Bioactive Compounds, Health Benefits and Food Applications of Grape
Abstract
:1. Introduction
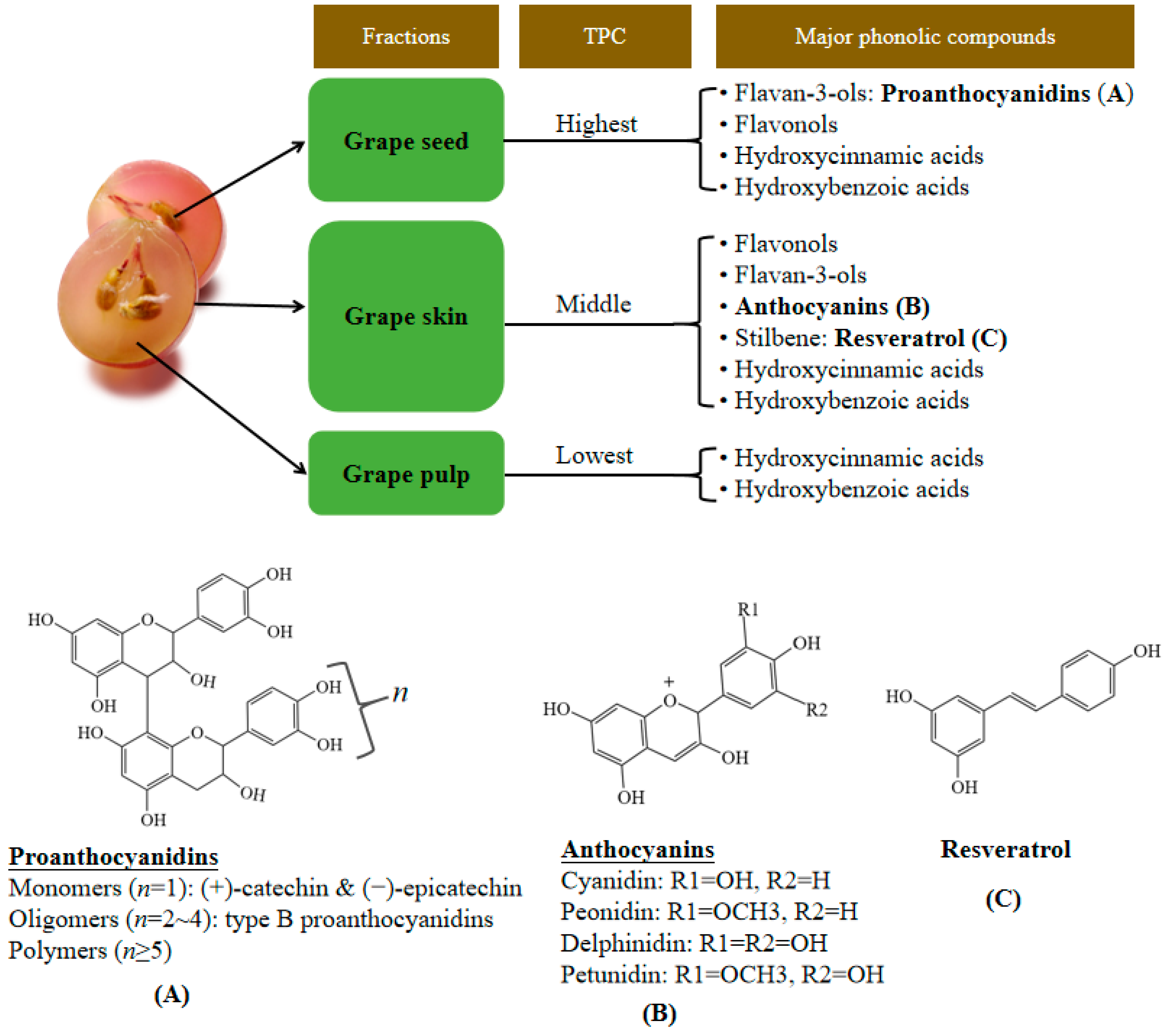
2. Bioactive Compounds in Grape
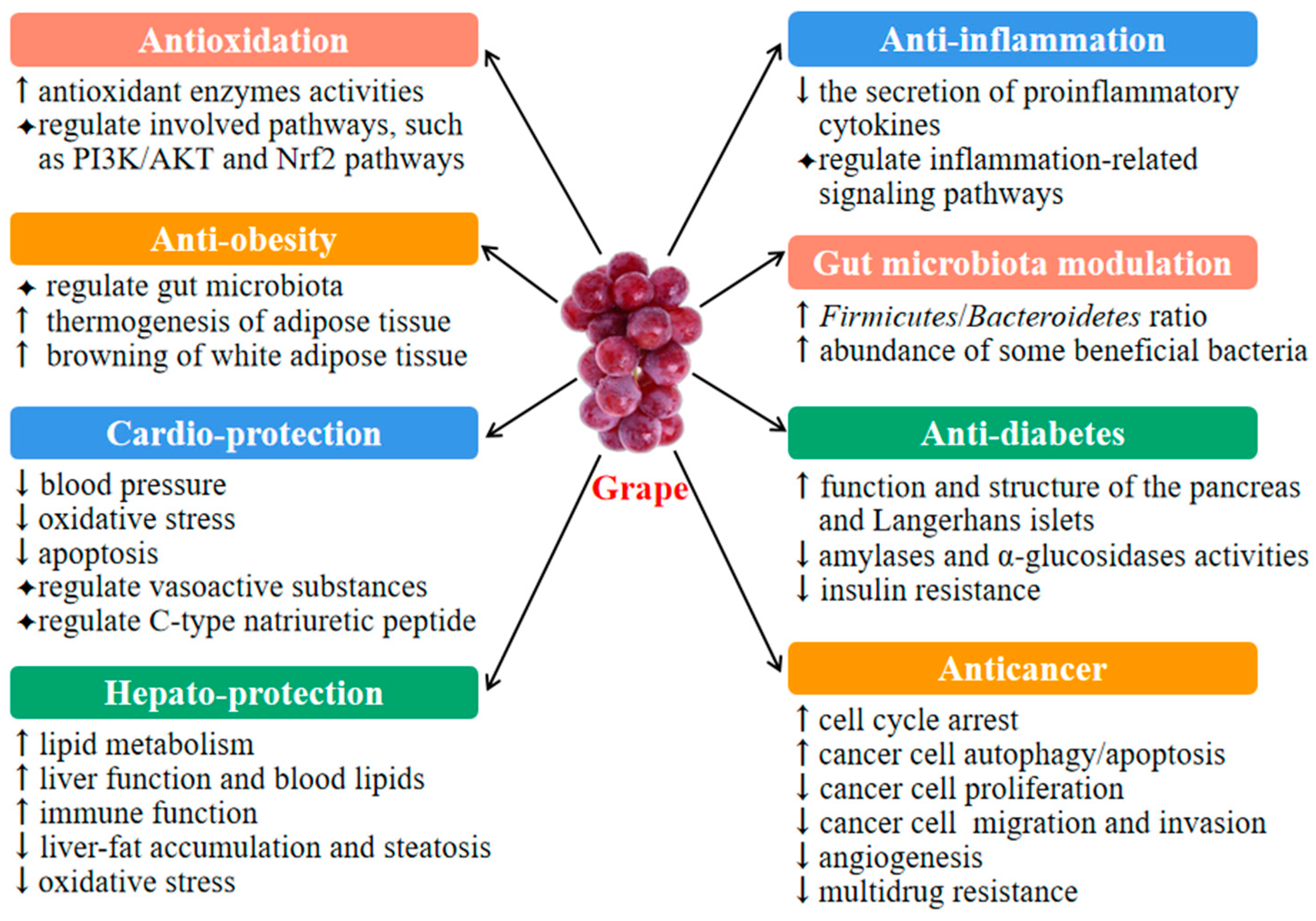
3. Health Benefits of Grape
3.1. Antioxidant Activity
3.2. Anti-Inflammatory Activity
3.3. Gut Microbiota Modulation
3.4. Antiobesity Activity
3.5. Cardioprotective Activity
3.6. Antidiabetic Activity
3.7. Hepatoprotective Activity
3.8. Anticancer Activity
3.9. Other Health Benefits
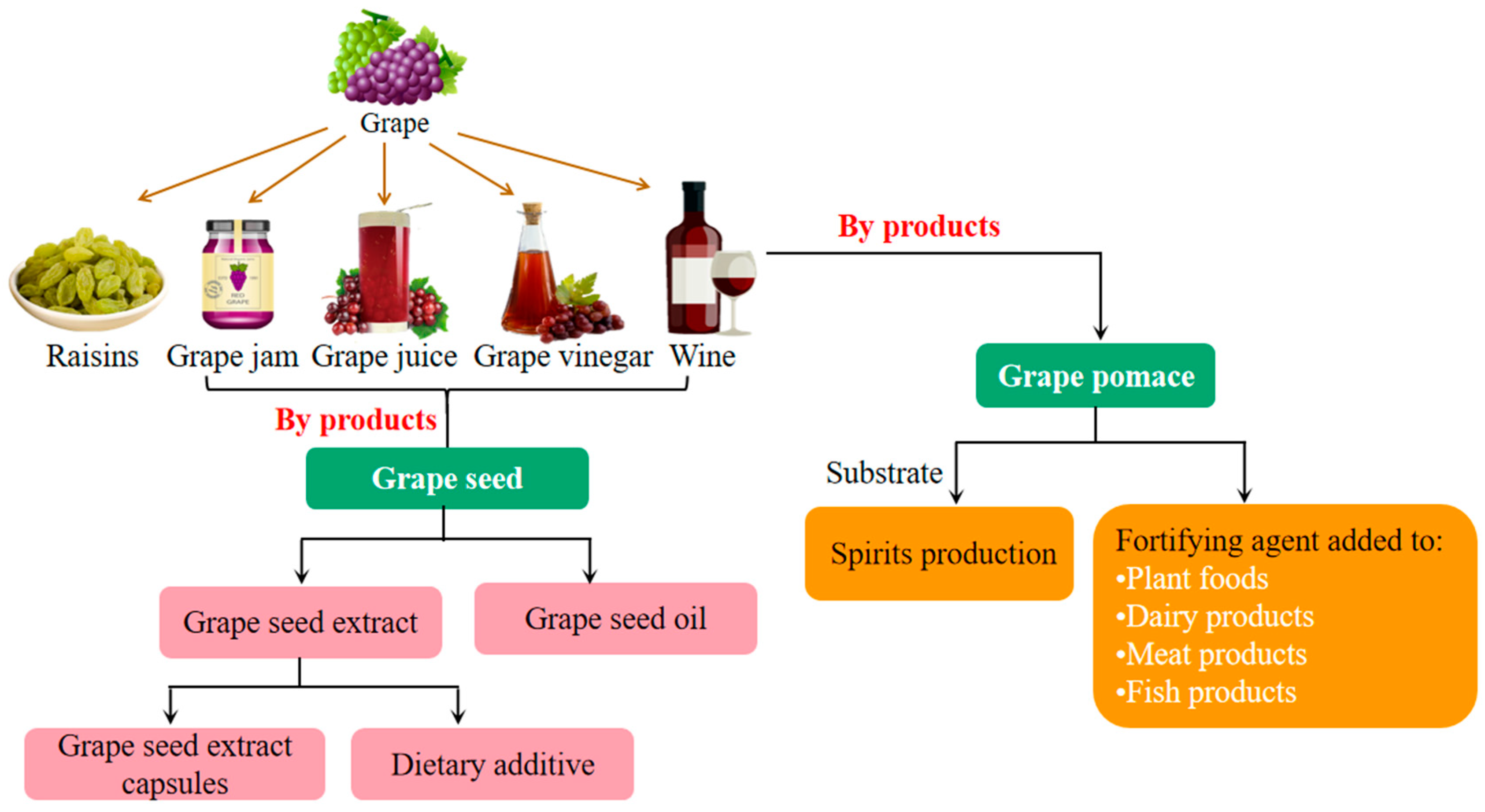
4. Applications of Grape in the Food Industry
4.1. Winemaking
4.2. Application of Grape Pomace in the Food Industry
4.3. Application of Grape Seed in the Food Industry
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Restani, P.; Fradera, U.; Ruf, J.C.; Stockley, C.; Teissedre, P.L.; Biella, S.; Colombo, F.; Di Lorenzo, C. Grapes and their derivatives in modulation of cognitive decline: A critical review of epidemiological and randomized-Controlled trials in humans. Crit. Rev. Food Sci. Nutr. 2021, 61, 566–576. [Google Scholar] [CrossRef] [PubMed]
- Colombo, F.; Di Lorenzo, C.; Regazzoni, L.; Fumagalli, M.; Sangiovanni, E.; de Sousa, L.P.; Bavaresco, L.; Tomasi, D.; Bosso, A.; Aldini, G.; et al. Phenolic profiles and anti-Inflammatory activities of sixteen table grape (Vitis vinifera L.) varieties. Food Funct. 2019, 10, 1797–1807. [Google Scholar] [CrossRef] [PubMed]
- Unusan, N. Proanthocyanidins in grape seeds: An updated review of their health benefits and potential uses in the food industry. J. Funct. Foods 2020, 67, 103861. [Google Scholar] [CrossRef]
- Liu, Q.; Tang, G.-Y.; Zhao, C.-N.; Feng, X.-L.; Xu, X.-Y.; Cao, S.-Y.; Meng, X.; Li, S.; Gan, R.-Y.; Li, H.-B. Comparison of antioxidant activities of different grape varieties. Molecules 2018, 23, 2432. [Google Scholar] [CrossRef] [PubMed]
- Andrade, M.A.; Lima, V.; Silva, A.S.; Vilarinho, F.; Castilho, M.C.; Khwaldia, K.; Ramos, F. Pomegranate and grape by-Products and their active compounds: Are they a valuable source for food applications? Trends Food Sci. Technol. 2019, 86, 68–84. [Google Scholar] [CrossRef]
- Han, X.; Guo, J.L.; Yin, M.W.; Liu, Y.W.; You, Y.L.; Zhan, J.C.; Huang, W.D. Grape extract activates brown adipose tissue through pathway involving the regulation of gut microbiota and bile acid. Mol. Nutr. Food Res. 2020, 64, 2000149. [Google Scholar] [CrossRef]
- Svezia, B.; Cabiati, M.; Matteucci, M.; Passino, C.; Pe, M.E.; Lionetti, V.; Del Ry, S. Tuscany sangiovese grape juice imparts cardioprotection by regulating gene expression of cardioprotective c-Type natriuretic peptide. Eur. J. Nutr. 2020, 59, 2953–2968. [Google Scholar] [CrossRef]
- Xia, E.-Q.; Deng, G.-F.; Guo, Y.-J.; Li, H.-B. Biological activities of polyphenols from grapes. Int. J. Mol. Sci. 2010, 11, 622–646. [Google Scholar] [CrossRef]
- Irak, K.; Yildirim, S.; Mert, H.; Mert, N. Grape seed extract effects on serum amylase levels and immunohistochemical alterations in streptozotocin-Induced diabetic rats. Cell. Mol. Biol. 2018, 64, 92–97. [Google Scholar] [CrossRef]
- El-Din, N.K.B.; Ali, D.A.; Abou-El-Magd, R.F. Grape seeds and skin induce tumor growth inhibition via g1-Phase arrest and apoptosis in mice inoculated with ehrlich ascites carcinoma. Nutrition 2019, 58, 100–109. [Google Scholar] [CrossRef]
- Yang, J.; Xiao, Y.-Y. Grape phytochemicals and associated health benefits. Crit. Rev. Food Sci. Nutr. 2013, 53, 1202–1225. [Google Scholar] [CrossRef] [PubMed]
- Barona, J.; Blesso, C.N.; Andersen, C.J.; Park, Y.; Lee, J.; Fernandez, M.L. Grape consumption increases anti-Inflammatory markers and upregulates peripheral nitric oxide synthase in the absence of dyslipidemias in men with metabolic syndrome. Nutrients 2012, 4, 1945–1957. [Google Scholar] [CrossRef] [PubMed]
- Mohammad, A.; Shahnaz, T.; Sorayya, K. Effect of 8 weeks’ supplementation grape seed extract on insulin resistance in iranian adolescents with metabolic syndrome: A randomized controlled trial. Diabetes Metab. Syndr.-Clin. Res. Rev. 2021, 15, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.X.; Du, C.G. Could grape-Based food supplements prevent the development of chronic kidney disease? Crit. Rev. Food Sci. Nutr. 2020, 60, 3054–3062. [Google Scholar] [CrossRef]
- Benbouguerra, N.; Richard, T.; Saucier, C.; Garcia, F. Voltammetric behavior, flavanol and anthocyanin contents, and antioxidant capacity of grape skins and seeds during ripening (Vitis vinifera var. Merlot, tannat, and Syrah). Antioxidants 2020, 9, 800. [Google Scholar] [CrossRef]
- Zhao, D.Y.; Simon, J.E.; Wu, Q.L. A critical review on grape polyphenols for neuroprotection: Strategies to enhance bioefficacy. Crit. Rev. Food Sci. Nutr. 2020, 60, 597–625. [Google Scholar] [CrossRef]
- de Oliveira, J.B.; Egipto, R.; Laureano, O.; de Castro, R.; Pereira, G.E.; Ricardo-da-Silva, J.M. Chemical characteristics of grapes cv. Syrah (Vitis vinifera L.) grown in the tropical semiarid region of brazil (pernambuco state): Influence of rootstock and harvest season. J. Sci. Food Agric. 2019, 99, 5050–5063. [Google Scholar] [CrossRef]
- Kandylis, P.; Dimitrellou, D.; Moschakis, T. Recent applications of grapes and their derivatives in dairy products. Trends Food Sci. Technol. 2021, 114, 696–711. [Google Scholar] [CrossRef]
- Tang, G.-Y.; Zhao, C.-N.; Liu, Q.; Feng, X.-L.; Xu, X.-Y.; Cao, S.-Y.; Meng, X.; Li, S.; Gan, R.-Y.; Li, H.-B. Potential of grape wastes as a natural source of bioactive compounds. Molecules 2018, 23, 2598. [Google Scholar] [CrossRef]
- Averilla, J.N.; Oh, J.; Kim, H.J.; Kim, J.S.; Kim, J.S. Potential health benefits of phenolic compounds in grape processing by-Products. Food Sci. Biotechnol. 2019, 28, 1607–1615. [Google Scholar] [CrossRef]
- Chen, Y.; Wen, J.Y.; Deng, Z.X.; Pan, X.Q.; Xie, X.F.; Peng, C. Effective utilization of food wastes: Bioactivity of grape seed extraction and its application in food industry. J. Funct. Foods 2020, 73, 104113. [Google Scholar] [CrossRef]
- Shinagawa, F.B.; de Santana, F.C.; Torres, L.R.O.; Mancini, J. Grape seed oil: A potential functional food? Food Sci. Technol. 2015, 35, 399–406. [Google Scholar] [CrossRef]
- Sabra, A.; Netticadan, T.; Wijekoon, C. Grape bioactive molecules, and the potential health benefits in reducing the risk of heart diseases. Food Chem. X 2021, 12, 100149. [Google Scholar] [CrossRef] [PubMed]
- Wongnarat, C.; Srihanam, P. Phytochemical and antioxidant activity in seeds and pulp of grape cultivated in thailand. Orient. J. Chem. 2017, 33, 113–121. [Google Scholar] [CrossRef]
- Kupe, M.; Karatas, N.; Unal, M.S.; Ercisli, S.; Baron, M.; Sochor, J. Nutraceutical and functional properties of peel, pulp, and seed extracts of six ‘kohnu’ grape clones. Horticulturae 2021, 7, 346. [Google Scholar] [CrossRef]
- Vo, G.T.; Liu, Z.Y.; Chou, O.; Zhong, B.M.; Barrow, C.J.; Dunshea, F.R.; Suleria, H.A.R. Screening of phenolic compounds in australian grown grapes and their potential antioxidant activities. Food Biosci. 2022, 47, 101644. [Google Scholar] [CrossRef]
- Mohamedshah, Z.; Chadwick-Corbin, S.; Wightman, J.D.; Ferruzzi, M.G. Comparative assessment of phenolic bioaccessibility from 100% grape juice and whole grapes. Food Funct. 2020, 11, 6433–6445. [Google Scholar] [CrossRef]
- Ledda, S.; Sanna, G.; Manca, G.; Franco, M.A.; Porcu, A. Variability in flavonol content of grapes cultivated in two mediterranean islands (sardinia and corsica). J. Food Compos. Anal. 2010, 23, 580–585. [Google Scholar] [CrossRef]
- Arapitsas, P.; Oliveira, J.; Mattivi, F. Do white grapes really exist? Food Res. Int. 2015, 69, 21–25. [Google Scholar] [CrossRef]
- Hasan, M.M.; Bae, H. An overview of stress-Induced resveratrol synthesis in grapes: Perspectives for resveratrol-Enriched grape products. Molecules 2017, 22, 294. [Google Scholar] [CrossRef] [Green Version]
- Perestrelo, R.; Lu, Y.; Santos, S.A.O.; Silvestre, A.J.D.; Neto, C.P.; Camara, J.S.; Rocha, S.M. Phenolic profile of sercial and tinta negra Vitis vinifera L. Grape skins by hplc-Dad-Esi-Msn novel phenolic compounds in vitis vinifera L. Grape. Food Chem. 2012, 135, 94–104. [Google Scholar] [CrossRef]
- Silva, L.R.; Queiroz, M. Bioactive compounds of red grapes from dao region (portugal): Evaluation of phenolic and organic profile. Asian Pac. J. Trop. Biomed. 2016, 6, 315–321. [Google Scholar] [CrossRef]
- Meng, J.F.; Shi, T.C.; Song, S.; Zhang, Z.W.; Fang, Y.L. Melatonin in grapes and grape-Related foodstuffs: A review. Food Chem. 2017, 231, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Yeung, A.W.K.; Aggarwal, B.B.; Orhan, I.E.; Barreca, D.; Battino, M.; Belwal, T.; Bishayee, A.; Daglia, M.; Devkota, H.P.; Echeverria, J.; et al. Resveratrol, a popular dietary supplement for human and animal health: Quantitative research literature analysis—A review. Anim. Sci. Pap. Rep. 2019, 37, 103–118. [Google Scholar]
- Xu, X.Y.; Meng, X.; Li, S.; Gan, R.Y.; Li, Y.; Li, H.B. Bioactivity, health benefits, and related molecular mechanisms of curcumin: Current progress, challenges, and perspectives. Nutrients 2018, 10, 1553. [Google Scholar] [CrossRef]
- Meng, X.; Zhou, J.; Zhao, C.N.; Gan, R.Y.; Li, H.B. Health benefits and molecular mechanisms of resveratrol: A narrative review. Foods 2020, 9, 340. [Google Scholar] [CrossRef]
- He, J.; Ye, S.; Correia, P.; Fernandes, I.; Zhang, R.; Wu, M.; Freitas, V.; Mateus, N.; Oliveira, H. Dietary polyglycosylated anthocyanins, the smart option? A comprehensive review on their health benefits and technological applications. Compr. Rev. Food Sci. Food Saf. 2022, 21, 3096–3128. [Google Scholar] [CrossRef]
- Meng, X.; Li, Y.; Li, S.; Zhou, Y.; Gan, R.Y.; Xu, D.P.; Li, H.B. Dietary sources and bioactivities of melatonin. Nutrients 2017, 9, 367. [Google Scholar] [CrossRef]
- Zhou, D.-D.; Luo, M.; Shang, A.; Mao, Q.-Q.; Li, B.-Y.; Gan, R.-Y.; Li, H.-B. Antioxidant food components for the prevention and treatment of cardiovascular diseases: Effects, mechanisms, and clinical studies. Oxidative Med. Cell. Longev. 2021, 2021, 6627355. [Google Scholar] [CrossRef]
- Fu, L.; Xu, B.-T.; Xu, X.-R.; Gan, R.-Y.; Zhang, Y.; Xia, E.-Q.; Li, H.-B. Antioxidant capacities and total phenolic contents of 62 fruits. Food Chem. 2011, 129, 345–350. [Google Scholar] [CrossRef]
- Elejalde, E.; Villaran, M.C.; Lopez-de-Armentia, I.; Ramon, D.; Murillo, R.; Alonso, R.M. Study of unpicked grapes valorization: A natural source of polyphenolic compounds and evaluation of their antioxidant capacity. Resources 2022, 11, 33. [Google Scholar] [CrossRef]
- Nallathambi, R.; Poulev, A.; Zuk, J.B.; Raskin, I. Proanthocyanidin-Rich grape seed extract reduces inflammation and oxidative stress and restores tight junction barrier function in caco-2 colon cells. Nutrients 2020, 12, 1623. [Google Scholar] [CrossRef] [PubMed]
- Maurer, L.H.; Cazarin, C.B.B.; Quatrin, A.; Minuzzi, N.M.; Nichelle, S.M.; Lamas, C.D.; Cagnon, V.H.A.; Morari, J.; Velloso, L.A.; Marostica, M.R.; et al. Grape peel powder attenuates the inflammatory and oxidative response of experimental colitis in rats by modulating the nf-Kappa b pathway and activity of antioxidant enzymes. Nutr. Res. 2020, 76, 52–70. [Google Scholar] [CrossRef] [PubMed]
- He, X.G.; Guo, X.D.; Ma, Z.J.; Li, Y.; Kang, J.H.; Zhang, G.Z.; Gao, Y.C.; Liu, M.Q.; Chen, H.W.; Kang, X.W. Grape seed proanthocyanidins protect pc12 cells from hydrogen peroxide-Induced damage via the pi3k/akt signaling pathway. Neurosci. Lett. 2021, 750, 135793. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, F.; Liang, M.; Chen, S.Z.; Zhu, Y.F.; Zou, Z.C.; Shi, B.K. Grape seed proanthocyanidin extract attenuates varicocele-Induced testicular oxidative injury in rats by activating the nrf2-Antioxidant system. Mol. Med. Rep. 2018, 17, 1799–1806. [Google Scholar]
- Shang, A.; Liu, H.-Y.; Luo, M.; Xia, Y.; Yang, X.; Li, H.-Y.; Wu, D.-T.; Sun, Q.; Geng, F.; Li, H.-B.; et al. Sweet tea (Lithocarpus polystachyus rehd.) as a new natural source of bioactive dihydrochalcones with multiple health benefits. Crit. Rev. Food Sci. Nutr. 2022, 62, 917–934. [Google Scholar] [CrossRef]
- Stamer, D.K.; Nizami, S.A.; Lee, F.Y.; Soung, D.Y. Whole grape alleviates inflammatory arthritis through inhibition of tumor necrosis factor. J. Funct. Foods 2017, 35, 458–465. [Google Scholar] [CrossRef]
- Pistol, G.C.; Marin, D.E.; Rotar, M.C.; Ropota, M.; Taranu, I. Bioactive compounds from dietary whole grape seed meal improved colonic inflammation via inhibition of mapks and nf-Kb signaling in pigs with dss induced colitis. J. Funct. Foods 2020, 66, 103708. [Google Scholar] [CrossRef]
- Liu, J.T.; Hu, S.L.; Zhu, B.Q.; Shao, S.M.; Yuan, L.B. Grape seed procyanidin suppresses inflammation in cigarette smoke-Exposed pulmonary arterial hypertension rats by the ppar-Gamma/cox-2 pathway. Nutr. Metab. Cardiovasc. Dis. 2020, 30, 347–354. [Google Scholar] [CrossRef]
- Bi, Y.J.; Yang, R.F. Human gut microbiota, nutrition and health. Chin. Sci. Bull. 2019, 64, 260–271. [Google Scholar] [CrossRef]
- Zhao, X.L.; Wu, Y.K.; Liu, H.C.; Hu, N.; Zhang, Y.; Wang, S. Grape seed extract ameliorates phip-Induced colonic injury by modulating gut microbiota, lipid metabolism, and nf-kappa b signaling pathway in rats. J. Funct. Foods 2021, 78, 104362. [Google Scholar] [CrossRef]
- Lu, F.; Liu, F.J.; Zhou, Q.; Hu, X.S.; Zhang, Y. Effects of grape pomace and seed polyphenol extracts on the recovery of gut microbiota after antibiotic treatment in high-Fat diet-Fed mice. Food Sci. Nutr. 2019, 7, 2897–2906. [Google Scholar] [CrossRef] [Green Version]
- Liu, M.; Yun, P.; Hu, Y.; Yang, J.; Khadka, R.B.; Peng, X.C. Effects of grape seed proanthocyanidin extract on obesity. Obes. Facts 2020, 13, 279–291. [Google Scholar] [CrossRef] [PubMed]
- Shang, A.O.; Gan, R.Y.; Xu, X.Y.; Mao, Q.Q.; Zhang, P.Z.; Li, H.B. Effects and mechanisms of edible and medicinal plants on obesity: An updated review. Crit. Rev. Food Sci. Nutr. 2021, 61, 2061–2077. [Google Scholar] [CrossRef]
- Zhou, F.; Yin, M.W.; Liu, Y.W.; Han, X.; Guo, J.L.; Ren, C.L.; Wang, W.; Huang, W.D.; Zhan, J.C.; You, Y.L. Grape seed flour intake decreases adiposity gain in high-Fat-Diet induced obese mice by activating thermogenesis. J. Funct. Foods 2019, 62, 103509. [Google Scholar] [CrossRef]
- Du, H.P.; Wang, Q.; Li, T.; Ren, D.Y.; Yang, X.B. Grape seed proanthocyanidins reduced the overweight of c57bl/6j mice through modulating adipose thermogenesis and gut microbiota. Food Funct. 2021, 12, 8467–8477. [Google Scholar] [CrossRef] [PubMed]
- Sierra-Cruz, M.; Miguens-Gomez, A.; Grau-Bove, C.; Rodriguez-Gallego, E.; Blay, M.; Pinent, M.; Ardevol, A.; Terra, X.; Beltran-Debon, R. Grape-Seed proanthocyanidin extract reverts obesity-Related metabolic derangements in aged female rats. Nutrients 2021, 13, 2059. [Google Scholar] [CrossRef] [PubMed]
- Parandoosh, M.; Yousefi, R.; Khorsandi, H.; Nikpayam, O.; Saidpour, A.; Babaei, H. The effects of grape seed extract (Vitis vinifera) supplement on inflammatory markers, neuropeptide y, anthropometric measures, and appetite in obese or overweight individuals: A randomized clinical trial. Phytother. Res. 2020, 34, 379–387. [Google Scholar] [CrossRef]
- Nunes, M.A.; Pimentel, F.; Costa, A.S.G.; Alves, R.C.; Oliveira, M.B.P.P. Cardioprotective properties of grape seed proanthocyanidins: An update. Trends Food Sci. Technol. 2016, 57, 31–39. [Google Scholar] [CrossRef]
- Ebrahimi-Mameghani, M.; Irandoost, P.; Pourmoradian, S. The effects of grape seed oil on the cardiovascular risk factors in overweight and obese women: A double-Blind randomized clinical trial. Curr. Top. Nutraceutical Res. 2020, 18, 221–226. [Google Scholar]
- Sato, A.; Nishioka, S.; Kiuchi, M.; Imada, Y.; Makino, K.; Nakagawa, K.; Tanaka, R.; Matsumura, Y.; Ohkita, M. Grape extract from chardonnay seeds restores deoxycorticosterone acetate-Salt-Induced endothelial dysfunction and hypertension in rats. Biol. Pharm. Bull. 2020, 43, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.F.; Song, S.J.; Zhang, X. Grape seed proanthocyanidin alleviates left ventricular remodeling by regulating systolic pressure, oxidative stress and vasoactive substances in spontaneously hypertensive rats. Trop. J. Pharm. Res. 2021, 20, 1663–1668. [Google Scholar] [CrossRef]
- Ruan, Y.X.; Jin, Q.K.; Zeng, J.J.; Ren, F.F.; Xie, Z.Y.; Ji, K.T.; Wu, L.P.; Wu, J.G.; Li, L. Grape seed proanthocyanidin extract ameliorates cardiac remodelling after myocardial infarction through pi3k/akt pathway in mice. Front. Pharmacol. 2020, 11, 585984. [Google Scholar] [CrossRef]
- Dillon, K.N.; Shariffi, B.; Thompson, B.; Steele, R.; Kim, J.K. Effects of acute grape seed extract supplementation on hemodynamics in normal body weight and obese males. J. Nutr. Sci. Vitaminol. 2020, 66, 427–431. [Google Scholar] [CrossRef]
- Foshati, S.; Nouripour, F.; Sadeghi, E.; Amani, R. The effect of grape (Vitis vinifera) seed extract supplementation on flow-Mediated dilation, blood pressure, and heart rate: A systematic review and meta-Analysis of controlled trials with duration- and dose analysis. Pharmacol. Res. 2022, 175, 105905. [Google Scholar] [CrossRef] [PubMed]
- Odai, T.; Terauchi, M.; Kato, K.; Hirose, A.; Miyasaka, N. Effects of grape seed proanthocyanidin extract on vascular endothelial function in participants with prehypertension: A randomized, double-Blind, placebo-Controlled study. Nutrients 2019, 11, 2844. [Google Scholar] [CrossRef]
- Yousefi, R.; Parandoosh, M.; Khorsandi, H.; Hosseinzadeh, N.; Tonekaboni, M.M.; Saidpour, A.; Babaei, H.; Ghorbani, A. Grape seed extract supplementation along with a restricted-Calorie diet improves cardiovascular risk factors in obese or overweight adult individuals: A randomized, placebo-Controlled trial. Phytother. Res. 2021, 35, 987–995. [Google Scholar] [CrossRef]
- Gupta, M.; Dey, S.; Marbaniang, D.; Pal, P.; Ray, S.; Mazumder, B. Grape seed extract: Having a potential health benefits. J. Food Sci. Technol.-Mysore 2020, 57, 1205–1215. [Google Scholar] [CrossRef] [PubMed]
- Kong, F.S.; Qin, Y.R.; Su, Z.P.; Ning, Z.X.; Yu, S.J. Optimization of extraction of hypoglycemic ingredients from grape seeds and evaluation of alpha-Glucosidase and alpha-Amylase inhibitory effects in vitro. J. Food Sci. 2018, 83, 1422–1429. [Google Scholar] [CrossRef] [PubMed]
- Li, X.J.; Shen, Y.B.; Zhu, J.H.; Xiao, J.Y.; Cong, R.H.; Zhang, H.; Wu, G.C.; Qi, X.G. Virgin grape seed oil alleviates insulin resistance and energy metabolism disorder in mice fed a high-Fat diet. Eur. J. Lipid Sci. Technol. 2020, 122, 1900158. [Google Scholar] [CrossRef]
- Gao, Z.L.; Liu, G.Y.; Hu, Z.; Shi, W.W.; Chen, B.B.; Zou, P.M.; Li, X.H. Grape seed proanthocyanidins protect against streptozotocin-Induced diabetic nephropathy by attenuating endoplasmic reticulum stress-Induced apoptosis. Mol. Med. Rep. 2018, 18, 1447–1454. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.S.; Li, H.Y.; Li, Y.; Liu, D.D.; Zhang, L.Y.; Wang, T.L.; Liu, T.; Ma, L. Protective effects of grape seed proanthocyanidins on the kidneys of diabetic rats through the nrf2 signalling pathway. Evid. Based Complement Altern. Med. 2020, 2020, 5205903. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.H.; Chu, Z.Y.; Li, W.N.; Sun, T.; Sun, X.J. Grape seed extracts attenuate retinal muller cell gliosis in streptozotocin-Diabetic rats. J. Funct. Foods 2018, 50, 45–52. [Google Scholar] [CrossRef]
- Ren, X.; Lu, H.Y.; Wang, N.; Zhang, C.H.; Ji, Y.P.; Cui, S.Q.; Dong, Y.C.; Yang, K.Y.; Du, M.Y.; Diao, F.S.; et al. Thioredoxin is implicated in the anti-Apoptotic effects of grape seed proanthocyanidin extract during hyperglycemia. Mol. Med. Rep. 2017, 16, 7731–7737. [Google Scholar] [CrossRef]
- Fan, M.Q.; Choi, Y.J.; Tang, Y.J.; Mun, S.; Yang, H.P.; Kim, E.K. Efficacy and mechanism of polymerized anthocyanin from grape-Skin extract on high-Fat-Diet-Induced nonalcoholic fatty liver disease. Nutrients 2019, 11, 2586. [Google Scholar] [CrossRef] [PubMed]
- Franklin, R.; Bispo, R.F.M.; Sousa-Rodrigues, C.F.; Pires, L.A.S.; Fonseca, A.; Babinski, M.A. Grape leucoanthocyanidin protects liver tissue in albino rabbits with nonalcoholic hepatic steatosis. Cells Tissues Organs 2018, 205, 129–136. [Google Scholar] [CrossRef]
- Zou, J.F.; Qi, F.J.; Ye, L.P.; Yao, S.Y. Protective role of grape seed proanthocyanidins against CCl4 induced acute liver injury in mice. Med. Sci. Monit. 2016, 22, 880–889. [Google Scholar] [CrossRef]
- Liu, B.Y.; Jiang, H.J.; Lu, J.J.; Baiyun, R.Q.; Li, S.Y.; Lv, Y.Y.; Li, D.; Wu, H.; Zhang, Z.G. Grape seed procyanidin extract ameliorates lead-Induced liver injury via mirna153 and akt/gsk-3 beta/fyn-Mediated nrf2 activation. J. Nutr. Biochem. 2018, 52, 115–123. [Google Scholar] [CrossRef]
- Mokni, M.; Hamlaoui, S.; Kadri, S.; Limam, F.; Amri, M.; Marzouki, L.; Aouani, E. Efficacy of grape seed and skin extract against doxorubicin-Induced oxidative stress in rat liver. Pak. J. Pharm. Sci. 2015, 28, 1971–1978. [Google Scholar]
- Wu, S.X.; Xiong, R.G.; Huang, S.Y.; Zhou, D.D.; Saimaiti, A.; Zhao, C.N.; Shang, A.; Zhang, Y.J.; Gan, R.Y.; Li, H.B. Effects and mechanisms of resveratrol for prevention and management of cancers: An updated review. Crit. Rev. Food Sci. Nutr. 2022; in press. [Google Scholar] [CrossRef]
- Li, C.H.; Zhang, L.L.; Liu, C.M.; He, X.M.; Chen, M.S.; Chen, J. Lipophilic grape seed proanthocyanidin exerts anti-Cervical cancer effects in hela cells and a hela-Derived xenograft zebrafish model. Antioxidants 2022, 11, 422. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.C.; Feng, Y.J.; Bai, Y.J.; Feng, Z.S.; Yang, X.J.; Dang, B.; Xiao, M.; Zhang, J.M.; Han, S.Q. Proanthocyanidins extracted from grape seeds inhibit the growth of hepatocellular carcinoma cells and induce apoptosis through the mapk/akt pathway. Food Biosci. 2022, 45, 101337. [Google Scholar] [CrossRef]
- Wang, L.H.; Huang, W.D.; Zhan, J.C. Grape seed proanthocyanidins induce autophagy and modulate survivin in hepg2 cells and inhibit xenograft tumor growth in vivo. Nutrients 2019, 11, 2983. [Google Scholar] [CrossRef] [PubMed]
- Joshi, T.; Patel, I.; Kumar, A.; Donovan, V.; Levenson, A.S. Grape powder supplementation attenuates prostate neoplasia associated with pten haploinsufficiency in mice fed high-Fat diet. Mol. Nutr. Food Res. 2020, 64, 2000326. [Google Scholar] [CrossRef]
- Yang, N.G.; Gao, J.; Hou, R.Z.; Xu, X.L.; Yang, N.Q.; Huang, S.S. Grape seed proanthocyanidins inhibit migration and invasion of bladder cancer cells by reversing emt through suppression of tgf-Beta signaling pathway. Oxidative Med. Cell. Longev. 2021, 2021, 5564312. [Google Scholar] [CrossRef]
- Lin, K.N.; Jiang, Y.L.; Zhang, S.G.; Huang, S.Y.; Li, H. Grape seed proanthocyanidin extract reverses multidrug resistance in hl-60/adr cells via inhibition of the pi3k/akt signaling pathway. Biomed. Pharmacother. 2020, 125, 109885. [Google Scholar] [CrossRef]
- Ravindranathan, P.; Pasham, D.; Goel, A. Oligomeric proanthocyanidins (opcs) from grape seed extract suppress the activity of abc transporters in overcoming chemoresistance in colorectal cancer cells. Carcinogenesis 2019, 40, 412–421. [Google Scholar] [CrossRef]
- Ben Youssef, S.; Brisson, G.; Doucet-Beaupre, H.; Castonguay, A.M.; Gora, C.; Amri, M.; Levesque, M. Neuroprotective benefits of grape seed and skin extract in a mouse model of parkinson’s disease. Nutr. Neurosci. 2021, 24, 197–211. [Google Scholar] [CrossRef]
- Grau-Bove, C.; Sierra-Cruz, M.; Miguens-Gomez, A.; Rodriguez-Gallego, E.; Beltran-Debon, R.; Blay, M.; Terra, X.; Pinent, M.; Ardevol, A. A ten-Day grape seed procyanidin treatment prevents certain ageing processes in female rats over the long term. Nutrients 2020, 12, 3647. [Google Scholar] [CrossRef]
- Kupe, M.; Karatas, N.; Unal, M.S.; Ercisli, S.; Baron, M.; Sochor, J. Phenolic composition and antioxidant activity of peel, pulp and seed extracts of different clones of the turkish grape cultivar ‘karaerik’. Plants 2021, 10, 2154. [Google Scholar] [CrossRef]
- Wang, Y.X.; Wang, Y.N.; Shen, W.L.; Wang, Y.D.; Cao, Y.N.; Nuerbulati, N.; Chen, W.; Lu, G.T.; Xiao, W.M.; Qi, R. Grape seed polyphenols ameliorated dextran sulfate sodium-Induced colitis via suppression of inflammation and apoptosis. Pharmacology 2020, 105, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Mezhibovsky, E.; Knowles, K.A.; He, Q.Y.; Sui, K.; Tveter, K.M.; Duran, R.M.; Roopchand, D.E. Grape polyphenols attenuate diet-Induced obesity and hepatic steatosis in mice in association with reduced butyrate and increased markers of intestinal carbohydrate oxidation. Front. Nutr. 2021, 8, 675267. [Google Scholar] [CrossRef]
- Mutlu, E.G.; Arslan, E.; Arikoglu, H.; Varol, S. The effect of grape seed extract on the pancreatic weight in diabetic rats. Med. J. Bakirkoy 2021, 17, 293–297. [Google Scholar] [CrossRef]
- Seo, K.H.; Kim, H.; Chon, J.W.; Kim, D.H.; Nah, S.Y.; Arvik, T.; Yokoyama, W. Flavonoid-Rich chardonnay grape seed flour supplementation ameliorates diet-Induced visceral adiposity, insulin resistance, and glucose intolerance via altered adipose tissue gene expression. J. Funct. Foods 2015, 17, 881–891. [Google Scholar] [CrossRef]
- Iuga, M.; Mironeasa, S. Potential of grape byproducts as functional ingredients in baked goods and pasta. Compr. Rev. Food Sci. Food Saf. 2020, 19, 2473–2505. [Google Scholar] [CrossRef] [PubMed]
- OIV (International Organisation of Vine and Wine). 2019 Statistical Report on World Vitiviniculture. Available online: http://www.oiv.int/public/medias/6782/oiv-2019-Statistical-Report-On-World-Vitiviniculture.pdf (accessed on 15 June 2020).
- Chedea, V.S.; Tomoiaga, L.L.; Macovei, S.O.; Magureanu, D.C.; Iliescu, M.L.; Bocsan, I.C.; Buzoianu, A.D.; Vosloban, C.M.; Pop, R.M. Antioxidant/pro-Oxidant actions of polyphenols from grapevine and wine by-Products-Base for complementary therapy in ischemic heart diseases. Front. Cardiovasc. Med. 2021, 8, 750508. [Google Scholar] [CrossRef]
- Baiano, A.; Scrocco, C.; Sepielli, G.; Del Nobile, M.A. Wine processing: A critical review of physical, chemical, and sensory implications of innovative vinification procedures. Crit. Rev. Food Sci. Nutr. 2016, 56, 2391–2407. [Google Scholar] [CrossRef]
- Nemzer, B.; Kalita, D.; Yashin, A.Y.; Yashin, Y.I. Chemical composition and polyphenolic compounds of red wines: Their antioxidant activities and effects on human health-A review. Beverages 2022, 8, 1. [Google Scholar] [CrossRef]
- Benbouguerra, N.; Hornedo-Ortega, R.; Garcia, F.; El Khawand, T.; Saucier, C.; Richard, T. Stilbenes in grape berries and wine and their potential role as anti-Obesity agents: A review. Trends Food Sci. Technol. 2021, 112, 362–381. [Google Scholar] [CrossRef]
- Antonic, B.; Jancikova, S.; Dordevic, D.; Tremlova, B. Grape pomace valorization: A systematic review and meta-Analysis. Foods 2020, 9, 1627. [Google Scholar] [CrossRef]
- Cilli, L.P.; Contini, L.R.F.; Sinnecker, P.; Lopes, P.S.; Andreo, M.A.; Neiva, C.R.P.; Nascimento, M.S.; Yoshida, C.M.P.; Venturini, A.C. Effects of grape pomace flour on quality parameters of salmon burger. J. Food Process. Preserv. 2020, 44, e14329. [Google Scholar] [CrossRef]
- Lee, H.-J.; Lee, J.-J.; Jung, M.-O.; Choi, J.-S.; Jung, J.-T.; Choi, Y.-I.; Lee, J.-K. Meat quality and storage characteristics of pork loin marinated in grape pomace. Korean J. Food Sci. Anim. Resour. 2017, 37, 726–734. [Google Scholar] [CrossRef]
- Zhou, Y.J.; Wang, Q.Y.; Wang, S.J. Effects of rosemary extract, grape seed extract and green tea polyphenol on the formation of n-Nitrosamines and quality of western-Style smoked sausage. J. Food Process. Preserv. 2020, 44, e14459. [Google Scholar] [CrossRef]
- Park, E.; Edirisinghe, I.; Choy, Y.Y.; Waterhouse, A.; Burton-Freeman, B. Effects of grape seed extract beverage on blood pressure and metabolic indices in individuals with pre-Hypertension: A randomised, double-Blinded, two-Arm, parallel, placebo-Controlled trial. Br. J. Nutr. 2016, 115, 226–238. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pasini, F.; Chinnici, F.; Caboni, M.F.; Verardo, V. Recovery of oligomeric proanthocyanidins and other phenolic compounds with established bioactivity from grape seed by-Products. Molecules 2019, 24, 677. [Google Scholar] [CrossRef]
- Yang, C.L.; Shang, K.; Lin, C.C.; Wang, C.; Shi, X.Q.; Wang, H.; Li, H. Processing technologies, phytochemical constituents, and biological activities of grape seed oil (gso): A review. Trends Food Sci. Technol. 2021, 116, 1074–1083. [Google Scholar] [CrossRef]
| Compound Name | Compound Name | Compound Name | Compound Name |
|---|---|---|---|
| Gallic acid 4-O-glucoside | Caffeic acid | Hesperetin 3′-sulfate | Quercetin 3-O-arabinoside |
| Ellagic acid arabinoside | 3-Caffeoylquinic acid | Neoeriocitrin | Violanone |
| Gallic acid | 3-p-Coumaroylquinic acid | Hesperidin | 2′-Hydroxyformononetin |
| Protocatechuic acid 4-O-glucoside | 3-Feruloylquinic acid | Hesperetin 3′-O-glucuronide | 5,6,7,3′,4′-Pentahydroxyisoflavone |
| 4-Hydroxybenzoic acid 4-O-glucoside | Ferulic acid | Rhoifolin | 3′-Hydroxygenistein |
| Schisandrin C | 1,5-Dicaffeoylquinic acid | Cirsilineol | 6”-O-Acetylglycitin |
| 2-Hydroxybenzoic acid | 3-Sinapoylquinic acid | Cirsilineol | Arbutin |
| Paeoniflorin | 3,4-Dihydroxyphenylacetic acid | Diosmin | Isopimpinellin |
| 3-O-Methylgallic acid | 2-Hydroxy-2-phenylacetic acid | Chrysoeriol 7-O-glucoside | 4-Hydroxybenzaldehyde |
| Cinnamic acid | Dihydroferulic acid 4-O-glucuronide | Myricetin 3-O-arabinoside | p-Anisaldehyde |
| Caffeoyl tartaric acid | Dihydrocaffeic acid 3-O-glucuronide | Procyanidin dimer B1 | p-Anisaldehyde |
| p-Coumaric acid 4-O-glucoside | Myricetin 3-O-rhamnoside | Kaempferol 3-O-xylosyl-glucoside | 4″-O-Methylepigallocatechin 3-Ogallate |
| m-Coumaric acid | 3-Hydroxyphloretin 2′-O-glucoside | Myricetin 3-O-rutinoside | Carnosic acid |
| Ferulic acid 4-O-glucoside | Phloridzin | Spinacetin 3-O-(2 | Hydroxytyrosol 4-O-glucoside |
| Cyanidin 3-O-(2-O-(6-O-(E)-caffeoyl-D glucoside)-D-glucoside)-5-O-Dglucoside | Kaempferol 3-O-(2″-rhamnosylgalactoside) 7-O-rhamnoside | Patuletin 3-O-glucosyl-(1->6)-[apiosyl (1->2)]-glucoside | 3′,4′,7-Trihydroxyisoflavanone |
| Hydroxycaffeic acid | Dihydromyricetin 3-O-rhamnoside | Myricetin 3-O-galactoside | 7-Oxomatairesinol |
| Caffeoyl glucose | Procyanidin trimer C1 | Kaempferol 3,7-O-diglucoside | Pinoresinol |
| Ferulic acid 4-O-glucuronide | Caffeic acid 3-O-glucuronide | Quercetin 3′-O-glucuronide | 2,3-Dihydroxybenzoic acid |
| Caffeic acid 4-sulfate | (+)-Catechin | Kaempferol 3-O-glucosyl-rhamnosylgalactoside | Resveratrol 5-O-glucoside |
| Feruloyl tartaric acid | (+)-Gallocatechin |
| Study Type | Model | Product (Component) | Treatment | Main Effects and Related Mechanisms | Ref. |
|---|---|---|---|---|---|
| Antioxidant ability | |||||
| In vitro | / | Grape extract (hydroxycinnamic acids, flavan-3-ols and tannins) | / | Exhibited antioxidant power, including ORAC, DPPH and ABTS radical-scavenging activities; FRAP, CUPRAC | [41] |
| In vitro | / | Grape pulp, peel and seed extracts (gallic acid, quercetin, catechin, chlorogenic acid, caffeic acid and p-coumaric acid) | / | Antioxidant capacity (determined by DPPH radical-scavenging activity and FRAP and TEAC assays): grape seed > grape peel > grape pulp | [90] |
| In vitro | / | Pulp, peel and seed extracts (epicatechin, catechin gallate, gallic acid, rutin and resveratrol) | / | Antioxidant capacity (determined by FRAP and TEAC assays): grape seed > grape peel > grape pulp; these results are in accordance with the respective TPC | [4,19] |
| In vitro | LPS-induced oxidation in human Caco-2 colon cells | Grape seed extract | Culture: 12.5 μg/mL for 24 h | Reduced the generation of intracellular ROS and mitochondrial superoxide and increased the gene expression of antioxidant enzymes (GSR, SOD and GPX) | [42] |
| In vitro | H2O2-induced oxidation in PC12 cells | Grape seed proanthocyanidins | Culture: 5, 10 or 25 μM for 24 h | Protected against oxidative damage via the PI3K/Akt signaling pathway | [44] |
| In vivo | TNBS-treated Wistar rats | Grape peel powder | Diet supplementation: 8% for 15 d | Ameliorated TNBS-induced oxidative damage by improving the activities of antioxidant enzymes (SOD and CAT) and decreasing oxidation and NO levels | [43] |
| In vivo | A varicocele model in Wistar rats | Grape seed proanthocyanidin extract | Oral administration: 250 mg/kg for 4 weeks | Reduced varicocele-induced testicular oxidative damage by activating the Nrf2 pathway | [45] |
| Anti-inflammatory activity | |||||
| In vitro | TNF-α-treated gastric epithelial cells | Grape extracts (phenolic compounds) | Culture: 5–100 μg/mL for 6 h | Inhibited TNF-α-induced IL-8 release | [2] |
| In vivo | Transgenic mice overexpressing TNF | Whole grape powder | Diet supplementation: 5% or 10% for 4 weeks | Suppressed human TNF-mediated inflammation and improved the symptoms of inflammatory arthritis | [47] |
| In vivo | TNBS-treated Wistar rats | Grape peel powder | Diet supplementation: 8% 15 d before and 7 d after TNBS treatment | Ameliorated inflammation by downregulating the NF-κB pathway | [43] |
| In vivo | DSS-induced inflammation in hybrid piglets | Grape seed meal (catechins, epicatechins and procyanidins) | Diet supplementation: 8% for 30 d | Counteracted the inflammatory response by decreasing the production of proinflammatory mediators, inhibiting the MAPK and NF-κB pathways | [48] |
| In vivo | DSS-induced inflammation in C57BL/6 mice | Grape seed polyphenol extract | Oral administration: 500 and 750 mg/kg for 6 d | Decreased inflammatory infiltration by inhibiting the mRNA expression of inflammatory cytokines (IL-1β, IL-6 and TNF-α) and reducing the phosphorylation of STAT3 | [91] |
| In vivo | Cigarette-smoke-exposed SD rats | Grape seed proanthocyanidin | Intratracheal injection: 30 mg, 2 mL/kg for 6 months | Inhibited inflammation through the PPAR-γ/COX-2 pathway | [49] |
| Gut microbiota modulatory ability | |||||
| In vivo | HFFD-fed C57BL/6 J mice | Grape extract | Drinking: 1% w/w dissolved in water | Restored the disturbance of gut microbiota by improving the ratio of Firmicutes/Bacteroidetes and the abundance of the Bifidobacteria, Clostridia and Akkermansia genera | [6] |
| In vivo | PhIP-treated Wistar rats | Grape seed extract | Intragastric administration: 60 mg/kg/d for 6 weeks | Maintained the homeostasis of gut microbiota, especially by preventing a decrease in Lactobacillus abundance induced by PhIP, thereby ameliorating colonic injury | [51] |
| In vivo | HFD-fed C57BL/6 J mice | Grape seed polyphenol extract | Oral administration: 200 mg/kg/d for 7 d | Promoted the recovery of gut microbiota after treatment with an antibiotic cocktail | [52] |
| Antiobesity activity | |||||
| In vivo | HFFD-fed C57BL/6 Cnc mice | Grape extract | Drinking: 1% w/w dissolved in water | Prevented obesity by restoring the dysbiosis of gut microbiota, subsequently modulating serum bile acid and promoting GPBAR1 in BAT, thereby activating thermogenesis of BAT | [6] |
| In vivo | Western-diet-fed C57BL/6 J mice | Grape polyphenol extract (B-type proanthocyanidins) | Diet supplementation: 1% for 23 weeks | Improved body composition and reduced adiposity, which were associated with a reduced level of gut butyrate and an increased abundance of Akkermansia muciniphila | [92] |
| In vivo | HFD-fed C57BL/6 J mice | Grape seed flour | Diet supplementation: 2.5% or 7.5% for 17 weeks | Reduced body weight gain and improved lipid profiles in hepatic and serum by increasing thermogenesis of BAT and energy expenditure | [55] |
| In vivo | HFD-fed C57BL/6 J mice | Grape seed proanthocyanidin extract | Oral administration: 200 mg/kg/d for 8 weeks | Reduced overweight by increasing the thermogenesis of adipose tissue, enhancing the browning of white adipose tissue and modulating gut microbiota | [56] |
| In vivo | Cafeteria-diet-treated aged rats | Grape seed proanthocyanidin extract | Intragastric administration: 500 mg/kg for a 10-day preventive treatment or an 11-week simultaneous treatment | Simultaneous treatment effectively reduced body weight, total adiposity and liver steatosis, whereas preventive treatment only decreased mesenteric adiposity | [57] |
| Double-blind RCT | 40 obese or overweight subjects | Grape seed extract | Capsules: 300 mg grape seed extract daily for 12 weeks | Reduced body weight, BMI, waist circumference and waist-to-hip ratio | [58] |
| Cardioprotective activity | |||||
| In vitro and in vivo | EA.hy926 endothelial cells; DOCA-salt-treated SD rats | Grape extract | Culture: 0–100 μg/mL; diet supplementation: 0.03% (w/w) for 3 weeks; oral administration: 500 mg/kg for 5 d | Increased the production of NO and restored endothelial dysfunction and hypertension induced by DOCA salt by activating endothelial NO synthase and the PI3K/Akt pathway | [61] |
| In vitro and in vivo | MCEC-1 cells; mouse model of myocardial infarction | Tuscany Sangiovese pure grape juice | Cell culture: 10–100% v/v; Diet supplementation for mice: 25% v/v, 200 µL/d for 4 weeks | Protected the myocardium from an ischemic microenvironment and showed a protective effect on infarcted hearts by regulating gene expression of CNP | [7] |
| In vitro and in vivo | H9C2 cells; C57BL/6J mice with myocardial infarction | Grape seed proanthocyanidin extract | Culture: 40 μg/mL; Intragastric administration: 200 mg/kg/d for 14 days; | Improved cardiac remodeling and dysfunction induced by myocardial infarction and prevented apoptosis of cardiomyocytes under hypoxic conditions via the PI3K/Akt pathway | [63] |
| Randomized, double-blind, crossover test | Normal-body-weight and obese male subjects | Grape seed extract | 300 mg in 2 capsules per administration twice, 1 week apart | Lowered SBP and MAP in both NBW and obese males subjects; the efficacy in obese male patients might be related to a reduction in cardiac output | [64] |
| Double-blind, placebo-controlled RCT | Middle-aged participants with prehypertension | Grape seed proanthocyanidin extract | 200 or 400 mg in tablets daily for 12 weeks | 400 mg/d: lowered mean SBP by 13 mmHg; 400 mg/d and non-smoking: significantly lowered mean SBP and DBP and improved PWV, distensibility, incremental elastic modulus and stiffness parameter β | [66] |
| Double-blind, placebo-controlled RCT | Obese or overweight adults | Grape seed extract | 300 mg/d for 12 weeks | Along with a calorie-restricted diet, improved cardiovascular risk factors, such as visceral adiposity index, blood lipid profile and plasma atherogenic index | [67] |
| Antidiabetic activity | |||||
| In vitro | Amylases and α-glucosidases | Grape seed aqueous extracts (catechin and epicatechin) | Culture: 25.25 or 66.68 μg/mL | Exerted a stronger effect than acarbose in inhibiting the activities of amylases and α-glucosidases | [69] |
| In vivo | STZ-induced diabetic rats | Grape seed extract | Intragastrical administration: 0.6 mL/rat for 20 d | Improved the functions and structures of pancreas and Langerhans islets and enhanced enzyme activities | [9] |
| In vivo | STZ-induced diabetic rats | Grape seed extract | Gavage: 400 mg/kg/d for 28 d | Increased pancreatic mass | [93] |
| In vivo | HFD-fed mice | Virgin grape seed oil (polyphenols) | Diet supplementation: 29% w/w total oil for 15 weeks | Reduced blood glucose and alleviated insulin resistance | [70] |
| In vivo | HFD-fed mice | Grape seed flour | Diet supplementation: 10% for 5 weeks | Lowered fasting glucose concentration and alleviated insulin resistance | [94] |
| In vivo | Diabetic rats | Grape seed proanthocyanidin extract | Intragastric administration: 250 mg/kg/d for 16 weeks | Prevented renal damage by lowering endoplasmic reticulum stress-mediated apoptosis via the caspase-12 pathway | [71] |
| In vivo | Diabetic rats | Grape seed proanthocyanidin extract | Intragastric administration: 125 or 250 mg/kg/d for 8 weeks | Lowered blood glucose and reduced renal injury by inhibiting oxidative stress-mediated damage by activating the Nrf2 signaling pathway | [72] |
| In vivo | Diabetic rats | Grape seed extract | Intragastric administration: 250 mg/kg/d for 16 weeks | Lowered blood glucose and prevented retinal injury by decreasing retinal Muller cell gliosis and oxidative stress by activating the Nrf2 signaling pathway | [73]. |
| In vitro and in vivo | Macroglial Muller cells; diabetic mice | Grape seed proanthocyanidin extract | Culture: 10 or 20 μg/mL for 72 h; Oral gavage: 200 or 300 mg/kg/d for 10 weeks | Inhibited photoreceptor cell damage by protecting them from hyperglycemia-induced degeneration and apoptosis via the inhibition of the Trx/ASK 1/Txnip signaling pathway | [74] |
| Hepatoprotective ability | |||||
| In vivo | HFD-induced NAFLD mice | Polymerized anthocyanin from grape skin extract | Oral gavage: 400 mg/kg/d for 4 weeks | Decreased hepatic fat accumulation and steatosis, improved liver function and blood lipids and regulated lipid metabolism | [75] |
| In vivo | Albino rabbits additionally administered 10 mL egg yolk and 1.5 g pure cholesterol | Grape leucoanthocyanidin | Oral gavage: 50 mg/kg/d for 100 d | Recovered hepatic tissue and reduced hepatic steatosis, which is reflected a reduced NAFLD activity score from 6 to 4 | [76] |
| In vivo | CCl4-treated Kunming mice | Grape seed proanthocyanidins | Oral gavage: 50, 250 or 500 mg/kg for 10 d | Protected liver from acute injury by scavenging free radicals, inhibiting lipid peroxidation, preserving immune function and improving antioxidant capacity | [77] |
| In vitro and in vivo | Lead-acetate-treated primary hepatocytes and rats | Grape seed procyanidin extract | Culture: 100 μg/mL for 2 h; Oral gavage: 200 mg/kg/d for 8 weeks | Increased cell viability, inhibited LDH release and decreased ROS levels in primary hepatocytes and alleviated liver injury in rats by activating the Nrf2 pathway | [78] |
| In vivo | Doxorubicin-treated rats | Grape seed and skin extract (polyphenols) | Oral gavage: 500 mg/kg/d for 8 d | Prevented hepatotoxicity induced by doxorubicin | [79] |
| Anticancer activity | |||||
| In vivo | Mice inoculated with Ehrlich ascites carcinoma | Mixed powder of grape seeds and grape skin | Diet supplementation: 10% (w/w) 14 d before inoculation and continued throughout the experiment | Prevented 47% of tumor development and reduced volume and weight of tumors by 93.9% and 86.3%, respectively, by inhibiting cell proliferation, inducing cell cycle arrest in the G1 phase and promoting apoptosis | [10] |
| In vitro and in vivo | HeLa cell lines; HeLa-derived xenograft tumors in zebrafish | Lipophilic grape seed proanthocyanidin | Cell culture: 25–200 µg/mL for 24 and 48 h; Fish water: 4 and 8 μg/mL for 48 h | Induced cell cycle arrest in the G2/M phase, promoted apoptosis in vitro by increasing ROS levels and inhibited cancer growth in zebrafish | [81] |
| In vitro | Hepatocellular carcinoma cells | Grape seed proanthocyanidin extract | Culture: 12.5, 25, 50 and 100 µg/mL for 24 or 48 h | Induced apoptosis and inhibited cancer cell growth by inhibiting the MAPK/Akt pathway | [82] |
| In vitro and in vivo | HepG2 cells; HepG2-derived mouse xenograft model | Grape seed proanthocyanidins | Culture: 10 mg/L for 24 h; Oral gavage: 100 and 200 mg/kg | Induced autophagy/apoptosis, reduced survivin expression and inhibited cancer cell growth via the MAPK pathway | [83] |
| In vivo | Pten-deficient mice | Grape powder | Diet supplementation: 10% for 33 weeks | Reduced angiogenesis, attenuated inflammation, improved the prostate neoplastic phenotype, inhibited hyperactive cell survival via the Akt and AR pathways and reduced circulating levels of oncogenic microRNAs | [84] |
| In vitro | T24 and 5637 bladder cancer cells | Grape seed proanthocyanidins | Culture: 0–200 μg/mL for 24, 48 or 72 h | Suppressed migration and invasion by reversing EMT via inhibition of the TGF-β pathway | [85] |
| In vitro | Human leukemia cell line HL-60 and HL-ADR cells | Grape seed proanthocyanidin extract | Culture: 25 μg/mL for 24 h | Reversed the MDR of cancer cells to some drugs by inhibiting the PI3K/Akt pathway and down-regulating the expression of MDR-associated protein 1, MDR protein 1 and lung resistance-related protein | [86] |
| In vitro and in vivo | Acquired and innately chemo-resistant colorectal cancer cells (HCT116-FOr cells and H716 cells); HCT116-FOr xenograft mice | Oligomeric proanthocyanidins from grape seed extract | Culture: 100 ng/µL for 48 h; Oral gavage: 100 mg/kg for 6 weeks | Sensitized cancer cells to oxaliplatin and 5-fluorouracil (chemotherapeutic drugs) and decreased chemoresistant xenograft tumor growth in mice via the inhibition of ABC transporter proteins | [87] |
| Other health benefits | |||||
| In vitro and in vivo | Dopamine neurons; Parkinson′s disease mouse model | Red grape seed and skin extract | Culture: 500 and 1000 μg/mL; Intraperitoneal injection: 250 mg/kg in 10% ethanol | Protected dopamine neurons from 6-OHDA-induced toxicity by reducing apoptosis, oxidative stress and inflammation; prevented neuronal loss; and improved motor function in a Parkinson’s disease mouse model | [88] |
| In vivo | Aged female rats | Grape seed procyanidin extract | Diet supply: 500 mg/kg for 10 d | Prevented certain aging processes, such as visceral adiposity, pancreas dysfunction and age-related tumor development | [89] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, D.-D.; Li, J.; Xiong, R.-G.; Saimaiti, A.; Huang, S.-Y.; Wu, S.-X.; Yang, Z.-J.; Shang, A.; Zhao, C.-N.; Gan, R.-Y.; et al. Bioactive Compounds, Health Benefits and Food Applications of Grape. Foods 2022, 11, 2755. https://doi.org/10.3390/foods11182755
Zhou D-D, Li J, Xiong R-G, Saimaiti A, Huang S-Y, Wu S-X, Yang Z-J, Shang A, Zhao C-N, Gan R-Y, et al. Bioactive Compounds, Health Benefits and Food Applications of Grape. Foods. 2022; 11(18):2755. https://doi.org/10.3390/foods11182755
Chicago/Turabian StyleZhou, Dan-Dan, Jiahui Li, Ruo-Gu Xiong, Adila Saimaiti, Si-Yu Huang, Si-Xia Wu, Zhi-Jun Yang, Ao Shang, Cai-Ning Zhao, Ren-You Gan, and et al. 2022. "Bioactive Compounds, Health Benefits and Food Applications of Grape" Foods 11, no. 18: 2755. https://doi.org/10.3390/foods11182755
APA StyleZhou, D.-D., Li, J., Xiong, R.-G., Saimaiti, A., Huang, S.-Y., Wu, S.-X., Yang, Z.-J., Shang, A., Zhao, C.-N., Gan, R.-Y., & Li, H.-B. (2022). Bioactive Compounds, Health Benefits and Food Applications of Grape. Foods, 11(18), 2755. https://doi.org/10.3390/foods11182755







