 |
| S/No. | (all-E)-β-Carotene * | Total Carotenoids (TC) * | % (all-E)-Lutein in TC | % (all-E)-β-Carotene in TC | RAE (µg) | % DRI from 100 g ** |
|---|---|---|---|---|---|---|
| 1 | 57.9 ± 4.75 | 247.8 ± 15.2 | 45.8 ± 0.76 | 23.4 ± 0.49 a | 4.83 ± 0.40 | 53.6 ± 4.40 |
| 2 | 46.0 ± 0.16 | 280.0 ± 13.0 | 49.5 ± 0.24 | 16.5 ± 0.71 | 3.83 ± 0.01 | 42.6 ± 0.14 |
| 3 | 61.6 ± 0.46 | 294.6 ± 1.69 | 44.4 ± 0.47 | 20.9 ± 0.04 | 5.13 ± 0.04 | 57.0 ± 0.43 |
| 4 | 42.5 ± 4.6 | 214.5 ± 17.2 | 52.1 ± 1.41 a | 19.8 ± 0.55 | 3.55 ± 0.38 | 39.4 ± 4.25 |
| 5 | 37.2 ± 0.44 | 207.5 ± 3.04 | 48.4 ± 0.36 | 17.9 ± 0.05 | 3.10 ± 0.04 | 34.4 ± 0.40 |
| 6 | 69.6 ± 3.92 a | 380.1 ± 17.5 a | 45.8 ± 0.40 | 18.3 ± 0.19 | 5.80 ± 0.33 a | 64.4 ± 3.63 a |
| 7 | 33.5 ± 0.47 | 193.3 ± 10.5 | 50.0 ± 1.00 | 17.4 ± 0.71 | 2.80 ± 0.04 | 31.1 ± 0.43 |
| 8 | 26.8 ± 1.86 | 197.9 ± 14.05 | 49.5 ± 1.58 | 13.5 ± 0.02 | 2.23 ± 0.15 | 24.8 ± 1.72 |
| 9 | 53.6 ± 0.53 | 283.9 ± 5.90 | 45.3 ± 0.60 | 18.9 ± 0.58 | 4.47 ± 0.04 | 49.7 ± 0.49 |
| 10 | 39.1 ± 3.68 | 229.6 ± 15.7 | 48.9 ± 0.85 | 17.0 ± 0.44 | 3.26 ± 0.31 | 36.2 ± 3.41 |
| 11 | 47.7 ± 1.98 | 277.0 ± 19.8 | 46.9 ± 0.03 | 17.3 ± 0.52 | 3.98 ± 0.17 | 44.2 ± 1.84 |
| 12 | 46.7 ± 1.94 | 234.5 ± 0.49 | 47.3 ± 0.12 | 19.9 ± 0.79 | 3.890 ± 0.16 | 43.2 ± 1.80 |
| 13 | 59.3 ± 2.58 | 300.2 ± 17.3 | 46.4 ± 0.83 | 19.8 ± 0.28 | 4.94 ± 0.22 | 54.9 ± 2.39 |
| 14 | 42.1 ± 3.17 | 261.7 ± 14.0 | 46.1 ± 0.74 | 16.1 ± 0.35 | 3.51 ± 0.26 | 39.0 ± 2.93 |
| 15 | 65.4 ± 1.27 | 348.4 ± 8.31 a | 47.0 ± 0.30 | 18.8 ± 0.08 | 5.45 ± 0.11 | 60.5 ± 1.17 |
| 16 | 46.3 ± 1.29 | 275.6 ± 8.99 | 45.9 ± 0.11 | 16.8 ± 0.08 | 3.86 ± 0.11 | 42.9 ± 1.19 |
| 17 | 43.0 ± 0.69 | 217.0 ± 13.3 | 46.3 ± 0.65 | 19.8 ± 0.90 | 3.58 ± 0.06 | 39.8 ± 0.64 |
| 18 | 31.1 ± 3.83 | 226.7 ± 14.1 | 47.6 ± 0.33 | 13.7 ± 0.84 | 2.59 ± 0.32 | 28.8 ± 3.55 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ko, E.-Y.; Lee, J.-H.; Sivanesan, I.; Choi, M.-J.; Keum, Y.-S.; Saini, R.K. Carotenoid and Tocopherol Profiling in 18 Korean Traditional Green Leafy Vegetables by LC-SIM-MS. Foods 2023, 12, 1312. https://doi.org/10.3390/foods12061312
Ko E-Y, Lee J-H, Sivanesan I, Choi M-J, Keum Y-S, Saini RK. Carotenoid and Tocopherol Profiling in 18 Korean Traditional Green Leafy Vegetables by LC-SIM-MS. Foods. 2023; 12(6):1312. https://doi.org/10.3390/foods12061312
Chicago/Turabian StyleKo, Eun-Young, Ji-Ho Lee, Iyyakkannu Sivanesan, Mi-Jung Choi, Young-Soo Keum, and Ramesh Kumar Saini. 2023. "Carotenoid and Tocopherol Profiling in 18 Korean Traditional Green Leafy Vegetables by LC-SIM-MS" Foods 12, no. 6: 1312. https://doi.org/10.3390/foods12061312
APA StyleKo, E.-Y., Lee, J.-H., Sivanesan, I., Choi, M.-J., Keum, Y.-S., & Saini, R. K. (2023). Carotenoid and Tocopherol Profiling in 18 Korean Traditional Green Leafy Vegetables by LC-SIM-MS. Foods, 12(6), 1312. https://doi.org/10.3390/foods12061312








