Nano- and Microplastics Migration from Plastic Food Packaging into Dairy Products: Impact on Nutrient Digestion, Absorption, and Metabolism
Abstract
1. Introduction
2. Effect of NMPs on Human Health—Overview
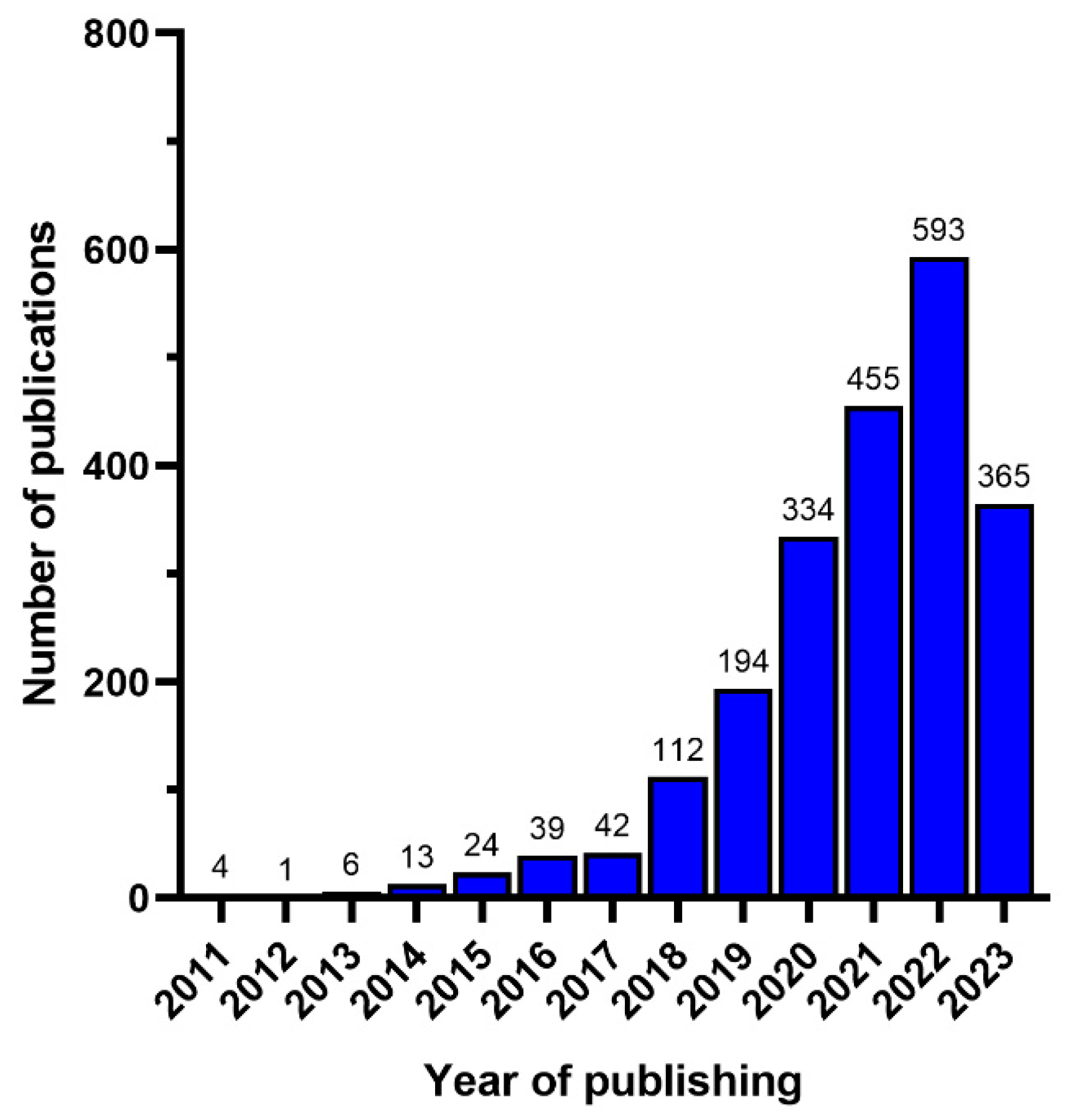
3. Bibliometric Analysis
4. Types of Polymers Used as Packaging in the Food Industry and Chemical Additives
| Type of Polymer | Application | Type of Monomer | Common Chemical Additives | Potential Health Effects of Chemical Additives | References |
|---|---|---|---|---|---|
| Polypropylene (PP) | Food packaging, sweet and snack wrappers, hinged caps | Propylene | Irgafos 168 (tris (2,4-di-tert-butylphenyl) phosphite) and Polystyrene-b-poly (ethylene-co-butylene)-b-polystyrene (SEBS) | Irgafos 168 is associated with cell growth inhibition and endocrine disruption | [24,25] |
| High-density polyethylene (HDPE) | Milk bottles | Ethylene | Calcium stearate, halogens, and metal catalysts. | No reported adverse health effects | [24,26] |
| Low-density polyethylene (LDPE) | Food packaging film, food containers, and trays | Ethylene | Calcium stearate, halogens, and Irgafos 168. | Irgafos 168 is associated with cell growth inhibition and endocrine disruption. | [24,27,28] |
| Polystyrene (PS) | Dairy and fishery food packaging, bottle caps, cups, and trays | Styrene | Hexabromocyclododecane | Hepatotoxicant, endocrine disruptor, and neurotoxic. | [26,29] |
| Polyethylene terephthalate (PET) | Water, soft drink, and juice bottles | Terephthalic acid and ethylene glycol | Benzothiazole, triphenyl phosphate (TPhP), Irgafos 168 (tris (2,4-di-tert-butylphenyl) phosphite), tripropyl phosphate (TPP), phthalide, tris (2-chloroethyl) phosphate (TCEP), benzophenone, and phthalimide | Neurotoxic (neuroinflammation and neuronal apoptosis); induces lipid peroxidation; causes lysosomal dysfunction, thyroid endocrine disruption; carcinogenic; hepatotoxic; and causes developmental disfunction. | [26,28,30] |
| Polyvinyl chloride (PVC) | Trays, bottles, containers, flexible films, caps, and can linings | Vinyl chloride | Plasticizers: dioctylphthalate esters; UV-stabilizers: barium, lead oxide, or cadmium carboxylates; Pigments: carbon black, titanium dioxide, organotin stabilizers, barbituric acid or thiobarbituric acid, dialkyltin maleates, 2-Benzimidazolylacetonitrile, naphthalene, and diisonyl phthalate. | Plasticizers may cause reproductive and developmental disorders, endocrine disruption, and cardiometabolic diseases. UV-stabilizers may cause oxidative stress and acute hepatotoxicity. Pigments may cause inflammation. | [25,26,31,32] |
5. Analytical Methods for the Detection of NMPs and Plastic Oligomers
6. Migration and Occurrence of NMPs in Dairy Products
| Type of Milk Product | Type of Packaging Material | Country of Study | Sample Processing and MPs Characterization | MPs’ Shape and Size | Quantity of MPs | References | ||
|---|---|---|---|---|---|---|---|---|
| MPs’ Extraction | Filter Pore Size | Polymer Characterization | ||||||
| Skimmed milk | Polyethylene | Ecuador | Filtration and digestion with 30% H2O2 for 72 h | 250 µm | FTIR | Fibers and fragments (2.48–183.37 µm) | 16–53 MPs/L | [35] |
| Whole, half fat, light, and lactose-free milk | Polysulfone | Mexico | Filtration (11 μm filter pore size) | 11 µm | Nikon epifluorescence microscope H6000L; SEM-EDS; Raman spectroscope | Fibers and fragments (>11 µm) | 3–11 MPs/L | [8] |
| Milk powder | Boxed with inner plastics | China | Enzyme digestion (gastric juice containing pepsin followed by the addition of a pancreatin enzyme) and filtration | 8 µm | FTIR | Fibers and fragments | 1–11 MPs/100 g | [41] |
| Yoghurt | NR | Turkey | Multi-enzyme and tetramethylammonium hydrate digestion (40 °C for 24 h) and filtration | 1 µm | SEM and ATR-FTIR | Fibers and fragments | 2–58 MPs/100 mL | [34] |
| Breastmilk | NR | Italy | Filtration and digestion with 10% KOH (40 °C for 48 h) | 1.6 µm | Raman microspectrometer | Fragment (2–12 µm) | 3–11 MPs/L | [33] |
| Liquid and powder milk | NR | Switzerland | Enzyme (Prozyme) and 25% tetramethyl ammonium hydroxide digestion (80 °C for 1 min) | 5 μm | µRaman and optical microscopy; SEM-EDX | Fragments | 204–1004 MPs/100 mL | [10] |
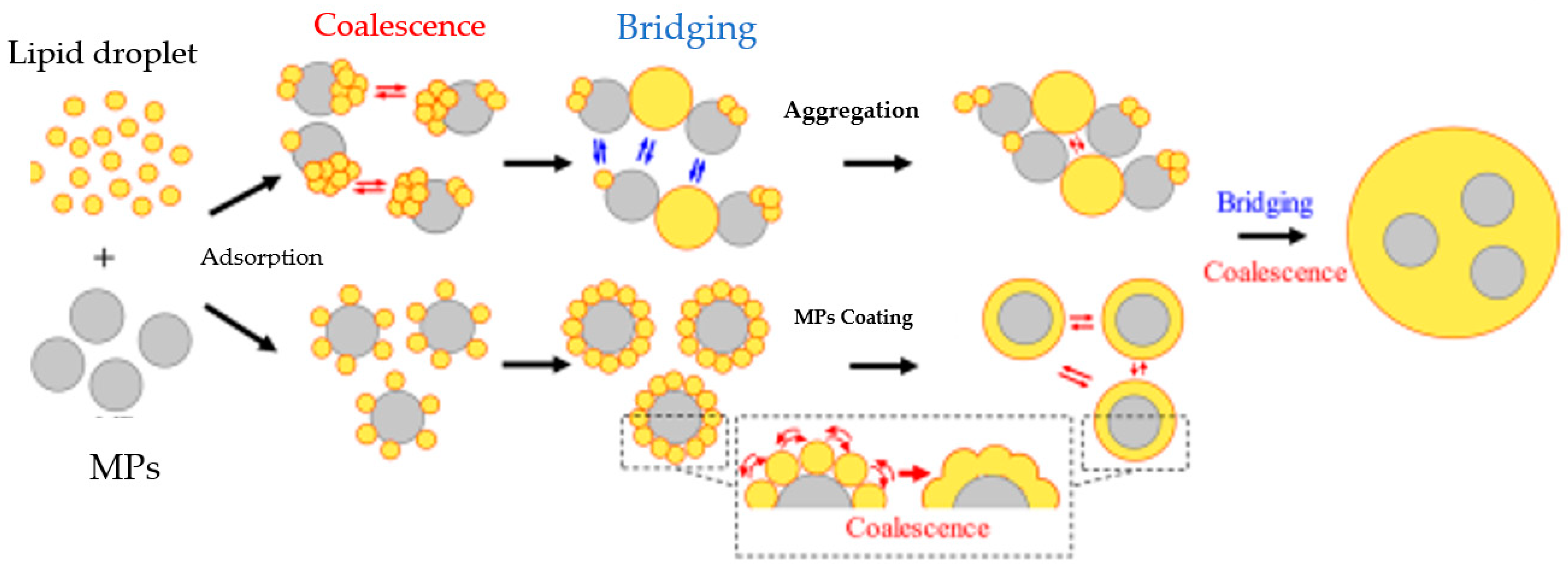
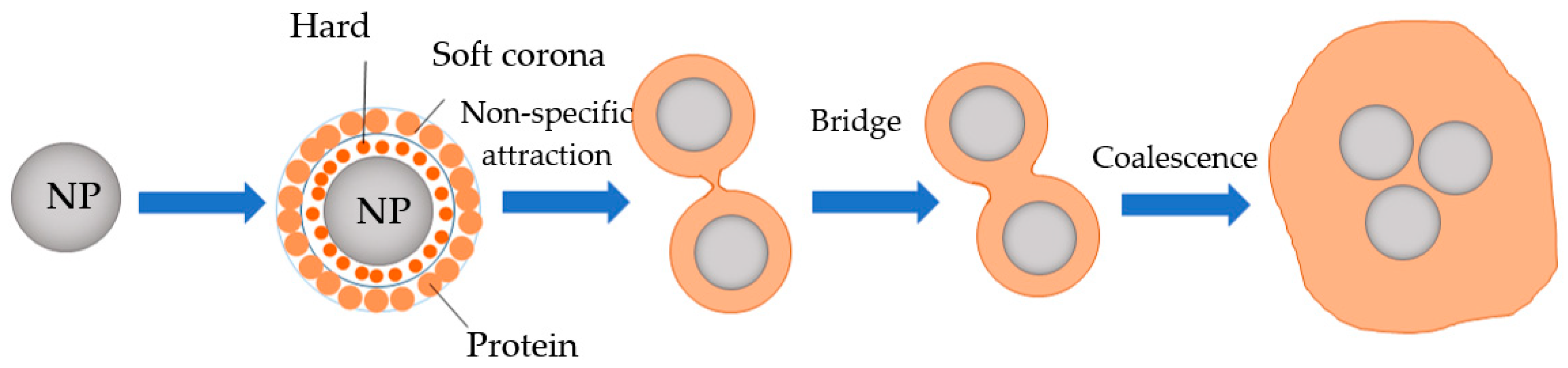
7. Potential Transformation of NMPs during Food Digestion
8. Interaction and Effect of NMPs on the Digestion and Absorption of Food Macro-Components
8.1. Carbohydrates
8.2. Fats
8.3. Proteins
| Nutrient Type | Type of Study | Experimental Model | Key Findings | References |
|---|---|---|---|---|
| Lipid | In vitro | Stock lipid emulsion (olive oil (4% w/w)+ phosphate buffer, pH = 7) was mixed with MPs (PE, PVC, and PET (100, 200, 300, 400 mg/L, and 50 nm, 1 μm, 10 μm)). Lipid digestion was carried out using in vitro simulated digestion. | All the MPs significantly reduced lipid digestion, with PS–MPs exhibiting the highest inhibition. Lipid digestion decreased with an increasing PS concentration. PS-based MPs interacted with both lipid droplets and lipase enzymes. | [12] |
| In vitro | A standardized food model (3.4% protein (sodium caseinate), 4.6% sugar (sucrose), 5.2% digestible carbohydrate (corn starch), 0.7% dietary fiber (pectin), 3.4% fat (corn oil), and 0.5% sodium chloride) and high-fat food (33.3% fat) models were mixed with PE–I PM0.1. In vitro simulated digestion was performed using a three-phase simulator. | PE–I increased fat digestion and absorption. Fatty acids in the small intestinal phase were enriched. | [49] | |
| In vivo | Rainbow trout fish (25.1 ± 8.1 g, 9.2 ± 2.2 cm) were exposed to PS–MPs (30 or 300 μg/L), chlorpyrifos (2 or 6 μg/L), or their combination. | The PS–MPs had a minimal effect on fatty acid composition. However, significant alterations in the fatty acid composition were observed in combined PS–MPs and chlorpyrifos. | [52] | |
| Protein | In vitro | A standardized food model (3.4% protein (sodium caseinate), 4.6% sugar (sucrose), 5.2% digestible carbohydrate (corn starch), 0.7% dietary fiber (pectin), 3.4% fat (corn oil), and 0.5% sodium chloride) and high-fat food (33.3% fat) models were mixed with PE–I PM0.1. In vitro simulated digestion was performed using a three-phase simulator. | Triacylglycerol lipase enrichment and β-casein depletion were observed. | [49] |
| In vitro | Cow’s milk was mixed with PS–MPs (10 µm, 14,200 particles) and subjected to simulated gastric digestion. | PS–MPs promoted the accumulation of larger peptides, suggesting their delayed digestion in the gastric phase. | [56] | |
| In vivo | Rainbow trout fish (25.1 ± 8.1 g, 9.2 ± 2.2 cm) were exposed to PS–MPs (30 or 300 μg/L), chlorpyrifos (2 or 6 μg/L), or their combination. | The PS-based MPs had no effect on the protein and amino acid contents of fish muscle. Nonetheless, significant changes were observed when PS-based MPs and chlorpyrifos were combined. | [52] | |
| In vivo | Healthy adult zebrafish (Danio rerio, 5-month-old) were exposed to PS–MPs (5 μm beads; 50 μg/L and 500 μg/L) for 21 days. | Phenylalanine, proline, lysine, leucine, threonine, alanine, glutamine, tyrosine, and ornithine were considerably altered. | [53] | |
| Carbohydrate | In vivo | Mussels of 5–7 cm lengths and 21 months of age were fed with PS spheres (10 μm, 55,000, and 110,000/L). | Exposure to higher levels of PS–MPs raised amylase activity and negatively affected the ability of mussels to digest starch. | [11] |
| In vivo | Mussels (M. coruscus (1.5 ± 0.90 g; 7.95 ± 0.32 cm)) were treated with different concentrations of PS microspheres that were 2 mm in diameter (0, 10, 104 and 106/L) at pH 7.7 and 8.1 for 14 days before a recovery acclimation of 7 days. | The alpha-amylase enzyme was significantly inhibited. | [47] |
9. Potential Effect of Ingested NMPs on Nutrient Metabolism
| Type of Study | Experimental Model | Key Findings | References |
|---|---|---|---|
| In vivo studies using mice | Five-week-old mice (n = 40) were exposed to 0.5 and 50 μm PS MPs (100 and 1000 μg/L) for 5 weeks. | MPs induced gut microbiota dysbiosis and hepatic lipid metabolism disorder. | [76] |
| Five-week-old mice were fed with PS–MPs (5 μm, 100 and 1000 μg/L) for 6 weeks. | PS–MPs induced gut microbiota dysbiosis, intestinal barrier dysfunction, and metabolic disorders. | [77] | |
| Seven-week-old C57BL/6J (wild type) male mice were fed with a high-fat diet together with MPs for 4 weeks. | Mice fed the high-fat diet with MPs showed a higher expression of genes related to the Na+/glucose cotransporter and long-chain fatty acid transporter. | [66] | |
| Forty C57 BL/6 female mice (ICR) and 20 male mice (seven-week-old) were treated with 1 and 10 mg/L polystyrene NPs. | Cholesterol metabolism was disturbed. Metabolic disorders which affected daidzein and sucrose concentrations were reported. | [78] | |
| Male C57BL/6 mice (six-week-old) were exposed to 100 μg/L or 1000 μg/L MPs, respectively, for 8 weeks. | Exposure to MPs induced the enrichment of genes expressed in the lipid metabolism pathway. | [70] | |
| PS–NPs were fed to mice at a dosage of 5 mg/kg and 15 mg/kg body weight. | Chronic exposure to PS–NPs increased plasma glucose levels. | [79] | |
| ICR female mice (7 week old) were administered with MPs (100 and 1000 μg/L) during pregnancy and lactation (∼6 weeks). | MPs caused the metabolic disorder in maternal MPs associated with gut microbiota dysbiosis and gut barrier dysfunction. | [80] | |
| One hundred male C57BL/6 mice (7–8 weeks old, 20–22 g) were administered for 8 weeks with PS–NPs at dosages of 1, 10, and 30 mg/kg/day alone or combined with a diet high in and injected with streptozocin (STZ). | Insulin resistance, glucose intolerance, and blood glucose level increased. | [67] | |
| PS–MPs (5 μm and 20 μm fluorescent) were fed to five-week-old male mice (Mus musculus, ICR) for 28 days | MPs exposure disturbed energy and lipid metabolism. | [68] | |
| Sixteen- to twenty-week-old male Hmox1 reporter mice were fed with carboxymethylcellulose (CMC, 0.5% w/v), a mixture of MPs (1, 4, 10 µm) and CMC (10 mL/kg body weight), for 28 days. | Energy metabolism was impaired. | [69] | |
| In vivo studies using fish | Juvenile M. nipponense (22.96 ± 3.87 mm in length and 0.14 ± 0.06 g in weight) were fed for 28 days with NPs of varying concentrations (0, 5, 10, 20, and 40 mg/L). | Metabolism-related genes including 6-phosphate glucokinase, Acetyl-CoA-binding protein (ACBP), HK, CPT-1, PK, ACC, and fatty-acid-binding protein 10 (FABP 10) had their expression altered. | [58] |
| Adult marine medaka (Oryzias melastigma, 8-month-old) were exposed to 2, 10, and 200 μm PS–MPs at a concentration of 10 mg/L for 60 days. | Exposure to 200 μm PS–MPs increased the bodyweight, hepatic lipid content, and adipocyte size. | [81] | |
| Juvenile G. laevifrons (n = 30, body size = 5.0 ± 0.4 cm SL; body weight = 1.5 g ± 0.2 g) were fed with 0.001 and 0.1 g of PS–MPs (8 μm) per 0.5 g of food weekly for 45 days. | Hyperemia was more severe in the higher-exposure group compared to the lower-exposure group. | [59] | |
| Nile Tilapia (Oreochromis niloticus) were fed with 1 mg/L PS NMPs (80 and 800 nm, 8.76 and 80 μm) for 14 days. | An imbalance of gut microbiota homeostasis and disordered liver metabolism were observed in fish fed with 80 nm NMPs. | [62] | |
| Healthy four-month-old zebrafish (Danio rerio, AB strain, 0.34 ± 0.03 g in wet weight, 33 ± 2 mm in body length) were exposed to pristine MPs (20 mg/L) for 24 h. | Increased metabolism disruption was observed. | [53] | |
| A total of 100 individuals of M. galloprovincialis (size 4.1 cm ± 0.9 SD) exposed to MPs were subjected to a synthetic polymer powder HDPE (1–50 μm) for 18 days. | Immune-related proteins were produced and growth energy was decreased. | [48] | |
| Large yellow croaker juveniles (about five months old) were fed with PS NPs suspensions of 0, 1, 10, and 100 mg/kg, respectively, for 21 days. | Liver lipid accumulation was observed. Fatty acid composition changes and lipid metabolism disruption were also observed. | [64] | |
| Zebrafish wild-type (AB/TL strain) larvae were exposed to 0, 0.2, 2, and 20 mg/ L PS–NPs. | PS–NP-induced disruption of glucose homoeostasis. | [63] | |
| In vivo studies using chickens | Sixty-one-day-old healthy Arbor Acres chickens (48 ± 4 g) were exposed to PE–MPs (200 mg/kg) in feed for 28 days. | PE–MPs exposure negatively affected gut microbial homeostasis and intestinal metabolism. | [82] |
| One-day-old (120) chickens were fed with polystyrene-based MPs (1, 10, and 100 mg/L) for 6 weeks | PS–MPs promoted lipopolysaccharide accumulation and promoted hepatic lipid metabolism disorders. | [83] | |
| Cornish-cross broilers were fed for 14 days with PS (2 mg/kg 50 nm) and carboxylated and undyed NPs. | Lower iron absorption was observed more in chickens exposed to carboxylated NPs. | [84] | |
| In vivo studies using bees | 10-day-old honeybees (Apis mellifera) were exposed to PS NMPs (104 and 105 particles/mL; 100 nm, 1 μm, and 10 μm) through a diet of pollen and 50% sucrose syrup. | Immune inhibitory genes were stimulated, while genes related to energy balance were depressed. | [71] |
| In vitro studies | In vitro simulated digestion models for gastric (6 mg PS–MPs were dispersed in 35 mL of gastric fluid; 0.1, 1, and 10 μm) and intestinal digestion were applied. | No significant effect on nutrient absorption or metabolism was observed. | [85] |
| RAW 264.7 macrophages and BV2 microglial cells were exposed to 200 nm NPs (1, 5, 10, 25, 50, 100, and 200 μg/mL) before incubation for 24 h. | The exposure of BV2 microglial cells to PS–NPs induced lipid accumulation. | [73] | |
| A549, HePG-2, and HCT116 cells were treated by 30 nm PS–NPs (25 μg/mL) and 30 nm Au–NPs (0.7875, 1.575, 3.15 ng/mL). | Distribution of cytokinesis-associated proteins was observed. | [74] | |
| Epithelial kidney cells and L929 mouse fibroblast cell lines were exposed to clear PE (1.0–4.0 μm) and PS (9.5–11.5 μm) microspheres. | The metabolic rate increased as the concentrations of PS and PE–MPs increased. | [75] | |
| Bronchial epithelial BEAS-2B cells were treated with 1 mg/mL PS–NPs. | Increases in amino acids and tricarboxylic acid cycle intermediate metabolites were observed. | [72] |
10. Migration of Plastic Oligomers and Their Potential Effect on Health
| Material Investigated | Material Composition | Conditions of the Migration Experiment | Key Findings | References |
|---|---|---|---|---|
| Tea bags | PET | Tea bags, from which the tea was removed prior to the migration experiment, were immersed for 5 min with constant temperature control in boiling water and 20% and 50% ethanol. | In pure water, only cyclic monomers, dimers, and trimer were found, and an up to first-series heptamer was found in 50% ethanol. In all simulants, the first-series cyclic trimer and second-series cyclic dimer were the most dominant. Linear oligomers have also been found. According to exposure assessment, when tea is prepared in water, the TTC threshold is not exceeded when one tea per day is consumed, but the threshold is exceeded for the second0series cyclic dimer in three out of five bags when five teas are consumed. In the same scenario, the EFSA threshold of 50 µg/kg food for the total oligomer migration is exceeded for four out of five tea bags in cases of cyclic PET oligomers. | [87] |
| Coffee capsules | PET, PBT, PP | In the first experiment, coffee capsules were placed in an instant coffee machine, and either water or 20% ethanol was passed through the capsules at a temperature of 80–85 °C. In the second experiment, an immersion test was performed by immersing the capsules in water at 92–93 °C or in 20% ethanol at 84–85 °C for 5 min. | Migration was higher in the immersion experiment than in the coffee machine one. The cyclic PBT dimer and pentamer had the highest migration level, while for PET, this was the case for the cyclic dimer and trimer. In addition, migration into 20% ethanol was higher than that into water. Consumption of one coffee per day does not exceed the TTC threshold. However, three out of five capsules do exceed the EFSA limit. | [88] |
| Various kitchen utensils | PA6, PA66 | A total immersion method was used. Sesame grinders and cake servers were treated at 60 °C for 30 min in water, 20% ethanol, and olive oil; ladles were treated at 95 °C for 30 min in water, 20% ethanol, and olive oil; and turners were treated at 121 °C for 30 min in olive oil. | Five PA6 cyclic oligomers and three PA66 cyclic monomer and oligomers were detected. High migration levels of the PA66 monomer, dimer, and trimer into 20% ethanol (260 to 1000 µg/cm2) were observed. | [89] |
| Cooking spoons and tea bags | PA6, PA66 | Cooking spoons were treated by boiling 3% acetic acid solution for 2 h in three subsequent migration experiments on three subsequent days. Filled or empty tea bags were inserted in cups with boiling water for 10 min. | From the cooking spoons, the PA 6 cyclic dimer up to hexamer contributed the most to the total PA 6 cyclic oligomer migration, while the cyclic PA 66 monomer and dimer contributed more than 90% of all migrated cyclic PA 66 oligomers. For the tea bags, there is no effect of the food matrix on the migration levels. The highest migration was found for PA 6 tetramer (189.6 µg/L). Based on the TTC approach, the threshold for maximum exposure is exceeded by both the cooking spoons and the tea bags. | [90] |
| Slotted spoons | PBT | Contact with boiling water (100 °C) for 2 h. Repeated three times for the same spoon. | In the first migrate, seven linear and the cyclic dimer were detected at a level of 0.86 mg per item, which decreased to 0.46 mg per item (second migrate) and 0.34 mg per item (third migrate), with the cyclic dimer making up only 15% of the total oligomers. The authors conclude that the migration of PBT oligomers could be considered safe when discussing aqueous foodstuffs. | [101] |
| Multilayer baby food packaging materials | PET/Al/PE | Baby food from commercial packaging was analyzed for oligomer content. A migration test on packaging was performed using 3% acetic acid and 20% ethanol for 10 days at 40 °C. | In total, 35 out of 39 NIAS detected were polyester oligomers, and 29 were cyclic, and 6 were linear oligomers. Several of the oligomers quantified exceeded the migration level of 0.01 mg/kg in some of the baby food analyzed. The authors give an example calculation on AA-DEG, where the consumption of one baby food pouch resulted in a daily intake that is 30 times higher than the TTC value (0.015 mg for a 10 kg infant). | [94] |
| Tin plates with Polyester-phenol-coatings | PE | Migration tests into the food simulants 20% ethanol, 50% ethanol, and water were performed in a sterilizer for 1 h at 121 °C with a 0.5 bar nitrogen excess pressure. The same was repeated with two types of commercial and two types of homemade infant food. | The following contents of cyclic polyester oligomers were detected: water—24.3 µg/dm3, 20% ethanol—101.2 µg/dm3, 50% ethanol—281.7 µg/dm3, commercial infant food (0.2% fat)—16.7 µg/dm3, commercial infant food (0.6% fat)—34.7 ug/dm3, homemade puree (0.2% fat)—11.6 ug/dm3, and homemade puree (5.2% fat)—68.4 ug/dm3. Both individual oligomers and polyester-based substances in total can be released in quantities of concern. | [95] |
| Plates | PBT | A migration experiment was performed in 20% ethanol for 30 days at 40 °C and 60 °C | The migration of cyclic and linear PBT dimers was observed at both temperatures, while the migration of the cyclic PBT trimer was detected only at 60 °C. After 3 days, the migration limit of 50 µg/kg was exceeded. There is a possible swelling effect of the studied material in 20% ethanol, which might cause the overestimation of migration levels. | [104] |
| Flexible multilayer packaging materials used for cured meat | PET//PA//CPP, PET//Al//PA//CPP, joint by PU layers, CPP on the food contact side | A migration test was performed on bags using the food simulants 10% ethanol, 3% acetic acid, and 95% ethanol. Bags were kept at 60 °C for 10 days. | Caprolactam oligomers from dimer to pentamer were detected in 95% ethanol, with the tetramer being the most abundant in both packaging materials. Several cyclic PU oligomers were also detected in concentrations above the migration limit. Generally, migration was higher in 95% ethanol than in the other two simulants. The cyclic oligomer AA-DEG was shown to hydrolyze into its linear form when in contact with 3% acetic acid. | [92] |
| Slotted spoons | PBT | Contact with boiling water (100 °C) for 2 h. Repeated three times for the same spoon. | In the first migrate, seven linear and the cyclic dimer were detected at a level of 0.86 mg per item, which decreased to 0.46 mg per item (second migrate) and 0.34 mg per item (third migrate), with the cyclic dimer making up only 15% of the total oligomers. The authors conclude that the migration of PBT oligomers could be considered safe when discussing aqueous foodstuffs. | [101] |
| Multilayer baby food packaging materials | PET/Al/PE | Baby food from commercial packaging was analyzed for oligomer content. A migration test on packaging was performed using 3% acetic acid and 20% ethanol for 10 days at 40 °C. | In total, 35 out of 39 NIAS detected were polyester oligomers, 29 were cyclic, and 6 were linear oligomers. Several of the oligomers quantified exceeded the migration level of 0.01 mg/kg in some of the baby food analyzed. The authors give an example calculation on AA-DEG, where the consumption of one baby food pouch resulted in a daily intake that is 30 times higher than the TTC value (0.015 mg for a 10 kg infant). | [94] |
| Tin plates with Polyester-phenol-coatings | PE | Migration tests into the food simulants 20% ethanol, 50% ethanol, and water were performed in a sterilizer for 1 h at 121 °C with a 0.5 bar nitrogen excess pressure. The same was repeated with two types of commercial and two types of homemade infant food. | The following contents of cyclic polyester oligomers were detected: water—24.3 µg/dm3, 20% ethanol—101.2 µg/dm3, 50% ethanol—281.7 µg/dm3, commercial infant food (0.2% fat)—16.7 µg/dm3, commercial infant food (0.6% fat)—34.7 ug/dm3, homemade puree (0.2% fat)—11.6 ug/dm3, and homemade puree (5.2% fat)—68.4 ug/dm3. Both individual oligomers and polyester-based substances in total can be released in quantities of concern. | [95] |
| Plates | PBT | A migration experiment was performed in 20% ethanol for 30 days at 40 °C and 60 °C | The migration of cyclic and linear PBT dimers was observed at both temperatures, while the migration of the cyclic PBT trimer was detected only at 60 °C. After 3 days, the migration limit of 50 µg/kg was exceeded. There is a possible swelling effect of the studied material in 20% ethanol, which might cause an overestimation of migration levels. | [104] |
| Flexible multilayer packaging materials used for cured meat | PET//PA//CPP, PET//Al//PA//CPP, joint by PU layers, CPP on the food contact side | A migration test was performed on bags using the food simulants 10% ethanol, 3% acetic acid, and 95% ethanol. Bags were kept at 60 °C for 10 days. | Caprolactam oligomers from dimer to pentamer were detected in 95% ethanol, with the tetramer being the most abundant in both packaging materials. Several cyclic PU oligomers were also detected in concentrations above the migration limit. Generally, migration was higher in 95% ethanol than in the other two simulants. The cyclic oligomer AA-DEG was shown to hydrolyze into its linear form when in contact with 3% acetic acid. | [92] |
| Multilayer packaging materials used for biological fluids and food contact | PET/Al/PA/PP/PE, joint by PU | If the packaging was in contact with the biological fluid, a migration test was performed in water for 3 days at 40 °C. For food contact, a material migration test was performed in 10% or 95% ethanol for 10 days at 60 °C, or in the case of pasteurized materials, for 30 min at 121 °C in 95% ethanol. | The migration values of the cyclic oligomers AA-DEG and AA-DEG-IPA-DEG were tested for 20 packaging materials. In most cases, the migration of cyclic oligomers exceeded the migration limit of 0.01 mg/kg. According to the TTC approach, 6 out of 20 materials complied with FDA guidelines, and 5 out of 20 complied with EFSA guidelines. | [93] |
| Kitchen utensils | PA6, PA66 | Migration experiments were performed in either foodstuffs, such as canned beans, chicken soup, whole milk, and sunflower oil, for 30 min at 100 °C or in the simulants 3% acetic acid or 10%, 50% and 95% ethanol. For the 10% ethanol, the same migration conditions were used as those used for food, and for 50% and 95% ethanol, a temperature of 60 °C was used for 2.5 h. | PA66 monomer was the most abundant oligomer found in both simulants and food, except for sunflower oil, where PA6 monomer was more abundant. The migration of oligomers was overestimated compared to that of real food. For the real foods, the sum of all oligomers was below the migration limit (5 mg/kg). The sums of oligomers in sunflower oil, beans, chicken soup, and whole milk were 4.6 mg/kg, 3.1 mg/kg, 4.4 mg/kg, and 3.6 mg/kg, respectively. | [96] |
| Shot glasses and reusable coffee cups | SAN and ABS copolymers | For reusable coffee cups, migration experiments were performed in water, acetic acid (3%), ethanol (10, 20, and 50%), coconut fat, sunflower oil, cream (30%), butter fat, Miglyol®812, and cow’s milk (3.5% fat) at 70 °C or 100 °C for 2 h. Shot glasses were treated with 50% or 70% ethanol for 30 min at 40 °C. | SAn2 and S2An trimers are the most abundant oligomers extracted from SAN and ABS. Migration was the highest in 50% ethanol, with a decrease from the first to the third migrate. Migration into oils, fats, and dairy was below the LOD. A product of oligomer degradation, AMNC, was also detected in all extracts. | [97] |
| Kitchenware | PBT | Migration experiments were performed using food simulants (95%, 50%, and 20% ethanol and 3% acetic acid) and sunflower oil and milk at different temperatures. | In general, cyclic oligomers were dominant in all migrates when performing migration experiments of a hot beverage simulation at 70 °C for 2 h. Migration decreased with every consecutive migration experiment. 50% ethanol overestimated the migration into milk by fourfold. In the case of sunflower oil, migration is affected by temperature. According to the authors, one can consume 410 mL of milk before reaching the threshold of concern. Also, 12–15 g of oil might be consumed before reaching the threshold, which should be equivalent to 100 g of french fries. | [98] |
11. Conclusions and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Yuan, Z.; Nag, R.; Cummins, E. Human Health Concerns Regarding Microplastics in the Aquatic Environment—From Marine to Food Systems. Sci. Total Environ. 2022, 823, 153730. [Google Scholar] [CrossRef]
- ISO/TR 21960:2020; Plastics—Environmental Aspects—State of Knowledge and Methodologies. International Organization for Standardization: Geneva, Switzerland, 2020.
- Amobonye, A.; Bhagwat, P.; Raveendran, S.; Singh, S.; Pillai, S. Environmental Impacts of Microplastics and Nanoplastics: A Current Overview. Front. Microbiol. 2021, 12, 768297. [Google Scholar] [CrossRef]
- Mitrano, D.M.; Beltzung, A.; Frehland, S.; Schmiedgruber, M.; Cingolani, A.; Schmidt, F. Synthesis of Metal-Doped Nanoplastics and Their Utility to Investigate Fate and Behaviour in Complex Environmental Systems. Nat. Nanotechnol. 2019, 14, 362–368. [Google Scholar] [CrossRef]
- Amankwa, M.; Tetteh, E.; Mohale, T.; Dagba, G.; Opoku, P. The Production of Valuable Products and Fuel from Plastic Waste in Africa. Discov. Sustain. 2021, 2, 31. [Google Scholar] [CrossRef]
- Plastics Europe, 2021. Plastics-the Fact 2021: An Analysis of European Plastics Production, Demand, and Waste Data. Available online: https://plasticseurope.org/knowledge-hub/plastics-the-facts2021/ (accessed on 25 April 2023).
- Górska-Warsewicz, H.; Rejman, K.; Laskowski, W.; Czeczotko, M. Milk and Dairy Products and Their Nutritional Contribution to the Average Polish Diet. Nutrients 2019, 11, 1771. [Google Scholar] [CrossRef] [PubMed]
- Kutralam-Muniasamy, G.; Pérez-Guevara, F.; Elizalde-Martínez, I.; Shruti, V.C. Branded Milks—Are They Immune from Microplastics Contamination? Sci. Total Environ. 2020, 714, 136823. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Shi, Y.; Yang, L.; Xiao, L.; Kehoe, D.K.; Gun’ko, Y.K.; Boland, J.J.; Wang, J.J. Microplastic Release from the Degradation of Polypropylene Feeding Bottles during Infant Formula Preparation. Nat. Food 2020, 1, 746–754. [Google Scholar] [CrossRef]
- Da Costa Filho, P.A.; Andrey, D.; Eriksen, B.; Peixoto, R.P.; Carreres, B.M.; Ambühl, M.E.; Descarrega, J.B.; Dubascoux, S.; Zbinden, P.; Panchaud, A.; et al. Detection and Characterization of Small-Sized Microplastics (≥5 Μm) in Milk Products. Sci. Rep. 2021, 11, 24046. [Google Scholar] [CrossRef]
- O’Brien, C.J.; Hong, H.C.; Bryant, E.E.; Connor, K.M. The Observation of Starch Digestion in Blue Mussel Mytilus Galloprovincialis Exposed to Microplastic Particles under Varied Food Conditions. PLoS ONE 2021, 16, e0253802. [Google Scholar] [CrossRef]
- Tan, H.; Yue, T.; Xu, Y.; Zhao, J.; Xing, B. Microplastics Reduce Lipid Digestion in Simulated Human Gastrointestinal System. Environ. Sci. Technol. 2020, 54, 12285–12294. [Google Scholar] [CrossRef]
- Jadhav, E.; Singh Sankhla, M.; Bhat, R.; Bhagat, D. Microplastics from Food Packaging: An Overview of Human Consumption, Health Threats, and Alternative Solutions. Environ. Nanotechnol. Monit. Manag. 2021, 16, 100608. [Google Scholar] [CrossRef]
- Stock, V.; Fahrenson, C.; Thuenemann, A.; Dönmez, M.H.; Voss, L.; Böhmert, L.; Braeuning, A.; Lampen, A.; Sieg, H. Impact of Artificial Digestion on the Sizes and Shapes of Microplastic Particles. Food Chem. Toxicol. 2020, 135, 111010. [Google Scholar] [CrossRef] [PubMed]
- Garrido Gamarro, E. Microplastics in Food Commodities: A Food Safety Review on Human Exposure through Dietary Sources; Série Sécurité sanitaire et qualité des aliments; FAO: Rome, Italy, 2022; ISBN 978-92-5-136982-1. [Google Scholar]
- Meng, X.; Zhang, J.; Wang, W.; Gonzalez-Gil, G.; Vrouwenvelder, J.S.; Li, Z. Effects of Nano- and Microplastics on Kidney: Physicochemical Properties, Bioaccumulation, Oxidative Stress and Immunoreaction. Chemosphere 2022, 288, 132631. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Zhang, Y.; Tan, W.; Zhang, Z. Microplastics as an Emerging Environmental Pollutant in Agricultural Soils: Effects on Ecosystems and Human Health. Front. Environ. Sci. 2022, 10, 855292. [Google Scholar] [CrossRef]
- He, Y.-J.; Qin, Y.; Zhang, T.-L.; Zhu, Y.-Y.; Wang, Z.-J.; Zhou, Z.-S.; Xie, T.-Z.; Luo, X.-D. Migration of (Non-) Intentionally Added Substances and Microplastics from Microwavable Plastic Food Containers. J. Hazard. Mater. 2021, 417, 126074. [Google Scholar] [CrossRef]
- Smith, M.; Love, D.C.; Rochman, C.M.; Neff, R.A. Microplastics in Seafood and the Implications for Human Health. Curr. Environ. Health Rep. 2018, 5, 375–386. [Google Scholar] [CrossRef] [PubMed]
- Ajaj, A.; J’Bari, S.; Ononogbo, A.; Buonocore, F.; Bear, J.C.; Mayes, A.G.; Morgan, H. An Insight into the Growing Concerns of Styrene Monomer and Poly(Styrene) Fragment Migration into Food and Drink Simulants from Poly(Styrene) Packaging. Foods 2021, 10, 1136. [Google Scholar] [CrossRef]
- Kontou, S.; Dessipri, E.; Lampi, E. Determination of Styrene Monomer Migrating in Foodstuffs from Polystyrene Food Contact Articles Using HS-SPME-GC-MS/MS: Results from the Greek Market. Food Addit. Contam. Part A 2022, 39, 415–427. [Google Scholar] [CrossRef]
- Guazzotti, V.; Hendrich, V.; Gruner, A.; Fiedler, D.; Störmer, A.; Welle, F. Migration of Styrene in Yogurt and Dairy Products Packaged in Polystyrene: Results from Market Samples. Foods 2022, 11, 2120. [Google Scholar] [CrossRef]
- Hoppe, M.; De Voogt, P.; Franz, R. Identification and Quantification of Oligomers as Potential Migrants in Plastics Food Contact Materials with a Focus in Polycondensates—A Review. Trends Food Sci. Technol. 2016, 50, 118–130. [Google Scholar] [CrossRef]
- Kadac-Czapska, K.; Knez, E.; Gierszewska, M.; Olewnik-Kruszkowska, E.; Grembecka, M. Microplastics Derived from Food Packaging Waste—Their Origin and Health Risks. Materials 2023, 16, 674. [Google Scholar] [CrossRef]
- Mamun, A.A.; Prasetya, T.A.E.; Dewi, I.R.; Ahmad, M. Microplastics in Human Food Chains: Food Becoming a Threat to Health Safety. Sci. Total Environ. 2023, 858, 159834. [Google Scholar] [CrossRef] [PubMed]
- Sridharan, S.; Kumar, M.; Saha, M.; Kirkham, M.B.; Singh, L.; Bolan, N.S. The Polymers and Their Additives in Particulate Plastics: What Makes Them Hazardous to the Fauna? Sci. Total Environ. 2022, 824, 153828. [Google Scholar] [CrossRef]
- Riquet, A.M.; Breysse, C.; Dahbi, L.; Loriot, C.; Severin, I.; Chagnon, M.C. The Consequences of Physical Post-Treatments (Microwave and Electron-Beam) on Food/Packaging Interactions: A Physicochemical and Toxicological Approach. Food Chem. 2016, 199, 59–69. [Google Scholar] [CrossRef]
- Fauser, P.; Vorkamp, K.; Strand, J. Residual Additives in Marine Microplastics and Their Risk Assessment—A Critical Review. Mar. Pollut. Bull. 2022, 177, 113467. [Google Scholar] [CrossRef]
- Schecter, A.; Szabo, D.T.; Miller, J.; Gent, T.L.; Malik-Bass, N.; Petersen, M.; Paepke, O.; Colacino, J.A.; Hynan, L.S.; Harris, T.R.; et al. Hexabromocyclododecane (HBCD) Stereoisomers in U.S. Food from Dallas, Texas. Environ. Health Perspect. 2012, 120, 1260–1264. [Google Scholar] [CrossRef]
- Cort, S.D.; Godts, F.; Moreau, A. Packaging Materials 1: Polyethylene Terephthalate (PET) for Food Packaging Applications; Updated Version; ILSI Europe: Brussels, Belgium, 2017. [Google Scholar]
- Callaghan, M.A.; Alatorre-Hinojosa, S.; Connors, L.T.; Singh, R.D.; Thompson, J.A. Plasticizers and Cardiovascular Health: Role of Adipose Tissue Dysfunction. Front. Pharmacol. 2020, 11, 626448. [Google Scholar] [CrossRef]
- Hlisníková, H.; Petrovičová, I.; Kolena, B.; Šidlovská, M.; Sirotkin, A. Effects and Mechanisms of Phthalates’ Action on Neurological Processes and Neural Health: A Literature Review. Pharmacol. Rep. 2021, 73, 386–404. [Google Scholar] [CrossRef] [PubMed]
- Ragusa, A.; Notarstefano, V.; Svelato, A.; Belloni, A.; Gioacchini, G.; Blondeel, C.; Zucchelli, E.; De Luca, C.; D’Avino, S.; Gulotta, A.; et al. Raman Microspectroscopy Detection and Characterisation of Microplastics in Human Breastmilk. Polymers 2022, 14, 2700. [Google Scholar] [CrossRef]
- Zipak, S.; Muratoğlu, K.; Büyükünal, S. Evaluation of Microplastic Presence in Yogurt Production Process. KAFKAS Univ. Vet. Fak. Derg. 2022, 28, 633–641. [Google Scholar] [CrossRef]
- Diaz-Basantes, M.F.; Conesa, J.A.; Fullana, A. Microplastics in Honey, Beer, Milk and Refreshments in Ecuador as Emerging Contaminants. Sustainability 2020, 12, 5514. [Google Scholar] [CrossRef]
- Li, J.; Liu, H.; Paul Chen, J. Microplastics in Freshwater Systems: A Review on Occurrence, Environmental Effects, and Methods for Microplastics Detection. Water Res. 2018, 137, 362–374. [Google Scholar] [CrossRef]
- Li, P.; Li, Q.; Hao, Z.; Yu, S.; Liu, J. Analytical Methods and Environmental Processes of Nanoplastics. J. Environ. Sci. 2020, 94, 88–99. [Google Scholar] [CrossRef] [PubMed]
- Prata, J.C.; Da Costa, J.P.; Duarte, A.C.; Rocha-Santos, T. Methods for Sampling and Detection of Microplastics in Water and Sediment: A Critical Review. Trends Anal. Chem. 2019, 110, 150–159. [Google Scholar] [CrossRef]
- Caldwell, J.; Taladriz-Blanco, P.; Lehner, R.; Lubskyy, A.; Ortuso, R.D.; Rothen-Rutishauser, B.; Petri-Fink, A. The Micro-, Submicron-, and Nanoplastic Hunt: A Review of Detection Methods for Plastic Particles. Chemosphere 2022, 293, 133514. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Zhang, X.; Jia, P.; He, S.; Dai, H.; Deng, S.; Han, J. Release of Microplastics from Breastmilk Storage Bags and Assessment of Intake by Infants: A Preliminary Study. Environ. Pollut. 2023, 323, 121197. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Liu, L.; Jiang, Y.; Zhang, Y.; Fan, Y.; Rao, W.; Qian, X. Microplastics in Infant Milk Powder. Environ. Pollut. 2023, 323, 121225. [Google Scholar] [CrossRef]
- Fournier, E.; Etienne-Mesmin, L.; Grootaert, C.; Jelsbak, L.; Syberg, K.; Blanquet-Diot, S.; Mercier-Bonin, M. Microplastics in the Human Digestive Environment: A Focus on the Potential and Challenges Facing in Vitro Gut Model Development. J. Hazard. Mater. 2021, 415, 125632. [Google Scholar] [CrossRef]
- Liu, S.; Wu, X.; Gu, W.; Yu, J.; Wu, B. Influence of the Digestive Process on Intestinal Toxicity of Polystyrene Microplastics as Determined by in Vitro Caco-2 Models. Chemosphere 2020, 256, 127204. [Google Scholar] [CrossRef]
- Roch, S.; Brinker, A. Rapid and Efficient Method for the Detection of Microplastic in the Gastrointestinal Tract of Fishes. Environ. Sci. Technol. 2017, 51, 4522–4530. [Google Scholar] [CrossRef]
- Tamargo, A.; Molinero, N.; Reinosa, J.J.; Alcolea-Rodriguez, V.; Portela, R.; Bañares, M.A.; Fernández, J.F.; Moreno-Arribas, M.V. PET Microplastics Affect Human Gut Microbiota Communities during Simulated Gastrointestinal Digestion, First Evidence of Plausible Polymer Biodegradation during Human Digestion. Sci. Rep. 2022, 12, 528. [Google Scholar] [CrossRef]
- Jeyavani, J.; Sibiya, A.; Gopi, N.; Mahboob, S.; Riaz, M.N.; Vaseeharan, B. Dietary Consumption of Polypropylene Microplastics Alter the Biochemical Parameters and Histological Response in Freshwater Benthic Mollusc Pomacea Paludosa. Environ. Res. 2022, 212, 113370. [Google Scholar] [CrossRef]
- Wang, X.; Huang, W.; Wei, S.; Shang, Y.; Gu, H.; Wu, F.; Lan, Z.; Hu, M.; Shi, H.; Wang, Y. Microplastics Impair Digestive Performance but Show Little Effects on Antioxidant Activity in Mussels under Low pH Conditions. Environ. Pollut. 2020, 258, 113691. [Google Scholar] [CrossRef]
- Détrée, C.; Gallardo-Escárate, C. Polyethylene Microbeads Induce Transcriptional Responses with Tissue-Dependent Patterns in the Mussel Mytilus Galloprovincialis. J. Molluscan Stud. 2017, 83, 220–225. [Google Scholar] [CrossRef]
- DeLoid, G.M.; Cao, X.; Coreas, R.; Bitounis, D.; Singh, D.; Zhong, W.; Demokritou, P. Incineration-Generated Polyethylene Micro-Nanoplastics Increase Triglyceride Lipolysis and Absorption in an In Vitro Small Intestinal Epithelium Model. Environ. Sci. Technol. 2022, 56, 12288–12297. [Google Scholar] [CrossRef] [PubMed]
- Wright, S.L.; Rowe, D.; Thompson, R.C.; Galloway, T.S. Microplastic Ingestion Decreases Energy Reserves in Marine Worms. Curr. Biol. CB 2013, 23, R1031–R1033. [Google Scholar] [CrossRef] [PubMed]
- Sussarellu, R.; Suquet, M.; Thomas, Y.; Lambert, C.; Fabioux, C.; Pernet, M.E.J.; Le Goïc, N.; Quillien, V.; Mingant, C.; Epelboin, Y.; et al. Oyster Reproduction Is Affected by Exposure to Polystyrene Microplastics. Proc. Natl. Acad. Sci. USA 2016, 113, 2430–2435. [Google Scholar] [CrossRef]
- Hanachi, P.; Karbalaei, S.; Yu, S. Combined Polystyrene Microplastics and Chlorpyrifos Decrease Levels of Nutritional Parameters in Muscle of Rainbow Trout (Oncorhynchus Mykiss). Environ. Sci. Pollut. Res. Int. 2021, 28, 64908–64920. [Google Scholar] [CrossRef] [PubMed]
- Qiao, R.; Deng, Y.; Zhang, S.; Wolosker, M.B.; Zhu, Q.; Ren, H.; Zhang, Y. Accumulation of Different Shapes of Microplastics Initiates Intestinal Injury and Gut Microbiota Dysbiosis in the Gut of Zebrafish. Chemosphere 2019, 236, 124334. [Google Scholar] [CrossRef]
- Gligorijevic, N.; Stanic-Vucinic, D.; Mutic, T.; Lujic, T.; Cirkovic Velickovic, T. Binding and Corona Formation of Ovalbumin to Polystyrene and Polyethylene Terephthalate Microplastics under Neutral and Acidic Conditions. In Proceedings of the XXII EuroFoodChem Congress, Belgrade, Serbia, 14 June 2023; p. 137. Available online: https://cherry.chem.bg.ac.rs/handle/123456789/5909 (accessed on 15 July 2023).
- Lujic, T.; Gligorijevic, N.; Stanic-Vucinic, D.; Cirkovic Velickovic, T. Investigation of Structural Changes in Ovalbumin Induced by Two Types of MPs and Its Impact on Protein Digestibility. In Proceedings of the XXII EuroFoodChem Congress, Belgrade, Serbia, 14 June 2023; p. 153. Available online: https://cherry.chem.bg.ac.rs/handle/123456789/5907 (accessed on 15 July 2023).
- de Guzman, M.K.; Stanic-Vucinic, D.; Gligorijevic, N.; Wimmer, L.; Gasparyan, M.; Lujic, T.; Vasovic, T.; Dailey, L.A.; Van Haute, S.; Velickovic, T.C. Small polystyrene microplastics interfere with the breakdown of milk proteins during static in vitro simulated human gastric digestion. Environ. Pollut. 2023, 335, 122282. [Google Scholar] [CrossRef]
- Gopinath, P.M.; Saranya, V.; Vijayakumar, S.; Mythili Meera, M.; Ruprekha, S.; Kunal, R.; Pranay, A.; Thomas, J.; Mukherjee, A.; Chandrasekaran, N. Assessment on Interactive Prospectives of Nanoplastics with Plasma Proteins and the Toxicological Impacts of Virgin, Coronated and Environmentally Released-Nanoplastics. Sci. Rep. 2019, 9, 8860. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Liu, Z.; Yang, Y.; Jiang, Q.; Wu, D.; Huang, Y.; Jiao, Y.; Chen, Q.; Huang, Y.; Zhao, Y. Effects of Nanoplastics on Energy Metabolism in the Oriental River Prawn (Macrobrachium Nipponense). Environ. Pollut. 2021, 268, 115890. [Google Scholar] [CrossRef] [PubMed]
- Ahrendt, C.; Perez-Venegas, D.J.; Urbina, M.; Gonzalez, C.; Echeveste, P.; Aldana, M.; Pulgar, J.; Galbán-Malagón, C. Microplastic Ingestion Cause Intestinal Lesions in the Intertidal Fish Girella Laevifrons. Mar. Pollut. Bull. 2020, 151, 110795. [Google Scholar] [CrossRef]
- Zhao, Y.; Qiao, R.; Zhang, S.; Wang, G. Metabolomic Profiling Reveals the Intestinal Toxicity of Different Length of Microplastic Fibers on Zebrafish (Danio Rerio). J. Hazard. Mater. 2021, 403, 123663. [Google Scholar] [CrossRef] [PubMed]
- Yin, L.; Liu, H.; Cui, H.; Chen, B.; Li, L.; Wu, F. Impacts of Polystyrene Microplastics on the Behavior and Metabolism in a Marine Demersal Teleost, Black Rockfish (Sebastes Schlegelii). J. Hazard. Mater. 2019, 380, 120861. [Google Scholar] [CrossRef]
- Wu, D.; Lu, X.; Dong, L.-X.; Tian, J.; Deng, J.; Wei, L.; Wen, H.; Zhong, S.; Jiang, M. Nano Polystyrene Microplastics Could Accumulate in Nile Tilapia (Oreochromis Niloticus): Negatively Impacts on the Liver and Intestinal Health through Water Exposure. J. Environ. Sci. 2024, 137, 604–614. [Google Scholar] [CrossRef]
- Brun, N.R.; van Hage, P.; Hunting, E.R.; Haramis, A.-P.G.; Vink, S.C.; Vijver, M.G.; Schaaf, M.J.M.; Tudorache, C. Polystyrene Nanoplastics Disrupt Glucose Metabolism and Cortisol Levels with a Possible Link to Behavioural Changes in Larval Zebrafish. Commun. Biol. 2019, 2, 382. [Google Scholar] [CrossRef]
- Lai, W.; Xu, D.; Li, J.; Wang, Z.; Ding, Y.; Wang, X.; Li, X.; Xu, N.; Mai, K.; Ai, Q. Dietary Polystyrene Nanoplastics Exposure Alters Liver Lipid Metabolism and Muscle Nutritional Quality in Carnivorous Marine Fish Large Yellow Croaker (Larimichthys Crocea). J. Hazard. Mater. 2021, 419, 126454. [Google Scholar] [CrossRef]
- Ašmonaitė, G.; Larsson, K.; Undeland, I.; Sturve, J.; Carney Almroth, B. Size Matters: Ingestion of Relatively Large Microplastics Contaminated with Environmental Pollutants Posed Little Risk for Fish Health and Fillet Quality. Environ. Sci. Technol. 2018, 52, 24. [Google Scholar] [CrossRef]
- Okamura, T.; Hamaguchi, M.; Hasegawa, Y.; Hashimoto, Y.; Majima, S.; Senmaru, T.; Ushigome, E.; Nakanishi, N.; Asano, M.; Yamazaki, M.; et al. Oral Exposure to Polystyrene Microplastics of Mice on a Normal or High-Fat Diet and Intestinal and Metabolic Outcomes. Environ. Health Perspect. 2023, 131, 027006. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wei, Z.; Xu, K.; Wang, X.; Gao, X.; Han, Q.; Wang, S.; Chen, M. The Effect and a Mechanistic Evaluation of Polystyrene Nanoplastics on a Mouse Model of Type 2 Diabetes. Food Chem. Toxicol. 2023, 173, 113642. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Zhang, Y.; Lemos, B.; Ren, H. Tissue Accumulation of Microplastics in Mice and Biomarker Responses Suggest Widespread Health Risks of Exposure. Sci. Rep. 2017, 7, 46687. [Google Scholar] [CrossRef]
- Stock, V.; Böhmert, L.; Lisicki, E.; Block, R.; Cara-Carmona, J.; Pack, L.K.; Selb, R.; Lichtenstein, D.; Voss, L.; Henderson, C.J.; et al. Uptake and Effects of Orally Ingested Polystyrene Microplastic Particles in Vitro and in Vivo. Arch. Toxicol. 2019, 93, 1817–1833. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Wu, Y.; Zhang, W.; Shen, T.; Li, H.; Wu, J.; Zhang, L.; Qin, L.; Chen, R.; Gu, W.; et al. Lipidomics and Transcriptomics Insight into Impacts of Microplastics Exposure on Hepatic Lipid Metabolism in Mice. Chemosphere 2022, 308, 136591. [Google Scholar] [CrossRef]
- Wang, K.; Zhu, L.; Rao, L.; Zhao, L.; Wang, Y.; Wu, X.; Zheng, H.; Liao, X. Nano- and Micro-Polystyrene Plastics Disturb Gut Microbiota and Intestinal Immune System in Honeybee. Sci. Total Environ. 2022, 842, 156819. [Google Scholar] [CrossRef]
- Lim, S.L.; Ng, C.T.; Zou, L.; Lu, Y.; Chen, J.; Bay, B.H.; Shen, H.-M.; Ong, C.N. Targeted Metabolomics Reveals Differential Biological Effects of Nanoplastics and NanoZnO in Human Lung Cells. Nanotoxicology 2019, 13, 1117–1132. [Google Scholar] [CrossRef]
- Florance, I.; Ramasubbu, S.; Mukherjee, A.; Chandrasekaran, N. Polystyrene Nanoplastics Dysregulate Lipid Metabolism in Murine Macrophages in Vitro. Toxicology 2021, 458, 152850. [Google Scholar] [CrossRef]
- Xia, L.; Gu, W.; Zhang, M.; Chang, Y.-N.; Chen, K.; Bai, X.; Yu, L.; Li, J.; Li, S.; Xing, G. Endocytosed Nanoparticles Hold Endosomes and Stimulate Binucleated Cells Formation. Part. Fibre Toxicol. 2016, 13, 63. [Google Scholar] [CrossRef]
- Palaniappan, S.; Sadacharan, C.M.; Rostama, B. Polystyrene and Polyethylene Microplastics Decrease Cell Viability and Dysregulate Inflammatory and Oxidative Stress Markers of MDCK and L929 Cells In Vitro. Expo. Health 2022, 14, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Wan, Z.; Luo, T.; Fu, Z.; Jin, Y. Polystyrene Microplastics Induce Gut Microbiota Dysbiosis and Hepatic Lipid Metabolism Disorder in Mice. Sci. Total Environ. 2018, 631–632, 449–458. [Google Scholar] [CrossRef]
- Jin, Y.; Lu, L.; Tu, W.; Luo, T.; Fu, Z. Impacts of Polystyrene Microplastic on the Gut Barrier, Microbiota and Metabolism of Mice. Sci. Total Environ. 2019, 649, 308–317. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Xiong, S.; Jing, Q.; van Gestel, C.A.M.; van Straalen, N.M.; Roelofs, D.; Sun, L.; Qiu, H. Maternal Exposure to Polystyrene Nanoparticles Retarded Fetal Growth and Triggered Metabolic Disorders of Placenta and Fetus in Mice. Sci. Total Environ. 2022, 854, 158666. [Google Scholar] [CrossRef]
- Fan, X.; Wei, X.; Hu, H.; Zhang, B.; Yang, D.; Du, H.; Zhu, R.; Sun, X.; Oh, Y.; Gu, N. Effects of Oral Administration of Polystyrene Nanoplastics on Plasma Glucose Metabolism in Mice. Chemosphere 2022, 288, 132607. [Google Scholar] [CrossRef]
- Luo, T.; Wang, C.; Pan, Z.; Jin, C.; Fu, Z.; Jin, Y. Maternal Polystyrene Microplastic Exposure during Gestation and Lactation Altered Metabolic Homeostasis in the Dams and Their F1 and F2 Offspring. Environ. Sci. Technol. 2019, 53, 10978–10992. [Google Scholar] [CrossRef]
- Zhang, X.; Wen, K.; Ding, D.; Liu, J.; Lei, Z.; Chen, X.; Ye, G.; Zhang, J.; Shen, H.; Yan, C.; et al. Size-Dependent Adverse Effects of Microplastics on Intestinal Microbiota and Metabolic Homeostasis in the Marine Medaka (Oryzias melastigma). Environ. Int. 2021, 151, 106452. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Wang, Y.; Kulyar, M.F.-E.-A.; Iqbal, M.; Lai, R.; Zhu, H.; Li, K. Environmental Microplastics Exposure Decreases Antioxidant Ability, Perturbs Gut Microbial Homeostasis and Metabolism in Chicken. Sci. Total Environ. 2023, 856, 159089. [Google Scholar] [CrossRef] [PubMed]
- Yin, K.; Wang, D.; Zhang, Y.; Lu, H.; Wang, Y.; Xing, M. Dose-Effect of Polystyrene Microplastics on Digestive Toxicity in Chickens (Gallus Gallus): Multi-Omics Reveals Critical Role of Gut-Liver Axis. J. Adv. Res. 2022, in press. [Google Scholar] [CrossRef]
- Mahler, G.J.; Esch, M.B.; Tako, E.; Southard, T.L.; Archer, S.D.; Glahn, R.P.; Shuler, M.L. Oral Exposure to Polystyrene Nanoparticles Affects Iron Absorption. Nat. Nanotechnol. 2012, 7, 264–271. [Google Scholar] [CrossRef]
- Ma, Y.; Koh, J.Y.C.; Lim, H.K.; Shi, P.; Tay, C.Y. Elucidating the Size-Dependency of In Vitro Digested Polystyrene Microplastics on Human Intestinal Cells Health and Function. Macromol. Chem. Phys. 2022, 223, 2100454. [Google Scholar] [CrossRef]
- European Commission (EU). Commission Regulation (EU) No 10/2011 of 14 January 2011 on Plastic Materials and Articles Intended to Come into Contact with Food. Off. J. Eur. Union 2011, 12, 89. Available online: https://eur-lex.europa.eu/EN/legal-content/summary/plastic-materials-and-articles-in-contact-with-food.html (accessed on 10 August 2023).
- Tsochatzis, E.D.; Alberto Lopes, J.; Kappenstein, O.; Tietz, T.; Hoekstra, E.J. Quantification of PET Cyclic and Linear Oligomers in Teabags by a Validated LC-MS Method—In Silico Toxicity Assessment and Consumer’s Exposure. Food Chem. 2020, 317, 126427. [Google Scholar] [CrossRef]
- Alberto Lopes, J.; Tsochatzis, E.D.; Karasek, L.; Hoekstra, E.J.; Emons, H. Analysis of PBT and PET Cyclic Oligomers in Extracts of Coffee Capsules and Food Simulants by a HPLC-UV/FLD Method. Food Chem. 2021, 345, 128739. [Google Scholar] [CrossRef]
- Abe, Y.; Mutsuga, M.; Ohno, H.; Kawamura, Y.; Akiyama, H. Isolation and Quantification of Polyamide Cyclic Oligomers in Kitchen Utensils and Their Migration into Various Food Simulants. PLoS ONE 2016, 11, e0159547. [Google Scholar] [CrossRef] [PubMed]
- Kappenstein, O.; Ebner, I.; Förster, C.; Richter, S.; Weyer, J.; Pfaff, K.; Luch, A. Validation and Application of an LC-MS/MS Method for the Determination of Cyclic Oligomers Originating from Polyamide 6 and Polyamide 66 in Food Simulant. Food Addit. Contam. Part A 2018, 35, 1410–1420. [Google Scholar] [CrossRef] [PubMed]
- Tsochatzis, E.D.; Lopes, J.A.; Kappenstein, O. Study of the Ionic Strength Effect on the Migration of Polyamide 6 and 66 Oligomers into Liquid Simulants by a LC-QTOF-MS Method. Food Packag. Shelf Life 2023, 35, 101015. [Google Scholar] [CrossRef]
- Ubeda, S.; Aznar, M.; Vera, P.; Nerín, C.; Henríquez, L.; Taborda, L.; Restrepo, C. Overall and Specific Migration from Multilayer High Barrier Food Contact Materials—Kinetic Study of Cyclic Polyester Oligomers Migration. Food Addit. Contam. Part A 2017, 34, 1784–1794. [Google Scholar] [CrossRef] [PubMed]
- Ubeda, S.; Aznar, M.; Rosenmai, A.K.; Vinggaard, A.M.; Nerín, C. Migration Studies and Toxicity Evaluation of Cyclic Polyesters Oligomers from Food Packaging Adhesives. Food Chem. 2020, 311, 125918. [Google Scholar] [CrossRef]
- Bauer, A.; Jesús, F.; Gómez Ramos, M.J.; Lozano, A.; Fernández-Alba, A.R. Identification of Unexpected Chemical Contaminants in Baby Food Coming from Plastic Packaging Migration by High Resolution Accurate Mass Spectrometry. Food Chem. 2019, 295, 274–288. [Google Scholar] [CrossRef]
- Eckardt, M.; Hetzel, L.; Brenz, F.; Simat, T.J. Release and Migration of Cyclic Polyester Oligomers from Bisphenol A Non-Intent Polyester–Phenol-Coatings into Food Simulants and Infant Food—A Comprehensive Study. Food Addit. Contam. Part A 2020, 37, 681–703. [Google Scholar] [CrossRef]
- Canellas, E.; Vera, P.; Song, X.-C.; Nerin, C.; Goshawk, J.; Dreolin, N. The Use of Ion Mobility Time-of-Flight Mass Spectrometry to Assess the Migration of Polyamide 6 and Polyamide 66 Oligomers from Kitchenware Utensils to Food. Food Chem. 2021, 350, 129260. [Google Scholar] [CrossRef]
- Kubicova, M.; Puchta, E.; Säger, S.; Hug, C.; Hofmann, S.; Simat, T.J. Styrene-Acrylonitrile-Copolymer and Acrylonitrile-Butadiene-Styrene-Copolymer: A Study on Extractable and Migratable Oligomers. Food Addit. Contam. Part A 2022, 39, 397–414. [Google Scholar] [CrossRef]
- Kubicova, M.; Eckardt, M.; Simat, T.J. Oligomers in Polybutylene Terephthalate for Food Contact—Strategies on Identification, Quantification, and Risk Assessment. Food Addit. Contam. Part A 2022, 39, 2002–2023. [Google Scholar] [CrossRef]
- Djapovic, M.; Milivojevic, D.; Ilic-Tomic, T.; Lješević, M.; Nikolaivits, E.; Topakas, E.; Maslak, V.; Nikodinovic-Runic, J. Synthesis and Characterization of Polyethylene Terephthalate (PET) Precursors and Potential Degradation Products: Toxicity Study and Application in Discovery of Novel PETases. Chemosphere 2021, 275, 130005. [Google Scholar] [CrossRef] [PubMed]
- Brenz, F.; Linke, S.; Simat, T.J. Linear and Cyclic Oligomers in PET, Glycol-Modified PET and TritanTM Used for Food Contact Materials. Food Addit. Contam. Part A 2021, 38, 160–179. [Google Scholar] [CrossRef] [PubMed]
- Brenz, F.; Linke, S.; Simat, T. Linear and Cyclic Oligomers in Polybutylene Terephthalate for Food Contact Materials. Food Addit. Contam. Part A 2018, 35, 583–598. [Google Scholar] [CrossRef] [PubMed]
- Eckardt, M.; Schneider, J.; Simat, T.J. In Vitro Intestinal Digestibility of Cyclic Aromatic Polyester Oligomers from Polyethylene Terephthalate (PET) and Polybutylene Terephthalate (PBT). Food Addit. Contam. Part A 2019, 36, 1882–1894. [Google Scholar] [CrossRef]
- Diamantidou, D.; Mastrogianni, O.; Tsochatzis, E.; Theodoridis, G.; Raikos, N.; Gika, H.; Kalogiannis, S. Liquid Chromatography-Mass Spectrometry Method for the Determination of Polyethylene Terephthalate and Polybutylene Terephthalate Cyclic Oligomers in Blood Samples. Anal. Bioanal. Chem. 2022, 414, 1503–1512. [Google Scholar] [CrossRef]
- Hoppe, M.; De Voogt, P.; Franz, R. Oligomers in Polyethylene Naphthalate and Polybutylene Terephthalate—Identification and Exploring Migration. Food Packag. Shelf Life 2018, 17, 171–178. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaseke, T.; Lujic, T.; Cirkovic Velickovic, T. Nano- and Microplastics Migration from Plastic Food Packaging into Dairy Products: Impact on Nutrient Digestion, Absorption, and Metabolism. Foods 2023, 12, 3043. https://doi.org/10.3390/foods12163043
Kaseke T, Lujic T, Cirkovic Velickovic T. Nano- and Microplastics Migration from Plastic Food Packaging into Dairy Products: Impact on Nutrient Digestion, Absorption, and Metabolism. Foods. 2023; 12(16):3043. https://doi.org/10.3390/foods12163043
Chicago/Turabian StyleKaseke, Tafadzwa, Tamara Lujic, and Tanja Cirkovic Velickovic. 2023. "Nano- and Microplastics Migration from Plastic Food Packaging into Dairy Products: Impact on Nutrient Digestion, Absorption, and Metabolism" Foods 12, no. 16: 3043. https://doi.org/10.3390/foods12163043
APA StyleKaseke, T., Lujic, T., & Cirkovic Velickovic, T. (2023). Nano- and Microplastics Migration from Plastic Food Packaging into Dairy Products: Impact on Nutrient Digestion, Absorption, and Metabolism. Foods, 12(16), 3043. https://doi.org/10.3390/foods12163043





