A Physicochemical Study of the Antioxidant Activity of Corn Silk Extracts
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methodology
2.2.1. Corn Silk Flavonoids Extraction and Determination
2.2.2. Corn Silk Polysaccharides Extraction and Determination
2.2.3. Corn Silk Steroids Extraction and Determination
2.2.4. EPR Spin-Trapping Measurement
2.2.5. DPPH Assay
2.2.6. ABTS Assay
2.2.7. FRAP Assay
2.2.8. CUPRAC Assay
2.2.9. HPLC Polyphenol Analysis
2.2.10. Statistical Data Analysis
3. Results and Discussion
3.1. Corn Silk Extraction Process Optimization
3.1.1. Corn Silk Flavonoids Extraction and Determination
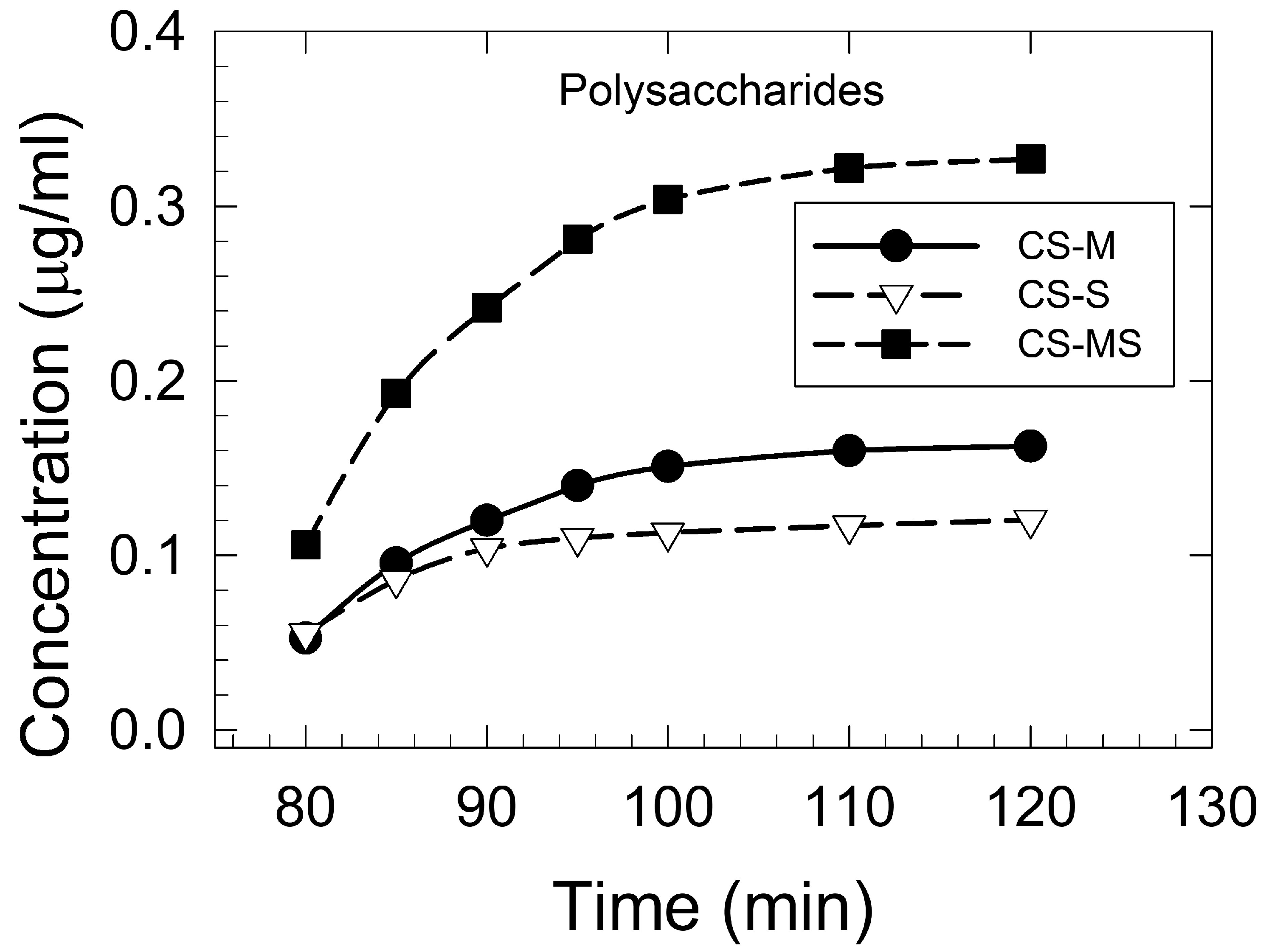
3.1.2. Corn Silk Polysaccharides Extraction and Determination
3.1.3. Corn Silk Steroids Extraction and Determination
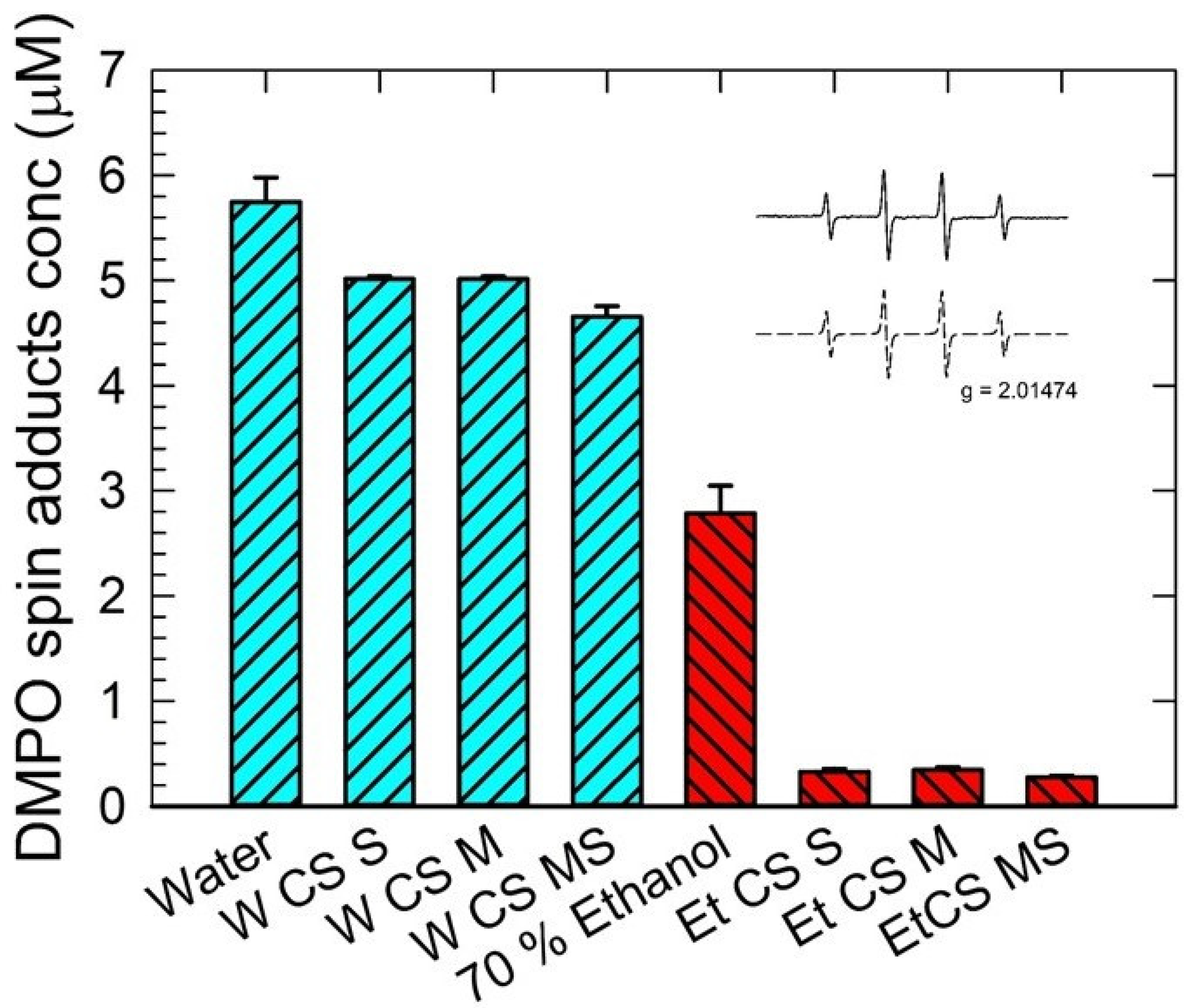
3.2. EPR Spin-Trapping Measurement
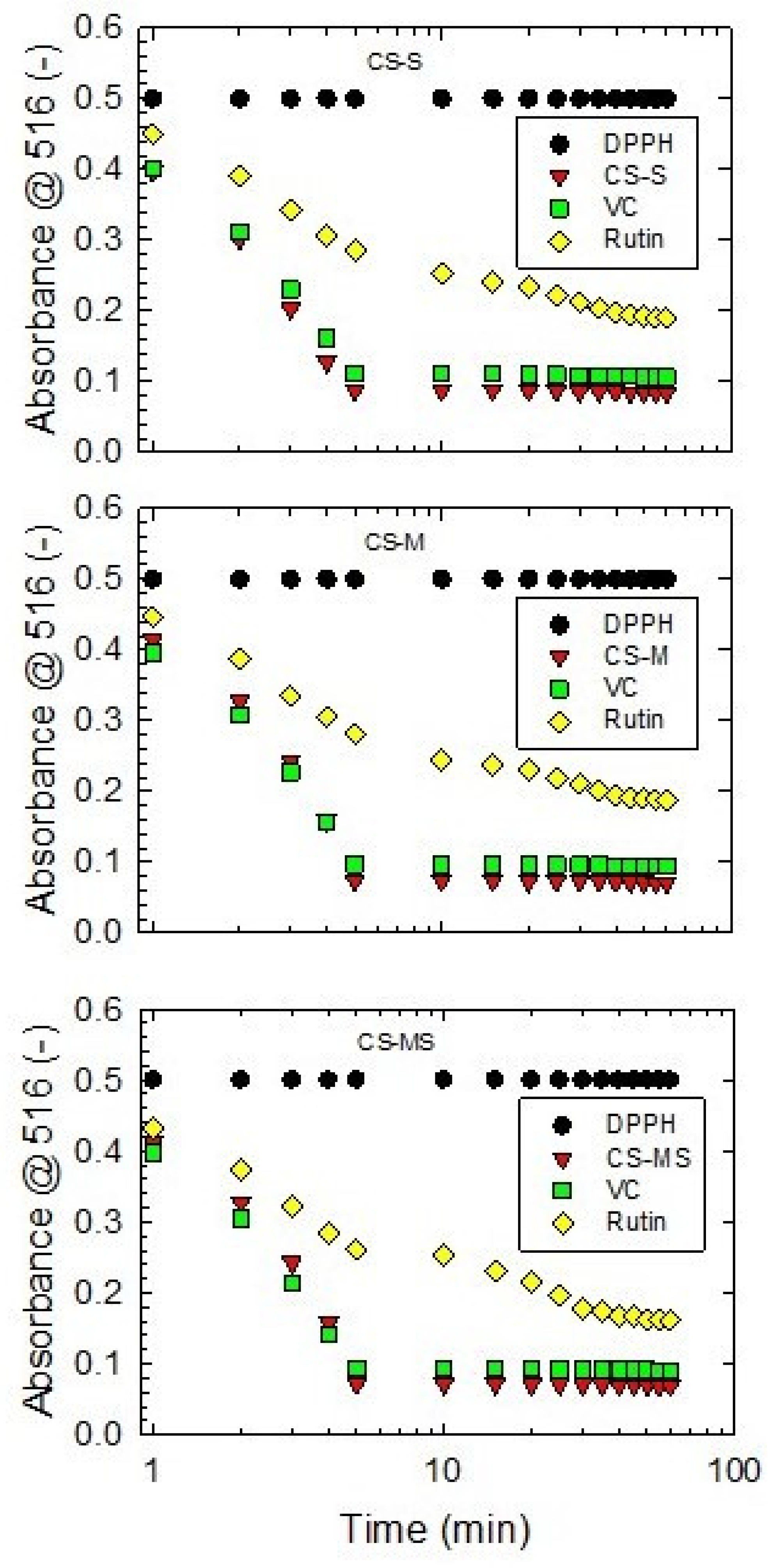
3.3. DPPH Assay
3.4. ABTS Assay
3.5. FRAP and CUPRAC Assay
3.6. HPLC Polyphenol Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Poprac, P.; Jomova, K.; Simunkova, M.; Kollar, V.; Rhodes, C.; Valko, M. Targeting free radicals in oxidative stress-related human diseases. Trends Pharmacol. Sci. 2017, 38, 592–607. [Google Scholar] [CrossRef]
- Cazzola, R.; Cestaro, B. Red wine polyphenols protect n−3 more than n−6 polyunsaturated fatty acid from lipid peroxidation. Food Res. Int. 2011, 44, 3065–3071. [Google Scholar] [CrossRef]
- Sarepoua, E.; Tangwongchai, R.; Suriharn, B.; Lertrat, K. Influence of variety and harvest maturity on phytochemical content in corn silk. Food Chem. 2015, 169, 424–429. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Hu, Z.; Wang, X.; Wu, M.; Zhou, H.; Zhang, Y. Characterization of a polysaccharide with antioxidant and anti-cervical cancer potentials from the corn silk cultivated in Jilin province. Int. J. Biol. Macromol. 2020, 155, 1105–1113. [Google Scholar] [CrossRef]
- Zhang, D.W.; Wang, Y.Z.; Liu, H.L. Corn silk extract inhibit the formation of N-epsilon-carboxymethyllysine by scavenging glyoxal/methyl glyoxal in a casein glucose-fatty acid model system. Food Chem. 2020, 309, 125708. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Li, Y.; Meng, X.; Zhou, T.; Zhou, Y.; Zheng, J.; Zhang, J.-J.; Li, H.-B. Natural antioxidants in foods and medicinal plants: Extraction, assessment and resources. Int. J. Mol. Sci. 2017, 18, 96. [Google Scholar] [CrossRef]
- Limmatvapirat, C.; Nateesathittarn, C.; Dechasathian, K.; Moohummad, T.; Chinajitphan, P.; Limmatvapirat, S. Phytochemical analysis of baby corn silk extracts. J. Ayurveda Integ. Med. 2020, 11, 344–351. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.N.; Wang, Y.J.; Li, R.L.; Li, S.; Zhang, M.; He, C.; Chen, H. The structural characteristic of acidic-hydrolyzed corn silk polysaccharides and its protection on the H2O2-injured intestinal epithelial cells. Food Chem. 2021, 356, 129691. [Google Scholar] [CrossRef]
- Li, P.; Lapčík, L.; Lapčíková, B.; Kalytchuk, S. Physico-chemical study of flavonoids from different matureness corn silk material. Potravinarstvo 2018, 12, 347–354. [Google Scholar] [CrossRef]
- Shahzad, M.K.; Shahzad, M.A.; Qadeer, U.; Mehmood, A. Investigation of phytochemical profiling and therapeutic effects of corn silk against diabetes in human male subjects. Pak. J. Pharmaceut. Sci. 2022, 35, 1699–1703. [Google Scholar]
- Liu, J.; Wang, C.N.; Wang, Z.Z.; Zhang, C.; Lu, S.; Liu, J. The antioxidant and free-radical scavenging activities of extract and fractions from corn silk (Zea mays L.) and related flavone glycosides. Food Chem. 2011, 126, 261–269. [Google Scholar] [CrossRef]
- Hasanudin, K.; Hashim, P.; Mustafa, S. Corn silk (Stigma maydis) in healthcare: A Phytochemical and Pharmacological Review. Molecules 2012, 17, 9697–9715. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.; Li, N.; Wang, Q.; Zhou, J.; Liu, J.; Zhang, M.; He, C.; Chen, H. Effect of Fe (III), Zn (II), and Cr (III) complexation on the physicochemical properties and bioactivities of corn silk polysaccharide. Int. J. Biol. Macromol. 2021, 189, 847–856. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.; Inbaraj, B.S.; Kaur, S.; Rasane, P.; Nanda, V. Phytochemical analysis and characterization of Corn Silk (Zea mays, G5417). Agronomy 2022, 12, 777. [Google Scholar] [CrossRef]
- Velazquez, D.; Xavier, H.S.; Batista, J.E.M.; de Castro-Chaves, C. Zea mays L. extracts modify glomerular function and potassium urinary excretion in conscious rats. Phytomedicine 2005, 12, 363–369. [Google Scholar] [CrossRef]
- Singh, J.; Rasane, P.; Nanda, V.; Kaur, S. Bioactive compounds of corn silk and their role in management of glycaemic response. J. Food Sci. Tech. 2022, 60, 1–16. [Google Scholar] [CrossRef]
- Hu, Q.; Zhang, L.-J.; Li, Y.-N.; Ding, Y.-J.; Li, F.-L. Purification and anti-fatigue activity of flavonoids from corn silk. Int. J. Phys. Sci. 2010, 5, 321–326. [Google Scholar]
- Bai, H.; Hai, C.; Xi, M.; Liang, X.; Liu, R. Protective effect of maize silks (Maydis stigma) ethanol extract on radiation-induced oxidative stress in mice. Plant Foods Hum. Nutr. 2010, 65, 271–276. [Google Scholar] [CrossRef]
- Vranješ, M.; Popović, B.M.; Štajner, D.; Ivetić, V.; Mandić, A.; Vranješ, D. Effects of bearberry, parsley and corn silk extracts on diuresis, electrolytes composition, antioxidant capacity and histopathological features in mice kidneys. J. Funct. Foods 2016, 21, 272–282. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, L.Y.; Ma, Z.S.; Cheng, J.; Liu, J.B. Anti-diabetic, anti-oxidant and anti-hyperlipidemic activities of flavonoids from corn silk on STZ-induced diabetic mice. Molecules 2016, 21, 7. [Google Scholar] [CrossRef]
- Mohsin, S.; Akhtar, N.; Mahmood, T.; Khan, H.; Mustafa, R. Formulation and stability of topical water in oil emulsion containing corn silk extract, Trop. J. Pharmaceut. Res. 2016, 15, 1115–1121. [Google Scholar]
- Aukkanit, N.; Kemngoen, T.; Ponharn, N. Utilization of corn silk in low fat meatballs and its characteristics. Procedia Soc. Behav. Sci. 2015, 197, 1403–1410. [Google Scholar] [CrossRef]
- Mačanga, J.; Popelka, P.; Koréneková, B.; Maskaľová, I.; Klempová, T.; Fečkaninová, A.; Mellen, M.; Marcinčáková, D.; Čertík, M.; Marcinčák, S. Effect of feeding of prefermented bioproduct containing gamma-linolenic acid and beta-carotene on selected parameters of broiler chicken meat quality. Potrvin. Slovak J. Food Sci. 2017, 11, 466–471. [Google Scholar] [CrossRef]
- Fiol, M.; Adermann, S.; Neugart, S.; Mügge, C.; Schreiner, M.; Krumbein, A.; Kroh, L.W. Highly glycosylated and acylated flavonols isolated from kale (Brassica oleracea var. sabellica)—Structure–antioxidant activity relationship. Food Res. Int. 2012, 47, 80–89. [Google Scholar] [CrossRef]
- Wang, J.; Zhou, W.-Y.; Huang, X.-X.; Song, S.-J. Flavonoids with antioxidant and tyrosinase inhibitory activity from corn silk (Stigma maydis). Nat. Prod. Res. 2023, 37, 835–839. [Google Scholar] [CrossRef]
- Finley, J.W.; Kong, A.-N.; Hintze, K.J.; Jeffery, E.; Ji, L.L.; Lei, X.G. Antioxidants in foods: State of the science important to the food industry. J. Agric. Food Chem. 2011, 59, 6837–6846. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Amy Cho, S.-A.K.; Kim, M.; Song, M.; Han, H.W.; Shin, E.-J.; Park, E.; Lee, S.-M. Potential photoprotective effect of dietary corn silk extract on ultraviolet B-induced skin damage. Molecules 2019, 24, 2587. [Google Scholar] [CrossRef]
- Zilic, S.; Jankovic, M.; Basic, Z.; Vancetovic, J.; Maksimovic, V. Antioxidant activity, phenolic profile, chlorophyll and mineral matter content of corn silk (Zea mays L): Comparison with medicinal herbs. J. Cereal Sci. 2016, 69, 363–370. [Google Scholar] [CrossRef]
- Maksimović, Z.; Malenčić, Đ.; Kovačević, N. Polyphenol contents and antioxidant activity of Maydis stigma extracts. Bioresour. Technol. 2005, 96, 873–877. [Google Scholar] [CrossRef]
- Fallovo, C.; Schreiner, M.; Schwarz, D.; Colla, G.; Krumbein, A. Phytochemical changes induced by different nitrogen supply forms and radiation levels in two leafy Brassica species. J. Agric. Food Chem. 2011, 59, 4198–4207. [Google Scholar] [CrossRef]
- Jia, Y.; Gao, X.; Xue, Z.; Wang, Y.; Lu, Y.; Zhang, M.; Panichayupakaranant, P.; Chen, H. Characterization, antioxidant activities, and inhibition on α-glucosidase activity of corn silk polysaccharides obtained by different extraction methods. Int. J. Biol. Macromol. 2020, 163, 1640–1648. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wang, M. Optimization of deep eutectic solvent-based ultrasound-assisted extraction of polysaccharides from Dioscorea opposita Thunb. Int. J. Biol. Macromol. 2017, 95, 675–681. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Geng, L.-L.; Lu, H.-Q.; Fan, X.-D. Ultrasound-synergized electrostatic field extraction of total flavonoids from Hemerocallis citrina baroni. Ultrason. Sonochem. 2017, 34, 571–579. [Google Scholar] [CrossRef] [PubMed]
- Périno, S.; Pierson, J.T.; Ruiz, K.; Cravotto, G.; Chemat, F. Laboratory to pilot scale: Microwave extraction for polyphenols lettuce. Food Chem. 2016, 204, 108–114. [Google Scholar] [CrossRef]
- Jing, S.; Li, Q.; Zheng, L.; Wang, S.; Yue, L.; Fan, S.; Tao, G. Dynamic high pressure microfluidization-assisted extraction and bioactivities of Cyperus esculentus (C. esculentus L.) leaves flavonoids. Food Chem. 2016, 192, 319–327. [Google Scholar] [CrossRef]
- Maran, J.P.; Manikandan, S.; Thirugnanasambandham, K.; Nivetha, C.V.; Dinesh, R. Box–Behnken design based statistical modeling for ultrasound-assisted extraction of corn silk polysaccharide. Carbohydr. Polym. 2013, 92, 604–611. [Google Scholar] [CrossRef]
- Chen, S.; Chen, H.; Tian, J.; Wang, J.; Wang, Y.; Xing, L. Enzymolysis-ultrasonic assisted extraction, chemical characteristics and bioactivities of polysaccharides from corn silk. Carbohydr. Polym. 2014, 101, 332–341. [Google Scholar] [CrossRef]
- Li, P.; Lapčík, L.; Lapčíková, B.; Kalytchuk, S. Physico-chemical study of steroids from different matureness corn silk material. Potravinarstvo 2019, 13, 658–664. [Google Scholar] [CrossRef]
- Cömert, E.D.; Gökmen, V. Evolution of food antioxidants as a core topic of food science for a century. Food Res. Int. 2018, 105, 76–93. [Google Scholar] [CrossRef]
- Joubert, E.; Winterton, P.; Britz, T.J.; Ferreira, D. Superoxide anion and α, α-diphenyl-β-picrylhydrazyl radical scavenging capacity of rooibos (Aspalathus linearis) aqueous extracts, crude phenolic fractions, tannin and flavonoids. Food Res. Int. 2004, 37, 133–138. [Google Scholar] [CrossRef]
- Oroian, M.; Escriche, I. Antioxidants: Characterization, natural sources, extraction and analysis. Food Res. Int. 2015, 74, 10–36. [Google Scholar] [CrossRef] [PubMed]
- Rahman, N.A.; Rosli, W.I.W. Nutritional compositions and antioxidative capacity of the silk obtained from immature and mature corn. J. King Saud Uni. Sci. 2014, 26, 119–127. [Google Scholar] [CrossRef]
- Peng, K.; Zhang, S.-Y.; Zhou, H.-L. Toxicological evaluation of the flavonoid-rich extract from Maydis stigma: Subchronic toxicity and genotoxicity studies in mice. J. Ethnopharmacol. 2016, 192, 161–169. [Google Scholar] [CrossRef]
- Marques, G.S.; Leão, W.F.; Lyra, M.A.; Peixoto, M.S.; Monteiro, R.P.M.; Rolim, L.A.; Xavier, H.S.; Neto, P.J.R. Comparative evaluation of UV/VIS and HPLC analytical methodologies applied for quantification of flavonoids from leaves of Bauhinia forficata. Rev. Bras. Farmacogn. 2013, 23, 51–57. [Google Scholar] [CrossRef]
- Hossain, M.B.; Tiwari, B.K.; Gangopadhyay, N.; O’Donnell, C.P.; Brunton, N.; Rai, D. Ultrasonic extraction of steroidal alkaloids from potato peel waste. Ultrason. Sonochem. 2014, 21, 1470–1476. [Google Scholar] [CrossRef] [PubMed]
- Jomova, K.; Hudecova, L.; Lauro, P.; Barbierikova, Z.; Malcek, M.; Alwasel, S.; Alhazza, I.; Rhodes, C.; Valko, M. The effect of luteolin on DNA damage mediated by a copper catalyzed Fenton reaction. J. Inorg. Biochem. 2022, 226, 111635. [Google Scholar] [CrossRef] [PubMed]
- Simunkova, M.; Barbierikova, Z.; Jomova, K.; Lauro, P.; Alwasel, S.; Alhazza, I.; Rhodes, C.; Valko, M. Antioxidant vs. prooxidant properties of the flavonoid, kaempferol, in the presence of Cu (II) ions: A ROS-scavenging activity, fenton reaction and DNA damage study. Int. J. Mol. Sci. 2021, 22, 1619. [Google Scholar] [CrossRef] [PubMed]
- Masek, A.; Chrzescijanska, E.; Latos, M.; Zaborski, M.; Podsedek, A. Antioxidant and antiradical properties of green tea extract compounds. Int. J. Electrochem. Sci. 2017, 12, 6600–6610. [Google Scholar] [CrossRef]
- Shahidi, F.; Zhong, Y. Measurement of antioxidant activity. J. Funct. Foods. 2015, 18, 757–781. [Google Scholar] [CrossRef]
- Berker, K.I.; Demirata, B.; Apak, R. Determination of total antioxidant capacity of lipophilic and hydrophilic antioxidants in the same solution by using ferric–ferricyanide assay. Food Anal. Methods 2012, 5, 1150–1158. [Google Scholar] [CrossRef]
- Pan, Y.; Wang, K.; Huang, S.; Wang, H.; Mu, X.; He, C.; Ji, X.; Zhang, J.; Huang, F. Antioxidant activity of microwave-assisted extract of longan (Dimocarpus Longan Lour.) peel. Food Chem. 2008, 106, 1264–1270. [Google Scholar] [CrossRef]
- Apak, R. Electron Transfer-Based Antioxidant Capacity Assays and the Cupric Ion Reducing Antioxidant Capacity (CUPRAC) Assay; Wiley-Blackwell Chichester: Chichester, UK, 2017; pp. 57–75. [Google Scholar]
- de Quiros, A.; Lage-Yusty, M.A.; Lopez-Hernandez, J. Determination of phenolic compounds in macroalgae for human consumption. Food Chem. 2010, 121, 634–638. [Google Scholar] [CrossRef]
- Lozano-Navarro, J.I.; Diaz-Zavala, N.P.; Velasco-Santos, C.; Melo-Banda, J.; Páramo-García, U.; Paraguay-Delgado, F.; García-Alamilla, R.; Martínez-Hernández, A.; Zapién-Castillo, S. Chitosan-starch films with natural extracts: Physical, chemical, morphological and thermal properties. Materials 2018, 11, 120. [Google Scholar] [CrossRef] [PubMed]
- Chaaban, H.; Ioannou, I.; Chebil, L.; Slimane, M.; Gérardin, C.; Paris, C.; Charbonnel, C.; Chekir, L.; Ghoul, M. Effect of heat processing on thermal stability and antioxidant activity of six flavonoids. J. Food Process. Preserv. 2017, 41, e13203. [Google Scholar] [CrossRef]
- Abirami, S.; Priyalakshmi, M.; Soundariya, A.; Samrot, A.V.; Saigeetha, S.; Emilin, R.R.; Dhiva, S.; Inbathamizh, L. Antimicrobial activity, antiproliferative activity, amylase inhibitory activity and phytochemical analysis of ethanol extract of corn (Zea mays L.) silk. Curr. Res. Green Sustain. Chem. 2021, 4, 100089. [Google Scholar] [CrossRef]
- da Hora, N.; Santana, L.F.; da Silva, V.; Costa, S.; Zambotti-Villela, L.; Colepicolo, P.; Ferraz, C.; Ribeiro, P. Identification of bioactive metabolites from corn silk extracts by a combination of metabolite profiling, univariate statistical analysis and chemometrics. Food Chem. 2021, 365, 130479. [Google Scholar] [CrossRef]
- Šukalović, V.H.; Veljović-Jovanović, S.; Maksimović, J.D.; Maksimović, V.; Pajić, Z. Characterisation of phenol oxidase and peroxidase from maize silk. Plant Biol. 2010, 12, 406–413. [Google Scholar] [CrossRef]
- Farah, A.; De Paulis, T.; Trugo, L.C.; Martin, P.R. Effect of roasting on the formation of chlorogenic acid lactones in coffee. J. Agric. Food Chem. 2005, 53, 1505–1513. [Google Scholar] [CrossRef]

| Corn Silk Sample | Ferric Ion-Reducing Power a | Copper ion Reductive Capability a |
|---|---|---|
| CS-S | 1.33 ± 0.32 a | 0.89 ± 0.09 a |
| CS-M | 0.53 ± 0.11 b | 0.78 ± 0.09 a |
| CS-MS | 2.63 ± 0.15 c | 1.21 ± 0.13 b |
| Polyphenol | Temperature | Concentration (μg/g) a | ||
|---|---|---|---|---|
| °C | CS-S | CS-M | CS-MS | |
| Caffeic acid | 60 | 0.74 ± 0.05 a | 1.01 ± 0.07 ab | 1.51 ± 0.10 c |
| 70 | 2.95 ± 0.15 d | 4.53 ± 0.18 e | 6.22 ± 0.17 f | |
| 80 | 1.28 ± 0.08 bc | 2.26 ± 0.11 h | 2.91 ± 0.14 d | |
| Chlorogenic acid | 60 | 6.24 ± 0.23 a | 15.21 ± 0.37 b | 11.12 ± 0.28 c |
| 70 | 22.67 ± 0.30 d | 33.55 ± 0.34 e | 47.06 ± 0.40 f | |
| 80 | 97.58 ± 0.52 g | 139.67 ± 0.81 h | 183.29 ± 1.09 i | |
| Elagic acid | 60 | 17.34 ± 0.28 a | 34.71 ± 0.33 b | 58.19 ± 0.41 c |
| 70 | 17.68 ± 0.25 a | 39.02 ± 0.34 d | 78.57 ± 0.48 e | |
| 80 | 28.82 ± 0.31 f | 74.16 ±0.44 g | 122.69 ± 0.70 h | |
| Epicatechin | 60 | 19.47 ± 0.19 a | 23.81 ± 0.29 b | 31.23 ± 0.30 c |
| 70 | 17.00 ± 0.18 d | 20.71 ± 0.25 e | 26.37 ± 0.24 f | |
| 80 | 8.55 ± 0.14 g | 10.99 ± 0.21 h | 14.04 ± 0.23 i | |
| Epigallocatechin | 60 | 70.70 ± 0.38 a | 97.73 ± 0.42 b | 137.60 ± 0.97 c |
| 70 | 83.39 ± 0.40 d | 116.04 ± 0.63 e | 162.05 ± 0.65 f | |
| 80 | 182.12 ± 0.69 g | 264.00 ± 1.28 h | 343.64 ± 1.55 i | |
| Ferullic acid | 60 | 0.69 ± 0.07 a | 1.27 ± 0.10 b | 2.38 ± 0.12 cd |
| 70 | 0.62 ± 0.09 a | 1.65 ± 0.11 be | 3.48 ± 0.16 f | |
| 80 | 0.98 ± 0.08 ab | 1.99 ± 0.10 ce | 2.75 ± 0.14 d | |
| p-Hydroxybenzoic acid | 60 | − | − | − |
| 70 | 0.30 ± 0.04 a | 0.38 ± 0.02 a | 0.57 ± 0.05 a | |
| 80 | 0.39 ± 0.03 a | 0.51 ± 0.05 a | 0.61 ± 0.04 a | |
| Kaempferol | 60 | − | − | 56.05 ± 0.37 a |
| 70 | 34.36 ± 0.33 b | 54.60 ± 0.42 c | 76.79 ± 0.50 d | |
| 80 | 76.16 ± 0.49 d | 123.11 ± 0.77 e | 165.60 ± 0.85 f | |
| Protocatechuic ethylester | 60 | 4.71 ± 0.17 a | 14.43 ± 0.16 b | 23.60 ± 0.22 c |
| 70 | 14.41 ± 0.11 b | 23.31 ± 0.18 c | 33.93 ± 0.28 d | |
| 80 | 4.11 ± 0.09 e | 6.99 ± 0.12 f | 9.62 ± 0.10 g | |
| Protocatechuic acid | 60 | 0.61 ± 0.08 a | 0.82 ± 0.10 a | 1.32 ± 0.12 b |
| 70 | 0.33 ± 0.07 c | 0.77 ± 0.11 a | 1.22 ± 0.14 b | |
| 80 | 0.29 ± 0.09 c | 0.64 ± 0.10 a | 0.90 ± 0.12 a | |
| Rutin | 60 | − | 7.49 ± 0.13 a | 10.79 ± 0.16 b |
| 70 | 3.32 ± 0.17 c | 4.79 ± 0.11 d | 6.48 ± 0.18 e | |
| 80 | − | − | − | |
| Sinapic acid | 60 | 0.10 ± 0.02 a | − | − |
| 70 | − | − | − | |
| 80 | 1.91 ± 0.08 b | 2.77 ± 0.14 c | 4.15 ± 0.13 d | |
| trans-2-Hydroxycinnamic acid | 60 | 3.59 ± 0.11 a | 4.67 ± 0.15 b | 7.13 ± 0.18 c |
| 70 | 2.88 ± 0.09 d | 4.46 ± 0.10 e | 6.90 ± 0.15 c | |
| 80 | 7.63 ± 0.16 f | 11.89 ± 0.18 g | 16.88 ± 0.13 h | |
| trans-Cinnamic acid | 60 | − | − | 8.48 ± 0.13 a |
| 70 | 5.20 ± 0.15 b | 6.20 ± 0.19 c | 11.62 ± 0.22 d | |
| 80 | 11.53 ± 0.24 d | 18.64 ± 0.27 e | 25.07 ± 0.30 f | |
| trans-p-Coumaric acid | 60 | 2.21 ± 0.14 a | 3.98 ± 0.06 b | 6.16 ± 0.10 c |
| 70 | 5.76 ± 0.17 d | 9.36 ± 0.15 e | 15.21 ± 0.22 f | |
| 80 | 4.87 ± 0.16 g | 7.68 ± 0.11 h | 10.36 ± 0.15 i | |
| Vanillic acid | 60 | 3.02 ± 0.09 a | 7.37 ± 0.14 b | 5.39 ± 0.12 c |
| 70 | 10.99 ± 0.15 d | 16.26 ± 0.20 e | 22.81 ± 0.16 f | |
| 80 | 47.31 ± 0.23 g | 67.71 ± 0.38 h | 88.86 ± 0.44 i | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lapčík, L.; Řepka, D.; Lapčíková, B.; Sumczynski, D.; Gautam, S.; Li, P.; Valenta, T. A Physicochemical Study of the Antioxidant Activity of Corn Silk Extracts. Foods 2023, 12, 2159. https://doi.org/10.3390/foods12112159
Lapčík L, Řepka D, Lapčíková B, Sumczynski D, Gautam S, Li P, Valenta T. A Physicochemical Study of the Antioxidant Activity of Corn Silk Extracts. Foods. 2023; 12(11):2159. https://doi.org/10.3390/foods12112159
Chicago/Turabian StyleLapčík, Lubomír, David Řepka, Barbora Lapčíková, Daniela Sumczynski, Shweta Gautam, Peng Li, and Tomáš Valenta. 2023. "A Physicochemical Study of the Antioxidant Activity of Corn Silk Extracts" Foods 12, no. 11: 2159. https://doi.org/10.3390/foods12112159
APA StyleLapčík, L., Řepka, D., Lapčíková, B., Sumczynski, D., Gautam, S., Li, P., & Valenta, T. (2023). A Physicochemical Study of the Antioxidant Activity of Corn Silk Extracts. Foods, 12(11), 2159. https://doi.org/10.3390/foods12112159







