Effect of the Degree of Hydrolysis on Nutritional, Functional, and Morphological Characteristics of Protein Hydrolysate Produced from Bighead Carp (Hypophthalmichthys nobilis) Using Ficin Enzyme
Abstract
:1. Introduction
2. Materials and Methods
2.1. Raw Fish Muscle Preparation
2.2. Enzyme and Reagents
2.3. Protein Hydrolysate Preparation
2.4. Determination of Hydrolysis Degree
2.5. Chemical Composition Analysis
2.6. Yield
2.7. Color Measurement
2.8. Amino Acid Determination
2.9. Molecular-Weight Distribution Profile
2.10. Scanning Electron Microscopy
2.11. Thermal Characterization Analysis
2.12. Functional Properties Analysis of FPH
2.12.1. Solubility
2.12.2. Emulsifying Properties
2.12.3. Water Holding Capacity
2.12.4. Oil Holding Capacity
2.13. Statistical Analysis
3. Results and Discussion
3.1. Production of Protein Hydrolysates
3.2. Proximate Chemical Composition
3.3. Yield
3.4. Color Measurements
3.5. Amino Acid Analysis
3.6. Molecular-Weight Distribution
3.7. Scanning Electron Microscopy Analysis
3.8. Differential Scanning Calorimeter Analysis
3.9. Functional Properties of FPH
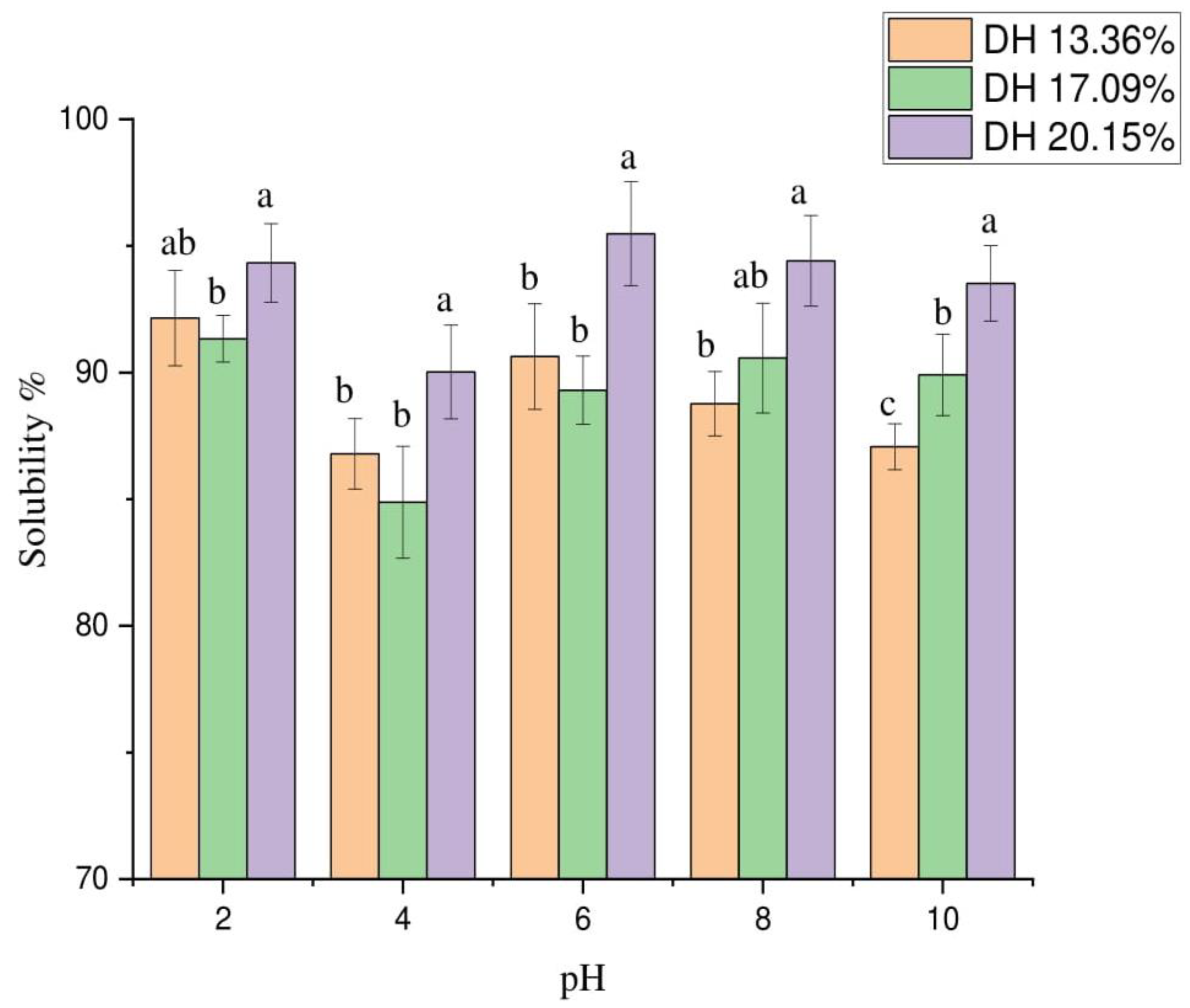
3.9.1. Protein Hydrolysate Solubility
3.9.2. Water and Oil Holding Capacity
3.9.3. Emulsifying Properties
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO. Fishery and Aquaculture Statistics Yearbook; Food & Agriculture Organization of the United Nations; FAO: Rome, Italy, 2020. [Google Scholar]
- Shi, X.; Zhang, J.; Shi, C.; Tan, Y.; Hong, H.; Luo, Y. Nondestructive prediction of freshness for bighead carp (Hypophthalmichthys nobilis) head by Excitation-Emission Matrix (EEM) analysis based on fish eye fluid: Comparison of BPNNs and RBFNNs. Food Chem. 2022, 382, 132341. [Google Scholar] [CrossRef] [PubMed]
- Cupp, A.R.; Erickson, R.A.; Fredricks, K.T.; Swyers, N.M.; Hatton, T.W.; Amberg, J.J. Responses of invasive silver and bighead carp to a carbon dioxide barrier in outdoor ponds. Can. J. Fish. Aquat. Sci. 2017, 74, 297–305. [Google Scholar] [CrossRef]
- Ashraf, S.A.; Adnan, M.; Patel, M.; Siddiqui, A.J.; Sachidanandan, M.; Snoussi, M.; Hadi, S. Fish-Based Bioactives as Potent Nutraceuticals: Exploring the Therapeutic Perspective of Sustainable Food from the Sea. Mar. Drugs 2020, 18, 265. [Google Scholar] [CrossRef] [PubMed]
- Chukwu, O. Influences of drying methods on nutritional properties of tilapia fish (Oreochromis nilotieus). World J. Agric. Sci. 2009, 5, 256–258. [Google Scholar]
- Chalamaiah, M.; Hemalatha, R.; Jyothirmayi, T. Fish protein hydrolysates: Proximate composition, amino acid composition, antioxidant activities and applications: A review. Food Chem. 2012, 135, 3020–3038. [Google Scholar] [CrossRef]
- Noman, A.; Xu, Y.; AL-Bukhaiti, W.Q.; Abed, S.M.; Ali, A.H.; Ramadhan, A.H.; Xia, W. Influence of enzymatic hydrolysis conditions on the degree of hydrolysis and functional properties of protein hydrolysate obtained from Chinese sturgeon (Acipenser sinensis) by using papain enzyme. Process Biochem. 2018, 67, 19–28. [Google Scholar] [CrossRef]
- Petreu, T.; Cotrutz, C.E.; Neamtu, M.; Maria Filioreanu, A.; Badescu, L.; Neamtu, A. Bioinformatic Lookup for Metalloproteinases and Their Natural Inhibitors. Structural Relationships. Ann. Rom. Soc. Cell Biol. 2011, 16, 175–180. [Google Scholar]
- Ismail, B.; Mohammed, H.; Nair, A.J. Influence of proteases on functional properties of food. In Green Bio-Processes; Springer: Singapore, 2019; pp. 31–53. [Google Scholar]
- Katsaros, G.; Katapodis, P.; Taoukis, P. High hydrostatic pressure inactivation kinetics of the plant proteases ficin and papain. J. Food Eng. 2009, 91, 42–48. [Google Scholar] [CrossRef]
- Arshad, M.S.; Kwon, J.-H.; Imran, M.; Sohaib, M.; Aslam, A.; Nawaz, I.; Amjad, Z.; Khan, U.; Javed, M. Plant and bacterial proteases: A key towards improving meat tenderization, a mini review. Cogent Food Agric. 2016, 2, 1261780. [Google Scholar] [CrossRef]
- Wasswa, J.; Tang, J.; Gu, X.-h.; Yuan, X.-q. Influence of the extent of enzymatic hydrolysis on the functional properties of protein hydrolysate from grass carp (Ctenopharyngodon idella) skin. Food Chem. 2007, 104, 1698–1704. [Google Scholar] [CrossRef]
- He, S.; Franco, C.; Zhang, W. Functions, applications and production of protein hydrolysates from fish processing co-products (FPCP). Food Res. Int. 2013, 50, 289–297. [Google Scholar] [CrossRef]
- Shahidi, S.; Jamili, S.; Mostafavi, P.G.; Rezaie, S.; Khorramizadeh, M. Assessment of the inhibitory effects of ficin-hydrolyzed gelatin derived from squid (Uroteuthis duvauceli) on breast cancer cell lines and animal model. Iran. J. Allergy Asthma Immunol. 2018, 17, 436–452. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schaafsma, G. The protein digestibility-corrected amino acid score (PDCAAS)—A concept for describing protein quality in foods and food ingredients: A critical review. J. AOAC Int. 2005, 88, 988–994. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- AOAC. Official Methods of Analysis; AOAC: Rockville, MD, USA, 1999. [Google Scholar]
- Romadhoni, A.R.; Afrianto, E.; Pratama, R.I.; Grandiosa, R. Extraction of snakehead fish [Ophiocephalus striatus (Bloch, 1793)] into fish protein concentrate as albumin source using various solvent. Aquat. Procedia 2016, 7, 4–11. [Google Scholar] [CrossRef]
- AOAC. Official methods of analysis. In Association of Official Analytical Chemists, 16th ed.; AOAC: Washington, DC, USA, 2005. [Google Scholar]
- Liu, W.; Gu, R.; Lin, F.; Lu, J.; Yi, W.; Ma, Y.; Dong, Z.; Cai, M. Isolation and identification of antioxidative peptides from pilot-scale black-bone silky fowl (Gallus gallus domesticus Brisson) muscle oligopeptides. J. Sci. Food Agric. 2013, 93, 2782–2788. [Google Scholar] [CrossRef]
- Alahmad, K.; Xia, W.; Jiang, Q.; Xu, Y. Influence of Drying Techniques on the Physicochemical, Nutritional, and Morphological Properties of Bighead Carp (Hypophthalmichthys nobilis) Fillets. Foods 2021, 10, 2837. [Google Scholar] [CrossRef]
- Jain, S.; Anal, A.K. Optimization of extraction of functional protein hydrolysates from chicken egg shell membrane (ESM) by ultrasonic assisted extraction (UAE) and enzymatic hydrolysis. LWT-Food Sci. Technol. 2016, 69, 295–302. [Google Scholar] [CrossRef]
- Noman, A.; Qixing, J.; Xu, Y.; Ali, A.H.; Al-Bukhaiti, W.Q.; Abed, S.M.; Xia, W. Influence of degree of hydrolysis on chemical composition, functional properties, and antioxidant activities of chinese sturgeon (Acipenser sinensis) hydrolysates obtained by using alcalase 2.4 L. J. Aquat. Food Prod. Technol. 2019, 28, 583–597. [Google Scholar] [CrossRef]
- Islam, M.S.; Hongxin, W.; Admassu, H.; Noman, A.; Ma, C.; An Wei, F. Degree of hydrolysis, functional and antioxidant properties of protein hydrolysates from Grass Turtle (Chinemys reevesii) as influenced by enzymatic hydrolysis conditions. Food Sci. Nutr. 2021, 9, 4031–4047. [Google Scholar] [CrossRef]
- Chalamaiah, M.; Rao, G.N.; Rao, D.G.; Jyothirmayi, T. Protein hydrolysates from meriga (Cirrhinus mrigala) egg and evaluation of their functional properties. Food Chem. 2010, 120, 652–657. [Google Scholar] [CrossRef]
- Ktari, N.; Jridi, M.; Bkhairia, I.; Sayari, N.; Salah, R.B.; Nasri, M. Functionalities and antioxidant properties of protein hydrolysates from muscle of zebra blenny (Salaria basilisca) obtained with different crude protease extracts. Food Res. Int. 2012, 49, 747–756. [Google Scholar] [CrossRef]
- Ovissipour, M.; Rasco, B.; Shiroodi, S.G.; Modanlow, M.; Gholami, S.; Nemati, M. Antioxidant activity of protein hydrolysates from whole anchovy sprat (Clupeonella engrauliformis) prepared using endogenous enzymes and commercial proteases. J. Sci. Food Agric. 2013, 93, 1718–1726. [Google Scholar] [CrossRef] [PubMed]
- Rivero-Pino, F.; Espejo-Carpio, F.J.; Guadix, E.M. Evaluation of the bioactive potential of foods fortified with fish protein hydrolysates. Food Res. Int. 2020, 137, 109572. [Google Scholar] [CrossRef] [PubMed]
- Dong, S.; Zeng, M.; Wang, D.; Liu, Z.; Zhao, Y.; Yang, H. Antioxidant and biochemical properties of protein hydrolysates prepared from Silver carp (Hypophthalmichthys molitrix). Food Chem. 2008, 107, 1485–1493. [Google Scholar] [CrossRef]
- Slizyte, R.; Rommi, K.; Mozuraityte, R.; Eck, P.; Five, K.; Rustad, T. Bioactivities of fish protein hydrolysates from defatted salmon backbones. Biotechnol. Rep. 2016, 11, 99–109. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Dai, Z.; Zhang, Y.; Dong, Y.; Hu, X. Structural characteristics and stability of salmon skin protein hydrolysates obtained with different proteases. LWT 2022, 153, 112460. [Google Scholar] [CrossRef]
- Elavarasan, K.; Shamasundar, B.A. Effect of oven drying and freeze drying on the antioxidant and functional properties of protein hydrolysates derived from freshwater fish (Cirrhinus mrigala) using papain enzyme. J. Food Sci. Technol. 2016, 53, 1303–1311. [Google Scholar] [CrossRef] [Green Version]
- Molina Ortiz, S.; Añón, M. Enzymatic hydrolysis of soy protein isolates. DSC study. J. Therm. Anal. Calorim. 2001, 66, 489–499. [Google Scholar] [CrossRef]
- Liu, Y.; Li, X.; Chen, Z.; Yu, J.; Wang, F.; Wang, J. Characterization of structural and functional properties of fish protein hydrolysates from surimi processing by-products. Food Chem. 2014, 151, 459–465. [Google Scholar] [CrossRef]
- Thiansilakul, Y.; Benjakul, S.; Shahidi, F. Compositions, functional properties and antioxidative activity of protein hydrolysates prepared from round scad (Decapterus maruadsi). Food Chem. 2007, 103, 1385–1394. [Google Scholar] [CrossRef]
- Gbogouri, G.; Linder, M.; Fanni, J.; Parmentier, M. Influence of hydrolysis degree on the functional properties of salmon byproducts hydrolysates. J. Food Sci. 2004, 69, C615–C622. [Google Scholar] [CrossRef]
- Foh, M.B.K.; Amadou, I.; Foh, B.M.; Kamara, M.T.; Xia, W. Functionality and antioxidant properties of tilapia (Oreochromis niloticus) as influenced by the degree of hydrolysis. Int. J. Mol. Sci. 2010, 11, 1851–1869. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.; Luo, Y.; Shen, H.; You, J. Antioxidant activities and functional properties of grass carp (Ctenopharyngodon idellus) protein hydrolysates. J. Sci. Food Agric. 2012, 92, 292–298. [Google Scholar] [CrossRef] [PubMed]
- Nalinanon, S.; Benjakul, S.; Kishimura, H.; Shahidi, F. Functionalities and antioxidant properties of protein hydrolysates from the muscle of ornate threadfin bream treated with pepsin from skipjack tuna. Food Chem. 2011, 124, 1354–1362. [Google Scholar] [CrossRef]
- Kristinsson, H.G.; Rasco, B.A. Fish protein hydrolysates: Production, biochemical, and functional properties. Crit. Rev. Food Sci. Nutr. 2000, 40, 43–81. [Google Scholar] [CrossRef] [PubMed]
- Mutilangi, W.; Panyam, D.; Kilara, A. Functional properties of hydrolysates from proteolysis of heat-denatured whey protein isolate. J. Food Sci. 1996, 61, 270–275. [Google Scholar] [CrossRef]
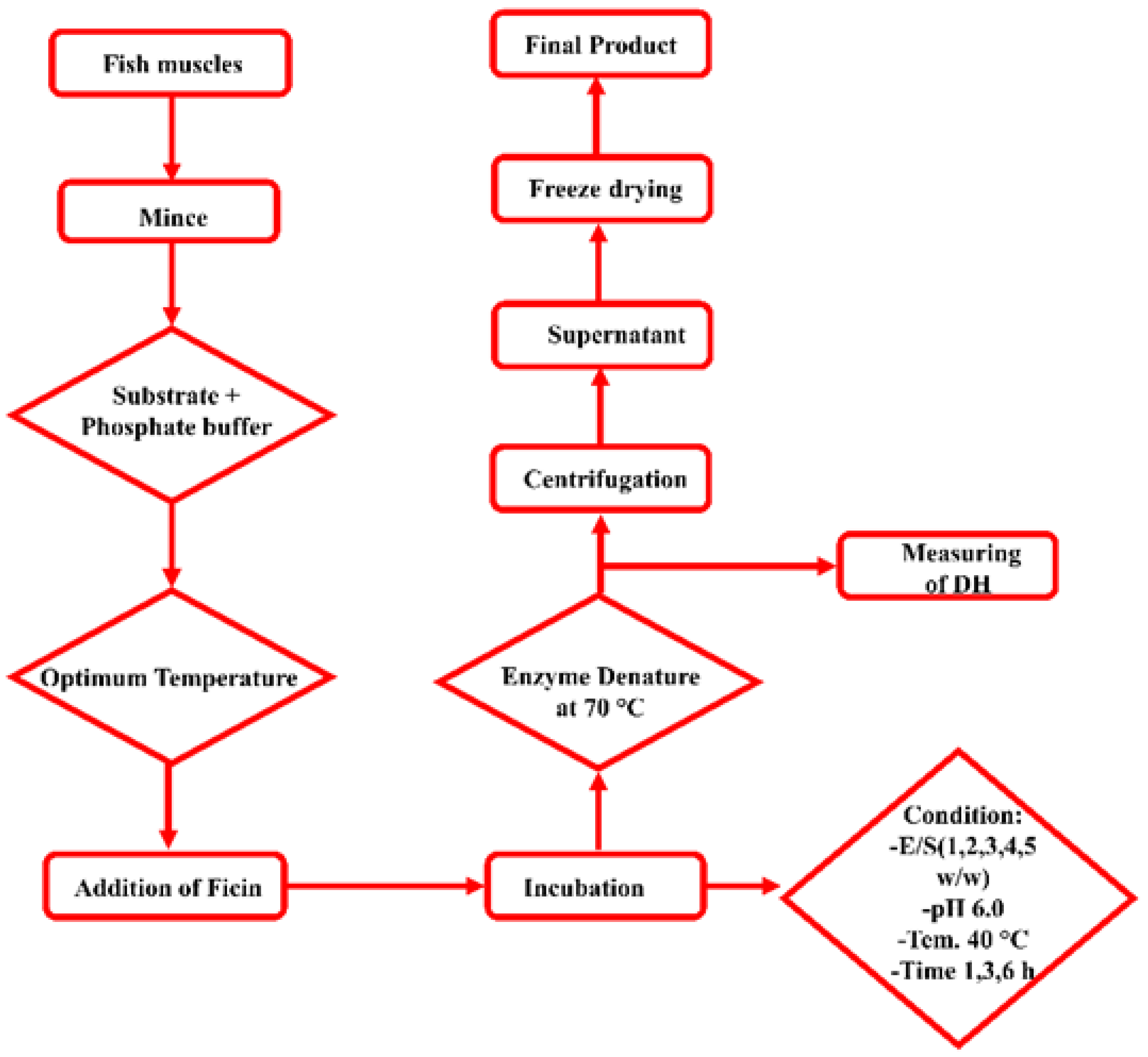
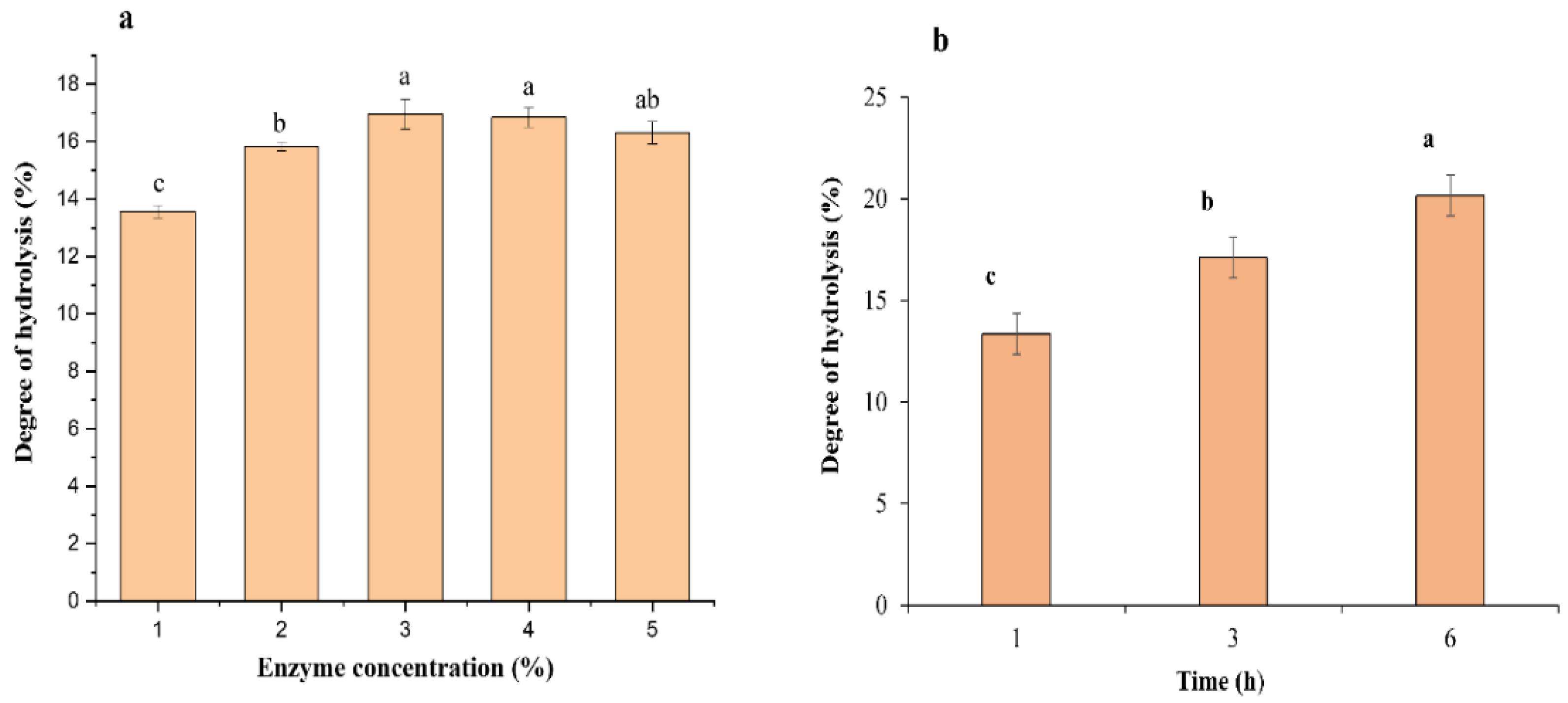
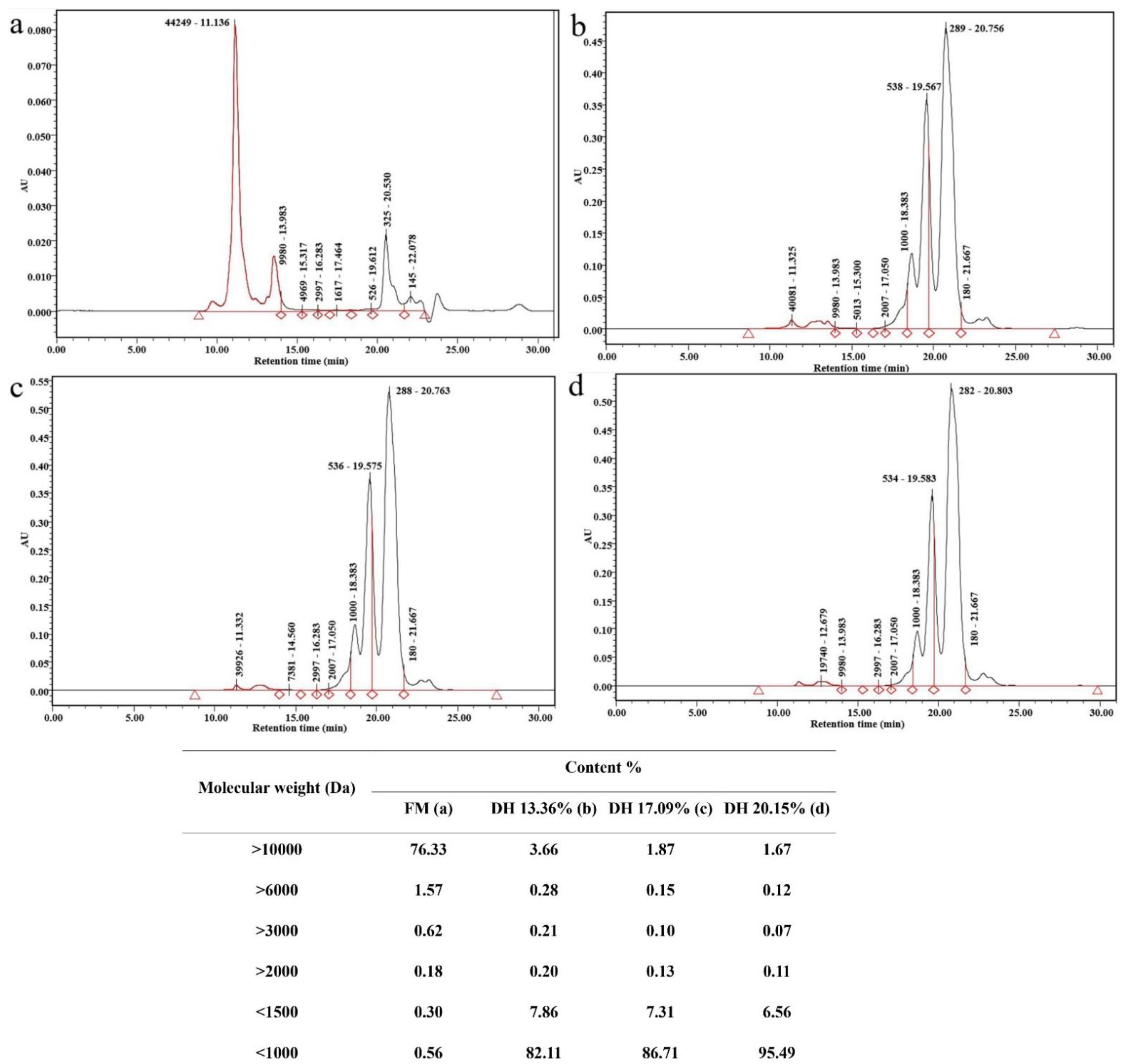

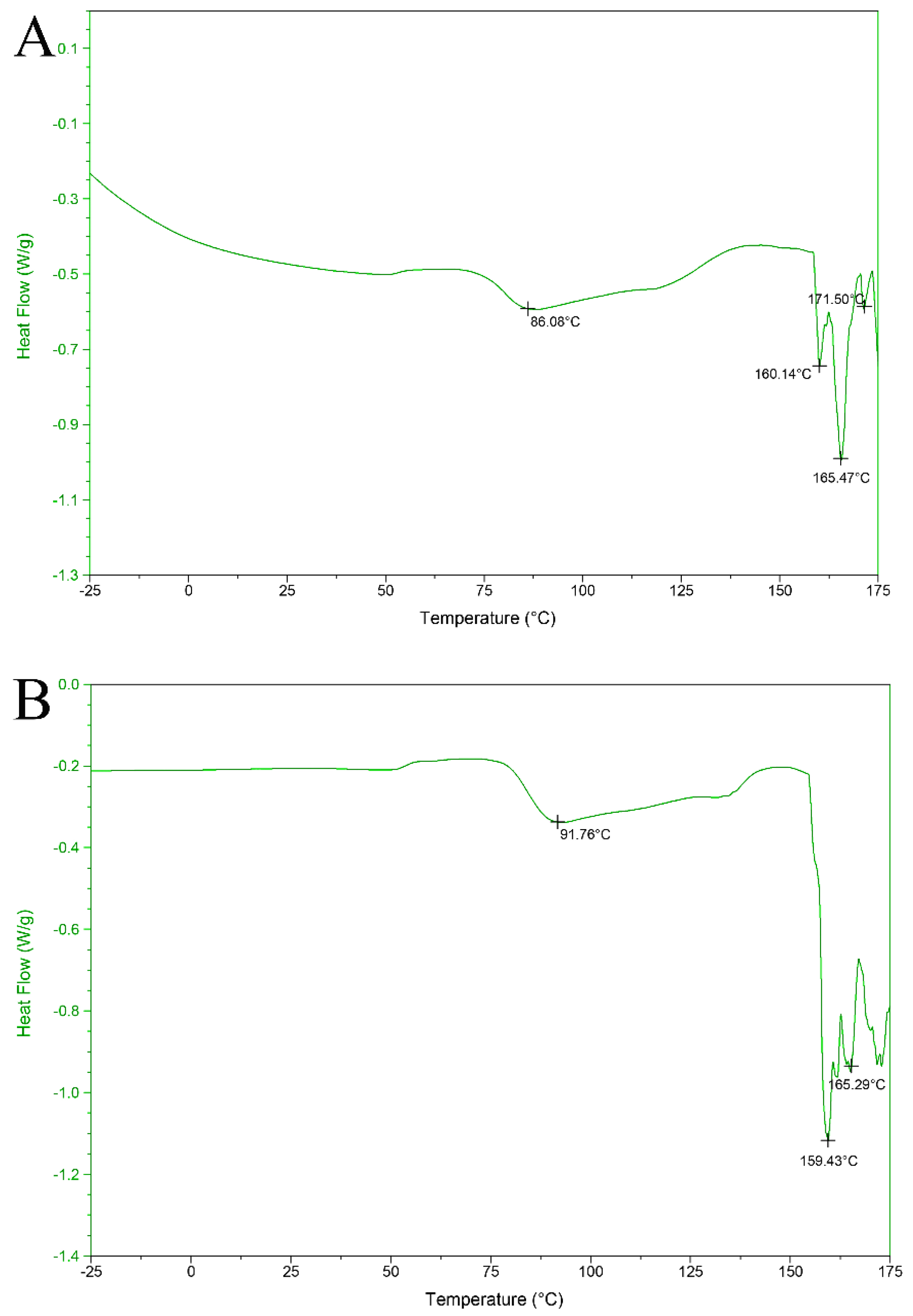

| Hydrolysis Conditions | Ficin Enzyme |
|---|---|
| Enzyme/substrate ratio (%) | 1, 2, 3, 4, 5 |
| Reaction pH | 6 |
| Temperature (°C) | 40 |
| Inactivation temperature of hydrolysis (°C) | 70 |
| Inactivation time (min) | 20 |
| Hydrolysis time (h) | 1, 3, 6 |
| Parameters | Hydrolysates | |||
|---|---|---|---|---|
| Fresh Sample | DH 13.36% (1 h) | DH 17.09% (3 h) | DH 20.15% (6 h) | |
| Moisture | 77.31 ± 0.93 | 4.26 ± 0.09 a | 4.21 ± 0.06 a | 4.28 ± 0.06 a |
| Protein | 15.78 ± 0.25 | 80.58 ± 0.65 c | 81.93 ± 1.15 b | 85.27 ± 0.73 a |
| Fat | 1.68 ± 0.07 | 4.13 ± 0.43 a | 4.59 ± 0.28 a | 2.84 ± 0.12 b |
| Ash | 1.34 ± 0.03 | 6.69 ± 0.18 a | 6.04 ± 0.11 b | 5.97 ± 0.16 b |
| Color determination | ||||
| L* | - | 89.23 ± 0.08 a | 88.82 ± 0.10 b | 86.55 ± 0.09 c |
| a* | - | 0.17 ± 0.01 b | 0.23 ± 0.05 ab | 0.31 ± 0.06 a |
| b* | - | 10.96 ± 0.56 b | 12.29 ± 0.10 a | 12.68 ± 0.03 a |
| ∆E | - | 19.85 ± 0.47 a | 18.71 ± 0.25 b | 18.92 ± 0.16 b |
| Hydrolysate yield | - | 11.25 ± 0.16 c | 14.67 ± 0.20 b | 15.92 ± 0.31 a |
| WHC (g/g FPH) | - | 2.18 ± 0.15 c | 2.79 ± 0.21 b | 3.93 ± 0.19 a |
| OHC (g/g FPH) | - | 3.85 ± 0.10 a | 3.72 ± 0.13 a | 2.94 ± 0.17 b |
| Amino Acids | Hydrolysates | ||
|---|---|---|---|
| Essential Amino Acids (EAAs) | DH 13.36% (1 h) | DH 17.09% (3 h) | DH 20.15% (6 h) |
| Histidine | 2.63 ± 0.02 b | 2.65 ± 0.02 b | 2.78 ± 0.05 a |
| Threonine | 3.13 ± 0.06 c | 3.24 ± 0.05 b | 3.36 ± 0.11 a |
| Valine | 4.23 ± 0.06 b | 4.32 ± 0.02 b | 4.43 ± 0.04 a |
| Methionine | 2.39 ± 0.16 b | 2.65 ± 0.02 a | 2.77 ± 0.10 a |
| Phenylalanine | 3.05 ± 0.02 c | 3.22 ± 0.01 b | 3.29 ± 0.02 a |
| Isoleucine | 3.80 ± 0.05 c | 3.86 ± 0.01 b | 4.01 ± 0.03 a |
| Leucine | 6.42 ± 0.05 c | 6.51 ± 0.02 b | 6.74 ± 0.02 a |
| Lysine | 8.12 ± 0.08 b | 8.01 ± 0.06 b | 8.49 ± 0.06 a |
| Arginine | 4.99 ± 0.01 b | 4.97 ± 0.01 b | 5.17 ± 0.07 a |
| Tyrosine | 2.03 ± 0.22 c | 2.34 ± 0.05 b | 2.62 ± 0.03 a |
| Nonessential amino acids (NAAs) | |||
| Cystenie | 0.23 ± 0.03 b | 0.28 ± 0.01 b | 0.36 ± 0.02 a |
| Aspartic acid | 9.72 ± 0.07 c | 10.00 ± 0.03 b | 10.45 ± 0.19 a |
| Glutamic acid | 13.79 ± 0.25 c | 14.02 ± 0.05 b | 14.46 ± 0.10 a |
| Serine | 2.26 ± 0.04 c | 2.59 ± 0.05 b | 2.70 ± 0.04 a |
| Glycine | 4.33 ± 0.07 a | 4.21 ± 0.04 b | 4.09 ± 0.01 c |
| Proline | 2.48 ± 0.02 a | 2.54 ± 0.04 a | 2.28 ± 0.02 b |
| Alanine | 4.70 ± 0.12 b | 4.91 ± 0.08 a | 5.01 ± 0.07 a |
| TEAA | 40.81 ± 0.45 c | 41.79 ± 0.29 b | 43.69 ± 0.36 a |
| TNAA | 37.52 ± 0.17 c | 38.57 ± 0.11 b | 39.38 ± 0.82 a |
| TAA | 78.33 ± 1.12 c | 80.36 ± 0.73 b | 83.07 ± 0.68 a |
| TEAA/TAA% | 51.97 ± 0.28 | 52.04 ± 0.41 | 52.85 ± 0.15 |
| DH % | Concentration % | EAI (m2/g) | ESI (min) |
|---|---|---|---|
| 13.36 | 0.1 | 92.75 ± 1.39 a | 51.39 ± 1.28 a |
| 0.5 | 27.36 ± 0.68 b | 29.94 ± 1.09 b | |
| 1 | 10.68 ± 0.51 c | 23.46 ± 0.75 c | |
| 17.09 | 0.1 | 76.82 ± 2.07 a | 45.87 ± 0.91 a |
| 0.5 | 14.98 ± 0.82 b | 20.16 ± 0.41 b | |
| 1 | 8.22 ± 0.45 c | 18.40 ± 0.34 c | |
| 20.15 | 0.1 | 72.04 ± 1.69 a | 36.03 ± 0.86 a |
| 0.5 | 16.11 ± 0.97 b | 19.78 ± 0.73 b | |
| 1 | 8.17 ± 0.65 c | 11.83 ± 0.24 c |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alahmad, K.; Xia, W.; Jiang, Q.; Xu, Y. Effect of the Degree of Hydrolysis on Nutritional, Functional, and Morphological Characteristics of Protein Hydrolysate Produced from Bighead Carp (Hypophthalmichthys nobilis) Using Ficin Enzyme. Foods 2022, 11, 1320. https://doi.org/10.3390/foods11091320
Alahmad K, Xia W, Jiang Q, Xu Y. Effect of the Degree of Hydrolysis on Nutritional, Functional, and Morphological Characteristics of Protein Hydrolysate Produced from Bighead Carp (Hypophthalmichthys nobilis) Using Ficin Enzyme. Foods. 2022; 11(9):1320. https://doi.org/10.3390/foods11091320
Chicago/Turabian StyleAlahmad, Kamal, Wenshui Xia, Qixing Jiang, and Yanshun Xu. 2022. "Effect of the Degree of Hydrolysis on Nutritional, Functional, and Morphological Characteristics of Protein Hydrolysate Produced from Bighead Carp (Hypophthalmichthys nobilis) Using Ficin Enzyme" Foods 11, no. 9: 1320. https://doi.org/10.3390/foods11091320
APA StyleAlahmad, K., Xia, W., Jiang, Q., & Xu, Y. (2022). Effect of the Degree of Hydrolysis on Nutritional, Functional, and Morphological Characteristics of Protein Hydrolysate Produced from Bighead Carp (Hypophthalmichthys nobilis) Using Ficin Enzyme. Foods, 11(9), 1320. https://doi.org/10.3390/foods11091320





