Saccharomyces cerevisiae Diversity in Arbutus unedo L. Fermentations in Association with the Volatile and Sensory Similarities of the Distillates
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experiment and Sampling
2.2. Microbiological Analysis
2.3. Yeast Isolation
2.4. Saccharomyces cerevisiae Characterization and Differentiation
2.4.1. Cell Lysis
2.4.2. Primer Specific for S. cerevisiae
2.4.3. Microsatellite Analysis with SSR Markers
2.4.4. Statistical Analysis
2.5. Distillation Description and Distillate Sampling
2.6. Analytical Distillate Determinations
Standards and Chemicals
2.7. Sensory Analysis of Distillate Samples
2.7.1. Sample Preparation and Presentation
2.7.2. Sorting Task
3. Results and Discussion
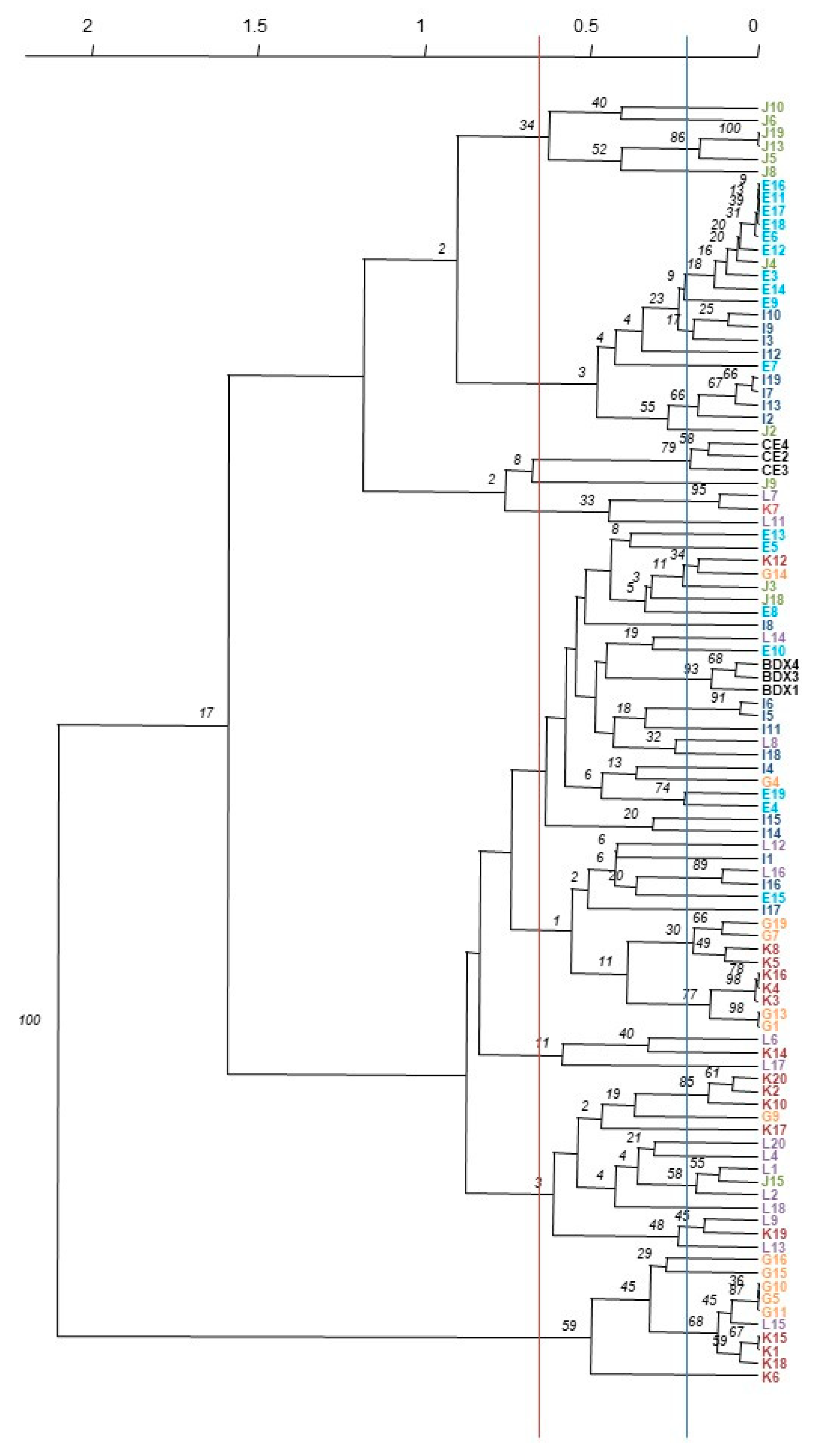
3.1. S. cerevisiae Characterization and Differentiation
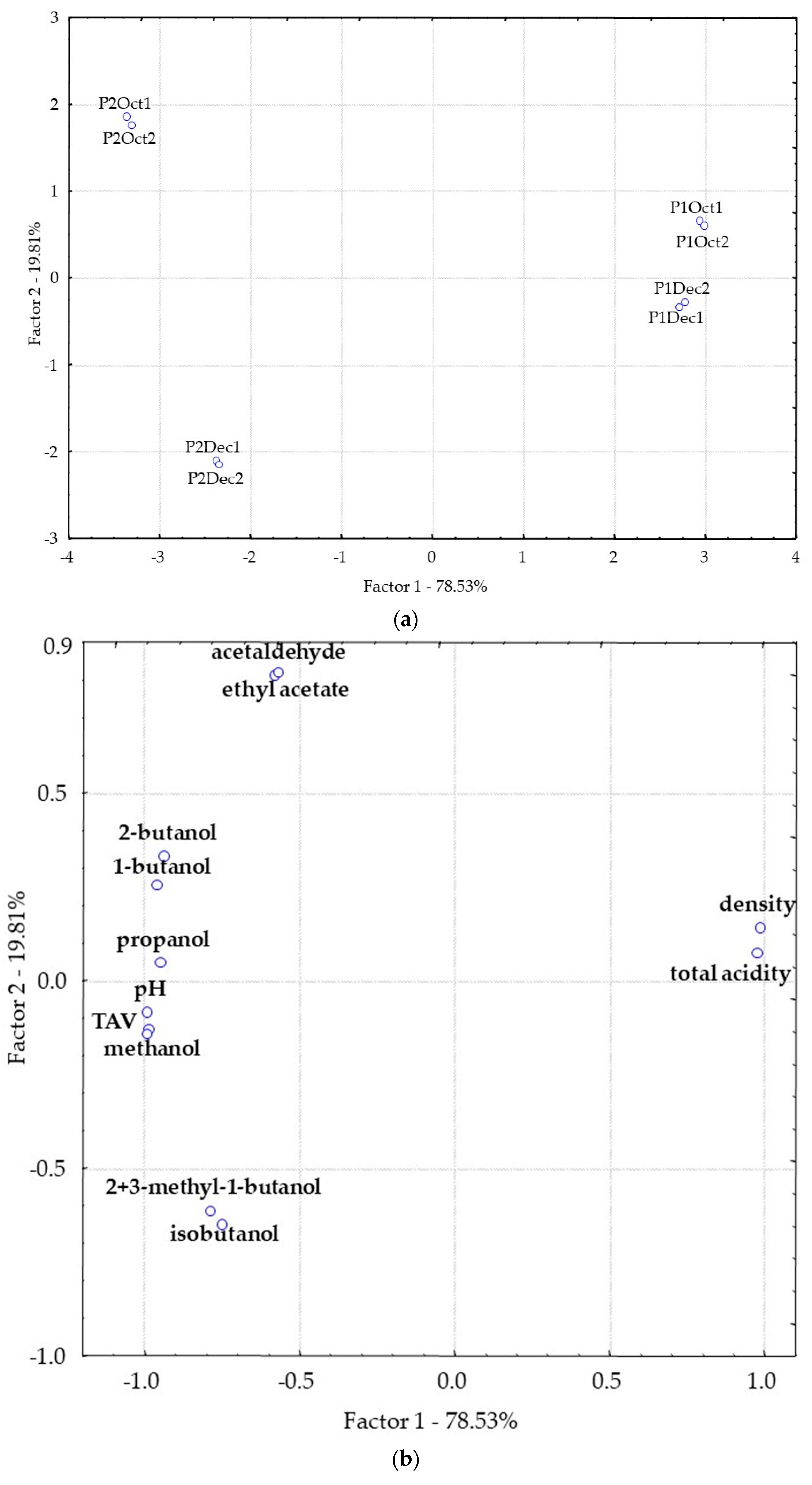
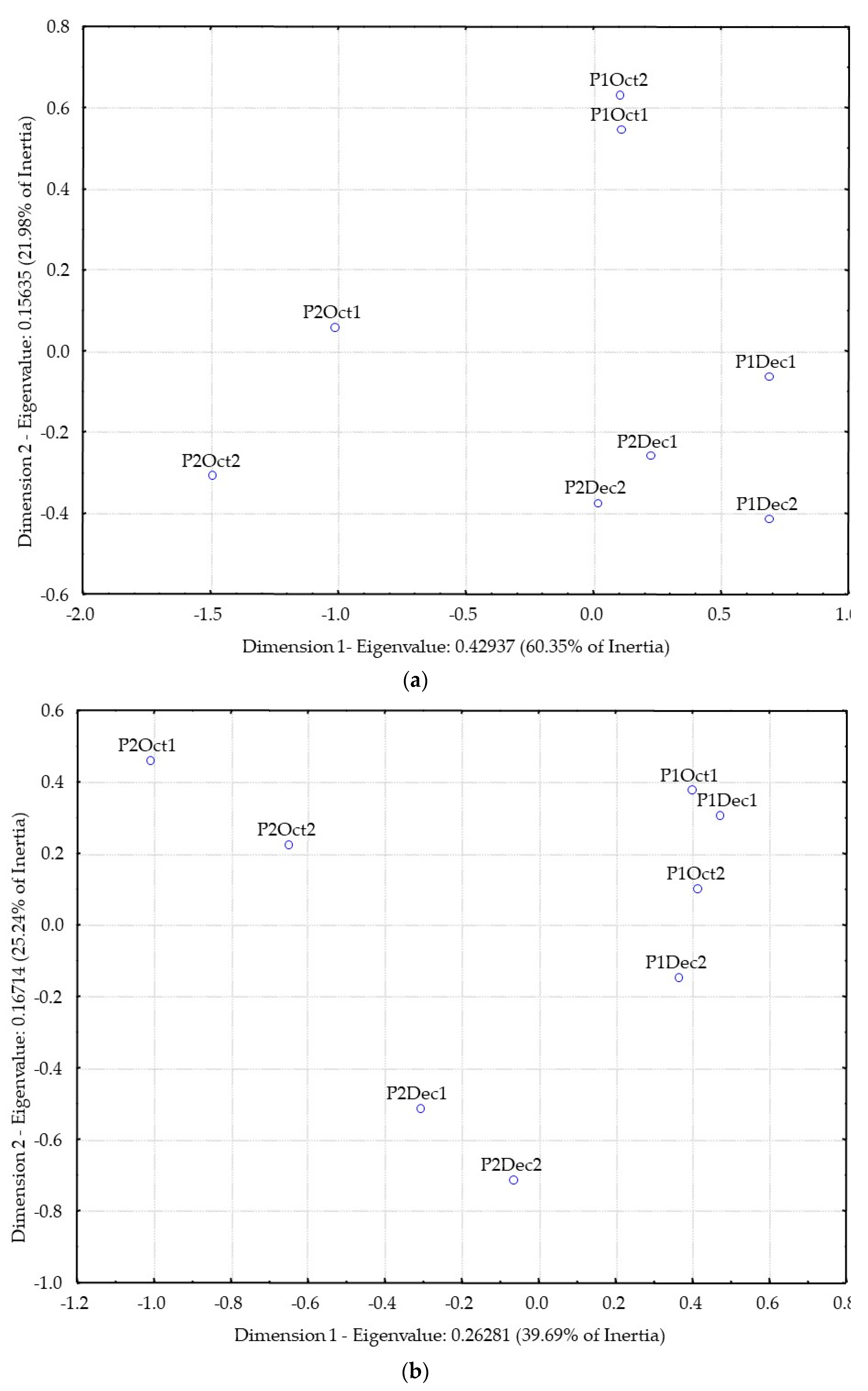
3.2. Distillates Characterization and Differentiation
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tenuta, M.C.; Tundis, R.; Xiao, J.; Loizzo, M.R.; Dugay, A.; Deguin, B. Arbutus species (Ericaceae) as source of valuable bioactive products. Crit. Rev. Food Sci. Nutr. 2018, 59, 864–881. [Google Scholar] [CrossRef] [PubMed]
- Pato, R.L.; Botelho, G.; Franco, J.; Santos, S.; Ressurreição, S.; Figueiredo, P.; Gama, J.; Gomes, F. Interaction between farming type, nutrient uptake and plant material in strawberry tree fruit production and quality. Acta Hortic. 2022, 1333, 275–284. [Google Scholar] [CrossRef]
- Garrido, N.; Silveira, T. Medronho: O fruto vermelho que Portugal está a (re)descobrir. In Público, Público—Comunicação Social, S.A. Lugar do Espido, Via Norte, Maia, Portugal; 2020; Available online: https://www.publico.pt/2020/11/20/sociedade/noticia/medronho-fruto-vermelho-futuro-portugal-redescobrir-1939941 (accessed on 5 April 2022).
- Celikel, G.; Demirsoy, L.; Demirsoy, H. The strawberry tree (Arbutus unedo L.) selection in Turkey. Sci. Hortic. 2008, 118, 115–119. [Google Scholar] [CrossRef]
- Ruiz-Rodríguez, B.-M.; Morales, P.; Fernández-Ruiz, V.; Sánchez-Mata, M.-C.; Cámara, M.; Díez-Marqués, C.; Pardo-de-Santayana, M.; Molina, M.; Tardío, J. Valorization of wild strawberry-tree fruits (Arbutus unedo L.) through nutritional assessment and natural production data. Food Res. Int. 2011, 44, 1244–1253. [Google Scholar] [CrossRef]
- Martins, J.; Pinto, G.; Canhoto, J. Biotechnology of the multipurpose tree species Arbutus unedo: A review. J. For. Res. 2022, 33, 377–390. [Google Scholar] [CrossRef]
- Caldeira, I.; Gomes, F.; Mira, H.; Botelho, G. Distillates composition obtained of fermented Arbutus unedo L. fruits from different seedlings and clonal plants. Ann. Agric. Sci. 2019, 64, 21–28. [Google Scholar] [CrossRef]
- Santo, D.E.; Galego, L.; Gonçalves, T.; Quintas, C. Yeast diversity in the Mediterranean strawberry tree (Arbutus unedo L.) fruits’ fermentations. Food Res. Int. 2012, 47, 45–50. [Google Scholar] [CrossRef]
- Botelho, G.; Galego, L. Manual de Boas Práticas de Fabrico de Aguardente de Medronho, 3rd ed.; Quântica Editora—Conteúdos Especializados, Lda.: Porto, Portugal, 2020; pp. 1–81. [Google Scholar]
- Cardoso, J.C.; Bessa, M.T.; Marado, M.B. Carta dos solos de Portugal (1: 1000000). Sep. Agron. Lusit. 1973, 33, 481–602. [Google Scholar]
- Soares da Silva, A.M. Carta Litológica—Notícia Explicativa I.13; 37, Atlas do Ambiente, Estação Agronómica Nacional, Comissão Nacional do Ambiente, (Ed.); Comissão Nacional do Ambiente: Lisboa, Portugal, 1983. [Google Scholar]
- RESOLUTION OIV/OENO 206/2010; Detection, Differentiation and Counting of Microorganisms—Microbiological Analysis of Wines and Musts. OIV, International Organisation of Vine and Wine: Paris, France, 2010. Available online: https://www.oiv.int/public/medias/1224/oiv-oeno-206-2010-en.pdf (accessed on 5 April 2022).
- Martorell, P.; Querol, A.; Fernández-Espinar, M.T. Rapid Identification and Enumeration of Saccharomyces cerevisiae Cells in Wine by Real-Time PCR. Appl. Environ. Microbiol. 2005, 71, 6823–6830. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Legras, J.L.; Ruh, O.; Merdinoglu, D.; Karst, F. Selection of hypervariable microsatellite loci for the characterization of Saccharomyces cerevisiae strains. Int. J. Food Microbiol. 2005, 102, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Duarte, F.L.; Alves, A.C.; Baleiras-Couto, M.M. Influence of red wine fermentation oenological additives on inoculated strain implantation. World J. Microbiol. Biotechnol. 2013, 29, 1139–1144. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019; Available online: https://www.R-project.org/ (accessed on 21 November 2019).
- Duarte, F.L.; Baleiras-Couto, M.M. Survey of Inoculated Commercial Saccharomyces cerevisiae in Winery-Based Trials. Fermentation 2021, 7, 176. [Google Scholar] [CrossRef]
- Botelho, G.; Gomes, F.; Ferreira, F.M.; Caldeira, I. Influence of maturation degree of Arbutus (Arbutus unedo L.) fruits in spirit composition and quality. Int. J. Biol. Biomol. Agric. Food Biotechnol. Eng. 2015, 9, 551–556. [Google Scholar]
- Luís, A.; Mota, D.; Anjos, O.; Caldeira, I. Single-laboratory validation of determination of acetaldehyde, ethyl acetate, methanol and fusel alcohols in wine spirits, brandies and grape marc spirits using GC-FID. Ciência Téc. Vitiv. 2011, 26, 69–76. [Google Scholar]
- ISO 3591; Sensory Analysis—Apparatus-Wine-Tasting Glass. ISO—International Organization for Standardization: Geneva, Switzerland, 1977.
- Macfie, H.J.M.; Bratchell, N.; Greenhoff, H.; Vallis, L.V. Designs to balance the effects of order of presentation and first order carryover effects in hall tests. J. Sens. Stud. 1989, 4, 129–148. [Google Scholar] [CrossRef]
- Cariou, V.; Qannari, E.M. Statistical treatment of free sorting data by means of correspondence and cluster analyses. Food Qual. Prefer. 2018, 68, 1–11. [Google Scholar] [CrossRef]
- Valentin, D.; Cholet, S.; Nestrud, M.; Abdi, H. Projective mapping and sorting tasks. In Descriptive Analysis in Sensory Evaluation; Hort, J., Kemp, S., Hollowood, T., Eds.; Wiley-Blackwell: London, UK, 2016; Volume 20, pp. 206–209. [Google Scholar]
- Cavaco, T.; Longuinho, C.; Quintas, C.; Carvalho, I.S. Chemical and microbial changes during the natural fermentation of strawberry tree (Arbutus Unedo L.) fruits. J. Food Biochem. 2007, 21, 715–725. [Google Scholar] [CrossRef]
- Baleiras Couto, M.M.; Reizinho, R.G.; Duarte, F.L. Partial 26S rDNA Restriction Analysis as a tool to Characterise non-Saccharomyces Yeasts present during Red Wine Fermentations. Int. J. Food Microbiol. 2005, 102, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Franco, W.; Benavides, S.; Valencia, P.; Ramírez, C.; Urtubia, A. Native Yeasts and Lactic Acid Bacteria Isolated from Spontaneous Fermentation of Seven Grape Cultivars from the Maule Region (Chile). Foods 2021, 10, 1737. [Google Scholar] [CrossRef]
- De Simone, N.; Russo, P.; Tufariello, M.; Fragasso, M.; Solimando, M.; Capozzi, V.; Grieco, F.; Spano, G. Autochthonous Biological Resources for the Production of Regional Craft Beers: Exploring Possible Contributions of Cereals, Hops, Microbes, and Other Ingredients. Foods 2021, 10, 1831. [Google Scholar] [CrossRef]
| Location | Farming Type | Year of Installation | Average Annual Temperature (°C) | Average Annual Rainfall (mm) | Soil Classification (WRB) |
|---|---|---|---|---|---|
| Estreito (P1) | Orchard from seedlings | 2005 | 7.5–10.0 | 1200–1400 | Cambisols |
| Signo Samo (P2) | Orchard from clonal plants | 2014 | 12.5–15.0 | 1200–1400 | Dystric Leptosols |
| Date of Harvest | Sampling Period | Producer 1 (P1) | Producer 2 (P2) |
|---|---|---|---|
| October 2019 | December 2019 (T1) | E (1–20) | G (1–20) |
| February 2020 (T2) | I (1–20) | K (1–20) | |
| December 2019 | December 2019 (T1) | F (1–20) | H (1–20) |
| February 2020 (T2) | J (1–20) | L (1–20) |
| Date of Harvest | Producer 1 (P1) | Producer 2 (P2) |
|---|---|---|
| October 2019 | P1 October (E,I) * | P2 October (G,K) * |
| December 2019 | P1 December (F,J) * | P2 December (H,L) * |
| Producer | Date of Harvest | Sampling Time | Sample Code | °Bx | Temperature (°C) |
|---|---|---|---|---|---|
| P1 | 31 October 2019 | T1 | E | 19.8 | 14.2 |
| T2 | I | 16.2 | 14.0 | ||
| P1 | 6 December 2019 | T1 | F | 18.3 | 14.1 |
| T2 | J | 8.3 | 14.0 | ||
| P2 | 23 October 2019 | T1 | G | 17.0 | 13.0 |
| T2 | K | 14.1 | 13.0 | ||
| P2 | 3 December 2019 | T1 | H | 18.8 | 13.0 |
| T2 | L | 9.5 | 13.0 |
| Producer | Sample Code | Sampling Period | Fermentation Days | Total Count (cfu/mL) | Yeast (cfu/mL) | Acetic Bacteria (cfu/mL) |
|---|---|---|---|---|---|---|
| E | T1 | 38 | 2.4 × 107 | 2.7 × 107 | <1 * | |
| I | T2 | 96 | 1.4 × 107 | 1.5 × 107 | <1 * | |
| P1 | F | T1 | 2 | 5.5 × 106 | 3.4 × 106 | 1.7 × 106 |
| J | T2 | 60 | 1.9 × 107 | 1.8 × 107 | <1 * | |
| G | T1 | 46 | 1.8 × 107 | 1.6 × 107 | <1 * | |
| K | T2 | 140 | 1.8 × 107 | 1.5 × 107 | <4 * | |
| P2 | H | T1 | 5 | 1.2 × 107 | 1.3 × 107 | 7.4 × 104 |
| L | T2 | 63 | 2.5 × 107 | 2.5 × 107 | <4 * |
| Code Samples | S. cerevisiae (Isolate Number) | Non-Saccharomyces cerevisiae (Isolate Number) |
|---|---|---|
| E | 1, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19 | 2, 20 |
| F | 1–20 | |
| G | 1, 4, 5, 7, 9, 10, 11, 13, 14, 15, 16, 19 | 2, 3, 6, 8, 12, 17, 18, 20 |
| H | 1–20 | |
| I | 1–19 | 20 |
| J | 2, 3, 4, 5, 6, 8, 9, 10, 13, 15, 18, 19 | 1, 7, 11, 12, 14, 16, 17, 20 |
| K | 1, 2, 3, 4, 5, 6, 7, 8, 10, 12, 14, 15, 16, 17, 18, 19, 20 | 6, 11, 13 |
| L | 1, 2, 4, 6, 7, 8, 9, 11, 12, 13, 14, 15, 16, 17, 18, 20 | 3, 5, 10, 19 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baleiras-Couto, M.M.; Caldeira, I.; Gomes, F.; Botelho, G.; Duarte, F.L. Saccharomyces cerevisiae Diversity in Arbutus unedo L. Fermentations in Association with the Volatile and Sensory Similarities of the Distillates. Foods 2022, 11, 1916. https://doi.org/10.3390/foods11131916
Baleiras-Couto MM, Caldeira I, Gomes F, Botelho G, Duarte FL. Saccharomyces cerevisiae Diversity in Arbutus unedo L. Fermentations in Association with the Volatile and Sensory Similarities of the Distillates. Foods. 2022; 11(13):1916. https://doi.org/10.3390/foods11131916
Chicago/Turabian StyleBaleiras-Couto, M. Margarida, Ilda Caldeira, Filomena Gomes, Goreti Botelho, and Filomena L. Duarte. 2022. "Saccharomyces cerevisiae Diversity in Arbutus unedo L. Fermentations in Association with the Volatile and Sensory Similarities of the Distillates" Foods 11, no. 13: 1916. https://doi.org/10.3390/foods11131916
APA StyleBaleiras-Couto, M. M., Caldeira, I., Gomes, F., Botelho, G., & Duarte, F. L. (2022). Saccharomyces cerevisiae Diversity in Arbutus unedo L. Fermentations in Association with the Volatile and Sensory Similarities of the Distillates. Foods, 11(13), 1916. https://doi.org/10.3390/foods11131916








