Dandelion (Taraxacum officinale L.) as a Source of Biologically Active Compounds Supporting the Therapy of Co-Existing Diseases in Metabolic Syndrome
Abstract
1. Introduction
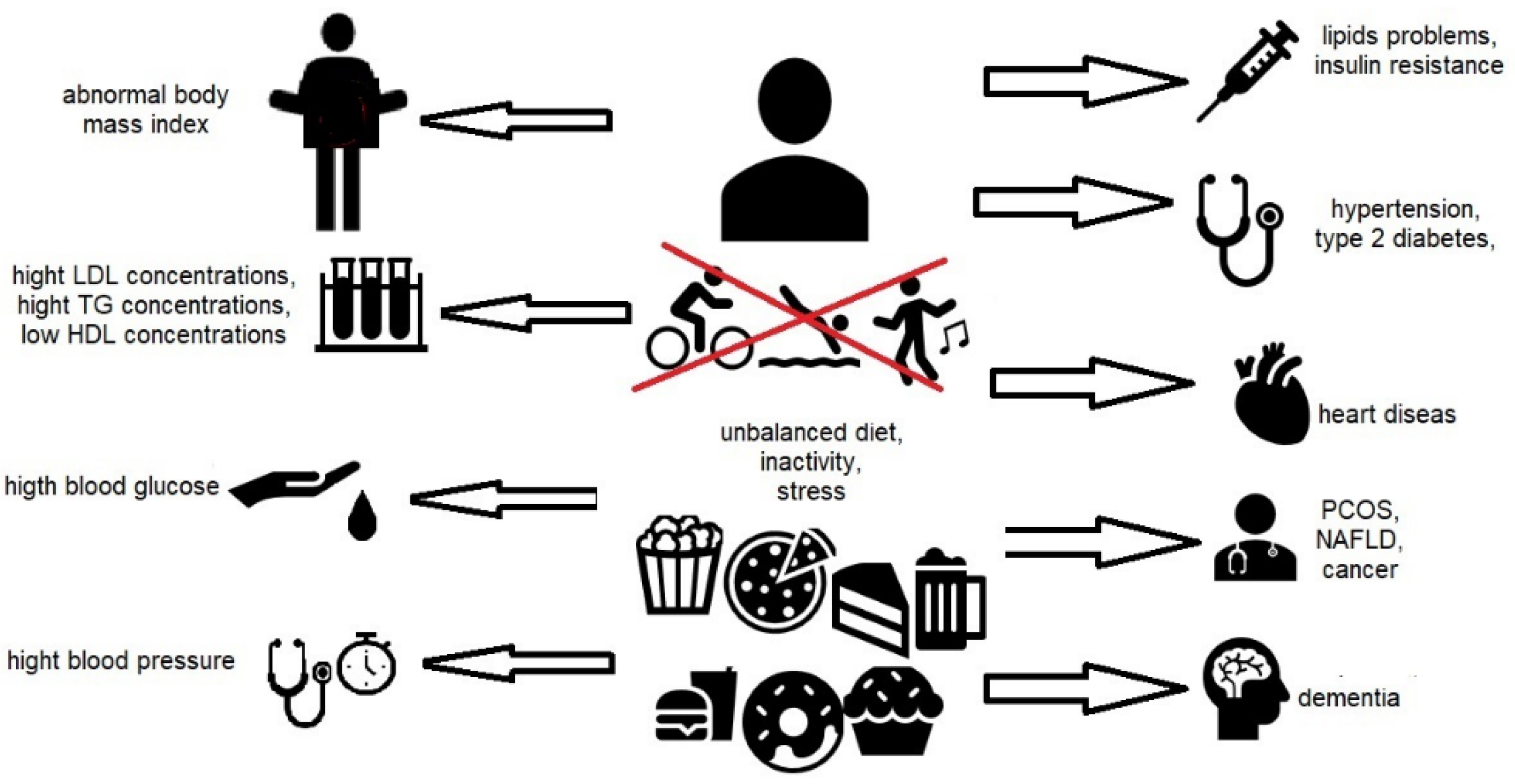
2. Definition and Etiology of Metabolic Syndrome (MetS)
3. Lifestyle vs. Metabolic Syndrome
4. Dandelion—Plant Characteristics
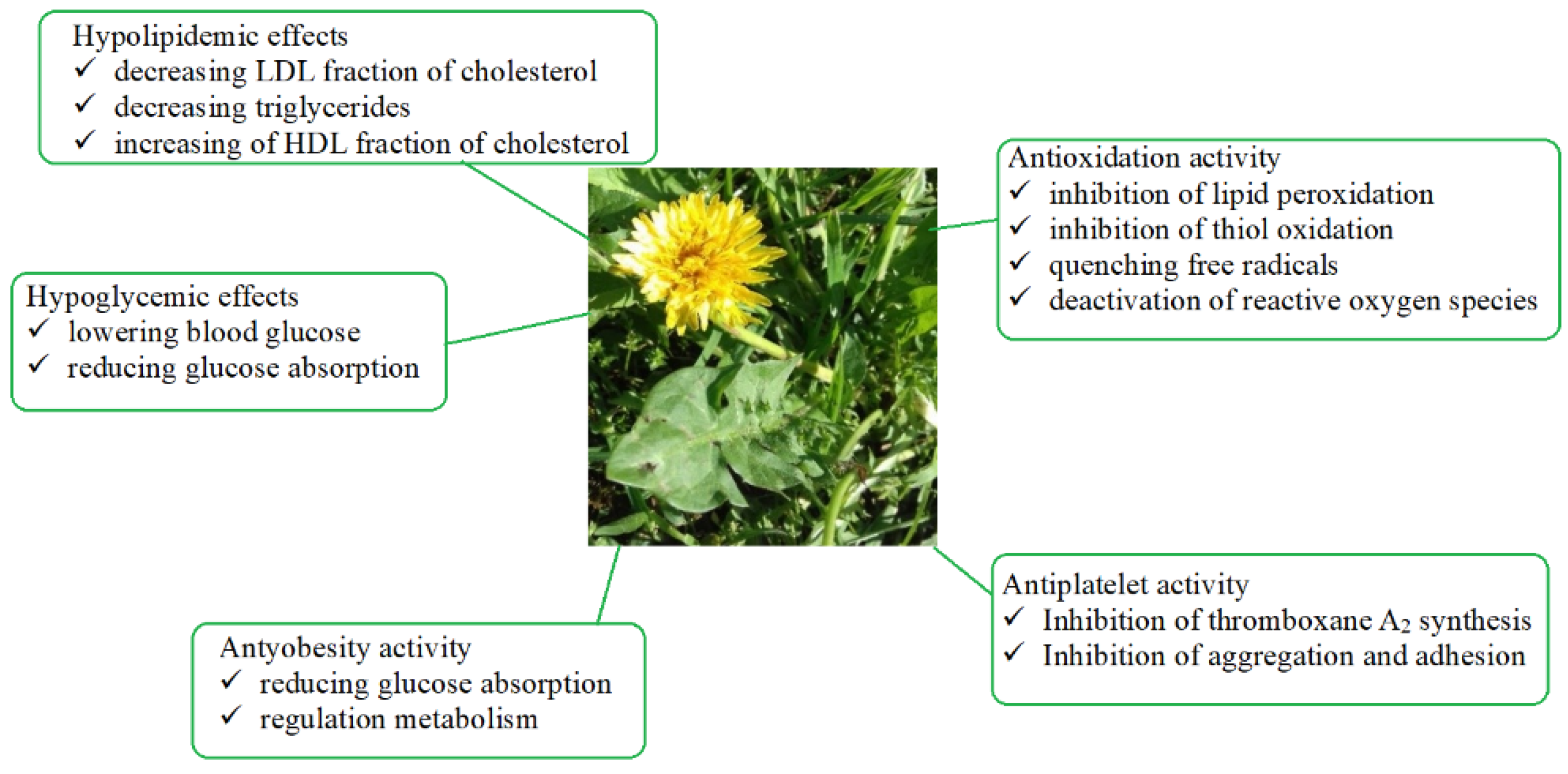
5. Pharmacological Activity of Dandelion for Potential Use in the Treatment of Metabolic Syndrome (MetS)
5.1. Antidiabetic Effect
5.2. Impact on Lipid Profile
5.3. Impact on Blood Pressure
5.4. Effects on Blood Coagulation
5.5. Dandelion vs. Obesity
6. Reports on the Toxic Effects of Dandelion and its Preparation
7. Final Remarks
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Alberti, K.G.; Zimmet, P.Z. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: Diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet. Med. 1998, 15, 539–553. [Google Scholar] [CrossRef]
- Alberti, K.G.; Zimmet, P.Z.; Shaw, J.; Grundy, S.M. International Diabetes Federation 2006: The IDF Consensus Worldwide Definition of the Metabolic Syndrome. Available online: http://www.idf.org/webdata/docs/IDF_Meta_def_final.pdf (accessed on 10 March 2016).
- Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive Summary of the Third Report of the National Cholesterol Education Program (NCEP) expert Panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III). JAMA 2001, 285, 2486–2497. [Google Scholar] [CrossRef] [PubMed]
- Alberti, K.G.; Zimmet, P.Z.; Shaw, J. Metabolic syndrome—A new world-wide definition. A Consensus Statement from the International Diabetes Federation. Diabet. Med. 2006, 23, 469–480. [Google Scholar] [CrossRef] [PubMed]
- Alberti, K.G.; Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z.; Cleeman, J.I.; Donato, K.A.; Fruchart, J.C.; James, W.P.; Loria, C.M.; Smith, S.C., Jr. Harmonizing the Metabolic Syndrome. Circulation 2009, 120, 1640–1645. [Google Scholar] [CrossRef]
- Grundy, S.M.; Cleeman, J.I.; Daniels, S.R.; Donato, K.A.; Eckel, R.H.; Franklin, B.A.; Gordon, D.J.; Krauss, R.M.; Savage, P.J.; Smith, S.C., Jr.; et al. Diagnosis and management of the metabolic syndrome: An American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation 2005, 112, 2735–2752. [Google Scholar] [CrossRef]
- Mamcarz, A.; Kopeć, G.; Podolec, J.; Godycki-Ćwirko, M.; Jankowski, P.; Knap, K.; Kozek, E.; Olszowska, M.; Pająk, A.; Czarnecka, D.; et al. Wytyczne Polskiego Forum Profilaktyki Chorób Układu Krążenia Dotyczące Zespołu Metabolicznego. Available online: http://polskieforumprofilaktyki.edu.pl/download/forum8.pdf (accessed on 3 September 2007).
- Knowler, W.C.; Barrett-Connor, E.; Fowler, S.E.; Hamman, R.F.; Lachin, J.M.; Walker, E.A.; Nathan, D.M.; Diabetes Prevention Program Research Group. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N. Engl. J. Med. 2002, 346, 393–403. [Google Scholar] [CrossRef]
- Yamaoka, K.; Tango, T. Effects of lifestyle modification on metabolic syndrome: A systematic review and meta-analysis. BMC Med. 2012, 10, 138. [Google Scholar] [CrossRef]
- Saboya, P.P.; Bodanese, L.C.; Zimmermann, P.R.; da Silva Gustavo, A.; Macagnan, F.E.; Feoli, A.P.; da Silva Oliveira, M. Lifestyle Intervention on Metabolic Syndrome and its Impact on Quality of Life: A Randomized Controlled Trial. Arq. Bras. Cardiol. 2017, 108, 60–69. [Google Scholar] [CrossRef]
- VanWormer, J.J.; Boucher, J.L.; Sidebottom, A.C.; Sillah, A.; Knickelbine, T. Lifestyle changes and prevention of metabolic syndrome in the Heart of New Ulm Project. Prev. Med. Rep. 2017, 6, 242–245. [Google Scholar] [CrossRef]
- Buscemi, S.; Sprini, D.; Grosso, G.; Galvano, F.; Nicolucci, A.; Lucisano, G.; Massenti, F.M.; Amodio, E.; Rini, G.B. Impact of lifestyle on metabolic syndrome in apparently healthy people. Eat. Weight Disord. 2014, 19, 225–232. [Google Scholar] [CrossRef]
- Kania, M.; Derebecka, N. Medicinal plants in type 2 diabetes mellitus. Postępy Fitoter. 2010, 2, 76–84. [Google Scholar]
- Jalili, C.; Taghadosi, M.; Pazhouhi, M.; Bahrehmand, F.; Miraghaee, S.S.; Pourmand, D.; Rashidi, I. An overview of therapeutic potentials of Taraxacum officinale (dandelion): A traditionally valuable herb with a reach historical background. WCRJ World Cancer Res. J. 2020, 7, e1679. [Google Scholar] [CrossRef]
- Esiyok, D.; Ötles, S.; Akcicek, E. Herbs as a food source in Turkey. Asian Pac. J. Cancer Prev. 2004, 5, 334–339. [Google Scholar]
- Biel, W.; Jaroszewska, A.; Łysoń, E.; Telesiński, A. The chemical composition and antioxidant properties of common dandelion leaves compared with sea buckthorn. Can. J. Plant Sci. 2017, 97, 1165–1174. [Google Scholar] [CrossRef]
- Gonzalez-Castejon, M.; Visioli, F.; Rodriguez-Casado, A. Diverse biological activities of dandelion. Nutr. Rev. 2012, 70, 534–547. [Google Scholar] [CrossRef]
- Kohlmünzer, S. Farmakognozja; PZWL: Warszawa, Poland, 1993; Volume 76, pp. 321–323. [Google Scholar]
- Wirngo, F.E.; Lambert, M.N.; Jeppesen, P.B. The Physiological Effects of Dandelion (Taraxacum officinale) in Type 2 Diabetes. Rev. Diabet. Stud. 2016, 13, 113–131. [Google Scholar] [CrossRef]
- Williams, C.A.; Goldstone, F.; Greenham, J. Flavonoids, cinnamic acids and coumarins from the different tissues and medicinal preparations of Taraxacum officinale. Phytochemistry 1996, 42, 121–127. [Google Scholar] [CrossRef]
- Schütz, K.; Kammerer, D.R.; Carle, R.; Schieber, A. Characterization of phenolic acids and flavonoids in dandelion (Taraxacum officinale WEB. ex WIGG.) root and herb by high-performance liquid chromatography/electrospray ionization mass spectrometry. Rapid Commun. Mass Spectrom. 2005, 19, 179–186. [Google Scholar] [CrossRef]
- Jędrejek, D.; Kontek, B.; Lis, B.; Stochmal, A.; Olas, B. Evaluation of antioxidant activity of phenolic fractions from the leaves and petals of dandelion in human plasma treated with H2O2 and H2O2/Fe. Chem.-Biol. Interact. 2017, 262, 29–37. [Google Scholar] [CrossRef]
- Hu, C.; Kitts, D.D. Luteolin and luteolin-7-O-glucoside from dandelion flower suppress iNOS and COX-2 in RAW264.7 cells. Mol. Cell. Biochem. 2004, 265, 107–113. [Google Scholar] [CrossRef]
- Tsai, K.; Kao, C.; Hung, C.; Cheng, Y.; Lin, H.; Chu, P. Chicoric acid is a potent anti-atherosclerotic ingredient by anti-oxidant action and anti-inflammation capacity. Oncotarget 2017, 8, 29600–29612. [Google Scholar] [CrossRef]
- Olas, B. New Perspectives on the Effect of Dandelion, Its Food Products and Other Preparations on the Cardiovascular System and Its Diseases. Nutrients 2022, 14, 1350. [Google Scholar] [CrossRef]
- Hook, I.; McGee, A.; Henman, M. Evaluation of dandelion for diuretic activity and variation in potassium content. Int J. Pharmacog. 1993, 31, 29–34. [Google Scholar] [CrossRef]
- Petlevski, R.; Hadžija, M.; Slijepčević, M.; Juretić, D.; Petrik, J. Glutathione S-transferase and malondialdehyde in the liver of NOD mice on short-term treatment with plant mixture extract P-980191. Phytother. Res. 2003, 17, 311–314. [Google Scholar] [CrossRef]
- Zhang, X.; Xiong, H.; Liu, L. Effects of taraxasterol on inflammatory responses in lipopolysaccharide-induced RAW 264.7 macrophages. J. Ethnopharmacol. 2012, 141, 206–211. [Google Scholar] [CrossRef]
- Yang, F.; Ye, X.J.; Chen, M.Y.; Li, H.C.; Wang, Y.F.; Zhong, M.Y.; Zhong, C.S.; Zeng, B.; Xu, L.H.; He, X.H.; et al. Inhibition of NLRP3 Inflammasome Activation and Pyroptosis in Macrophages by Taraxasterol Is Associated With Its Regulation on mTOR Signaling. Front. Immunol. 2021, 17, 632606. [Google Scholar] [CrossRef]
- Yang, Y.; Ying, G.; Wu, S.; Wu, F.; Chen, Z. In vitro inhibition effects of hepatitis B virus by dandelion and taraxasterol. Infect. Agents Cancer 2020, 15, 44. [Google Scholar] [CrossRef]
- Hagymasi, K.; Blazovics, A.; Feher, J.; Lugasi, A.; Kristo, S.T.; Kery, A. The in vitro effect of dandelions antioxidants on microsomal lipid peroxidation. Phytother. Res. 2000, 14, 43–44. [Google Scholar] [CrossRef]
- Tousch, D.; Lajoix, A.D.; Hosy, E.; Azay-Milhau, J.; Ferrare, K.; Jahannault, C.; Cros, G.; Petit, P. Chicoric acid, a new compound able to enhance insulin release and glucose uptake. Biochem. Biophys. Res. Commun. 2008, 377, 131–135. [Google Scholar] [CrossRef]
- Majewski, M.; Lis, B.; Juśkiewicz, J.; Ognik, K.; Borkowska-Sztachańska, M.; Jedrejek, D.; Stochmal, A.; Olas, B. Phenolic Fractions from Dandelion Leaves and Petals as Modulators of the Antioxidant Status and Lipid Profile in an In Vivo Study. Antioxidants 2020, 9, 131. [Google Scholar] [CrossRef]
- Esatbeyoglu, T.; Obermair, B.; Dorn, T.; Siems, K.; Rimbach, G.; Birringer, M. Sesquiterpene Lactone Composition and Cellular Nrf2 Induction of Taraxacum officinale Leaves and Roots and Taraxinic Acid β-d-Glucopyranosyl Ester. J. Med. Food 2017, 20, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Kenny, O.; Smyth, T.J.; Hewage, C.M.; Brunton, N.P. Antioxidant properties and quantitative UPLC-MS/MS analysis of phenolic compounds in dandelion (Taraxacum officinale) root extracts. Free. Radic. Antioxid. 2014, 4, 55–61. [Google Scholar] [CrossRef]
- Miao, M.; Xiang, L. Chapter Three—Pharmacological action and potential targets of chlorogenic acid. In Advances in Pharmacology; Guanhua, D., Ed.; Academic Press: London, OH, USA, 2020; Volume 87, pp. 71–88. [Google Scholar] [CrossRef]
- Lis, B.; Jedrejek, D.; Rywaniak, J.; Soluch, A.; Stochmal, A.; Olas, B. Flavonoid Preparations from Taraxacum officinale L. Fruits—A Phytochemical, Antioxidant and Hemostasis Studies. Molecules 2020, 25, 5402. [Google Scholar] [CrossRef]
- Park, C.M.; Park, J.Y.; Song, Y.S. Luteolin and chicoric acid, two major constituents of dandelion leaf, inhibit nitric oxide and lipid peroxide formation in lipopolysaccharide-stimulated RAW 264.7 cells. Prev. Nutr. Food Sci. 2010, 15, 92–97. [Google Scholar] [CrossRef][Green Version]
- Chambers, E.S.; Byrne, C.S.; Morrison, D.J.; Murphy, K.G.; Preston, T.; Tedford, C.; Garcia-Perez, I.; Fountana, S.; Serrano-Contreras, J.I.; Holmes, E.; et al. Dietary supplementation with inulin-propionate ester or inulin improves insulin sensitivity in adults with overweight and obesity with distinct effects on the gut microbiota, plasma metabolome and systemic inflammatory responses: A randomised cross-over trial. Gut 2019, 68, 1430–1438. [Google Scholar] [CrossRef] [PubMed]
- Juee, L.Y.M.; Naqishbandi, A.M. In vivo and in vitro antidiabetic potential of Taraxacum officinale root extracts. Curr. Issues Pharm. Med. Sci. 2020, 33, 168–175. [Google Scholar] [CrossRef]
- Sekhon-Loodu, S.; Rupasinghe, H.P.V. Evaluation of Antioxidant, Antidiabetic and Antiobesity Potential of Selected Traditional Medicinal Plants. Front. Nutr. 2019, 6, 53. [Google Scholar] [CrossRef]
- Hussain, Z.; Waheed, A.; Qureshi, R.A.; Burdi, D.K.; Verspohl, E.J.; Khan, N.; Hasan, M. The effect of medicinal plants of Islamabad and Murree region of Pakistan on insulin secretion from INS-1 cells. Phytother Res. 2004, 18, 73–77. [Google Scholar] [CrossRef]
- Önal, S.; Timur, S.; Okutucu, B.; Zihnioğlu, F. Inhibition of -glucosidase by aqueous extract of some potent antidiabetic medicinal herbs. Prep. Biochem. Biotechnol. 2005, 35, 29–36. [Google Scholar] [CrossRef]
- Petlevski, R.; Hadžija, M.; Slijepčević, M.; Juretić, D. Effect of ‘antidiabetis’ herbal preparation on serum glucose and fructosamine in NOD mice. J. Ethnopharmacol. 2001, 75, 181–184. [Google Scholar] [CrossRef]
- Cho, S.Y.; Park, J.Y.; Park, E.M.; Choi, M.S.; Lee, M.K.; Jeon, S.M.; Jang, M.K.; Kim, M.J.; Park, Y.B. Alternation of hepatic antioxidant enzyme activities and lipid profile in streptozotocin-induced diabetic rats by supplementation of dandelion water extract. Clin. Chim. Acta 2002, 317, 109–117. [Google Scholar] [CrossRef]
- Villiger, A.; Sala, F.; Suter, A.; Butterweck, V. In vitro inhibitory potential of Cynara scolymus, Silybum marianum, Taraxacum officinale, and Peumus boldus on key enzymes relevant to metabolic syndrome. Phytomedicine 2015, 22, 138–144. [Google Scholar] [CrossRef]
- Mir, M.A.; Sawhney, S.S.; Jassal, M.M.S. In-vitro antidiabetic studies of various extracts of Taraxacum officinale. Pharma Innov. J. 2015, 4, 61–66. [Google Scholar]
- Li, J.; Luo, J.; Chai, Y.; Guo, Y.; Tianzhi, Y.; Bao, Y. Hypoglycemic effect of Taraxacum officinale root extract and its synergism with Radix Astragali extract. Food Sci. Nutr. 2021, 9, 2075–2085. [Google Scholar] [CrossRef]
- Choi, J.; Yoon, K.D.; Kim, J. Chemical constituents from Taraxacum officinale and their α-glucosidase inhibitory activities. Bioorg. Med. Chem. Lett. 2018, 28, 476–481. [Google Scholar] [CrossRef]
- Perumal, N.; Nallappan, M.; Shohaimi, S.; Kassim, N.K.; Tee, T.T.; Cheah, Y.H. Synergistic antidiabetic activity of Taraxacum officinale (L.) Weber ex FH Wigg and Momordica charantia L. polyherbal combination. Biomed. Pharmacother. 2022, 145, 112401. [Google Scholar] [CrossRef]
- Nnamdi, C.C.; Uwakwe, A.; Chuku, L. Hypoglycemic effects of aqueous and ethanolic extracts of dandelion (Taraxacum officinale F.H. Wigg.) leaves and roots on streptozotocin-induced albino rats. Glob. J. Res. Med. Plants Indig. Med. 2012, 1, 211–217. [Google Scholar]
- Pisonero-Vaquero, S.; González-Gallego, J.; Sánchez-Campos, S.; García-Mediavilla, M.V. Flavonoids and Related Compounds in Non-Alcoholic Fatty Liver Disease Therapy. Curr. Med. Chem. 2015, 22, 2991–3012. [Google Scholar] [CrossRef]
- Masarone, M.; Rosato, V.; Aglitti, A.; Bucci, T.; Caruso, R.; Salvatore, T.; Sasso, F.C.; Tripodi, M.F.; Persico, M. Liver biopsy in type 2 diabetes mellitus: Steatohepatitis represents the sole feature of liver damage. PLoS ONE 2017, 12, e0178473. [Google Scholar] [CrossRef]
- Ignat, M.V.; Coldea, T.E.; Salanță, L.C.; Mudura, E. Plants of the Spontaneous Flora with Beneficial Action in the Management of Diabetes, Hepatic Disorders, and Cardiovascular Disease. Plants 2021, 10, 216. [Google Scholar] [CrossRef]
- McCarty, M.F. A chlorogenic acid-induced increase in GLP-1 production may mediate the impact of heavy coffee consumption on diabetes risk. Med. Hypotheses 2005, 64, 848–853. [Google Scholar] [CrossRef] [PubMed]
- Scheen, A.J. Is There a Role for α-Glucosidase Inhibitors in the Prevention of Type 2 Diabetes Mellitus? Drugs 2003, 63, 933–951. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Kang, M.J.; Kim, M.J.; Kim, M.E.; Song, J.H.; Lee, Y.M.; Kim, J.I. Pancreatic lipase inhibitory activity of Taraxacum officinale in vitro and in vivo. Nutr. Res. Pract. 2008, 2, 200–203. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.D.; Duan, Y.Q.; Gao, J.M.; Ruan, Z.G. Screening for anti-lipase properties of 37 traditional Chinese medicinal herbs. J. Chin. Med. Assoc. 2010, 73, 319–324. [Google Scholar] [CrossRef] [PubMed]
- Davaatseren, M.; Hur, H.J.; Yang, H.J.; Hwang, J.T.; Park, J.H.; Kim, H.J.; Kim, M.J.; Kwon, D.Y.; Sung, M.J. Taraxacum officiale (dandelion) leaf extract alleviates high-fat diet-induced nonalcoholic fatty liver. Food Chem. Toxicol. 2013, 58, 30–36. [Google Scholar] [CrossRef]
- Choi, U.-K.; Lee, O.-H.; Yim, J.H.; Cho, C.-W.; Rhee, Y.K.; Lim, S.-I.; Kim, Y.-C. Hypolipidemic and Antioxidant Effects of Dandelion (Taraxacum officinale) Root and Leaf on Cholesterol-Fed Rabbits. Int. J. Mol. Sci. 2010, 11, 67–78. [Google Scholar] [CrossRef] [PubMed]
- González-Castejón, M.; García-Carrasco, B.; Fernández-Dacosta, R.; Dávalos, A.; Rodriguez-Casado, A. Reduction of adipogenesis and lipid accumulation by Taraxacum officinale (Dandelion) extracts in 3T3L1 adipocytes: An in vitro study. Phytother. Res. 2014, 28, 745–752. [Google Scholar] [CrossRef]
- Colitti, M.; Stefanon, B. Different anti-adipogenic effects of bio-compounds on primary visceral pre-adipocytes and adipocytes. EXCLI J. 2016, 15, 362–377. [Google Scholar] [CrossRef]
- Sirakov, I.; Velichkova, K.; Stoyanova, S.; Zhelyazkov, G.; Staykov, Y. The effect of diet supplemented with dandelion’s (Taraxacum officinale) extract on the productive and blood parameters of common carp (Cyprinus carpio L.), cultivated in the recirculation system. Maced. Vet. Rev. 2019, 42, 131–139. [Google Scholar] [CrossRef]
- Aremu, O.O.; Oyedeji, A.O.; Oyedeji, O.O.; Nkeh-Chungag, B.N.; Rusike, C.R.S. In Vitro and In Vivo Antioxidant Properties of Taraxacum officinale in Nω-Nitro-l-Arginine Methyl Ester (L-NAME)-Induced Hypertensive Rats. Antioxidants 2019, 15, 309. [Google Scholar] [CrossRef]
- Neef, H.; Cilli, F.; Declerck, P.J.; Laekeman, G. Platelet antiaggregating activity of Taraxacum officinale Weber. Phytother. Res. 1996, 10, 138–140. [Google Scholar]
- Lis, B.; Jędrejek, D.; Stochmal, A.; Olas, B. Assessment of effects of phenolic fractions from leaves and petals of dandelion in selected components of hemostasis. Food Res. Int. 2018, 107, 605–612. [Google Scholar] [CrossRef]
- Jedrejek, D.; Lis, B.; Rolnik, A.; Stochmal, A.; Olas, B. Comparative phytochemical, cytotoxicity, antioxidant and haemostatic studies of Taraxacum officinale root preparations. Food Chem. Toxicol. 2019, 126, 233–247. [Google Scholar] [CrossRef]
- Lis, B.; Jedrejek, D.; Moldoch, J.; Stochmal, A.; Olas, B. The anti-oxidative and hemostasis-related multifunctionality of L-chicoric acid, the main component of dandelion: An in vitro study of its cellular safety, antioxidant and anti-platelet properties, and effect on coagulation. J. Funct. Foods 2019, 62, 103524. [Google Scholar] [CrossRef]
- Lis, B.; Stochmal, A.; Jędrejek, D.; Rywaniak, J.; Olas, B.; Szustka, A. Anti-platelet activity of phytocompounds in various dandelion organs in human whole blood model in vitro. J. Funct. Foods 2021, 80, 104438. [Google Scholar] [CrossRef]
- Rao, P.R.M.; Jyothi, Y.; Rabban, S.I. Anti-obesity activity of Taraxacum officinale in high fat diet induced obese rats. J. Chem. Pharm. Res. 2015, 7, 244–248. [Google Scholar]
- Aabideen, Z.U.; Mumtaz, M.W.; Akhtar, M.T.; Mukhtar, H.; Raza, S.A.; Touqeer, T.; Saari, N. Anti-Obesity Attributes; UHPLC-QTOF-MS/MS-Based Metabolite Profiling and Molecular Docking Insights of Taraxacum officinale. Molecules 2020, 25, 4935. [Google Scholar] [CrossRef]
- Majewski, M.; Lis, B.; Juśkiewicz, J.; Ognik, K.; Jedrejek, D.; Stochmal, A.; Olas, B. The composition and vascular/antioxidant properties of Taraxacum officinale flower water syrup in a normal-fat diet using an obese rat model. J. Ethnopharmacol. 2021, 30, 113393. [Google Scholar] [CrossRef]
- Yu, H.; Li, J.; Yu, L.; Liu, J.; Wu, K.; Gao, M. Mineral analysis and animal toxicology assessement of wild dandelion (Taraxcum mongolicum). Stud. Trace Elem. Health 2004, 21, 4–5. [Google Scholar]
- Yarnell, E.; Abascal, K. Dandelion (Taraxacum officinale and T. mongolicum). Integr. Med. 2009, 8, 35–38. [Google Scholar]
- Schütz, K.; Carle, R.; Schieber, A. Taraxacum—A review on its phytochemical and pharmacological profile. J. Ethnopharmacol. 2006, 107, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Dufay, S.; Worsley, A.; Monteillier, A.; Avanzi, C.; Sy, J.; Ng, T.F.; Wong, I.C. Herbal tea extracts inhibit Cytochrome P450 3A4 in vitro. J. Pharm. Pharmacol. 2014, 66, 1478–1490. [Google Scholar] [CrossRef] [PubMed]
- Benalia, A.; Djjebar, A.A. Evaluation of the Acute Toxicity of Dandelion (Taraxacum officinale) Roots. J. Drug Deliv. Ther. 2020, 10, 159–163. [Google Scholar] [CrossRef]


| Name of the Phyto-Component and Parts of the Plant | Structure | Actions | References |
|---|---|---|---|
| taraxasterol (phytosterol) root |  C30H50O C30H50O | antihyperglycemic and anti-inflammatory properties | [27] |
| anti-inflammatory activity | [28,29] | ||
| decreases protein expression levels of PTBP1 and SIRT1, and may inhibit HBV and be a potential anti-HBV drug, | [30] | ||
| stigma sterol (phytosterols) leaf and steam | 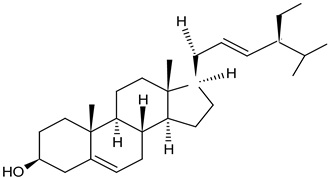 C29H48O C29H48O | anti-inflammatory, anti-hyperglycemic, antimicrobial properties | [31] |
| chicoric acid all parts of the plant |  C22H18O12 C22H18O12 | antidiabetic agent with both insulin-sensitizing and insulin-secreting properties, preventing the formation and/or progression of atherosclerosis, antiradical and protective actions against oxidation processes, meanwhile, flavonoids inhibit the formation of reactive oxygen and/or nitrogen species by suppressing NO synthase and COX-2 protein expression | [32,33] |
| tetrahydroridentin B sesquiterpen lactone root | 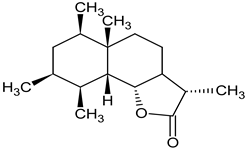 C15H24O4 C15H24O4 | activated the transcription factor nuclear factor erythroid 2-related factor 2 (Nrf2) in human hepatocytes, induced the Nrf2 target gene heme oxygenase | [34] |
| chlorogenic acid flower | 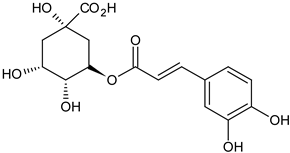 C16H18O9 C16H18O9 | antioxidant properties | [35] |
| anti-inflammatory, antibacterial, antiviral, hypoglycemic, lipid-lowering, anticardiovascular, antimutagenic, anticancer, immunomodulatory | [36] | ||
| caffeic acid flower and root | 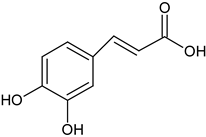 C9H8O4 C9H8O4 | anti-oxidative and immunostimulatory properties | [35] |
| hydroxycinnamic acids fruit | 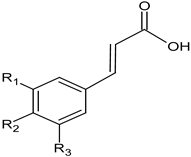 R2-OH p-coumaric acid R2-OH p-coumaric acidR3-OCH3; R2-OH ferulic acid R1-OCH3; R2-OH; R3-OCH3 sinapic acid | in experiments on plasma and platelets, using several different parameters (lipid peroxidation, protein carbonylation, oxidation of thiols, and platelet adhesion), the highest antioxidant and antiplatelet potential was demonstrated | [37] |
| luteolin aboveground plant parts | 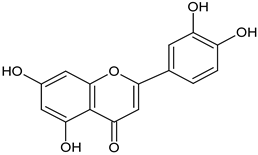 C15H10O6 C15H10O6 | important role in the amelioration of LPS-induced oxidative stress and inflammation. | [38] |
| inulin root | 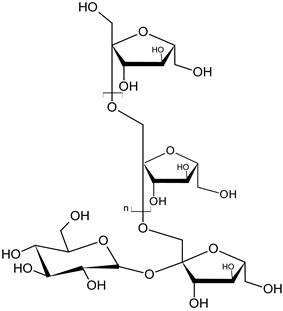 C6nH10n+2O5n+1 C6nH10n+2O5n+1 | influences the development of normal intestinal microflora | [39] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kania-Dobrowolska, M.; Baraniak, J. Dandelion (Taraxacum officinale L.) as a Source of Biologically Active Compounds Supporting the Therapy of Co-Existing Diseases in Metabolic Syndrome. Foods 2022, 11, 2858. https://doi.org/10.3390/foods11182858
Kania-Dobrowolska M, Baraniak J. Dandelion (Taraxacum officinale L.) as a Source of Biologically Active Compounds Supporting the Therapy of Co-Existing Diseases in Metabolic Syndrome. Foods. 2022; 11(18):2858. https://doi.org/10.3390/foods11182858
Chicago/Turabian StyleKania-Dobrowolska, Małgorzata, and Justyna Baraniak. 2022. "Dandelion (Taraxacum officinale L.) as a Source of Biologically Active Compounds Supporting the Therapy of Co-Existing Diseases in Metabolic Syndrome" Foods 11, no. 18: 2858. https://doi.org/10.3390/foods11182858
APA StyleKania-Dobrowolska, M., & Baraniak, J. (2022). Dandelion (Taraxacum officinale L.) as a Source of Biologically Active Compounds Supporting the Therapy of Co-Existing Diseases in Metabolic Syndrome. Foods, 11(18), 2858. https://doi.org/10.3390/foods11182858







