Sustainable Applications for the Valorization of Cereal Processing By-Products
Abstract
:1. Introduction
2. Cereal Processing By-Products and Target Compounds
3. Recovery Approaches
4. Emerging Technologies
5. The Biorefinery Concept
6. Applications
7. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pallas, L. Rice Processing: Beyond the Farm Gate. In Ency-Clopedia of Food Grains, Second Edition, Volume 3 Grain-Based Products and Their Processing; Wrigley, C., Corke, H., Seetharaman, K., Faubion, J., Eds.; Elsevier Academic Press: Oxford, UK, 2016; pp. 446–452. [Google Scholar]
- Awika, J.M. Health Promoting Effects of Cereal and Cereal Products. In Fruit and Cereal Bioactives: Sources, Chemistry, and Applications; Tokuşoğlu, Ö., Hall, C., III, Eds.; CRC Press: Boca Raton, FL, USA, 2011; pp. 9–17. [Google Scholar]
- Preedy, V.R.; Watson, R.R.; Patel, V.B. Flour and Breads and Their Fortification in Health and Disease Prevention; Academic Press: Cambridge, MA, USA, 2011. [Google Scholar]
- Arzami, A.N.; Ho, T.M.; Mikkonen, K.S. Valorization of cereal by-product hemicelluloses: Fractionation and purity considerations. Food Res. Int. 2022, 151, 110818. [Google Scholar] [CrossRef]
- Dapcevic-Hadadev, T.; Hadnadev, M.; Pojic, M. The healthy components of cereal by-products and their functional properties. In Sustainable Recovery and Reutilization of Cereal Processing By-Products; Galanakis, C.M., Ed.; Elsevier Inc.: Waltham, MA, USA, 2018; pp. 27–61. [Google Scholar]
- Skendi, A.; Zinoviadou, K.; Papageorgiou, M.; Rocha, J. Advances on the Valorisation and Functionalization of By-Products and Wastes from Cereal-Based Processing Industry. Foods 2020, 9, 1243. [Google Scholar] [CrossRef]
- Tian, S.; Sun, Y.; Chen, Z.; Zhao, R. Bioavailability and Bioactivity of Alkylresorcinols from Different Cereal Products. J. Food Qual. 2020, 1–6. [Google Scholar] [CrossRef]
- Galanakis, C.M. The Food Systems in the Era of the Coronavirus (COVID-19) Pandemic Crisis. Foods 2020, 9, 523. [Google Scholar] [CrossRef] [PubMed]
- Galanakis, C.M.; Rizou, M.; Aldawoud, T.M.S.; Ucak, I.; Rowan, N.J. Innovations and technology disruptions in the food sector within the COVID-19 pandemic and post-lockdown era. Trends Food Sci. Technol. 2021, 110, 193–200. [Google Scholar] [CrossRef]
- Boyaci-Gunduz, C.P.; Ibrahim, S.A.; Wei, O.C.; Galanakis, C.M. Transformation of the Food Sector: Security and Resilience after the COVID-19 Pandemic. Foods 2021, 10, 497. [Google Scholar] [CrossRef]
- Galanakis, C.M.; Aldawoud, T.M.; Rizou, M.; Rowan, N.J.; Ibrahim, S.A. Food Ingredients and Active Compounds against the Coronavirus Disease (COVID-19) Pandemic: A Comprehensive Review. Foods 2020, 9, 1701. [Google Scholar] [CrossRef] [PubMed]
- Papageorgiou, M.; Skendi, A. Introduction to cereal processing and by-products. In Sustainable Recovery and Reutilization of Cereal Processing By-Products; Galanakis, C.M., Ed.; Elsevier Inc.: Waltham, MA, USA, 2018; pp. 1–25. [Google Scholar]
- Esa, N.M.; Ling, T.B.; Peng, L.S. By-products of Rice Processing: An Overview of Health Benefits and Applications. Rice Res. Open Access 2013, 4. [Google Scholar] [CrossRef] [Green Version]
- Prasad, N.M.N.; Sanjay, K.R.; Shravya Khatokar, M.; Vismaya, M.N.; Nanjunda Swamy, S. Health benefits of rice bran—A review. J. Nutr. Food Sci. 2011, 1, 1–7. [Google Scholar]
- Gul, K.; Yousuf, B.; Singh, A.K.; Singh, P.; Wani, A.A. Rice bran: Nutritional values and its emerging potential for development of functional food—A review. Bioact. Carbohydr. Diet. Fibre 2015, 6, 24–30. [Google Scholar] [CrossRef]
- Rose, D.J.; Inglett, G.E. Production of feruloylated arabinoxylo-oligosaccharides from maize (Zea mays) bran by microwave-assisted autohydrolysis. Food Chem. 2010, 119, 1613–1618. [Google Scholar] [CrossRef]
- Rahmanian, N.; Jafari, S.M.; Galanakis, C.M. Recovery and Removal of Phenolic Compounds from Olive Mill Wastewater. J. Am. Oil Chem. Soc. 2013, 91, 1–18. [Google Scholar] [CrossRef]
- Galanakis, C.M. Concluding remarks and future perspectives. In Sustainable Recovery and Reutilization of Cereal Processing By-Products; Galanakis, C.M., Ed.; Elsevier Inc.: Waltham, MA, USA, 2018; pp. 319–328. [Google Scholar]
- Galanakis, C.M. Phenols recovered from olive mill wastewater as additives in meat products. Trends Food Sci. Technol. 2018, 79, 98–105. [Google Scholar] [CrossRef]
- Galanakis, C.M.; Tsatalas, P.; Galanakis, I.M. Implementation of phenols recovered from olive mill wastewater as UV booster in cosmetics. Ind. Crop. Prod. 2018, 111, 30–37. [Google Scholar] [CrossRef]
- Hernández, N.; Rodriguez-Alegría, M.E.; Gonzalez, F.; Lopez-Munguia, A. Enzymatic treatment of rice bran to improve processing. JAOCS J. Am. Oil Chem. Soc. 2000, 77, 177–180. [Google Scholar] [CrossRef]
- Jiang, Y.; Wang, T. Phytosterols in cereal by-products. JAOCS J. Am. Oil Chem. Soc. 2005, 82, 439–444. [Google Scholar] [CrossRef]
- Moreau, R.A.; Singh, V.; Eckhoff, S.R.; Powell, M.J.; Hicks, K.B.; Norton, R.A. Comparison of Yield and Composition of Oil Extracted from Corn Fiber and Corn Bran. Cereal Chem. 1999, 76, 449–451. [Google Scholar] [CrossRef] [Green Version]
- Carr, T.P.; Weller, C.L.; Schlegel, V.L.; Cuppett, S.L.; Guderian, D.M.; Johnson, K.R. Grain Sorghum Lipid Extract Reduces Cholesterol Absorption and Plasma Non-HDL Cholesterol Concentration in Hamsters. J. Nutr. 2005, 135, 2236–2240. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prückler, M.; Siebenhandl-Ehn, S.; Apprich, S.; Höltinger, S.; Haas, C.; Schmid, E.; Kneifel, W. Wheat bran-based biore-finery 1: Composition of wheat bran and strategies of functionalization. LWT Food Sci. Technol. 2014, 56, 211–221. [Google Scholar] [CrossRef]
- Zangenberg, M.; Hansen, H.B.; Jørgensen, J.R.; Hellgren, L.I. Cultivar and Year-to-Year Variation of Phytosterol Content in Rye (Secale cereale L.). J. Agric. Food Chem. 2004, 52, 2593–2597. [Google Scholar] [CrossRef]
- Moreau, R.A.; Flores, R.A.; Hicks, K.B. Composition of Functional Lipids in Hulled and Hulless Barley in Fractions Obtained by Scarification and in Barley Oil. Cereal Chem. 2007, 84, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Webster, F.H. Whole-grain oats and oat products. In Whole-Grain Foods in Health and Disease; Marquart, L., Slavin, J.L., Fulcher, R.G., Eds.; AACC Press: St. Paul, MN, USA, 2002; pp. 84–119. [Google Scholar]
- Lazaridou, A.; Biliaderis, C.G.; Izydorczyk, M.S. Cereal β-glucan: Structures, physical properties and physiological functions. In Functional Food Carbohydrates; Biliaderis, C.G., Izydorczyk, M.S., Eds.; CRC Press: Boca Raton, FL, USA, 2007; p. 41. [Google Scholar]
- Braaten, J.T.; Scott, F.W.; Wood, P.J.; Riedel, K.D.; Wolynetz, M.S.; Brule, D.; Collins, M.W. High beta-glucan oat bran and oat gum reduce postprandial blood-glucose and insulin in subjects with and without type-2 diabetes. Diabet. Med. 1994, 11, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Andersson, K.E.; Immerstrand, T.; Swärd, K.; Bergenståhl, B.; Lindholm, M.W.; Öste, R.; Hellstrand, P. Effects of oats on plasma cholesterol and lipoproteins in C57BL/6 mice are substrain specific. Br. J. Nutr. 2010, 103, 513–521. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patsioura, A.; Galanakis, C.M.; Gekas, V. Ultrafiltration optimization for the recovery of β-glucan from oat mill waste. J. Membr. Sci. 2011, 373, 53–63. [Google Scholar] [CrossRef]
- Ananey-Obiri, D.; Matthews, L.; Azahrani, M.H.; Ibrahim, S.A.; Galanakis, C.M.; Tahergorabi, R. Application of protein-based edible coatings for fat uptake reduction in deep-fat fried foods with an emphasis on muscle food proteins. Trends Food Sci. Technol. 2018, 80, 167–174. [Google Scholar] [CrossRef]
- Lindahl, L.; Ahldén, I.; Öste, R.; Sjöholm, I. Homogenous and Stable Cereal Suspension and a Method of Making the Same. U.S. Patent 5686123, 11 November 1997. [Google Scholar]
- Galanakis, C.M.; Patsioura, A.; Gekas, V. Enzyme kinetics modeling as a tool to optimize food biotechnology ap-plications: A pragmatic approach based on amylolytic enzymes. Crit. Rev. Food Sci. Technol. 2015, 55, 1758–1770. [Google Scholar] [CrossRef] [PubMed]
- Prado, F.C.; Parada, J.L.; Pandey, A.; Soccol, C.R. Trends in non-dairy probiotic beverages. Food Res. Int. 2008, 41, 111–123. [Google Scholar] [CrossRef]
- Del Re, G.; Di Giacomo, G.; Spera, L.; Vegliò, F. Integrated approach in the biotreatment of starch wastes by Rhizopus oligosporus: Kinetic analysis. Desalination 2003, 156, 389–396. [Google Scholar] [CrossRef]
- Volikakis, P.; Biliaderis, C.G.; Vamvakas, C.; Zerfiridis, G.K. Effect of a commercial oat-β-glucan concentrate on the chemical, physic-chemical and sensory attributes of a low-fat white-brined cheese product. Food Res. Int. 2004, 37, 83–94. [Google Scholar] [CrossRef]
- Domagała, J.; Sady, M.; Grega, T.; Bonczar, G. Rheological Properties and Texture of Yoghurts When Oat-Maltodextrin is Used as a Fat Substitute. Int. J. Food Prop. 2006, 9, 1–11. [Google Scholar] [CrossRef]
- Galanakis, C.M. Separation of functional macromolecules and micromolecules: From ultrafiltration to the border of nanofiltration. Trends Food Sci. Technol. 2015, 42, 44–63. [Google Scholar] [CrossRef]
- Sibakov, J.; Myllymäki, O.; Holopainen, U.; Kaukovirta-Norja, A.; Hietaniemi, V.; Pihlava, J.M.; Poutanen, K.; Lehtinen, P. Lipid removal enhances separation of oat grain cell wall material from starch and protein. J. Cereal Sci. 2011, 54, 104–109. [Google Scholar] [CrossRef]
- Ma, C.-Y.; Rout, M.K.; Chan, W.-M.; Phillips, D.L. Raman Spectroscopic Study of Oat Globulin Conformation. J. Agric. Food Chem. 2000, 48, 1542–1547. [Google Scholar] [CrossRef]
- Ma, C.-Y.; Rout, M.K.; Mock, W.-Y. Study of Oat Globulin Conformation by Fourier Transform Infrared Spectroscopy. J. Agric. Food Chem. 2001, 49, 3328–3334. [Google Scholar] [CrossRef]
- Loponen, J.; Laine, P.; Sontag-Strohm, T.; Salovaara, H. Behaviour of oat globulins in lactic acid fermentation of oat bran. Eur. Food Res. Technol. 2007, 225, 105–110. [Google Scholar] [CrossRef]
- Galanakis, C.M. Functionality of Food Components and Emerging Technologies. Foods 2021, 10, 128. [Google Scholar] [CrossRef]
- Galanakis, C.M. Recovery of high added-value components from food wastes: Conventional, emerging technologies and commercialized applications. Trends Food Sci. Technol. 2012, 26, 68–87. [Google Scholar] [CrossRef]
- Sapirstein, H.D. Bioactive compounds in wheat bran. In Encyclopedia of Food Grains, Second Edition, Vol. 2 Nutrition and Food Grains; Wrigley, C., Corke, H., Seetharaman, K., Faubion, J., Eds.; Elsevier Academic Press: Oxford, UK, 2016; pp. 268–276. [Google Scholar]
- Hell, J.; Kneifel, W.; Rosenau, T.; Böhmdorfer, S. Analytical techniques for the elucidation of wheat bran constituents and their structural features with emphasis on dietary fiber—A review. Trends Food Sci. Technol. 2014, 35, 102–113. [Google Scholar] [CrossRef]
- Aguedo, M.; Fougnies, C.; Dermience, M.; Richel, A. Extraction by three processes of arabinoxylans from wheat bran and characterization of the fractions obtained. Carbohydr. Polym. 2014, 105, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Ma, S.; Fan, L.; Zhang, C.; Pu, X.; Zheng, X.; Wang, X. The influence of ultrasonic modification on arabinoxylans properties obtained from wheat bran. Int. J. Food Sci. Technol. 2016, 51, 2338–2344. [Google Scholar] [CrossRef]
- Onipe, O.O.; Jideani, A.I.O.; Beswa, D. Composition and functionality of wheat bran and its application in some cereal food products. Int. J. Food Sci. Technol. 2015, 50, 2509–2518. [Google Scholar] [CrossRef]
- Kamal-Eldin, A.; Lærke, H.N.; Knudsen, K.E.B.; Lampi, A.M.; Piironen, V.; Adlercreutz, H.; Katina, K.; Poutanen, K.; Åman, P. Physical, microscopic and chemical characterisation of industrial rye and wheat brans from the Nordic countries. Food Nutr. Res. 2009, 53, 1912. [Google Scholar] [CrossRef] [Green Version]
- Thebaudin, J.Y.; Lefebvre, A.C.; Harrington, M.; Bourgeois, C.M. Dietary fibres: Nutritional and technological in-terest. Trends Food Sci. Technol. 1997, 8, 41–48. [Google Scholar] [CrossRef]
- Mehta, N.; Ahlawat, S.S.; Sharma, D.P.; Dabur, R.S. Novel trends in development of dietary fiber rich meat products—A critical review. J. Food Sci. Technol. 2015, 52, 633–647. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kemppainen, K.; Rommi, K.; Holopainen, U.; Kruus, K. Steam explosion of Brewer’s spent grain improves enzymatic digestibility of carbohydrates and affects solubility and stability of proteins. Appl. Biochem. Biotechnol. 2016, 180, 94–108. [Google Scholar] [CrossRef]
- Koirala, P.; Maina, N.H.; Nihtilä, H.; Katina, K.; Coda, R. Brewers’ spent grain as substrate for dextran biosynthesis by Leuconostoc pseudomesenteroides DSM20193 and Weissella confusa A16. Microb. Cell Factories 2021, 20, 1–13. [Google Scholar] [CrossRef]
- Naibaho, J.; Butula, N.; Jonuzi, E.; Korzeniowska, M.; Laaksonen, O.; Foste, M.; Kutt, M.-L.; Yang, B. Potential of brewers’ spent grain in yogurt fermentation and evaluation of its impact in rheological behaviour, consistency, microstructural properties and acidity profile during the refrigerated storage. Food Hydrocoll. 2022, 125, 107412. [Google Scholar] [CrossRef]
- Neylon, E.; Arendt, E.K.; Zannini, E.; Sahin, A.W. Fundamental study of the application of brewers spent grain and fermented brewers spent grain on the quality of pasta. Food Struct. 2021, 30, 100225. [Google Scholar] [CrossRef]
- Neylon, E.; Arendt, E.; Zannini, E.; Sahin, A. Fermentation as a Tool to Revitalise Brewers’ Spent Grain and Elevate Techno-Functional Properties and Nutritional Value in High Fibre Bread. Foods 2021, 10, 1639. [Google Scholar] [CrossRef]
- Ferri, M.; Graen-Heedfeld, J.; Bretz, K.; Guillon, F.; Michelini, E.; Calabretta, M.M.; Lamborghini, M.; Gruarin, N.; Roda, A.; Kraft, A.; et al. Peptide Fractions Obtained from Rice By-Products by Means of an Environment-Friendly Process Show In Vitro Health-Related Bioactivities. PLoS ONE 2017, 12, e0170954. [Google Scholar] [CrossRef] [Green Version]
- Idris, W.H.; Babiker, E.E.; El Tinay, A.H. Fractionation, solubility and functional properties of wheat bran proteins as influenced by pH and/or salt concentration. Food/Nahrung 2003, 47, 425–429. [Google Scholar] [CrossRef] [PubMed]
- Tang, D.S.; Yin, G.M.; He, Y.Z.; Hu, S.Q.; Li, B.; Li, L.; Liang, H.L.; Borthakur, D. Recovery of protein from brewer’s spent grain by ultrafiltration. Biochem. Eng. J. 2009, 48, 1–5. [Google Scholar] [CrossRef]
- Mandalari, G.; Faulds, C.B.; Sancho, A.I.; Saija, A.; Bisignano, G.; Locurto, R.; Waldron, K.W. Fractionation and characterisation of arabinoxylans from brewers’ spent grain and wheat bran. J. Cereal Sci. 2005, 42, 205–212. [Google Scholar] [CrossRef]
- Phongthai, S.; Lim, S.T.; Rawdkuen, S. Optimization of microwave-assisted extraction of rice bran protein and its hydrolysates properties. J. Cereal Sci. 2016, 70, 146–154. [Google Scholar] [CrossRef]
- Castro-Muñoz, R.; Yáñez-Fernández, J. Valorization of Nixtamalization wastewaters (Nejayote) by integrated membrane process. Food Bioprod. Process. 2015, 95, 7–18. [Google Scholar] [CrossRef]
- Celus, I.; Brijs, K.; Delcour, J. The effects of malting and mashing on barley protein extractability. J. Cereal Sci. 2006, 44, 203–211. [Google Scholar] [CrossRef]
- Diptee, R.; Smith, J.P.; Alli, I.; Khanizadeh, S. Application of response surface methodology in protein extraction studies from brewer’s spent grain. J. Food Process. Preserv. 1989, 13, 457–474. [Google Scholar] [CrossRef]
- Zhang, Z.; Smith, C.; Li, W. Extraction and modification technology of arabinoxylans from cereal by-products: A critical review. Food Res. Int. 2014, 65, 423–436. [Google Scholar] [CrossRef]
- Zheng, X.; Li, L.; Wang, X. Molecular Characterization of Arabinoxylans from Hull-Less Barley Milling Fractions. Molecules 2011, 16, 2743–2753. [Google Scholar] [CrossRef]
- Deeslie, W.D.; Cheryan, M. Functional properties of soy protein hydrolyzates from a continuous ultrafiltration reactor. J. Agric. Food Chem. 1988, 36, 26–31. [Google Scholar] [CrossRef]
- McCarthy, A.L.; O’Callaghan, Y.C.; Neugart, S.; Piggott, C.O.; Connolly, A.; Jansen, M.A.K.; Krumbein, A.; Schreiner, M.; FitzGerald, R.J.; O’Brien, N.M. The hydroxycinnamic acid content of barley and brewers’ spent grain (BSG) and the potential to incorporate phenolic extracts of BSG as antioxidants into fruit beverages. Food Chem. 2013, 141, 2567–2574. [Google Scholar] [CrossRef] [PubMed]
- Galanakis, C.M. Emerging technologies for the production of nutraceuticals from agricultural by-products: A viewpoint of opportunities and challenges. Food Bioprod. Process. 2013, 91, 575–579. [Google Scholar] [CrossRef]
- Roselló-Soto, E.; Galanakis, C.M.; Brnčić, M.; Orlien, V.; Trujillo, F.J.; Mawson, R.; Knoerzer, K.; Barba, F.J. Clean recovery of antioxidant compounds from plant foods, by-products and algae assisted by ultrasounds processing. Modeling approaches to optimize processing conditions. Trends Food Sci. Technol. 2015, 42, 134–149. [Google Scholar] [CrossRef]
- Roselló-Soto, E.; Barba, F.J.; Parniakov, O.; Galanakis, C.M.; Grimi, N.; Lebovka, N.; Vorobiev, E. High voltage electrical discharges, pulsed electric field and ultrasounds assisted extraction of protein and phenolic compounds from olive kernel. Food Bioprocess Technol. 2015, 8, 885–894. [Google Scholar] [CrossRef]
- Kovacevic, D.B.; Barba, F.J.; Granato, D.; Galanakis, C.M.; Herceg, Z.; Dragovic-Uzelac, V.; Prutnik, P. Pressurized hot water extraction (PHWE) for the green recovery of bioactive compounds from steviol glycosides from Stevis rebaudiana Bertoni Leaves. Food Chem. 2018, 254, 150–157. [Google Scholar] [CrossRef]
- Nagarajan, J.; Prasad, N.K.; Ramakrishnan, N.R.; Raghunandan, M.E.; Galanakis, C.M.; Wei, O.C. A facile water-induced complexation of lycopene and pectin from pink guava by-product: Extraction, characterization and kinetic studies. Food Chem. 2019, 296, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Sarfarazi, M.; Jafari, S.M.; Rajabzadeh, G.; Galanakis, C. Evaluation of microwave-assisted extraction technology for separation of bioactive components of saffron (Crocus sativus L.). Ind. Crop. Prod. 2020, 145, 111978. [Google Scholar] [CrossRef]
- Barba, F.J.; Galanakis, C.; Esteve, M.J.; Frigola, A.; Vorobiev, E. Potential use of pulsed electric technologies and ultrasounds to improve the recovery of high-added value compounds from blackberries. J. Food Eng. 2015, 167, 38–44. [Google Scholar] [CrossRef]
- Deng, Q.; Zinoviadou, K.G.; Galanakis, C.M.; Orlien, V.; Grimi, N.; Vorobiev, E.; Lebovka, N.; Barba, F.J. The effects of conventional and non-conventional processing on glucosinolates and its derived forms, isothiocyanates: Extraction, degradation and applications. Food Eng. Rev. 2015, 7, 357–381. [Google Scholar] [CrossRef]
- Zinoviadou, K.G.; Galanakis, C.M.; Brnčić, M.; Grimi, N.; Boussetta, N.; Mota, M.J.; Saraiva, J.A.; Patras, A.; Tiwari, B.; Barba, F.J. Fruit juice sonication: Implications on food safety and physicochemical and nutritional properties. Food Res. Int. 2015, 77, 743–752. [Google Scholar] [CrossRef]
- Phongthai, S.; Lim, S.-T.; Rawdkuen, S. Ultrasonic-Assisted Extraction of Rice Bran Protein Using Response Surface Methodology. J. Food Biochem. 2016, 41, e12314. [Google Scholar] [CrossRef]
- Delolitte. Biomass Golden Age. 2014. Available online: https://www2.deloitte.com/tr/tr/pages/energy-and-resources/articles/golden-age-of-biomass-article.html (accessed on 3 November 2016).
- Kwiatkowski, J.R.; McAloon, A.J.; Taylor, F.; Johnston, D.B. Modeling the process and costs of fuel ethanol production by the corn dry-grind process. Ind. Crop. Prod. 2006, 23, 288–296. [Google Scholar] [CrossRef]
- Dien, B.S.; Bothast, R.J.; Nichols, N.N.; Cotta, M.A. The US corn ethanol industry: An overview of current technology and future prospects. Int. Sugar J. 2002, 103, 204–208. [Google Scholar]
- Martin, M.A. First generation biofuels compete. New Biotechnol. 2010, 27, 596–608. [Google Scholar] [CrossRef] [PubMed]
- Chatzifragkou, A.; Kosik, O.; Prabhakumari, P.C.; Lovegrove, A.; Frazier, R.A.; Shewry, P.R.; Charalampopoulos, D. Biorefinery strategies for upgrading Distillers’ Dried Grains with Solubles (DDGS). Process Biochem. 2015, 50, 2194–2207. [Google Scholar] [CrossRef]
- Belyea, R.L.; Rausch, K.D.; Clevenger, T.E.; Singh, V.; Johnston, D.B.; Tumbleson, M.E. Sources of variation in composition of DDGS. Anim. Feed Sci. Technol. 2010, 159, 122–130. [Google Scholar] [CrossRef]
- Böttger, C.; Südekum, K.-H. Within plant variation of distillers dried grains with solubles (DDGS) produced from multiple raw materials in varying proportions: Chemical composition and in vitro evaluation of feeding value for ruminants. Anim. Feed Sci. Technol. 2017, 229, 79–90. [Google Scholar] [CrossRef]
- Singh, V.; Eckhoff, S.R. Economics of Germ Preseparation for Dry-Grind Ethanol Facilities. Cereal Chem. 1997, 74, 462–466. [Google Scholar] [CrossRef]
- Hughes, S.A.; Shewry, P.R.; Li, L.; Gibson, G.R.; Sanz, M.L.; Rastall, R.A. In vitro fermentation by human fecal microflora of wheat arabinoxylans. J. Agric. Food Chem. 2007, 55, 4589–4595. [Google Scholar] [CrossRef]
- Abecassis, J.; de Vries, H.; Rouau, X. New perspective for biorefining cereals. Biofuels Bioprod. Biorefining 2013, 8, 462–474. [Google Scholar] [CrossRef]
- Harris, P.J.; Chavan, R.R.; Ferguson, L.R. Production and characterisation of two wheat-bran fractions: An aleurone-rich and a pericarp-rich fraction. Mol. Nutr. Food Res. 2005, 49, 536–545. [Google Scholar] [CrossRef]
- Apprich, S.; Tirpanalan, Ö.; Hell, J.; Reisinger, M.; Böhmdorfer, S.; Siebenhandl-Ehn, S.; Novalin, S.; Kneifel, W. Wheat bran-based biorefinery 2: Valorization of products. LWT Food Sci. Technol. 2014, 56, 222–231. [Google Scholar] [CrossRef]
- Benamrouche, S.; Crônier, D.; Debeire, P.; Chabbert, B. A Chemical and Histological Study on the Effect of (1→4)-β-endo-xylanase Treatment on Wheat Bran. J. Cereal Sci. 2002, 36, 253–260. [Google Scholar] [CrossRef]
- Reisinger, M.; Tirpanalan, Ö.; Huber, F.; Kneifel, W.; Novalin, S. Investigations on a wheat bran biorefinery involving organosolv fractionation and enzymatic treatment. Bioresour. Technol. 2014, 170, 53–61. [Google Scholar] [CrossRef]
- Kumar, A.; Gautam, A.; Dutt, D. Biotechnological Transformation of Lignocellulosic Biomass in to Industrial Products: An Overview. Adv. Biosci. Biotechnol. 2016, 7, 149–168. [Google Scholar] [CrossRef] [Green Version]
- Ravindran, R.; Jaiswal, A.K. Microbial Enzyme Production Using Lignocellulosic Food Industry Wastes as Feedstock: A Review. Bioengineering 2016, 3, 30. [Google Scholar] [CrossRef] [Green Version]
- Mussatto, S.; Dragone, G.; Roberto, I. Brewers’ spent grain: Generation, characteristics and potential applications. J. Cereal Sci. 2006, 43, 1–14. [Google Scholar] [CrossRef]
- Aliyu, S.; Bala, M. Brewer’s spent grain: A review of its potentials and applications. Afr. J. Biotechnol. 2011, 10, 324–331. [Google Scholar]
- Xiros, C.; Christakopoulos, P. Biotechnological Potential of Brewers Spent Grain and its Recent Applications. Waste Biomass Valorization 2012, 3, 213–232. [Google Scholar] [CrossRef]
- Nigam, P.S. An overview: Recycling of solid barley waste generated as a by-product in distillery and brewery. Waste Manag. 2017, 62, 255–261. [Google Scholar] [CrossRef]
- Gomathi, D.; Muthulakshmi, C.; Kumar, D.; Ravikumar, G.; Kalaiselvi, M.; Uma, C. Submerged fermentation of wheat bran by Apergillus flavus for production and characterization of carboxy methyl cellulose. Asian Pac. J. Trop. Biomed. 2012, 2, S67–S73. [Google Scholar] [CrossRef]
- Hashemi, M.; Hadi Razavi, S.; Abbas Shojaosadati, S.; Mohammed Mousavi, S. The potential of brewer’s spent grain to improve the production of α-amylase by Bacillus sp. Kr-8104 in submerged fermentation system. New Biotechnol. 2011, 28, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Sanghi, A.; Garg, N.; Sharma, J.; Kuhar, K.; Kuhad, R.; Gupta, V. Optimization of xylanase production using inexpensive agro-residues by alkalophilic Bacillus subtilis ASH in sold-state fermentation. World J. Microbiol. Biotechnol. 2008, 24, 633–640. [Google Scholar] [CrossRef]
- Kapoor, M.; Nair, L.M.; Kuhad, R.C. Cost-effective xylanase production from free and immobilized Bacillus pumilus strain MK001 and its application in saccharification of Prosopis juliflora. Biochem. Eng. J. 2008, 38, 88–97. [Google Scholar] [CrossRef]
- Tanasković, S.J.; Šekuljic, N.; Jovanović, J.; Gazikalović, I.; Grbavčić, S.; Đorđević, N.; Vukašinović Sekulića, M.; Hao, J.; Lu-kovića, N.; Knežević-Jugovića, Z. Upgrading of valuable food component contents and anti-nutritional factors depletion by solid-state fermentation: A way to valorize wheat bran for nutrition. J. Cereal Sci. 2021, 99, 103159. [Google Scholar] [CrossRef]
- Heiniö, R.-L.; Liukkonen, K.-H.; Myllymäki, O.; Pihlava, J.-M.; Adlercreutz, H.; Heinonen, S.-M.; Poutanen, K. Quantities of phenolic compounds and their impacts on the perceived flavour attributes of rye grain. J. Cereal Sci. 2008, 47, 566–575. [Google Scholar] [CrossRef]
- Heiniö, R.L.; Noort, M.W.J.; Katina, K.; Alam, S.A.; Sozer, N.; de Kock, H.L.; Hersleth, M.; Poutanen, K. Sensory characteristics of wholegrain and bran-rich cereal foods—A review. Trends Food Sci. Technol. 2016, 47, 25–38. [Google Scholar] [CrossRef] [Green Version]
- Hemdane, S.; Jacobs, P.J.; Bosmans, G.M.; Verspreet, J.; Delcour, J.; Courtin, C.M. Study on the effects of wheat bran incorporation on water mobility and biopolymer behavior during bread making and storage using time-domain 1 H NMR relaxometry. Food Chem. 2017, 236, 76–86. [Google Scholar] [CrossRef] [PubMed]
- Coda, R.; Kärki, I.; Nordlund, E.; Heiniö, R.-L.; Poutanen, K.; Katina, K. Influence of particle size on bioprocess induced changes on technological functionality of wheat bran. Food Microbiol. 2014, 37, 69–77. [Google Scholar] [CrossRef]
- Le Bleis, F.; Chaunier, L.; Chiron, H.; Della Valle, G.; Saulnier, L. Rheological properties of wheat flour dough and French bread enriched with wheat bran. J. Cereal Sci. 2015, 65, 167–174. [Google Scholar] [CrossRef]
- Boita, E.R.F.; Oro, T.; Bressiani, J.; Santetti, G.S.; Bertolin, T.E.; Gutkoski, L.C. Rheological properties of wheat flour dough and pan bread with wheat bran. J. Cereal Sci. 2016, 71, 177–182. [Google Scholar] [CrossRef]
- Sanz Penella, J.M.; Collar, C.; Haros, M. Effect of wheat bran and enzyme addition on dough functional performance and phytic acid levels in bread. J. Cereal Sci. 2008, 48, 715–721. [Google Scholar] [CrossRef]
- Nascimento, T.A.; Calado, V.; Carvalho, C.W. Effect of brewer’s spent grain and temperature on physical properties of expanded extrudates from rice. LWT Food Sci. Technol. 2017, 79, 145–151. [Google Scholar] [CrossRef]
- Stojceska, V.; Ainsworth, P.; Plunkett, A.; İbanogˇlu, S. The recycling of brewer’s processing by-product into ready-to-eat snacks using extrusion technology. J. Cereal Sci. 2008, 47, 469–479. [Google Scholar] [CrossRef]
- Robin, F.; Dubois, C.; Pineau, N.; Schuchmann, H.P.; Palzer, S. Expansion mechanism of extruded foams supplemented with wheat bran. J. Food Eng. 2011, 107, 80–89. [Google Scholar] [CrossRef]
- Santala, O.; Kiran, A.; Sozer, N.; Poutanen, K.; Nordlund, E. Enzymatic modification and particle size reduction of wheat bran improves the mechanical properties and structure of bran-enriched expanded extrudates. J. Cereal Sci. 2014, 60, 448–456. [Google Scholar] [CrossRef]
- Laureati, M.; Conte, A.; Padalino, L.; Del Nobile, M.A.; Pagliarini, E. Effect of fiber information on consumer’s expectation and liking of wheat bran enriched pasta. J. Sens. Stud. 2016, 31, 348–359. [Google Scholar] [CrossRef]
- Aravind, N.; Sissons, M.; Egan, N.; Fellows, C. Effect of insoluble dietary fibre addition on technological, sensory, and structural properties of durum wheat spaghetti. Food Chem. 2012, 130, 299–309. [Google Scholar] [CrossRef]
- Padalino, L.; Mastromatteo, M.; Sepielli, G.; Del Nobile, M.A. Formulation optimization of gluten-free functional spaghetti based on maize flour and oat bran enriched in β-glucans. Materials 2011, 4, 2119–2135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shao, M.; Wang, J.; Zhang, Y.; Yu, Q. Exploitation and utilization of rice wine lees. China Brew. 2011, 9, 4. [Google Scholar]
- Ertaş, N. Effect of wheat bran stabilization methods on nutritional and physico-mechanical characteristics of cookies. J. Food Qual. 2015, 38, 184–191. [Google Scholar] [CrossRef]

| Processes | Dry Milling | Wet Milling | ||||
|---|---|---|---|---|---|---|
| Corn | Paddy Rice | Wheat | Oat | Corn | Barley | |
| Cleaning | Yes | Yes | Yes | |||
| Steeping | Yes | Yes | ||||
| Conditioning | Yes | Yes | ||||
| Evaporation | Yes | |||||
| Germination | Yes | |||||
| Processing/Breaking | Yes | Yes | ||||
| Coarse grinding | Yes, germ separation | |||||
| Degermination | Yes, germ separation | |||||
| Drying/Dehusking | Yes | Yes, husk separation, delivering brown rice | Yes, root separation, delivering malt | |||
| Aspiration | Yes | |||||
| Grading | Yes | |||||
| Sieving | Yes, twice | Yes | ||||
| Polishing | Yes, wastewater removal | |||||
| Oil extraction | Yes | |||||
| Destoning | Yes | |||||
| Grinding | Yes | Yes, delivering white rice | Yes, delivering flour | Yes, bran separation, delivering flour | Yes, bran separation | |
| Starch washing | Yes, starch separation | |||||
| Cereal | By-Product | Target Functional Compounds | Health Properties | Examples |
|---|---|---|---|---|
| Rice | Bran, husk | Vitamins, proteins, dietary fiber, oil | Vitamins possess antioxidant activities, proteins show hypoallergenic properties, fiber prevents cardiovascular diseases | Rice bran contains 179–389 mg tocopherols and tocotrienols (Vitamin E compounds)/kg [5] |
| Corn | Bran, germ | Oil, insoluble dietary fiber | Oil reduces cholesterol levels, fiber prevents cardiovascular diseases | Corn kernels, bran and fiber contain 98–113 mg, 10.4–15.3 mg and 38–84 mg of ferulate phytosterol esters/kg, respectively [23] |
| Sorghum and millet | Bran | Phenolic compounds, phytosterols and policosanons | Phenolic compounds possess antioxidant properties, phytosterols and policosanons reduce cholesterol levels | Due to the high policosanol content, sorghum dry distiller grain hexane extracts significantly reduced cholesterol absorption by up to 17% and non-HDL plasma cholesterol by up to 70% in animal models [24] |
| Oat | Bran, oat mill waste | Soluble dietary fiber, β-glucan | Dietary fiber contributes to an increase in fecal bulk, β-glucan has been shown to reduce blood cholesterol | Oat bran contains at least 5.5% of β-glucan per dry mater and a total dietary fiber content of 16.0% dry matter [5] |
| Wheat | Bran, germ | Fiber, arabinoxylans | Bran fiber contributes to an acceleration of intestinal transit, and an increase in fecal bulk. Arabinoxylans contribute to a reduction of glucose level in the blood | Arabinoxylans are accounting for 10.9–26.0% of dry matter of bran. Health benefits of arabinoxylans are attributed to their prebiotic effects for obesity and other metabolic malfunctions, and ability to lower blood cholesterol and the post-prandial glycemic response [25] |
| Rye | Bran | Fiber, arabinoxylans, phytosterols | Dietary fiber contributes to normal bowel function. Arabinoxylans contribute to a reduction of glucose level in the blood | Phytosterol content in rye is 700–100 µg/g [26]. Daily doses of 1–3 g of plant sterols can reduce blood cholesterol in humans [5] |
| Barley | Spent grains | Dietary fiber, β-glucan | β-glucan contributes to the reduction of the blood glucose rise after meal, dietary fiber reduces cholesterol levels | Total phytosterols in barley oils (0.18–1.44 g/15 g oil) are able to significantly lower low-density lipoprotein (LDL) cholesterol at reasonable dosages of 15 mL/d (1 tablespoon/d) [27] |
| Cereal Processing By-Product | Target Compound | Recovery Method | Results | References |
|---|---|---|---|---|
| Oat mill waste | β-Glucan | Polysulfone membrane was applied in the pilot cross-flow module for the ultrafiltration of β-glucan containing feeds (<600 mg/L) recovered from the industrial oat mill waste | Two thirds (~67%) of β-glucan had been recovered | [32] |
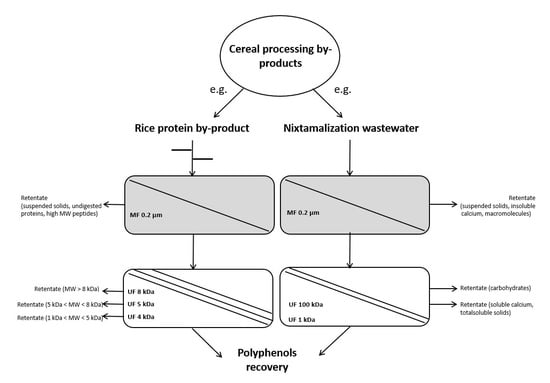
| Rice by-products | Peptides | Combined treatment with proteases and sequential fractionation with one microfiltration (0.2 μm) and three polyethersulfone ultrafiltration (8, 5, and 4 kDa, respectively) steps | The retentate with the highest peptide content was the one recovered with the 8 kDa membrane | [60] |
| Wheat bran | Proteins | The adherent endosperm was removed from wheat bran with brushing before being extracted using the alkaline treatment and isoelectric precipitation | High nitrogen dispensability with minimum and maximum protein solubility pH values of 5.5 and 11.5, respectively. | [61] |
| Barley spent grain | Proteins | Coupling ultrasonic-assisted extraction with ultrafiltration | Recovery yield of 146 mg/g under the optimum conditions (solid-to-liquid ratio of 2 g/100 mL, intensity of 88.2 W/100 mL of extractant, and application of a 30-kDa membrane | [62] |
| Βrewers’ spent grains (BSG) | Arabinoxylans | Sequential fractionation using KOH of increasing strength (0.5, 1.0, and 4.0 M) of growing strength and 50 mM Na2CO3 | Extraction of arabinoxylans with a lower ratio of arabinose/xylose as a factor of alkali’s power. | [63] |
| Rice bran | Proteins | Coupling alkaline extraction with microwave-assisted extraction | The optimum conditions were found to be a power of 1000 W, a water solid-to-liquid ratio of 0.89 g bran/10 mL, and an extraction time of 90 s. Microwaves enhanced by 1.5 fold the protein yield of alkaline extraction. | [64] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Galanakis, C.M. Sustainable Applications for the Valorization of Cereal Processing By-Products. Foods 2022, 11, 241. https://doi.org/10.3390/foods11020241
Galanakis CM. Sustainable Applications for the Valorization of Cereal Processing By-Products. Foods. 2022; 11(2):241. https://doi.org/10.3390/foods11020241
Chicago/Turabian StyleGalanakis, Charis M. 2022. "Sustainable Applications for the Valorization of Cereal Processing By-Products" Foods 11, no. 2: 241. https://doi.org/10.3390/foods11020241
APA StyleGalanakis, C. M. (2022). Sustainable Applications for the Valorization of Cereal Processing By-Products. Foods, 11(2), 241. https://doi.org/10.3390/foods11020241