Application of a Newly Developed Chitosan/Oleic Acid Edible Coating for Extending Shelf-Life of Fresh Pork
Abstract
:1. Introduction
2. Materials and Methods
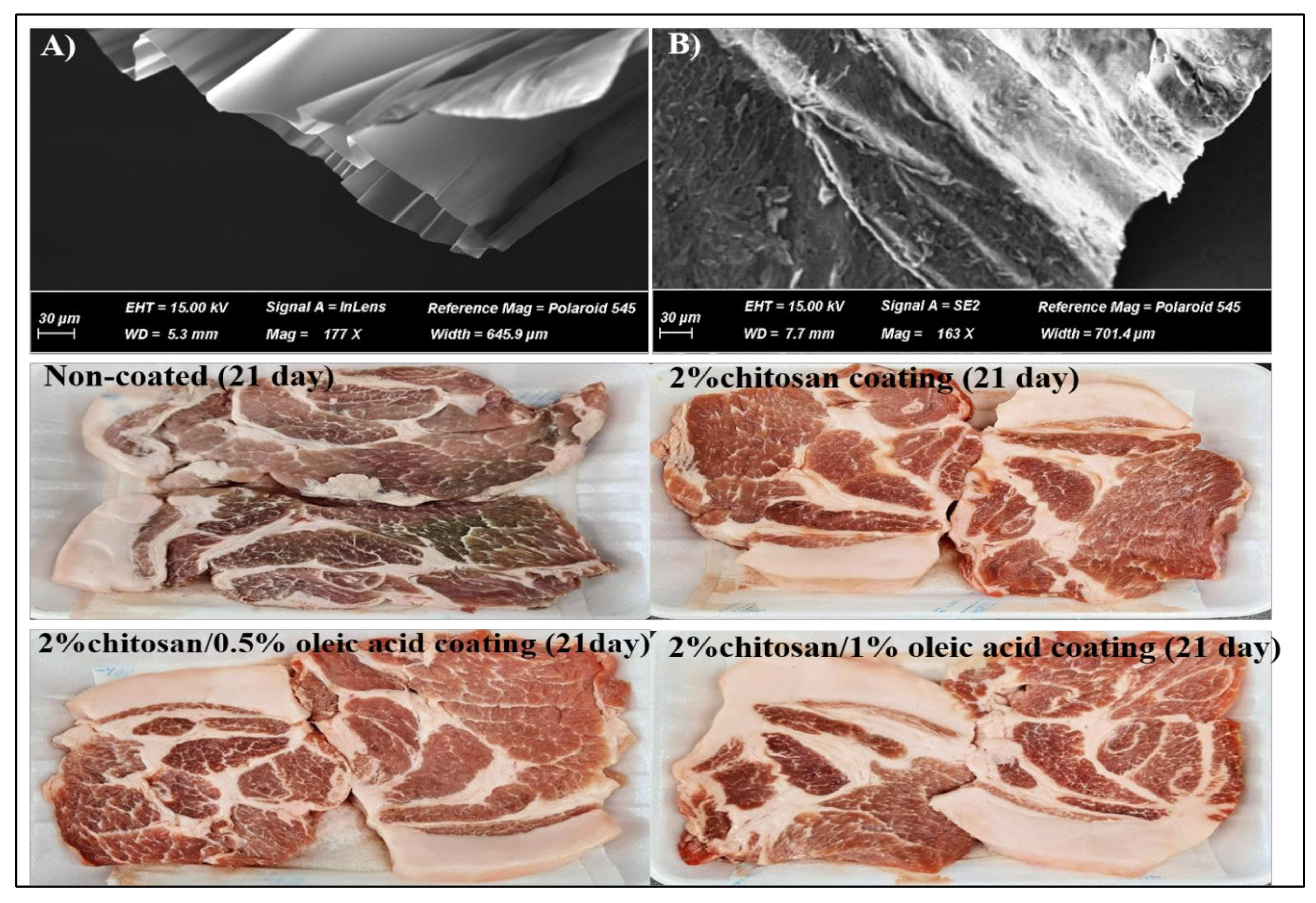
2.1. Preparation of Coating Solutions and Application
2.2. Shelf-Life Measurements
2.2.1. Microbiology
2.2.2. pH Measurement
2.2.3. Total Volatile Basic Nitrogen (TVBN)
2.2.4. Thiobarbituric Acid Reactive Substances (TBARS)
2.2.5. Meat Color
2.3. Fatty Acid Composition
2.4. Aroma Volatiles
2.5. Statistical Analysis
3. Results and Discussion
3.1. Fatty Acid Profiles
3.2. Effects on Microbial Quality
3.3. Effects on TBARS and TVBN Contents
3.4. Effects on Color and pH
3.5. Effects on Aroma Volatiles
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bekhit, A.E.A.; Holman, B.W.B.; Giteru, S.G.; Hopkins, D.L. Total volatile basic nitrogen (TVB-N) and its role in meat spoilage: A review. Trends Food Sci. Technol. 2021, 109, 280–302. [Google Scholar] [CrossRef]
- EFSA. The European Union summary report on trends and source of zoonoses, zoonotic agents and foodborne outbreaks in 2017. EFSA J. 2018, 16, 5500. [Google Scholar]
- OECD. Meat Consumption (Indicator). Available online: https://data.oecd.org/agroutput/meat-consumption.htm (accessed on 15 December 2021).
- Li, N.; Zhang, Y.; Wu, Q.; Gu, Q.; Chen, M.; Zhang, Y.; Sun, X.; Zhang, J. High-throughput sequencing analysis of bacterial community composition and quality characteristics in refrigerated pork during storage. Food Microbiol. 2019, 83, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Gao, F.; Xu, X.; Ye, K.; Zhou, G. Changes in the composition of the bacterial flora on tray-packaged pork during chilled storage analyzed by PCR-DGGE and real-time PCR. J. Food Sci. 2011, 76, 27–33. [Google Scholar] [CrossRef]
- Cao, L.; Sun, G.; Zhang, C.; Liu, W.; Li, J.; Wang, L. An intelligent film based on cassia gum containing bromothymol blue-anchored cellulose fibers for real-time detection of meat freshness. J. Agric. Food Chem. 2019, 67, 2066–2074. [Google Scholar] [CrossRef]
- Bekhit, A.E.A.; Giteru, S.G.; Holman, B.W.B.; Hopkins, D.L. Total volatile basic nitrogen and trimethylamine in muscle foods: Potential formation pathways and effects on human health: Compr. Rev. Food Sci. Food Saf. 2021, 20, 3620–3666. [Google Scholar] [CrossRef]
- Papuc, C.; Goran, G.V.; Predescu, C.N.; Nicorescu, V. Mechanisms of oxidative processes in meat and toxicity induced by postprandial degradation products: A review. Compr. Rev. Food Sci. Food Saf. 2017, 16, 96–122. [Google Scholar] [CrossRef]
- Silva, M.M.; Lidon, F.C. Food preservatives-an overview on applications and side effects. Emir. J. Food Agric. 2016, 28, 366–373. [Google Scholar] [CrossRef]
- Varraso, R.; Camargo, C.A. Processed meat consumption and lung health: More evidence for harm. Eur. Respir. Soc. 2014, 43, 943–946. [Google Scholar] [CrossRef] [Green Version]
- Zhou, G.H.; Xu, X.L.; Liu, Y. Preservation technologies for fresh meat: A review. Meat Sci. 2010, 86, 119–128. [Google Scholar] [CrossRef]
- Song, D.H.; Hoa, V.B.; Kim, H.W.; Kang, S.M.; Cho, S.H.; Ham, J.S.; Seol, K.H. Edible Films on Meat and Meat Products. Coatings 2021, 11, 1344. [Google Scholar] [CrossRef]
- Grande-Tovar, C.D.; Chaves-Lopez, C.; Serio, A.; Rossi, C.; Paparella, A. Chitosan coatings enriched with essential oils: Effects on fungi involved in fruit decay and mechanism of action. Trends Food Sci. Technol. 2018, 78, 61–71. [Google Scholar] [CrossRef]
- No, K.H.; Meyers, S.P.; Prinyawiwatkul, W.; Xu, Z. Applications of chitosan for improvement of quality and shelf-life of foods: A review. Food Sci. 2007, 72, 87–99. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Mukherjee, A.; Dutta, J. Chitosan based nanocomposite films and coattings: Emerging antimicrobial food packaging alternatives. Trends Food Sci. Technol. 2020, 97, 196–209. [Google Scholar] [CrossRef]
- Priyadarshi, R.; Rhim, J.W. Chitosan-based biodegradable functional films for food packaging applications. Innov. Food Sci. Emerg. Technol. 2020, 62, 102346. [Google Scholar] [CrossRef]
- Kanatt, S.R.; Rao, M.S.; Chawla, S.P.; Sharma, A. Effects of chitosan coating on shelf-life of ready-to-cook meat products during chilled storage. LWT-Food Sci. Technol. 2013, 53, 321–326. [Google Scholar] [CrossRef]
- Duran, A.; Kahve, H.I. The effect of chitosan coating and vacuum packaging on the microbiological and chemical properties of beef. Meat Sci. 2020, 162, 107961. [Google Scholar] [CrossRef]
- Alirezalu, K.; Pirouzi, S.; Yaghoubi, M.; Karimi-Dehkordi, M.; Jafarzadeh, S.; Khaneghah, A.M. Packaging of beef fillet with active chitosan film incorporated with ε-polylysine: An assessment of quality indices and shelf life. Meat Sci. 2021, 176, 108475. [Google Scholar] [CrossRef]
- Fang, Z.; Lin, D.; Warner, D.; Ha, M. Effect of gallic acid/chitosan coating on fresh pork quality in modified atmosphere packaging. Food Chem. 2018, 260, 90–96. [Google Scholar] [CrossRef]
- Desbois, A.P.; Smith, V.J. Antibacterial free fatty acids: Activities, mechanisms of action and biotechnological potential. Appl. Microbiol. Biotechnol. 2010, 85, 1629–1642. [Google Scholar] [CrossRef] [Green Version]
- Kumar, P.; Lee, J.; Beyenal, H.; Lee, J. Fatty acids as antibiofilm and antivirulence agents. Trends Microbiol. 2020, 28, 753–768. [Google Scholar] [CrossRef] [PubMed]
- Casillas-Vargas, G.; Ocasio-Malave, C.; Medina, S.; Morales-Guzman, C.; Del Valle, R.G.; Carballeira, N.M.; Sanabria-Ríos, D.J. Antibacterial fatty acids: An update of possible mechanisms of action and implications in the development of the next-generation of antibacterial agents. Prog. Lipid Res. 2021, 82, 101093. [Google Scholar] [CrossRef] [PubMed]
- Carballeira, N.; Montano, N.; Morales, C.; Mooney, J.; Torres, X.; Díaz, D.; Sanabria-Rios, D.J. 2- Methoxylated FA display unusual antibacterial activity towards clinical isolates of methicillin-resistant Staphylococcus aureus (CIMRSA) and Escherichia coli. Lipids 2017, 52, 535–548. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.B. Marbling and its nutritional impact on risk factors for cardiovascular disease. J. Food Sci. Anim. Resour. 2016, 36, 435–444. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoa, V.B.; Amna, T.; Hwang, I.H. Significant influence of particular unsaturated fatty acids and pH on the volatile compounds in meat-like model systems. Meat Sci. 2013, 94, 480–488. [Google Scholar]
- Vargas, M.; Albors, A.; Chiralt, A.; Gonzaler-Martinez, C. Quality of cold stored strawberries as affected by chitosan-oleic acid edible coatings. Postharvest Biol. Technol. 2006, 41, 164–171. [Google Scholar] [CrossRef]
- Stenz, L.; François, P.; Fischer, A.; Huyghe, A.; Tangomo, M.; Hernandez, D.; Cassat, J.; Linder, P.; Schrenzel, J. Impact of oleic acid (cis-9-octadecenoic acid) on bacterial viability and biofilm production in Staphylococcus aureus. FEMS Microbiol. Lett. 2008, 287, 149–155. [Google Scholar] [CrossRef] [Green Version]
- ISO-International Organization for Standardization, ISO. Meat and Meat Products—Enumeration of Presumptive Pseudomonas spp.; International Organization for Standardization, ISO: Geneva, Switzerland, 2010; p. 13720. [Google Scholar]
- Hoa, V.B.; Song, D.H.; Seol, K.H.; Kang, S.M.; Kim, H.W.; Kim, J.H.; Cho, S.H. Coating with chitosan containing lauric acid (C12:0) significantly extends the shelf-life of aerobically-Packaged beef steaks during refrigerated storage. Meat Sci. 2022, 184, 108696. [Google Scholar] [CrossRef]
- Buege, J.A.; Aust, J.D. Microsomal lipid peroxidation. Methods Enzymol. 1987, 52, 302–309. [Google Scholar]
- Hoa, V.B.; Seong, P.N.; Cho, S.H.; Kang, S.M.; Kim, Y.S.; Moon, S.S.; Choi, Y.M.; Kim, J.H.; Seol, K.H. Quality characteristics and flavor compounds of pork meat as a function of carcass quality grade. Asian-Australas. J. Anim. Sci. 2019, 32, 1448–1457. [Google Scholar] [CrossRef]
- Mottram, D.S. Flavor formation in meat and meat products: A review. Food Chem. 1998, 62, 415–424. [Google Scholar] [CrossRef]
- Dinh, T.T.N.; To, K.V.; Schilling, M.W. Fatty acid composition of meat animals as flavor precursors. Meat Muscle Biol. 2021, 5, 1–16. [Google Scholar] [CrossRef]
- Kim, J.; Yim, D.G. Assessment of the microbial level for livestock products in retail meat shops implementing HACCP system. Korean J. Food Sci. Anim. Resour. 2016, 36, 594–600. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghafir, Y.; China, B.; Dierick, K.; De Zutter, L.; Daube, G. Hygiene indicator microorganisms for selected pathogens on beef, pork, and poultry meats in Belgium. J. Food Prot. 2008, 71, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Doulgeraki, A.I.; Ercolini, D.; Villani, F.; Nychas, G.J.E. Spoilage microbiota associated to the storage of raw meat in different conditions. A review. Int. J. Food Microbiol. 2012, 157, 130–141. [Google Scholar] [CrossRef]
- Aijuka, M.; Buys, E.M. Persistence of foodborne diarrheagenic Escherichia coli in the agricultural and food production environment: Implications for food safety and public health. Food Microbiol. 2019, 82, 363–370. [Google Scholar] [CrossRef]
- Fiore, A.; Park, S.; Volpe, S.; Torrieri, E.; Masi, P. Active packaging based on PLA and chitosan-caseinate enriched rosemary essential oil coating for fresh minced chicken breast application. Food Packag. Shelf Life 2021, 29, 100708. [Google Scholar] [CrossRef]
- Liu, H.; Du, Y.; Wang, X.; Sun, L. Chitosan kills bacteria through cell membrane damage. Int. J. Food Microbiol. 2004, 95, 147–155. [Google Scholar] [CrossRef]
- Kim, H.J.; Kim, D.; Kim, H.J.; Song, S.O.; Song, Y.H. Evaluation of the microbiological status of raw beef in Korea: Considering the suitability of aerobic plate count guideline. Korean J. Food Sci. Anim. Resour. 2018, 38, 43–51. [Google Scholar]
- Georgantelis, D.; Ambrosiadis, I.; Katikou, P.; Blekas, G.; Georgakis, S.A. Effect of rosemary extract, chitosan and a-tocopherol on microbiological parameters and lipid oxidation of fresh pork sausages stored at 4 °C. Meat Sci. 2007, 76, 172–181. [Google Scholar] [CrossRef]
- Cheng, Y.; Hu, J.; Wu, S. Chitosan based coatings extend the shelf-life of beef slices during refrigerated storage. LWT-Food Sci. Technol. 2021, 138, 110694. [Google Scholar] [CrossRef]
- Mancini, R.A.; Hunt, M.C. Current research in meat color. Meat Sci. 2005, 71, 100–121. [Google Scholar] [CrossRef] [PubMed]
- Brantley, R.E.; Smerdon, S.J.; Wilkinson, A.J.; Singleton, E.W.; Jonh, S.H. The mechanism of autooxidation of myoglobin. J. Biol. Chem. 1993, 268, 6995–7010. [Google Scholar] [CrossRef]
- Elmore, J.S.; Mottram, D.S.; Enser, M.; Wood, J.D. Effect of the polyunsaturated fatty acid composition of beef muscle on the profile of aroma volatiles. J. Agric. Food Chem. 1999, 47, 1619–1625. [Google Scholar] [CrossRef]
- Machiels, D.; Istasse, L.; van Ruth, S.M. Gas chromatography-olfactometry analysis of beef meat originating from differently fed Belgian Blue, Limousin and Aberdeen Angus bulls. Food Chem. 2004, 86, 377–383. [Google Scholar] [CrossRef]
- Rochat, S.; Chaintreau, A. Carbonyl odorants contributing to the in-oven roast beef top note. J. Agric. Food Chem. 2005, 53, 9578–9585. [Google Scholar] [CrossRef]
- Casaburi, A.; Piombino, P.; Nychas, G.; Villani, F.; Ercolini, D. Bacterial populations and the volatilome associated to meat spoilage. Food Microbiol. 2015, 45, 83–102. [Google Scholar] [CrossRef]
| Items | CON | CHI | CHI/0.5% OA | CHI/1% OA |
|---|---|---|---|---|
| C14:0 (Myristic acid) | 1.85 ± 0.01 | 1.70 ± 0.18 | 1.57 ± 0.05 | 1.52 ± 0.06 |
| C16:0 (Palmitic acid) | 27.45 ± 0.04 | 26.16 ± 0.80 | 26.13 ± 0.43 | 25.78 ± 0.75 |
| C16:1n7 (Palmitoleic acid) | 2.71 ± 0.02 | 2.06 ± 0.28 | 2.14 ± 0.45 | 2.09 ± 0.51 |
| C18:0 (Stearic acid) | 13.67 ± 0.75 | 14.42 ± 0.29 | 14.99 ± 0.35 | 13.95 ± 1.01 |
| C18:1n9 (Oleic acid) | 37.54 ± 0.74 c | 38.09 ± 1.43 c | 39.24 ± 0.28 ab | 40.66 ± 1.44 a |
| C18:1n7 (Cis-vaccenic acid) | 0.22 ± 0.03 | 0.24 ± 0.05 | 0.21 ± 0.03 | 0.23 ± 0.03 |
| C18:2n6 (Linoleic acid) | 14.79 ± 0.02 | 15.57 ± 0.14 | 14.06 ± 0.77 | 14.07 ± 0.59 |
| C18:3n6 (Gamma linoleic acid) | 0.01 ± 0.00 | 0.01 ± 0.00 | 0.01 ± 0.00 | 0.01 ± 0.00 |
| C18:3n3 (Linolenic acid) | 0.35 ± 0.03 | 0.36 ± 0.02 | 0.33 ± 0.03 | 0.34 ± 0.05 |
| C20:1n9 (Eicosenoic acid) | 0.90 ± 0.03 | 0.89 ± 0.01 | 0.89 ± 0.02 | 0.87 ± 0.03 |
| C20:4n6 (Arachidonic acid) | 0.35 ± 0.02 | 0.34 ± 0.06 | 0.32 ± 0.02 | 0.34 ± 0.04 |
| C20:5n3 (Eicosapentaenoic acid) | 0.01 ± 0.00 | 0.01 ± 0.00 | ND | ND |
| C22:4n6 (Adrenic acid) | 0.13 ± 0.02 | 0.13 ± 0.02 | 0.11 ± 0.01 | 0.13 ± 0.00 |
| SFA | 42.97 ± 0.76 | 42.29 ± 1.28 | 42.69 ± 0.55 | 41.25 ± 1.81 |
| UFA | 57.03 ± 0.76 | 57.71 ± 1.28 | 57.31 ± 0.55 | 58.75 ± 1.81 |
| MUFA | 41.37 ± 0.71 b | 41.28 ± 1.21 b | 42.47 ± 0.42 ab | 43.85 ± 1.16 a |
| PUFA | 15.65 ± 0.06 ab | 16.42 ± 0.09 a | 14.84 ± 0.82 b | 14.90 ± 0.68 b |
| n6/n3 | 41.27 ± 3.53 | 44.09 ± 2.17 | 43.52 ± 2.38 | 43.56 ± 4.38 |
| MUFA/SFA | 0.96 ± 0.03 b | 0.98 ± 0.06 b | 1.00 ± 0.01 ab | 1.07 ± 0.07 a |
| PUFA/SFA | 0.36 ± 0.01 | 0.39 ± 0.01 | 0.35 ± 0.02 | 0.36 ± 0.03 |
| Treatment | Aerobic Plate Count (log cfu/g) | E. coli (log cfu/g) | Pseudomonas spp. (log cfu/g) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 Day | 7 Day | 14 Day | 21 Day | 1 Day | 7 Day | 14 Day | 21 Day | 1 Day | 7 Day | 14 Day | 21 Day | |
| CON | 2.43 ± 0.17 d | 4.05 ± 0.03 c,A | 5.71 ± 0.13 b,A | 7.61 ± 0.01 a,A | 1.59 ± 0.26 b | 2.29 ± 0.03 a,A | 2.42 ± 0.07 a,A | 2.61 ± 0.03 aA | 1.97 ± 0.07 d | 3.96 ± 0.02 c,A | 4.57 ± 0.01 b,A | 5.58 ± 0.01 a,A |
| CHI | 2.35 ± 0.23 c | 3.65 ± 0.02 b,B | 4.04 ± 0.05 a,B | 4.11 ± 0.02 a,B | 1.62 ± 0.15 a,b | 1.82 ± 0.11 a,B | 1.72 ± 0.12 a,b,B | 1.36 ± 0.10 b,B | 1.88 ± 0.09 c | 3.52 ± 0.06 a,B | 3.41 ± 0.06 a,B | 2.25 ± 0.04 b,B |
| CHI/0.5% OA | 2.33 ± 0.10 c | 3.46 ± 0.04 b,C | 3.65 ± 0.02 a,b,C | 3.78 ± 0.01 a,C | 1.59 ± 0.11 a,b | 1.73 ± 0.05 a,C,B | 1.59 ± 0.05 b,C | ND | 1.84 ± 0.12 c | 3.47 ± 0.02 aC | 3.28 ± 0.02 b,C | 1.90 ± 0.05 c,C |
| CHI/1% OA | 2.31 ± 0.03 c | 3.38 ± 0.07 b,C | 3.55 ± 0.04 a,C | 3.62 ± 0.02 a,D | 1.53 ± 0.21 | 1.67 ± 0.06 C | 1.51 ± 0.19 C | ND | 1.88 ± 0.06 c | 3.40 ± 0.01 a,C | 3.14 ± 0.09 b,D | 1.52 ± 0.07 d,D |
| Treatment | TBARS (mg MAD/kg) | TVBN (mg/100 g) | ||||||
|---|---|---|---|---|---|---|---|---|
| 1 Day | 7 Day | 14 Day | 21 Day | 1 Day | 7 Day | 14 Day | 21 Day | |
| CON | 0.33 ± 0.01 d | 0.41 ± 0.04 c,A | 0.79 ± 1.20 b,A | 1.03 ± 0.04 a,A | 7.86 ± 0.77 d | 15.35 ± 0.94 c,A | 26.03 ± 1.86 b,A | 56.01 ± 2.40 a,A |
| CHI | 0.34 ± 0.01 d | 0.36 ± 0.02 c,B | 0.40 ± 0.04 B,b | 0.49 ± 0.03 a,B | 7.42 ± 0.98 d | 10.67 ± 0.31 c,B | 16.29 ± 0.69 b,B | 27.53 ± 1.84 a,B |
| CHI/0.5% OA | 0.32 ± 0.01 c | 0.36 ± 0.03 c,b,C | 0.39 ± 0.06 b,B,C | 0.46 ± 0.04 a,C | 7.36 ± 0.71 c | 9.73 ± 0.92 c,B | 15.73 ± 1.07 b,B | 19.79 ± 1.05 a,B,C |
| CHI/1% OA | 0.33 ± 0.01 c | 0.35 ± 0.02 c,B | 0.38 ± 0.03 b,C | 0.45 ± 0.04 a,C | 7.40 ± 0.62 c | 8.61 ± 0.68 c,B | 14.42 ± 0.55 b,B | 17.41 ± 1.85 a,C |
| Treatment | L* (Lightness) | a* (Redness) | ||||||
|---|---|---|---|---|---|---|---|---|
| 1 Day | 7 Day | 14 Day | 21 Day | 1 Day | 7 Day | 14 Day | 21 Day | |
| CON | 52.15 ± 2.27 a | 51.81 ± 2.87 b | 50.29 ± 2.77 Bb | 48.29 ± 3.58 B,c | 17.35 ± 1.26 a | 15.12 ± 1.42 B,b | 13.85 ± 1.67 C,c | 10.65 ± 1.42 C,d |
| CHI | 50.88 ± 3.43 b | 53.72 ± 3.14 a | 51.80 ± 1.88 Aab | 52.50 ± 3.80A a,b | 16.74 ± 1.46 a | 16.11 ± 1.44 A,B,a | 15.05 ± 1.57 B,b | 14.12 ± 1.02 B,b |
| CHI/0.5% OA | 50.88 ± 2.29 | 52.73 ± 3.60 | 53.19 ± 3.23 A | 53.31 ± 2.98 A | 17.13 ± 1.62 a | 16.45 ± 1.60 A,a | 16.05 ± 1.11 A,B,a | 15.78 ± 1.33 A,B,b |
| CHI/1% OA | 52.81 ± 2.89 | 53.83 ± 2.00 | 51.98 ± 3.07 A | 52.24 ± 3.71 A | 17.16 ± 1.81 | 17.10 ± 1.36 A | 16.69 ± 1.18 A | 16.19 ± 2.01 A |
| Reduction percentage (%) of a* (redness) values | pH | |||||||
| CON | 0 | 12.87 ± 1.94 c,A | 20.14 ± 1.76 b,A | 38.59 ± 2.25 a,A | 6.03 ± 0.13 c | 6.10 ± 0.09 b | 6.28 ± 0.04 a,A | 6.33 ± 0.11 a,A |
| CHI | 0 | 3.76 ± 0.91 c,B | 10.04 ± 022 b,B | 15.61 ± 1.99 a,B | 6.02 ± 0.10 b | 6.03 ± 0.09 a,b | 6.06 ± 0.06 a,b,B | 6.12 ± 0.33 a,B |
| CHI/0.5% OA | 0 | 3.99 ± 0.98 b,B | 6.28 ± 0.58 a,B,C | 7.90 ± 0.16 a,B,C | 6.02 ± 0.10 | 6.03 ± 0.09 | 6.09 ± 0.06 C,B | 6.09 ± 0.06 C |
| CHI/1% OA | 0 | 0.36 ± 0.09 B | 2.71 ± 0.16 C | 5.63 ± 0.62 C | 6.02 ± 0.14 | 6.03 ± 0.06 | 6.03 ± 0.02 C | 6.06 ± 0.08 C |
| Items | Retention Time (min) | Sotage Day | CON | CHI | CHI/0.5% OA | CHI/1% OA | Identying Method |
|---|---|---|---|---|---|---|---|
| Aldehydes | |||||||
| 2-Methyl pentanal | 1.660 | 1 | 0.008 ± 0.000 | 0.012 ± 0.001 | 0.012 ± 0.000 | 0.019 ± 0.000 | MS, STD |
| 21 | NF | NF | NF | NF | |||
| 2-Methyl propanal | 1.824 | 1 | 0.001 ± 0.000 b | 0.004 ± 0.000 a | 0.002 ± 0.000 a,b | 0.002 ± 0.000 a,b | MS, STD |
| 21 | 0.001 ± 0.000 | 0.005 ± 0.000 | 0.002 ± 0.000 | 0.003 ± 0.000 | |||
| Butanal | 1.995 | 1 | NF | NF | NF | NF | MS, STD |
| 21 | 0.018 ± 0.000 | 0.008 ± 0.000 | NF | NF | |||
| 2-Ethylhexanal | 2.023 | 1 | NF | NF | NF | NF | MS, STD |
| 21 | 0.033 ± 0.004 | NF | NF | NF | |||
| 3-Methyl butanal | 2.501 | 1 | 0.002 ± 0.000 | 0.036 ± 0.000 | 0.022 ± 0.008 B | 0.019 ± 0.001 | MS, STD |
| 21 | 0.030 ± 0.001 | 0.014 ± 0.003 | 0.040 ± 0.005 A | 0.007 ± 0.000 | |||
| 2-Methyl butanal | 2.599 | 1 | 0.005 ± 0.000 | 0.008 ± 0.000 | 0.003 ± 0.000 | 0.005 ± 0.000 | MS, STD |
| 21 | 0.018 ± 0.006 | 0.009 ± 0.000 | 0.013 ± 0.004 | 0.009 ± 0.000 | |||
| Pentanal | 3.035 | 1 | 0.223 ± 0.034 B | 0.227 ± 0.073 | 0.193 ± 0.001 B | 0.260 ± 0.042 | MS, STD |
| 21 | 0.085 ± 0.004 b,A | 0.314 ± 0.000 a,b | 0.461 ± 0.003 a,A | 0.349 ± 0.001 a,b | |||
| Hexanal | 5.843 | 1 | 1.681 ± 0.064 b,A | 1.250 ± 0.138 b | 2.725 ± 0.006 a,b,B | 3.250 ± 0.408 a | MS, STD |
| 21 | 0.841 ± 0.193 c,B | 1.948 ± 0.648 b | 3.543 ± 0.836 a,A | 3.952 ± 0.775 a | |||
| E,2-Hexenal | 7.354 | 1 | 0.007 ± 0.000 | 0.004 ± 0.000 | 0.008 ± 0.000B | 0.013 ± 0.008 | MS, STD |
| 21 | 0.001 ± 0.000 | 0.011 ± 0.00 | 0.017 ± 0.00A | 0.014 ± 0.00 | |||
| Heptanal | 8.799 | 1 | 0.161 ± 0.039 b,A | 0.142 ± 0.049 b | 0.196 ± 0.022 b,B | 0.315 ± 0.073 a | MS, STD |
| 21 | 0.033 ± 0.001 c,B | 0.261 ± 0.005 b | 0.378 ± 0.049 a,b,A | 0.453 ± 0.079 a | |||
| E,E-2,4-Heptadienal | 9.526 | 1 | NF | NF | 0.002 ± 0.000 b | 0.009 ± 0.000 a | MS, STD |
| 21 | NF | NF | NF | NF | |||
| E,2-Heptenal | 10.279 | 1 | 0.027 ± 0.009 a,b,B | 0.022 ± 0.013 b | 0.036 ± 0.007 a,b | 0.059 ± 0.029 a | MS, STD |
| 21 | 0.056 ± 0.001 A | 0.043 ± 0.002 | 0.076 ± 0.003 | 0.073 ± 0.006 | |||
| Benzaldehyde | 10.349 | 1 | 0.043 ± 0.001 | 0.013 ± 0.001 | 0.014 ± 0.006 | 0.024 ± 0.005 | MS, STD |
| 21 | 0.006 ± 0.000 c | 0.016 ± 0.003 b | 0.034 ± 0.007 a | 0.034 ± 0.002 a | |||
| Octanal | 11.449 | 1 | 0.146 ± 0.016 c,A | 0.151 ± 0.015 b,c | 0.162 ± 0.009 b,B | 0.275 ± 0.088 a,B | MS, STD |
| 21 | 0.081 ± 0.005 d,B | 0.179 ± 0.005 c | 0.281 ± 0.060 b,A | 0.355 ± 0.009 a,A | |||
| Benzeneacetaldehyde | 12.391 | 1 | 0.003 ± 0.002 | 0.005 ± 0.002 | 0.002 ± 0.000 | 0.008 ± 0.000 | MS, STD |
| 21 | 0.007 ± 0.000 | 0.011 ± 0.002 | 0.022 ± 0.005 | 0.011 ± 0.005 | |||
| Nonanal | 1 | 0.076 ± 0.018 c | 0.072 ± 0.015 c | 0.120 ± 0.021 b | 0.179 ± 0.065 a,B | MS, STD | |
| 21 | 0.102 ± 0.009 b | 0.110 ± 0.035 b | 0.204 ± 0.106 b | 0.264 ± 0.017 a,A | |||
| E,2-Octenal | 12.708 | 1 | 0.017 ± 0.005 | 0.013 ± 0.005 | 0.026 ± 0.007 | 0.033 ± 0.008 | MS, STD |
| 21 | 0.008 ± 0.007 | 0.020 ± 0.008 | 0.032 ± 0.002 | 0.047 ± 0.008 | |||
| E,2-Nonenal | 14.826 | 1 | 0.05 ± 0.000 | 0.003 ± 0.000 | 0.005 ± 0.000 | 0.009 ± 0.000 | MS, STD |
| 21 | 0.001 ± 0.000 | 0.006 ± 0.000 | 0.014 ± 0.002 | 0.015 ± 0.000 | |||
| Decanal | 15.711 | 1 | 0.008 ± 0.000 | 0.002 ± 0.000 | 0.006 ± 0.000 | 0.004 ± 0.000 | MS, STD |
| 21 | 0.001 ± 0.000 | 0.008 ± 0.000 | 0.003 ± 0.000 | 0.003 ± 0.000 | |||
| E,E,2,4-Nonadienal | 15.87 | 1 | 0.001 ± 0.000 | 0.001 ± 0.001 | 0.001 ± 0.000 | 0.011 ± 0.016 | MS, STD |
| 21 | 0.001 ± 0.000 | 0.001 ± 0.000 | 0.003 ± 0.000 | 0.003 ± 0.000 | |||
| E,2-Decenal | 16.745 | 1 | 0.004 ± 0.000 | 0.004 ± 0.000 | 0.003 ± 0.000 | 0.006 ± 0.000 | MS, STD |
| 21 | 0.001 ± 0.000 | 0.004 ± 0.000 | 0.010 ± 0.003 | 0.009 ± 0.000 | |||
| E,E,2,4-Decadienal | 17.321 | 1 | 0.001 ± 0.000 | 0.001 ± 0.000 | 0.005 ± 0.005 | 0.016 ± 0.011 | MS, STD |
| 21 | 0.004 ± 0.000 | 0.001 ± 0.000 | 0.004 ± 0.000 | 0.003 ± 0.000 | |||
| 2-Undecenal | 18.519 | 1 | 0.002 ± 0.000 | 0.002 ± 0.000 | 0.001 ± 0.000 | 0.003 ± 0.000 | MS, STD |
| 21 | 0.001 ± 0.000 | 0.002 ± 0.000 | 0.005 ± 0.000 | 0.005 ± 0.000 | |||
| Alcohols | |||||||
| 2-propanol | 1.532 | 1 | NF | NF | NF | NF | MS |
| 21 | 0.032 ± 0.000 | NF | NF | NF | |||
| 2-Propen-1-ol | 2.823 | 1 | 0.003 ± 0.000 B | 0.003 ± 0.000 | 0.003 ± 0.000 | 0.004 ± 0.000 | MS |
| 21 | 0.01 ± 0.001 A | 0.007 ± 0.000 | 0.010 ± 0.000 | 0.008 ± 0.000 | |||
| 1-Pentanol | 4.619 | 1 | 0.191 ± 0.004 | 0.165 ± 0.008 | 0.167 ± 0.008 B | 0.246 ± 0.001 B | MS, STD |
| 21 | 0.048 ± 0.002 c | 0.245 ± 0.021 b | 0.324 ± 0.003 a,b,A | 0.413 ± 0.001 a,A | |||
| 3-Methyl-1-butanol | 3.746 | 1 | NF | NF | NF | NF | MS |
| 21 | 0.019 ± 0.008 | NF | NF | NF | |||
| 1-Hexanol | 7.879 | 1 | 0.025 ± 0.004 b | 0.025 ± 0.005 b | 0.029 ± 0.000 b | 0.045 ± 0.005 a | MS, STD |
| 21 | 0.040 ± 0.021 | 0.116 ± 0.090 | 0.139 ± 0.005 | 0.453 ± 0.006 | |||
| 1-Heptanol | 10.655 | 1 | 0.021 ± 0.007 b,B | 0.017 ± 0.006 b | 0.032 ± 0.002 b | 0.056 ± 0.018 a | MS, STD |
| 21 | 0.04 ± 0.000 A | 0.030 ± 0.003 | 0.048 ± 0.004 | 0.084 ± 0.006 | |||
| 1-Octen-3-ol | 10.885 | 1 | 0.060 ± 0.000 B | 0.037 ± 0.002 B | 0.005 ± 0.000 B | 0.011 ± 0.002 B | MS, STD |
| 21 | 0.18 ± 0.002 a,b,A | 0.142 ± 0.053 b,A | 0.163 ± 0.027 b,A | 0.259 ± 0.065 a,A | |||
| E,2-Octen-1-ol | 12.929 | 1 | 0.005 ± 0.000 | 0.004 ± 0.000 | 0.012 ± 0.004 | 0.015 ± 0.007 | MS |
| 21 | 0.003 ± 0.000 | 0.009 ± 0.000 | 0.011 ± 0.000 | 0.038 ± 0.005 | |||
| 1-Octanol | 12.989 | 1 | 0.012 ± 0.004 b | 0.010 ± 0.002 b | 0.017 ± 0.005 b | 0.027 ± 0.005 a | MS, STD |
| 21 | 0.017 ± 0.005 | 0.015 ± 0.007 | 0.024 ± 0.008 | 0.041 ± 0.000 | |||
| Ketones | |||||||
| 2-Butanone | 2.022 | 1 | 0.004 ± 0.000 | 0.006 ± 0.000 | 0.005 ± 0.000 | 0.005 ± 0.000 | MS, STD |
| 21 | 0.005 ± 0.000 | 0.005 ± 0.000 | 0.004 ± 0.000 | 0.005 ± 0.000 | |||
| Heptanone | 8.394 | 1 | 0.05 ± 0.006 | 0.01 ± 0.000 | 0.021 ± 0.000 B | 0.02 ± 0.000 B | MS, STD |
| 21 | 0.033 ± 0.002 b | 0.026 ± 0.002 b | 0.054 ± 0.005 a,b,A | 0.070 ± 0.000 aA | |||
| 2,3-Butanedione | 1.972 | 1 | NF | NF | NF | NF | MS, STD |
| 21 | 0.020 ± 0.005 | NF | NF | NF | |||
| Pyrazines and sulfur-containing compounds | |||||||
| Methylpyrazine | 6.383 | 1 | 0.001 ± 0.000 b | 0.001 ± 0.000 b | 0.004 ± 0.000 b | 0.022 ± 0.009 a | MS, STD |
| 21 | NF | 0.002 ± 0.00 | 0.003 ± 0.000 | 0.031 ± 0.006 | |||
| 2,5-Dimethylpyrazine | 9.014 | 1 | 0.010 ± 0.009 | 0.020 ± 0.008 | 0.008 ± 0.000 | 0.025 ± 0.009 | MS, STD |
| 21 | NF | 0.008 ± 0.004 | 0.007 ± 0.000 | 0.035 ± 0.007 | |||
| 3-Ethyl-2,5-dimethylpyrazine | 13.172 | 1 | 0.001 ± 0.000 b | 0.005 ± 0.000 a,b | 0.004 ± 0.000 a,b | 0.008 ± 0.000 a | MS |
| 21 | 0.06 ± 0.000 | 0.05 ± 0.000 | 0.06 ± 0.000 | 0.09 ± 0.000 | |||
| Methanethiol | 1.486 | 1 | 0.002 ± 0.000 | 0.004 ± 0.000 | 0.005 ± 0.000 | 0.003 ± 0.000 | MS |
| 21 | 0.002 ± 0.000 | 0.004 ± 0.000 | 0.003 ± 0.000 | 0.002 ± 0.000 | |||
| Carbon disulfide | 1.754 | 1 | 0.002 ± 0.000 | 0.006 ± 0.000 | 0.004 ± 0.000 | 0.005 ± 0.000 | MS, STD |
| 21 | 0.002 ± 0.000 | 0.005 ± 0.000 | 0.026 ± 0.034 | 0.006 ± 0.000 | |||
| Hydrocarbons | |||||||
| 1,2-Propanediol | 1.523 | 1 | 0.072 ± 0.001 b | 0.254 ± 0.161 a | 0.116 ± 0.008 a,b | 0.135 ± 0.001 a,b | MS |
| 21 | 0.004 ± 0.000 b | 0.083 ± 0.001 b | 0.035 ± 0.001 b | 0.291 ± 0.004 a | |||
| Butane | 1.609 | 1 | NF | NF | NF | NF | MS |
| 21 | 0.031 ± 0.013 | NF | NF | NF | |||
| 2-Octene | 5.318 | 1 | 0.003 ± 0.000 | 0.004 ± 0.000 | 0.001 ± 0.000 | 0.004 ± 0.000 | MS |
| 21 | 0.004 ± 0.000 | 0.003 ± 0.000 | 0.008 ± 0.000 | 0.005 ± 0.000 | |||
| Xylene | 7.803 | 1 | 0.003 ± 0.000 B | 0.004 ± 0.000 | 0.004 ± 0.000 | 0.008 ± 0.000 B | MS |
| 21 | 0.024 ± 0.001 A | 0.011 ± 0.005 | 0.028 ± 0.008 | 0.016 ± 0.000 A | |||
| Ethylbenzene | 7.553 | 1 | 0.001 ± 0.000 | 0.002 ± 0.000 | 0.002 ± 0.000 B | 0.003 ± 0.000 | MS, STD |
| 21 | 0.016 ± 0.000 | 0.004 ± 0.000 | 0.009 ± 0.000 A | 0.006 ± 0.000 | |||
| Styrene | 8.439 | 1 | NF | NF | NF | NF | MS, STD |
| 21 | 0.010 ± 0.000 a | 0.003 ± 0.000 b | NF | NF | |||
| Heptanoic acid | 11.149 | 1 | 0.010 ± 0.000 | 0.028 ± 0.008 B | 0.064 ± 0.006 | 0.160 ± 0.005 | MS, STD |
| 21 | 0.014 ± 0.000 | 0.290 ± 0.008 A | 0.345 ± 0.009 | 0.249 ± 0.005 | |||
| Octanoic acid | 15.079 | 1 | 0.004 ± 0.000 B | 0.008 ± 0.000 | 0.009 ± 0.000 | 0.004 ± 0.000 | MS, STD |
| 21 | 0.010 ± 0.000 A | 0.030 ± 0.000 | 0.026 ± 0.002 | 0.005 ± 0.000 | |||
| Dodecane | 15.58 | 1 | 0.017 ± 0.001 | 0.049 ± 0.000 | 0.050 ± 0.001 | 0.064 ± 0.009 | MS, STD |
| 21 | 0.004 ± 0.000 | 0.006 ± 0.000 | 0.004 ± 0.000 | 0.067 ± 0.001 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hoa, V.-B.; Song, D.-H.; Seol, K.-H.; Kang, S.-M.; Kim, H.-W.; Kim, J.-H.; Moon, S.-S.; Cho, S.-H. Application of a Newly Developed Chitosan/Oleic Acid Edible Coating for Extending Shelf-Life of Fresh Pork. Foods 2022, 11, 1978. https://doi.org/10.3390/foods11131978
Hoa V-B, Song D-H, Seol K-H, Kang S-M, Kim H-W, Kim J-H, Moon S-S, Cho S-H. Application of a Newly Developed Chitosan/Oleic Acid Edible Coating for Extending Shelf-Life of Fresh Pork. Foods. 2022; 11(13):1978. https://doi.org/10.3390/foods11131978
Chicago/Turabian StyleHoa, Van-Ba, Dong-Heon Song, Kuk-Hwan Seol, Sun-Moon Kang, Hyun-Wook Kim, Jin-Hyoung Kim, Sung-Sil Moon, and Soo-Hyun Cho. 2022. "Application of a Newly Developed Chitosan/Oleic Acid Edible Coating for Extending Shelf-Life of Fresh Pork" Foods 11, no. 13: 1978. https://doi.org/10.3390/foods11131978
APA StyleHoa, V.-B., Song, D.-H., Seol, K.-H., Kang, S.-M., Kim, H.-W., Kim, J.-H., Moon, S.-S., & Cho, S.-H. (2022). Application of a Newly Developed Chitosan/Oleic Acid Edible Coating for Extending Shelf-Life of Fresh Pork. Foods, 11(13), 1978. https://doi.org/10.3390/foods11131978






