Flaxseed Bioactive Compounds: Chemical Composition, Functional Properties, Food Applications and Health Benefits-Related Gut Microbes
Abstract
1. Introduction
2. Nutritional Composition of Flaxseeds
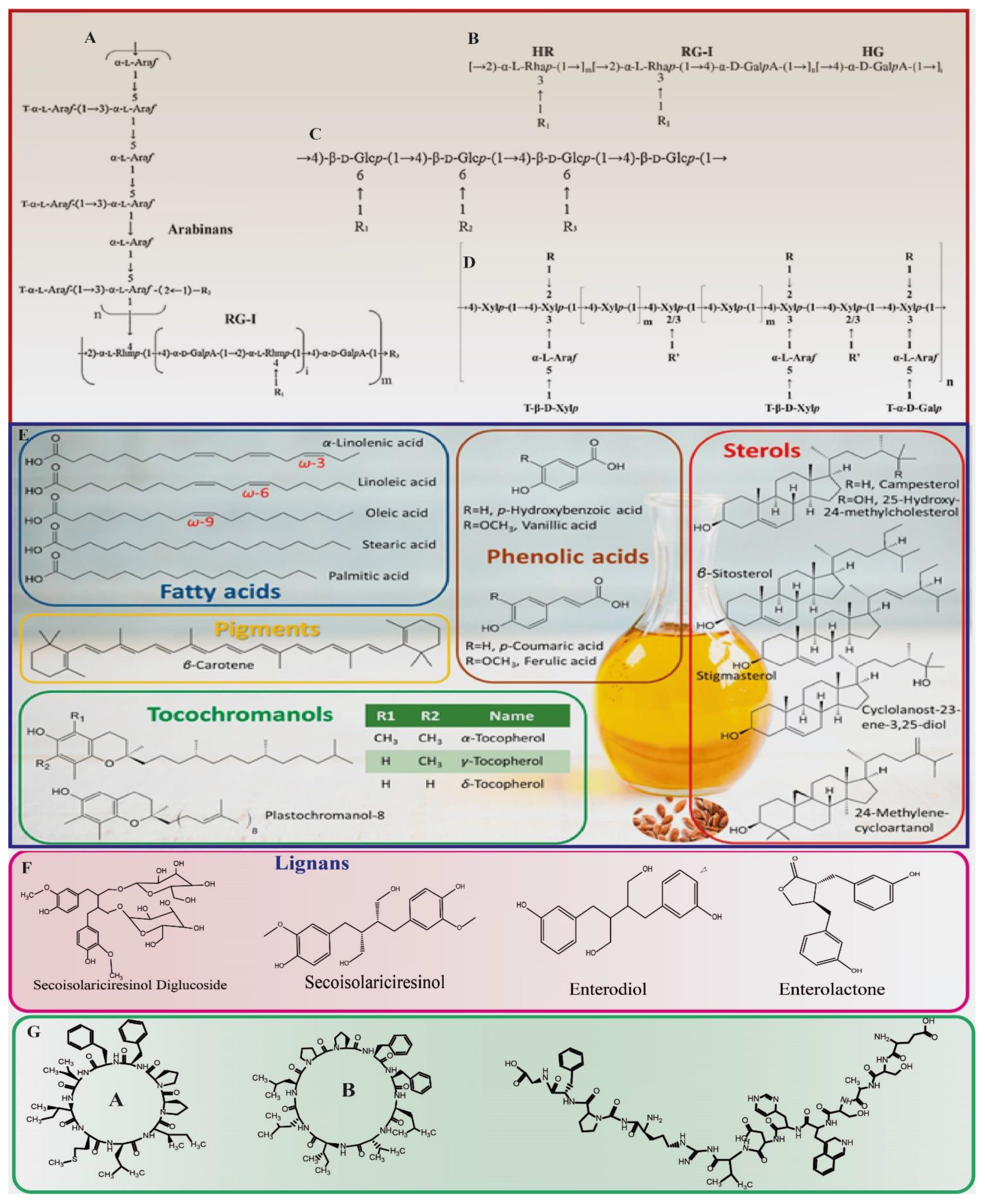
2.1. Lipids
| Composition of protein [16,17] | ||
| Amino acids | Flaxseed protein meal (g/100 g) | Flaxseed protein hydrolysate (mg/g) |
| Alanine | 4.59 | n.a * |
| Arginine | 10.63 | 33.12 |
| Asparagine | 9.76 | 496.51 |
| Cysteine | 3.80 | n.a * |
| Glutamic acid | 26.92 | 911.05 |
| Glycine | 6.14 | 288.43 |
| Histidine | 2.45 | 118.63 |
| Isoleucine | 5.21 | 207.49 |
| Leucine | 6.82 | 261.79 |
| Lysine | 4.18 | 191.41 |
| Methionine | 2.20 | 104.09 |
| Phenylalanine | 5.33 | 284.49 |
| Proline | 5.24 | n.a * |
| Serine | 5.88 | n.a * |
| Threonine | 4.19 | 169.94 |
| Tryptophan | 1.38 | n.a * |
| Tyrosine | 2.94 | 500.33 |
| Valine | 5.17 | 131.43 |
| Composition of oil (%) [15] | ||
| Fatty acids | Flaxseed oil | Soybean oil |
| Myristic (C14:0) | 0.03–0.05 | n.a *–0.12 |
| Pentadecanoic acid (C15:0) | n.a *–0.01 | n.a * |
| Palmitic (C16:0) | 4.58–6.42 | 10.80–11.50 |
| Palmitoleic (C16:1) | 0.04–0.20 | n.a *–0.16 |
| Margaric (C17:0) | n.a *–0.04 | n.a *–0.04 |
| Margaroleic (C17:1) | n.a *–0.03 | n.a * |
| Stearic (C18:0) | 3.65–5.96 | 3.62–4.11 |
| Oleic (C18:1n-9) | 16.33–22.56 | 20.80–23.50 |
| Linoleic (C18:2n-6) | 9.18–15.88 | 50.23–53.33 |
| Linolenic (C18:3n-3) | 42.97–61.06 | 6.76–7.65 |
| Arachidic (C20:0) | 0.01–0.20 | n.a *–0.32 |
| Gadoleic (C20:1) | n.a *–0.21 | n.a *–0.22 |
| Eicosanoic (C20:2) | n.a *–0.09 | n.a * |
| Behenic (C22:0) | 0.11–0.14 | n.a *–0.27 |
| Lignoceric (C24:0) | 0.04–0.13 | n.a *–0.13 |
| Saturated | 8.42–12.90 | 14.42–16.18 |
| Total Monounsaturated | 16.37–23.00 | 20.8–23.88 |
| Total Polyunsaturated | 52.15–76.94 | 56.99–60.98 |
| Composition of phenolic acids and lignans (mg/100 g) [16,18] | ||
| Non-defatted extracts | Defatted extracts | |
| p-Hydroxybenzoic acid | 1719 | 6454 |
| Chlorogenic acid | 720 | 1435 |
| Ferulic acid | 161 | 313 |
| Coumaric acid | 87 | 130 |
| Gallic acid | 29 | 17 |
| Vanillic acid | 22 | 42 |
| Sinapic acid | 18 | 27 |
| Protocatechuic acid | 7 | 7 |
| Caffeic acid | 4 | 15 |
| Diphyllin | 4.2 | n.a * |
| Secoisolariciresinol diglucoside | 1300 | n.a * |
| Secoisolariciresinol | 156 | n.a * |
| Laricinesol | 1.7 | n.a * |
| Matairesinol | 3.1 | n.a * |
| Pinoresinol | 0.8 | n.a * |
| Composition of vitamins and pigments (µg/g) [19,20] | ||
| Raw flaxseed | Boiled flaxseed | |
| α-Tocopherol | 6.26 | 4.56 |
| β-Tocopherol | 1.07 | 0.89 |
| γ-Tocopherol | 302.0 | 256.2 |
| δ-Tocopherol | 2.26 | 2.09 |
| β-Carotenoid | 0.52 | 0.48 |
| Xanthophyll | 27.1 | 20.2 |
| Sprouted | LT001 a | Zhongya 4 a |
| Lutein | 56.03 | 8.14 |
| Zeaxanthin | 2.38 | 2.76 |
| β-Cryptoxanthin | 1.64 | 1.86 |
| ε-Carotene | 1.44 | 1.61 |
| β-Carotene | 5.18 | 5.87 |
2.2. Polysaccharides
2.3. Protein/Peptides
2.4. Phenolic Compounds and Carotenoids
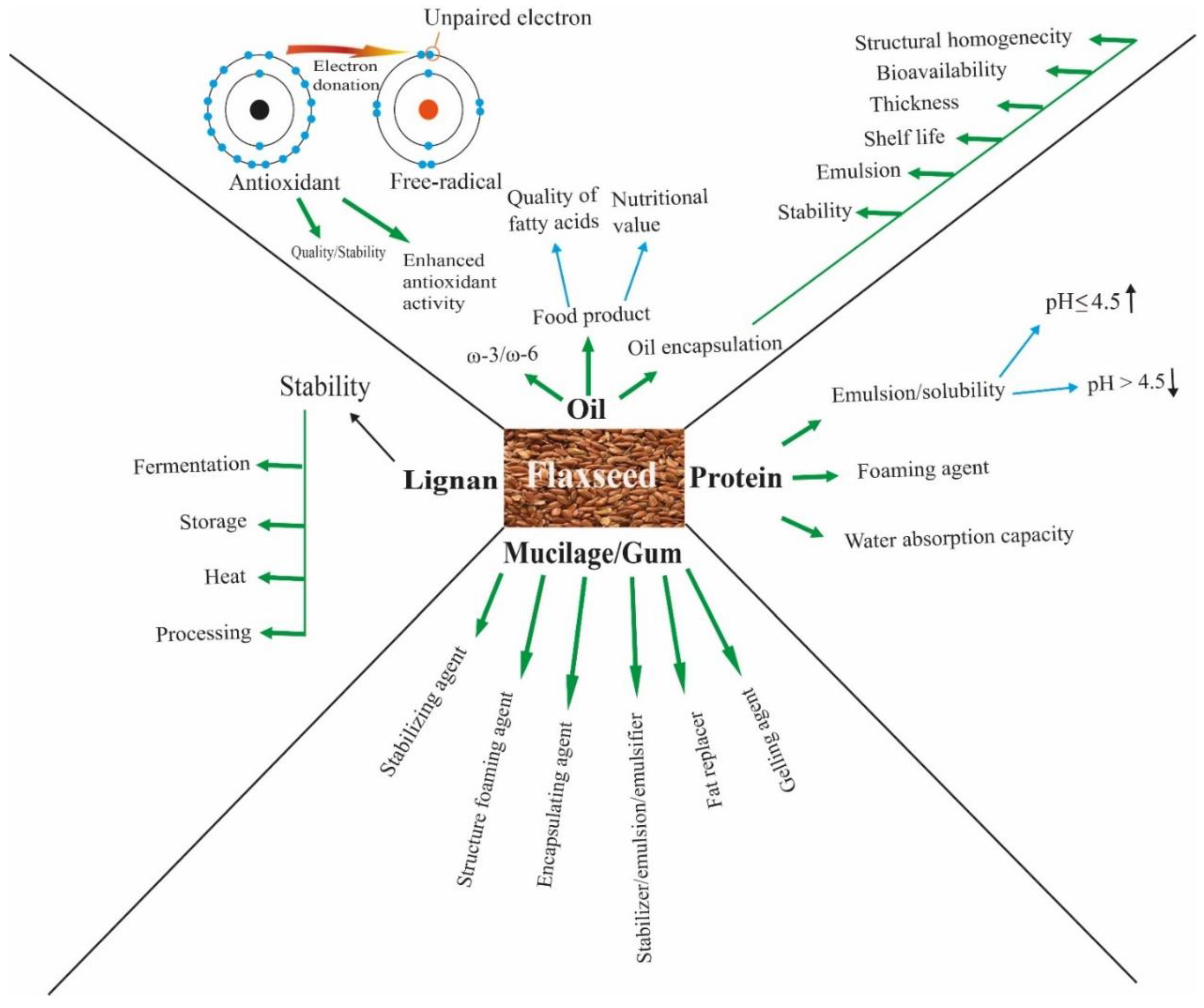
3. Food Applications of Flaxseed and Its Components
3.1. Flaxseed Kernel
3.2. Flaxseed Oil
3.3. Protein
3.4. Polysaccharide/Mucilage
3.5. Lignans
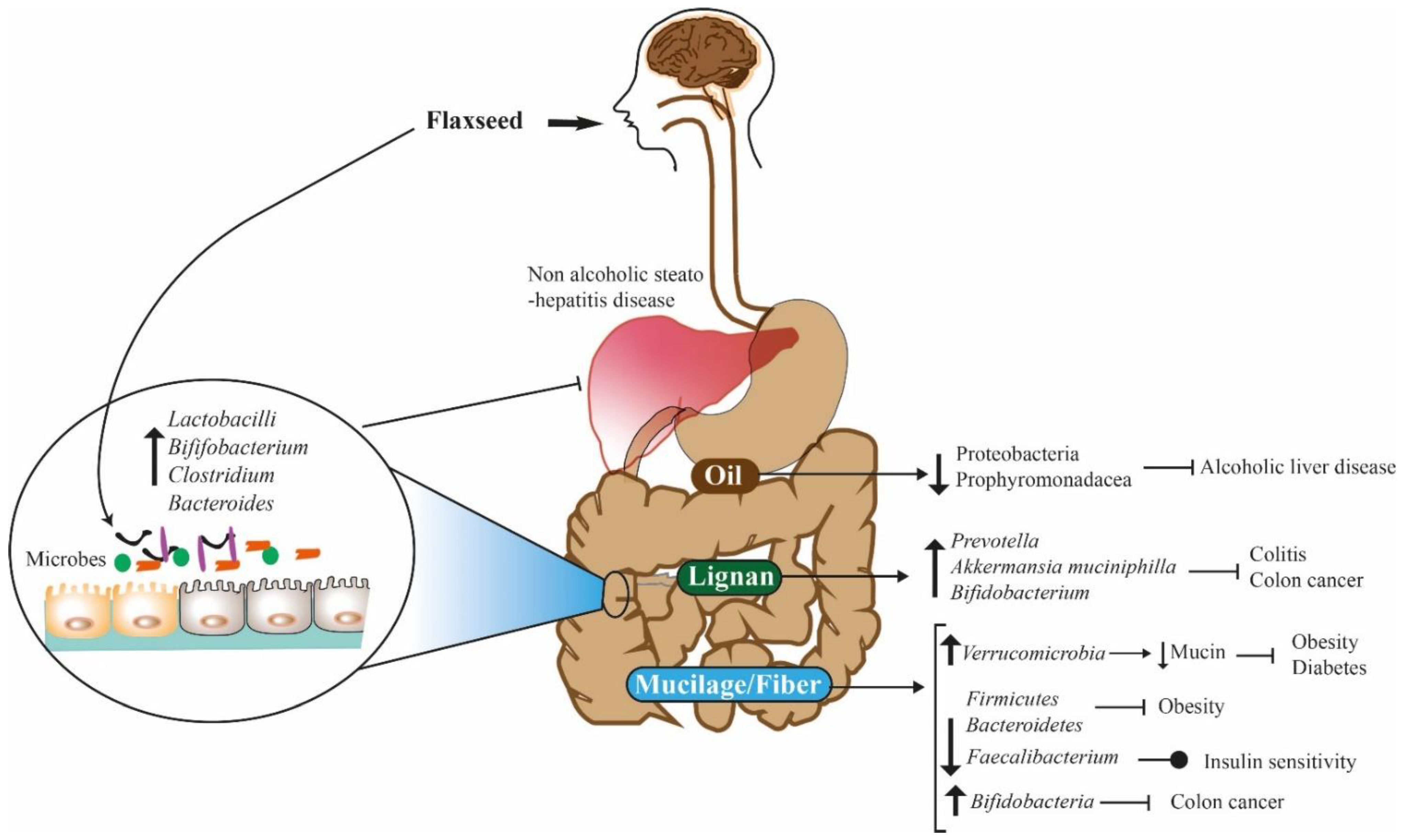
4. Role of Flaxseed Compounds in the Maintenance of Gut Microbes and Human Health
5. Conclusions and Future Perspective
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- de Lourdes, R.; Giada, M. Food applications for flaxseed and its components: Products and processing. Recent Pat. Food Nutr. Agric. 2010, 2, 181–186. [Google Scholar]
- Sanmartin, C.; Taglieri, I.; Venturi, F.; Macaluso, M.; Zinnai, A.; Tavarini, S.; Botto, A.; Serra, A.; Conte, G.; Flamini, G. Flaxseed cake as a tool for the improvement of nutraceutical and sensorial features of sourdough bread. Foods 2020, 9, 204. [Google Scholar] [CrossRef] [PubMed]
- Fale, S.K.; Umekar, M.J.; Das, R.; Alaspure, M. A comprehensive study of herbal cosmetics prepared from flaxseed. Multidiscip. Int. Res. J. Gujarat Technol. Univ. 2022, 4, 106–112. [Google Scholar]
- Oomah, B.D. Processing of flaxseed fiber, oil, protein, and lignan. In Flaxseed in Human Nutrition; American Botanical Council: Austin, TX, USA, 2003; pp. 363–386. [Google Scholar]
- Parikh, M.; Maddaford, T.G.; Austria, J.A.; Aliani, M.; Netticadan, T.; Pierce, G.N. Dietary flaxseed as a strategy for improving human health. Nutrients 2019, 11, 1171. [Google Scholar] [CrossRef] [PubMed]
- Dzuvor, C.K.O.; Taylor, J.T.; Acquah, C.; Pan, S.; Agyei, D. Bioprocessing of functional ingredients from flaxseed. Molecules 2018, 23, 2444. [Google Scholar] [CrossRef]
- Saka, İ.; Baumgartner, B.; Özkaya, B. Usability of microfluidized flaxseed as a functional additive in bread. J. Sci. Food Agric. 2022, 102, 505–513. [Google Scholar] [CrossRef]
- Puligundla, P.; Lim, S. A Review of Extraction Techniques and Food Applications of Flaxseed Mucilage. Foods 2022, 11, 1677. [Google Scholar] [CrossRef]
- Tvrzicka, E.; Kremmyda, L.-S.; Stankova, B.; Zak, A. Fatty acids as biocompounds: Their role in human metabolism, health and disease-a review. Part 1: Classification, dietary sources and biological functions. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc Czech Repub. 2011, 155, 117–130. [Google Scholar] [CrossRef]
- Yaqoob, N.; Bhatti, I.A.; Anwar, F.; Mushtaq, M.; Artz, W.E. Variation in physico-chemical/analytical characteristics of oil among different flaxseed (Linum usitatissimum L.) cultivars. Ital. J. Food Sci. 2016, 28, 83–89. [Google Scholar]
- Ishag, O.A.O.; Khalid, A.A.; Abdi, A.; Erwa, I.Y.; Omer, A.B.; Nour, A.H. Proximate Composition, Physicochemical Properties and Antioxidant Activity of Flaxseed. Annu. Res. Rev. Biol. 2019, 34, 1–10. [Google Scholar]
- Rubilar, M.; Gutiérrez, C.; Verdugo, M.; Shene, C.; Sineiro, J. Flaxseed as a source of functional ingredients. J. Soil. Sci Plant Nutr. 2010, 10, 373–377. [Google Scholar] [CrossRef]
- Zhang, Z.-S.; Wang, L.-J.; Li, D.; Li, S.-J.; Özkan, N. Characteristics of flaxseed oil from two different flax plants. Int. J. Food Prop. 2011, 14, 1286–1296. [Google Scholar] [CrossRef]
- Herchi, W.; Sakouhi, F.; Khaled, S.; Xiong, Y.; Boukhchina, S.; Kallel, H.; Curtis, J.M. Characterisation of the glycerophospholipid fraction in flaxseed oil using liquid chromatography–mass spectrometry. Food Chem. 2011, 129, 437–442. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Wen, C.; Duan, Y.; Deng, Q.; Peng, D.; Zhang, H.; Ma, H. The composition, extraction, analysis, bioactivities, bioavailability and applications in food system of flaxseed (Linum usitatissimum L.) oil: A review. Trends Food Sci. Technol. 2021, 118, 252–260. [Google Scholar] [CrossRef]
- Bekhit, A.E.-D.A.; Shavandi, A.; Jodjaja, T.; Birch, J.; Teh, S.; Ahmed, I.A.M.; Al-Juhaimi, F.Y.; Saeedi, P.; Bekhit, A.A. Flaxseed: Composition, detoxification, utilization, and opportunities. Biocatal. Agric. Biotechnol. 2018, 13, 129–152. [Google Scholar] [CrossRef]
- Guimarães Drummond e Silva, F.; Hernández-Ledesma, B.; Amigo, L.; Netto, F.M.; Miralles, B. Identification of peptides released from flaxseed (Linum usitatissimum) protein by Alcalase® hydrolysis: Antioxidant activity. LWT Food Sci. Technol. 2017, 76, 140–146. [Google Scholar] [CrossRef]
- El-Beltagi, H.; Salama, Z.; El-Hariri, D. Evaluation of fatty acids profile and the content of some secondary metabolites in seeds of different flax cultivars (Linum usitatissimum L.). Gen. Appl. Plant Physiol. 2007, 33, 187–202. [Google Scholar]
- Guo, X.; Wu, Y.; Xiang, N.; Gao, F.; Qiu, C. Effect of Ultrasonic Pretreatment on the Biosynthesis of Tocopherols, Tocotrienols and Carotenoids in Flax Sprouts (Linum Usitatissimum L.). J. Nat. Fibers 2021, 1–10. [Google Scholar] [CrossRef]
- Kiczorowska, B.; Samolińska, W.; Andrejko, D.; Kiczorowski, P.; Antoszkiewicz, Z.; Zając, M.; Winiarska-Mieczan, A.; Bąkowski, M. Comparative analysis of selected bioactive components (fatty acids, tocopherols, xanthophyll, lycopene, phenols) and basic nutrients in raw and thermally processed camelina, sunflower, and flax seeds (Camelina sativa L. Crantz, Helianthus L., and Linum L.). J. Food Sci. Technol. 2019, 56, 4296–4310. [Google Scholar] [CrossRef]
- Dąbrowski, G.; Czaplicki, S.; Konopka, I. Fractionation of sterols, tocols and squalene in flaxseed oils under the impact of variable conditions of supercritical CO2 extraction. J. Food Compost. Anal. 2019, 83, 103261. [Google Scholar] [CrossRef]
- Han, J.; Yang, Y.; Feng, M.; Wang, G. Analysis of phytosterol contents in Chinese plant food and primary estimation of its intake of people. Wei Sheng Yen Chiu J. Hyg. Res. 2007, 36, 301–305. [Google Scholar]
- Choo, W.-S.; Birch, J.; Dufour, J.-P. Physicochemical and quality characteristics of cold-pressed flaxseed oils. J. Food Compost. Anal. 2007, 20, 202–211. [Google Scholar] [CrossRef]
- Mène-Saffrané, L.; DellaPenna, D. Biosynthesis, regulation and functions of tocochromanols in plants. Plant Physiol. Biochem. 2010, 48, 301–309. [Google Scholar] [CrossRef]
- Obranović, M.; Škevin, D.; Kraljić, K.; Pospišil, M.; Neđeral, S.; Blekić, M.; Putnik, P. Influence of climate, variety and production process on tocopherols, plastochromanol-8 and pigments in flaxseed oil. Food Technol. Biotechnol. 2015, 53, 496. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Shim, Y.Y.; Timothy, J.T.; Wang, Y.; Reaney, M.J. Flaxseed gum a versatile natural hydrocolloid for food and non-food applications. Trends Food Sci. Technol. 2018, 75, 146–157. [Google Scholar] [CrossRef]
- Cui, W.; Kenaschuk, E.; Mazza, G. Influence of genotype on chemical composition and rheological properties of flaxseed gums. Food Hydrocoll. 1996, 10, 221–227. [Google Scholar] [CrossRef]
- Qian, K.; Cui, S.; Wu, Y.; Goff, H. Flaxseed gum from flaxseed hulls: Extraction, fractionation, and characterization. Food Hydrocoll. 2012, 28, 275–283. [Google Scholar] [CrossRef]
- Qian, K.-Y.; Cui, S.W.; Nikiforuk, J.; Goff, H.D. Structural elucidation of rhamnogalacturonans from flaxseed hulls. Carbohydr. Res. 2012, 362, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Elboutachfaiti, R.; Delattre, C.; Quéro, A.; Roulard, R.; Duchêne, J.; Mesnard, F.; Petit, E. Fractionation and structural characterization of six purified rhamnogalacturonans type I from flaxseed mucilage. Food Hydrocoll. 2017, 62, 273–279. [Google Scholar] [CrossRef]
- Roulard, R.; Petit, E.; Mesnard, F.; Rhazi, L. Molecular investigations of flaxseed mucilage polysaccharides. Int. J. Biol. Macromol. 2016, 86, 840–847. [Google Scholar] [CrossRef]
- Wu, S.; Wang, X.; Qi, W.; Guo, Q. Bioactive protein/peptides of flaxseed: A review. Trends Food Sci. Technol. 2019, 92, 184–193. [Google Scholar] [CrossRef]
- Madhusudhan, K.; Singh, N. Isolation and characterization of the major fraction (12 S) of linseed proteins. J. Agric. Food Chem. 1985, 33, 673–677. [Google Scholar] [CrossRef]
- Youle, R.J.; Huang, A.H. Occurrence of low molecular weight and high cysteine containing albumin storage proteins in oilseeds of diverse species. Am. J. Bot. 1981, 68, 44–48. [Google Scholar] [CrossRef]
- Marcone, M.F.; Kakuda, Y.; Yada, R.Y. Salt-soluble seed globulins of various dicotyledonous and monocotyledonous plants—I. Isolation/purification and characterization. Food Chem. 1998, 62, 27–47. [Google Scholar] [CrossRef]
- Madhusudhan, K.; Singh, N. Isolation and characterization of a small molecular weight protein of linseed meal. Phytochemistry 1985, 24, 2507–2509. [Google Scholar] [CrossRef]
- Shim, Y.Y.; Song, Z.; Jadhav, P.D.; Reaney, M.J. Orbitides from flaxseed (Linum usitatissimum L.): A comprehensive review. Trends Food Sci. Technol. 2019, 93, 197–211. [Google Scholar]
- Herchi, W.; Sakouhi, F.; Boukhchina, S.; Kallel, H.; Pepe, C. Changes in fatty acids, tocochromanols, carotenoids and chlorophylls content during flaxseed development. J. Am. Oil Chem. Soc. 2011, 88, 1011–1017. [Google Scholar] [CrossRef]
- Johnsson, P.; Kamal-Eldin, A.; Lundgren, L.N.; Åman, P. HPLC method for analysis of secoisolariciresinol diglucoside in flaxseeds. J. Agric. Food Chem. 2000, 48, 5216–5219. [Google Scholar] [CrossRef]
- Thompson, L.U.; Robb, P.; Serraino, M.; Cheung, F. Mammalian lignan production from various foods. Nutr. Cancer 1991, 16, 43–52. [Google Scholar] [CrossRef]
- Gutiérrez, C.; Rubilar, M.; Jara, C.; Verdugo, M.; Sineiro, J.; Shene, C. Flaxseed and flaxseed cake as a source of compounds for food industry. J. Soil Sci. Plant Nutr. 2010, 10, 454–463. [Google Scholar] [CrossRef]
- Gul, K.; Tak, A.; Singh, A.; Singh, P.; Yousuf, B.; Wani, A.A. Chemistry, encapsulation, and health benefits of β-carotene-A review. Cogent Food Agric. 2015, 1, 1018696. [Google Scholar] [CrossRef]
- Yonekura, L.; Nagao, A. Intestinal absorption of dietary carotenoids. Mol. Nutr. Food Res. 2007, 51, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Farag, M.A.; Elimam, D.M.; Afifi, S.M. Outgoing and potential trends of the omega-3 rich linseed oil quality characteristics and rancidity management: A comprehensive review for maximizing its food and nutraceutical applications. Trends Food Sci. Technol. 2021, 114, 292–309. [Google Scholar] [CrossRef]
- Parikh, M.; Netticadan, T.; Pierce, G.N. Flaxseed: Its bioactive components and their cardiovascular benefits. Am. J. Physiol. Heart Circ. Physiol. 2018, 314, H146–H159. [Google Scholar] [CrossRef] [PubMed]
- Hyvärinen, H.K.; Pihlava, J.-M.; Hiidenhovi, J.A.; Hietaniemi, V.; Korhonen, H.J.; Ryhänen, E.-L. Effect of processing and storage on the stability of flaxseed lignan added to dairy products. J. Agric. Food Chem. 2006, 54, 8788–8792. [Google Scholar] [CrossRef]
- Dev, D.; Quensel, E. Functional properties of linseed protein products containing different levels of mucilage in selected food systems. J. Food Sci. 1989, 54, 183–186. [Google Scholar] [CrossRef]
- Goyal, A.; Sharma, V.; Sihag, M.K.; Singh, A.; Arora, S.; Sabikhi, L. Fortification of dahi (Indian yoghurt) with omega-3 fatty acids using microencapsulated flaxseed oil microcapsules. J. Food Sci. Technol. 2016, 53, 2422–2433. [Google Scholar] [CrossRef]
- Goh, K.K.; Ye, A.; Dale, N. Characterisation of ice cream containing flaxseed oil. Int. J. Food Sci. Technol. 2006, 41, 946–953. [Google Scholar] [CrossRef]
- Ivanov, S.; Rashevskaya, T.; Makhonina, M. Flaxseed additive application in dairy products production. Procedia Food Sci 2011, 1, 275–280. [Google Scholar] [CrossRef]
- Aliani, M.; Ryland, D.; Pierce, G.N. Effect of flax addition on the flavor profile and acceptability of bagels. J. Food Sci. 2012, 77, S62–S70. [Google Scholar] [CrossRef]
- Alpaslan, M.; Hayta, M. The effects of flaxseed, soy and corn flours on the textural and sensory properties of a bakery product. J. Food Qual. 2006, 29, 617–627. [Google Scholar] [CrossRef]
- Conforti, F.D.; Davis, S.F. The effect of soya flour and flaxseed as a partial replacement for bread flour in yeast bread. Int. J. Food Sci. Technol. 2006, 41, 95–101. [Google Scholar] [CrossRef]
- Conforti, F.D.; Cachaper, K.F. Effects of selected antioxidants on physical and sensory characteristics of yeast bread containing flaxseed meal. Int. J. Consum. Stud. 2009, 33, 89–93. [Google Scholar] [CrossRef]
- Marpalle, P.; Sonawane, S.K.; Arya, S.S. Effect of flaxseed flour addition on physicochemical and sensory properties of functional bread. LWT Food Sci. Technol. 2014, 58, 614–619. [Google Scholar] [CrossRef]
- Hao, M.; Beta, T. Development of Chinese steamed bread enriched in bioactive compounds from barley hull and flaxseed hull extracts. Food Chem. 2012, 133, 1320–1325. [Google Scholar] [CrossRef]
- Menteş, Ö.; Bakkalbaşşi, E.; Ercan, R. Effect of the use of ground flaxseed on quality and chemical composition of bread. Food Sci. Technol. Int. 2008, 14, 299–306. [Google Scholar] [CrossRef]
- Roozegar, M.; Shahedi, M.; Keramet, J.; Hamdami, N.; Roshanak, S. Effect of coated and uncoated ground flaxseed addition on rheological, physical and sensory properties of Taftoon bread. J. Food Sci. Technol. 2015, 52, 5102–5110. [Google Scholar] [CrossRef] [PubMed]
- Lipilina, E.; Ganji, V. Incorporation of ground flaxseed into bakery products and its effect on sensory and nutritional characteristics—A pilot study. J. Foodserv. 2009, 20, 52–59. [Google Scholar] [CrossRef]
- Ahmed, M.; Header, E.; El-Sherif, F.A.; El-Dashlouty, M.; El-Brollose, S. Sensory, chemical and biological evaluation of some products fortified by whole flaxseed. Egypt. J. Agric. Res. 2010, 88, 257–271. [Google Scholar]
- Hussain, S.; Muhammad Anjum, F.; Butt, M.S.; Alamri, M.S.; Rafiq Khan, M. Biochemical and Nutritional Evaluation of Unleavened Flat Breads Fortified with Healthy Flaxseed. Int. J. Agric. Biol. 2012, 14, 190–196. [Google Scholar]
- Khattab, R.; Zeitoun, M.; Barbary, O.M. Evaluation of pita bread fortified with defatted flaxseed flour. Curr. Nutr. Food Sci. 2012, 8, 91–101. [Google Scholar] [CrossRef]
- Khouryieh, H.; Aramouni, F. Physical and sensory characteristics of cookies prepared with flaxseed flour. J. Sci. Food Agric. 2012, 92, 2366–2372. [Google Scholar] [CrossRef] [PubMed]
- Khouryieh, H.; Aramouni, F. Effect of flaxseed flour incorporation on the physical properties and consumer acceptability of cereal bars. Food Sci. Technol. Int. 2013, 19, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Masoodi, L.; Bashir, V. Fortification of biscuit with flaxseed: Biscuit production and quality evaluation. IOSR J. Environ. Sci. Toxicol. Food Technol. 2012, 1, 6–9. [Google Scholar] [CrossRef]
- Rathi, P.; Mogra, R. Development and sensory evaluation of superior products with flaxseed. Int. J. Food Sci. Nutr. 2013, 2, 12. [Google Scholar]
- Aliani, M.; Ryland, D.; Pierce, G.N. Effect of flax addition on the flavor profile of muffins and snack bars. Food Res. Int. 2011, 44, 2489–2496. [Google Scholar] [CrossRef]
- Fofana, B.; Cloutier, S.; Kirby, C.W.; McCallum, J.; Duguid, S. A well balanced omega-6/omega-3 ratio in developing flax bolls after heating and its implications for use as a fresh vegetable by humans. Food Res. Int. 2011, 44, 2459–2464. [Google Scholar] [CrossRef]
- Mervat, E.-D.; Mahmoud, K.F.; Bareh, G.F.; Albadawy, W. Effect of fortification by full fat and defatted flaxseed flour sensory properties of wheat bread and lipid profile laste. Int. J. Curr. Microbiol. Appl. Sci. 2015, 4, 581–598. [Google Scholar]
- Cameron, S.J.; Hosseinian, F. Potential of flaxseed in the development of omega-3 rice paper with antioxidant activity. LWT Food Sci. Technol. 2013, 53, 170–175. [Google Scholar] [CrossRef]
- Kaur, A.; Sandhu, V.; Sandhu, K. Effects of flaxseed addition on sensory and baking quality of whole wheat bread. Int. J. Food Nutr. Saf. 2013, 4, 43–54. [Google Scholar]
- Pohjanheimo, T.A.; Hakala, M.A.; Tahvonen, R.L.; Salminen, S.J.; Kallio, H.P. Flaxseed in breadmaking: Effects on sensory quality, aging, and composition of bakery products. J. Food Sci. 2006, 71, S343–S348. [Google Scholar] [CrossRef]
- Ahmed, Z.S. Physico-chemical, structural and sensory quality of corn-based flax-snack. Food/Nahrung 1999, 43, 253–258. [Google Scholar] [CrossRef]
- Manthey, F.; Lee, R.; Kegode, R. Quality of spaghetti containing ground flaxseed. In Proceedings of the 58th Flax Institute of the US, Fargo, ND, USA, 23–25 March 2000; pp. 92–99. [Google Scholar]
- Shearer, A.E.; Davies, C.G. Physicochemical properties of freshly baked and stored whole-wheat muffins with and without flaxseed meal. J. Food Qual. 2005, 28, 137–153. [Google Scholar] [CrossRef]
- Koca, A.F.; Anil, M. Effect of flaxseed and wheat flour blends on dough rheology and bread quality. J. Sci. Food Agric. 2007, 87, 1172–1175. [Google Scholar] [CrossRef]
- Stewart, S.; Mazza, G. Effect of flaxseed gum on quality and stability of a model salad dressing 1. J. Food Qual. 2000, 23, 373–390. [Google Scholar] [CrossRef]
- Xu, Y.; Hall Iii, C.; Wolf-Hall, C. Antifungal activity stability of flaxseed protein extract using response surface methodology. J. Food Sci. 2008, 73, M9–M14. [Google Scholar] [CrossRef]
- Waszkowiak, K.; Rudzińska, M. Effect of flaxseed meals and extracts on lipid stability in a stored meat product. J. Am. Oil Chem. Soc. 2014, 91, 979–987. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, Y.; Li, P.; Li, Z. Antibacterial properties of cyclolinopeptides from flaxseed oil and their application on beef. Food Chem. 2022, 385, 132715. [Google Scholar] [CrossRef]
- Ghafouri-Oskuei, H.; Javadi, A.; Asl, M.R.S.; Azadmard-Damirchi, S.; Armin, M. Quality properties of sausage incorporated with flaxseed and tomato powders. Meat Sci. 2020, 161, 107957. [Google Scholar] [CrossRef] [PubMed]
- Bilska, A.; Waszkowiak, K.; Błaszyk, M.; Rudzińska, M.; Kowalski, R. Effect of liver pâté enrichment with flaxseed oil and flaxseed extract on lipid composition and stability. J. Sci. Food Agric. 2018, 98, 4112–4120. [Google Scholar] [CrossRef] [PubMed]
- Liu, K. Expanding soybean food utilization. Food Technol. 2000, 54, 46–58. [Google Scholar]
- Ramcharitar, A.; Badrie, N.; Mattfeldt-Beman, M.; Matsuo, H.; Ridley, C. Consumer acceptability of muffins with flaxseed (Linum usitatissimum). J. Food Sci. 2005, 70, s504–s507. [Google Scholar] [CrossRef]
- Mercier, S.; Villeneuve, S.; Moresoli, C.; Mondor, M.; Marcos, B.; Power, K.A. Flaxseed-enriched cereal-based products: A review of the impact of processing conditions. Compr. Rev. Food Sci. Food Saf. 2014, 13, 400–412. [Google Scholar] [CrossRef] [PubMed]
- Condori, M.A.V.; Chagman, G.J.P.; Barriga-Sanchez, M.; Vilchez, L.F.V.; Ursetta, S.; Pérez, A.G.; Hidalgo, A. Effect of tomato (Solanum lycopersicum L.) lycopene-rich extract on the kinetics of rancidity and shelf-life of linseed (Linum usitatissimum L.) oil. Food Chem. 2020, 302, 125327. [Google Scholar] [CrossRef] [PubMed]
- Pham, L.B.; Wang, B.; Zisu, B.; Truong, T.; Adhikari, B. Microencapsulation of flaxseed oil using polyphenol-adducted flaxseed protein isolate-flaxseed gum complex coacervates. Food Hydrocoll. 2020, 107, 105944. [Google Scholar] [CrossRef]
- Lamothe, S.; Desroches, V.; Britten, M. Effect of milk proteins and food-grade surfactants on oxidation of linseed oil-in-water emulsions during in vitro digestion. Food Chem. 2019, 294, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Cheng, S.-F.; Bhattacharya, B.; Chakkaravarthi, S. Efficacy of free and encapsulated natural antioxidants in oxidative stability of edible oil: Special emphasis on nanoemulsion-based encapsulation. Trends Food Sci. Technol. 2019, 91, 305–318. [Google Scholar] [CrossRef]
- Nasrabadi, M.N.; Goli, S.A.H.; Doost, A.S.; Dewettinck, K.; Van der Meeren, P. Bioparticles of flaxseed protein and mucilage enhance the physical and oxidative stability of flaxseed oil emulsions as a potential natural alternative for synthetic surfactants. Colloids Surf. B Biointerfaces 2019, 184, 110489. [Google Scholar] [CrossRef]
- Li, R.; Dai, T.; Tan, Y.; Fu, G.; Wan, Y.; Liu, C.; McClements, D.J. Fabrication of pea protein-tannic acid complexes: Impact on formation, stability, and digestion of flaxseed oil emulsions. Food Chem. 2020, 310, 125828. [Google Scholar] [CrossRef] [PubMed]
- Leskovec, J.; Levart, A.; Perić, L.; Stojčić, M.Đ.; Tomović, V.; Pirman, T.; Salobir, J.; Rezar, V. Antioxidative effects of supplementing linseed oil-enriched diets with α-tocopherol, ascorbic acid, selenium, or their combination on carcass and meat quality in broilers. Poult. Sci. 2019, 98, 6733–6741. [Google Scholar] [CrossRef] [PubMed]
- Pi, Y.; Gao, S.; Ma, L.; Zhu, Y.; Wang, J.; Zhang, J.; Xu, J.; Bu, D. Effectiveness of rubber seed oil and flaxseed oil to enhance the α-linolenic acid content in milk from dairy cows. J. Dairy Sci. 2016, 99, 5719–5730. [Google Scholar] [CrossRef]
- Moallem, U.; Lehrer, H.; Livshits, L.; Zachut, M. The effects of omega-3 α-linolenic acid from flaxseed oil supplemented to high-yielding dairy cows on production, health, and fertility. Livest. Sci. 2020, 242, 104302. [Google Scholar] [CrossRef]
- Tonial, I.B.; Matsushita, M.; Furuya, W.M.; de Souza, N.E.; Visentainer, J.V. Fatty acid contents in fractions of neutral lipids and phospholipids of fillets of tilapia treated with flaxseed oil. J. Am. Oil Chem. Soc. 2012, 89, 1495–1500. [Google Scholar] [CrossRef]
- Huang, C.; Chiba, L.; Bergen, W. Bioavailability and metabolism of omega-3 polyunsaturated fatty acids in pigs and omega-3 polyunsaturated fatty acid-enriched pork: A review. Livest. Sci. 2021, 243, 104370. [Google Scholar] [CrossRef]
- Shin, J.-A.; Akoh, C.C.; Lee, K.-T. Enzymatic interesterification of anhydrous butterfat with flaxseed oil and palm stearin to produce low-trans spreadable fat. Food Chem. 2010, 120, 1–9. [Google Scholar] [CrossRef]
- Vassel, B.; Nesbitt, L. The nitrogenous constituents of flaxseed II. The isolation of a purified protein fraction. J. Biol. Chem. 1945, 159, 571–584. [Google Scholar] [CrossRef]
- Oomah, B.D.; Mazza, G. Compositional changes during commercial processing of flaxseed. Ind. Crops Prod. 1998, 9, 29–37. [Google Scholar] [CrossRef]
- Ye, X.-P.; Xu, M.-F.; Tang, Z.-X.; Chen, H.-J.; Wu, D.-T.; Wang, Z.-Y.; Songzhen, Y.-X.; Hao, J.; Wu, L.-M.; Shi, L.-E. Flaxseed protein: Extraction, functionalities and applications. Food Sci. Technol. 2022, 42, e22021. [Google Scholar] [CrossRef]
- Hu, Y.; Shim, Y.Y.; Reaney, M.J. Flaxseed gum solution functional properties. Foods 2020, 9, 681. [Google Scholar] [CrossRef]
- Cui, W.; Mazza, G.; Biliaderis, C. Chemical structure, molecular size distributions, and rheological properties of flaxseed gum. J. Agric. Food Chem. 1994, 42, 1891–1895. [Google Scholar] [CrossRef]
- Kaushik, P.; Dowling, K.; Adhikari, R.; Barrow, C.J.; Adhikari, B. Effect of extraction temperature on composition, structure and functional properties of flaxseed gum. Food Chem. 2017, 215, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Ding, H.H.; Cui, S.W.; Goff, H.D.; Wang, Q.; Chen, J.; Han, N.F. Soluble polysaccharides from flaxseed kernel as a new source of dietary fibres: Extraction and physicochemical characterization. Food Res. Int. 2014, 56, 166–173. [Google Scholar] [CrossRef]
- Fabre, J.-F.; Lacroux, E.; Valentin, R.; Mouloungui, Z. Ultrasonication as a highly efficient method of flaxseed mucilage extraction. Ind. Crops Prod. 2015, 65, 354–360. [Google Scholar] [CrossRef]
- Mazza, G.; Biliaderis, C. Functional properties of flax seed mucilage. J. Food Sci. 1989, 54, 1302–1305. [Google Scholar] [CrossRef]
- Warrand, J.; Michaud, P.; Picton, L.; Muller, G.; Courtois, B.; Ralainirina, R.; Courtois, J. Flax (Linum usitatissimum) seed cake: A potential source of high molecular weight arabinoxylans? J. Agric. Food Chem. 2005, 53, 1449–1452. [Google Scholar] [CrossRef]
- Fedeniuk, R.W.; Biliaderis, C.G. Composition and physicochemical properties of linseed (Linum usitatissimum L.) mucilage. J. Agric. Food Chem. 1994, 42, 240–247. [Google Scholar] [CrossRef]
- Chen, H.-H.; Xu, S.-Y.; Wang, Z. Interaction between flaxseed gum and meat protein. J. Food Eng. 2007, 80, 1051–1059. [Google Scholar] [CrossRef]
- Sun, J.; Li, X.; Xu, X.; Zhou, G. Influence of various levels of flaxseed gum addition on the water-holding capacities of heat-induced porcine myofibrillar protein. J. Food Sci. 2011, 76, C472–C478. [Google Scholar] [CrossRef]
- Zhou, W.W.; Meng, L.; Li, X.; Ma, L.; Dai, R. Effect of the interaction between carrageenan, gellan gum and flaxseed gum on quality attributes of starch-free emulsion-type sausage. J. Muscle Foods 2010, 21, 255–267. [Google Scholar] [CrossRef]
- Qin, L.; Xu, S.Y.; Zhang, W.B. Effect of enzymatic hydrolysis on the yield of cloudy carrot juice and the effects of hydrocolloids on color and cloud stability during ambient storage. J. Sci. Food Agric. 2005, 85, 505–512. [Google Scholar] [CrossRef]
- Zhang, F.; Cui, S.; Li, Z.; Yuan, Y.; Li, C.; Li, R. A combination of metabolite profiling and network pharmacology to explore the potential pharmacological changes of secoisolariciresinol-diglycoside. RSC Adv. 2020, 10, 34847–34858. [Google Scholar] [CrossRef]
- Long, J.-J.; Zu, Y.-G.; Fu, Y.-J.; Luo, M.; Mu, P.-S.; Zhao, C.-J.; Li, C.-Y.; Wang, W.; Li, J. Oil removal from oily water systems using immobilized flaxseed gum gel beads. RSC Adv. 2012, 2, 5172–5177. [Google Scholar] [CrossRef]
- Thakur, G.; Mitra, A.; Pal, K.; Rousseau, D. Effect of flaxseed gum on reduction of blood glucose and cholesterol in type 2 diabetic patients. Int. J. Food Sci. Nutr. 2009, 60, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Ibrügger, S.; Kristensen, M.; Mikkelsen, M.S.; Astrup, A. Flaxseed dietary fiber supplements for suppression of appetite and food intake. Appetite 2012, 58, 490–495. [Google Scholar] [CrossRef] [PubMed]
- Muir, A.D.; Westcott, N.D. Quantitation of the lignan secoisolariciresinol diglucoside in baked goods containing flax seed or flax meal. J. Agric. Food Chem. 2000, 48, 4048–4052. [Google Scholar] [CrossRef] [PubMed]
- Simbalista, R.L.; Frota, K.d.M.G.; Soares, R.A.M.; Arêas, J.A.G. Effect of storage and processing of Brazilian flaxseed on lipid and lignan contents. J. Food Sci. Technol. 2012, 32, 374–380. [Google Scholar] [CrossRef]
- Hall, C.A., III; Manthey, F.A.; Lee, R.E.; Niehaus, M. Stability of α-linolenic acid and secoisolariciresinol diglucoside in flaxseed-fortified macaroni. J. Food Sci. 2005, 70, c483–c489. [Google Scholar] [CrossRef]
- Shim, Y.Y.; Olivia, C.M.; Liu, J.; Boonen, R.; Shen, J.; Reaney, M.J. Secoisolariciresinol diglucoside and cyanogenic glycosides in gluten-free bread fortified with flaxseed meal. J. Agric. Food Chem. 2016, 64, 9551–9558. [Google Scholar] [CrossRef]
- Hornung, B.; Dos Santos, V.A.M.; Smidt, H.; Schaap, P.J. Studying microbial functionality within the gut ecosystem by systems biology. Genes Nutr. 2018, 13, 5. [Google Scholar] [CrossRef]
- Turroni, F.; Milani, C.; Duranti, S.; Mancabelli, L.; Mangifesta, M.; Viappiani, A.; Lugli, G.A.; Ferrario, C.; Gioiosa, L.; Ferrarini, A. Deciphering bifidobacterial-mediated metabolic interactions and their impact on gut microbiota by a multi-omics approach. ISME J. 2016, 10, 1656–1668. [Google Scholar] [CrossRef]
- Zhou, X.; Zhang, Z.; Huang, F.; Yang, C.; Huang, Q. In vitro digestion and fermentation by human fecal microbiota of polysaccharides from flaxseed. Molecules 2020, 25, 4354. [Google Scholar] [CrossRef] [PubMed]
- Rivière, A.; Gagnon, M.; Weckx, S.; Roy, D.; De Vuyst, L. Mutual cross-feeding interactions between Bifidobacterium longum subsp. longum NCC2705 and Eubacterium rectale ATCC 33656 explain the bifidogenic and butyrogenic effects of arabinoxylan oligosaccharides. Appl. Environ. Microbiol. 2015, 81, 7767–7781. [Google Scholar] [CrossRef] [PubMed]
- Clavel, T.; Borrmann, D.; Braune, A.; Doré, J.; Blaut, M. Occurrence and activity of human intestinal bacteria involved in the conversion of dietary lignans. Anaerobe 2006, 12, 140–147. [Google Scholar] [CrossRef]
- Clavel, T.; Doré, J.; Blaut, M. Bioavailability of lignans in human subjects. Nutr. Res. Rev. 2006, 19, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Pusceddu, M.M.; El Aidy, S.; Crispie, F.; O’Sullivan, O.; Cotter, P.; Stanton, C.; Kelly, P.; Cryan, J.F.; Dinan, T.G. N-3 polyunsaturated fatty acids (PUFAs) reverse the impact of early-life stress on the gut microbiota. PLoS ONE 2015, 10, e0139721. [Google Scholar]
- Wall, R.; Ross, R.P.; Shanahan, F.; O’Mahony, L.; Kiely, B.; Quigley, E.; Dinan, T.G.; Fitzgerald, G.; Stanton, C. Impact of administered bifidobacterium on murine host fatty acid composition. Lipids 2010, 45, 429–436. [Google Scholar] [CrossRef]
- Golding, M.; Wooster, T.J. The influence of emulsion structure and stability on lipid digestion. Curr. Opin. Colloid Interface Sci. 2010, 15, 90–101. [Google Scholar] [CrossRef]
- Singh, H.; Ye, A.; Horne, D. Structuring food emulsions in the gastrointestinal tract to modify lipid digestion. Prog. Lipid Res. 2009, 48, 92–100. [Google Scholar] [CrossRef]
- Hussain, M.M. Intestinal lipid absorption and lipoprotein formation. Curr. Opin. Lipidol. 2014, 25, 200. [Google Scholar] [CrossRef]
- Wang, T.Y.; Liu, M.; Portincasa, P.; Wang, D.Q.H. New insights into the molecular mechanism of intestinal fatty acid absorption. Eur. J. Clin. Investig. 2013, 43, 1203–1223. [Google Scholar] [CrossRef]
- Couëdelo, L.; Boué-Vaysse, C.; Fonseca, L.; Montesinos, E.; Djoukitch, S.; Combe, N.; Cansell, M. Lymphatic absorption of α-linolenic acid in rats fed flaxseed oil-based emulsion. Br. J. Nutr. 2011, 105, 1026–1035. [Google Scholar] [CrossRef]
- Nishimukai, M.; Hara, H.; Aoyama, Y. Enteral administration of soyabean lecithin enhanced lymphatic absorption of triacylglycerol in rats. Br. J. Nutr. 2003, 90, 565–571. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Luo, J.; Li, Y.; Mai, Y.; Gao, L.; Ou, S.; Wang, Y.; Liu, L.; Peng, X. Flaxseed gum reduces body weight by regulating gut microbiota. J. Funct. Foods 2018, 47, 136–142. [Google Scholar] [CrossRef]
- Gao, R.; Zhu, C.; Li, H.; Yin, M.; Pan, C.; Huang, L.; Kong, C.; Wang, X.; Zhang, Y.; Qu, S. Dysbiosis signatures of gut microbiota along the sequence from healthy, young patients to those with overweight and obesity. Obesity 2018, 26, 351–361. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Xu, Z.; Deng, Q.; Huang, Q.; Wang, X.; Huang, F. Beneficial effects of flaxseed polysaccharides on metabolic syndrome via gut microbiota in high-fat diet fed mice. Food Res. Int. 2020, 131, 108994. [Google Scholar] [CrossRef]
- Yang, C.; Wan, M.; Xu, D.; Pan, D.; Xia, H.; Yang, L.; Sun, G. Flaxseed powder attenuates non-alcoholic steatohepatitis via modulation of gut microbiota and bile acid metabolism through gut–liver axis. Int. J. Mol. Sci. 2021, 22, 10858. [Google Scholar] [CrossRef]
- Pittler, M.; Ernst, E. Complementary therapies for reducing body weight: A systematic review. Int. J. Obes. 2005, 29, 1030–1038. [Google Scholar] [CrossRef][Green Version]
- Ogawa, A.; Kadooka, Y.; Kato, K.; Shirouchi, B.; Sato, M. Lactobacillus gasseri SBT2055 reduces postprandial and fasting serum non-esterified fatty acid levels in Japanese hypertriacylglycerolemic subjects. Lipids Health Dis. 2014, 13, 36. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.J.; Jae, G.Y.; Lee, I.A.; Liu, M.J.; Shen, Y.F.; Sharma, S.P.; Jamal, M.A.; Yoo, J.H.; Kim, H.J.; Hong, S.T. Intestinal removal of free fatty acids from hosts by Lactobacilli for the treatment of obesity. FEBS Open Bio 2016, 6, 64–76. [Google Scholar] [CrossRef] [PubMed]
- Drissi, F.; Raoult, D.; Merhej, V. Metabolic role of lactobacilli in weight modification in humans and animals. Microb. Pathog. 2017, 106, 182–194. [Google Scholar] [CrossRef] [PubMed]
- Tamargo, A.; Cueva, C.; Laguna, L.; Moreno-Arribas, M.V.; Muñoz, L.A. Understanding the impact of chia seed mucilage on human gut microbiota by using the dynamic gastrointestinal model simgi®. J. Funct. Foods 2018, 50, 104–111. [Google Scholar] [CrossRef]
- Power, K.A.; Lepp, D.; Zarepoor, L.; Monk, J.M.; Wu, W.; Tsao, R.; Liu, R. Dietary flaxseed modulates the colonic microenvironment in healthy C57Bl/6 male mice which may alter susceptibility to gut-associated diseases. J. Nutr. Biochem. 2016, 28, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Pulkrabek, M.; Rhee, Y.; Gibbs, P.; Hall, C. Flaxseed-and buckwheat-supplemented diets altered Enterobacteriaceae diversity and prevalence in the cecum and feces of obese mice. J. Diet. Suppl. 2017, 14, 667–678. [Google Scholar] [CrossRef] [PubMed]
- Xia, H.; Shi, X.; Zhou, B.; Sui, J.; Yang, C.; Liu, H.; Yang, L.; Wang, S.; Sun, G. Milled flaxseed-added diets ameliorated hepatic inflammation by reducing gene expression of TLR4/NF-κB pathway and altered gut microbiota in STZ-induced type 1 diabetic mice. Food Sci. Hum. Wellness 2022, 11, 32–40. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, H.; Yin, P.; Fan, H.; Sun, L.; Liu, Y. Flaxseed oil ameliorates alcoholic liver disease via anti-inflammation and modulating gut microbiota in mice. Lipids Health Dis. 2017, 16, 44. [Google Scholar] [CrossRef] [PubMed]
- de Freitas Gomides, A.F.; Gonçalves, R.V.; de Paula, S.O.; Ferreira, C.L.d.L.F.; Comastri, D.S.; Peluzio, M.d.C.G. Defatted flaxseed meal prevents the appearance of aberrant crypt foci in the colon of mice increasing the gene expression of p53. Nutr. Hosp. 2015, 31, 1675–1681. [Google Scholar]
- Lagkouvardos, I.; Kläring, K.; Heinzmann, S.S.; Platz, S.; Scholz, B.; Engel, K.H.; Schmitt-Kopplin, P.; Haller, D.; Rohn, S.; Skurk, T. Gut metabolites and bacterial community networks during a pilot intervention study with flaxseeds in healthy adult men. Mol. Nutr. Food Res. 2015, 59, 1614–1628. [Google Scholar] [CrossRef] [PubMed]
- van Kranen, H.J.; Mortensen, A.; Sorensen, I.K.; van den Berg-Wijnands, J.; Beems, R.; Nurmi, T.; Adlercreutz, H.; van Kreijl, C.F. Lignan precursors from flaxseed or rye bran do not protect against the development of intestinal neoplasia in ApcMin mice. Nutr. Cancer 2003, 45, 203–210. [Google Scholar] [CrossRef]
- Määttänen, P.; Lurz, E.; Botts, S.R.; Wu, R.Y.; Yeung, C.W.; Li, B.; Abiff, S.; Johnson-Henry, K.C.; Lepp, D.; Power, K.A. Ground flaxseed reverses protection of a reduced-fat diet against Citrobacter rodentium-induced colitis. Am. J. Physiol. Gastrointest. Liver Physiol. 2018, 315, G788–G798. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Xing, C.; Long, W.; Wang, H.Y.; Liu, Q.; Wang, R.-F. Impact of microbiota on central nervous system and neurological diseases: The gut-brain axis. J. Neuroinflamm. 2019, 16, 53. [Google Scholar] [CrossRef]
- Senizza, A.; Rocchetti, G.; Mosele, J.I.; Patrone, V.; Callegari, M.L.; Morelli, L.; Lucini, L. Lignans and gut microbiota: An interplay revealing potential health implications. Molecules 2020, 25, 5709. [Google Scholar] [CrossRef] [PubMed]
- Bonaz, B.; Sinniger, V.; Pellissier, S. The vagus nerve in the neuro-immune axis: Implications in the pathology of the gastrointestinal tract. Front. Immunol. 2017, 8, 1452. [Google Scholar] [CrossRef] [PubMed]
- Bonaz, B.; Sinniger, V.; Pellissier, S. Vagus nerve stimulation: A new promising therapeutic tool in inflammatory bowel disease. J. Intern. Med. 2017, 282, 46–63. [Google Scholar] [CrossRef] [PubMed]
- Rieder, R.; Wisniewski, P.J.; Alderman, B.L.; Campbell, S.C. Microbes and mental health: A review. Brain. Behav. Immun. 2017, 66, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Farzi, A.; Fröhlich, E.E.; Holzer, P. Gut microbiota and the neuroendocrine system. Neurotherapeutics 2018, 15, 5–22. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D. Interactions between gut microbes and host cells control gut barrier and metabolism. Int. J. Obes. Suppl. 2016, 6, S28–S31. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Sha, L.; Li, Y.; Zhu, L.; Wang, Z.; Li, K.; Lu, H.; Bao, T.; Guo, L.; Zhang, X. Dietary α-linolenic acid-rich flaxseed oil exerts beneficial effects on polycystic ovary syndrome through sex steroid hormones—Microbiota—Inflammation axis in rats. Front. Endocrinol. 2020, 11, 284. [Google Scholar] [CrossRef] [PubMed]
| Flaxseed Form | Method of Processing/ Products Name | Dose of Flaxseed Supplementation | Mechanism | Reference |
|---|---|---|---|---|
| Fortification of flaxseed in dairy products | ||||
| Flaxseed lignan SDG | SDG stability in milk, yogurt, whey drinks, and cheese | 1 g/10 L | ↓SDG hydrolysis in cheese by the presence of lactic acid bacteria and enzymes. ↓SDG 25% due to the ↓pH of the whey drinks. ↑Temperature pasteurization of milk and whey. SDG was found stable. | [46] |
| HMPC and LMPI | Ice cream | 0.5% and 1% | ↑HMPC ↑Viscosity, LMPI did not affect the viscosity of ice cream; LMPI reduced sensory score more than gelatin. ↑Ice cream is overrun by ↑HMPC but ↓by higher LMPI. LMPI provides a better overrun than gelatin. | [47] |
| MEFOP | Fermentation or Indian yogurt | 1–3% | ↑The acidity of fortified yogurt samples may be due to lactose in the MFOP. ↑Gelling properties may be due to the capability of proteins to entrap without syneresis. ↑Peroxide value during storage due to FO, susceptible to oxidation. | [48] |
| FO | Ice cream | 0–12% | ↑Meltdown rate, ↓ice cream hardness linear to concentration. | [49] |
| Flaxseed as additive | Microstructure of flaxseed in butter | 0.8–1.6% | Microstructure of flaxseed, globules, and cellular microstructure ↑butter structure, ↓degree of destruction. | [50] |
| Fortification of flaxseed in baked products | ||||
| Whole flaxseed flour | Bagels | 30 g per bagel | Flax and grain ↑aroma and flavor, cinnamon raisin bagel ↓aroma and flavor, ↑sweet aroma and taste. Cinnamon raisin bagels had a higher acceptance rate of flavor compared to sunflower, sesame, and plain bagels. | [51] |
| Flaxseed meal and flour | Bagels and pretzel-type bakery products | 5, 10, and 15% | The sample with 15% of flaxseed; ↓flavor 5% flaxseed ↓lightness or brightness values (L), whereas 10 and 15% flaxseed supplementation: ↓Fracture force, Formulation up to 10%; ↑crumb redness and darkness, has significant overall acceptability, nutritious and healthy substitute to consumers. | [52] |
| Flaxseed flour | Bread | 15% | ↓Loaf volume of bread, bright crust, and darker crumb | [53] |
| Flaxseed meal | Bread | 15% | Flaxseed bread was evaluated during a period of 8 weeks in storage ↑crumb firmness, no significant differences in sensory attributes. | [54] |
| Raw and roasted ground flaxseed | Bread | 10 g/100 g | Flaxseed enriched bread ↑water absorption, dough stickiness, and crumb softness. ↓Protein digestibility than the control. | [55] |
| Flaxseed hull extracts | Chinese steamed bread | 1% (w/w) | ↑Phytochemical content, ↑DPPH, ↑total phenolic content, ↑antioxidant activity. | [56] |
| Flaxseed flour | Bread | 10, 15, 20, and 25% | Ground flaxseed 10%: ↑loaf volume, Dallman degree, nutritional content (linolenic acid and γ-tocopherol), and ↓staling brad. Flaxseed flour used with 15% ↑sensory acceptability. | [57] |
| Coated and uncoated ground flaxseed | Taftoon bread | 5, 15, and 25% | ↑Coated and uncoated ground flaxseed, ↓water absorption due to rich in oil can coat starch and gluten ↑stability, ↑dough development, and relaxation time. Ground flaxseed with Arabic gum ↑water absorption, ↑oxidative stability for 80 days at 25 °C. | [58] |
| Ground flaxseed | Yeast bread and muffins | 15, 25, and 30% | Flaxseed improved the color of both bread and muffins due to the presence of leutin or zeaxanthin and the high protein content of flaxseed. 30% ground flaxseed appropriate formulations for bread. | [59] |
| Roasted flaxseed flour | Pan bread and pizza | 10, 15, and 20% | Flaxseed with 15% ↑protein, ↑fat, ↑fiber, ↓carbohydrates, and ↓total serum cholesterol. | [60] |
| Full fat and partially defatted flaxseed flour | Unleavened flatbread | 4, 8, 12, 16, and 20% | ↑Acceptability of unleavened flatbreads with maximum flaxseed containing 12% full-fat flour and 16% partially defatted flour, ↑soluble and insoluble total dietary fiber, and ↑essential amino acids. | [61] |
| Flaxseed cake flour | Pita bread | 5, 10, 15, and 20% | 15 and 20% flaxseed: ↑Water absorption due to protein and mucilage, ↑mixing time (4.43 min) of dough, ↑extension (elasticity) of dough, ↑water holding capacity, ↑moisture content, ↑flaxseed cake flour, ↑alkaline water retention capacity. | [62] |
| Whole flaxseed flour | High Protein Cookies | 12% (w/w) | ↑Cookies hardness due to high protein, ↑darker and browner appearance, ↑sensory of 6 and 12% flaxseed, up to 12% flaxseed without negatively affecting the quality, | [63] |
| Golden flaxseed flour | Cereal bars | 6, 12, and 18% | ↑Nutritional qualities incorporated up to 12% without affecting their sensory and quality, ↑consumer acceptability, and no distinction between the control and 12% flaxseed cereal bars. | [64] |
| Roasted flaxseed, flour | Biscuits | 10, 25, and 43% | 10% flaxseed ↑quality (Moisture content, fortification, dark color, texture) and nutritional value without undesirable change. | [65] |
| Flaxseed flour | Biscuits | 20, 30, and 40% | Flaxseed flour with 30% acceptable and 40% unacceptable, and product appearance was affected, i.e., the darker color and bitter taste were found by the panel. | [66] |
| Whole flaxseed flour | Muffin or snack bar | 30 g per muffin or bar | ↑Flax aroma, ↓sweetness, ↓vanilla aroma, ↑bitter taste, while no intensities on gingerbread raisin snack, ↑spice aroma, ↑nutritional value. | [67] |
| Flaxseed | Flaxseed boll | 1 g per each boll | Flax balls under a cooking treatment balanced ω-3/ω-6 ratio, stable fatty acids profile, ↓CG, 16 days after anthesis bolls were more stable compared to 8 days after anthesis under heat treatment with good taste, texture, and aroma. | [68] |
| Defatted and non-defatted flaxseed flour | Wheat bread | 10% NDF, 15% DF | DF and NDF fortified wheat flour: ↑protein, ↑fiber, ↑ash, ↓and carbohydrates, while 15% DF: ↓fat, ↑carbohydrates, ↑High-density lipoprotein-cholesterol, ↑triglycerides, ↓very low-density lipoprotein, ↓total cholesterol, ↓low-density lipoprotein. | [69] |
| Flaxseed flour | ω-3 rice paper | 10% (w/w) | ↑Antioxidant activity (231.7 mmol TE/g), ↑nutritional value. | [70] |
| Germinated and non-germinated flaxseed flour | Whole wheat bread | 0, 5, 10, 15, and 20 % (w/w) | Germinated and non-germinated flaxseed: 10% acceptable for bread baking and sensory, ↑loaf volume, ↑and overall acceptability. Germinate flaxseed used in bread: ↑nutritional value, ↓decreased anti-nutrients ↑bioavailability, ↑nutritional absorption. | [71] |
| Whole flaxseed and crushed; FO and crushed flaxseed | Flaxseed bread roll and cinnamon roll | 5% whole seed flour + 3% crushed seed, 13% FO + 1% crushed | In storage for 5 days at room temperature with no rancid odor detected, soft and remaining moisture content based on sensory panel and instrumental measurement. | [72] |
| Flaxseed flour | Corn snack | Up to 20% | ↓Puffy extruded, probably due to protein and fat competition for water with starch | [73] |
| Ground flaxseed | Spaghetti | 2.5–15% | ↑Dough development time and strength, ↑dark color. Smaller flaxseed flour size, better food quality produced | [74] |
| Flaxseed flour | Muffins | 2%, 5% | ↑Flaxseed flour proportion, ↑viscosity, ↑Firmness, ↓elastic texture, ↑dark color with less redness, yellowness based on the Hunter scale, and no anti-staling effect. | [75] |
| Flaxseed flour | White bread | 5–20% | Addition of flaxseed flour above 10%: ↑water absorption capacity, ↓dough stability and strength, ↑dough development time, ↑bread volume, ↓darker color of the crust, and ↑the value of crumb. | [76] |
| Fortification of flaxseed in other products | ||||
| FM extract | Salad dressing | 0–1.5% | ↑Protein content, ↑surface active properties, ↑emulsion stability at pH 6.0 and 2.0, ↑FM extract concentration in salad dressing, ↑viscosity, ↓and oil droplet size. Above mucilage concentration 0.45% (w/w), ↑stabilizing effect. | [77] |
| FP extract (high in protein and carbohydrate) | Potato dextrose agar | 5% | Antifungal activity against (50%) Penicillium sp, Fusarium gramineaum, Aspergillus flavus, and 40% against Pencillium chrysogenum (under the conditions: 72 °C and 15 s), ↓acidic pH, ↑fungistatic activity. | [78] |
| Flaxseed extracts and meal | Pork meat | 1.5% and 3% | ↑Extended shelf life of meat, ↓oxidation of cholesterol and fatty acid, ↓peroxide value, and thiobarbituric acid-reactive substance. | [79] |
| Flaxseed cyclolinopeptides | Beef | 100–200 µM | ↓Listeria monocytogene activity during beef storage. | [80] |
| Flaxseed powder | Beef sausages | 0,3, and 6% | ↓Nitric content during storage, ↑linolenic acid, and no adverse effect on the sensory evaluation. | [81] |
| Flaxseed oil and extract | Liver pate | 20% and 0.5–0.25% | ↓Lipid oxidation, ↓monoenoic and saturated fatty acids, ↑polyene fatty acids, ↑phytosterols, and improve the oxidative stability of the product. | [82] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mueed, A.; Shibli, S.; Korma, S.A.; Madjirebaye, P.; Esatbeyoglu, T.; Deng, Z. Flaxseed Bioactive Compounds: Chemical Composition, Functional Properties, Food Applications and Health Benefits-Related Gut Microbes. Foods 2022, 11, 3307. https://doi.org/10.3390/foods11203307
Mueed A, Shibli S, Korma SA, Madjirebaye P, Esatbeyoglu T, Deng Z. Flaxseed Bioactive Compounds: Chemical Composition, Functional Properties, Food Applications and Health Benefits-Related Gut Microbes. Foods. 2022; 11(20):3307. https://doi.org/10.3390/foods11203307
Chicago/Turabian StyleMueed, Abdul, Sahar Shibli, Sameh A. Korma, Philippe Madjirebaye, Tuba Esatbeyoglu, and Zeyuan Deng. 2022. "Flaxseed Bioactive Compounds: Chemical Composition, Functional Properties, Food Applications and Health Benefits-Related Gut Microbes" Foods 11, no. 20: 3307. https://doi.org/10.3390/foods11203307
APA StyleMueed, A., Shibli, S., Korma, S. A., Madjirebaye, P., Esatbeyoglu, T., & Deng, Z. (2022). Flaxseed Bioactive Compounds: Chemical Composition, Functional Properties, Food Applications and Health Benefits-Related Gut Microbes. Foods, 11(20), 3307. https://doi.org/10.3390/foods11203307













