Effects of Different Amounts of Corn Silk Polysaccharide on the Structure and Function of Peanut Protein Isolate Glycosylation Products
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Chemicals
2.2. Preparation of PPI-CSP Complexes
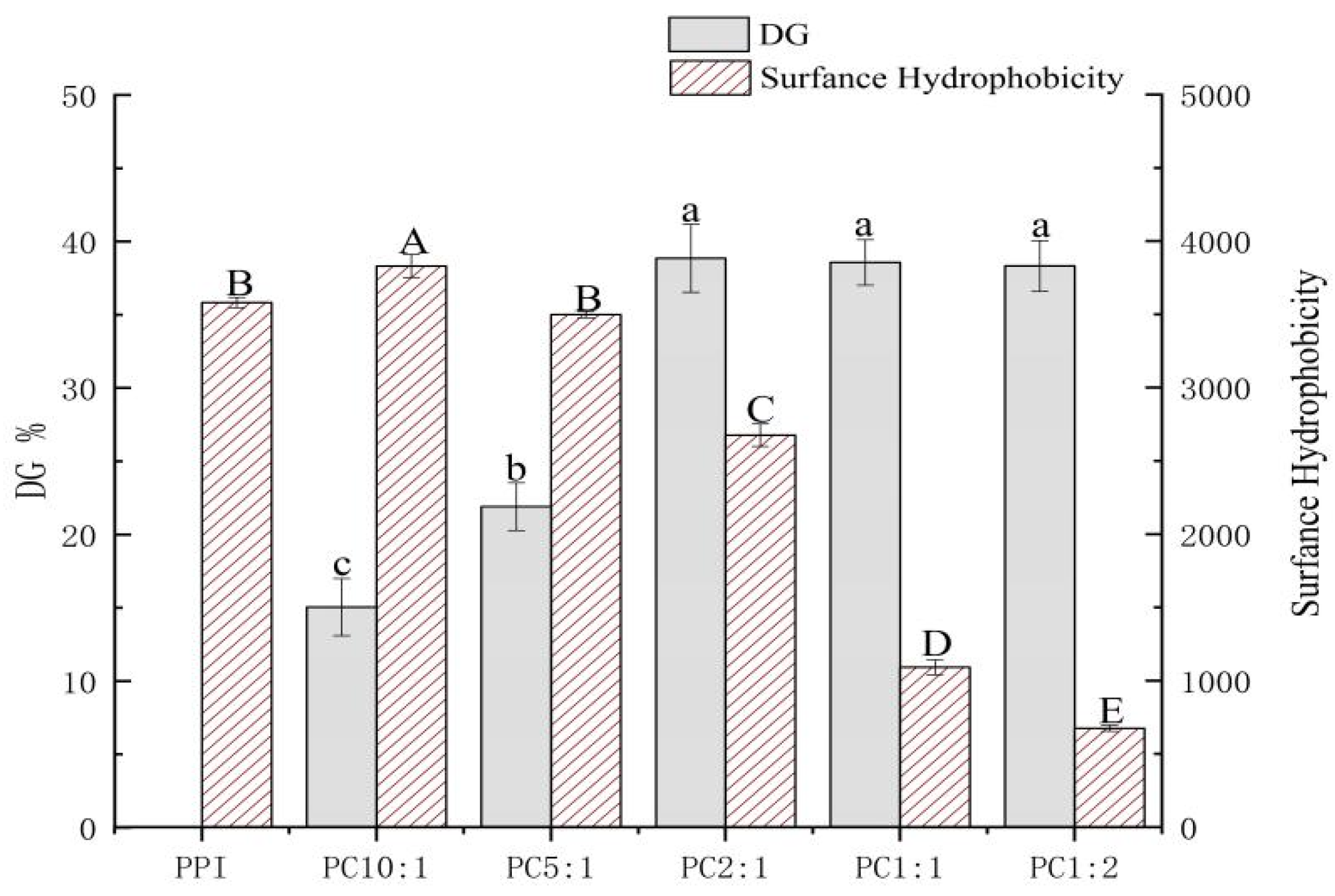
2.3. Degree of Graft (DG)
2.4. Measurement of Surface Hydrophobicity (H0)
2.5. Differential Scanning Calorimetry (DSC)
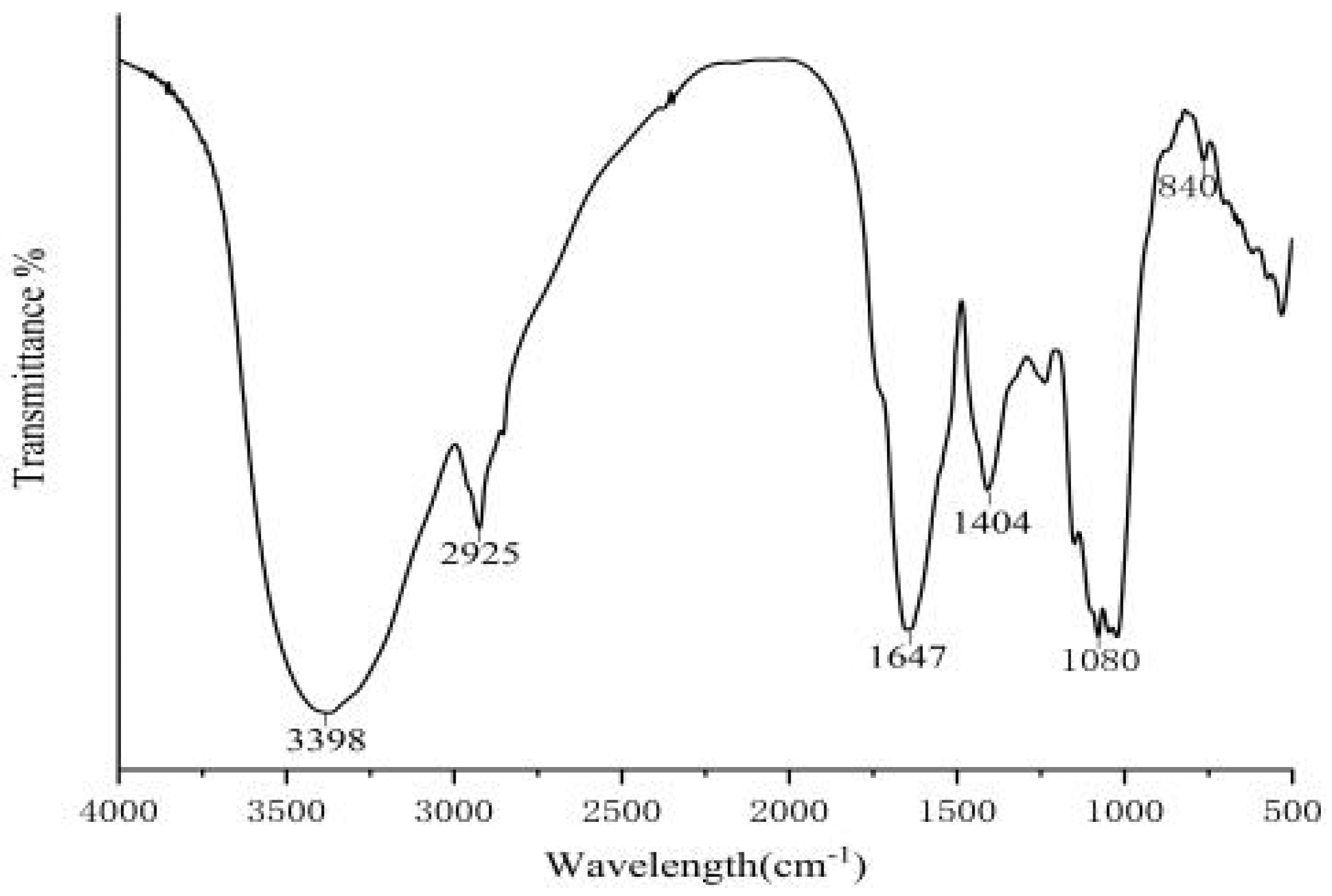
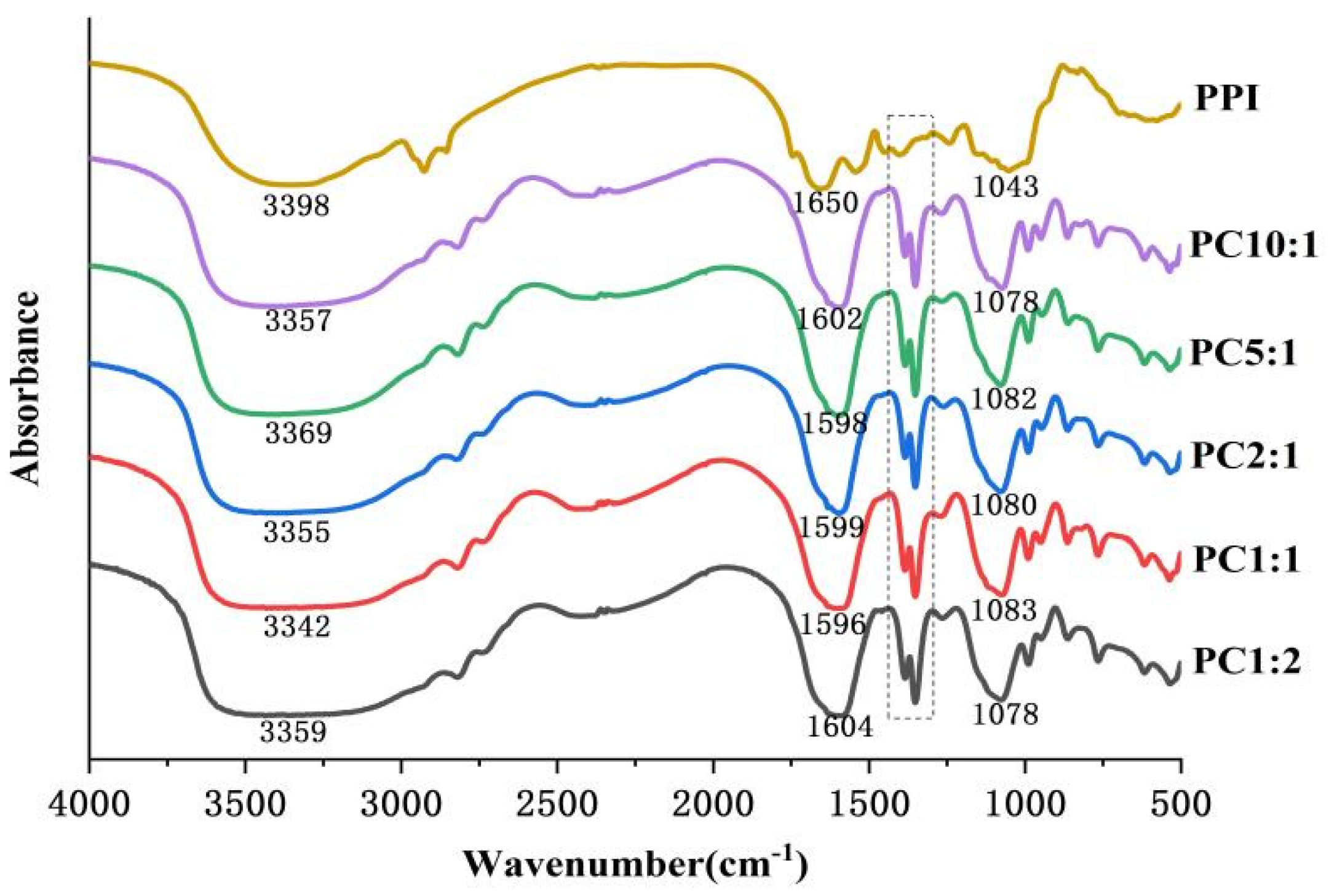
2.6. Fourier Transform Infrared Spectroscopy (FTIR)
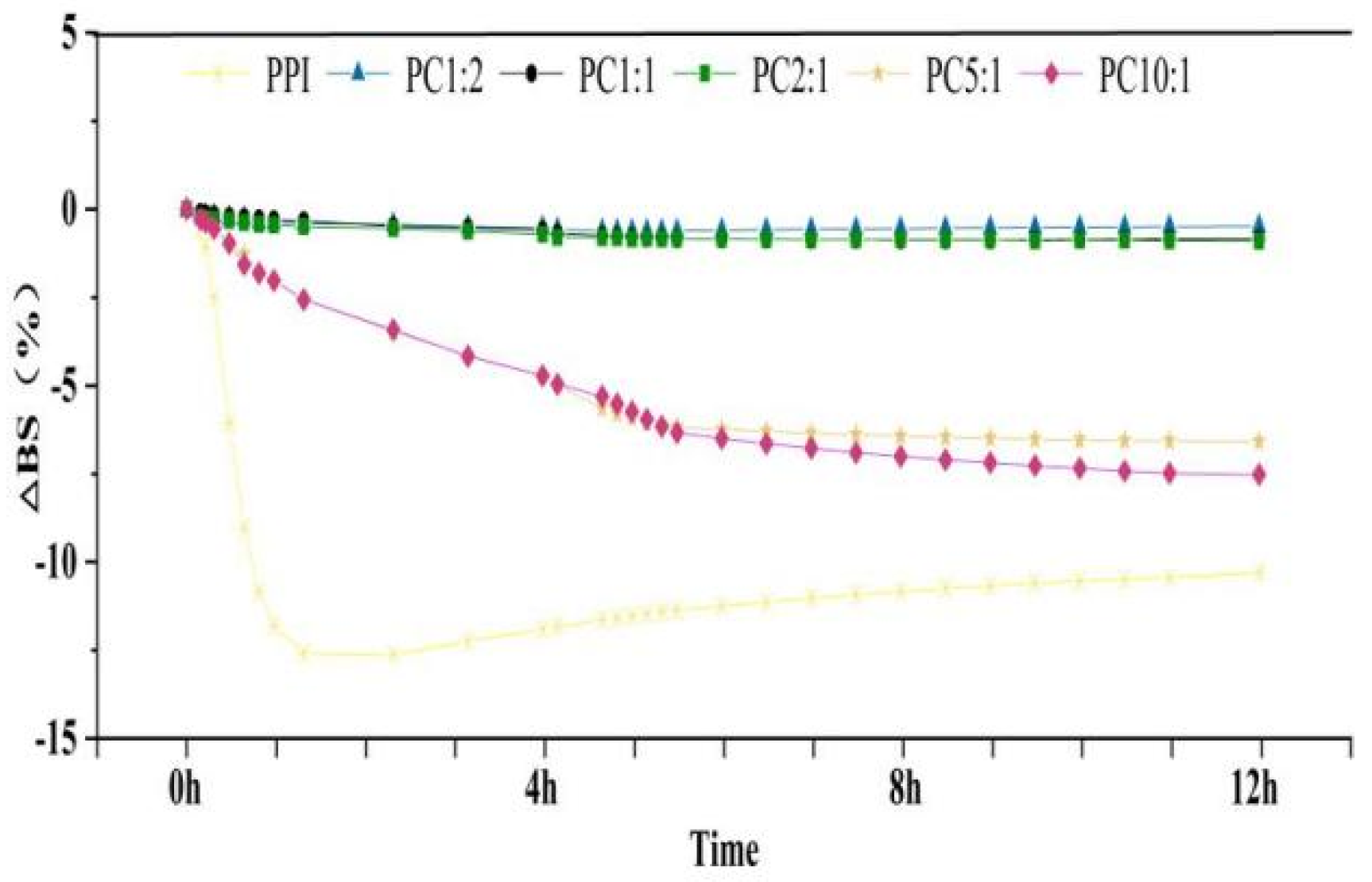
2.7. Solution Stability Analysis via Multiple Light Scattering
2.8. Foaming Capacity (FC) and Stability (FS)
2.9. Protein Solubility
2.10. Statistical Analysis
3. Results and Discussion
3.1. Effect of Polysaccharide Quality on Grafting Reaction
3.2. Surface Hydrophobicity of PPI-CSP Complexes (H0)
3.3. Thermal Properties
3.4. FTIR Spectroscopy
3.5. Stability Analysis by Multiple Light Scatterer
3.6. Foaming Properties
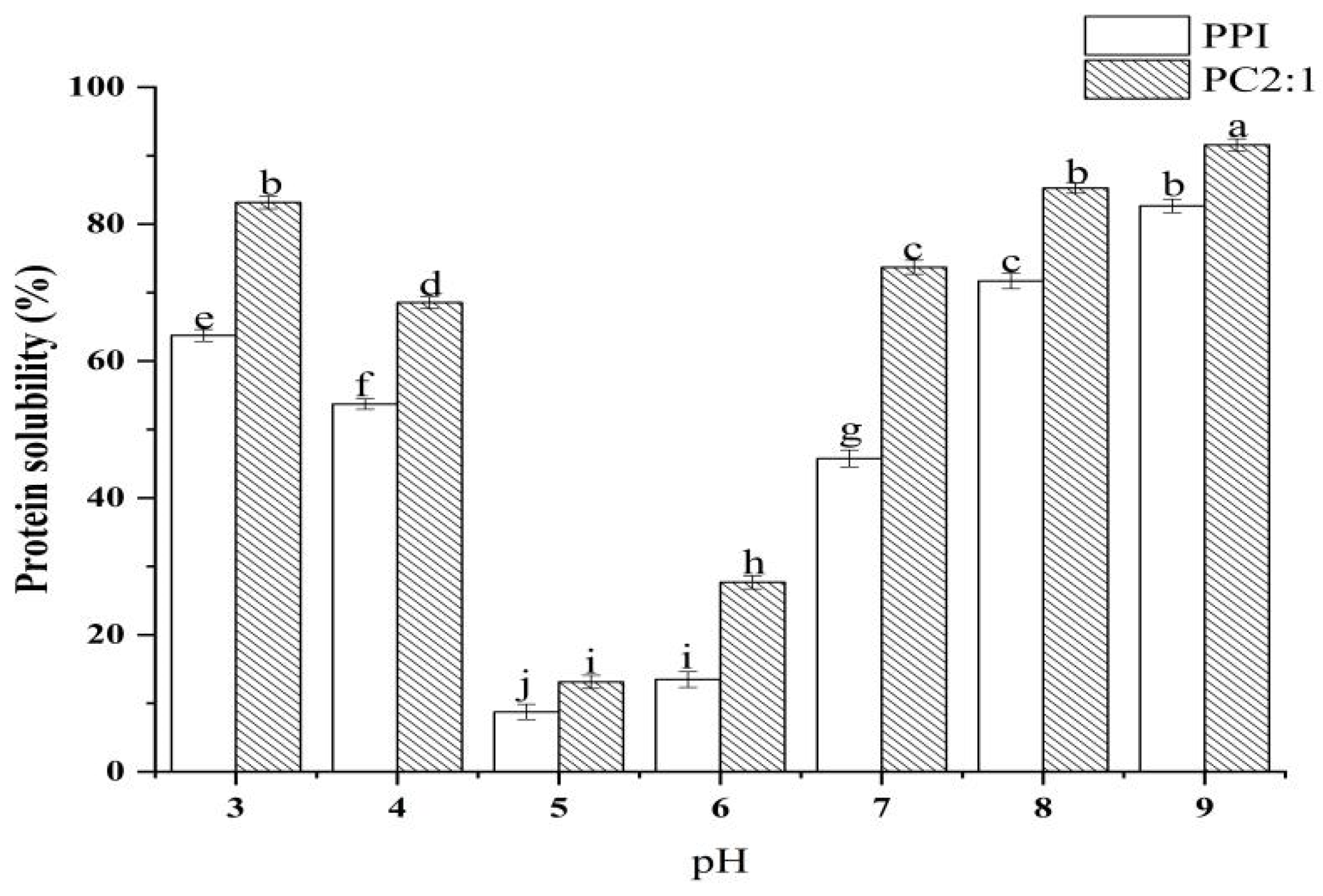
3.7. Protein Solubility at Different pH Conditions
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
Appendix B
References
- Zhao, G.; Liu, Y.; Zhao, M.; Ren, J.; Yang, B. Enzymatic Hydrolysis and Their Effects on Conformational and Functional Properties of Peanut Protein Isolate. Food Chem. 2011, 127, 1438–1443. [Google Scholar] [CrossRef]
- Wu, H.; Wang, Q.; Ma, T.; Ren, J. Comparative Studies on the Functional Properties of Various Protein Concentrate Preparations of Peanut Protein. Food Res. Int. 2009, 42, 343–348. [Google Scholar] [CrossRef]
- Liu, Y.; Zhao, G.; Zhao, M.; Ren, J.; Yang, B. Improvement of Functional Properties of Peanut Protein Isolate by Conjugation with Dextran through Maillard Reaction. Food Chem. 2012, 131, 901–906. [Google Scholar] [CrossRef]
- Guan, J.-J.; Qiu, A.-Y.; Liu, X.-Y.; Hua, Y.-F.; Ma, Y.-H. Microwave Improvement of Soy Protein Isolate–Saccharide Graft Reactions. Food Chem. 2006, 97, 577–585. [Google Scholar] [CrossRef]
- Jia, Y.; Gao, X.; Xue, Z.; Wang, Y.; Lu, Y.; Zhang, M.; Panichayupakaranant, P.; Chen, H. Characterization, Antioxidant Activities, and Inhibition on Alpha-Glucosidase Activity of Corn Silk Polysaccharides Obtained by Different Extraction Methods. Int. J. Biol. Macromol. 2020, 163, 1640–1648. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.; Zhao, M.; Shi, J.; Yang, B.; Li, J.; Luo, D.; Jiang, G.; Jiang, Y. Effects of Combined High-Pressure Homogenization and Enzymatic Treatment on Extraction Yield, Hydrolysis and Function Properties of Peanut Proteins. Innov. Food Sci. Emerg. Technol. 2011, 12, 478–483. [Google Scholar] [CrossRef]
- Jiménez-Castaño, L.; Villamiel, M.; López-Fandiño, R. Glycosylation of Individual Whey Proteins by Maillard Reaction Using Dextran of Different Molecular Mass. Food Hydrocoll. 2007, 21, 433–443. [Google Scholar] [CrossRef]
- Su, G.; Ren, J.; Yang, B.; Cui, C.; Zhao, M. Comparison of Hydrolysis Characteristics on Defatted Peanut Meal Proteins between a Protease Extract from Aspergillus Oryzae and Commercial Proteases. Food Chem. 2011, 126, 1306–1311. [Google Scholar] [CrossRef]
- Yang, Z.H.; Li, C.; Li, Y.Y.; Wang, Z.H. Effects of Maillard Reaction on Allergenicity of Buckwheat Allergen Fag T 3 During Thermal Processing. J. Sci. Food Agric. 2013, 93, 1510–1515. [Google Scholar] [CrossRef]
- Cheng, Y.-H.; Mu, D.-C.; Feng, Y.-Y.; Xu, Z.; Wen, L.; Chen, M.-L.; Ye, J. Glycosylation of Rice Protein with Dextran Via the Maillard Reaction in a Macromolecular Crowding Condition to Improve Solubility. J. Cereal Sci. 2022, 103, 103374. [Google Scholar] [CrossRef]
- Ma, X.; Chen, W.; Yan, T.; Wang, D.; Hou, F.; Miao, S.; Liu, D. Comparison of Citrus Pectin and Apple Pectin in Conjugation with Soy Protein Isolate (Spi) under Controlled Dry-Heating Conditions. Food Chem. 2020, 309, 125501. [Google Scholar] [CrossRef]
- Zhang, B.; Chi, Y.J.; Li, B. Effect of Ultrasound Treatment on the Wet Heating Maillard Reaction between Β-Conglycinin and Maltodextrin and on the Emulsifying Properties of Conjugates. Eur. Food Res. Technol. 2013, 238, 129–138. [Google Scholar] [CrossRef]
- Zhang, H.; Yang, J.; Zhao, Y. High Intensity Ultrasound Assisted Heating to Improve Solubility, Antioxidant and Antibacterial Properties of Chitosan-Fructose Maillard Reaction Products. LWT—Food Sci. Technol. 2015, 60, 253–262. [Google Scholar] [CrossRef]
- Bouyer, E.; Mekhloufi, G.; Rosilio, V.; Grossiord, J.L.; Agnely, F. Proteins, Polysaccharides, and Their Complexes Used as Stabilizers for Emulsions: Alternatives to Synthetic Surfactants in the Pharmaceutical Field? Int. J. Pharm. 2012, 436, 359–378. [Google Scholar] [CrossRef]
- Zhao, C.; Yin, H.; Yan, J.; Niu, X.; Qi, B.; Liu, J. Structure and Acid-Induced Gelation Properties of Soy Protein Isolate–Maltodextrin Glycation Conjugates with Ultrasonic Pretreatment. Food Hydrocoll. 2021, 112, 106278. [Google Scholar] [CrossRef]
- Vigo, M.S.; Malec, L.S.; Gomez, R.G.; Llosa, R.A. Spectrophotometric Assay Using O-Phthaldialdehyde for Determination of Reactive Lysine in Dairy Products. Food Chem. 1992, 44, 363–365. [Google Scholar] [CrossRef]
- Chandrapala, J.; Zisu, B.; Palmer, M.; Kentish, S.; Ashokkumar, M. Effects of Ultrasound on the Thermal and Structural Characteristics of Proteins in Reconstituted Whey Protein Concentrate. Ultrason. Sonochem. 2011, 18, 951–957. [Google Scholar] [CrossRef]
- Basse, B.; Bosc, V.; Saiter, J.M.; Chan-Huot, M.; Dupas, J.P.; Maillard, M.N.; Menut, P. Combined Effects of Ionic Strength and Enzymatic Pre-Treatment in Thermal Gelation of Peanut Proteins Extracts. Food Res. Int. 2020, 137, 109362. [Google Scholar] [CrossRef]
- Zhao, Y.; Han, X.; Yin, H.; Li, Q.; Zhou, J.; Zhang, H.; Zhang, W.; Zhao, C.; Liu, J. Preparation and Characterisation of Curcumin-Loaded Pea Protein-Zein Nanocomplexes Using Ph-Driven Method. Int. J. Food Sci. Technol. 2022, 57, 3589–3603. [Google Scholar] [CrossRef]
- Hu, Q.; Wu, Y.; Zhong, L.; Ma, N.; Zhao, L.; Ma, G.; Cheng, N.; Nakata, P.A.; Xu, J. In Vitro Digestion and Cellular Antioxidant Activity of Β-Carotene-Loaded Emulsion Stabilized by Soy Protein Isolate-Pleurotus Eryngii Polysaccharide Conjugates. Food Hydrocoll. 2021, 112, 106340. [Google Scholar] [CrossRef]
- Du, F.; Qi, Y.; Huang, H.; Wang, P.; Xu, X.; Yang, Z. Stabilization of O/W Emulsions Via Interfacial Protein Concentrating Induced by Thermodynamic Incompatibility between Sarcoplasmic Proteins and Xanthan Gum. Food Hydrocoll. 2022, 124, 107242. [Google Scholar] [CrossRef]
- Wang, Q.; Jiang, J.; Xiong, Y.L. High Pressure Homogenization Combined with Ph Shift Treatment: A Process to Produce Physically and Oxidatively Stable Hemp Milk. Food Res. Int. 2018, 106, 487–494. [Google Scholar] [CrossRef]
- Watanabe, M.; Shimada, A.; Arai, S. Enzymatic Modification of Protein Functionality: Implantation of Potent Amphiphilicity to Succinylated Proteins by Covalent Attachment of Leucine Alkyl Esters. Agric. Biol. Chem. 2014, 45, 1621–1625. [Google Scholar] [CrossRef]
- Perusko, M.; Al-Hanish, A.; Cirkovic Velickovic, T.; Stanic-Vucinic, D. Macromolecular Crowding Conditions Enhance Glycation and Oxidation of Whey Proteins in Ultrasound-Induced Maillard Reaction. Food Chem. 2015, 177, 248–257. [Google Scholar] [CrossRef]
- Lowry, O.; Rosebrough, N.; Farr, A.L.; Randall, R. Protein Measurement with the Folin Phenol Reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef]
- Liu, L.; Li, X.; Du, L.; Zhang, X.; Yang, W.; Zhang, H. Effect of Ultrasound Assisted Heating on Structure and Antioxidant Activity of Whey Protein Peptide Grafted with Galactose. LWT 2019, 109, 130–136. [Google Scholar] [CrossRef]
- Xue, F.; Li, C.; Adhikari, B. Physicochemical Properties of Soy Protein Isolates-Cyanidin-3-Galactoside Conjugates Produced Using Free Radicals Induced by Ultrasound. Ultrason. Sonochem. 2020, 64, 104990. [Google Scholar] [CrossRef]
- Sun, W.-W.; Yu, S.-J.; Yang, X.-Q.; Wang, J.-M.; Zhang, J.-B.; Zhang, Y.; Zheng, E.-L. Study on the Rheological Properties of Heat-Induced Whey Protein Isolate–Dextran Conjugate Gel. Food Res. Int. 2011, 44, 3259–3263. [Google Scholar] [CrossRef]
- Zhao, C.B.; Zhou, L.Y.; Liu, J.Y.; Zhang, Y.; Chen, Y.; Wu, F. Effect of Ultrasonic Pretreatment on Physicochemical Characteristics and Rheological Properties of Soy Protein/Sugar Maillard Reaction Products. J. Food Sci. Technol. 2016, 53, 2342–2351. [Google Scholar] [CrossRef]
- He, W.; Tian, L.; Zhang, S.; Pan, S. A Novel Method to Prepare Protein-Polysaccharide Conjugates with High Grafting and Low Browning: Application in Encapsulating Curcumin. LWT 2021, 145, 111349. [Google Scholar] [CrossRef]
- Nakai, S. Structure-Function Relationships of Food Proteins: With an Emphasis on the Importance of Protein Hydrophobicity. J. Agric. Food Chem. 1983, 31, 676–683. [Google Scholar] [CrossRef]
- Mu, L.; Zhao, M.; Yang, B.; Zhao, H.; Cui, C.; Zhao, Q. Effect of Ultrasonic Treatment on the Graft Reaction between Soy Protein Isolate and Gum Acacia and on the Physicochemical Properties of Conjugates. J. Agric. Food Chem. 2010, 58, 4494–4499. [Google Scholar] [CrossRef] [PubMed]
- Achouri, A.; Boye, J.; Yaylayan, V.; Yeboah, F. Functional Properties of Glycated Soy 11s Glycinin. J. Food Sci. 2005, 70, C269–C274. [Google Scholar] [CrossRef]
- Feng, J.L.; Qi, J.R.; Yin, S.W.; Wang, J.M.; Guo, J.; Weng, J.Y.; Liu, Q.R.; Yang, X.Q. Fabrication and Characterization of Stable Soy Beta-Conglycinin-Dextran Core-Shell Nanogels Prepared Via a Self-Assembly Approach at the Isoelectric Point. J. Agric. Food Chem. 2015, 63, 6075–6083. [Google Scholar] [CrossRef]
- Dong, X.; Du, S.; Deng, Q.; Tang, H.; Yang, C.; Wei, F.; Chen, H.; Quek, S.Y.; Zhou, A.; Liu, L. Study on the Antioxidant Activity and Emulsifying Properties of Flaxseed Gum-Whey Protein Isolate Conjugates Prepared by Maillard Reaction. Int. J. Biol. Macromol. 2020, 153, 1157–1164. [Google Scholar] [CrossRef]
- Choi, S.; Kim, H.; Park, K.; Moon, T. Molecular Characteristics of Ovalbumin?Dextran Conjugates Formed through the Maillard Reaction. Food Chem. 2005, 92, 93–99. [Google Scholar] [CrossRef]
- Ibanoglu, E. Effect of Hydrocolloids on the Thermal Denaturation of Proteins. Food Chem. 2005, 90, 621–626. [Google Scholar] [CrossRef]
- Wang, J.S.; Zhao, M.M.; Yang, X.Q.; Jiang, Y.M. Improvement of Emulsifying Properties of Wheat Gluten Hydrolysate/Lambda-Carrageenan Conjugates. Food Technol. Biotechnol. 2006, 44, 25–32. [Google Scholar] [CrossRef]
- Huang, X.; Tu, Z.; Xiao, H.; Wang, H.; Zhang, L.; Hu, Y.; Zhang, Q.; Niu, P. Characteristics and Antioxidant Activities of Ovalbumin Glycated with Different Saccharides under Heat Moisture Treatment. Food Res. Int. 2012, 48, 866–872. [Google Scholar] [CrossRef]
- Shu, Y.W.; Sahara, S.; Nakamura, S.; Kato, A. Effects of the Length of Polysaccharide Chains on the Functional Properties of the Maillard-Type Lysozymepolysaccharide Conjugate. J. Agric. Food Chem. 1996, 44, 2544–2548. [Google Scholar] [CrossRef]
- Xue, F.; Gu, Y.; Wang, Y.; Li, C.; Adhikari, B. Encapsulation of Essential Oil in Emulsion Based Edible Films Prepared by Soy Protein Isolate-Gum Acacia Conjugates. Food Hydrocoll. 2019, 96, 178–189. [Google Scholar] [CrossRef]
- Su, J.-F.; Huang, Z.; Yuan, X.-Y.; Wang, X.-Y.; Li, M. Structure and Properties of Carboxymethyl Cellulose/Soy Protein Isolate Blend Edible Films Crosslinked by Maillard Reactions. Carbohydr. Polym. 2010, 79, 145–153. [Google Scholar] [CrossRef]
- Jia, Y.; Xue, Z.; Wang, Y.; Lu, Y.; Li, R.; Li, N.; Wang, Q.; Zhang, M.; Chen, H. Chemical Structure and Inhibition on Alpha-Glucosidase of Polysaccharides from Corn Silk by Fractional Precipitation. Carbohydr. Polym. 2021, 252, 117185. [Google Scholar] [CrossRef]
- Jiang, Z.; Wang, C.; Li, T.; Sun, D.; Gao, H.; Gao, Z.; Mu, Z. Effect of Ultrasound on the Structure and Functional Properties of Transglutaminase-Crosslinked Whey Protein Isolate Exposed to Prior Heat Treatment. Int. Dairy J. 2019, 88, 79–88. [Google Scholar] [CrossRef]
- Zhao, C.; Yin, H.; Yan, J.; Qi, B.; Liu, J. Structural and Physicochemical Properties of Soya Bean Protein Isolate/Maltodextrin Mixture and Glycosylation Conjugates. Int. J. Food Sci. Technol. 2020, 55, 3315–3326. [Google Scholar] [CrossRef]
- Hattori, M.; Okada, Y.; Takahashi, K. Functional Changes in Beta-Lactoglobulin Upon Conjugation with Carboxymethyl Cyclodextrin. J. Agric. Food Chem. 2000, 48, 3789–3794. [Google Scholar] [CrossRef][Green Version]
- Chen, L.; Chen, J.; Ren, J.; Zhao, M. Effects of Ultrasound Pretreatment on the Enzymatic Hydrolysis of Soy Protein Isolates and on the Emulsifying Properties of Hydrolysates. J. Agric. Food Chem. 2011, 59, 2600–2609. [Google Scholar] [CrossRef]
- Li, C.; Huang, X.; Peng, Q.; Shan, Y.; Xue, F. Physicochemical Properties of Peanut Protein Isolate-Glucomannan Conjugates Prepared by Ultrasonic Treatment. Ultrason. Sonochem. 2014, 21, 1722–1727. [Google Scholar] [CrossRef]
- Wu, C.S.; Guo, J.H.; Lin, M.J. Stability Evaluation of Ph-Adjusted Goat Milk for Developing Ricotta Cheese with a Mixture of Cow Cheese Whey and Goat Milk. Foods 2020, 9, 366. [Google Scholar] [CrossRef]
- Zhao, L.; Zhang, S.; Lu, J.; Lv, J. Effects of Heat Treatment and Stabilizing Salts Supplementation on the Physicochemical Properties, Protein Structure and Salts Balance of Goat Milk. LWT 2020, 132, 109878. [Google Scholar] [CrossRef]
- Matos, M.; Marefati, A.; Gutiérrez, G.; Wahlgren, M.; Rayner, M.J.P.O. Comparative Emulsifying Properties of Octenyl Succinic Anhydride (Osa)-Modified Starch: Granular Form Vs Dissolved State. PLoS ONE 2016, 11, e0160140. [Google Scholar] [CrossRef]
- Polowczyk, I.; Bastrzyk, A.; Koźlecki, T.; Sadowski, Z. Stability of Three-Phase Water-Particle-Oil Systems. Chem. Eng. Technol. 2015, 38, 715–720. [Google Scholar] [CrossRef]
- Rodríguez Patino, J.M.; Pilosof, A.M.R. Protein–Polysaccharide Interactions at Fluid Interfaces. Food Hydrocoll. 2011, 25, 1925–1937. [Google Scholar] [CrossRef]
- Moro, A.; Báez, G.D.; Busti, P.A.; Ballerini, G.A.; Delorenzi, N.J. Effects of Heat-Treated Β-Lactoglobulin and Its Aggregates on Foaming Properties. Food Hydrocoll. 2011, 25, 1009–1015. [Google Scholar] [CrossRef]
- Mingzhe, P.; Xianjun, M.; Lianzhou, J.; Dianyu, Y.; Tianyi, L. Effect of Cosolvents (Polyols) on Structural and Foaming Properties of Soy Protein Isolate. Czech J. Food 2017, 35, 57–66. [Google Scholar] [CrossRef]
- Asokapandian, S.; Venkatachalam, S.; Swamy, G.J.; Kuppusamy, K. Optimization of Foaming Properties and Foam Mat Drying of Muskmelon Using Soy Protein. J. Food Process Eng. 2016, 39, 692–701. [Google Scholar] [CrossRef]
- Gharibzahedi, S.M.T.; Smith, B. The Functional Modification of Legume Proteins by Ultrasonication: A Review. Trends Food Sci. Technol. 2020, 98, 107–116. [Google Scholar] [CrossRef]
- Deng, W.; Yang, X.; Zhu, Y.; Yu, J.; Xu, X. Structural Characterization and Hypolipidemic Activities of Purified Stigma Maydis Polysaccharides. Food Sci. Nutr. 2019, 7, 2674–2683. [Google Scholar] [CrossRef]
- Jin, X.; Wang, Q.; Yang, X.; Guo, M.; Li, W.; Shi, J.; Adu-Frimpong, M.; Xu, X.; Deng, W.; Yu, J. Chemical Characterisation and Hypolipidaemic Effects of Two Purified Pleurotus Eryngii. Polysaccharides. Int. J. Food Sci. Technol. 2018, 53, 2298–2307. [Google Scholar] [CrossRef]
- Yuan, D.; Li, C.; Huang, Q.; Fu, X.J.C.P. Ultrasonic Degradation Effects on the Physicochemical, Rheological and Antioxidant Properties of Polysaccharide from Sargassum Pallidum. Carbohydr. Polym. 2020, 239, 116230. [Google Scholar] [CrossRef] [PubMed]
- Muhidinov, Z.K.; Bobokalonov, J.T.; Ismoilov, I.B.; Strahan, G.D.; Chau, H.K.; Hotchkiss, A.T.; Liu, L. Characterization of Two Types of Polysaccharides from Eremurus Hissaricus Roots Growing in Tajikistan. Food Hydrocoll. 2020, 105, 105768. [Google Scholar] [CrossRef]

| Sample | Conarachin | Arachin | ||
|---|---|---|---|---|
| Td (°C) | ΔH(J/g) | Td (°C) | ΔH (J/g) | |
| PPI | 64.55 ± 0.23 e | 1221.6 ± 0.09 d | 104.22 ± 0.27 c | 281.7 ± 0.02 d |
| PC10:1 | 69.83 ± 0.38 f | 1328.1 ± 0.10 c | 110.29 ± 0.31 b | 418.5 ± 0.08 c |
| PC5:1 | 74.98 ± 0.21 d | 1430.3 ± 0.03 b | 110.31 ± 0.18 b | 486.1 ± 0.05 b |
| PC2:1 | 86.06 ± 0.26 c | 1533.5 ± 0.07 a | 112.58 ± 0.24 a | 544.7 ± 0.13 a |
| PC1:1 | 89.31 ± 0.15 b | 1553.7 ± 0.04 a | 112.43 ± 0.19 a | 543.3 ± 0.11 a |
| PC1:2 | 90.56 ± 0.19 a | 1512.3 ± 0.06 a | 112.23 ± 0.32 a | 544.4 ± 0.14 a |
| Sample | α-Helix (%) | β-Sheet (%) | β-Turns (%) | Random Coil (%) |
|---|---|---|---|---|
| PPI | 27.03 ± 0.13 a | 18.39 ± 0.27 d | 20.05 ± 0.15 d | 34.53 ± 0.12 d |
| PC10:1 | 20.53 ± 0.21 b | 19.84 ± 0.19 c | 24.10 ± 0.24 c | 35.53 ± 0.17 c |
| PC5:1 | 15.90 ± 0.18 c | 21.31 ± 0.23 b | 25.95 ± 0.11 b | 36.84 ± 0.06 b |
| PC2:1 | 13.44 ± 0.29 d | 22.06 ± 0.08 a | 27.21 ± 0.34 a | 37.29 ± 0.25 a |
| PC1:1 | 13.11 ± 0.05 d | 22.13 ± 0.12 a | 27.45 ± 0.15 a | 37.31 ± 0.06 a |
| PC1:2 | 13.06 ± 0.34 d | 22.14 ± 0.16 a | 27.47 ± 0.09 a | 37.33 ± 0.14 a |
| Sample | PPI | PC10:1 | PC5:1 | PC2:1 | PC1:1 | PC1:2 |
|---|---|---|---|---|---|---|
| TSI | 41.46 | 9.8 | 8.85 | 3.55 | 4.14 | 6.18 |
| BS | 16.88 | 12.3 | 12.04 | 9.27 | 9.26 | 9.25 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, X.; Zhao, Y.; Mao, S.; Hu, N.; Sun, D.; Yang, Q.; Chu, Z.; Zheng, Q.; Xiu, L.; Liu, J. Effects of Different Amounts of Corn Silk Polysaccharide on the Structure and Function of Peanut Protein Isolate Glycosylation Products. Foods 2022, 11, 2214. https://doi.org/10.3390/foods11152214
Han X, Zhao Y, Mao S, Hu N, Sun D, Yang Q, Chu Z, Zheng Q, Xiu L, Liu J. Effects of Different Amounts of Corn Silk Polysaccharide on the Structure and Function of Peanut Protein Isolate Glycosylation Products. Foods. 2022; 11(15):2214. https://doi.org/10.3390/foods11152214
Chicago/Turabian StyleHan, Xinxin, Yilin Zhao, Sining Mao, Nannan Hu, Dijing Sun, Qi Yang, Zejun Chu, Qihang Zheng, Lin Xiu, and Jingsheng Liu. 2022. "Effects of Different Amounts of Corn Silk Polysaccharide on the Structure and Function of Peanut Protein Isolate Glycosylation Products" Foods 11, no. 15: 2214. https://doi.org/10.3390/foods11152214
APA StyleHan, X., Zhao, Y., Mao, S., Hu, N., Sun, D., Yang, Q., Chu, Z., Zheng, Q., Xiu, L., & Liu, J. (2022). Effects of Different Amounts of Corn Silk Polysaccharide on the Structure and Function of Peanut Protein Isolate Glycosylation Products. Foods, 11(15), 2214. https://doi.org/10.3390/foods11152214






