Influence of Natural Polysaccharides on Intestinal Microbiota in Inflammatory Bowel Diseases: An Overview
Abstract
:1. Introduction
2. Overview of Role of Intestinal Microbiota in IBD
3. NPs Impact on Intestinal Microbiota
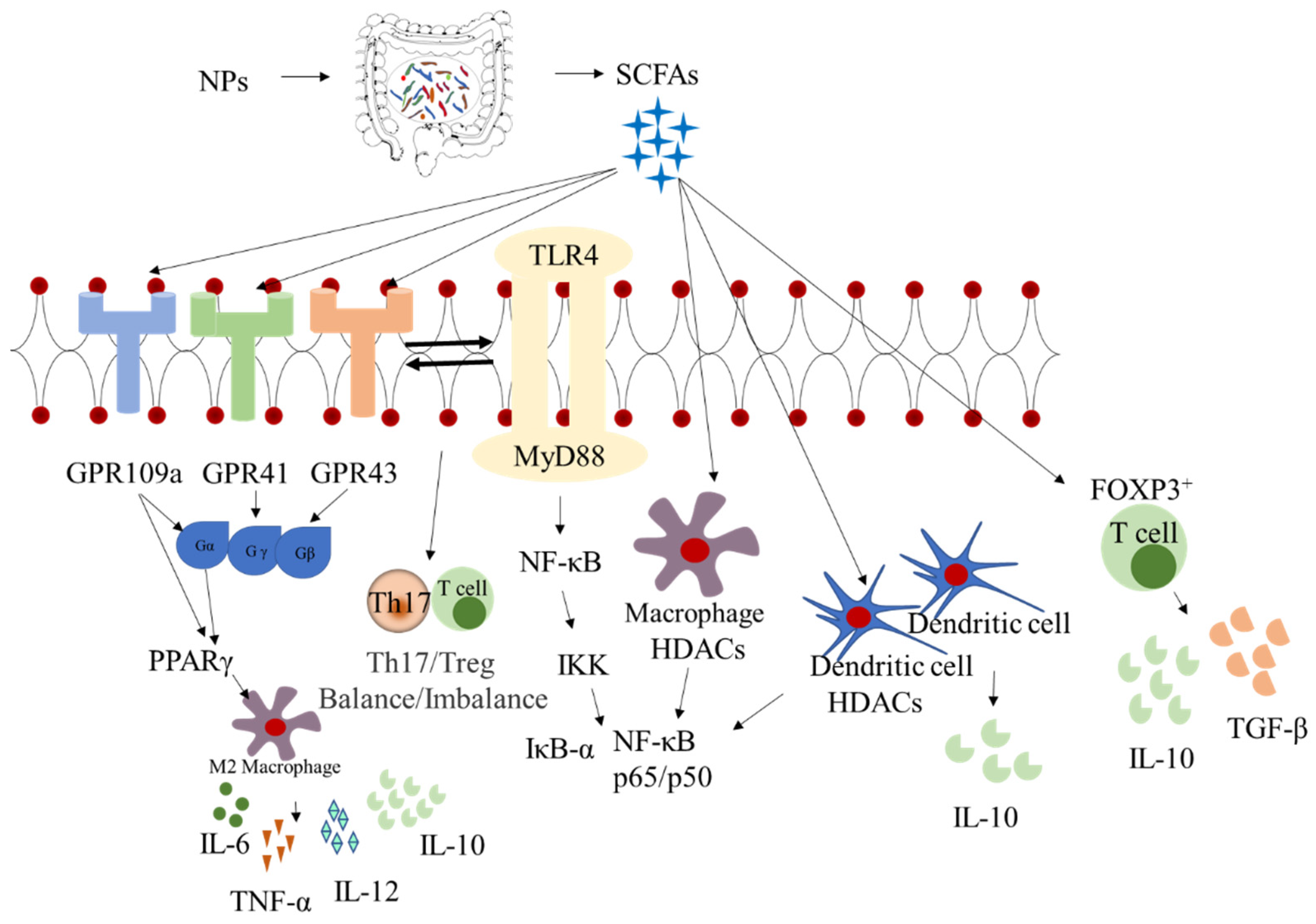
4. Synergistic Effects between Intestinal Microbiota and Metabolites
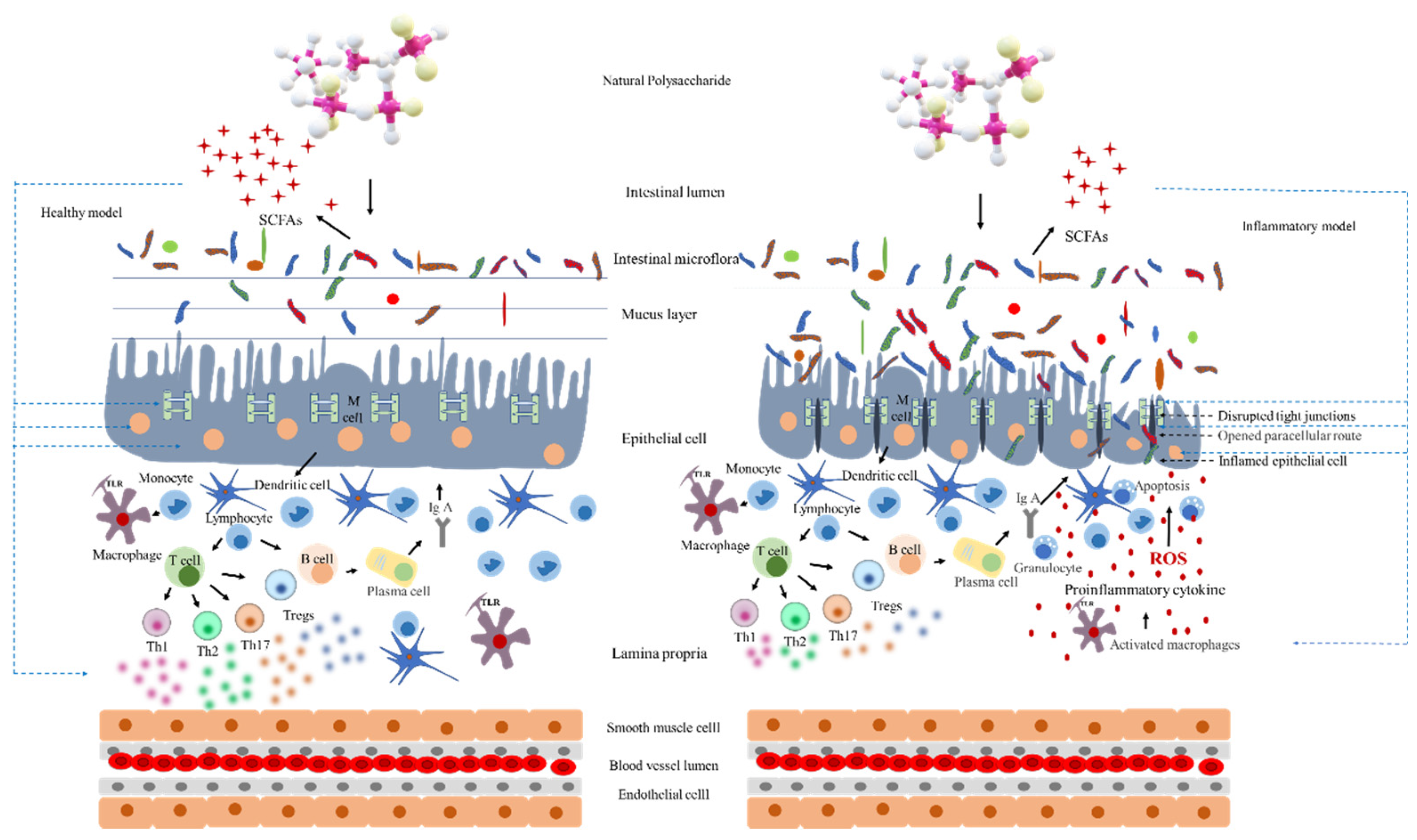
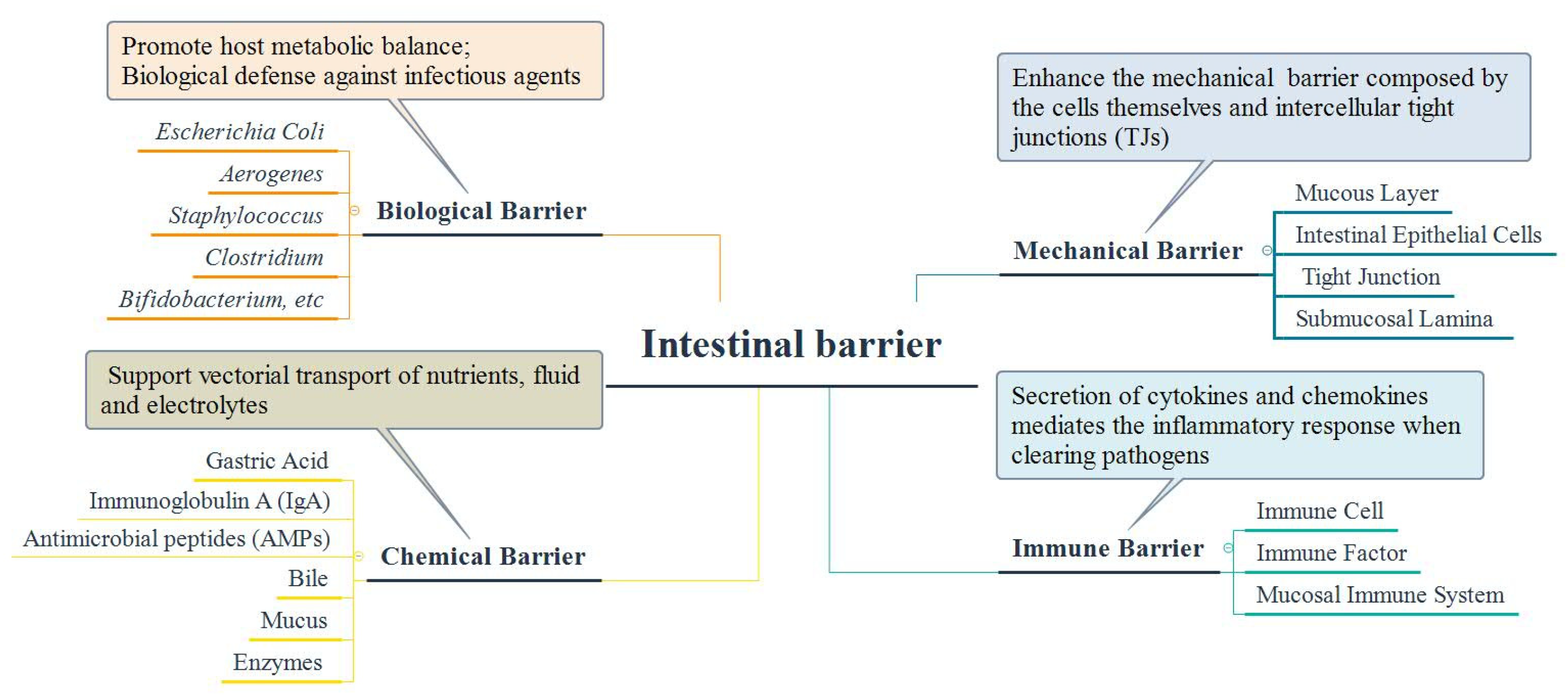
5. Interaction between Intestinal Microbiota and Intestinal Barrier
6. Effect on Intestinal Immunity and Inflammatory Responses
7. Conclusions and Recommendations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, K.; Jin, X.; Li, Q.; Sawaya, A.C.H.F.; Leu, R.K.L.; Conlon, M.A.; Wu, L.; Hu, F. Propolis from different geographic origins decreases intestinal inflammation and Bacteroides spp. populations in a model of DSS-induced colitis. Mol. Nutr. Food Res. 2018, 62, e1800080. [Google Scholar] [CrossRef] [PubMed]
- Piovani, D.; Danese, S.; Peyrin-Biroulet, L.; Bonovas, S. Inflammatory bowel disease: Estimates from the global burden of disease 2017 study. Aliment. Pharm. 2020, 51, 261–270. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Liang, W.; Wang, T.; Sui, J.; Wang, J.; Deng, Z.; Chen, D. Saponins regulate intestinal inflammation in colon cancer and IBD. Pharm. Res. 2019, 144, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Glassner, K.L.; Abraham, B.P.; Quigley, E.M.M. The microbiome and inflammatory bowel disease. J. Allergy Clin. Immunol. 2020, 145, 16–27. [Google Scholar] [CrossRef] [Green Version]
- Zhang, S.L.; Wang, S.N.; Miao, C.Y. Influence of microbiota on intestinal immune system in ulcerative colitis and its intervention. Front. Immunol. 2017, 8, 1674. [Google Scholar] [CrossRef] [Green Version]
- Shen, X.; Wan, Q.; Zhao, R.; Wu, Y.; Wang, Y.; Cui, Y.; Su, X.; Wu, X. Inflammatory bowel diseases and the risk of adverse health outcomes: Umbrella review of meta-analyses of observational studies. Dig. Liver Dis. 2021, 53, 809–816. [Google Scholar] [CrossRef]
- Yadav, H.; Karthikeyan, C. Natural polysaccharides: Structural features and properties. In Polysaccharide Carriers for Drug Delivery; Maiti, S., Jana, S., Eds.; Woodhead Publishing: Cambridge, UK, 2019; pp. 1–17. [Google Scholar]
- Ji, X.; Hou, C.; Guo, X. Physicochemical properties, structures, bioactivities and future prospective for polysaccharides from Plantago L. (Plantaginaceae): A review. Int. J. Biol. Macromol. 2019, 135, 637–646. [Google Scholar] [CrossRef]
- Gao, H.; Wen, J.J.; Hu, J.L.; Nie, Q.X.; Chen, H.H.; Xiong, T.; Nie, S.P.; Xie, M.Y. Polysaccharide from fermented Momordica charantia L. with Lactobacillus plantarum NCU116 ameliorates type 2 diabetes in rats. Carbohydr. Polym. 2018, 201, 624–633. [Google Scholar] [CrossRef]
- Nie, Q.; Hu, J.; Gao, H.; Fan, L.; Chen, H.; Nie, S. Polysaccharide from Plantago asiatica L. attenuates hyperglycemia, hyperlipidemia and affects colon microbiota in type 2 diabetic rats. Food Hydrocoll. 2019, 86, 34–42. [Google Scholar] [CrossRef]
- Mirzadeh, M.; Lelekami, A.K.; Khedmat, L. Plant/algal polysaccharides extracted by microwave: A review on hypoglycemic, hypolipidemic, prebiotic, and immune-stimulatory effect. Carbohydr. Polym. 2021, 266, 118134. [Google Scholar] [CrossRef]
- Zhang, S.; Zhang, H.; Shi, L.; Li, Y.; Tuerhong, M.; Abudukeremu, M.; Cui, J.; Li, Y.; Jin, D.-Q.; Xu, J.; et al. Structure features, selenylation modification, and improved anti-tumor activity of a polysaccharide from Eriobotrya japonica. Carbohydr. Polym. 2021, 273, 118496. [Google Scholar] [CrossRef] [PubMed]
- Xiao, B.; Chen, S.; Huang, Q.; Tan, J.; Zeng, J.; Yao, J.; Feng, T.; Wang, G.; Zhang, Y. The lipid lowering and antioxidative stress potential of polysaccharide from Auricularia auricula prepared by enzymatic method. Int. J. Biol. Macromol. 2021, 187, 651–663. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, J.R.; Junior, R.N.C. Polysaccharides obtained from natural edible sources and their role in modulating the immune system: Biologically active potential that can be exploited against COVID-19. Trends Food Sci. Technol. 2021, 108, 223–235. [Google Scholar] [CrossRef] [PubMed]
- Maria-Ferreira, D.; Dallazen, J.L.; Corso, C.R.; Nascimento, A.M.; Cipriani, T.R.; Watanabe, P.S.; Sant’Ana, D.M.G.; Baggio, C.H.; Werner, M.F.P. Rhamnogalacturonan polysaccharide inhibits inflammation and oxidative stress and alleviates visceral pain. J. Funct. Foods 2021, 82, 104483. [Google Scholar] [CrossRef]
- Nie, S.; Wang, J.; Xie, M. Chapter 32—Beta-glucan from Lentinus, Cordyceps, and Ganoderma. In Handbook of Hydrocolloids, 3rd ed.; Phillips, G.O., Williams, P.A., Eds.; Woodhead Publishing: Cambridge, UK, 2021; pp. 1019–1035. [Google Scholar]
- Schupfer, E.; Pak, S.C.; Wang, S.; Micalos, P.S.; Jeffries, T.; Ooi, S.L.; Golombick, T.; Harris, G.; El-Omar, E. The effects and benefits of arabinoxylans on human gut microbiota—A narrative review. Food Biosci. 2021, 43, 101267. [Google Scholar] [CrossRef]
- Nie, Q.; Hu, J.; Chen, H.; Geng, F.; Nie, S. Arabinoxylan ameliorates type 2 diabetes by regulating the gut microbiota and metabolites. Food Chem. 2022, 371, 131106. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Nie, Q.; Hu, J.; Huang, X.; Zhang, K.; Pan, S.; Nie, S. Hypoglycemic and Hypolipidemic Effects of Glucomannan Extracted from Konjac on Type 2 Diabetic Rats. J. Agr. Food Chem. 2019, 67, 5278–5288. [Google Scholar] [CrossRef]
- Munoz-Almagro, N.; Montilla, A.; Villamiel, M. Role of pectin in the current trends towards low-glycaemic food consumption. Food Res. Int. 2021, 140, 109851. [Google Scholar] [CrossRef]
- Barak, S.; Mudgil, D. Locust bean gum: Processing, properties and food applications—A review. Int. J. Biol. Macromol. 2014, 66, 74–80. [Google Scholar] [CrossRef]
- Fang, X.; Zhou, P. Clinical observation of astragalus polysaccharide for injection to reduce the side effects of chemotherapy of stage II colon cancer. Chin. Herb Med. 2010, 34, 657–659. [Google Scholar]
- Preter, V.D.; Joossens, M.; Ballet, V.; Shkedy, Z.; Rutgeerts, P.; Vermeire, S.; Verbeke, K. Metabolic profiling of the impact of oligofructose-enriched inulin in Crohn’s disease patients: A double-blinded randomized controlled trial. Clin. Transl. Gastroenterol. 2013, 4, e30. [Google Scholar] [CrossRef]
- Wei, Y.; Gong, J.; Zhu, W.; Tian, H.; Ding, C.; Gu, L.; Li, N.; Li, J. Pectin enhances the effect of fecal microbiota transplantation in ulcerative colitis by delaying the loss of diversity of gut flora. BMC Microbiol. 2016, 16, 255. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, W.; Zhao, P.; Li, X.; Guo, L.; Gao, W. The potential roles of natural plant polysaccharides in inflammatory bowel disease: A review. Carbohydr. Polym. 2022, 277, 118821. [Google Scholar] [CrossRef] [PubMed]
- Besednova, N.N.; Zaporozhets, T.S.; Kuznetsova, T.A.; Makarenkova, I.D.; Kryzhanovsky, S.P.; Fedyanina, L.N.; Ermakova, S.P. Extracts and marine algae polysaccharides in therapy and prevention of inflammatory diseases of the intestine. Mar. Drugs 2020, 18, 289. [Google Scholar] [CrossRef] [PubMed]
- Niu, W.; Chen, X.; Xu, R.; Dong, H.; Yang, F.; Wang, Y.; Zhang, Z.; Ju, J. Polysaccharides from natural resources exhibit great potential in the treatment of ulcerative colitis: A review. Carbohydr. Polym. 2021, 254, 117189. [Google Scholar] [CrossRef]
- Chelakkot, C.; Ghim, J.; Ryu, S.H. Mechanisms regulating intestinal barrier integrity and its pathological implications. Exp. Mol. Med. 2018, 50, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Shao, S.; Wang, D.; Zheng, W.; Li, X.; Zhang, H.; Zhao, D.; Wang, M. A unique polysaccharide from Hericium erinaceus mycelium ameliorates acetic acid-induced ulcerative colitis rats by modulating the composition of the gut microbiota, short chain fatty acids levels and GPR41/43 respectors. Int. Immunopharmacol. 2019, 71, 411–422. [Google Scholar] [CrossRef]
- Barko, P.C.; McMichael, M.A.; Swanson, K.S.; Williams, D.A. The gastrointestinal microbiome: A review. J. Vet. Intern. Med. 2018, 32, 9–25. [Google Scholar] [CrossRef]
- Sarin, S.K.; Pande, A.; Schnabl, B. Microbiome as a therapeutic target in alcohol-related liver disease. J. Hepatol. 2019, 70, 260–272. [Google Scholar] [CrossRef] [Green Version]
- Singh, R.K.; Chang, H.W.; Yan, D.; Lee, K.M.; Ucmak, D.; Wong, K.; Abrouk, M.; Farahnik, B.; Nakamura, M.; Zhu, T.H.; et al. Influence of diet on the gut microbiome and implications for human health. J. Transl. Med. 2017, 15, 73. [Google Scholar] [CrossRef] [Green Version]
- Do, M.H.; Seo, Y.S.; Park, H.Y. Polysaccharides: Bowel health and gut microbiota. Crit. Rev. Food Sci. Nutr. 2021, 61, 1212–1224. [Google Scholar] [CrossRef]
- Ndeh, D.; Gilbert, H.J. Biochemistry of complex glycan depolymerisation by the human gut microbiota. FEMS Microbiol. Rev. 2018, 42, 146–164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cockburn, D.W.; Koropatkin, N.M. Polysaccharide degradation by the intestinal microbiota and its influence on human health and disease. J. Mol. Biol. 2016, 428, 3230–3252. [Google Scholar] [CrossRef] [PubMed]
- Sartor, R.B. Microbial influences in inflammatory bowel diseases. Gastroenterology 2008, 134, 577–594. [Google Scholar] [CrossRef] [PubMed]
- Han, R.; Ma, Y.; Xiao, J.; You, L.; Pedisic, S.; Liao, L. The possible mechanism of the protective effect of a sulfated polysaccharide from Gracilaria lemaneiformis against colitis induced by dextran sulfate sodium in mice. Food Chem. Toxicol. 2021, 149, 112001. [Google Scholar] [CrossRef]
- Chow, J.; Tang, H.; Mazmanian, S.K. Pathobionts of the gastrointestinal microbiota and inflammatory disease. Curr. Opin. Immunol. 2011, 23, 473–480. [Google Scholar] [CrossRef] [Green Version]
- Gomes-Neto, J.C.; Kittana, H.; Mantz, S.; Munoz, R.R.S.; Schmaltz, R.J.; Bindels, L.B.; Clarke, J.; Hostetter, J.M.; Benson, A.K.; Walter, J.; et al. A gut pathobiont synergizes with the microbiota to instigate inflammatory disease marked by immunoreactivity against other symbionts but not itself. Sci. Rep. 2017, 7, 17707. [Google Scholar] [CrossRef] [Green Version]
- You, S.; Liu, X.; Xu, G.; Ye, M.; Bai, L.; Lin, R.; Sha, X.; Liang, L.; Huang, J.; Zhou, C.; et al. Identification of bioactive polysaccharide from Pseudostellaria heterophylla with its anti-inflammatory effects. J. Funct. Foods 2021, 78, 104353. [Google Scholar] [CrossRef]
- Liu, Y.; Zhao, X.; Lin, T.; Wang, Q.; Zhang, Y.; Xie, J. Molecular mechanisms of polysaccharides from Ziziphus jujuba Mill var. spinosa seeds regulating the bioavailability of spinosin and preventing colitis. Int. J. Biol. Macromol. 2020, 163, 1393–1402. [Google Scholar] [CrossRef]
- Zhang, X.; Zhao, S.; Song, X.; Jia, J.; Zhang, Z.; Zhou, H.; Fu, H.; Cui, H.; Hu, S.; Fang, M.; et al. Inhibition effect of glycyrrhiza polysaccharide (GCP) on tumor growth through regulation of the gut microbiota composition. J. Pharm. Sci. 2018, 137, 324–332. [Google Scholar] [CrossRef]
- Xue, M.; Liang, H.; Ji, X.; Zhou, Z.; Liu, Y.; Sun, T.; Zhang, L. Effects of fucoidan on gut flora and tumor prevention in 1,2-dimethylhydrazine-induced colorectal carcinogenesis. J. Nutr. Biochem. 2020, 82, 108396. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Li, S.; Wang, C.; Lou, Y.; Xia, X.; Xu, H. Whole and polysaccharide powdered Sporisorium reilianum improves DSS-induced colitis in BALB/c mice by modulating gut microbiota. J. Funct. Foods 2021, 79, 104409. [Google Scholar] [CrossRef]
- Shao, X.; Sun, C.Z.; Tang, X.; Zhang, X.S.; Han, D.; Liang, S.; Qu, R.; Hui, X.D.; Shan, Y.W.; Hu, L.H.; et al. Anti-inflammatory and intestinal microbiota modulation properties of Jinxiang garlic (Allium sativum L.) polysaccharides toward dextran sodium sulfate-induced colitis. J. Agric. Food Chem. 2020, 68, 12295–12309. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Zhang, M.; Wang, X.; Ren, Y.; Yue, T.; Wang, Z.; Gao, Z. Edible fungal polysaccharides, the gut microbiota, and host health. Carbohydr. Polym. 2021, 273, 118558. [Google Scholar] [CrossRef]
- Feng, W.; Liu, J.; Tan, Y.; Ao, H.; Wang, J.; Peng, C. Polysaccharides from Atractylodes macrocephala Koidz. Ameliorate ulcerative colitis via extensive modification of gut microbiota and host metabolism. Food Res. Int. 2020, 138, 109777. [Google Scholar] [CrossRef]
- Chen, D.; Chen, G.; Ding, Y.; Wan, P.; Peng, Y.; Chen, C.; Ye, H.; Zeng, X.; Ran, L. Polysaccharides from the flowers of tea (Camellia sinensis L.) modulate gut health and ameliorate cyclophosphamide-induced immunosuppression. J. Funct. Foods 2019, 61, 103470. [Google Scholar] [CrossRef]
- Li, P.; Xiao, N.; Zeng, L.; Xiao, J.; Huang, J.; Xu, Y.; Chen, Y.; Ren, Y.; Du, B. Structural characteristics of a mannoglucan isolated from Chinese yam and its treatment effects against gut microbiota dysbiosis and DSS-induced colitis in mice. Carbohydr. Polym. 2020, 250, 116958. [Google Scholar] [CrossRef]
- Guo, C.; Wang, Y.; Zhang, S.; Zhang, X.; Du, Z.; Li, M.; Ding, K. Crataegus pinnatifida polysaccharide alleviates colitis via modulation of gut microbiota and SCFAs metabolism. Int. J. Biol. Macromol. 2021, 181, 357–368. [Google Scholar] [CrossRef]
- Wu, T.; Shen, M.; Yu, Q.; Chen, Y.; Chen, X.; Yang, J.; Huang, L.; Guo, X.; Xie, J. Cyclocarya paliurus polysaccharide improves metabolic function of gut microbiota by regulating short-chain fatty acids and gut microbiota composition. Food Res. Int. 2021, 141, 110119. [Google Scholar] [CrossRef]
- Wang, Y.J.; Li, Q.M.; Zha, X.Q.; Luo, J.P. Dendrobium fimbriatum Hook polysaccharide ameliorates dextran-sodium-sulfate-induced colitis in mice via improving intestinal barrier function, modulating intestinal microbiota, and reducing oxidative stress and inflammatory responses. Food Funct. 2022, 13, 143–160. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, Z.; Liu, J.; Zheng, Z.; Li, Q.; Wang, H.; Chen, Z.; Wang, K. Identification of the core active structure of a Dendrobium officinale polysaccharide and its protective effect against dextran sulfate sodium-induced colitis via alleviating gut microbiota dysbiosis. Food Res. Int. 2020, 137, 109641. [Google Scholar] [CrossRef] [PubMed]
- Xia, S.; Zhai, Y.; Wang, X.; Fan, Q.; Dong, X.; Chen, M.; Han, T. Phosphorylation of polysaccharides: A review on the synthesis and bioactivities. Int. J. Biol. Macromol. 2021, 184, 946–954. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Dong, J.; Jiang, S.; Liang, Q.; Zhang, Y.; Liu, Z.; Ma, C.; Wang, J.; Kang, W. Effect of Durio zibethinus rind polysaccharide on functional constipation and intestinal microbiota in rats. Food Res. Int. 2020, 136, 109316. [Google Scholar] [CrossRef] [PubMed]
- Zou, Q.H.; Zhang, X.; Liu, X.S.; Li, Y.T.; Tan, Q.L.; Dan, Q.; Yuan, T.; Liu, X.B.; Liu, R.H.; Liu, Z.G. Ficus carica polysaccharide attenuates DSS-induced ulcerative colitis in C57BL/6 mice. Food Funct. 2020, 11, 6666–6679. [Google Scholar] [CrossRef]
- Hino, S.; Ito, H.; Bito, H.; Kawagishi, H.; Morita, T. Ameliorating effects of short-chain inulin-like fructans on the healing stage of Trinitrobenzene Sulfonic Acid-induced colitis in rats. Biosci. Biotechnol. Biochem. 2011, 75, 2169–2174. [Google Scholar] [CrossRef] [Green Version]
- Zeng, Z.Q.; Xie, Z.Y.; Chen, G.J.; Sun, Y.; Zeng, X.X.; Liu, Z.H. Anti-inflammatory and gut microbiota modulatory effects of polysaccharides from Fuzhuan brick tea on colitis in mice induced by dextran sulfate sodium. Food Funct. 2022, 13, 649–663. [Google Scholar] [CrossRef]
- Zhou, X.N.; Lu, Q.Q.; Kang, X.Z.; Tian, G.; Ming, D.G.; Yang, J.L. Protective role of a new polysaccharide extracted from Lonicera japonica Thunb in mice with ulcerative colitis induced by Dextran Sulphate Sodium. Biomed. Res. Int. 2021, 2021, 1–9. [Google Scholar] [CrossRef]
- Gao, L.L.; Ma, J.M.; Fan, Y.N.; Zhang, Y.N.; Ge, R.; Tao, X.J.; Zhang, M.W.; Gao, Q.H.; Yang, J.J. Lycium barbarum polysaccharide combined with aerobic exercise ameliorated nonalcoholic fatty liver disease through restoring gut microbiota, intestinal barrier and inhibiting hepatic inflammation. Int. J. Biol. Macromol. 2021, 183, 1379–1392. [Google Scholar] [CrossRef]
- Jin, M.Y.; Wu, X.Y.; Li, M.Y.; Li, X.T.; Huang, R.M.; Sun, Y.M.; Xu, Z.L. Noni (Morinda citrifolia L.) fruit polysaccharides regulated IBD mice via targeting gut microbiota: Association of JNK/ERK/NF-kappa B signaling pathways. J. Agric. Food Chem. 2021, 69, 10151–10162. [Google Scholar] [CrossRef]
- Gou, Y.; Sun, J.; Liu, J.; Chen, H.; Kan, J.; Qian, C.; Zhang, N.; Jin, C. Structural characterization of a water-soluble purple sweet potato polysaccharide and its effect on intestinal inflammation in mice. J. Funct. Foods 2019, 61, 103502. [Google Scholar] [CrossRef]
- Feng, Z.; Peng, S.; Wu, Z.; Jiao, L.; Xu, S.; Wu, Y.; Liu, Z.; Hu, Y.; Liu, J.; Wu, Y.; et al. Ramulus mori polysaccharide-loaded PLGA nanoparticles and their anti-inflammatory effects in vivo. Int. J. Biol. Macromol. 2021, 182, 2024–2036. [Google Scholar] [CrossRef] [PubMed]
- Su, L.; Mao, C.; Wang, X.; Li, L.; Tong, H.; Mao, J.; Ji, D.; Lu, T.; Hao, M.; Huang, Z.; et al. The anti-colitis effect of Schisandra chinensis polysaccharide is associated with the regulation of the composition and metabolism of gut microbiota. Front. Cell. Infect. Microbiol. 2020, 10, 519479. [Google Scholar] [CrossRef] [PubMed]
- Cui, L.; Guan, X.; Ding, W.; Luo, Y.; Wang, W.; Bu, W.; Song, J.; Tan, X.; Sun, E.; Ning, Q.; et al. Scutellaria baicalensis Georgi polysaccharide ameliorates DSS-induced ulcerative colitis by improving intestinal barrier function and modulating gut microbiota. Int. J. Biol. Macromol. 2021, 166, 1035–1045. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Chen, X.; Hao, L.; Du, P.; Li, C.; Han, H.; Xu, H.; Liu, L. The bioavailability of soybean polysaccharides and their metabolites on gut microbiota in the simulator of the human intestinal microbial ecosystem (SHIME). Food Chem. 2021, 362, 130233. [Google Scholar] [CrossRef] [PubMed]
- Zha, Z.Q.; Lv, Y.; Tang, H.L.; Li, T.T.; Miao, Y.H.; Cheng, J.W.; Wang, G.Q.; Tan, Y.F.; Zhu, Y.; Xing, X.; et al. An orally administered butyrate-releasing xylan derivative reduces inflammation in dextran sulphate sodium-induced murine colitis. Int. J. Biol. Macromol. 2020, 156, 1217–1233. [Google Scholar] [CrossRef]
- Ji, X.; Hou, C.; Zhang, X.; Han, L.; Yin, S.; Peng, Q.; Wang, M. Microbiome-metabolomic analysis of the impact of Zizyphus jujuba cv. Muzao polysaccharides consumption on colorectal cancer mice fecal microbiota and metabolites. Int. J. Biol. Macromol. 2019, 131, 1067–1076. [Google Scholar] [CrossRef]
- Ji, X.L.; Hou, C.Y.; Gao, Y.G.; Xue, Y.Q.; Yan, Y.Z.; Guo, X.D. Metagenomic analysis of gut microbiota modulatory effects of jujube (Ziziphus jujuba Mill.) polysaccharides in a colorectal cancer mouse model. Food Funct. 2020, 11, 163–173. [Google Scholar] [CrossRef]
- Mou, J.; Li, Q.; Shi, W.; Qi, X.; Song, W.; Yang, J. Chain conformation, physicochemical properties of fucosylated chondroitin sulfate from sea cucumber Stichopus chloronotus and its in vitro fermentation by human gut microbiota. Carbohydr. Polym. 2020, 228, 115359. [Google Scholar] [CrossRef]
- Liu, Z.; Zhang, Y.; Ai, C.; Wen, C.; Dong, X.; Sun, X.; Cao, C.; Zhang, X.; Zhu, B.; Song, S. Gut microbiota response to sulfated sea cucumber polysaccharides in a differential manner using an in vitro fermentation model. Food Res. Int. 2021, 148, 110562. [Google Scholar] [CrossRef]
- Cai, B.; Pan, J.; Chen, H.; Chen, X.; Ye, Z.; Yuan, H.; Sun, H.; Wan, P. Oyster polysaccharides ameliorate intestinal mucositis and improve metabolism in 5-fluorouracil-treated S180 tumour-bearing mice. Carbohydr. Polym. 2021, 256, 117545. [Google Scholar] [CrossRef]
- Liu, W.; Tang, S.; Zhao, Q.; Zhang, W.; Li, K.; Yao, W.; Gao, X. The α-D-glucan from marine fungus Phoma herbarum YS4108 ameliorated mice colitis by repairing mucosal barrier and maintaining intestinal homeostasis. Int. J. Biol. Macromol. 2020, 149, 1180–1188. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Dai, W.J.; Tao, H.; Zhuang, W.; Qu, M.; Chang, Y.N. Polysaccharide isolated from Auricularia auricular-judae (Bull.) prevents dextran sulfate sodium-induced colitis in mice through modulating the composition of the gut microbiota. J. Food Sci. 2020, 85, 2943–2951. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; Guo, D.; Fang, L.; Sang, T.; Wu, J.; Guo, C.; Wang, Y.; Wang, Y.; Chen, C.; Chen, J.; et al. Ganoderma lucidum polysaccharide modulates gut microbiota and immune cell function to inhibit inflammation and tumorigenesis in colon. Carbohydr. Polym. 2021, 267, 118231. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Liu, Y.; Chen, B.; Zhang, G.; Ou, S.; Luo, J.; Peng, X. Ganoderma lucidum polysaccharide improves rat DSS-induced colitis by altering cecal microbiota and gene expression of colonic epithelial cells. Food Nutr. Res. 2019, 63, 1559. [Google Scholar] [CrossRef]
- Sang, T.; Guo, C.; Guo, D.; Wu, J.; Wang, Y.; Wang, Y.; Chen, J.; Chen, C.; Wu, K.; Na, K.; et al. Suppression of obesity and inflammation by polysaccharide from sporoderm-broken spore of Ganoderma lucidum via gut microbiota regulation. Carbohydr. Polym. 2021, 256, 117594. [Google Scholar] [CrossRef]
- Xu, Y.; Xie, L.; Zhang, Z.; Zhang, W.; Tang, J.; He, X.; Zhou, J.; Peng, W. Tremella fuciformis polysaccharides inhibited colonic inflammation in Dextran Sulfate Sodium-treated mice via Foxp3+ T cells, gut microbiota, and bacterial metabolites. Front. Immunol. 2021, 12, 648162. [Google Scholar] [CrossRef]
- Kanwal, S.; Joseph, T.P.; Aliya, S.; Song, S.; Saleem, M.Z.; Nisar, M.A.; Wang, Y.; Meyiah, A.; Ma, Y.; Xin, Y. Attenuation of DSS induced colitis by Dictyophora indusiata polysaccharide (DIP) via modulation of gut microbiota and inflammatory related signaling pathways. J. Funct. Foods 2020, 64, 103641. [Google Scholar] [CrossRef]
- Zhang, R.; Yuan, S.; Ye, J.; Wang, X.; Zhang, X.; Shen, J.; Yuan, M.; Liao, W. Polysaccharide from Flammuliana velutipes improves colitis via regulation of colonic microbial dysbiosis and inflammatory responses. Int. J. Biol. Macromol. 2020, 149, 1252–1261. [Google Scholar] [CrossRef]
- Zhou, X.; Hong, T.; Yu, Q.; Nie, S.; Gong, D.; Xiong, T.; Xie, M. Exopolysaccharides from Lactobacillus plantarum NCU116 induce c-Jun dependent Fas/Fasl-mediated apoptosis via TLR2 in mouse intestinal epithelial cancer cells. Sci. Rep. 2017, 7, 14247. [Google Scholar] [CrossRef]
- Ren, X.; Liu, L.; Liu, P.; Gamallat, Y.; Xin, Y.; Shang, D. Polysaccharide extracted from Enteromorpha ameliorates cisplastin-induced small intestine injury in mice. J. Funct. Foods 2018, 49, 154–161. [Google Scholar] [CrossRef]
- Liu, X.; Xi, X.; Jia, A.; Zhang, M.; Cui, T.; Bai, X.; Shi, Y.; Liu, C. A fucoidan from Sargassum fusiforme with novel structure and its regulatory effects on intestinal microbiota in high-fat diet-fed mice. Food Chem. 2021, 358, 129908. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.Y.; Aweya, J.J.; Li, N.; Deng, R.Y.; Chen, W.Y.; Tang, J.; Cheong, K.L. Microbial catabolism of Porphyra haitanensis polysaccharides by human gut microbiota. Food Chem. 2019, 289, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Nie, Y.; Lin, Q.L.; Luo, F.J. Effects of non-starch polysaccharides on inflammatory bowel disease. Int. J. Mol. Sci. 2017, 18, 1372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gonçalves, P.; Araújo, J.R.; Santo, J.P.D. A cross-talk between microbiota-derived short-chain fatty acids and the host mucosal immune system regulates intestinal homeostasis and inflammatory bowel disease. Inflamm. Bowel Dis. 2018, 24, 558–572. [Google Scholar] [CrossRef] [Green Version]
- Thangaraju, M.; Cresci, G.A.; Liu, K.; Ananth, S.; Gnanaprakasam, J.P.; Browning, D.D.; Mellinger, J.D.; Smith, S.B.; Digby, G.J.; Lambert, N.A.; et al. GPR109A is a G-protein-coupled receptor for the bacterial fermentation product butyrate and functions as a tumor suppressor in colon. Cancer Res. 2009, 69, 2826–2832. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Furusawa, Y.; Obata, Y.; Fukuda, S.; Endo, T.A.; Nakato, G.; Takahashi, D.; Nakanishi, Y.; Uetake, C.; Kato, K.; Kato, T.; et al. Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells. Nature 2013, 504, 446–450. [Google Scholar] [CrossRef]
- Singh, N.; Gurav, A.; Sivaprakasam, S.; Brady, E.; Padia, R.; Shi, H.; Thangaraju, M.; Puttur, D.P.; Manicassamy, S.; Munn, D.H.; et al. Activation of Gpr109a, receptor for niacin and the commensal metabolite butyrate, suppresses colonic inflammation and carcinogenesis. Immunity 2014, 40, 128–139. [Google Scholar] [CrossRef] [Green Version]
- Canani, R.B.; Costanzo, M.D.; Leone, L. The epigenetic effects of butyrate: Potential therapeutic implications for clinical practice. Clin. Epigenetics 2012, 4, 4. [Google Scholar] [CrossRef]
- Tan, J.; McKenzie, C.; Potamitis, M.; Thorburn, A.N.; Mackay, C.R.; Macia, L. The role of short-chain fatty acids in health and disease. In Advances in Immunology; Alt, F.W., Ed.; Academic Press: Cambridge, MA, USA, 2014; Volume 121, pp. 91–119. [Google Scholar]
- Lu, S.Y.; Liu, Y.; Tang, S.; Zhang, W.; Yu, Q.; Shi, C.; Cheong, K.-L. Gracilaria lemaneiformis polysaccharides alleviate colitis by modulating the gut microbiota and intestinal barrier in mice. Food Chem. X 2022, 13, 100197. [Google Scholar] [CrossRef]
- Kang, Y.; Yang, G.; Zhang, S.; Ross, C.F.; Zhu, M.J. Goji berry modulates gut microbiota and alleviates colitis in IL-10-deficient mice. Mol. Nutr. Food Res. 2018, 62, e1800535. [Google Scholar] [CrossRef]
- Lavelle, A.; Sokol, H. Gut microbiota-derived metabolites as key actors in inflammatory bowel disease. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 223–237. [Google Scholar] [CrossRef] [PubMed]
- Menni, C.; Hernandez, M.M.; Vital, M.; Mohney, R.P.; Spector, T.D.; Valdes, A.M. Circulating levels of the anti-oxidant indoleproprionic acid are associated with higher gut microbiome diversity. Gut Microbes 2019, 10, 688–695. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, M.; Ma, N.; He, T.; Johnston, L.J.; Ma, X. Tryptophan (Trp) modulates gut homeostasis via aryl hydrocarbon receptor (AhR). Crit. Rev. Food Sci. Nutr. 2019, 60, 1760–1768. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, T.; Natoli, G. Transcriptional regulation of macrophage polarization: Enabling diversity with identity. Nat. Rev. Immunol. 2011, 11, 750–761. [Google Scholar] [CrossRef]
- Lee, M.; Chang, E.B. Inflammatory Bowel Diseases (IBD) and the microbiome—Searching the crime scene for clues. Gastroenterology 2021, 160, 524–537. [Google Scholar] [CrossRef]
- Wu, J.; Zhao, Y.; Wang, X.; Kong, L.; Johnston, L.J.; Lu, L.; Ma, X. Dietary nutrients shape gut microbes and intestinal mucosa via epigenetic modifications. Crit. Rev. Food Sci. Nutr. 2022, 62, 783–797. [Google Scholar] [CrossRef]
- Tsilimigras, M.C.B.; Fodor, A.; Jobin, C. Carcinogenesis and therapeutics: The microbiota perspective. Nat. Microbiol. 2017, 2, 17008. [Google Scholar] [CrossRef]
- Bergstrom, K.; Shan, X.; Casero, D.; Batushansky, A.; Lagishetty, V.; Jacobs, J.P.; Hoover, C.; Kondo, Y.; Shao, B.; Gao, L.J.S. Proximal colon-derived O-glycosylated mucus encapsulates and modulates the microbiota. Science 2020, 370, 467–472. [Google Scholar] [CrossRef]
- Huo, J.; Wu, Z.; Sun, W.; Wang, Z.; Wu, J.; Huang, M.; Wang, B.; Sun, B. Protective effects of natural polysaccharides on intestinal barrier injury: A review. J. Agric. Food Chem. 2022, 70, 711–735. [Google Scholar] [CrossRef]
- Wong, C.; Harris, P.; Ferguson, L. Potential benefits of dietary fibre intervention in inflammatory bowel disease. Int. J. Mol. Sci. 2016, 17, 919. [Google Scholar] [CrossRef] [Green Version]
- de Vrese, M.; Marteau, P.R. Probiotics and prebiotics: Effects on diarrhea. J. Nutr. 2007, 137, 803S–811S. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ren, Y.; Geng, Y.; Du, Y.; Li, W.; Lu, Z.M.; Xu, H.Y.; Xu, G.H.; Shi, J.S.; Xu, Z.H. Polysaccharide of Hericium erinaceus attenuates colitis in C57BL/6 mice via regulation of oxidative stress, inflammation-related signaling pathways and modulating the composition of the gut microbiota. J. Nutr. Biochem. 2018, 57, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.; Yeoh, B.S.; Walker, R.E.; Xiao, X.; Saha, P.; Golonka, R.M.; Cai, J.; Bretin, A.C.A.; Cheng, X.; Liu, Q.; et al. Microbiota fermentation-NLRP3 axis shapes the impact of dietary fibres on intestinal inflammation. Gut 2019, 68, 1801–1812. [Google Scholar] [CrossRef] [PubMed]
- Landy, J.; Ronde, E.; English, N.; Clark, S.K.; Hart, A.L.; Knight, S.C.; Ciclitira, P.J.; Al-Hassi, H.O. Tight junctions in inflammatory bowel diseases and inflammatory bowel disease associated colorectal cancer. World J. Gastroenterol. 2016, 22, 3117–3126. [Google Scholar] [CrossRef] [PubMed]
- Hou, W.; Meng, Q. lnjury of intestinal mucosaI barrier function and its restitution. Chin. Gen. Pract. 2010, 13, 2295–2297. [Google Scholar]
- Belkaid, Y.; Hand, T.W. Role of the microbiota in immunity and inflammation. Cell 2014, 157, 121–141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kamada, N.; Núñez, G. Role of the gut microbiota in the development and function of lymphoid cells. J. Immunol. 2013, 190, 1389–1395. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Zhang, C.; Guo, C.; Li, X. Chitosan ameliorates DSS-induced ulcerative colitis mice by enhancing intestinal barrier function and improving microflora. Int. J. Mol. Sci. 2019, 20, 5751. [Google Scholar] [CrossRef] [Green Version]
- Yang, W.; Ren, D.; Zhao, Y.; Liu, L.; Yang, X. Fuzhuan brick tea polysaccharide improved ulcerative colitis in association with gut microbiota-derived tryptophan metabolism. J. Agric. Food Chem. 2021, 69, 8448–8459. [Google Scholar] [CrossRef]
- Birchenough, G.M.H.; Johansson, M.E.; Gustafsson, J.K.; Bergström, J.H.; Hansson, G.C. New developments in goblet cell mucus secretion and function. Mucosal Immunol. 2015, 8, 712–719. [Google Scholar] [CrossRef] [Green Version]
- Sommer, F.; Rühlemann, M.C.; Bang, C.; Höppner, M.; Rehman, A.; Kaleta, C.; Schmitt-Kopplin, P.; Dempfle, A.; Weidinger, S.; Ellinghaus, E.; et al. Microbiomarkers in inflammatory bowel diseases: Caveats come with caviar. Gut 2017, 66, 1734–1738. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.; Ding, R.; Sun, J.; Liu, J.; Kan, J.; Jin, C. The mpacts of natural polysaccharides on intestinal microbiota and immune responses—A review. Food Funct. 2019, 10, 2290–2312. [Google Scholar] [CrossRef] [PubMed]
- Song, Q.; Wang, Y.; Huang, L.; Shen, M.; Yu, Y.; Yu, Q.; Chen, Y.; Xie, J. Review of the relationships among polysaccharides, gut microbiota, and human health. Food Res. Int. 2021, 140, 109858. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, I.I.; Frutos, R.L.; Manel, N.; Yoshinaga, K.; Rifkin, D.B.; Sartor, R.B.; Finlay, B.B.; Littman, D.R. Specific microbiota direct the differentiation of IL-17-producing T-Helper cells in the mucosa of the small intestine. Cell Host Microbe 2008, 4, 337–349. [Google Scholar] [CrossRef] [Green Version]
- Tao, J.-H.; Duan, J.-A.; Zhang, W.; Jiang, S.; Guo, J.-M.; Wei, D.-D. Polysaccharides from Chrysanthemum morifolium Ramat ameliorate colitis rats via regulation of the metabolic profiling and NF-κ B/TLR4 and IL-6/JAK2/STAT3 signaling pathways. Front. Pharm. 2018, 9, 746. [Google Scholar] [CrossRef]
- Zhou, X.; Zhang, D.; Qi, W.; Hong, T.; Xiong, T.; Wu, T.; Geng, F.; Xie, M.; Nie, S. Exopolysaccharides from Lactobacillus plantarum NCU116 facilitate intestinal homeostasis by modulating intestinal epithelial regeneration and microbiota. J. Agric. Food Chem. 2021, 69, 7863–7873. [Google Scholar] [CrossRef]
- Sun, J.; Chen, H.; Kan, J.; Gou, Y.; Liu, J.; Zhang, X.; Wu, X.; Tang, S.; Sun, R.; Qian, C.; et al. Anti-inflammatory properties and gut microbiota modulation of an alkali-soluble polysaccharide from purple sweet potato in DSS-induced colitis mice. Int. J. Biol. Macromol. 2020, 153, 708–722. [Google Scholar] [CrossRef]
- Wang, Y.; Ji, X.; Yan, M.; Chen, X.; Kang, M.; Teng, L.; Wu, X.; Chen, J.; Deng, C. Protective effect and mechanism of polysaccharide from Dictyophora indusiata on dextran sodium sulfate-induced colitis in C57BL/6 mice. Int. J. Biol. Macromol. 2019, 140, 973–984. [Google Scholar] [CrossRef]
- Kostic, A.D.; Xavier, R.J.; Gevers, D. The microbiome in inflammatory bowel disease: Current status and the future ahead. Gastroenterology 2014, 146, 1489–1499. [Google Scholar] [CrossRef] [Green Version]
- Lv, J.; Zhang, Y.; Tian, Z.; Liu, F.; Shi, Y.; Liu, Y.; Xia, P. Astragalus polysaccharides protect against dextran sulfate sodium-induced colitis by inhibiting NF-κB activation. Int. J. Biol. Macromol. 2017, 98, 723–729. [Google Scholar] [CrossRef]
- Zhang, L.J.; Huang, X.J.; Shi, X.D.; Chen, H.H.; Cui, S.W.; Nie, S.P. Protective effect of three glucomannans from different plants against DSS induced colitis in female BALB/c mice. Food Funct. 2019, 10, 1928–1939. [Google Scholar] [CrossRef] [PubMed]
- Gibson, D.L.; Ma, C.; Bergstrom, K.S.; Huang, J.T.; Man, C.; Vallance, B.A. MyD88 signalling plays a critical role in host defence by controlling pathogen burden and promoting epithelial cell homeostasis during Citrobacter rodentium-induced colitis. Cell Microbiol. 2008, 10, 618–631. [Google Scholar] [CrossRef] [PubMed]
- Dong, N.; Li, X.; Xue, C.; Wang, C.; Xu, X.; Bi, C.; Shan, A.; Li, D. Astragalus polysaccharides attenuated inflammation and balanced the gut microflora in mice challenged with Salmonella typhimurium. Int. Immunopharmacol. 2019, 74, 105681. [Google Scholar] [CrossRef] [PubMed]
- Tostanoski, L.H.; Eppler, H.B.; Xia, B.; Zeng, X.; Jewell, C.M. Engineering release kinetics with polyelectrolyte multilayers to modulate TLR signaling and promote immune tolerance. Biomater. Sci. 2019, 7, 798–808. [Google Scholar] [CrossRef]
- Cui, L.; Wang, W.; Luo, Y.; Ning, Q.; Xia, Z.; Chen, J.; Feng, L.; Wang, H.; Song, J.; Tan, X.; et al. Polysaccharide from Scutellaria baicalensis Georgi ameliorates colitis via suppressing NF-κB signaling and NLRP3 inflammasome activation. Int. J. Biol. Macromol. 2019, 132, 393–405. [Google Scholar] [CrossRef]
- Jia, J.H.; Zhang, P.P.; Zhang, C.X.; Jiang, G.P.; Zheng, W.Y.; Song, S.; Ai, C.Q. Sulfated polysaccharides from pacific abalone attenuated DSS-induced acute and chronic ulcerative colitis in mice via regulating intestinal micro-ecology and the NF-kappa B pathway. Food Funct. 2021, 12, 11351–11365. [Google Scholar] [CrossRef]
- Anish, C.; Beurret, M.; Poolman, J. Combined effects of glycan chain length and linkage type on the immunogenicity of glycoconjugate vaccines. Npj Vaccines 2021, 6, 150. [Google Scholar] [CrossRef]
| Natural Source | Intestinal Microbiota Modulation | Changes in SCFAs and LA | References |
|---|---|---|---|
| Plant | |||
| Allium sativum L. | ↑Lactobacillaceae ↓Lachnospiraceae, ↓Muribaculaceae, ↓Marinifilaceae | ↑AA> PA> IBA> IVA | [45] |
| Arctium lappa | ↑Firmicutes, ↑Ruminococcaceae, ↑Lachnospiraceae, ↑Lactobacillus ↓Proteobacteria, ↓Alcaligenaceae, ↓Staphylococcus, ↓Bacteroidetes | - | [46] |
| Atractylodes macrocephala Koidz | ↑Butyricicoccus, ↑Lactobacillus ↓Actinobacteria, ↓Akkermansia, ↓Anaeroplasma, ↓Bifidobacterium, ↓Erysipelatoclostridium, ↓Faecalibaculum, ↓Parasutterella, ↓Parvibacter, ↓Tenericutes, ↓Verrucomicrobia | ↑PA | [47] |
| Camellia sinensis L. | ↑Bacteroidaceae, ↑Prevotellaceae | ↑AA> PA> BA | [48] |
| Chinese yam | ↓Alistipes, ↓Helicobacter, ↓Enterobacteriaceae | - | [49] |
| Crataegus pinnatifida | ↑Alistipes, ↑Odoribacter | ↑AA | [50] |
| Cyclocarya Palinurus | ↑Lactobacillus, ↑Lactobacillaceae, ↓Streptococcaceae | ↑AA> PA> BA> VA | [51] |
| Dendrobium fimbriatum Hook | ↑Romboutsia, ↑Lactobacillus, ↑Odoribacter ↓Parasutterella, ↓Burkholderia, ↓Caballeronia, ↓Paraburkholderia, ↓Acinetobacter | ↑AA> PA> BA | [52] |
| Dendrobium officinale | ↑Bacteroides, ↑Lactobacillus, ↑Ruminococcaceae ↓Proteobacteria | ↑AA> IBA | [53] |
| Diospyros kaki L. | ↑Lactobacillus | ↑PA> BA | [54] |
| Durio zibethinus Murr rind | ↑Lachnospiraceae NK4A136 group | ↑AA> BA> PA | [55] |
| Ficus carica | ↑S24-7, ↑Bacteroides, ↑Coprococus ↓Escherichia, ↓Clostridium | ↑AA> BA | [56] |
| Fructan | ↑Lactobacilli, ↑Bifidobacteria | ↑PA> LA | [57] |
| Fuzhuan brick tea | ↑Bacteroides, ↑Parasutterella, ↑Collinsella | - | [58] |
| Lonicera japonica Thunb | ↑Bifidobacterium, ↑Lactobacilli ↓Escherichia coli, ↓Enterococcus | - | [59] |
| Lycium barbarum | ↓Enterococcaceae ↑Deferribacteraceae | ↑AA> BA> VA | [60] |
| Morinda citrifolia L. | ↑Dubosiella, ↑Muribaculaceae, ↑Ruminococcaceae_UGG-014, ↑Ruminococcus_1 ↓Campylobacter, ↓Escherichia-Shigella, ↓Bilophila, ↓Ochrobactrum, ↓Vibrio | ↑AA> PA> BA | [61] |
| Pseudostellaria heterophylla | ↑Bacteroides, ↑Pseudomonas ↓Verrucomicrobia, ↓Akkermansia | ↑AA> PA> BA | [39] |
| Purple sweet potato | ↑Bifidobacterium, ↑Lactobacillus, ↑Roseburia ↓Bacteroides, ↓Proteobacteria | ↑AA> PA | [62] |
| Ramulus mori | ↑Clostridium XIVa, ↑Mucispirillum, ↑Paraprevotella | ↑AA> PA> BA | [63] |
| Schisandra chinensis | ↑Norank_f_Bacteroidales_S24-7_group, ↑Desulfovibrio, ↑Alistipes ↓Lactobacillus, ↓Turicibacter | ↑PA> BA> VA | [64] |
| Scutellaria baicalensisGeorgi. | ↑Firmicutes, ↑Bifidobacterium, ↑Lactobacillus, ↑Roseburia | ↑AA> PA> BA | [65] |
| Soybean | ↑Bifidobacterium, ↑Lactobacillus | ↑AA> LA> BA> PA> | [66] |
| Xylan (corn cobs) | ↓Oscillibacter, ↓Ruminococcaceae UGC-009, ↓Erysipdatoclostridium, ↓Defluviitaleaceae UCG-01 | ↑BA | [67] |
| Zizyphus jujuba cv. Muzao | ↑Bifidobacterium, ↑Bacteroides, ↑Lactobacillus | ↑AA | [68] |
| Ziziphus jujuba Mill. | ↓Firmicutes ↑Bacteroidetes | - | [69] |
| Animal | |||
| Stichopus chloronotus | ↑Megamonas, ↑Bacteroides, ↑Fusobacterium, ↑Parabacteroides, ↑Prevotella, ↑Faecalibacterium | ↑AA> BA> IVA | [70] |
| Sea cucumber | ↑Parabacteroides distasonis | ↑BA | [71] |
| Oyster | ↑Akkermansia | ↑PA> BA | [72] |
| Fungus | |||
| Marine fungus Phoma herbarum YS4108 | ↑Bacteroidetes ↓Firmicutes, ↓Proteobacteria, ↓Clostridiales, ↓Lachnospiraceae | ↑BA> IVA | [73] |
| Auricularia auricular-judae | ↑Bacteroidetes ↓Firmicutes, ↓Ruminococcus, ↓Deferribacteres, ↓Actinobacteria | - | [74] |
| Ganoderma lucidum | ↑Allobaculum, ↑Bifidobacterium, ↑Christensenellaceae R-7, ↑Choerinum, ↑Lactobacillus, ↑Firmicutes, ↑Paraprevotella, ↑Ruminococcus_1, ↑Fusicatenibacter, ↑Ruminiclostridium_5, ↑Clostridiales ↓Proteobacteria, ↓Escherichia-Shigella, ↓Barnesiella, ↓Anaerotruncus, ↓Tyzzerella | ↑AA> PA> BA | [75,76,77] |
| Tremella fuciformis | ↑Lactobacillus, ↑Ruminococcaceae, ↑Odoribacter, ↑Helicobacter, ↑Marinifilaceae | - | [78] |
| Dictyophora indusiata | ↑Lactobacillus ↓Proteobacteria, ↓Gammaproteobacteria, ↓Bacteroides, ↓Bacteroidaceae, ↓Enterobacteriaceae | - | [79] |
| Flammuliana velutipes | ↑Ruminal butyrivibrios, ↑Roseburia, ↑Bacteroidales family S24-7 | ↑BA> IVA> VA | [80] |
| Hericium erinaceus | ↑Ruminococcaceae, ↑Allobaculum, ↑Desulfovibrionales | ↑AA> BA | [41] |
| Bacteria | |||
| Lactobacillus planta-rum NCU116 | ↑Lactobacillaceae, ↑Bifidobacteriaceae | ↑LA | [81] |
| Algae | |||
| Enteromorpha | ↑Lactobacillus | - | [82] |
| Sargassum fusiforme | - | [83] | |
| Porphyra haitanensis | ↑Bacteroides thetaiotaomicron, ↑Bacteroides ovatus, ↑Defluviitalea saccharophila, ↑Faecalibacterium prausnitzii | ↑AA> PA> BA | [84] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Q.; Li, L.; Li, Q.; Wang, J.; Nie, S.; Xie, M. Influence of Natural Polysaccharides on Intestinal Microbiota in Inflammatory Bowel Diseases: An Overview. Foods 2022, 11, 1084. https://doi.org/10.3390/foods11081084
Li Q, Li L, Li Q, Wang J, Nie S, Xie M. Influence of Natural Polysaccharides on Intestinal Microbiota in Inflammatory Bowel Diseases: An Overview. Foods. 2022; 11(8):1084. https://doi.org/10.3390/foods11081084
Chicago/Turabian StyleLi, Qi, Linyan Li, Qiqiong Li, Junqiao Wang, Shaoping Nie, and Mingyong Xie. 2022. "Influence of Natural Polysaccharides on Intestinal Microbiota in Inflammatory Bowel Diseases: An Overview" Foods 11, no. 8: 1084. https://doi.org/10.3390/foods11081084
APA StyleLi, Q., Li, L., Li, Q., Wang, J., Nie, S., & Xie, M. (2022). Influence of Natural Polysaccharides on Intestinal Microbiota in Inflammatory Bowel Diseases: An Overview. Foods, 11(8), 1084. https://doi.org/10.3390/foods11081084






