Effects of Octenyl-Succinylated Chitosan—Whey Protein Isolated on Emulsion Properties, Astaxanthin Solubility, Stability, and Bioaccessibility
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of OSA-Modified Chitosan Oligosaccharide (OSA-COS)
2.3. Characterizations of OSA-Modified Chitosan Oligosaccharide
2.3.1. 1H Nuclear Magnetic Resonance (NMR) Spectroscopy
2.3.2. Fourier-Transform Infrared (FTIR) Spectroscopy
2.3.3. X-ray Diffraction Analysis
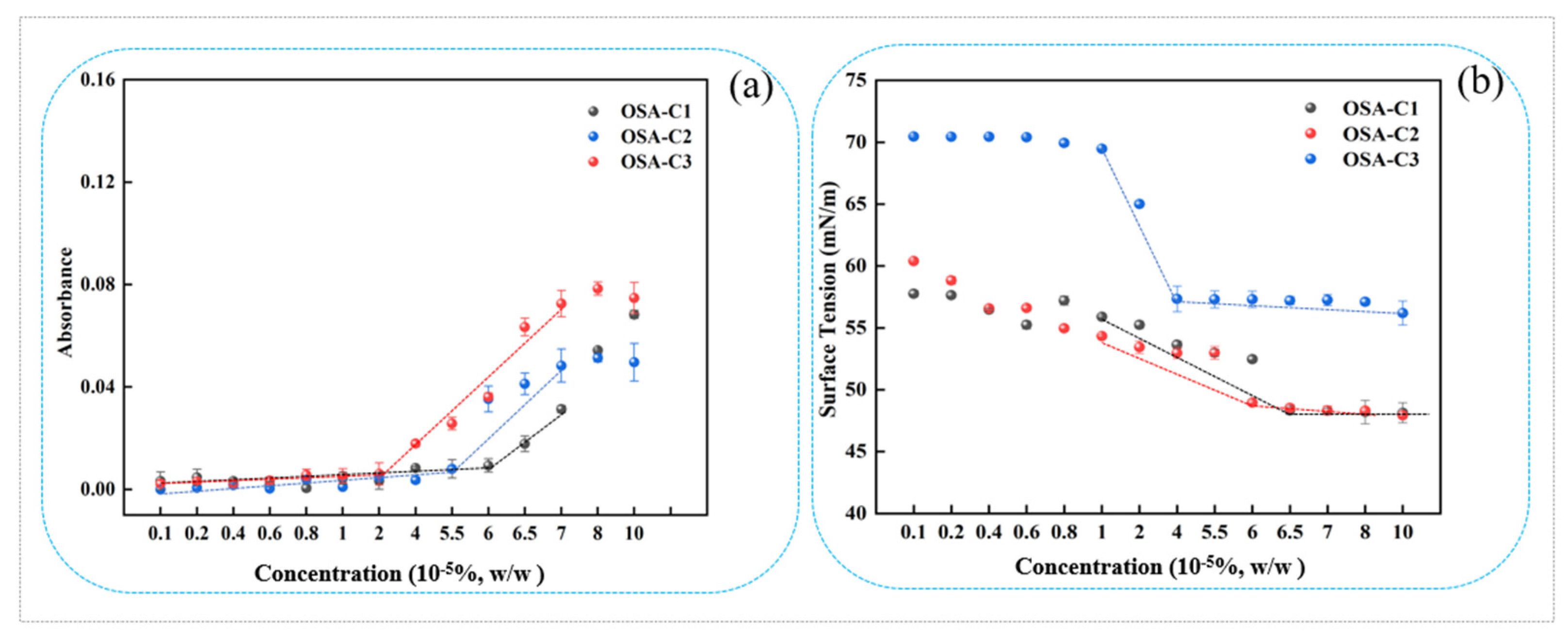
2.4. Critical Aggregation Concentration (CAC)
2.4.1. Dye Solubilization
2.4.2. Surface Tension
2.5. Antimicrobial Activity
2.6. Emulsification Properties
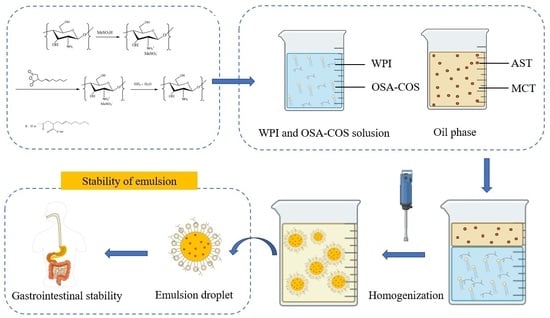
2.6.1. Emulsion Preparation
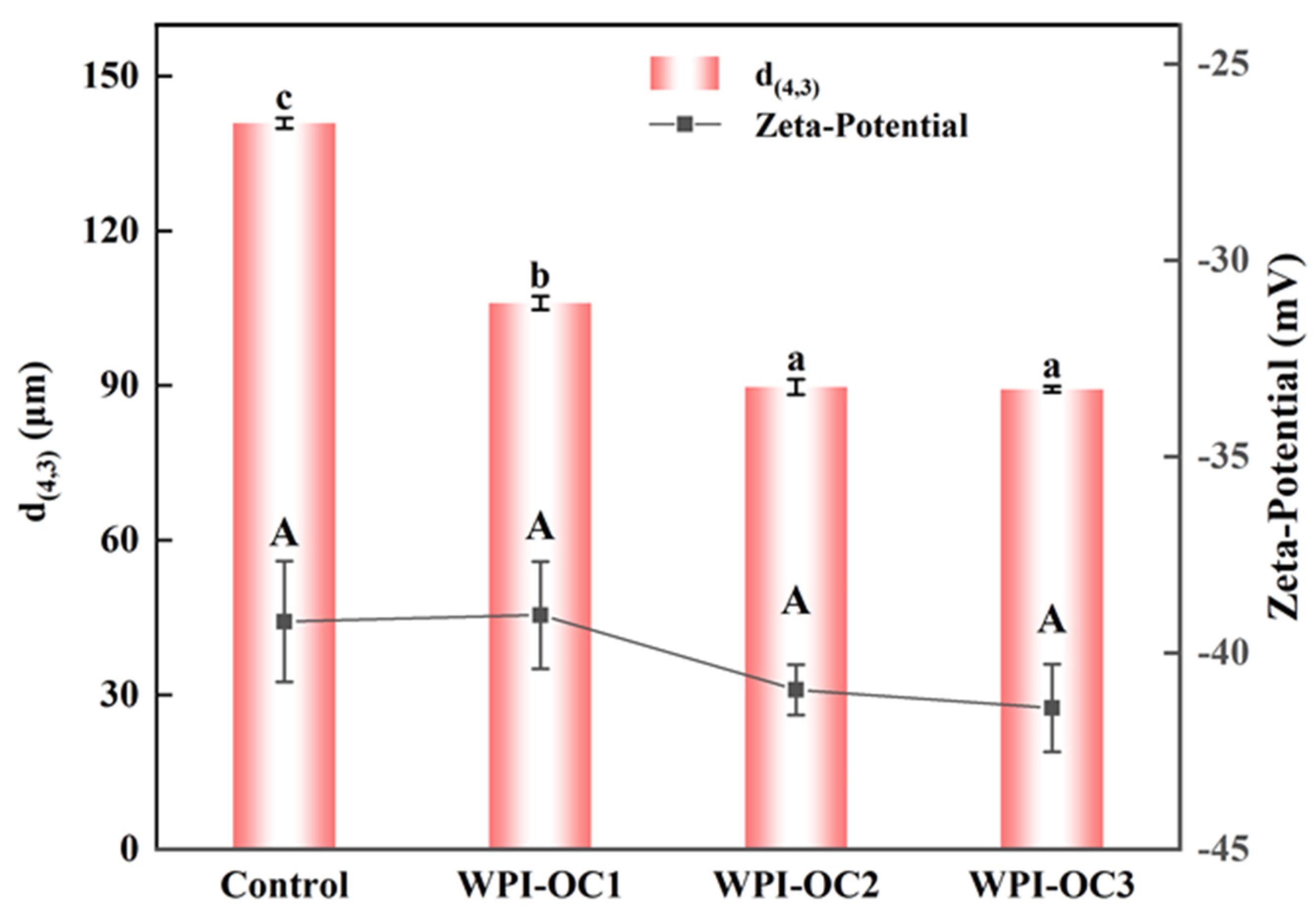
2.6.2. Emulsion Particle Sizes and Zeta Potentials
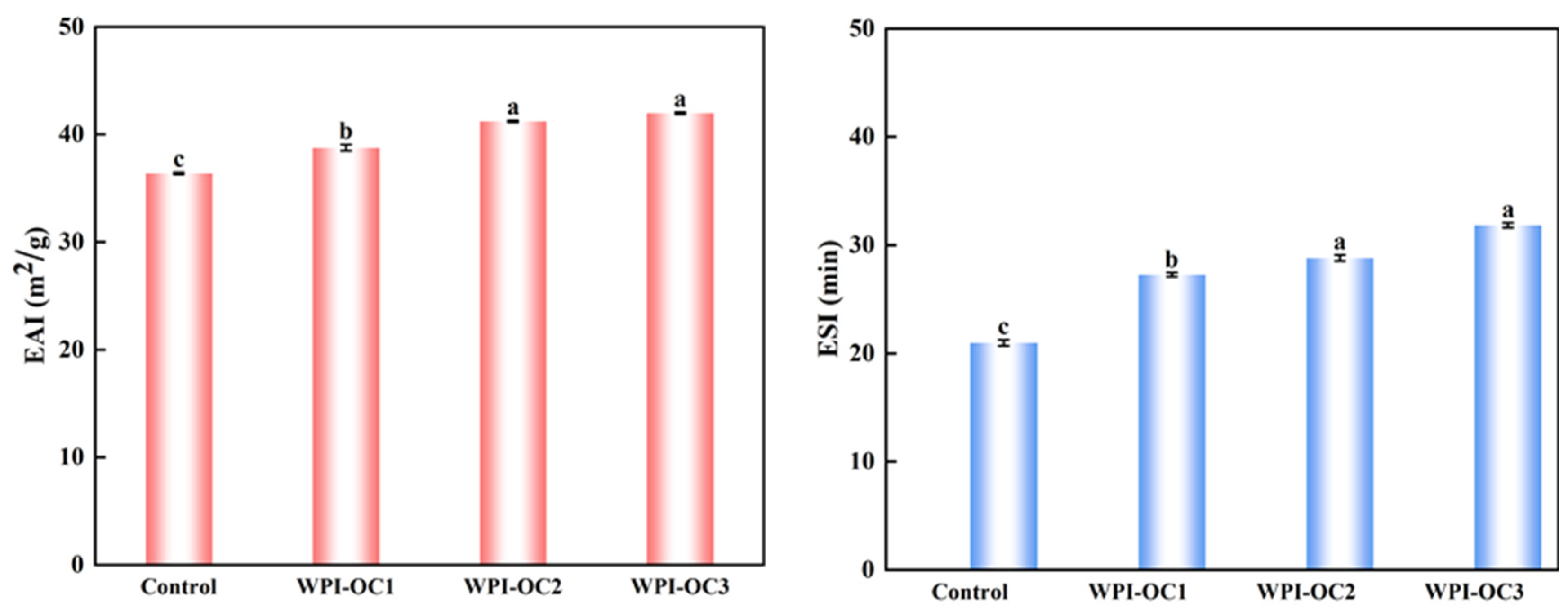
2.6.3. Emulsifying Activity Index (EAI) and Emulsifying Stability Index (ESI)
2.7. In Vitro Digestion
2.8. Astaxanthin Loading Efficiency
3. Results and Discussion
3.1. Synthesis and Characterization of OSA-COS
3.2. CAC
3.3. Emulsion Properties
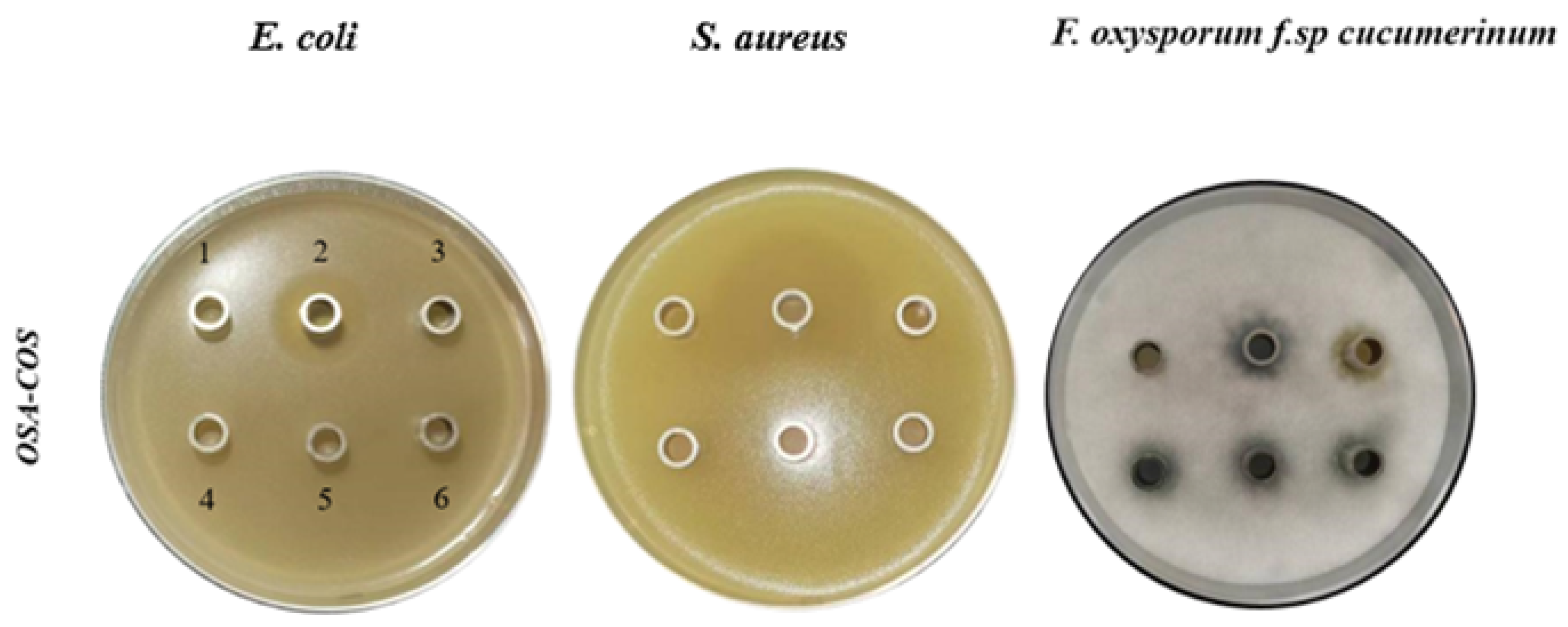
3.4. Antimicrobial Activity of OSA-COS
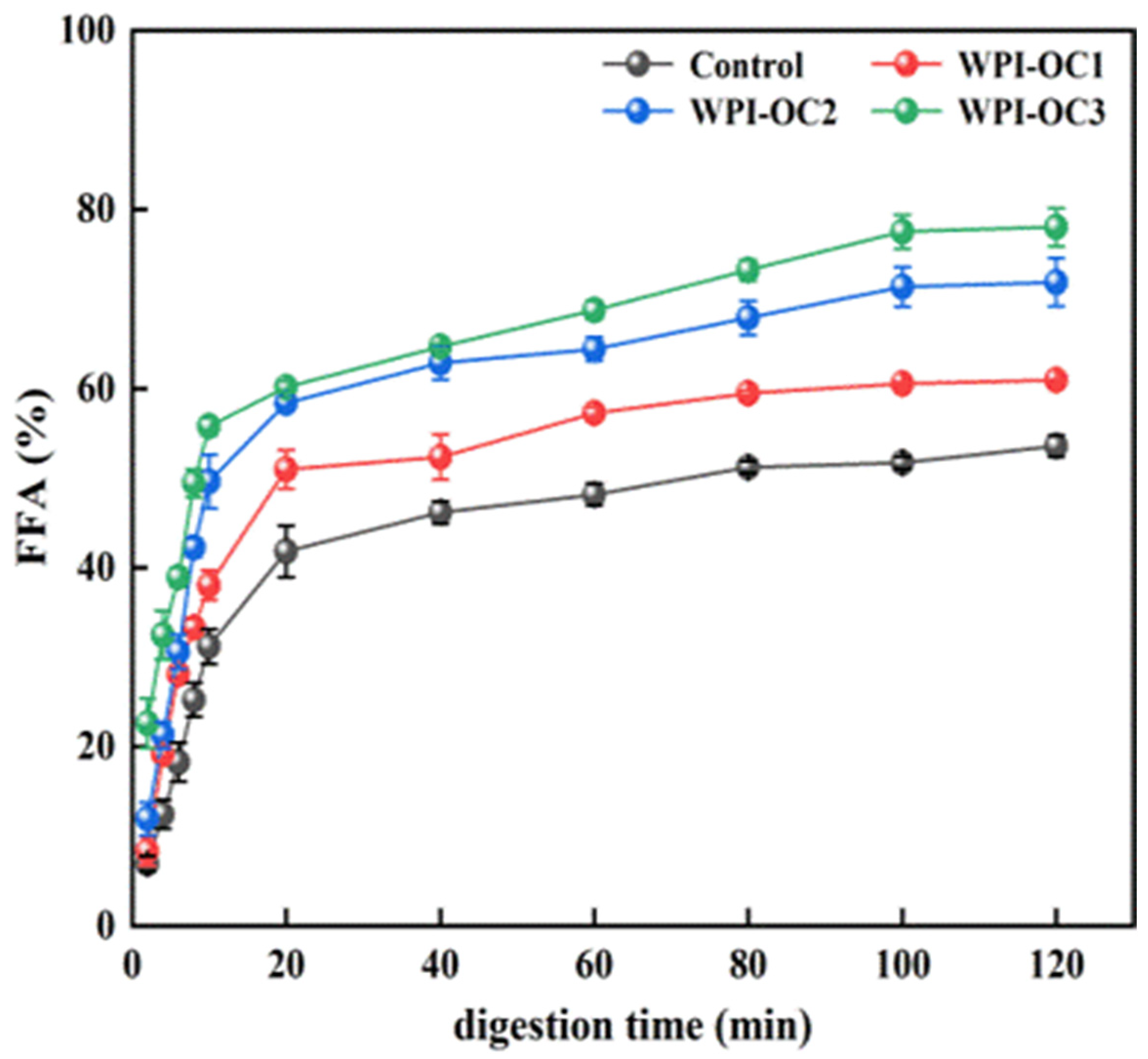
3.5. In Vitro Digestion of the Emulsions
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- López-Cervantes, J.; Sánchez-Machado, D.I. Astaxanthin, lutein, and zeaxanthin. In Nonvitamin Nonmineral Nutritionak Supplements; Nabavi, S.M., Silva, A.S., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 19–25. [Google Scholar]
- Kim, J.H.; Nam, S.W.; Kim, B.W.; Kim, W.J.; Choi, Y.H. Astaxanthin improves the proliferative capacity as well as the osteogenic and adipogenic differentiation potential in neural stem cells. Food Chem. Toxicol. 2010, 48, 1741–1745. [Google Scholar] [CrossRef]
- Jayakumar, R.; Chennazhi, K.P.; Muzzarelli, R.A.A.; Tamura, H.; Nair, S.V.; Selvamurugan, N. Chitosan conjugated DNA nanoparticles in gene therapy. Carbohydr. Polym. 2010, 79, 1–8. [Google Scholar] [CrossRef]
- Zhou, J.; Wen, B.; Xie, H.; Zhang, C.; Bai, Y.; Cao, H.; Che, Q.; Guo, J.; Su, Z. Advances in the preparation and assessment of the biological activities of chitosan oligosaccharides with different structural characteristics. Food Funct. 2021, 12, 926–951. [Google Scholar] [CrossRef]
- Affes, S.; Aranaz, I.; Acosta, N.; Heras, Á.; Nasri, M.; Maalej, H. Physicochemical and biological properties of chitosan derivatives with varying molecular weight produced by chemical depolymerization. Biomass Convers. Biorefinery 2022. [Google Scholar] [CrossRef]
- Hu, K.; Yuan, X.; He, H.; Zhang, H.; Wang, F.; Qiao, J. Pharmacological mechanisms of chitotriose as a redox regulator in the treatment of rat inflammatory bowel disease. Biomed Pharmacother. 2022, 150, 112988. [Google Scholar] [CrossRef]
- Li, R.; Zhu, L.; Liu, D.; Wang, W.; Zhang, C.; Jiao, S.; Wei, J.; Ren, L.; Zhang, Y.; Gou, X.; et al. High molecular weight chitosan oligosaccharide exhibited antifungal activity by misleading cell wall organization via targeting PHR transglucosidases. Carbohydr. Polym. 2022, 285, 119253. [Google Scholar] [CrossRef]
- Gurovic, M.S.V.; Staffolo, M.D.; Montero, M.; Debbaudt, A.; Albertengo, L.; Rodríguez, M.S. Chitooligosaccharides as novel ingredients of fermented foods. Food Funct. 2015, 6, 3437–3443. [Google Scholar] [CrossRef] [PubMed]
- Karagozlu, M.Z.; Karadeniz, F.; Kong, C.S.; Kim, S.K. Aminoethylated chitooligomers and their apoptotic activity on AGS human cancer cells. Carbohydr. Polym. 2012, 87, 1383–1389. [Google Scholar] [CrossRef]
- Liu, X.L.; Xia, W.S.; Jiang, Q.X.; Xu, Y.S.; Yu, P.P. Synthesis, characterization, and antimicrobial activity of kojic acid grafted chitosan oligosaccharide. J. Agric. Food Chem. 2014, 62, 297–303. [Google Scholar] [CrossRef]
- Feng, Y.; Xia, W.S. Preparation, characterization and antibacterial activity of water-soluble O-fumaryl-chitosan. Carbohydr. Polym. 2011, 83, 1169–1173. [Google Scholar] [CrossRef]
- Khana, I.; Ullahb, S.; Oha, D.H. Chitosan grafted monomethyl fumaric acid as a potential food preservative. Carbohydr. Polym. 2016, 152, 87–96. [Google Scholar] [CrossRef]
- Hu, B.; Hu, J.; Han, L.Y.; Cao, J.J.; Nishinari, K.; Yang, J.X.; Fang, Y.P.; Li, D.M. Conformational transition and gelation of κ-carrageenan in electrostatic complexation with β-lactoglobulin aggregates. Food Hydrocoll. 2021, 118, 106764. [Google Scholar] [CrossRef]
- Bosnea, L.A.; Moschakis, T.; Biliaderis, C.G. Complex coacervation as a novel microencapsulation technique to improve viability of probiotics under different stresses. Food Bioprocess Technol. 2014, 7, 2767–2781. [Google Scholar] [CrossRef]
- Devi, N.; Sarmah, M.; Khatun, B.; Maji, T.K. Encapsulation of active ingredients in polysaccharide-protein complex coacervates. Adv. Colloid Interface Sci. 2017, 239, 136–145. [Google Scholar] [CrossRef]
- Eratte, D.; Dowling, K.; Barrow, C.J.; Adhikari, B.P. In-vitro digestion of probiotic bacteria and omega-3 oil co-microencapsulated in whey protein isolate-gum Arabic complex coacervates. Food Chem. 2017, 227, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, S.M.H.; Emam-Djomeh, Z.; Sabatino, P.; Meeren, P.V. Nanocomplexes arising from protein-polysaccharide electrostatic interaction as a promising carrier for nutraceutical compounds. Food Hydrocoll. 2015, 50, 16–26. [Google Scholar] [CrossRef]
- Jain, A.; Thakur, D.; Ghoshal, G.; Katare, O.P.; Shivhare, U.S. Characterization of microcapsulated beta-carotene formed by complex coacervation using casein and gum tragacanth. Int. J. Biol. Macromol. 2016, 87, 101–113. [Google Scholar] [CrossRef]
- Koupantsis, T.; Pavlidou, E.; Paraskevopoulou, A. Flavour encapsulation in milk proteins—CMC coacervate-type complexes. Food Hydrocoll. 2014, 37, 134–142. [Google Scholar] [CrossRef]
- Xiao, Z.B.; Liu, W.L.; Zhu, G.Y.; Zhou, R.J.; Niu, Y.W. A review of the preparation and application of flavour and essential oils microcapsules based on complex coacervation technology. J. Sci. Food Agric. 2014, 94, 1482–1494. [Google Scholar] [CrossRef]
- Cuevas-Bernardino, J.C.; Leyva-Gutierrez, F.M.A.; Vernon-Carter, E.J.; Lobato-Calleros, C.; Román-Guerrero, A.; Davidov-Pardo, G. Formation of biopolymer complexes composed of pea protein and mesquite gum—Impact of quercetin addition on their physical and chemical stability. Food Hydrocoll. 2018, 77, 736–745. [Google Scholar] [CrossRef]
- Huang, J.; Feng, X.; Zhang, S.; Wang, L.Z.; Yue, J.J.; Chu, L.L. Preparation and characterization of astaxanthin-loaded microcapsules and its application in effervescent tablets. J. Sci. Food Agric. 2023, 103, 1421–1431. [Google Scholar] [CrossRef]
- Liu, Q.R.; Qi, J.R.; Yin, S.W.; Wang, J.M.; Guo, J.; Feng, J.L.; Cheng, M.; Cao, J.; Weng, J.Y.; Yang, X.Q. The influence of heat treatment on acid-tolerant emulsions prepared from acid soluble soy protein and soy soluble polysaccharide complexes. Food Res. Int. 2016, 89 Pt 1, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Zeeb, B.; Mi-Yeon, L.; Gibis, M.; Weiss, J. Growth phenomena in biopolymer complexes composed of heated WPI and pectin. Food Hydrocoll. 2018, 74, 53–61. [Google Scholar] [CrossRef]
- Puerta-Gomez, A.; Castell-Perez, M.E. Visual spectroscopy method to evaluate entrapment efficiency of electrostatically precipitated proteins in combination with octenyl succinic anhydrate (OSA)-modified polysaccharides. Food Hydrocoll. 2017, 63, 160–169. [Google Scholar] [CrossRef]
- Puerta-Gomez, A.F.; Castell-Perez, M.E. Studies on self-assembly interactions of proteins and octenyl succinic anhydrate (OSA)-modified depolymerized waxy rice starch using rheological principles. J. Appl. Polym. Sci. 2016, 133, 43603–43613. [Google Scholar] [CrossRef]
- Han, L.Y.; Sun, J.; Williams, P.A.; Yang, J.X.; Zhang, S.B. Octenyl-succinylated inulins for the delivery of hydrophobic drug. Int. J. Biol. Macromol. 2022, 221, 1112–1120. [Google Scholar] [CrossRef] [PubMed]
- Han, L.Y.; Hu, B.; Ratcliffe, I.; Senanc, C.; Yang, J.X.; Williams, P.A. Octenyl-succinylated inulin for the encapsulation and release of hydrophobic compounds. Carbohydr. Polym. 2020, 238, 116199. [Google Scholar] [CrossRef] [PubMed]
- Han, L.Y.; Ratcliffe, I.; Williams, P.A. Self-assembly and emulsification properties of hydrophobically modified inulin. J. Agric. Food Chem. 2015, 63, 3709–3715. [Google Scholar] [CrossRef]
- Han, L.Y.; Ratcliffe, I.; Williams, P.A. Synthesis, characterisation and physicochemical properties of hydrophobically modified inulin using long-chain fatty acyl chlorides. Carbohydr. Polym. 2017, 178, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.A.; Liu, T.; Zhong, G.Q. Synthesis, characterization and applications of copper (II) complexes with Schiff bases derived from chitooligosaccharide and iodosubstituted salicylaldehyde. Carbohydr. Polym. 2019, 224, 115151. [Google Scholar] [CrossRef]
- Sun, J.; Liu, T.M.; Zhang, F.; Huang, Y.Q.; Zhang, Y.; Xu, B. Tea polyphenols on emulsifying and antioxidant properties of egg white protein at acidic and neutral pH conditions. LWT 2022, 153, 112537. [Google Scholar] [CrossRef]
- Li, R.Y.; Dai, T.T.; Tan, Y.B.; Fu, G.M.; Wan, Y.; Liu, C.M.; McClements, D.J. Fabrication of pea protein-tannic acid complexes: Impact on formation, stability, and digestion of flaxseed oil emulsions. Food Chem. 2020, 310, 125828. [Google Scholar] [CrossRef] [PubMed]
- Xia, Z.Y.; Han, Y.H.; Du, H.J.; McClements, D.J.; Tang, Z.H.; Xiao, H. Exploring the effects of carrier oil type on in vitro bioavailability of β-carotene: A cell culture study of carotenoid-enriched nanoemulsions. LWT 2020, 134, 110224. [Google Scholar] [CrossRef]
- Ahmed, K.; Li, Y.; McClements, D.J.; Xiao, H. Nanoemulsion- and emulsion-based delivery systems for curcumin: Encapsulation and release properties. Food Chem. 2012, 132, 799–807. [Google Scholar] [CrossRef]
- Xu, G.R.; Wang, C.N.; Yao, P. Stable emulsion produced from casein and soy polysaccharide compacted complex for protection and oral delivery of curcumin. Food Hydrocoll. 2017, 71, 108–117. [Google Scholar] [CrossRef]
- Bai, Y.; Shi, Y.C. Structure and preparation of octenyl succinic esters of granular starch, microporous starch and soluble maltodextrin. Carbohydr. Polym. 2011, 83, 520–527. [Google Scholar] [CrossRef]
- Wei, L.J.; Zhang, J.J.; Luan, F.; Tan, W.Q.; Li, Q.; Dong, F.; Guo, Z.Y. Synthesis, characterization, and the antifungal property of aminoethyl chitosan quaternary ammonium salts. Starch 2018, 70, 1700266. [Google Scholar] [CrossRef]
- Cai, J.P.; Dang, Q.F.; Liu, C.S.; Fan, B.; Yan, J.Q.; Xu, Y.Y.; Li, J.J. Preparation and characterization of N-benzoyl-O-acetyl-chitosan. Int. J. Biol. Macromol. 2015, 77, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Ping, Q.N.; Zhang, H.J.; Shen, J. Synthesis and characterization of water-soluble O-succinyl-chitosan. Eur. Polym. J. 2003, 39, 1629–1634. [Google Scholar] [CrossRef]
- Xiao, B.; Wan, Y.; Wang, X.Y.; Zha, Q.C.; Liu, H.M.; Qiu, Z.Y.; Zhang, S.M. Synthesis and characterization of N-(2-hydroxy)propyl-3-trimethyl ammonium chitosan chloride for potential application in gene delivery. Colloids Surf. B Biointerfaces 2012, 91, 168–174. [Google Scholar] [CrossRef]
- Feng, F.; Liu, Y.; Zhao, B.Y.; Hu, K. Characterization of half N-acetylated chitosan powders and films. Procedia Eng. 2012, 27, 718–732. [Google Scholar] [CrossRef]
- Anbinder, P.; Macchi, C.; Amalvy, J.; Somoza, A. Chitosan-graft-poly (n-butyl acrylate) copolymer: Synthesis and characterization of a natural/synthetic hybrid material. Carbohydr. Polym. 2016, 145, 86–94. [Google Scholar] [CrossRef]
- Ogawa, K.; Yui, T.; Miya, M. Dependence on the preparation procedure of the polymorphism and crystallinity of chitosan membranes. Biosci. Biotechnol. Biochem. 1992, 56, 858–862. [Google Scholar] [CrossRef] [PubMed]
- Kokubun, S.; Ratcliffe, I.; Williams, P.A. Synthesis, characterization and self-assembly of biosurfactants based on hydrophobically modified inulins. Biomacromolecules 2013, 14, 2830–2836. [Google Scholar] [CrossRef] [PubMed]
- Kokubun, S.; Ratcliffe, I.; Williams, P.A. The emulsification properties of octenyl- and dodecenyl- succinylated inulins. Food Hydrocoll. 2015, 50, 145–149. [Google Scholar] [CrossRef]
- Kokubun, S.; Ratcliffe, I.; Williams, P.A. The interfacial, emulsification and encapsulation properties of hydrophobically modified inulin. Carbohydr. Polym. 2018, 194, 18–23. [Google Scholar] [CrossRef]
- Yue, L.; Wang, M.; Khan, I.M.; Niazi, S.; Wang, B.; Ma, X.Y.; Wang, Z.P.; Xia, W.S. Preparation and characterization of chitosan oligosaccharide derivatives containing cinnamyl moieties with enhanced antibacterial activities. LWT 2021, 147, 111663. [Google Scholar] [CrossRef]
- Jiang, W.; Zhou, X.H.; Su, Y. Preparation of viscose fiber modified with silkworm pupa chitosan oligosaccharide by β-glucosaccharase. Text. Res. J. 2019, 89, 4461–4475. [Google Scholar] [CrossRef]
- Liu, W.L.; Ye, A.Q.; Liu, C.M.; Liu, W.; Singh, H. Structure and integrity of liposomes prepared from milk- or soybean-derived phospholipids during in vitro digestion. Food Res. Int. 2012, 48, 499–506. [Google Scholar] [CrossRef]
- Li, P.; Li, H.; Hou, L.F.; Chang, Z.G.; Li, Y.Y.; Li, X.Q. Gastrointestinal digestive fate of whey protein isolate coated liposomes loading astaxanthin: Lipolysis, release, and bioaccessibility. Food Biosci. 2022, 45, 101464. [Google Scholar] [CrossRef]


| Sample | Mol (OSA:COS) | DS | CAC (10−5%) |
|---|---|---|---|
| OSA-C1 | 1:6 | 0.14 | 6.5 |
| OSA-C2 | 1:8 | 0.16 | 6.0 |
| OSA-C3 | 1:10 | 0.19 | 4 |
| Sample | Bacteria Name/Inhibition Zone (mm ± SD) | ||
|---|---|---|---|
| E. coli | S. aureus | Fusarium oxysporum f.sp cucumerinum | |
| potassium sorbate | 27.1 ± 0.66 | 17.5 ± 0.57 | |
| streptomycin | 45.7 ± 0.66 | ||
| COS | NA | NA | 15.6 ± 0.33 |
| OSA-C1 | NA | NA | 13.1 ± 0.09 |
| OSA-C2 | NA | NA | 14.3 ± 0.04 |
| OSA-C3 | NA | NA | 13.5 ± 0.09 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, L.; Zhai, R.; Hu, B.; Yang, J.; Li, Y.; Xu, Z.; Meng, Y.; Li, T. Effects of Octenyl-Succinylated Chitosan—Whey Protein Isolated on Emulsion Properties, Astaxanthin Solubility, Stability, and Bioaccessibility. Foods 2023, 12, 2898. https://doi.org/10.3390/foods12152898
Han L, Zhai R, Hu B, Yang J, Li Y, Xu Z, Meng Y, Li T. Effects of Octenyl-Succinylated Chitosan—Whey Protein Isolated on Emulsion Properties, Astaxanthin Solubility, Stability, and Bioaccessibility. Foods. 2023; 12(15):2898. https://doi.org/10.3390/foods12152898
Chicago/Turabian StyleHan, Lingyu, Ruiyi Zhai, Bing Hu, Jixin Yang, Yaoyao Li, Zhe Xu, Yueyue Meng, and Tingting Li. 2023. "Effects of Octenyl-Succinylated Chitosan—Whey Protein Isolated on Emulsion Properties, Astaxanthin Solubility, Stability, and Bioaccessibility" Foods 12, no. 15: 2898. https://doi.org/10.3390/foods12152898
APA StyleHan, L., Zhai, R., Hu, B., Yang, J., Li, Y., Xu, Z., Meng, Y., & Li, T. (2023). Effects of Octenyl-Succinylated Chitosan—Whey Protein Isolated on Emulsion Properties, Astaxanthin Solubility, Stability, and Bioaccessibility. Foods, 12(15), 2898. https://doi.org/10.3390/foods12152898