Effect of High Hydrostatic Pressure and Pulsed Electric Fields Processes on Microbial Safety and Quality of Black/Red Raspberry Juice
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Plant Material and Juice Preparation
2.3. Physicochemical Analysis
2.4. HHP Treatments
2.5. PEF Treatments
2.6. Microbiological Evaluation
2.7. Pectin Methylesterase (PME)
Wt
PME untreated sample
2.8. Vitamin C
2.9. Total Phenolic Compounds (TPC)
2.10. Statistical Analyses
3. Results
3.1. Inactivation of Molds and Yeast by HHP
3.2. Inactivation of Molds and Yeast by PEF
3.3. Physicochemical Parameters of the Black/Red Raspberry Juice
3.3.1. pH Value
3.3.2. % Acidity
3.3.3. TSS (°Brix)
3.3.4. Water Activity (aw)
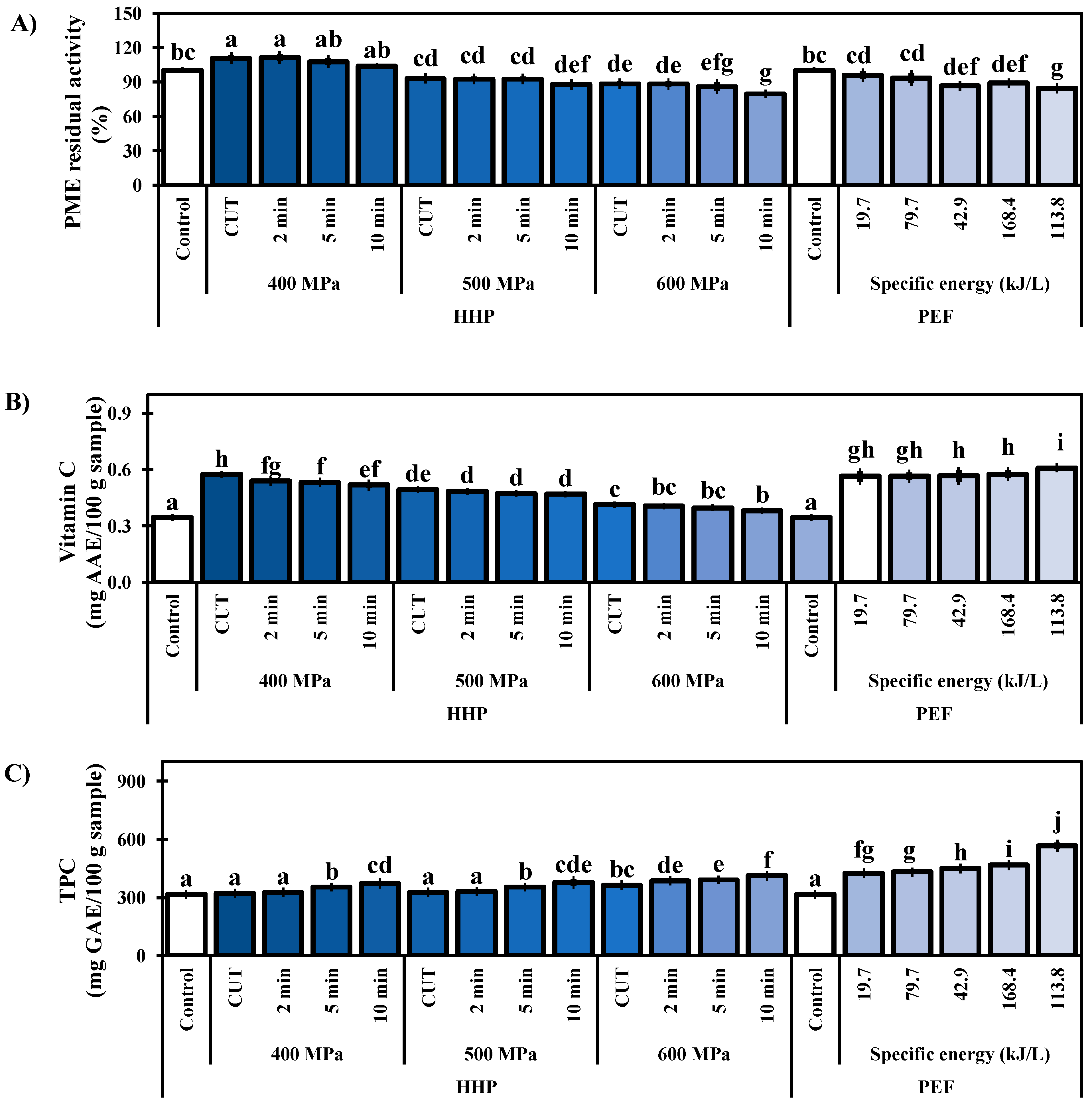
3.4. PME Activity
3.5. Vitamin C Content
3.6. Total Phenolic Content
4. Discussion
Putative Effect of the HHP and PEF on Black/Red Raspberry Juice
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pohlmann, A.R.; Fonseca, F.N.; Paese, K.; Detoni, C.B.; Coradini, K.; Beck, R.C.; Guterres, S.S. Poly (ϵ-caprolactone) microcapsules and nanocapsules in drug delivery. Expert Opin. Drug Deliv. 2013, 10, 623–638. [Google Scholar] [CrossRef] [PubMed]
- Hurtado, A.; Dolors Guàrdia, M.; Picouet, P.; Jofré, A.; Bañón, S.; Ros, J.M. Shelf-life extension of multi-vegetables smoothies by high-pressure processing compared with thermal treatment. Part I: Microbial and enzyme inhibition, antioxidant status, and physical stability. J. Food Process. Preserv. 2019, 43, e14139. [Google Scholar] [CrossRef]
- Skrovankova, S.; Sumczynski, D.; Mlcek, J.; Jurikova, T.; Sochor, J. Bioactive compounds and antioxidant activity in different types of berries. Int. J. Mol. Sci. 2015, 16, 24673–24706. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Akimov, M.Y.; Bessonov, V.V.; Kodentsova, V.M.; Eller, K.I.; Vrzhesinskaya, O.A.; Beketova, N.A.; Kosheleva, O.V.; Bogachuk, M.N.; Malinkin, A.D.; Makarenko, M.A.; et al. Biological value of fruits and berries of Russian production. Probl. Nutr. 2020, 89, 220–232. [Google Scholar]
- Oliveira, M.; Rodrigues, C.M.; Teixeira, P. Microbiological quality of raw berries and their products: A focus on foodborne pathogens. Heliyon 2019, 5, e02992. [Google Scholar] [CrossRef] [Green Version]
- Gonzalez, M.E.; Barrett, D.M. Thermal, high pressure, and electric field processing effects on plant cell membrane integrity and relevance to fruit and vegetable quality. J. Food Sci. 2010, 75, R121–R130. [Google Scholar] [CrossRef] [Green Version]
- Morata, A. Emerging trends in beverage processing. Beverages 2021, 7, 8. [Google Scholar] [CrossRef]
- Welti-Chanes, J.; Ochoa-Velasco, C.E.; Guerrero-Beltrán, J.Á. High-pressure homogenization of orange juice to inactivate pectinmethylesterase. Innov. Food Sci. Emerg. Technol. 2009, 10, 457–462. [Google Scholar] [CrossRef]
- Escobedo-Avellaneda, Z.; Moure, M.P.; Chotyakul, N.; Torres, J.A.; Welti-Chanes, J.; Lamela, C.P. Benefits and limitations of food processing by high-pressure technologies: Effects on functional compounds and abiotic contaminants Beneficios y limitaciones del procesamiento de alimentos por tecnologías de alta presión: Efectos en componentes funcionales y contaminantes abióticos. CyTa-J. Food. 2011, 9, 351–364. [Google Scholar]
- Escobedo-Avellaneda, Z.; Pérez-Simón, I.; Lavilla-Martín, M.; Baranda-González, A.; Welti-Chanes, J. Enzymatic and phytochemical stabilization of orange–strawberry–banana beverages by high hydrostatic pressure and mild heat. Food Sci. Technol. Int. 2017, 23, 185–193. [Google Scholar] [CrossRef]
- Buitimea-Cantúa, G.V.; Welti-Chanes, J.; Escobedo-Avellaneda, Z. Metabolite transformation and β-D-glucosidase activity during the high hydrostatic pressure assisted the curing process of vanilla beans (Vanilla planifolia) to improve phenolic compounds formation. Food Chem. 2022, 384, 132497. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Maqueo, A.; Welti-Chanes, J.; Cano, M.P. Release mechanisms of bioactive compounds in fruits submitted to high hydrostatic pressure: A dynamic microstructural analysis based on prickly pear cells. Food Resch. Int. 2020, 130, 108909. [Google Scholar] [CrossRef] [PubMed]
- Nowosad, K.; Sujka, M.; Pankiewicz, U.; Kowalski, R. The application of PEF technology in food processing and human nutrition. J. Food Sci, Tech. 2021, 58, 397–411. [Google Scholar] [CrossRef] [PubMed]
- Genovese, J.; Kranjc, M.; Serša, I.; Petracci, M.; Rocculi, P.; Miklavčič, D.; Mahnič-Kalamiza, S. PEF-treated plant and animal tissues: Insights by approaching with different electroporation assessment methods. Innov. Food Sci. Emerg. Technol. 2021, 74, 102872. [Google Scholar] [CrossRef]
- Soliva-Fortuny, R.; Balasa, A.; Knorr, D.; Martín-Belloso, O. Effects of pulsed electric fields on bioactive compounds in foods: A review. Trends Food Sci. Technol. 2009, 20, 544–556. [Google Scholar] [CrossRef]
- Mor-Mur, M. High pressure treated food products. Nutritional aspects. Act. Diet. 2020, 14, 53–58. [Google Scholar]
- Aguiló-Aguayo, I.; Sobrino-López, Á.; Soliva-Fortuny, R.; Martín-Belloso, O. Influence of high-intensity pulsed electric field processing on lipoxygenase and β-glucosidase activities in strawberry juice. Innov. Food Sci. Emerg. Technol. 2008, 9, 455–462. [Google Scholar] [CrossRef]
- Patras, A.; Brunton, N.P.; Da Pieve, S.; Butler, F. Impact of high pressure processing on total antioxidant activity, phenolic, ascorbic acid, anthocyanin content and colour of strawberry and blackberry purées. Innov. Food Sci. Emerg. Technol. 2009, 10, 308–313. [Google Scholar] [CrossRef]
- Gurtler, J.B.; Bailey, R.B.; Geveke, D.J.; Zhang, H.Q. Pulsed electric field inactivation of E. coli O157: H7 and non-pathogenic surrogate E. coli in strawberry juice as influenced by sodium benzoate, potassium sorbate, and citric acid. Food Control. 2011, 22, 1689–1694. [Google Scholar] [CrossRef]
- Geveke, D.J.; Aubuchon, I.; Zhang, H.Q.; Boyd, G.; Sites, J.E.; Bigley, A.B. Validation of a pulsed electric field process to pasteurize strawberry purée. J. Food Eng. 2015, 166, 384–389. [Google Scholar] [CrossRef]
- Aaby, K.; Grimsbo, I.H.; Hovda, M.B.; Rode, T.M. Effect of high pressure and thermal processing on shelf life and quality of strawberry purée and juice. Food Chem. 2018, 260, 115–123. [Google Scholar] [CrossRef]
- Yildiz, S.; Pokhrel, P.R.; Unluturk, S.; Barbosa-Cánovas, G.V. Identification of equivalent processing conditions for pasteurization of strawberry juice by high pressure, ultrasound, and pulsed electric fields processing. Innov. Food Sci. Emerg. Technol. 2019, 57, 102195. [Google Scholar] [CrossRef]
- Yildiz, S.; Pokhrel, P.R.; Unluturk, S.; Barbosa-Cánovas, G.V. Shelf life extension of strawberry juice by equivalent ultrasound, high pressure, and pulsed electric fields processes. Food Res. Int. 2021, 140, 110040. [Google Scholar] [CrossRef] [PubMed]
- Castaldo, D.; Laratta, B.; Loiudice, R.; Giovane, A.; Quagliuolo, L.; Servillo, L. Presence of residual pectin methylesterase activity in thermally stabilized industrial fruit preparations. LWT-Food Sci. Technol. 1997, 30, 479–484. [Google Scholar] [CrossRef] [Green Version]
- Croak, S.; Corredig, M. The role of pectin in orange juice stabilization: Effect of pectin methylesterase and pectinase activity on the size of cloud particles. Food Hydrocoll 2006, 20, 961–965. [Google Scholar] [CrossRef]
- Bermejo-Prada, A.; Segovia-Bravo, K.A.; Guignon, B.; Otero, L. Effect of hyperbaric storage at room temperature on pectin methylesterase activity and serum viscosity of strawberry juice. Innov. Food Sci. Emerg. Technol. 2015, 30, 170–176. [Google Scholar] [CrossRef]
- NOM-092-SSA1-1994; Bienes y Servicios. Método Para la Cuenta de Bacterias Aerobias en Placa. NORMA Oficial Mexicana: Ciudad de México, México, 1994.
- NOM-110-SSA1-1994; Bienes y Servicios. Preparación y Dilución de Muestras de Alimentos Para su Análisis Microbiológico. NORMA Oficial Mexicana: Ciudad de México, México, 1994.
- US-FDA. Kinetics of Microbial Inactivation for Alternative Food Processing Technologies-High Pressure Processing; United Sattes Food and Drug Administration: Silver Spring, MD, USA, 2014; p. 55.
- Escobedo-Avellaneda, Z.; Gutiérrez-Uribe, J.; Valdez-Fragoso, A.; Torres, J.A.; Welti-Chanes, J. Phytochemicals and antioxidant activity of juice, flavedo, albedo and comminuted orange. J. Funct. Foods 2014, 6, 470–481. [Google Scholar] [CrossRef]
- Daher, D.; le Gourrierec, S.; Pérez-Lamela, C. Effect of High Pressure Processing on the Microbial Inactivation in Fruit Preparations and Other Vegetable Based Beverages. Agriculture 2017, 7, 72. [Google Scholar] [CrossRef] [Green Version]
- Cheftel, J.C. High-pressure, microbial inactivation and food preservation. Food Sci. Technol. 1995, 1, 2–3. [Google Scholar] [CrossRef]
- Li, L.; Yang, R.; Zhao, W. The Effect of Pulsed Electric Fields (PEF) Combined with Temperature and Natural Preservatives on the Quality and Microbiological Shelf-Life of Cantaloupe Juice. Foods 2021, 10, 2606. [Google Scholar] [CrossRef]
- Esser, A.T.; Smith, K.C.; Gowrishankar, T.R.; Weaver, J.C. Towards solid tumor treatment by irreversible electroporation: Intrinsic redistribution of fields and currents in tissue. Technol. Cancer Res. Treat. 2007, 6, 261–273. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weaver, J.C. Electroporation of biological membranes from multicellular to nano scales. IEEE Trans. Dielectr. Electr. Insul. 2003, 10, 754–768. [Google Scholar] [CrossRef]
- Garcia-Rubio, R.; de Oliveira, H.C.; Rivera, J.; Trevijano-Contador, N. The fungal cell wall: Candida, Cryptococcus, and Aspergillus species. Front. Microbiol. 2020, 10, 2993. [Google Scholar] [CrossRef] [PubMed]
- Aganovic, K.; Hertel, C.; Vogel, R.F.; Johne, R.; Schlüter, O.; Schwarzenbolz, U.; Jäger, H.; Holzhauser, T.; Bergmair, J.; Roth, A.; et al. Aspects of high hydrostatic pressure food processing: Perspectives on technology and food safety. Compr. Rev. Food Sci. Food Saf. 2021, 20, 3225–3266. [Google Scholar] [CrossRef]
- Giampieri, F.; Forbes-Hernandez, T.Y.; Gasparrini, M.; Alvarez-Suarez, J.M.; Afrin, S.; Bompadre, S.; Quiles, J.L.; Mezzetti, B.; Battino, M. Strawberry as a health promoter: An evidence based review. Food Funct. 2015, 6, 1386–1398. [Google Scholar] [CrossRef] [Green Version]
- Sadler, G.D.; Murphy, P.A. pH and titratable acidity. Food Anal. 2010, 219–238. [Google Scholar] [CrossRef]
- Hussain, I.; Zeb, A.; Shakir, I.; Shah, A.S. Combined effect of potassium sorbate and sodium benzoate on individual and blended juices of apricot and apple fruits grown in Azad Jammu and Kashmir. Pak. J. Nutr. 2008, 7, 181–185. [Google Scholar] [CrossRef] [Green Version]
- Iqbal, S.A.; Yasmin, S.; Wadud, S.; Shah, W.H. Production storage packing and quality evaluation of guava nectar. Pak. J. Food Sci. 2001, 11, 33–36. [Google Scholar]
- Jouquand, C.; Chandler, C.; Plotto, A.; Goodner, K. A sensory and chemical analysis of fresh strawberries over harvest dates and seasons reveals factors that affect eating quality. J. Am. Soc. Hortic. Sci. 2008, 133, 859–867. [Google Scholar] [CrossRef] [Green Version]
- Pistón, F.; Pérez, A.G.; Sanz, C.; Refoyo, A. Relationship between sugar content and Brix as influenced by cultivar and ripening stages of strawberry. In Proceedings of the VIII International Strawberry Symposium, Québec City, QC, Canada, 13–17 August 2016; Volume 1156, pp. 491–496. [Google Scholar]
- Menéndez A., O.; Lozano, S.E.; Ocampo, M.L.A.; Torres, K.B.; del Villar Martínez, A.A.; Aparicio, A.R.J. Cambios en la actividad de a-amilasa, pectinmetilesterasa y poligalacturonasa durante la maduración del maracuyá amarillo (Passiflora edulis var. flavicarpa Degener). Interciencia 2006, 1, 728–733. [Google Scholar]
- Franco, G.; Valenzuela, J.R.C.; Londoño, G.A.C.; Rojano, B.A.; Correa, A.M.P.; Arias, M.L. Physicochemical properties of gulupa fruits (Passiflora edulis Sims) during pre and postharvest. Rev. Ibe. Tec. Post. 2014, 15, 15–30. [Google Scholar]
- Quek, S.Y.; Chok, N.K.; Swedlund, P. The physicochemical properties of spray-dried watermelon powders. Chem. Eng. Process. 2007, 46, 386–392. [Google Scholar] [CrossRef]
- Sandulachi, E. Water activity concept and its role in food preservation. Meridian Ing. 2012, 4, 40–48. [Google Scholar]
- Anese, M.; NicolI, M.C.; Dall’aglio, G.; Lerici, C.R. Effect of high pressure treatments on peroxidase and polyphenoloxidase activities. J. Food Biochem. 1994, 18, 285–293. [Google Scholar] [CrossRef]
- Butz, P.; García, A.F.; Lindauer, R.; Dieterich, S.; Bognar, A.; Tauscher, B. Influence of ultra high pressure processing on fruit and vegetable products. J. Food Eng. 2003, 56, 233–236. [Google Scholar] [CrossRef]
- Basak, S.; Ramaswamy, H.S. Effect of high pressure processing on the texture of selected fruits and vegetables. J. Texture Stud. 1998, 29, 587–601. [Google Scholar] [CrossRef]
- Chakraborty, S.; Mishra, H.N.; Knorr, D. Strawberry Enzyme Inactivation by HPP: Models and Contours; LAP LAMBERT Academic Publishing: Chisinau, Republic of Moldova, 2012. [Google Scholar]
- Ali, N.; Popović, V.; Koutchma, T.; Warriner, K.; Zhu, Y. Effect of thermal, high hydrostatic pressure, and ultraviolet-C processing on the microbial inactivation, vitamins, chlorophyll, antioxidants, enzyme activity, and color of wheatgrass juice. J. Food Process. Eng. 2020, 43, e13036. [Google Scholar] [CrossRef]
- Horemans, N.; Foyer, C.H.; Potters, G.; Asard, H. Ascorbate function and associated transport systems in plants. Plant. Physiol. Biochem. 2000, 38, 531–540. [Google Scholar] [CrossRef]
- Escobedo-Avellaneda, Z.; Gutiérrez-Uribe, J.; Valdez-Fragoso, A.; Torres, J.A.; Welti-Chanes, J. High hydrostatic pressure combined with mild temperature for the preservation of comminuted orange: Effects on functional compounds and antioxidant activity. Food Bioprocess Technol. 2015, 8, 1032–1044. [Google Scholar] [CrossRef]
- Ribas-Agustí, A.; Martín-Belloso, O.; Soliva-Fortuny, R.; Elez-Martínez, P. Food processing strategies to enhance phenolic compounds bioaccessibility and bioavailability in plant-based foods. Crit. Rev. Food Sci. Nutr. 2018, 58, 2531–2548. [Google Scholar] [CrossRef] [Green Version]
- Navarro-Baez, J.E.; Martínez, L.M.; Welti-Chanes, J.; Buitimea-Cantúa, G.V.; Escobedo-Avellaneda, Z. High Hydrostatic Pressure to Increase the Biosynthesis and Extraction of Phenolic Compounds in Food: A Review. Molecules 2022, 27, 1502. [Google Scholar] [CrossRef] [PubMed]
- Bouarab-Chibane, L.; Forquet, V.; Lantéri, P.; Clément, Y.; Léonard-Akkari, L.; Oulahal, N.; Degraeve, P.; Bordes, C. Antibacterial properties of polyphenols: Characterization and QSAR (Quantitative structure–activity relationship) models. Front. Microbiol. 2019, 10, 829. [Google Scholar] [CrossRef] [PubMed]
- Mousavi, S.; Bereswill, S.; Heimesaat, M.M. Immunomodulatory and antimicrobial effects of vitamin C. Eur. J. Microbiol. Immunol. 2019, 9, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Wormit, A.; Usadel, B. The multifaceted role of pectin methylesterase inhibitors (PMEIs). Int. J. Mol. Sci. 2018, 19, 2878. [Google Scholar] [CrossRef] [Green Version]
- Fraeye, I.; Duvetter, T.; Doungla, E.; Van Loey, A.; Hendrickx, M. Fine-tuning the properties of pectin–calcium gels by control of pectin fine structure, gel composition and environmental conditions. Trends Food Sci. Technol. 2010, 21, 219–228. [Google Scholar] [CrossRef]
- Ellerbee, L.; Wicker, L. Calcium and pH influence on orange juice cloud stability. J. Sci Food Agric. 2011, 91, 171–177. [Google Scholar] [CrossRef]
- Chakraborty, S.; Kaushik, N.; Rao, P.S.; Mishra, H.N. High-pressure inactivation of enzymes: A review on its recent applications on fruit purees and juices. Compr. Rev. Food Sci. Food Saf. 2014, 13, 578–596. [Google Scholar] [CrossRef]
- Galanakis, C.M. Functionality of food components and emerging technologies. Foods 2021, 10, 128. [Google Scholar] [CrossRef]
- Poojary, M.; Roohinejad, S.; Koubaa, M.; Barba, F.; Passamonti, P.; Jambrak, A.R.; Oey, I.; Greiner, R. Impact of Pulsed Electric Fields on Enzymes. In Handbook of Electroporation; Miklavcic, D., Ed.; Springer International Publishing: Cham, Switzerland, 2016; pp. 1–21. [Google Scholar]
- Patterson, M.F. Microbiology of pressure-treated foods. J. Appl. Microb. 2005, 98, 1400–1409. [Google Scholar] [CrossRef]

| High Hydrostatic Pressure (HHP) | |||||
|---|---|---|---|---|---|
| Conditions | Molds | Yeast | |||
| Pressure (MPa) | Time (min) | CFU/mL | Log (Nt/N0) | CFU/mL | Log (Nt/N0) |
| Control | 5.30 × 103 ± 0.01 c | 3.72 ± 0.01 c | 1.55 × 103 ± 0.12 b | 3.19 ± 0.00 b | |
| 400 | CUT | 7.50 × 101 ± 0.21 b | −1.85 ± 0.21 b | 1.00 × 100 ± 0.00 a | −3.19 ± 0.00 a |
| 2 | 1.00 × 100 ± 0.00 a | −3.72 ± 0.00 a | 1.00 × 100 ± 0.00 a | −3.19 ± 0.00 a | |
| 5 | 1.00 × 100 ± 0.00 a | −3.72 ± 0.00 a | 1.00 × 100 ± 0.00 a | −3.19 ± 0.00 a | |
| 10 | 1.00 × 100 ± 0.00 a | −3.72 ± 0.00 a | 1.00 × 100 ± 0.00 a | −3.19 ± 0.00 a | |
| 500 | CUT | 1.00 × 100 ± 0.00 a | −3.72 ± 0.00 a | 1.00 × 100 ± 0.00 a | −3.19 ± 0.00 a |
| 2 | 1.00 × 100 ± 0.00 a | −3.72 ± 0.00 a | 1.00 × 100 ± 0.00 a | −3.19 ± 0.00 a | |
| 5 | 1.00 × 100 ± 0.00 a | −3.72 ± 0.00 a | 1.00 × 100 ± 0.00 a | −3.19 ± 0.00 a | |
| 10 | 1.00 × 100 ± 0.00 a | −3.72 ± 0.00 a | 1.00 × 100 ± 0.00 a | −3.19 ± 0.00 a | |
| 600 | CUT | 1.00 × 100 ± 0.00 a | −3.72 ± 0.00 a | 1.00 × 100 ± 0.00 a | −3.19 ± 0.00 a |
| 2 | 1.00 × 100 ± 0.00 a | −3.72 ± 0.00 a | 1.00 × 100 ± 0.00 a | −3.19 ± 0.00 a | |
| 5 | 1.00 × 100 ± 0.00 a | −3.72 ± 0.00 a | 1.00 × 100 ± 0.00 a | −3.19 ± 0.00 a | |
| 10 | 1.00 × 100 ± 0.00 a | −3.72 ± 0.00 a | 1.00 × 100 ± 0.00 a | −3.19 ± 0.00 a | |
| Pulsed Electric Field (PEF) | |||||
| Conditions | Molds | Yeast | |||
| Frequency (Hz) | Specific Energy (kJ/L) | CFU/mL | Log (Nt/N0) | CFU/mL | Log (Nt/N0) |
| Control | 5.30 × 103 ± 0.01 e | 3.72 ± 0.01 e | 1.55 × 103 ± 0.002 c | 3.19 ± 0.00 c | |
| 100 | 19.7 | 3.45 × 103 ± 0.03 e | −0.19 ± 0.03 e | 1.25 × 103 ± 0.07 c | −0.09 ± 0.007 c |
| 79.7 | 2.50 × 102 ± 0.002 c | −1.33 ± 0.002 c | 6.50 × 101 ± 0.14 b | −1.38 ± 0.14 b | |
| 200 | 42.9 | 6.30 × 102 ± 0.06 d | −0.92 ± 0.06 d | 1.00 × 100 ± 0.00 a | −3.19 ± 0.00 a |
| 168.4 | 4.00 × 101 ± 0.16 a | −2.12 ± 0.16 a | 1.00 × 100 ± 0.00 a | −3.19 ± 0.00 a | |
| 500 | 113.8 | 1.00 × 102 ± 0.12 b | −1.72 ± 0.12 b | 1.00 × 100 ± 0.00 a | −3.19 ± 0.00 a |
| High Hydrostatic Pressure (HHP) | |||||
|---|---|---|---|---|---|
| Conditions | pH | Titratable Acidity (% Citric Acid) | Water Activity (aw) | Total Soluble Solids (°Brix) | |
| Pressure (MPa) | Time (min) | ||||
| Control | 1.00 ± 0.00 c | 0.98 ± 0.00 b | 7.5 ± 0.00 a | ||
| 400 | CUT | 3.43 ± 0.01 g | 0.80 ± 0.00 a | 0.98 ± 0.00 a, b | 8.0 ± 0.00 b |
| 2 | 3.41 ± 0.01 f, g | 0.80 ± 0.00 a | 0.97 ± 0.00 a, b | 8.0 ± 0.00 b | |
| 5 | 3.36 ± 0.01 c | 0.80 ± 0.00 a | 0.98 ± 0.00 a, b | 8.0 ± 0.00 b | |
| 10 | 3.41 ± 0.01 f | 0.80 ± 0.00 a | 0.98 ± 0.00 b | 8.0 ± 0.00 b | |
| 500 | CUT | 3.45 ± 0.00 h | 0.80 ± 0.00 a | 0.97 ± 0.00 a, b | 8.0 ± 0.00 b |
| 2 | 3.42 ± 0.00 f, g | 0.80 ± 0.00 a | 0.97 ± 0.00 a, b | 8.0 ± 0.00 b | |
| 5 | 3.41 ± 0.00 f | 0.80 ± 0.00 a | 0.98 ± 0.00 a, b | 8.0 ± 0.00 b | |
| 10 | 3.39 ± 0.00 e | 0.80 ± 0.00 a | 0.98 ± 0.00 a, b | 8.0 ± 0.00 b | |
| 600 | CUT | 3.41 ± 0.00 f | 0.80 ± 0.00 a | 0.97 ± 0.00 a, b | 8.0 ± 0.00 b |
| 2 | 3.39 ± 0.00 e | 0.80 ± 0.00 a | 0.97 ± 0.00 a, b | 8.0 ± 0.00 b | |
| 5 | 3.41 ± 0.00 f | 0.80 ± 0.00 a | 0.96 ± 0.00 a, | 8.0 ± 0.00 b | |
| 10 | 3.43 ± 0.00 g | 0.80 ± 0.00 a | 0.97 ± 0.00 a, b | 8.0 ± 0.00 b | |
| Pulsed Electric Field (PEF) | |||||
| Conditions | pH | Titratable Acidity (% Citric Acid) | Water Activity (aw) | Total Soluble Solids (°Brix) | |
| Frequency (Hz) | Specific Energy (kJ/L) | ||||
| Control | 3.31 ± 0.00 b | 1.00 ± 0.00 c | 0.97 ± 0.00 a, b | 7.5 ± 0.00 a | |
| 100 | 19.7 | 3.38 ± 0.00 d, e | 0.90 ± 0.00 b | 0.98 ± 0.00 b | 8.5 ± 0.00 c |
| 79.7 | 3.38 ± 0.00 d, e | 0.90 ± 0.00 b | 0.98 ± 0.00 b | 8.5 ± 0.00 c | |
| 200 | 42.9 | 3.36 ± 0.00 c | 0.90 ± 0.00 b | 0.97 ± 0.00 a, b | 8.5 ± 0.00 c |
| 168.4 | 3.37 ± 0.00 c, d | 0.90 ± 0.00 b | 0.98 ± 0.00 b | 8.5 ± 0.00 c | |
| 500 | 113.8 | 3.38 ± 0.00 d, e | 0.90 ± 0.00 b | 0.98 ± 0.00 b | 8.5 ± 0.00 c |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buitimea-Cantúa, G.V.; Rico-Alderete, I.A.; Rostro-Alanís, M.d.J.; Welti-Chanes, J.; Escobedo-Avellaneda, Z.J.; Soto-Caballero, M.C. Effect of High Hydrostatic Pressure and Pulsed Electric Fields Processes on Microbial Safety and Quality of Black/Red Raspberry Juice. Foods 2022, 11, 2342. https://doi.org/10.3390/foods11152342
Buitimea-Cantúa GV, Rico-Alderete IA, Rostro-Alanís MdJ, Welti-Chanes J, Escobedo-Avellaneda ZJ, Soto-Caballero MC. Effect of High Hydrostatic Pressure and Pulsed Electric Fields Processes on Microbial Safety and Quality of Black/Red Raspberry Juice. Foods. 2022; 11(15):2342. https://doi.org/10.3390/foods11152342
Chicago/Turabian StyleBuitimea-Cantúa, Génesis V., Iván Alejandro Rico-Alderete, Magdalena de Jesús Rostro-Alanís, Jorge Welti-Chanes, Zamantha J. Escobedo-Avellaneda, and Mayra Cristina Soto-Caballero. 2022. "Effect of High Hydrostatic Pressure and Pulsed Electric Fields Processes on Microbial Safety and Quality of Black/Red Raspberry Juice" Foods 11, no. 15: 2342. https://doi.org/10.3390/foods11152342
APA StyleBuitimea-Cantúa, G. V., Rico-Alderete, I. A., Rostro-Alanís, M. d. J., Welti-Chanes, J., Escobedo-Avellaneda, Z. J., & Soto-Caballero, M. C. (2022). Effect of High Hydrostatic Pressure and Pulsed Electric Fields Processes on Microbial Safety and Quality of Black/Red Raspberry Juice. Foods, 11(15), 2342. https://doi.org/10.3390/foods11152342








