Lentil (Lens culinaris Medik.) Flour Varieties as Promising New Ingredients for Gluten-Free Cookies
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Methods Used in Both Cookie and Flour Samples
Colour Measurement
Determination of Moisture and Crude Protein Content
Total Polyphenol, Total Flavonoid Content and Antioxidant Capacity
2.2.2. Methods Used in Cookie Samples
Baking Loss, Geometry
Texture Analysis
Sensory Tests
2.3. Data Analysis
3. Results and Discussion
3.1. Results Obtained for Raw Materials
3.1.1. Colour (L*, a*, b*) of Raw Materials
3.1.2. Flour Yield
3.1.3. Physicochemical Properties of Raw Materials
3.2. Results Obtained for Cookies
3.2.1. Baking Loss, Geometry of the Cookies
3.2.2. Colour (L*, a*, b*) of the Cookies
3.2.3. Physicochemical Properties of the Cookies
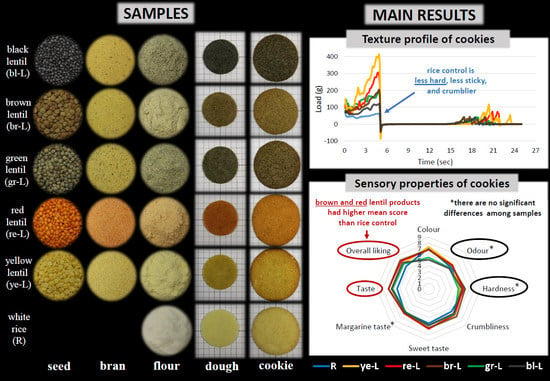
3.3. Textural Properties of Doughs and Cookies
3.4. Sensory Properties of Cookies
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Di Cairano, M.; Galgano, F.; Tolve, R.; Caruso, M.C.; Condelli, N. Focus on Gluten Free Biscuits: Ingredients and Issues. Trends Food Sci. Technol. 2018, 81, 203–212. [Google Scholar] [CrossRef]
- Melini, V.; Melini, F. Gluten-Free Diet: Gaps and Needs for a Healthier Diet. Nutrients 2019, 11, 170. [Google Scholar] [CrossRef] [PubMed]
- Jnawali, P.; Kumar, V.; Tanwar, B. Celiac Disease: Overview and Considerations for Development of Gluten-Free Foods. Food Sci. Hum. Wellness 2016, 5, 169–176. [Google Scholar] [CrossRef]
- Naqash, F.; Gani, A.; Gani, A.; Masoodi, F.A. Gluten-Free Baking: Combating the Challenges—A Review. Trends Food Sci. Technol. 2017, 66, 98–107. [Google Scholar] [CrossRef]
- Vici, G.; Belli, L.; Biondi, M.; Polzonetti, V. Gluten Free Diet and Nutrient Deficiencies: A Review. Clin. Nutr. 2016, 35, 1236–1241. [Google Scholar] [CrossRef]
- Cardo, A.; Churruca, I.; Lasa, A.; Navarro, V.; Vázquez-Polo, M.; Perez-Junkera, G.; Larretxi, I. Nutritional Imbalances in Adult Celiac Patients Following a Gluten-Free Diet. Nutrients 2021, 13, 2877. [Google Scholar] [CrossRef]
- Szűcs, V.; Fazakas, Z.; Farr, A.M.; Tarcea, M. Quality of Life of Consumers Following a Gluten-Free Diet. Results of a Questionnaire Survey in Hungary and Romania. Orv. Hetil. 2019, 160, 980–986. [Google Scholar] [CrossRef]
- Valitutti, F.; Iorfida, D.; Anania, C.; Trovato, C.M.; Montuori, M.; Cucchiara, S.; Catassi, C. Cereal Consumption among Subjects with Celiac Disease: A Snapshot for Nutritional Considerations. Nutrients 2017, 9, 396. [Google Scholar] [CrossRef]
- Khazaei, H.; Subedi, M.; Nickerson, M.; Martínez-Villaluenga, C.; Frias, J.; Vandenberg, A. Seed Protein of Lentils: Current Status, Progress, and Food Applications. Foods 2019, 8, 391. [Google Scholar] [CrossRef]
- Turfani, V.; Narducci, V.; Durazzo, A.; Galli, V.; Carcea, M. Technological, Nutritional and Functional Properties of Wheat Bread Enriched with Lentil or Carob Flours. LWT-Food Sci. Technol. 2017, 78, 361–366. [Google Scholar] [CrossRef]
- Mitchell, D.C.; Marinangeli, C.P.F.; Pigat, S.; Bompola, F.; Campbell, J.; Pan, Y.; Curran, J.M.; Cai, D.J.; Jaconis, S.Y.; Rumney, J. Pulse Intake Improves Nutrient Density among US Adult Consumers. Nutrients 2021, 13, 2668. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Agriculture Food Data Central. Available online: https://fdc.nal.usda.gov/ (accessed on 20 May 2022).
- Sen Gupta, D.; Thavarajah, D.; Knutson, P.; Thavarajah, P.; McGee, R.J.; Coyne, C.J.; Kumar, S. Lentils (Lens culinaris L.), a Rich Source of Folates. J. Agric. Food Chem. 2013, 61, 7794–7799. [Google Scholar] [CrossRef] [PubMed]
- Hall, C.; Hillen, C.; Garden Robinson, J. Composition, Nutritional Value, and Health Benefits of Pulses. Cereal Chem. 2017, 94, 11–31. [Google Scholar] [CrossRef]
- Ferawati, F.; Hefni, M.; Witthöft, C. Flours from Swedish Pulses: Effects of Treatment on Functional Properties and Nutrient Content. Food Sci. Nutr. 2019, 7, 4116–4126. [Google Scholar] [CrossRef] [PubMed]
- Siva, N.; Thavarajah, P.; Thavarajah, D. The Impact of Processing and Cooking on Prebiotic Carbohydrates in Lentil. J. Food Compos. Anal. 2018, 70, 72–77. [Google Scholar] [CrossRef]
- Siva, N.; Johnson, C.R.; Richard, V.; Jesch, E.D.; Whiteside, W.; Abood, A.A.; Thavarajah, P.; Duckett, S.; Thavarajah, D. Lentil (Lens culinaris Medikus) Diet Affects the Gut Microbiome and Obesity Markers in Rat. J. Agric. Food Chem. 2018, 66, 8805–8813. [Google Scholar] [CrossRef]
- Patterson, C.A.; Curran, J.; Der, T. Effect of Processing on Antinutrient Compounds in Pulses. Cereal Chem. 2017, 94, 2–10. [Google Scholar] [CrossRef]
- Landi, N.; Pacifico, S.; Piccolella, S.; Di Giuseppe, A.M.A.; Mezzacapo, M.C.; Ragucci, S.; Iannuzzi, F.; Zarrelli, A.; Di Maro, A. Valle Agricola Lentil, an Unknown Lentil (Lens culinaris Medik.) Seed from Southern Italy as a Novel Antioxidant and Prebiotic Source. Food Fuction 2015, 6, 3155–3164. [Google Scholar] [CrossRef]
- Margier, M.; Georgé, S.; Hafnaoui, N.; Remond, D.; Nowicki, M.; Du Chaffaut, L.; Amiot, M.J.; Reboul, E. Nutritional Composition and Bioactive Content of Legumes: Characterization of Pulses Frequently Consumed in France and Effect of the Cooking Method. Nutrients 2018, 10, 1668. [Google Scholar] [CrossRef]
- Foschia, M.; Horstmann, S.W.; Arendt, E.K.; Zannini, E. Legumes as Functional Ingredients in Gluten-Free Bakery and Pasta Products. Annu. Rev. Food Sci. Technol. 2017, 8, 75–96. [Google Scholar] [CrossRef]
- Ganesan, K.; Xu, B. Polyphenol-Rich Lentils and Their Health Promoting Effects. Int. J. Mol. Sci. 2017, 18, 2390. [Google Scholar] [CrossRef] [PubMed]
- Paucean, A.; Moldovan, O.P.; Mureșan, V.; Socaci, S.A.; Dulf, F.V.; Alexa, E.; Man, S.M.; Mureșan, A.E.; Muste, S. Folic Acid, Minerals, Amino-Acids, Fatty Acids and Volatile Compounds of Green and Red Lentils. Folic Acid Content Optimization in Wheat-Lentils Composite Flours. Chem. Cent. J. 2018, 12, 88. [Google Scholar] [CrossRef] [PubMed]
- Bubelová, Z.; Sumczynski, D.; Salek, R.N. Effect of Cooking and Germination on Antioxidant Activity, Total Polyphenols and Flavonoids, Fiber Content, and Digestibility of Lentils (Lens culinaris L.). J. Food Process. Preserv. 2018, 42, e13388. [Google Scholar] [CrossRef]
- Liu, Y.; Ragaee, S.; Marcone, M.F.; Abdel-Aal, E.S.M. Composition of Phenolic Acids and Antioxidant Properties of Selected Pulses Cooked with Different Heating Conditions. Foods 2020, 9, 908. [Google Scholar] [CrossRef] [PubMed]
- Portman, D.; Maharjan, P.; McDonald, L.; Laskovska, S.; Walker, C.; Irvin, H.; Blanchard, C.; Naiker, M.; Panozzo, J.F. Nutritional and Functional Properties of Cookies Made Using Down-Graded Lentil—A Candidate for Novel Food Production and Crop Utilization. Cereal Chem. 2020, 97, 95–103. [Google Scholar] [CrossRef]
- AACC (American Association of Cereal Chemists). Approved Methods of the American Association of Cereal Chemists, 10th ed.; Method 10-50D; American Association of Cereal Chemists: Saint Paul, MN, USA, 2000. [Google Scholar]
- CIE (Commission Internationale de l’Éclairage). Technical Report: Colorimetry, 3rd ed.; CIE 15: Washington, DC, WA, USA, 2004; ISBN 390-190-633-9. [Google Scholar]
- FAO (Food and Agriculture Organization of the United Nations). Food Energy—Methods of Analysis and Conversion Factors; FAO Food and Nutrition Paper 77; FAO: Rome, Italy, 2003. [Google Scholar]
- Singleton, V.L.; Rossi, J.A. Colorimetry of Total Phenolics with Phosphomolybdic-Phosphotungstic Acid Reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Chang, C.-C.; Yang, M.-H.; Wen, H.-M.; Chern, J.-C. Estimation of Total Flavonoid Content in Propolis by Two Complementary Colorimetric Methods. J. Food Drug Anal. 2002, 10, 178–182. [Google Scholar] [CrossRef]
- Apak, R.; Güçlü, K.; Özyürek, M.; Karademir, S.E. Novel Total Antioxidant Capacity Index for Dietary Polyphenols and Vitamins C and E, Using Their Cupric Ion Reducing Capability in the Presence of Neocuproine: CUPRAC Method. J. Agric. Food Chem. 2004, 52, 7970–7981. [Google Scholar] [CrossRef]
- Zappalà, M.; Fallico, B.; Arena, E.; Verzera, A. Methods for the Determination of HMF in Honey: A Comparison. Food Control 2005, 16, 273–277. [Google Scholar] [CrossRef]
- ISO 8589:2007; Sensory Analysis—General Guidance for the Design of Test Rooms. ISO (International Organization for Standardization): Geneva, Switzerland, 2007.
- ISO 8589:2007/AMD 1:2014; Sensory Analysis—General Guidance for the Design of Test Rooms—Amendment 1. ISO (International Organization for Standardization): Geneva, Switzerland, 2014.
- ISO 11136:2014; Sensory Analysis—Methodology—General Guidance for Conducting Hedonic Tests with Consumers in a Controlled Area. ISO (International Organization for Standardization): Geneva, Switzerland, 2014.
- ISO 11136:2014/AMD 1:2020; Sensory Analysis—Methodology—General Guidance for Conducting Hedonic Tests with Consumers in a Controlled Area—Amendment 1. ISO (International Organization for Standardization): Geneva, Switzerland, 2020.
- ISO 4121:2003; Sensory Analysis—Guidelines for the Use of Quantitative Response Scales. ISO (International Organization for Standardization): Geneva, Switzerland, 2003.
- Oskaybaş-Emlek, B.; Özbey, A.; Kahraman, K. Effects of Germination on the Physicochemical and Nutritional Characteristics of Lentil and Its Utilization Potential in Cookie-Making. J. Food Meas. Charact. 2021, 15, 4245–4255. [Google Scholar] [CrossRef]
- Romano, A.; Gallo, V.; Ferranti, P.; Masi, P. Lentil Flour: Nutritional and Technological Properties, in Vitro Digestibility and Perspectives for Use in the Food Industry. Curr. Opin. Food Sci. 2021, 40, 157–167. [Google Scholar] [CrossRef]
- Fujiwara, N.; Hall, C.; Jenkins, A.L. Development of Low Glycemic Index (GI) Foods by Incorporating Pulse Ingredients into Cereal-Based Products: Use of in Vitro Screening and in Vivo Methodologies. Cereal Chem. 2017, 94, 110–116. [Google Scholar] [CrossRef]
- Zou, Y.; Chang, S.K.C.; Gu, Y.; Qian, S.Y. Antioxidant Activity and Phenolic Compositions of Lentil (Lens culinaris Var. Morton) Extract and Its Fractions. J. Agric. Food Chem. 2011, 59, 2268–2276. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-Y.; Yeo, Y.-S.; Park, S.-Y.; Lee, S.-G.; Lee, S.-M.; Cho, H.-S.; Chung, N.-J.; Oh, S.-W. Compositional Analysis of Lentil (Lens culinaris) Cultivars Related to Colors and Their Antioxidative Activity. Plant Breed. Biotechnol. 2017, 5, 192–203. [Google Scholar] [CrossRef]
- Polat, H.; Dursun Capar, T.; Inanir, C.; Ekici, L.; Yalcin, H. Formulation of Functional Crackers Enriched with Germinated Lentil Extract: A Response Surface Methodology Box-Behnken Design. LWT 2020, 123, 109065. [Google Scholar] [CrossRef]
- Rezagholi, F.; Hesarinejad, M.A. Integration of Fuzzy Logic and Computer Vision in Intelligent Quality Control of Celiac-Friendly Products. Procedia Comput. Sci. 2017, 120, 325–332. [Google Scholar] [CrossRef]
- Shapla, U.M.; Solayman, M.; Alam, N.; Khalil, M.I.; Gan, S.H. 5-Hydroxymethylfurfural (HMF) Levels in Honey and Other Food Products: Effects on Bees and Human Health. Chem. Cent. J. 2018, 12, 35. [Google Scholar] [CrossRef]
- Delgado-Andrade, C.; Rufián-Henares, J.A.; Morales, F.J. Hydroxymethylfurfural in Commercial Biscuits Marketed in Spain. J. Food Nutr. Res. 2009, 48, 14–19. [Google Scholar]
- Sharma, P.; Gujral, H.S. Cookie Making Behavior of Wheat-Barley Flour Blends and Effects on Antioxidant Properties. LWT-Food Sci. Technol. 2014, 55, 301–307. [Google Scholar] [CrossRef]
- Švecová, B.; Mach, M. Content of 5-Hydroxymethyl-2-Furfural in Biscuits for Kids. Interdiscip. Toxicol. 2017, 10, 66–69. [Google Scholar] [CrossRef]
- Han, J.; Janz, J.A.M.; Gerlat, M. Development of Gluten-Free Cracker Snacks Using Pulse Flours and Fractions. Food Res. Int. 2010, 43, 627–633. [Google Scholar] [CrossRef]
| Ingredient | Amount |
|---|---|
| Lentil flour (or rice flour in the case of control) | 100.00 g |
| Powdered sugar | 57.80 g |
| Margarine (with 70% fat content) | 28.40 g |
| Salt | 0.93 g |
| Sodium bicarbonate | 1.11 g |
| Distilled water | 7.11 g |
| Glucose solution (5 g/100 mL) | 14.60 g |
| Flour | Bran | Dough | Cookies | |
|---|---|---|---|---|
| Black lentil | bl-LF | bl-LB | bl-LF-D | bl-LF-C |
| Brown lentil | br-LF | br-LB | br-LF-D | br-LF-C |
| Green lentil | gr-LF | gr-LB | gr-LF-D | gr-LF-C |
| Red lentil | re-LF | re-LB | re-LF-D | re-LF-C |
| Yellow lentil | ye-LF | ye-LB | ye-LF-D | ye-LF-C |
| Rice (control) | RF | – | RF-D | RF-C |
| Sample | L* | a* | b* |
|---|---|---|---|
| bl-WLS | 25.26 ± 0.11 a | 1.09 ± 0.03 b | −2.25 ± 0.04 a |
| br-WLS | 39.70 ± 0.59 d | 1.12 ± 0.17 b | 11.37 ± 0.32 c |
| gr-WLS | 31.10 ± 0.36 b | −0.03 ± 0.11 a | 4.76 ± 0.22 b |
| re-WLS | 38.13 ± 0.55 c | 20.03 ± 0.19 d | 28.68 ± 0.56 e |
| ye-WLS | 46.89 ± 0.45 e | 3.18 ± 0.17 c | 26.82 ± 0.70 d |
| RF | 92.27 ± 1.19 f | −0.60 ± −0.03 d | 11.01 ± 0.11 b |
| bl-LF | 46.19 ± 0,67 a | −1.56 ± 0.02 ab | 10.23 ± 0.21 a |
| br-LF | 53.22 ± 0.27 c | −1.49 ± 0.02 bc | 15.48 ± 0.20 d |
| gr-LF | 51.96 ± 0.22 b | −1.75 ± 0.13 a | 12.62 ± 0.15 c |
| ye-LF | 56.63 ± 0.31 e | −1.30 ± 0.05 c | 20.69 ± 0.21 e |
| re-LF | 54.54 ± 0.19 d | 7.36 ± 0.22 e | 21.65 ± 0.19 f |
| bl-LB | 49.17 ± 0.83 b | 2.16 ± 0.20 c | 26.30 ± 0.51 a |
| br-LB | 50.11 ± 0.32 b | 1.14 ± 0.05 a | 26.51 ± 0.31 a |
| gr-LB | 50.13 ± 0.72 b | 1.72 ± 0.31 bc | 25.98 ± 0.32 a |
| re-LB | 47.31 ± 0.70 a | 14.69 ± 0.44 d | 26.68 ± 0.62 a |
| ye-LB | 51.69 ± 0.95 c | 1.56 ± 0.21 ab | 28.01 ± 0.60 b |
| Sample | Moisture (g/100 g) n = 3 | CP (g/100 g) n = 3 | CUPRAC (mmol TE/100 g) n = 5 | TPC (mg GAE/100 g) n = 5 | TFC (mg CE/100 g) n = 5 |
|---|---|---|---|---|---|
| RF | 9.3 ± 0.1 b | 6.5 ± 0.1 a | 0.29 ± 0.02 a | 44.0 ± 3.8 a | 14.3 ± 1.9 a |
| bl-LF | 9.9 ± 0.1 d | 25.7 ± 0.1 c | 1.09 ± 0.02 d | 244.4 ± 7.1 e | 34.2 ± 2.6 c |
| br-LF | 9.9 ± 0.1 d | 21.4 ± 0.3 b | 0.77 ± 0.04 c | 197.2 ± 5.7 d | 39.0 ± 2.7 c |
| gr-LF | 10.0 ± 0.1 d | 22.5 ± 0.1 b | 1.58 ± 0.04 e | 337.3 ± 5.2 f | 63.3 ± 2.7 d |
| re-LF | 9.5 ± 0.0 c | 24.8 ± 1.1 c | 0.35 ± 0.01 ab | 151.1 ± 2.6 c | 24.6 ± 5.1 b |
| ye-LF | 8.2 ± 0.1 a | 24.8 ± 0.2 c | 0.36 ± 0.03 b | 113.2 ± 4.3 b | 24.7 ± 3.2 b |
| Sample | Baking Loss, (%), n = 10 | Diameter (mm), n = 20 | Height (mm), n = 30 | Diameter/ Height |
|---|---|---|---|---|
| RF-C | 14.0 ± 0.8 a | 59.9 ± 1.3 a | 9.8 ± 1.0 d | 6.1 |
| bl-LF-C | 13.3 ± 0.7 a | 63.0 ± 2.9 b | 9.1 ± 0.7 c | 6.9 |
| br-LF-C | 13.7 ± 0.7 a | 62.0 ± 1.9 ab | 8.6 ± 0.7 c | 7.2 |
| gr-LF-C | 14.1 ± 0.7 a | 62.6 ± 2.1 b | 9.0 ± 0.6 c | 7.0 |
| re-LF-C | 14.2 ± 0.8 a | 68.6 ± 2.8 c | 7.3 ± 0.5 b | 9.4 |
| ye-LF-C | 13.9 ± 0.8 a | 68.9 ± 2.6 c | 6.5 ± 0.6 a | 10.6 |
| Sample | L* | a* | b* |
|---|---|---|---|
| RF-C | 72.68 ± 1.98 f | 5.23 ± 1.22 b | 34.37 ± 1.22 d |
| bl-LF-C | 33.79 ± 0.61 a | 1.69 ± 0.36 a | 9.31 ± 0.35 a |
| br-LF-C | 45.20 ± 0.60 c | 8.96 ± 0.65 c | 24.28 ± 0.29 c |
| gr-LF-C | 38.25 ± 0.55 b | 4.62 ± 0.49 b | 15.48 ± 0.23 b |
| re-LF-C | 51.50 ± 1.08 d | 18.24 ± 0.32 e | 35.72 ± 0.91 d |
| ye-LF-C | 55.12 ± 2.30 e | 11.63 ± 1.60 d | 38.30 ± 2.04 e |
| Sample | Moisture (g/100 g) n = 3 | CP (g/100 g) n = 3 | CUPRAC (mmol TE/100 g) n = 5 | TPC (mg GAE/100 g) n = 5 | TFC (mg CE/100 g) n = 5 |
|---|---|---|---|---|---|
| RF-C | 5.4 ± 0.0 d | 3.8 ± 0.1 a | 0.35 ± 0.04 a | 61.5 ± 1.7 a | 13.1 ± 2.9 a |
| bl-LF-C | 4.3 ± 0.1 a | 14.6 ± 0.1 de | 1.57 ± 0.08 e | 342.3 ± 13.3 e | 56.9 ± 2.6 c |
| br-LF-C | 4.5 ± 0.0 b | 12.1 ± 0.2 b | 1.29 ± 0.08 d | 269.5 ± 4.8 d | 56.7 ± 3.4 c |
| gr-LF-C | 4.5 ± 0.0 b | 13.2 ± 0.1 c | 1.81 ± 0.09 f | 399.9 ± 14.6 f | 75.9 ± 6.9 d |
| re-LF-C | 4.4 ± 0.0 b | 14.8 ± 0.0 e | 0.82 ± 0.02 c | 196.3 ± 10.1 c | 24.6 ± 1.4 b |
| ye-LF-C | 4.9 ± 0.0 c | 14.4 ± 0.2 d | 0.60 ± 0.01 b | 136.5 ± 3.5 b | 23.8 ± 2.9 b |
| pH n = 3 | HMF (mg/kg) n = 6 | |
|---|---|---|
| RF-C | 9.27 ± 0.02 c | 26.2 ± 3.2 |
| bl-LF-C | 7.26 ± 0.01 a | <1 |
| br-LF-C | 7.63 ± 0.08 b | <1 |
| gr-LF-C | 7.20 ± 0.05 a | <1 |
| re-LF-C | 7.72 ± 0.02 b | <1 |
| ye-LF-C | 7.64 ± 0.01 b | <1 |
| Hardness Work (mJ) | Adhesive Force (g) | Cohesiveness (-) | Springiness (mm) | |
|---|---|---|---|---|
| RF-D | 2.8 ± 0.4 d | 3.7 ± 0.9 a | 0.06 ± 0.01 a | 5.9 ± 0.3 bc |
| bl-LF-D | 1.5 ± 0.1 b | 7.3 ± 0.9 bc | 0.14 ± 0.01 c | 5.3 ± 0.4 a |
| br-LF-D | 1.5 ± 0.1 b | 7.7 ± 0.7 c | 0.14 ± 0.01 c | 5.9 ± 0.4 c |
| gr-LF-D | 2.5 ± 0.3 c | 9.1 ± 1.3 d | 0.12 ± 0.00 b | 5.0 ± 0.5 a |
| re-LF-D | 1.3 ± 0.1 b | 6.3 ± 0.8 b | 0.17 ± 0.01 d | 5.4 ± 0.2 ab |
| ye-LF-D | 0.8 ± 0.1 a | 8.1 ± 0.8 cd | 0.21 ± 0.02 e | 5.3 ± 0.5 a |
| Hardness (g) | Adhesive Force (g) | Cohesiveness (-) | Springiness (mm) | Gumminess (g) | Chewiness (mJ) | |
|---|---|---|---|---|---|---|
| RF-C | 73.0 ± 45.9 a | 11.3 ± 6.2 a | 0.09 ± 0.01 a | 1.0 ± 0.3 a | 7.3 ± 4.4 a | 0.08 ± 0.06 a |
| bl-LF-C | 143.9 ± 39.8 ab | 38.4 ± 9.9 b | 0.15 ± 0.02 bc | 1.9 ± 0.7 ab | 20.5 ± 5.2 ab | 0.40 ± 0.20 a |
| br-LF-C | 216.5 ± 99.2 b | 43.9 ± 11.7 bc | 0.16 ± 0.02 c | 2.0 ± 0.7 ab | 34.3 ± 16.4 b | 0.77 ± 0.45 ab |
| gr-LF-C | 240.5 ± 89.5 bc | 53.1 ± 24.1 bc | 0.12 ± 0.02 b | 2.1 ± 0.8 b | 29.7 ± 13.5 b | 0.68 ± 0.61 a |
| re-LF-C | 363.8 ± 164.2 cd | 55.6 ± 23.2 bc | 0.15 ± 0.01 bc | 2.9 ± 1.3 b | 55.6 ± 27.7 c | 1.59 ± 1.17 c |
| ye-LF-C | 469.4 ± 184.8 d | 60.5 ± 17.3 c | 0.15 ± 0.03 c | 2.5 ± 1.2 b | 66.3 ± 15.2 c | 1.54 ± 0.80 bc |
| RF-C | bl-LF-C | br-LF-C | gr-LF-C | re-LF-C | ye-LF-C | |
|---|---|---|---|---|---|---|
| Colour | 7.1 ± 1.4 b | 5.1 ± 2.5 a | 6.8 ± 1.6 b | 5.5 ± 2.0 a | 6.8 ± 1.9 b | 7.3 ± 1.6 b |
| Odour | 5.3 ± 1.7 a | 5.0 ± 1.8 a | 5.7 ± 1.8 a | 5.1 ± 1.7 a | 5.8 ± 1.7 a | 5.9 ± 1.8 a |
| Hardness | 6.3 ± 2.0 ab | 5.8 ± 2.3 ab | 6.3 ± 2.0 b | 5.2 ± 2.3 a | 6.0 ± 2.1 ab | 5.5 ± 2.2 ab |
| Crumbliness | 5.6 ± 2.4 a | 6.1 ± 2.0 ab | 6.8 ± 1.7 b | 6.3 ± 2.0 ab | 6.4 ± 1.9 ab | 6.3 ± 2.2 ab |
| Taste | 5.5 ± 2.4 a | 6.5 ± 1.7 b | 7.0 ± 1.7 b | 6.3 ± 1.7 ab | 7.0 ± 1.5 b | 6.5 ± 2.0 b |
| Sweet taste | 6.0 ± 2.0 a | 6.4 ± 1.9 ab | 6.8 ± 1.9 ab | 6.3 ± 1.9 ab | 7.0 ± 1.6 b | 7.0 ± 1.7 b |
| Margarine taste | 5.8 ± 1.8 a | 5.8 ± 2.0 a | 6.0 ± 2.0 a | 5.7 ± 2.0 a | 6.0 ± 1.7 a | 5.7 ± 2.0 a |
| Overall liking | 5.6 ± 2.3 a | 6.5 ± 1.8 ab | 7.0 ± 1.6 b | 6.1 ± 1.7 ab | 6.8 ± 1.6 b | 6.2 ± 1.9 ab |
| Purchase intention | 2.6 ± 1.3 a | 2.8 ± 1.3 ab | 3.6 ± 1.1 c | 3.0 ± 1.1 abc | 3.4 ± 1.1 cb | 3.1 ± 1.2 abc |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hajas, L.; Sipos, L.; Csobod, É.C.; Bálint, M.V.; Juhász, R.; Benedek, C. Lentil (Lens culinaris Medik.) Flour Varieties as Promising New Ingredients for Gluten-Free Cookies. Foods 2022, 11, 2028. https://doi.org/10.3390/foods11142028
Hajas L, Sipos L, Csobod ÉC, Bálint MV, Juhász R, Benedek C. Lentil (Lens culinaris Medik.) Flour Varieties as Promising New Ingredients for Gluten-Free Cookies. Foods. 2022; 11(14):2028. https://doi.org/10.3390/foods11142028
Chicago/Turabian StyleHajas, Lívia, László Sipos, Éva Csajbókné Csobod, Márta Veresné Bálint, Réka Juhász, and Csilla Benedek. 2022. "Lentil (Lens culinaris Medik.) Flour Varieties as Promising New Ingredients for Gluten-Free Cookies" Foods 11, no. 14: 2028. https://doi.org/10.3390/foods11142028
APA StyleHajas, L., Sipos, L., Csobod, É. C., Bálint, M. V., Juhász, R., & Benedek, C. (2022). Lentil (Lens culinaris Medik.) Flour Varieties as Promising New Ingredients for Gluten-Free Cookies. Foods, 11(14), 2028. https://doi.org/10.3390/foods11142028