Emerging Postharvest Technologies to Enhance the Shelf-Life of Fruit and Vegetables: An Overview
Abstract
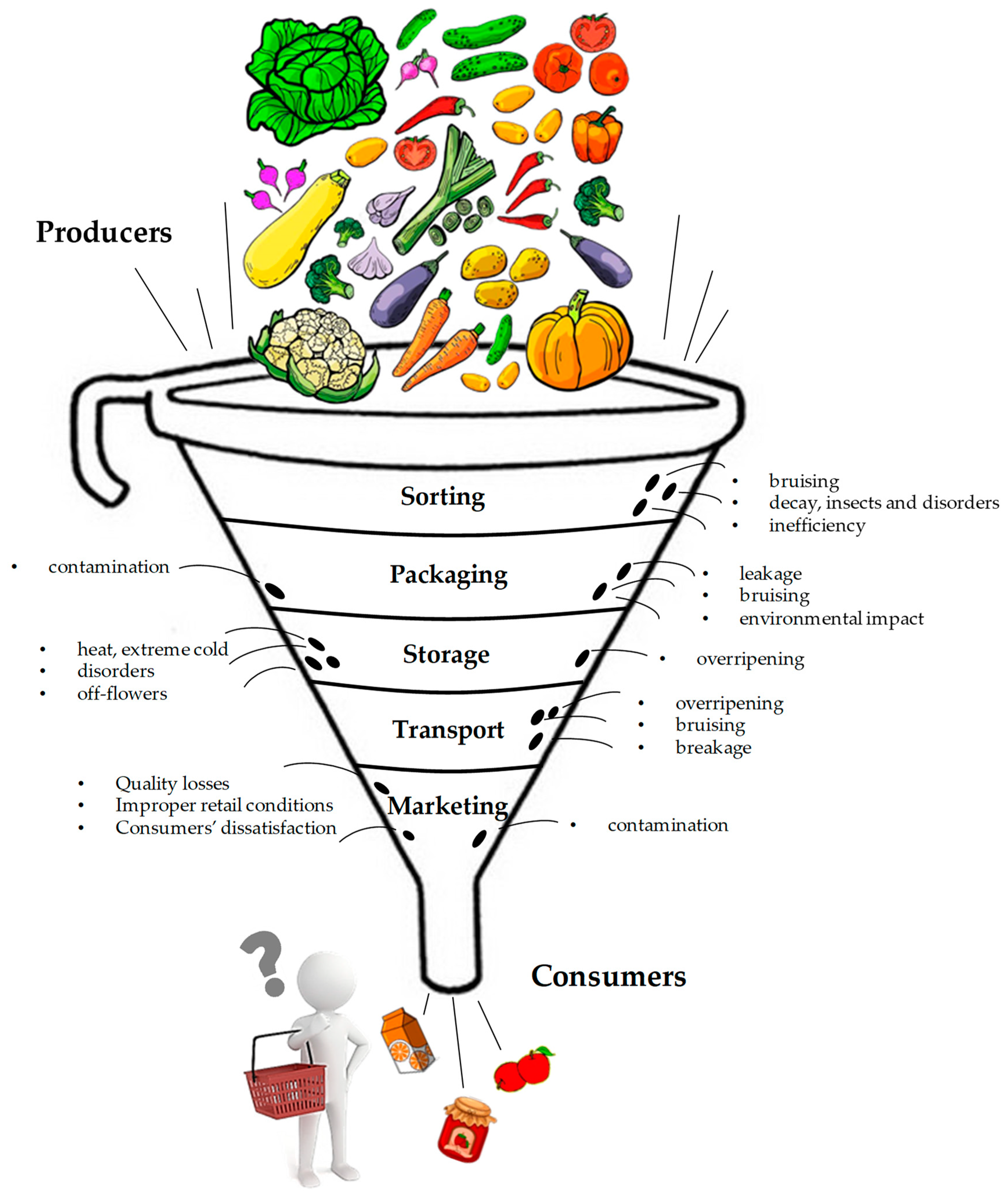
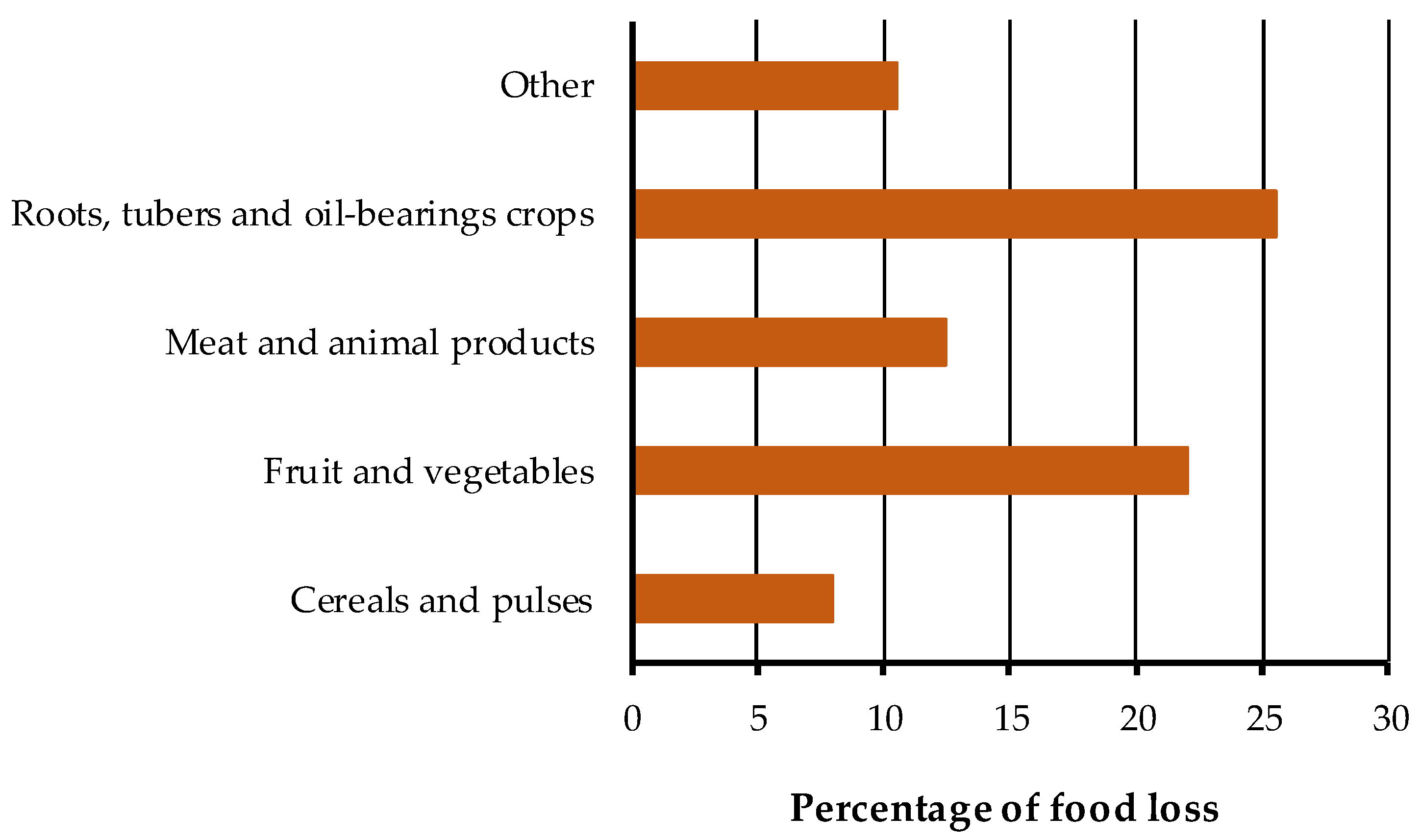
1. Introduction
2. Postharvest Strategies to Extend the Shelf-Life of Fruit and Vegetables
2.1. Physical Treatments
2.1.1. Microwave
2.1.2. Pulsed Electric Field
2.1.3. High Hydrostatic Pressure
2.1.4. Cold Plasma
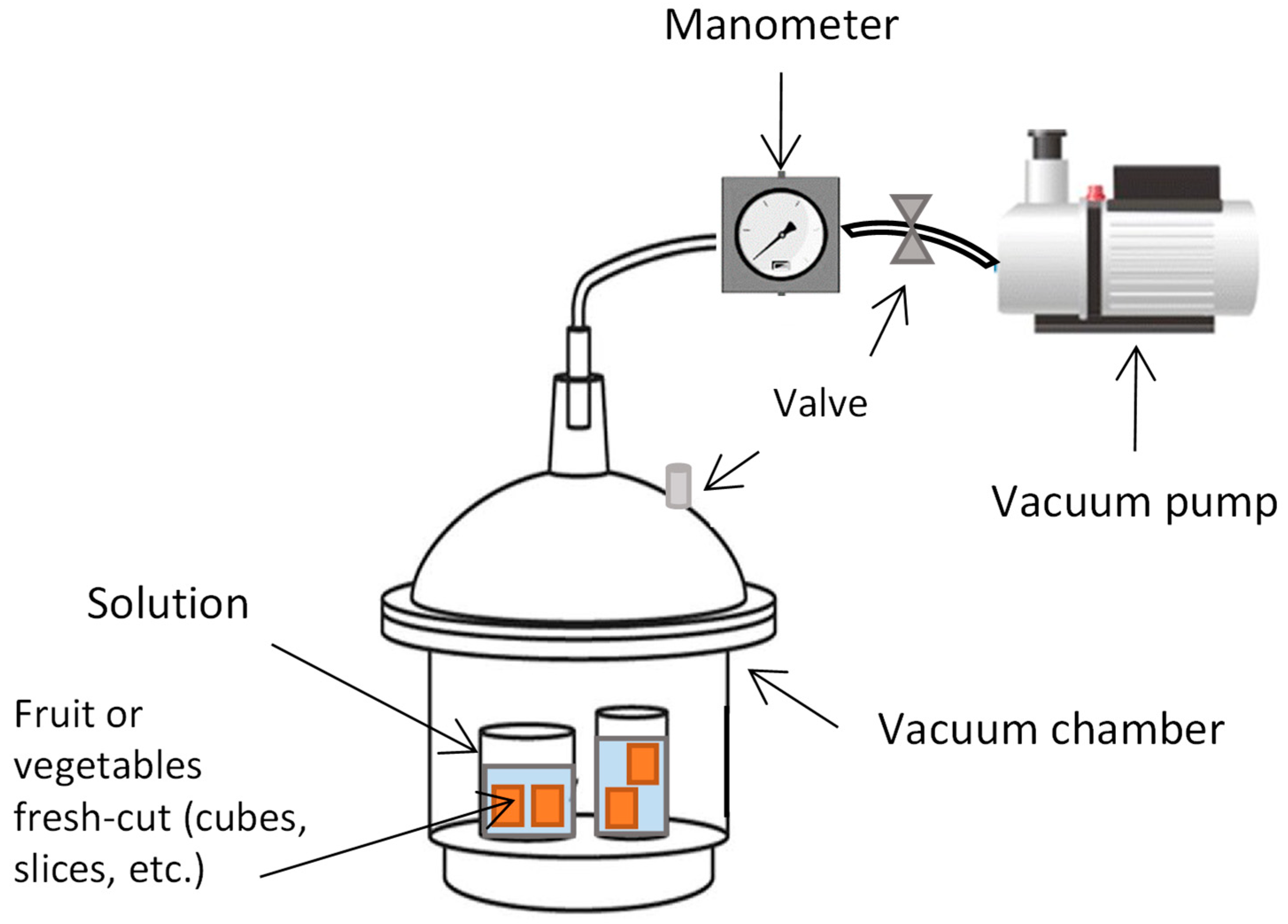
2.2. Dipping and Vacuum Impregnation
2.3. Edible Active Packaging, Based on Natural Compounds
2.4. Strategies of Biocontrol
3. Innovative Non-Destructive Techniques for the Quality Monitoring of Fruit and Vegetables
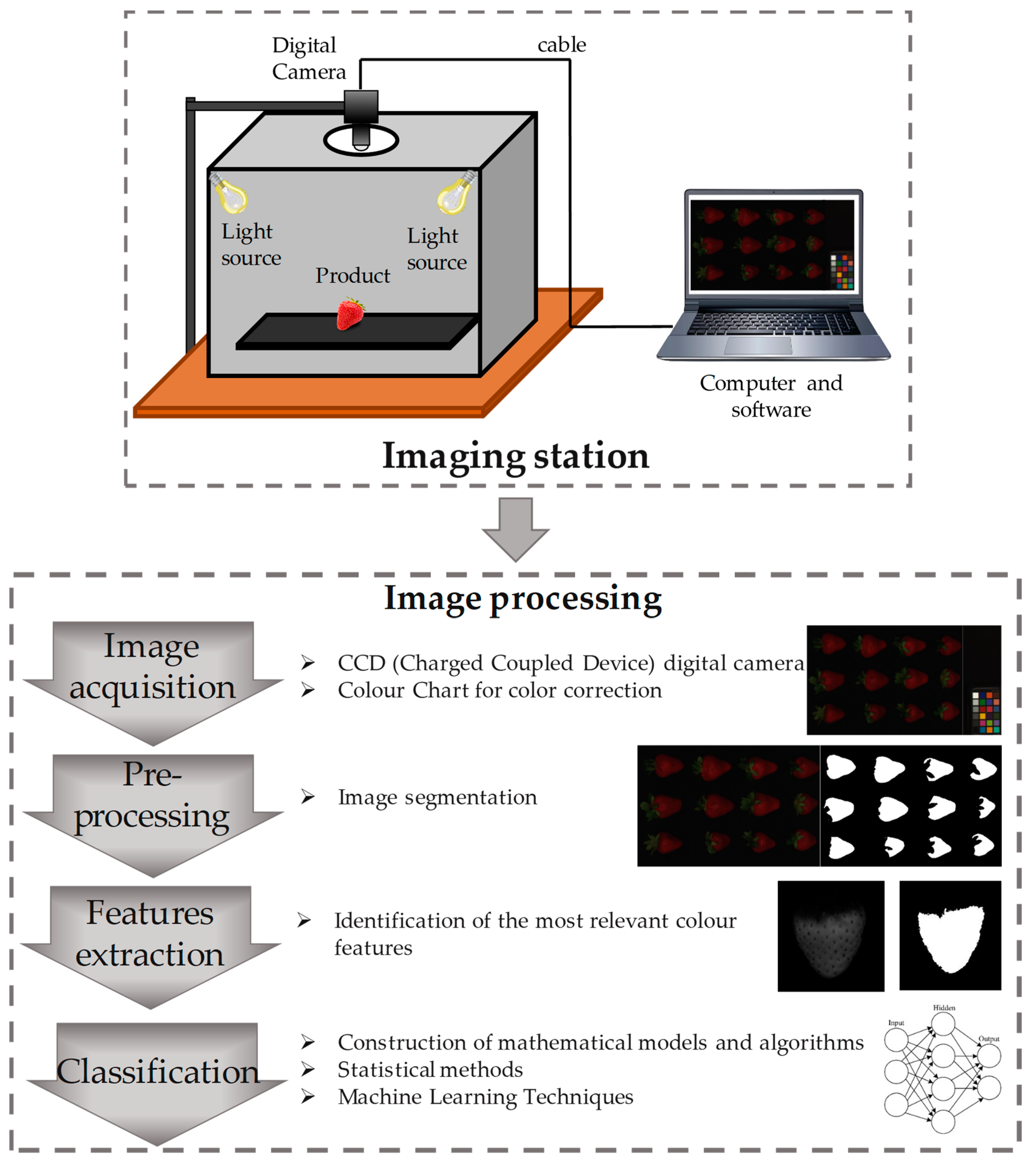
3.1. Image Analysis through a Computer Vision System
3.2. E-nose
3.3. Near-Infrared Spectroscopy
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mahajan, P.V.; Caleb, O.J.; Singh, Z.; Watkins, C.B.; Geyer, M. Postharvest Treatments of Fresh Produce. Philos. Trans. R. Soc. Math. Phys. Eng. Sci. 2014, 372, 20130309. [Google Scholar] [CrossRef] [PubMed]
- Brasil, I.M.; Siddiqui, M.W. Postharvest Quality of Fruits and Vegetables: An Overview. In Preharvest Modulation of Postharvest Fruit and Vegetable Quality; Elsevier: Amsterdam, The Netherlands, 2018; pp. 1–40. ISBN 978-0-12-809807-3. [Google Scholar]
- Chauhan, O.P.; Lakshmi, S.; Pandey, A.K.; Ravi, N.; Gopalan, N.; Sharma, R.K. Non-Destructive Quality Monitoring of Fresh Fruits and Vegetables. Def. Life Sci. J. 2017, 2, 103. [Google Scholar] [CrossRef]
- Barth, M.; Hankinson, T.R.; Zhuang, H.; Breidt, F. Microbiological Spoilage of Fruits and Vegetables. In Compendium of the Microbiological Spoilage of Foods and Beverages; Sperber, W.H., Doyle, M.P., Eds.; Springer New York: New York, NY, USA, 2009; pp. 135–183. ISBN 978-1-4419-0825-4. [Google Scholar]
- De Corato, U. Improving the Shelf-Life and Quality of Fresh and Minimally-Processed Fruits and Vegetables for a Modern Food Industry: A Comprehensive Critical Review from the Traditional Technologies into the Most Promising Advancements. Crit. Rev. Food Sci. Nutr. 2020, 60, 940–975. [Google Scholar] [CrossRef]
- Berger, C.N.; Sodha, S.V.; Shaw, R.K.; Griffin, P.M.; Pink, D.; Hand, P.; Frankel, G. Fresh Fruit and Vegetables as Vehicles for the Transmission of Human Pathogens: Fresh Produce as Vehicles for Transmission of Human Pathogens. Environ. Microbiol. 2010, 12, 2385–2397. [Google Scholar] [CrossRef] [PubMed]
- Srisamran, J.; Atwill, E.R.; Chuanchuen, R.; Jeamsripong, S. Detection and Analysis of Indicator and Pathogenic Bacteria in Conventional and Organic Fruits and Vegetables Sold in Retail Markets. Food Qual. Saf. 2022, 6, fyac013. [Google Scholar] [CrossRef]
- Gustavsson, J.; Cederberg, C.; Sonesson, U. The Methodology of the FAO Study: “Global Food Losses and Food Waste—Extent, Causes and Prevention”—FAO. Available online: https://www.diva-portal.org/smash/get/diva2:944159/FULLTEXT01.pdf (accessed on 29 August 2022).
- Bourne, M. Post Harvest Food Losses—The Neglected Dimension in Increasing the World Food Supply. In Cornell International Agriculture Mimeograph 53; Cornell University: Ithaca, NY, USA, 1977. [Google Scholar]
- SOFA 2019—The State of Food and Agriculture in the World. Available online: https://www.fao.org/state-of-food-agriculture/2019/en/ (accessed on 1 October 2022).
- Singh, V.; Hedayetullah, M.; Zaman, P.; Meher, J. Postharvest Technology of Fruits and Vegetables: An Overview, Journal of Post Harvest Technology. J. Postharvest Technol. 2014, 2, 124–135. [Google Scholar]
- Ali, A.; Yeoh, W.K.; Forney, C.; Siddiqui, M.W. Advances in Postharvest Technologies to Extend the Storage Life of Minimally Processed Fruits and Vegetables. Crit. Rev. Food Sci. Nutr. 2018, 58, 2632–2649. [Google Scholar] [CrossRef]
- Fatchurrahman, D.; Amodio, M.L.; Colelli, G. Quality of Goji Berry Fruit (Lycium barbarum L.) Stored at Different Temperatures. Foods 2022, 11, 3700. [Google Scholar] [CrossRef]
- Kader, A.A. Postharvest Technology of Horticultural Crops; University of California Agriculture and Natural Resources: Oakland, CA, USA, 2002; ISBN 978-1-879906-51-8. [Google Scholar]
- Fahmy, K.; Nakano, K. Effective Transport and Storage Condition for Preserving The Quality of ‘Jiro’ Persimmon in Export Market. Agric. Agric. Sci. Procedia 2016, 9, 279–290. [Google Scholar] [CrossRef][Green Version]
- Bhargava, A.; Bansal, A. Fruits and Vegetables Quality Evaluation Using Computer Vision: A Review. J. King Saud Univ. - Comput. Inf. Sci. 2021, 33, 243–257. [Google Scholar] [CrossRef]
- García-Oliveira, P.; Fraga-Corral, M.; Pereira, A.G.; Prieto, M.A.; Simal-Gandara, J. Solutions for the Sustainability of the Food Production and Consumption System. Crit. Rev. Food Sci. Nutr. 2022, 62, 1765–1781. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Wang, W. Quality of Fresh and Fresh-Cut Produce Impacted by Nonthermal Physical Technologies Intended to Enhance Microbial Safety. Crit. Rev. Food Sci. Nutr. 2022, 62, 362–382. [Google Scholar] [CrossRef] [PubMed]
- Ganesan, A.R.; Tiwari, U.; Ezhilarasi, P.N.; Rajauria, G. Application of Cold Plasma on Food Matrices: A Review on Current and Future Prospects. J. Food Process. Preserv. 2021, 45, e15070. [Google Scholar] [CrossRef]
- Dar, A.H.; Shams, R.; ul Eain Hyder Rizvi, Q.; Majid, I. Microwave and Ohmic Heating of Fresh Cut Fruits and Vegetable Products; Elsevier Inc.: Amsterdam, The Netherlands, 2019; ISBN 9780128161845. [Google Scholar]
- Usall, J.; Ippolito, A.; Sisquella, M.; Neri, F. Physical Treatments to Control Postharvest Diseases of Fresh Fruits and Vegetables. Postharvest Biol. Technol. 2016, 122, 30–40. [Google Scholar] [CrossRef]
- Martínez-Hernández, G.B.; Amodio, M.L.; Colelli, G. Potential Use of Microwave Treatment on Fresh-Cut Carrots: Physical, Chemical and Microbiological Aspects. J. Sci. Food Agric. 2016, 96, 2063–2072. [Google Scholar] [CrossRef]
- Song, L.; Luo, H.; Cheng, X.; Yan, F.; Yang, Z.; Yu, Z. Effects of Microwave Treatment on Physiology and Quality of Minimally Processed Bok Choy (Brassica campestris L.) during Storage at 5 °C. J. Food Meas. Charact. 2018, 12, 913–922. [Google Scholar] [CrossRef]
- Colelli, G.; Amodio, M.L.; de Chiara, M.L.V. Operating Conditions for Microwave Application throughout Production Process to Reduce Microbial Load of Fresh-Cut Apples. Acta Hortic. 2021, 1319, 223–230. [Google Scholar] [CrossRef]
- Vadivambal, R.; Jayas, D.S. Non-Uniform Temperature Distribution During Microwave Heating of Food Materials—A Review. Food Bioprocess Technol. 2010, 3, 161–171. [Google Scholar] [CrossRef]
- Li, J.; Shi, J.; Huang, X.; Wang, T.; Zou, X.; Li, Z.; Zhang, D.; Zhang, W.; Xu, Y. Effects of Pulsed Electric Field Pretreatment on Frying Quality of Fresh-Cut Lotus Root Slices. LWT 2020, 132, 109873. [Google Scholar] [CrossRef]
- López-Gámez, G.; Elez-Martínez, P.; Martín-Belloso, O.; Soliva-Fortuny, R. Pulsed Electric Fields Affect Endogenous Enzyme Activities, Respiration and Biosynthesis of Phenolic Compounds in Carrots. Postharvest Biol. Technol. 2020, 168, 111284. [Google Scholar] [CrossRef]
- López-Gámez, G.; Elez-Martínez, P.; Martín-Belloso, O.; Soliva-Fortuny, R. Enhancing Phenolic Content in Carrots by Pulsed Electric Fields during Post-Treatment Time: Effects on Cell Viability and Quality Attributes. Innov. Food Sci. Emerg. Technol. 2020, 59, 102252. [Google Scholar] [CrossRef]
- López-Gámez, G.; Elez-Martínez, P.; Martín-Belloso, O.; Soliva-Fortuny, R. Changes of Carotenoid Content in Carrots after Application of Pulsed Electric Field Treatments. LWT 2021, 147, 111408. [Google Scholar] [CrossRef]
- Rux, G.; Gelewsky, R.; Schlüter, O.; Herppich, W.B. High Hydrostatic Pressure Treatment Effects on Selected Tissue Properties of Fresh Horticultural Products. Innov. Food Sci. Emerg. Technol. 2020, 61, 102326. [Google Scholar] [CrossRef]
- Ramos-Parra, P.A.; García-Salinas, C.; Rodríguez-López, C.E.; García, N.; García-Rivas, G.; Hernández-Brenes, C.; Díaz de la Garza, R.I. High Hydrostatic Pressure Treatments Trigger de Novo Carotenoid Biosynthesis in Papaya Fruit (Carica papaya Cv. Maradol). Food Chem. 2019, 277, 362–372. [Google Scholar] [CrossRef]
- Hu, X.; Ma, T.; Ao, L.; Kang, H.; Hu, X.; Song, Y.; Liao, X. Effect of High Hydrostatic Pressure Processing on Textural Properties and Microstructural Characterization of Fresh-cut Pumpkin (Cucurbita pepo). J. Food Process Eng. 2020, 43, e13379. [Google Scholar] [CrossRef]
- Hu, K.; Peng, D.; Wang, L.; Liu, H.; Xie, B.; Sun, Z. Effect of Mild High Hydrostatic Pressure Treatments on Physiological and Physicochemical Characteristics and Carotenoid Biosynthesis in Postharvest Mango. Postharvest Biol. Technol. 2021, 172, 111381. [Google Scholar] [CrossRef]
- Kundukulangara Pulissery, S.; Kallahalli Boregowda, S.; Suseela, S.; Jaganath, B. A Comparative Study on the Textural and Nutritional Profile of High Pressure and Minimally Processed Pineapple. J. Food Sci. Technol. 2021, 58, 3734–3742. [Google Scholar] [CrossRef]
- Paciulli, M.; Rinaldi, M.; Rodolfi, M.; Ganino, T.; Morbarigazzi, M.; Chiavaro, E. Effects of High Hydrostatic Pressure on Physico-Chemical and Structural Properties of Two Pumpkin Species. Food Chem. 2019, 274, 281–290. [Google Scholar] [CrossRef]
- Paciulli, M.; Medina Meza, I.G.; Rinaldi, M.; Ganino, T.; Pugliese, A.; Rodolfi, M.; Barbanti, D.; Morbarigazzi, M.; Chiavaro, E. Improved Physicochemical and Structural Properties of Blueberries by High Hydrostatic Pressure Processing. Foods 2019, 8, 272. [Google Scholar] [CrossRef]
- Paciulli, M.; Ganino, T.; Meza, I.G.M.; Rinaldi, M.; Rodolfi, M.; Morbarigazzi, M.; Chiavaro, E. High Pressure and Thermal Processing on the Quality of Zucchini Slices. Eur. Food Res. Technol. 2021, 247, 475–484. [Google Scholar] [CrossRef]
- Tappi, S.; Ramazzina, I.; Rizzi, F.; Sacchetti, G.; Ragni, L.; Rocculi, P. Effect of Plasma Exposure Time on the Polyphenolic Profile and Antioxidant Activity of Fresh-Cut Apples. Appl. Sci. 2018, 8, 1939. [Google Scholar] [CrossRef]
- Tappi, S.; Ragni, L.; Tylewicz, U.; Romani, S.; Ramazzina, I.; Rocculi, P. Browning Response of Fresh-Cut Apples of Different Cultivars to Cold Gas Plasma Treatment. Innov. Food Sci. Emerg. Technol. 2019, 53, 56–62. [Google Scholar] [CrossRef]
- Zhou, D.; Li, T.; Cong, K.; Suo, A.; Wu, C. Influence of Cold Plasma on Quality Attributes and Aroma Compounds in Fresh-Cut Cantaloupe during Low Temperature Storage. LWT 2022, 154, 112893. [Google Scholar] [CrossRef]
- Ahmadnia, M.; Sadeghi, M.; Abbaszadeh, R.; Ghomi Marzdashti, H.R. Decontamination of Whole Strawberry via Dielectric Barrier Discharge Cold Plasma and Effects on Quality Attributes. J. Food Process. Preserv. 2021, 45, e15019. [Google Scholar] [CrossRef]
- Rana, S.; Mehta, D.; Bansal, V.; Shivhare, U.S.; Yadav, S.K. Atmospheric Cold Plasma (ACP) Treatment Improved in-Package Shelf-Life of Strawberry Fruit. J. Food Sci. Technol. 2020, 57, 102–112. [Google Scholar] [CrossRef]
- Li, X.; Li, M.; Ji, N.; Jin, P.; Zhang, J.; Zheng, Y.; Zhang, X.; Li, F. Cold Plasma Treatment Induces Phenolic Accumulation and Enhances Antioxidant Activity in Fresh-Cut Pitaya (Hylocereus Undatus) Fruit. LWT 2019, 115, 108447. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, J.; Zhang, Y.; Hu, H.; Luo, S.; Zhang, L.; Zhou, H.; Li, P. Effects of In-package Atmospheric Cold Plasma Treatment on the Qualitative, Metabolic and Microbial Stability of Fresh-cut Pears. J. Sci. Food Agric. 2021, 101, 4473–4480. [Google Scholar] [CrossRef]
- Giannoglou, M.; Stergiou, P.; Dimitrakellis, P.; Gogolides, E.; Stoforos, N.G.; Katsaros, G. Effect of Cold Atmospheric Plasma Processing on Quality and Shelf-Life of Ready-to-Eat Rocket Leafy Salad. Innov. Food Sci. Emerg. Technol. 2020, 66, 102502. [Google Scholar] [CrossRef]
- Hu, X.; Sun, H.; Yang, X.; Cui, D.; Wang, Y.; Zhuang, J.; Wang, X.; Ma, R.; Jiao, Z. Potential Use of Atmospheric Cold Plasma for Postharvest Preservation of Blueberries. Postharvest Biol. Technol. 2021, 179, 111564. [Google Scholar] [CrossRef]
- Gavahian, M.; Sheu, F.; Tsai, M.; Chu, Y. The Effects of Dielectric Barrier Discharge Plasma Gas and Plasma-activated Water on Texture, Color, and Bacterial Characteristics of Shiitake Mushroom. J. Food Process. Preserv. 2020, 44, e14316. [Google Scholar] [CrossRef]
- Liu, C.; Chen, C.; Jiang, A.; Sun, X.; Guan, Q.; Hu, W. Effects of Plasma-Activated Water on Microbial Growth and Storage Quality of Fresh-Cut Apple. Innov. Food Sci. Emerg. Technol. 2020, 59, 102256. [Google Scholar] [CrossRef]
- Perinban, S.; Orsat, V.; Raghavan, V. Influence of Plasma Activated Water Treatment on Enzyme Activity and Quality of Fresh-Cut Apples. Food Chem. 2022, 393, 133421. [Google Scholar] [CrossRef] [PubMed]
- Vanga, S.K.; Wang, J.; Jayaram, S.; Raghavan, V. Effects of Pulsed Electric Fields and Ultrasound Processing on Proteins and Enzymes: A Review. Processes 2021, 9, 722. [Google Scholar] [CrossRef]
- González-Casado, S.; Martín-Belloso, O.; Elez-Martínez, P.; Soliva-Fortuny, R. Enhancing the Carotenoid Content of Tomato Fruit with Pulsed Electric Field Treatments: Effects on Respiratory Activity and Quality Attributes. Postharvest Biol. Technol. 2018, 137, 113–118. [Google Scholar] [CrossRef]
- Sotelo, K.A.G.; Hamid, N.; Oey, I.; Pook, C.; Gutierrez-Maddox, N.; Ma, Q.; Ying Leong, S.; Lu, J. Red Cherries (Prunus avium Var. Stella) Processed by Pulsed Electric Field—Physical, Chemical and Microbiological Analyses. Food Chem. 2018, 240, 926–934. [Google Scholar] [CrossRef]
- Ribas-Agustí, A.; Martín-Belloso, O.; Soliva-Fortuny, R.; Elez-Martínez, P. Enhancing Hydroxycinnamic Acids and Flavan-3-Ol Contents by Pulsed Electric Fields without Affecting Quality Attributes of Apple. Food Res. Int. 2019, 121, 433–440. [Google Scholar] [CrossRef]
- Pokhrel, P.R.; Boulet, C.; Yildiz, S.; Sablani, S.; Tang, J.; Barbosa-Cánovas, G.V. Effect of High Hydrostatic Pressure on Microbial Inactivation and Quality Changes in Carrot-Orange Juice Blends at Varying PH. LWT 2022, 159, 113219. [Google Scholar] [CrossRef]
- Bagheri, H.; Abbaszadeh, S. Effect of Cold Plasma on Quality Retention of Fresh-Cut Produce. J. Food Qual. 2020, 8866369. [Google Scholar] [CrossRef]
- Li, M.; Li, X.; Han, C.; Ji, N.; Jin, P.; Zheng, Y. Physiological and Metabolomic Analysis of Cold Plasma Treated Fresh-Cut Strawberries. J. Agric. Food Chem. 2019, 67, 4043–4053. [Google Scholar] [CrossRef]
- Ziuzina, D.; Misra, N.N.; Han, L.; Cullen, P.J.; Moiseev, T.; Mosnier, J.P.; Keener, K.; Gaston, E.; Vilaró, I.; Bourke, P. Investigation of a Large Gap Cold Plasma Reactor for Continuous In-Package Decontamination of Fresh Strawberries and Spinach. Innov. Food Sci. Emerg. Technol. 2020, 59, 102229. [Google Scholar] [CrossRef]
- Hozák, P.; Jirešová, J.; Khun, J.; Scholtz, V.; Julák, J. Shelf Life Prolongation of Fresh Strawberries by Nonthermal Plasma Treatment. J. Food Process. Preserv. 2022, 46, e16150. [Google Scholar] [CrossRef]
- Puligundla, P.; Lee, T.; Mok, C. Effect of Intermittent Corona Discharge Plasma Treatment for Improving Microbial Quality and Shelf Life of Kumquat (Citrus Japonica) Fruits. LWT 2018, 91, 8–13. [Google Scholar] [CrossRef]
- Guo, J.; Qin, D.; Li, W.; Wu, F.; Li, L.; Liu, X. Inactivation of Penicillium Italicum on Kumquat via Plasma-Activated Water and Its Effects on Quality Attributes. Int. J. Food Microbiol. 2021, 343, 109090. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.; Karwe, M.V. Inactivation of Enterobacter Aerogenes on the Surfaces of Fresh-Cut Purple Lettuce, Kale, and Baby Spinach Leaves Using Plasma Activated Mist (PAM). Innov. Food Sci. Emerg. Technol. 2021, 74, 102868. [Google Scholar] [CrossRef]
- Laurita, R.; Gozzi, G.; Tappi, S.; Capelli, F.; Bisag, A.; Laghi, G.; Gherardi, M.; Cellini, B.; Abouelenein, D.; Vittori, S.; et al. Effect of Plasma Activated Water (PAW) on Rocket Leaves Decontamination and Nutritional Value. Innov. Food Sci. Emerg. Technol. 2021, 73, 102805. [Google Scholar] [CrossRef]
- Silvetti, T.; Pedroni, M.; Brasca, M.; Vassallo, E.; Cocetta, G.; Ferrante, A.; De Noni, I.; Piazza, L.; Morandi, S. Assessment of Possible Application of an Atmospheric Pressure Plasma Jet for Shelf Life Extension of Fresh-Cut Salad. Foods 2021, 10, 513. [Google Scholar] [CrossRef]
- Sudarsan, A.; Keener, K. Inactivation of Spoilage Organisms on Baby Spinach Leaves Using High Voltage Atmospheric Cold Plasma (HVACP) and Assessment of Quality. Innov. Food Sci. Emerg. Technol. 2022, 79, 103023. [Google Scholar] [CrossRef]
- Dong, X.Y.; Yang, Y.L. A Novel Approach to Enhance Blueberry Quality During Storage Using Cold Plasma at Atmospheric Air Pressure. Food Bioprocess Technol. 2019, 12, 1409–1421. [Google Scholar] [CrossRef]
- Ji, Y.; Hu, W.; Liao, J.; Jiang, A.; Xiu, Z.; Gaowa, S.; Guan, Y.; Yang, X.; Feng, K.; Liu, C. Effect of Atmospheric Cold Plasma Treatment on Antioxidant Activities and Reactive Oxygen Species Production in Postharvest Blueberries during Storage. J. Sci. Food Agric. 2020, 100, 5586–5595. [Google Scholar] [CrossRef]
- Pathak, N.; Grossi Bovi, G.; Limnaios, A.; Fröhling, A.; Brincat, J.; Taoukis, P.; Valdramidis, V.P.; Schlüter, O. Impact of Cold Atmospheric Pressure Plasma Processing on Storage of Blueberries. J. Food Process. Preserv. 2020, 44, e14581. [Google Scholar] [CrossRef]
- Segura-Ponce, L.A.; Reyes, J.E.; Troncoso-Contreras, G.; Valenzuela-Tapia, G. Effect of Low-Pressure Cold Plasma (LPCP) on the Wettability and the Inactivation of Escherichia Coli and Listeria Innocua on Fresh-Cut Apple (Granny Smith) Skin. Food Bioprocess Technol. 2018, 11, 1075–1086. [Google Scholar] [CrossRef]
- Zhou, R.; Zhou, R.; Mai-Prochnow, A.; Zhang, X.; Xian, Y.; Cullen, P.J.; Ostrikov, K. (Ken) Surface Plasma Discharges for the Preservation of Fresh-Cut Apples: Microbial Inactivation and Quality Attributes. J. Phys. Appl. Phys. 2020, 53, 174003. [Google Scholar] [CrossRef]
- Chen, C.; Liu, C.; Jiang, A.; Guan, Q.; Sun, X.; Liu, S.; Hao, K.; Hu, W. The Effects of Cold Plasma-Activated Water Treatment on the Microbial Growth and Antioxidant Properties of Fresh-Cut Pears. Food Bioprocess Technol. 2019, 12, 1842–1851. [Google Scholar] [CrossRef]
- Zhao, Z.; Wang, X.; Ma, T. Properties of Plasma-Activated Water with Different Activation Time and Its Effects on the Quality of Button Mushrooms (Agaricus Bisporus). LWT 2021, 147, 111633. [Google Scholar] [CrossRef]
- Lee, T.; Puligundla, P.; Mok, C. Intermittent Corona Discharge Plasma Jet for Improving Tomato Quality. J. Food Eng. 2018, 223, 168–174. [Google Scholar] [CrossRef]
- Zhao, Y.; Chen, R.; Liu, D.; Wang, W.; Niu, J.; Xia, Y.; Qi, Z.; Zhao, Z.; Song, Y. Effect of Nonthermal Plasma-Activated Water on Quality and Antioxidant Activity of Fresh-Cut Kiwifruit. IEEE Trans. Plasma Sci. 2019, 47, 4811–4817. [Google Scholar] [CrossRef]
- Limnaios, A.; Pathak, N.; Grossi, G.; Fr, A.; Valdramidis, P.; Taoukis, P.S.; Schlüter, O. Effect of Cold Atmospheric Pressure Plasma Processing on Quality and Shelf Life of Red Currants. LWT 2021, 151, 112213. [Google Scholar] [CrossRef]
- Siddiqui, W.; Chakraborty, I.; Ayala-Zavala, J.F.; Dhua, R.S. Advances in Minimal Processing of Fruits and Vegetables: A Review. J. Sci. Ind. Res. 2011, 70, 12. [Google Scholar]
- Escobedo-Avellaneda, Z.; García-García, R.; Valdez-Fragoso, A.; Mújica-Paz, H.; Welti-Chanes, J. Fruit Preservation and Design of Functional Fruit Products by Vacuum Impregnation. In Fruit Preservation; Rosenthal, A., Deliza, R., Welti-Chanes, J., Barbosa-Cánovas, G.V., Eds.; Food Engineering Series; Springer New York: New York, NY, USA, 2018; pp. 335–349. ISBN 978-1-4939-3309-9. [Google Scholar]
- Chinnaswamy, S.; Rudra, S.G.; Sharma, R.R. Texturizers for Fresh-Cut Fruit and Vegetable Products. In Fresh-Cut Fruits and Vegetables; Elsevier: Amsterdam, The Netherlands, 2020; pp. 121–149. ISBN 978-0-12-816184-5. [Google Scholar]
- Joshi, A.; Prajapati, U.; Sethi, S.; Arora, B.; Sharma, R.R. Fortification in Fresh and Fresh-Cut Horticultural Products. In Fresh-Cut Fruits and Vegetables; Elsevier: Amsterdam, The Netherlands, 2020; pp. 183–204. ISBN 978-0-12-816184-5. [Google Scholar]
- Martín-Diana, A.B.; Rico, D.; Frías, J.M.; Barat, J.M.; Henehan, G.T.M.; Barry-Ryan, C. Calcium for Extending the Shelf Life of Fresh Whole and Minimally Processed Fruits and Vegetables: A Review. Trends Food Sci. Technol. 2007, 18, 210–218. [Google Scholar] [CrossRef]
- Soliva-Fortuny, R.C.; Martín-Belloso, O. New Advances in Extending the Shelf-Life of Fresh-Cut Fruits: A Review. Trends Food Sci. Technol. 2003, 14, 341–353. [Google Scholar] [CrossRef]
- Alzamora, S.M.; Salvatori, D.; Tapia, M.S.; López-Malo, A.; Welti-Chanes, J.; Fito, P. Novel Functional Foods from Vegetable Matrices Impregnated with Biologically Active Compounds. J. Food Eng. 2005, 67, 205–214. [Google Scholar] [CrossRef]
- Mu, B.; Xue, J.; Zhang, S.; Li, Z. Effects of the Use of Different Temperature and Calcium Chloride Treatments during Storage on the Quality of Fresh-Cut “Xuebai” Cauliflowers. Foods 2022, 11, 442. [Google Scholar] [CrossRef] [PubMed]
- Mola, S.; Uthairatanakij, A.; Srilaong, V.; Aiamla-or, S.; Jitareerat, P. Impacts of Sodium Chlorite Combined with Calcium Chloride, and Calcium Ascorbate on Microbial Population, Browning, and Quality of Fresh-Cut Rose Apple. Agric. Nat. Resour. 2016, 50, 331–337. [Google Scholar] [CrossRef]
- Giacalone, G.; Chiabrando, V. Effect of Different Treatments with Calcium Salts on Sensory Quality of Fresh-Cut Apple. J. Food Nutr. Res. 2013, 52, 79–86. [Google Scholar]
- Albertini, S.; Lai Reyes, A.E.; Trigo, J.M.; Sarriés, G.A.; Spoto, M.H.F. Effects of Chemical Treatments on Fresh-Cut Papaya. Food Chem. 2016, 190, 1182–1189. [Google Scholar] [CrossRef]
- Yan, R.; Han, C.; Fu, M.; Jiao, W.; Wang, W. Inhibitory Effects of CaCl2 and Pectin Methylesterase on Fruit Softening of Raspberry during Cold Storage. Horticulturae 2021, 8, 1. [Google Scholar] [CrossRef]
- Hamdan, N.; Lee, C.H.; Wong, S.L.; Fauzi, C.E.N.C.A.; Zamri, N.M.A.; Lee, T.H. Prevention of Enzymatic Browning by Natural Extracts and Genome-Editing: A Review on Recent Progress. Molecules 2022, 27, 1101. [Google Scholar] [CrossRef]
- Martínez-Hernández, G.B.; Castillejo, N.; Artés-Hernández, F. Effect of Fresh–Cut Apples Fortification with Lycopene Microspheres, Revalorized from Tomato by-Products, during Shelf Life. Postharvest Biol. Technol. 2019, 156, 110925. [Google Scholar] [CrossRef]
- Supapvanich, S.; Mitsang, P.; Srinorkham, P. Effects of ‘Queen’ and ‘Smooth Cayenne’ Pineapple Fruit Core Extracts on Browning Inhibition of Fresh-Cut Wax Apple Fruit during Storage. Int. Food Res. J. 2017, 24, 559–564. [Google Scholar]
- Turrini, F.; Malaspina, P.; Giordani, P.; Catena, S.; Zunin, P.; Boggia, R. Traditional Decoction and PUAE Aqueous Extracts of Pomegranate Peels as Potential Low-Cost Anti-Tyrosinase Ingredients. Appl. Sci. 2020, 10, 2795. [Google Scholar] [CrossRef]
- Jirasuteeruk, C.; Theerakulkait, C. Ultrasound-Assisted Extraction of Phenolic Compounds from Mango (Mangifera Indica Cv. Chok Anan) Peel and Its Inhibitory Effect on Enzymatic Browning of Potato Puree. Food Technol. Biotechnol. 2019, 57, 350–357. [Google Scholar] [CrossRef] [PubMed]
- Supapvanich, S.; Mitrsang, P.; Srinorkham, P.; Boonyaritthongchai, P.; Wongs-Aree, C. Effects of Fresh Aloe Vera Gel Coating on Browning Alleviation of Fresh Cut Wax Apple (Syzygium Samarangenese) Fruit Cv. Taaptimjaan. J. Food Sci. Technol. 2016, 53, 2844–2850. [Google Scholar] [CrossRef] [PubMed]
- Wessels, B.; Damm, S.; Kunz, B.; Schulze-Kaysers, N. Effect of Selected Plant Extracts on the Inhibition of Enzymatic Browning in Fresh-Cut Apple. J. Appl. Bot. Food Qual. 2014, 87, 16–23. [Google Scholar] [CrossRef]
- Supapvanich, S.; Yimpong, A.; Srisuwanwichan, J. Browning Inhibition on Fresh-Cut Apple by the Immersion of Liquid Endosperm from Mature Coconuts. J. Food Sci. Technol. 2020, 57, 4424–4431. [Google Scholar] [CrossRef]
- Gras, M.; Vidal-Brotóns, N.; Betoret, A.; Chiralt; Fito, P. The Response of Some Vegetables to Vacuum Impregnation. Innov. Food Sci. Emerg. Technol. 2002, 3, 263–269. [Google Scholar] [CrossRef]
- Fito, P. Modelling of Vacuum Osmotic Dehydration of Food. J. Food Eng. 1994, 22, 313–328. [Google Scholar] [CrossRef]
- Andrés, A.; Salvatori, D.; Albors, A.; Chiralt, A.; Fito, P. Vacuum Impregnation Viability of Some Fruits and Vegetables. In Osmotic Dehydration & Vacuum Impregnation; Fito, P., Chiralt, A., Barat, J.M., Spiess, W.E.L., Behsnilian, D., Eds.; CRC Press: Boca Raton, FL, USA, 2019; pp. 53–60. ISBN 978-0-429-13221-6. [Google Scholar]
- Kang, J.-W.; Kang, D.-H. Enhanced Antimicrobial Effect of Organic Acid Washing against Foodborne Pathogens on Broccoli by Vacuum Impregnation. Int. J. Food Microbiol. 2016, 217, 85–93. [Google Scholar] [CrossRef]
- Yılmaz, F.M.; Ersus Bilek, S. Ultrasound-Assisted Vacuum Impregnation on the Fortification of Fresh-Cut Apple with Calcium and Black Carrot Phenolics. Ultrason. Sonochem. 2018, 48, 509–516. [Google Scholar] [CrossRef]
- Santana Moreira, M.; de Almeida Paula, D.; Maurício Furtado Martins, E.; Nascif Rufino Vieira, É.; Mota Ramos, A.; Stringheta, P.C. Vacuum Impregnation of β-Carotene and Lutein in Minimally Processed Fruit Salad. J. Food Process. Preserv. 2018, 42, e13545. [Google Scholar] [CrossRef]
- Lech, K.; Michalska, A.; Wojdyło, A.; Nowicka, P.; Figiel, A. The Influence of Physical Properties of Selected Plant Materials on the Process of Osmotic Dehydration. LWT 2018, 91, 588–594. [Google Scholar] [CrossRef]
- Peng, J.; Bi, J.; Yi, J.; Allaf, K.; Besombes, C.; Jin, X.; Wu, X.; Lyu, J.; Asghar Ali, M.N.H. Apple Juice Concentrate Impregnation Enhances Nutritional and Textural Attributes of the Instant Controlled Pressure Drop (DIC)-dried Carrot Chips. J. Sci. Food Agric. 2019, 99, 6248–6257. [Google Scholar] [CrossRef]
- Derossi, A.; Ricci, I.; Fiore, A.G.; Severini, C. Apple Sclices Enriched With Aloe Vera By Vacuum Impregnation. Ital. J. Food Sci. 2017, 30, 256–257. [Google Scholar] [CrossRef]
- González-Pérez, J.E.; Jiménez-González, O.; Ramírez-Corona, N.; Guerrero-Beltrán, J.A.; López-Malo, A. Vacuum Impregnation on Apples with Grape Juice Concentrate: Effects of Pressure, Processing Time, and Juice Concentration. Innov. Food Sci. Emerg. Technol. 2022, 77, 102981. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, P.; Chen, F.; Lai, S.; Yu, H.; Yang, H. Effects of Calcium and Pectin Methylesterase on Quality Attributes and Pectin Morphology of Jujube Fruit under Vacuum Impregnation during Storage. Food Chem. 2019, 289, 40–48. [Google Scholar] [CrossRef]
- Yang, H.; Wu, Q.; Ng, L.Y.; Wang, S. Effects of Vacuum Impregnation with Calcium Lactate and Pectin Methylesterase on Quality Attributes and Chelate-Soluble Pectin Morphology of Fresh-Cut Papayas. Food Bioprocess Technol. 2017, 10, 901–913. [Google Scholar] [CrossRef]
- Pandey, V.K.; Islam, R.U.; Shams, R.; Dar, A.H. A Comprehensive Review on the Application of Essential Oils as Bioactive Compounds in Nano-Emulsion Based Edible Coatings of Fruits and Vegetables. Appl. Food Res. 2022, 2, 100042. [Google Scholar] [CrossRef]
- M. Rangaraj, V.; Rambabu, K.; Banat, F.; Mittal, V. Natural Antioxidants-Based Edible Active Food Packaging: An Overview of Current Advancements. Food Biosci. 2021, 43, 101251. [Google Scholar] [CrossRef]
- Zam, W. Effect of Alginate and Chitosan Edible Coating Enriched with Olive Leaves Extract on the Shelf Life of Sweet Cherries (Prunus avium L.). J. Food Qual. 2019, 2019, 8192964. [Google Scholar] [CrossRef]
- Robles-Sánchez, R.M.; Rojas-Graü, M.A.; Odriozola-Serrano, I.; González-Aguilar, G.; Martin-Belloso, O. Influence of Alginate-Based Edible Coating as Carrier of Antibrowning Agents on Bioactive Compounds and Antioxidant Activity in Fresh-Cut Kent Mangoes. LWT - Food Sci. Technol. 2013, 50, 240–246. [Google Scholar] [CrossRef]
- Özdemir, K.S.; Gökmen, V. Effect of Chitosan-Ascorbic Acid Coatings on the Refrigerated Storage Stability of Fresh-Cut Apples. Coatings 2019, 9, 503. [Google Scholar] [CrossRef]
- Liu, J.; Liu, S.; Zhang, X.; Kan, J.; Jin, C. Effect of Gallic Acid Grafted Chitosan Film Packaging on the Postharvest Quality of White Button Mushroom (Agaricus Bisporus). Postharvest Biol. Technol. 2019, 147, 39–47. [Google Scholar] [CrossRef]
- Carvalho, R.L.; Cabral, M.F.; Germano, T.A.; de Carvalho, W.M.; Brasil, I.M.; Gallão, M.I.; Moura, C.F.H.; Lopes, M.M.A.; de Miranda, M.R.A. Chitosan Coating with Trans-Cinnamaldehyde Improves Structural Integrity and Antioxidant Metabolism of Fresh-Cut Melon. Postharvest Biol. Technol. 2016, 113, 29–39. [Google Scholar] [CrossRef]
- Jagannath, J.H.; Nanjappa, C.; Gupta, D.D.; Bawa, A.S. Studies on the Stability of an Edible Film and Its Use for the Preservation of Carrot (Daucus Carota). Int. J. Food Sci. Technol. 2006, 41, 498–506. [Google Scholar] [CrossRef]
- Imeneo, V.; Piscopo, A.; Martín-Belloso, O.; Soliva-Fortuny, R. Efficacy of Pectin-Based Coating Added with a Lemon Byproduct Extract on Quality Preservation of Fresh-Cut Carrots. Foods 2022, 11, 1314. [Google Scholar] [CrossRef] [PubMed]
- Perdones, A.; Sánchez-González, L.; Chiralt, A.; Vargas, M. Effect of Chitosan–Lemon Essential Oil Coatings on Storage-Keeping Quality of Strawberry. Postharvest Biol. Technol. 2012, 70, 32–41. [Google Scholar] [CrossRef]
- Ghafoor, K.; Al-Juhaimi, F.Y.; Babiker, E.E.; Mohamed Ahmed, I.A.; Shahzad, S.A.; Alsawmahi, O.N. Quality Attributes of Refrigerated Barhi Dates Coated with Edible Chitosan Containing Natural Functional Ingredients. Foods 2022, 11, 1584. [Google Scholar] [CrossRef]
- Vieira, T.M.; Alves, V.D.; Moldão Martins, M. Application of an Eco-Friendly Antifungal Active Package to Extend the Shelf Life of Fresh Red Raspberry (Rubus idaeus L. Cv. ‘Kweli’). Foods 2022, 11, 1805. [Google Scholar] [CrossRef]
- Jurburg, S.D.; Eisenhauer, N.; Buscot, F.; Chatzinotas, A.; Chaudhari, N.M.; Heintz-Buschart, A.; Kallies, R.; Küsel, K.; Litchman, E.; Macdonald, C.A.; et al. Potential of Microbiome-Based Solutions for Agrifood Systems. Nat. Food 2022, 3, 557–560. [Google Scholar] [CrossRef]
- Wassermann, B.; Müller, H.; Berg, G. An Apple a Day: Which Bacteria Do We Eat With Organic and Conventional Apples? Front. Microbiol. 2019, 10, 1629. [Google Scholar] [CrossRef]
- Kłapeć, T.; Wójcik-Fatla, A.; Farian, E.; Kowalczyk, K.; Cholewa, G.; Cholewa, A.; Dutkiewicz, J. Mycobiota of Berry Fruits—Levels of Filamentous Fungi and Mycotoxins, Composition of Fungi, and Analysis of Potential Health Risk for Consumers. Ann. Agric. Environ. Med. 2022, 29, 28–37. [Google Scholar] [CrossRef]
- Vermote, L.; Verce, M.; Mozzi, F.; De Vuyst, L.; Weckx, S. Microbiomes Associated With the Surfaces of Northern Argentinian Fruits Show a Wide Species Diversity. Front. Microbiol. 2022, 13, 872281. [Google Scholar] [CrossRef]
- Sellitto, V.M.; Zara, S.; Fracchetti, F.; Capozzi, V.; Nardi, T. Microbial Biocontrol as an Alternative to Synthetic Fungicides: Boundaries between Pre- and Postharvest Applications on Vegetables and Fruits. Fermentation 2021, 7, 60. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, Q.; Chen, S.; Zhang, Z.; Song, J.; Long, Z.; Yu, Y.; Fang, H. Enterobacteriaceae Predominate in the Endophytic Microbiome and Contribute to the Resistome of Strawberry. Sci. Total Environ. 2020, 727, 138708. [Google Scholar] [CrossRef] [PubMed]
- De Simone, N.; Pace, B.; Grieco, F.; Chimienti, M.; Tyibilika, V.; Santoro, V.; Capozzi, V.; Colelli, G.; Spano, G.; Russo, P. Botrytis Cinerea and Table Grapes: A Review of the Main Physical, Chemical, and Bio-Based Control Treatments in Post-Harvest. Foods 2020, 9, 1138. [Google Scholar] [CrossRef] [PubMed]
- De Simone, N.; Capozzi, V.; Amodio, M.L.; Colelli, G.; Spano, G.; Russo, P. Microbial-Based Biocontrol Solutions for Fruits and Vegetables: Recent Insight, Patents, and Innovative Trends. Recent Pat. Food Nutr. Agric. 2021, 12, 3–18. [Google Scholar] [CrossRef]
- Capozzi, V.; Fragasso, M.; Bimbo, F. Microbial Resources, Fermentation and Reduction of Negative Externalities in Food Systems: Patterns toward Sustainability and Resilience. Fermentation 2021, 7, 54. [Google Scholar] [CrossRef]
- Díaz, M.A.; Pereyra, M.M.; Picón-Montenegro, E.; Meinhardt, F.; Dib, J.R. Killer Yeasts for the Biological Control of Postharvest Fungal Crop Diseases. Microorganisms 2020, 8, 1680. [Google Scholar] [CrossRef]
- Hernandez-Montiel, L.G.; Droby, S.; Preciado-Rangel, P.; Rivas-García, T.; González-Estrada, R.R.; Gutiérrez-Martínez, P.; Ávila-Quezada, G.D. A Sustainable Alternative for Postharvest Disease Management and Phytopathogens Biocontrol in Fruit: Antagonistic Yeasts. Plants 2021, 10, 2641. [Google Scholar] [CrossRef]
- Carmona-Hernandez, S.; Reyes-Pérez, J.; Chiquito-Contreras, R.; Rincon-Enriquez, G.; Cerdan-Cabrera, C.; Hernandez-Montiel, L. Biocontrol of Postharvest Fruit Fungal Diseases by Bacterial Antagonists: A Review. Agronomy 2019, 9, 121. [Google Scholar] [CrossRef]
- Aiello, D.; Restuccia, C.; Stefani, E.; Vitale, A.; Cirvilleri, G. Postharvest Biocontrol Ability of Pseudomonas Synxantha against Monilinia Fructicola and Monilinia Fructigena on Stone Fruit. Postharvest Biol. Technol. 2019, 149, 83–89. [Google Scholar] [CrossRef]
- Bu, S.; Munir, S.; He, P.; Li, Y.; Wu, Y.; Li, X.; Kong, B.; He, P.; He, Y. Bacillus Subtilis L1-21 as a Biocontrol Agent for Postharvest Gray Mold of Tomato Caused by Botrytis Cinerea. Biol. Control 2021, 157, 104568. [Google Scholar] [CrossRef]
- Wang, X.; Zhou, X.; Cai, Z.; Guo, L.; Chen, X.; Chen, X.; Liu, J.; Feng, M.; Qiu, Y.; Zhang, Y.; et al. A Biocontrol Strain of Pseudomonas Aeruginosa CQ-40 Promote Growth and Control Botrytis Cinerea in Tomato. Pathogens 2020, 10, 22. [Google Scholar] [CrossRef]
- Raman, J.; Kim, J.-S.; Choi, K.R.; Eun, H.; Yang, D.; Ko, Y.-J.; Kim, S.-J. Application of Lactic Acid Bacteria (LAB) in Sustainable Agriculture: Advantages and Limitations. Int. J. Mol. Sci. 2022, 23, 7784. [Google Scholar] [CrossRef] [PubMed]
- Linares-Morales, J.R.; Gutiérrez-Méndez, N.; Rivera-Chavira, B.E.; Pérez-Vega, S.B.; Nevárez-Moorillón, G.V. Biocontrol Processes in Fruits and Fresh Produce, the Use of Lactic Acid Bacteria as a Sustainable Option. Front. Sustain. Food Syst. 2018, 2, 50. [Google Scholar] [CrossRef]
- EFSA Panel on Biological Hazards (BIOHAZ); Koutsoumanis, K.; Allende, A.; Alvarez-Ordóñez, A.; Bolton, D.; Bover-Cid, S.; Chemaly, M.; Davies, R.; De Cesare, A.; Hilbert, F.; et al. Update of the List of QPS-recommended Biological Agents Intentionally Added to Food or Feed as Notified to EFSA 13: Suitability of Taxonomic Units Notified to EFSA until September 2020. EFSA J. 2021, 19. [Google Scholar] [CrossRef]
- Russo, P.; Spano, G.; Capozzi, V. Safety Evaluation of Starter Cultures. In Starter Cultures in Food Production; Speranza, B., Bevilacqua, A., Corbo, M.R., Sinigaglia, M., Eds.; John Wiley & Sons, Ltd: Chichester, UK, 2017; pp. 101–128. ISBN 978-1-118-93379-4. [Google Scholar]
- Mani-López, E.; Arrioja-Bretón, D.; López-Malo, A. The Impacts of Antimicrobial and Antifungal Activity of Cell-free Supernatants from Lactic Acid Bacteria in Vitro and Foods. Compr. Rev. Food Sci. Food Saf. 2022, 21, 604–641. [Google Scholar] [CrossRef]
- Agriopoulou, S.; Stamatelopoulou, E.; Sachadyn-Król, M.; Varzakas, T. Lactic Acid Bacteria as Antibacterial Agents to Extend the Shelf Life of Fresh and Minimally Processed Fruits and Vegetables: Quality and Safety Aspects. Microorganisms 2020, 8, 952. [Google Scholar] [CrossRef]
- Tenea, G.N.; Olmedo, D.; Ortega, C. Peptide-Based Formulation from Lactic Acid Bacteria Impairs the Pathogen Growth in Ananas Comosus (Pineapple). Coatings 2020, 10, 457. [Google Scholar] [CrossRef]
- Dong, A.; Malo, A.; Leong, M.; Ho, V.T.T.; Turner, M.S. Control of Listeria Monocytogenes on Ready-to-Eat Ham and Fresh Cut Iceberg Lettuce Using a Nisin Containing Lactococcus Lactis Fermentate. Food Control 2021, 119, 107420. [Google Scholar] [CrossRef]
- De Simone, N.; Capozzi, V.; de Chiara, M.L.V.; Amodio, M.L.; Brahimi, S.; Colelli, G.; Drider, D.; Spano, G.; Russo, P. Screening of Lactic Acid Bacteria for the Bio-Control of Botrytis Cinerea and the Potential of Lactiplantibacillus Plantarum for Eco-Friendly Preservation of Fresh-Cut Kiwifruit. Microorganisms 2021, 9, 773. [Google Scholar] [CrossRef]
- Yin, H.-B.; Chen, C.-H.; Colorado-Suarez, S.; Patel, J. Biocontrol of Listeria Monocytogenes and Salmonella Enterica on Fresh Strawberries with Lactic Acid Bacteria During Refrigerated Storage. Foodborne Pathog. Dis. 2022, 19, 324–331. [Google Scholar] [CrossRef]
- Yap, P.-C.; MatRahim, N.-A.; AbuBakar, S.; Lee, H.Y. Antilisterial Potential of Lactic Acid Bacteria in Eliminating Listeria Monocytogenes in Host and Ready-to-Eat Food Application. Microbiol. Res. 2021, 12, 234–257. [Google Scholar] [CrossRef]
- Luz, C.; D’Opazo, V.; Quiles, J.M.; Romano, R.; Mañes, J.; Meca, G. Biopreservation of Tomatoes Using Fermented Media by Lactic Acid Bacteria. LWT 2020, 130, 109618. [Google Scholar] [CrossRef]
- Ranjith, F.H.; Muhialdin, B.J.; Yusof, N.L.; Mohammed, N.K.; Miskandar, M.H.; Hussin, A.S.M. Effects of Lacto-Fermented Agricultural By-Products as a Natural Disinfectant against Post-Harvest Diseases of Mango (Mangifera indica L.). Plants 2021, 10, 285. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, M.; Chouhan, D.; Kamra, A.; Sharma, V. Sustainable Utilization of Potato Industry Waste for Antifungal Biopolymer Production by Lactobacillus Helveticus and Its Application on Pomegranates (Punica granatum L.). Circ. Econ. Sustain. 2021, 1, 1297–1312. [Google Scholar] [CrossRef]
- de Oliveira, K.Á.R.; Fernandes, K.F.D.; de Souza, E.L. Current Advances on the Development and Application of Probiotic-Loaded Edible Films and Coatings for the Bioprotection of Fresh and Minimally Processed Fruit and Vegetables. Foods 2021, 10, 2207. [Google Scholar] [CrossRef]
- Fernandes, K.F.D.; de Oliveira, K.Á.R.; de Souza, E.L. Application of Potentially Probiotic Fruit-Derived Lactic Acid Bacteria Loaded into Sodium Alginate Coatings to Control Anthracnose Development in Guava and Mango During Storage. Probiotics Antimicrob. Proteins 2021. [Google Scholar] [CrossRef]
- Álvarez, A.; Manjarres, J.J.; Ramírez, C.; Bolívar, G. Use of an Exopolysaccharide-Based Edible Coating and Lactic Acid Bacteria with Antifungal Activity to Preserve the Postharvest Quality of Cherry Tomato. LWT 2021, 151, 112225. [Google Scholar] [CrossRef]
- Rocchetti, M.T.; Russo, P.; Capozzi, V.; Drider, D.; Spano, G.; Fiocco, D. Bioprospecting Antimicrobials from Lactiplantibacillus Plantarum: Key Factors Underlying Its Probiotic Action. Int. J. Mol. Sci. 2021, 22, 12076. [Google Scholar] [CrossRef]
- Khalil, O.A.A.; Mounir, A.M.; Hassanien, R.A. Effect of Gamma Irradiated Lactobacillus Bacteria as an Edible Coating on Enhancing the Storage of Tomato under Cold Storage Conditions. J. Radiat. Res. Appl. Sci. 2020, 13, 318–330. [Google Scholar] [CrossRef]
- Pace, B.; Cefola, M.; Da Pelo, P.; Renna, F.; Attolico, G. Non-Destructive Evaluation of Quality and Ammonia Content in Whole and Fresh-Cut Lettuce by Computer Vision System. Food Res. Int. 2014, 64, 647–655. [Google Scholar] [CrossRef]
- Mohammadi, V.; Kheiralipour, K.; Ghasemi-Varnamkhasti, M. Detecting Maturity of Persimmon Fruit Based on Image Processing Technique. Sci. Hortic. 2015, 184, 123–128. [Google Scholar] [CrossRef]
- Kaur, H.; Sawhney, B.K.; Jawandha, S.K. Evaluation of Plum Fruit Maturity by Image Processing Techniques. J. Food Sci. Technol. 2018, 55, 3008–3015. [Google Scholar] [CrossRef]
- Cavallo, D.P.; Cefola, M.; Pace, B.; Logrieco, A.F.; Attolico, G. Non-Destructive and Contactless Quality Evaluation of Table Grapes by a Computer Vision System. Comput. Electron. Agric. 2019, 156, 558–564. [Google Scholar] [CrossRef]
- Fashi, M.; Naderloo, L.; Javadikia, H. The Relationship between the Appearance of Pomegranate Fruit and Color and Size of Arils Based on Image Processing. Postharvest Biol. Technol. 2019, 154, 52–57. [Google Scholar] [CrossRef]
- Ireri, D.; Belal, E.; Okinda, C.; Makange, N.; Ji, C. A Computer Vision System for Defect Discrimination and Grading in Tomatoes Using Machine Learning and Image Processing. Artif. Intell. Agric. 2019, 2, 28–37. [Google Scholar] [CrossRef]
- Fan, S.; Li, J.; Zhang, Y.; Tian, X.; Wang, Q.; He, X.; Zhang, C.; Huang, W. On Line Detection of Defective Apples Using Computer Vision System Combined with Deep Learning Methods. J. Food Eng. 2020, 286, 110102. [Google Scholar] [CrossRef]
- Cavallo, D.P.; Cefola, M.; Pace, B.; Logrieco, A.F.; Attolico, G. Contactless and Non-Destructive Chlorophyll Content Prediction by Random Forest Regression: A Case Study on Fresh-Cut Rocket Leaves. Comput. Electron. Agric. 2017, 140, 303–310. [Google Scholar] [CrossRef]
- Palumbo, M.; Pace, B.; Cefola, M.; Montesano, F.F.; Serio, F.; Colelli, G.; Attolico, G. Self-Configuring CVS to Discriminate Rocket Leaves According to Cultivation Practices and to Correctly Attribute Visual Quality Level. Agronomy 2021, 11, 1353. [Google Scholar] [CrossRef]
- Amodio, M.L.; Cabezas-Serrano, A.B.; Peri, G.; Colelli, G. Post-Cutting Quality Changes of Fresh-Cut Artichokes Treated with Different Anti-Browning Agents as Evaluated by Image Analysis. Postharvest Biol. Technol. 2011, 62, 213–220. [Google Scholar] [CrossRef]
- Pace, B.; Cefola, M.; Renna, F.; Attolico, G. Relationship between Visual Appearance and Browning as Evaluated by Image Analysis and Chemical Traits in Fresh-Cut Nectarines. Postharvest Biol. Technol. 2011, 61, 178–183. [Google Scholar] [CrossRef]
- Pace, B.; Cavallo, D.P.; Cefola, M.; Colella, R.; Attolico, G. Adaptive Self-Configuring Computer Vision System for Quality Evaluation of Fresh-Cut Radicchio. Innov. Food Sci. Emerg. Technol. 2015, 32, 200–207. [Google Scholar] [CrossRef]
- Subhashree, S.N.; Sunoj, S.; Xue, J.; Bora, G.C. Quantification of Browning in Apples Using Colour and Textural Features by Image Analysis. Food Qual. Saf. 2017, 1, 221–226. [Google Scholar] [CrossRef]
- Hongyang, T.; Daming, H.; Xingyi, H.; Aheto, J.H.; Yi, R.; Yu, W.; Ji, L.; Shuai, N.; Mengqi, X. Detection of Browning of Fresh-cut Potato Chips Based on Machine Vision and Electronic Nose. J. Food Process Eng. 2021, 44. [Google Scholar] [CrossRef]
- Cavallo, D.P.; Cefola, M.; Pace, B.; Logrieco, A.F.; Attolico, G. Non-Destructive Automatic Quality Evaluation of Fresh-Cut Iceberg Lettuce through Packaging Material. J. Food Eng. 2018, 223, 46–52. [Google Scholar] [CrossRef]
- Palumbo, M.; Pace, B.; Cefola, M.; Montesano, F.F.; Colelli, G.; Attolico, G. Non-Destructive and Contactless Estimation of Chlorophyll and Ammonia Contents in Packaged Fresh-Cut Rocket Leaves by a Computer Vision System. Postharvest Biol. Technol. 2022, 189, 111910. [Google Scholar] [CrossRef]
- Xia, Z.; Wu, D.; Nie, P.; He, Y. Non-Invasive Measurement of Soluble Solid Content and PH in Kyoho Grapes Using a Computer Vision Technique. Anal. Methods 2016, 8, 3242–3248. [Google Scholar] [CrossRef]
- Pace, B.; Cefola, M.; Renna, F.; Renna, M.; Serio, F.; Attolico, G. Multiple Regression Models and Computer Vision Systems to Predict Antioxidant Activity and Total Phenols in Pigmented Carrots. J. Food Eng. 2013, 117, 74–81. [Google Scholar] [CrossRef]
- Nadafzadeh, M.; Abdanan Mehdizadeh, S.; Soltanikazemi, M. Development of Computer Vision System to Predict Peroxidase and Polyphenol Oxidase Enzymes to Evaluate the Process of Banana Peel Browning Using Genetic Programming Modeling. Sci. Hortic. 2018, 231, 201–209. [Google Scholar] [CrossRef]
- Sabzi, S.; Javadikia, H.; Arribas, J.I. A Three-Variety Automatic and Non-Intrusive Computer Vision System for the Estimation of Orange Fruit PH Value. Measurement 2020, 152, 107298. [Google Scholar] [CrossRef]
- Palumbo, M.; Cozzolino, R.; Laurino, C.; Malorni, L.; Picariello, G.; Siano, F.; Stocchero, M.; Cefola, M.; Corvino, A.; Romaniello, R.; et al. Rapid and Non-Destructive Techniques for the Discrimination of Ripening Stages in Candonga Strawberries. Foods 2022, 11, 1534. [Google Scholar] [CrossRef]
- Basak, J.K.; Madhavi, B.G.K.; Paudel, B.; Kim, N.E.; Kim, H.T. Prediction of Total Soluble Solids and PH of Strawberry Fruits Using RGB, HSV and HSL Colour Spaces and Machine Learning Models. Foods 2022, 11, 2086. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Lv, S.; Feng, L.; Peng, P.; Hu, L.; Liu, Z.; Hati, S.; Bimal, C.; Mo, H. Smartphone-Based Image Analysis for Rapid Evaluation of Kiwifruit Quality during Cold Storage. Foods 2022, 11, 2113. [Google Scholar] [CrossRef] [PubMed]
- Farneti, B.; Alarcón, A.A.; Papasotiriou, F.G.; Samudrala, D.; Cristescu, S.M.; Costa, G.; Harren, F.J.M.; Woltering, E.J. Chilling-Induced Changes in Aroma Volatile Profiles in Tomato. Food Bioprocess Technol. 2015, 8, 1442–1454. [Google Scholar] [CrossRef]
- Zhu, D.; Ren, X.; Wei, L.; Cao, X.; Ge, Y.; Liu, H.; Li, J. Collaborative Analysis on Difference of Apple Fruits Flavour Using Electronic Nose and Electronic Tongue. Sci. Hortic. 2020, 260, 108879. [Google Scholar] [CrossRef]
- Barbosa-Pereira, L.; Rojo-Poveda, O.; Ferrocino, I.; Giordano, M.; Zeppa, G. Assessment of Volatile Fingerprint by HS-SPME/GC-QMS and E-Nose for the Classification of Cocoa Bean Shells Using Chemometrics. Food Res. Int. 2019, 123, 684–696. [Google Scholar] [CrossRef]
- Gaggiotti, S.; Mascini, M.; Pittia, P.; Della Pelle, F.; Compagnone, D. Headspace Volatile Evaluation of Carrot Samples—Comparison of GC/MS and AuNPs-HpDNA-Based E-Nose. Foods 2019, 8, 293. [Google Scholar] [CrossRef]
- Applying Electronic Nose Based on Odour Classification and Identification Technology in Detecting the Shelf Life of Fresh Fruits. Chem. Eng. Trans. 2018, 68, 217–222. [CrossRef]
- Bonah, E.; Huang, X.; Aheto, J.H.; Osae, R. Application of Electronic Nose as a Non-Invasive Technique for Odor Fingerprinting and Detection of Bacterial Foodborne Pathogens: A Review. J. Food Sci. Technol. 2020, 57, 1977–1990. [Google Scholar] [CrossRef]
- Du, D.; Wang, J.; Wang, B.; Zhu, L.; Hong, X. Ripeness Prediction of Postharvest Kiwifruit Using a MOS E-Nose Combined with Chemometrics. Sensors 2019, 19, 419. [Google Scholar] [CrossRef]
- Galvan, D.; Aquino, A.; Effting, L.; Mantovani, A.C.G.; Bona, E.; Conte-Junior, C.A. E-Sensing and Nanoscale-Sensing Devices Associated with Data Processing Algorithms Applied to Food Quality Control: A Systematic Review. Crit. Rev. Food Sci. Nutr. 2022, 62, 6605–6645. [Google Scholar] [CrossRef]
- Aghilinategh, N.; Dalvand, M.J.; Anvar, A. Detection of Ripeness Grades of Berries Using an Electronic Nose. Food Sci. Nutr. 2020, 8, 4919–4928. [Google Scholar] [CrossRef] [PubMed]
- Nategh, N.A.; Dalvand, M.J.; Anvar, A. Detection of Toxic and Non-Toxic Sweet Cherries at Different Degrees of Maturity Using an Electronic Nose. J. Food Meas. Charact. 2021, 15, 1213–1224. [Google Scholar] [CrossRef]
- Cozzolino, R.; Cefola, M.; Laurino, C.; Pellicano, M.P.; Palumbo, M.; Stocchero, M.; Pace, B. Electronic-Nose as Non-Destructive Tool to Discriminate “Ferrovia” Sweet Cherries Cold Stored in Air or Packed in High CO2 Modified Atmospheres. Front. Nutr. 2021, 8, 720092. [Google Scholar] [CrossRef] [PubMed]
- Cozzolino, R.; Martignetti, A.; Cefola, M.; Pace, B.; Capotorto, I.; De Giulio, B.; Montemurro, N.; Pellicano, M.P. Volatile Metabolites, Quality and Sensory Parameters of “Ferrovia” Sweet Cherry Cold Stored in Air or Packed in High CO2 Modified Atmospheres. Food Chem. 2019, 286, 659–668. [Google Scholar] [CrossRef]
- Cozzolino, R.; Pace, B.; Palumbo, M.; Laurino, C.; Picariello, G.; Siano, F.; De Giulio, B.; Pelosi, S.; Cefola, M. Profiles of Volatile and Phenolic Compounds as Markers of Ripening Stage in Candonga Strawberries. Foods 2021, 10, 3102. [Google Scholar] [CrossRef]
- Qiao, J.; Su, G.; Liu, C.; Zou, Y.; Chang, Z.; Yu, H.; Wang, L.; Guo, R. Study on the Application of Electronic Nose Technology in the Detection for the Artificial Ripening of Crab Apples. Horticulturae 2022, 8, 386. [Google Scholar] [CrossRef]
- Li, A.; Duan, S.; Dang, Y.; Zhang, X.; Xia, K.; Liu, S.; Han, X.; Wen, J.; Li, Z.; Wang, X.; et al. Origin Identification of Chinese Maca Using Electronic Nose Coupled with GC-MS. Sci. Rep. 2019, 9, 12216. [Google Scholar] [CrossRef]
- Longobardi, F.; Casiello, G.; Centonze, V.; Catucci, L.; Agostiano, A. Electronic Nose in Combination with Chemometrics for Characterization of Geographical Origin and Agronomic Practices of Table Grape. Food Anal. Methods 2019, 12, 1229–1237. [Google Scholar] [CrossRef]
- Liu, K.; Zhang, C. Volatile Organic Compounds Gas Sensor Based on Quartz Crystal Microbalance for Fruit Freshness Detection: A Review. Food Chem. 2021, 334, 127615. [Google Scholar] [CrossRef]
- Ghasemi-Varnamkhasti, M.; Mohammad-Razdari, A.; Yoosefian, S.H.; Izadi, Z.; Rabiei, G. Selection of an Optimized Metal Oxide Semiconductor Sensor (MOS) Array for Freshness Characterization of Strawberry in Polymer Packages Using Response Surface Method (RSM). Postharvest Biol. Technol. 2019, 151, 53–60. [Google Scholar] [CrossRef]
- Huang, X.; Yu, S.; Xu, H.; Aheto, J.H.; Bonah, E.; Ma, M.; Wu, M.; Zhang, X. Rapid and Nondestructive Detection of Freshness Quality of Postharvest Spinaches Based on Machine Vision and Electronic Nose. J. Food Saf. 2019, 39. [Google Scholar] [CrossRef]
- Nicolaï, B.M.; Beullens, K.; Bobelyn, E.; Peirs, A.; Saeys, W.; Theron, K.I.; Lammertyn, J. Nondestructive Measurement of Fruit and Vegetable Quality by Means of NIR Spectroscopy: A Review. Postharvest Biol. Technol. 2007, 46, 99–118. [Google Scholar] [CrossRef]
- McClure, W.F. 204 Years of near Infrared Technology: 1800–2003. J. Infrared Spectrosc. 2003, 11, 487–518. [Google Scholar] [CrossRef]
- Reusch, W. Table of Contents. Available online: https://www2.chemistry.msu.edu/faculty/reusch/VirtTxtJml/intro1.htm (accessed on 1 October 2022).
- Peirs, A.; Tirry, J.; Verlinden, B.; Darius, P.; Nicolaï, B.M. Effect of Biological Variability on the Robustness of NIR Models for Soluble Solids Content of Apples. Postharvest Biol. Technol. 2003, 28, 269–280. [Google Scholar] [CrossRef]
- Schaare, P.N.; Fraser, D.G. Comparison of Reflectance, Interactance and Transmission Modes of Visible-near Infrared Spectroscopy for Measuring Internal Properties of Kiwifruit (Actinidia Chinensis). Postharvest Biol. Technol. 2000, 20, 175–184. [Google Scholar] [CrossRef]
- Clark, C.J.; McGlone, V.A.; De Silva, H.N.; Manning, M.A.; Burdon, J.; Mowat, A.D. Prediction of Storage Disorders of Kiwifruit (Actinidia Chinensis) Based on Visible-NIR Spectral Characteristics at Harvest. Postharvest Biol. Technol. 2004, 32, 147–158. [Google Scholar] [CrossRef]
- McGlone, V.A.; Kawano, S. Firmness, Dry-Matter and Soluble-Solids Assessment of Postharvest Kiwifruit by NIR Spectroscopy. Postharvest Biol. Technol. 1998, 13, 131–141. [Google Scholar] [CrossRef]
- Liu, Y.; Sun, X.; Ouyang, A. Nondestructive Measurement of Soluble Solid Content of Navel Orange Fruit by Visible–NIR Spectrometric Technique with PLSR and PCA-BPNN. LWT - Food Sci. Technol. 2010, 43, 602–607. [Google Scholar] [CrossRef]
- Gómez, A.H.; He, Y.; Pereira, A.G. Non-Destructive Measurement of Acidity, Soluble Solids and Firmness of Satsuma Mandarin Using Vis/NIR-Spectroscopy Techniques. J. Food Eng. 2006, 77, 313–319. [Google Scholar] [CrossRef]
- Lee, K.J.; Kim, G.Y.; Kang, S.W.; Son, J.R.; Choi, D.S.; Choi, K.H. Measurement of Sugar Contents in Citrus Using Near Infrared Transmittance. Key Eng. Mater. 2004, 270–273, 1014–1019. [Google Scholar]
- Schulz, H.; Baranska, M.; Quilitzsch, R.; Schütze, W.; Lösing, G. Characterization of Peppercorn, Pepper Oil, and Pepper Oleoresin by Vibrational Spectroscopy Methods. J. Agric. Food Chem. 2005, 53, 3358–3363. [Google Scholar] [CrossRef] [PubMed]
- Amodio, M.L.; Ceglie, F.; Chaudhry, M.M.A.; Piazzolla, F.; Colelli, G. Potential of NIR Spectroscopy for Predicting Internal Quality and Discriminating among Strawberry Fruits from Different Production Systems. Postharvest Biol. Technol. 2017, 125, 112–121. [Google Scholar] [CrossRef]
- Braue, E.H.; Pannella, M.G. Consistency in Circle Cell FT-IR Analysis of Aqueous Solutions. Appl. Spectrosc. 1987, 41, 1057–1067. [Google Scholar] [CrossRef]



| Treatment | Experimental Conditions | Food Matrix | Effects | Reference |
|---|---|---|---|---|
| Microwave | 454 W/5 s | Minimally processed bok choy | Microwaving decreases respiration rate, while retarding decay occurrence, and improves cell membrane integrity. | [23] |
| Microwave | 300–100 W/35–10 s | Fresh cut apples | Application of the treatments at the highest intensity after minimal processing led to the best mesophiles and psychrophiles control. | [24] |
| Pulsed Electric Field | 10,000 pulses at 0.5, 1.0 and 1.5 kV cm−1 | Fresh-cut lotus root | Sugar content was reduced after PEF application in fresh samples, thus lowering the browning index and reducing acrylamide. | [26] |
| Pulsed Electric Field | 5 pulses of 350 kVm−1 | Carrot | Twelve hours after the treatment, high CO2 and volatiles production was observed; after 24 h, the largest total phenolic increase occurred. | [27] |
| Pulsed Electric Field | 0.8, 2 and 3.5 kV cm−1 and 5, 12 and 30 pulses | Carrot | Treatment did not affect color, while an increase in the phenolic and carotenoid content and softening was observed. | [28,29] |
| High hydrostatic pressure | Red cabbage: 150–200 MPa at 35–55 °C, 5–20 min/Radish: 100–200 MPa, at 20–40 °C, 5–10 min | Red cabbage leaves and radish tubers | Variations in the HHP treatments affected the integrity of the tissues and cell turgor. | [30] |
| High hydrostatic pressure | 50–400 MPa for 3–60 min | Fresh-cut papaya fruit | After HHP treatment and storage, the enhancement of carotenoid precursors and carotene content was observed. | [31] |
| High hydrostatic pressure | 100–600 MPa for 2 min | Fresh-cut pumpkin | Color parameters, firmness, electrical conductance, and pectin esterification were positively affected by HHP. | [32] |
| Mild high hydrostatic pressure | 20–80 MPa for 10 min | Mango | HHP reduced the respiration rate, prevented tissue damage, and positively affected the content of bioactive compounds. | [33] |
| High pressure processing | 100–300 MPa for 5–20 min | Minimally processed pineapple | HPP significantly affected firmness, flavonoids, polyphenols, vitamin C content, and colorimetric parameters. | [34] |
| High pressure treatments | 200, 400, 600 MPa for 5 min | Pumpkin | After applying 400 MPa, the pectinmethylesterase enzyme was inactivated. Color parameters decreased and an increase in antioxidant activity was observed. | [35] |
| High hydrostatic pressures | 400–600 MPa; 1–5 min | Blueberries | Treatment caused high tissue damage, thus resulting in the leakage of bioactive cellular components. | [36] |
| High-pressure treatments | 400,600 MPa; 1, 5 min | Zucchini slices | The longest treatment led to more severe cell lysis, browning and dehydration occurrence. | [37] |
| Dielectric Barrier Discharge (DBD) | 5 + 5 min on each side, 10 + 10, and 15 + 15; 30 and 60 min | Fresh cut apple | Plasma treatment caused less browning incidence and enhanced phenols and antioxidant activity after 10 min of treatment. | [38,39] |
| Cold plasma | 40 kV/90 s | Fresh cut cantaloupe | Cold plasma treatment significantly reduced bacteria and mold development during storage. Final product showed higher quality, firmness, and sensory attributes. | [40] |
| Dielectric barrier discharge cold plasma | Plasma levels of 7% and 14% duty cycle for 5, 10, 20 min. | Strawberry | Plasma treatment of 20 minutes reduced the mesophilic bacteria and yeasts and molds, while not affecting texture and color. | [41] |
| Atmospheric cold plasma (ACP) | ACP at 60 kV for 10, 15, 30 min | Strawberry | ACP treatment was able to prolong the product shelf-life in treated strawberries. A 15-minute treatment resulted in 2 log bacteria reduction, also enhancing the phenolic content and antioxidant activity. TSS, pH, and moisture were not affected. | [42] |
| Dielectric barrier discharge cold plasma | 60 kV/5 min | Fresh cut pitaya | Total aerobic bacterial count was significantly reduced by treatment, while phenolic content and antioxidant activity increased. | [43] |
| Atmospheric double barrier discharge plasma | 1 or 5 min at 45 and 65 kV | Fresh cut pears | Treatment effectively inhibited the growth of mesophiles and yeast and mold. The 65 kV/1 min treatment slowed respiration rate and maintained organoleptic properties and quality. | [44] |
| Cold atmospheric dielectric barrier discharge plasma | 5, 10, 15 and 20 min | Ready-to-eat rocket leafy salad | A treatment length of 10 min was optimal for the adequate reduction of the microbial load while maintaining color and firmness. | [45] |
| Atmospheric cold plasma (ACP) | 0, 5, 10, 15 and 20 min | Blueberries | ACP treatment inhibited microbial development and decay occurrence. Treatments of 5 and 10 min showed negligible effects on firmness, pH, ORP and anthocyanin concentration, but darkened the color. | [46] |
| Dielectric barrier discharge gas plasma and arc plasma-activated water (PAW) | 20-min DBD treatment and 20 min PAW immersion | Shiitake mushroom | Treatment reduced the total bacterial load after 7 days of storage, also slowing down the overall color modification and positively affecting firmness. | [47] |
| Plasma-activated water (PAW) | 7.0 kHz at 6 kV, 8 kV (PAW-8), 10 kV for 5 min | Fresh cut apple | PAW-8 treatment inhibited bacterial development and the reduced browning of the cut surface without affecting firmness, titratable acidity, radical scavenging activity, and antioxidant content. | [48] |
| Plasma activated water (PAW) | Activation times of 10, 20, 30, 45 and 60 min, 5 min washing | Fresh cut apple | PAW variably affected enzyme activities, while it did not show an effect on total phenolic content and antioxidant activity. Significant reductions in the aerobic bacteria and yeast and mold loads were observed. | [49] |
| Product | Active Ingredient | Country/Company | Fruit/Vegetable | Target |
|---|---|---|---|---|
| Bio-fungicides recommended for postharvest applications | ||||
| Biosave® | Pseudomonas syringae | Jet Harvest Solutions USA | Pome Fruit, Citrus, Strawberry, Cherry, Potato | Penicillium, Botrytis, Mucor |
| Nexy® | Candida oleophila | Lesaffre Belgium | Pome Fruit | Botrytis, Penicillium |
| BoniProtect® | Aureobasisium pullulans (2 strains) | Bio-ferm, Austria | Pome Fruit | Penicillium, Botrytis, Monilinia |
| BlossomProtect® | Grape | |||
| Botector® | ||||
| Noli | Metschnikowia fructicola | Koppert The Netherlands | Table Grape, Pome Fruit, Strawberry, Stone Fruit, Sweet Potato | Botrytis, Penicillium, Rhizopus, Aspergillus |
| Bio-fungicides developed for preharvest applications, also recommended for postharvest | ||||
| Serenade® Opti | Bacillus subtilis | Bayer | Grape, Berry Fruits, Potato | Botrytis, Silver scarf |
| Amylo-x® | Bacillus amyloliquefaciens | Biogard, Italy | Grape, Apple, Pear, Kiwifruit | Botrytis, Pseudomonas syringae |
| CBC-Europe, Germany | ||||
| Bio-protection agents developed for food processing, also recommended for postharvest | ||||
| Gaia™ | Metschnikowia fructicola | IOC, France | Harvested Grape, Withering Grape, Grape Musts | Botrytis, non-Saccharomyces spoiling yeasts |
| Nymphea™ | Torulaspora delbrueckii | ICV/ Lallemand, France | Harvested Grapes, Grape Musts | Botrytis, non-Saccharomyces spoiling yeasts |
| Adsorption Peaks | |||
|---|---|---|---|
| Functional Group | Wavenumber (cm−1) | Wavelength (µm) | |
| O-H | aliphatic and aromatic | 3600–3000 | 2.8–3.3 |
| NH2 | amine | 3600–3100 | 2.8–3.2 |
| CH | aromatic | 3150–3000 | 3.2–3.3 |
| CH | aliphatic | 3000–2850 | 3.3–3.5 |
| C≡N | nitril | 2400–2200 | 4.2–4.6 |
| C≡C- | alkyne | 2260–2100 | 4.4–4.8 |
| COOR | ester | 1750–1700 | 5.7–5.9 |
| COOH | carboxylic acid | 1740–1670 | 5.7–6.0 |
| C=O | aldehydes and ketones | 1740–1660 | 5.7–6.0 |
| CONH2 | amide | 1720–1640 | 5.8–6.1 |
| C=C- | alkene | 1670–1610 | 6.0–6.2 |
| Ø-O-R | aromatic | 1300–1180 | 7.7–8.5 |
| R-O-R | aliphatic | 1160–1060 | 8.6–9.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palumbo, M.; Attolico, G.; Capozzi, V.; Cozzolino, R.; Corvino, A.; de Chiara, M.L.V.; Pace, B.; Pelosi, S.; Ricci, I.; Romaniello, R.; et al. Emerging Postharvest Technologies to Enhance the Shelf-Life of Fruit and Vegetables: An Overview. Foods 2022, 11, 3925. https://doi.org/10.3390/foods11233925
Palumbo M, Attolico G, Capozzi V, Cozzolino R, Corvino A, de Chiara MLV, Pace B, Pelosi S, Ricci I, Romaniello R, et al. Emerging Postharvest Technologies to Enhance the Shelf-Life of Fruit and Vegetables: An Overview. Foods. 2022; 11(23):3925. https://doi.org/10.3390/foods11233925
Chicago/Turabian StylePalumbo, Michela, Giovanni Attolico, Vittorio Capozzi, Rosaria Cozzolino, Antonia Corvino, Maria Lucia Valeria de Chiara, Bernardo Pace, Sergio Pelosi, Ilde Ricci, Roberto Romaniello, and et al. 2022. "Emerging Postharvest Technologies to Enhance the Shelf-Life of Fruit and Vegetables: An Overview" Foods 11, no. 23: 3925. https://doi.org/10.3390/foods11233925
APA StylePalumbo, M., Attolico, G., Capozzi, V., Cozzolino, R., Corvino, A., de Chiara, M. L. V., Pace, B., Pelosi, S., Ricci, I., Romaniello, R., & Cefola, M. (2022). Emerging Postharvest Technologies to Enhance the Shelf-Life of Fruit and Vegetables: An Overview. Foods, 11(23), 3925. https://doi.org/10.3390/foods11233925










