Identification of Genetic Markers for the Detection of Bacillus thuringiensis Strains of Interest for Food Safety
Abstract
1. Introduction
2. Materials and Methods
2.1. Whole-Genome Sequencing
2.2. Dataset Definition
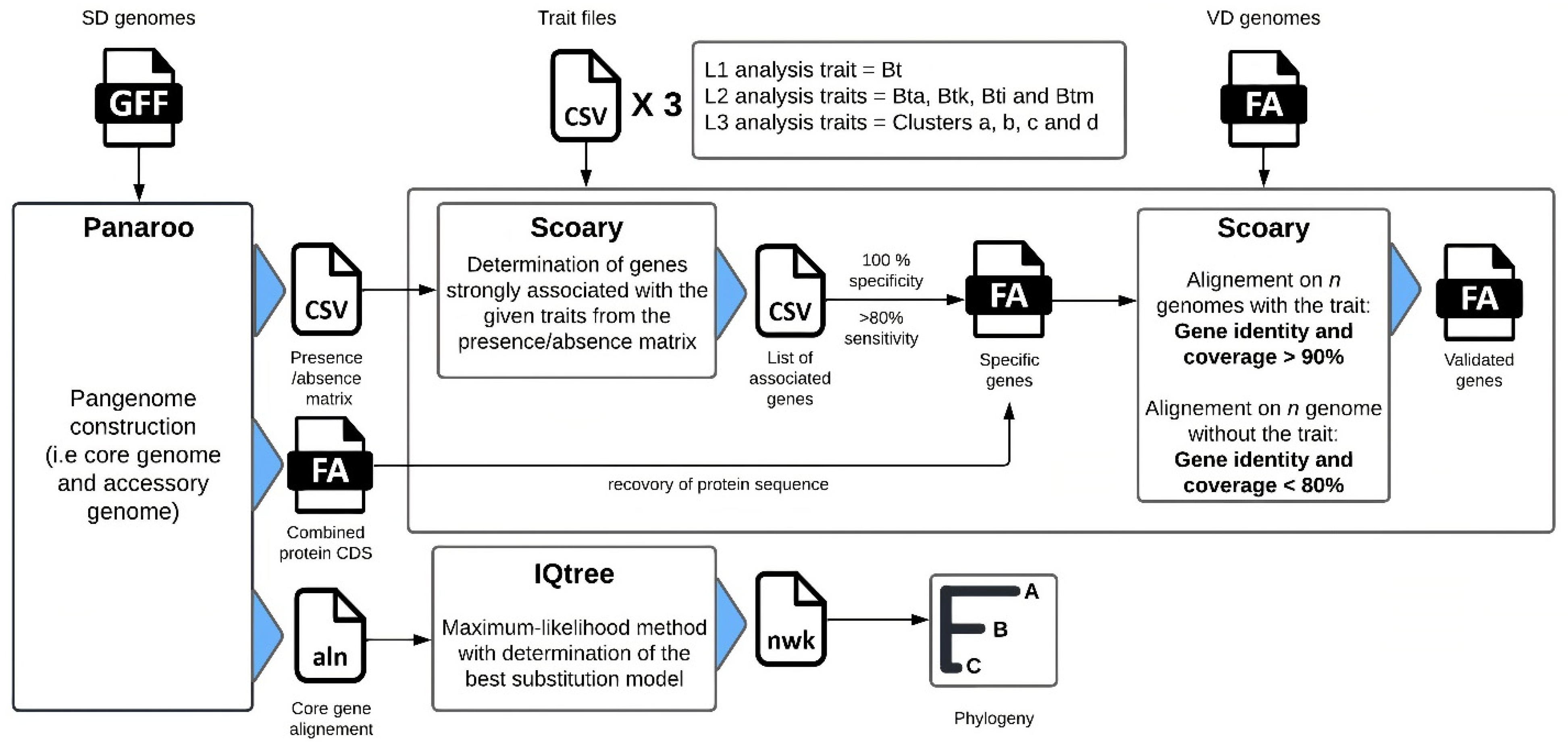
2.3. Pangenome Analysis
2.3.1. Pangenome Construction
2.3.2. Genotype Association
2.3.3. Core Genome Phylogeny
2.3.4. In Silico Validation of Genetic Markers
3. Results
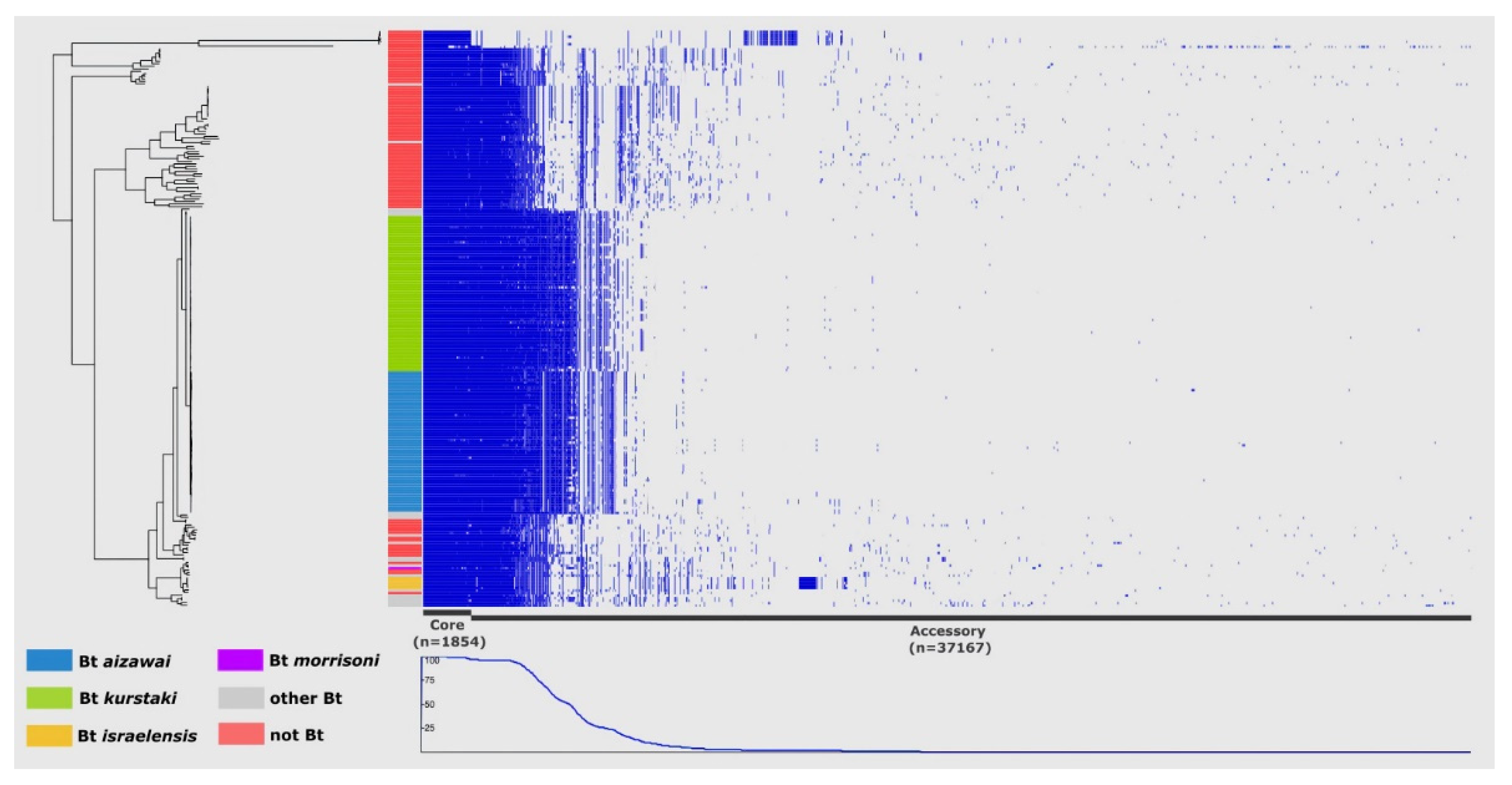
3.1. Bacillus cereus Group Pangenome
3.2. Pan-GWAS Analysis
3.2.1. Gene-Based GWAS
3.2.2. Validation of GWAS Results
3.2.3. Functions and Distribution of Specific Markers
3.2.4. Workflow for Bt Identification
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Miller, R.A.; Beno, S.M.; Kent, D.J.; Carroll, L.M.; Martin, N.H.; Boor, K.J.; Kovac, J. Bacillus wiedmannii sp. nov., a psychrotolerant and cytotoxic Bacillus cereus group species isolated from dairy foods and dairy environments. Int. J. Syst. Evol. Microbiol. 2016, 66, 4744–4753. [Google Scholar] [CrossRef] [PubMed]
- Ehling-Schulz, M.; Lereclus, D.; Koehler, T.M. The Bacillus cereus group: Bacillus species with pathogenic potential. Microbiol. Spectr. 2019, 7. [Google Scholar] [CrossRef]
- Carroll, L.M.; Cheng, R.A.; Wiedmann, M.; Kovac, J. Keeping up with the Bacillus cereus group: Taxonomy through the genomics era and beyond. Crit. Rev. Food Sci. Nutr. 2021, 62, 7677–7702. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Du, J.; Lai, Q.; Zeng, R.; Ye, D.; Xu, J.; Shao, Z. Proposal of nine novel species of the Bacillus cereus group. Int. J. Syst. Evol. Microbiol. 2017, 67, 2499–2508. [Google Scholar] [CrossRef]
- EFSA. The European Union One Health 2019 zoonoses report. EFSA J. 2021, 19, e06406. [Google Scholar] [CrossRef]
- Dietrich, R.; Jessberger, N.; Ehling-Schulz, M.; Martlbauer, E.; Granum, P.E. The food poisoning toxins of Bacillus cereus. Toxins 2021, 13, 98. [Google Scholar] [CrossRef]
- Ceuppens, S.; Rajkovic, A.; Heyndrickx, M.; Tsilia, V.; Van De Wiele, T.; Boon, N.; Uyttendaele, M. Regulation of toxin production by Bacillus cereus and its food safety implications. Crit. Rev. Microbiol. 2011, 37, 188–213. [Google Scholar] [CrossRef]
- David, D.B.; Kirkby, G.R.; Noble, B.A. Bacillus cereus endophthalmitis. Br. J. Ophthalmol. 1994, 78, 577–580. [Google Scholar] [CrossRef]
- Glasset, B.; Herbin, S.; Granier, S.A.; Cavalié, L.; Lafeuille, E.; Guérin, C.; Ruimy, R.; Casagrande-Magne, F.; Levast, M.; Chautemps, N.; et al. Bacillus cereus, a serious cause of nosocomial infections: Epidemiologic and genetic survey. PLoS ONE 2018, 13, e0194346. [Google Scholar] [CrossRef]
- Jovanovic, J.; Ornelis, V.F.M.; Madder, A.; Rajkovic, A. Bacillus cereus food intoxication and toxicoinfection. Compr. Rev. Food Sci. Food Saf. 2021, 20, 3719–3761. [Google Scholar] [CrossRef]
- Hofte, H.; Whiteley, H.R. Insecticidal crystal proteins of Bacillus thuringiensis. Microbiol. Rev. 1989, 53, 242–255. [Google Scholar] [CrossRef] [PubMed]
- Roh, J.-Y.; Choi, J.-Y.; Li, M.-S.; Jin, B.-R.; Je, Y.-H. Bacillus thuringiensis as a specific, safe, and effective tool for insect pest control. J. Microbiol. Biotechnol. 2007, 17, 547–559. [Google Scholar] [PubMed]
- Crickmore, N.; Zeigler, D.R.; Feitelson, J.; Schnepf, E.; Van Rie, J.; Lereclus, D.; Baum, J.; Dean, D.H. Revision of the nomenclature for the Bacillus thuringiensis pesticidal crystal proteins. Microbiol. Mol. Biol. Rev. MMBR 1998, 62, 807–813. [Google Scholar] [CrossRef] [PubMed]
- Crickmore, N.; Berry, C.; Panneerselvam, S.; Mishra, R.; Connor, T.R.; Bonning, B.C. A structure-based nomenclature for Bacillus thuringiensis and other bacteria-derived pesticidal proteins. J. Invertebr. Pathol. 2020, 186, 107438. [Google Scholar] [CrossRef] [PubMed]
- Palma, L.; Munoz, D.; Berry, C.; Murillo, J.; Caballero, P. Bacillus thuringiensis toxins: An overview of their biocidal activity. Toxins 2014, 6, 3296–3325. [Google Scholar] [CrossRef]
- Lacey, L.; Grzywacz, D.; Shapiro-Ilan, D.; Frutos, R.; Brownbridge, M.; Goettel, M. Insect pathogens as biological control agents: Back to the future. J. Invertebr. Pathol. 2015, 132, 1–41. [Google Scholar] [CrossRef] [PubMed]
- Bonis, M.; Felten, A.; Pairaud, S.; Dijoux, A.; Maladen, V.; Mallet, L.; Radomski, N.; Duboisset, A.; Arar, C.; Sarda, X.; et al. Comparative phenotypic, genotypic and genomic analyses of Bacillus thuringiensis associated with foodborne outbreaks in France. PLoS ONE 2021, 16, e0246885. [Google Scholar] [CrossRef]
- De Bock, T.; Zhao, X.; Jacxsens, L.; Devlieghere, F.; Rajkovic, A.; Spanoghe, P.; Höfte, M.; Uyttendaele, M. Evaluation of B. thuringiensis-based biopesticides in the primary production of fresh produce as a food safety hazard and risk. Food Control 2021, 130, 108390. [Google Scholar] [CrossRef]
- Frederiksen, K.; Rosenquist, H.; Jørgensen, K.; Wilcks, A. Occurrence of natural Bacillus thuringiensis contaminants and residues of Bacillus thuringiensis-based insecticides on fresh fruits and vegetables. Appl. Environ. Microbiol. 2006, 72, 3435. [Google Scholar] [CrossRef]
- Frentzel, H.; Juraschek, K.; Pauly, N.; Kelner-Burgos, Y.; Wichmann-Schauer, H. Indications of biopesticidal Bacillus thuringiensis strains in bell pepper and tomato. Int. J. Food Microbiol. 2020, 321, 108542. [Google Scholar] [CrossRef]
- McIntyre, L.; Bernard, K.; Beniac, D.; Isaac-Renton, J.L.; Naseby, D.C. Identification of Bacillus cereus group species associated with food poisoning outbreaks in British Columbia, Canada. Appl. Environ. Microbiol. 2008, 74, 7451. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Risks for public health related to the presence of Bacillus cereus and other Bacillus spp. including Bacillus thuringiensis in foodstuffs. EFSA J. 2016, 14, e04524. [Google Scholar] [CrossRef]
- Bazinet, A.L. Pan-genome and phylogeny of Bacillus cereus sensu lato. BMC Evol. Biol. 2017, 17, 176. [Google Scholar] [CrossRef]
- Guinebretière, M.H.; Thompson, F.L.; Sorokin, A.; Normand, P.; Dawyndt, P.; Ehling-Schulz, M.; Svensson, B.; Sanchis, V.; Nguyen-The, C.; Heyndrickx, M. Ecological diversification in the Bacillus cereus group. Environ. Microbiol. 2008, 10, 851–865. [Google Scholar] [CrossRef] [PubMed]
- Carroll, L.M.; Kovac, J.; Miller, R.A.; Wiedmann, M. Rapid, high-throughput identification of anthrax-causing and emetic Bacillus cereus group genome assemblies via BTyper, a computational tool for virulence-based classification of Bacillus cereus group isolates by using nucleotide sequencing data. Appl. Environ. Microbiol. 2017, 83, e01096-17. [Google Scholar] [CrossRef] [PubMed]
- Carroll, L.M.; Wiedmann, M.; Kovac, J. Proposal of a taxonomic nomenclature for the Bacillus cereus group which reconciles genomic definitions of bacterial species with clinical and industrial phenotypes. mBio 2020, 11, e00034-20. [Google Scholar] [CrossRef]
- Power, R.A.; Parkhill, J.; de Oliveira, T. Microbial genome-wide association studies: Lessons from human GWAS. Nat. Rev. Genet. 2017, 18, 41–50. [Google Scholar] [CrossRef]
- Vila Nova, M.; Durimel, K.; La, K.; Felten, A.; Bessieres, P.; Mistou, M.Y.; Mariadassou, M.; Radomski, N. Genetic and metabolic signatures of Salmonella enterica subsp. enterica associated with animal sources at the pangenomic scale. BMC Genom. 2019, 20, 814. [Google Scholar] [CrossRef]
- Desjardins, C.A.; Cohen, K.A.; Munsamy, V.; Abeel, T.; Maharaj, K.; Walker, B.J.; Shea, T.P.; Almeida, D.V.; Manson, A.L.; Salazar, A. Genomic and functional analyses of Mycobacterium tuberculosis strains implicate ald in D-cycloserine resistance. Nat. Genet. 2016, 48, 544–551. [Google Scholar] [CrossRef]
- Fritsch, L.; Felten, A.; Palma, F.; Mariet, J.F.; Radomski, N.; Mistou, M.Y.; Augustin, J.C.; Guillier, L. Insights from genome-wide approaches to identify variants associated to phenotypes at pan-genome scale: Application to L. monocytogenes ability to grow in cold conditions. Int. J. Food Microbiol. 2019, 291, 181–188. [Google Scholar] [CrossRef]
- Mahamat Abdelrahim, A.; Radomski, N.; Delannoy, S.; Djellal, S.; Le Negrate, M.; Hadjab, K.; Fach, P.; Hennekinne, J.A.; Mistou, M.Y.; Firmesse, O. Large-scale genomic analyses and toxinotyping of Clostridium perfringens implicated in foodborne outbreaks in France. Front. Microbiol. 2019, 10, 777. [Google Scholar] [CrossRef]
- Liu, H.; Zheng, J.; Bo, D.; Yu, Y.; Ye, W.; Peng, D.; Sun, M. BtToxin_Digger: A comprehensive and high-throughput pipeline for mining toxin protein genes from Bacillus thuringiensis. Bioinformatics 2021, 38, 250–251. [Google Scholar] [CrossRef] [PubMed]
- Seemann, T. Prokka: Rapid prokaryotic genome annotation. Bioinformatics 2014, 30, 2068–2069. [Google Scholar] [CrossRef] [PubMed]
- Tonkin-Hill, G.; MacAlasdair, N.; Ruis, C.; Weimann, A.; Horesh, G.; Lees, J.A.; Gladstone, R.A.; Lo, S.; Beaudoin, C.; Floto, R.A.; et al. Producing polished prokaryotic pangenomes with the Panaroo pipeline. Genome Biol. 2020, 21, 180. [Google Scholar] [CrossRef]
- Li, W.; Godzik, A. Cd-hit: A fast program for clustering and comparing large sets of protein or nucleotide sequences. Bioinformatics 2006, 22, 1658–1659. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.T.; Schmidt, H.A.; von Haeseler, A.; Minh, B.Q. IQ-TREE: A fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 2015, 32, 268–274. [Google Scholar] [CrossRef]
- Brynildsrud, O.; Bohlin, J.; Scheffer, L.; Eldholm, V. Rapid scoring of genes in microbial pan-genome-wide association studies with scoary. Genome Biol. 2016, 17, 238. [Google Scholar] [CrossRef]
- Abdi, H. The Bonferonni and Šidák corrections for multiple comparisons. In Encyclopedia of Measurement and Statistics; Sage: Thousand Oaks, CA, USA, 2007; pp. 103–107. [Google Scholar]
- Wright, S.P. Adjusted p-values for simultaneous inference. Biometrics 1992, 48, 1005–1013. [Google Scholar] [CrossRef]
- Katoh, K.; Misawa, K.; Kuma, K.i.; Miyata, T. MAFFT: A novel method for rapid multiple sequence alignment based on fast Fourier transform. Nucleic Acids Res. 2002, 30, 3059–3066. [Google Scholar] [CrossRef]
- Kalyaanamoorthy, S.; Minh, B.Q.; Wong, T.K.F.; von Haeseler, A.; Jermiin, L.S. ModelFinder: Fast model selection for accurate phylogenetic estimates. Nat. Methods 2017, 14, 587–589. [Google Scholar] [CrossRef]
- Hadfield, J.; Croucher, N.J.; Goater, R.J.; Abudahab, K.; Aanensen, D.M.; Harris, S.R. Phandango: An interactive viewer for bacterial population genomics. Bioinformatics 2018, 34, 292–293. [Google Scholar] [CrossRef] [PubMed]
- Letunic, I.; Bork, P. Interactive Tree Of Life (iTOL) v4: Recent updates and new developments. Nucleic Acids Res. 2019, 47, W256–W259. [Google Scholar] [CrossRef] [PubMed]
- Galata, V.; Fehlmann, T.; Backes, C.; Keller, A. PLSDB: A resource of complete bacterial plasmids. Nucleic Acids Res. 2019, 47, D195–D202. [Google Scholar] [CrossRef] [PubMed]
- Zwick, M.E.; Joseph, S.J.; Didelot, X.; Chen, P.E.; Bishop-Lilly, K.A.; Stewart, A.C.; Willner, K.; Nolan, N.; Lentz, S.; Thomason, M.K.; et al. Genomic characterization of the Bacillus cereus sensu lato species: Backdrop to the evolution of Bacillus anthracis. Genome Res. 2012, 22, 1512–1524. [Google Scholar] [CrossRef] [PubMed]
- The UniProt, C. UniProt: The universal protein knowledgebase. Nucleic Acids Res. 2017, 45, D158–D169. [Google Scholar] [CrossRef]
- Yang, J.; Peng, Q.; Chen, Z.; Deng, C.; Shu, C.; Zhang, J.; Huang, D.; Song, F. Transcriptional regulation and characteristics of a novel N-acetylmuramoyl-L-alanine amidase gene involved in Bacillus thuringiensis mother cell lysis. J. Bacteriol. 2013, 195, 2887–2897. [Google Scholar] [CrossRef]
- Park, S.S.; Wong, S.L.; Wang, L.F.; Doi, R.H. Bacillus subtilis subtilisin gene (aprE) is expressed from a sigma A (sigma 43) promoter in vitro and in vivo. J. Bacteriol. 1989, 171, 2657–2665. [Google Scholar] [CrossRef]
- Au, N.; Kuester-Schoeck, E.; Mandava, V.; Bothwell, L.E.; Canny, S.P.; Chachu, K.; Colavito, S.A.; Fuller, S.N.; Groban, E.S.; Hensley, L.A.; et al. Genetic composition of the Bacillus subtilis SOS system. J. Bacteriol. 2005, 187, 7655–7666. [Google Scholar] [CrossRef]
- Krüger, E.; Witt, E.; Ohlmeier, S.; Hanschke, R.; Hecker, M. The clp proteases of Bacillus subtilis are directly involved in degradation of misfolded proteins. J. Bacteriol. 2000, 182, 3259–3265. [Google Scholar] [CrossRef]
- Bongiorni, C.; Ishikawa, S.; Stephenson, S.; Ogasawara, N.; Perego, M. Synergistic regulation of competence development in Bacillus subtilis by two Rap-Phr systems. J. Bacteriol. 2005, 187, 4353–4361. [Google Scholar] [CrossRef]
- Waterhouse, A.; Bertoni, M.; Bienert, S.; Studer, G.; Tauriello, G.; Gumienny, R.; Heer, F.T.; de Beer, T.A.P.; Rempfer, C.; Bordoli, L.; et al. SWISS-MODEL: Homology modelling of protein structures and complexes. Nucleic Acids Res. 2018, 46, W296–W303. [Google Scholar] [CrossRef] [PubMed]
- Knight, D.R.; Imwattana, K.; Kullin, B.; Guerrero-Araya, E.; Paredes-Sabja, D.; Didelot, X.; Dingle, K.E.; Eyre, D.W.; Rodríguez, C.; Riley, T.V. Major genetic discontinuity and novel toxigenic species in Clostridioides difficile taxonomy. eLife 2021, 10, e64325. [Google Scholar] [CrossRef] [PubMed]
- Page, A.J.; Cummins, C.A.; Hunt, M.; Wong, V.K.; Reuter, S.; Holden, M.T.; Fookes, M.; Falush, D.; Keane, J.A.; Parkhill, J. Roary: Rapid large-scale prokaryote pan genome analysis. Bioinformatics 2015, 31, 3691–3693. [Google Scholar] [CrossRef]
- Fayad, N.; Kallassy Awad, M.; Mahillon, J. Diversity of Bacillus cereus sensu lato mobilome. BMC Genom. 2019, 20, 436. [Google Scholar] [CrossRef]
- Meric, G.; Mageiros, L.; Pascoe, B.; Woodcock, D.J.; Mourkas, E.; Lamble, S.; Bowden, R.; Jolley, K.A.; Raymond, B.; Sheppard, S.K. Lineage-specific plasmid acquisition and the evolution of specialized pathogens in Bacillus thuringiensis and the Bacillus cereus group. Mol. Ecol. 2018, 27, 1524–1540. [Google Scholar] [CrossRef]
- Zhu, L.; Peng, D.; Wang, Y.; Ye, W.; Zheng, J.; Zhao, C.; Han, D.; Geng, C.; Ruan, L.; He, J.; et al. Genomic and transcriptomic insights into the efficient entomopathogenicity of Bacillus thuringiensis. Sci. Rep. 2015, 5, 14129. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Lai, Q.; Göker, M.; Meier-Kolthoff, J.P.; Wang, M.; Sun, Y.; Wang, L.; Shao, Z. Genomic insights into the taxonomic status of the Bacillus cereus group. Sci. Rep. 2015, 5, 14082. [Google Scholar] [CrossRef]
- Gillis, A.; Fayad, N.; Makart, L.; Bolotin, A.; Sorokin, A.; Kallassy, M.; Mahillon, J. Role of plasmid plasticity and mobile genetic elements in the entomopathogen Bacillus thuringiensis serovar israelensis. FEMS Microbiol. Rev. 2018, 42, 829–856. [Google Scholar] [CrossRef]
- San, J.E.; Baichoo, S.; Kanzi, A.; Moosa, Y.; Lessells, R.; Fonseca, V.; Mogaka, J.; Power, R.; de Oliveira, T. Current affairs of microbial genome-wide association studies: Approaches, bottlenecks and analytical pitfalls. Front. Microbiol. 2019, 10, 3119. [Google Scholar] [CrossRef]
- Chang, Y.-H.; Shangkuan, Y.-H.; Lin, H.-C.; Liu, H.-W. PCR assay of the groEL gene for detection and differentiation of Bacillus cereus group cells. Appl. Environ. Microbiol. 2003, 69, 4502–4510. [Google Scholar] [CrossRef]
- Chelliah, R.; Wei, S.; Park, B.-J.; Rubab, M.; Dalirii, E.B.-M.; Barathikannan, K.; Jin, Y.-G.; Oh, D.-H. Whole genome sequence of Bacillus thuringiensis ATCC 10792 and improved discrimination of Bacillus thuringiensis from Bacillus cereus group based on novel biomarkers. Microb. Pathog. 2019, 129, 284–297. [Google Scholar] [CrossRef] [PubMed]
- Dzieciol, M.; Fricker, M.; Wagner, M.; Hein, I.; Ehling-Schulz, M. A novel diagnostic real-time PCR assay for quantification and differentiation of emetic and non-emetic Bacillus cereus. Food Control 2013, 32, 176–185. [Google Scholar] [CrossRef]
- Burnham, K.P.; Overton, W.S. Estimation of the size of a closed population when capture probabilities vary among animals. Biometrika 1978, 65, 625–633. [Google Scholar] [CrossRef]
| Species | Subspecies | No. of Genome(s) | Representative Genome | NCBI Accession Number |
|---|---|---|---|---|
| Bacillus anthracis | 1 | Ames | NC_003997.3 | |
| B. cereus (sensu stricto) (Bc s.s.) | 2 | ATCC 14579 | NC_004722.1 | |
| B. cytotoxicus | 1 | NVH 391-98 | NC_009674.1 | |
| B. mycoides | 1 | DSM 2048 | NZ_CM000742.1 | |
| B. pseudomycoides | 1 | DSM 12442 | NZ_CM000745.1 | |
| B. thuringiensis (Bt) | Bt ssp. aizawai | 56 | Leapi01 | AMXS00000000.2 |
| Bt ssp. kurstaki | 57 | HD-1 | NZ_CP004870.1 | |
| Bt ssp. israelensis | 5 | AM6552 | NZ_CP013275.1 | |
| Bt ssp. morrisoni | 1 | BGSC 4AA1 | NZ_CP010577.1 | |
| other and/or unknown | 25 | ATCC 10792 | NZ_CP021061.1 | |
| B. toyonensis | 1 | BCT-7112 | NC_022781.1 | |
| B. weihenstephanensis | 1 | WSBC 10204 | NZ_CP009746.1 | |
| B. wiedmannii | 1 | MM3 | NZ_CM000718.1 | |
| other B. cereus (sensu lato) * | 77 |
| \ | Scoary | TBLASTN | |||
|---|---|---|---|---|---|
| GWAS Analysis | Trait | No. of Genome(s) Associated with Trait | No. of Genome(s) Exempt from Trait | No. of Genes Strongly Associated with Trait * | No. of Genes Validated In Silico (Annotated) |
| L1 (n = 230) | Bacillus thuringiensis (Bt) | 144 | 86 | 99 | 32 (22) |
| L2 (n = 230) | Bt ssp. aizawai | 56 | 169 | 27 | 17 (4) |
| Bt ssp. kurstaki | 57 | 168 | 5 | 3 (0) | |
| Bt ssp. israelensis | 5 | 220 | 809 | NA (78) | |
| Bt ssp. morrisoni | 1 | 224 | 246 | NA (101) | |
| L3 (n = 104) | Cluster a | 11 | 93 | 10 | 6 (0) |
| Cluster b | 40 | 64 | 14 | 10 (1) | |
| Cluster c | 24 | 80 | 46 | 30 (1) | |
| Cluster d | 29 | 75 | 48 | 30 (3) | |
| GWAS Analysis | Gene Name | Replicon | Annotation | UniprotKB | Trait | Marker Sensitivity (%) | Cumulative Sensitivity (%) | p-Value * | NCBI Accession Number |
|---|---|---|---|---|---|---|---|---|---|
| L1 | cwlA | Chromosome | N-acetylmuramoyl-L-alanine amidase CwlA | P24808 | Bt | 93.8 | 93.8 | 1.72 × 10−51 | WP_021728236.1 |
| intQ | Chromosome | Putative defective protein IntQ | P76168 | Bt | 91.7 | 94.4 | 1.64 × 10−48 | WP_000237488.1 | |
| group_3916 | Chromosome | Hypothetical protein | N/A | Bt | 88.9 | 95.1 | 3.29 × 10−47 | WP_000858032.1 | |
| group_20749 | Chromosome | Hypothetical protein | N/A | Bt | 86.8 | 97.2 | 6.36 × 10−45 | WP_042596929.1 | |
| group_20361 | Plasmid | Hypothetical protein | N/A | Bt | 84.7 | 98.6 | 8.43 × 10−43 | WP_002101540.1 | |
| sdpR | Plasmid | Transcriptional repressor SdpR | O32242 | Bt | 84.0 | 100.0 | 4.00 × 10−42 | WP_000998670.1 | |
| L2 | apr | Chromosome | Subtilisin | P04189 | Bta | 100.0 | N/A | 9.10 × 10−53 | WP_021728520.1 |
| group_27293 | Plasmid | Hypothetical protein | N/A | Btk | 98.2 | 98.2 | 5.83 × 10−52 | WP_003273526.1 | |
| group_27336 | Plasmid | Hypothetical protein | N/A | Btk | 89.5 | 100.0 | 1.05 × 10−43 | WP_001293418.1 | |
| L3 | group_10114 | Chromosome | Hypothetical protein | N/A | Cluster a | 100.0 | N/A | 4.48 × 10−15 | WP_000415284.1 |
| rapF | Chromosome | Response regulator aspartate phosphatase F | P71002 | Cluster b | 92.5 | 92.5 | 4.82 × 10−25 | WP_050062578.1 | |
| group_20667 | Plasmid | Hypothetical protein | N/A | Cluster b | 82.5 | 100.0 | 1.34 × 10−20 | WP_131256056.1 | |
| lexA | Chromosome | LexA repressor | P31080 | Cluster c | 100.0 | N/A | 4.31 × 10−24 | AHZ54004.1 | |
| clpP1 | Chromosome | ATP-dependent Clp subunit 1 | B0B803 | Cluster d | 100.0 | N/A | 2.13 × 10−26 | WP_000791073.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fichant, A.; Felten, A.; Gallet, A.; Firmesse, O.; Bonis, M. Identification of Genetic Markers for the Detection of Bacillus thuringiensis Strains of Interest for Food Safety. Foods 2022, 11, 3924. https://doi.org/10.3390/foods11233924
Fichant A, Felten A, Gallet A, Firmesse O, Bonis M. Identification of Genetic Markers for the Detection of Bacillus thuringiensis Strains of Interest for Food Safety. Foods. 2022; 11(23):3924. https://doi.org/10.3390/foods11233924
Chicago/Turabian StyleFichant, Arnaud, Arnaud Felten, Armel Gallet, Olivier Firmesse, and Mathilde Bonis. 2022. "Identification of Genetic Markers for the Detection of Bacillus thuringiensis Strains of Interest for Food Safety" Foods 11, no. 23: 3924. https://doi.org/10.3390/foods11233924
APA StyleFichant, A., Felten, A., Gallet, A., Firmesse, O., & Bonis, M. (2022). Identification of Genetic Markers for the Detection of Bacillus thuringiensis Strains of Interest for Food Safety. Foods, 11(23), 3924. https://doi.org/10.3390/foods11233924






