Chemical, Functional, and Technological Features of Grains, Brans, and Semolina from Purple and Red Durum Wheat Landraces
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
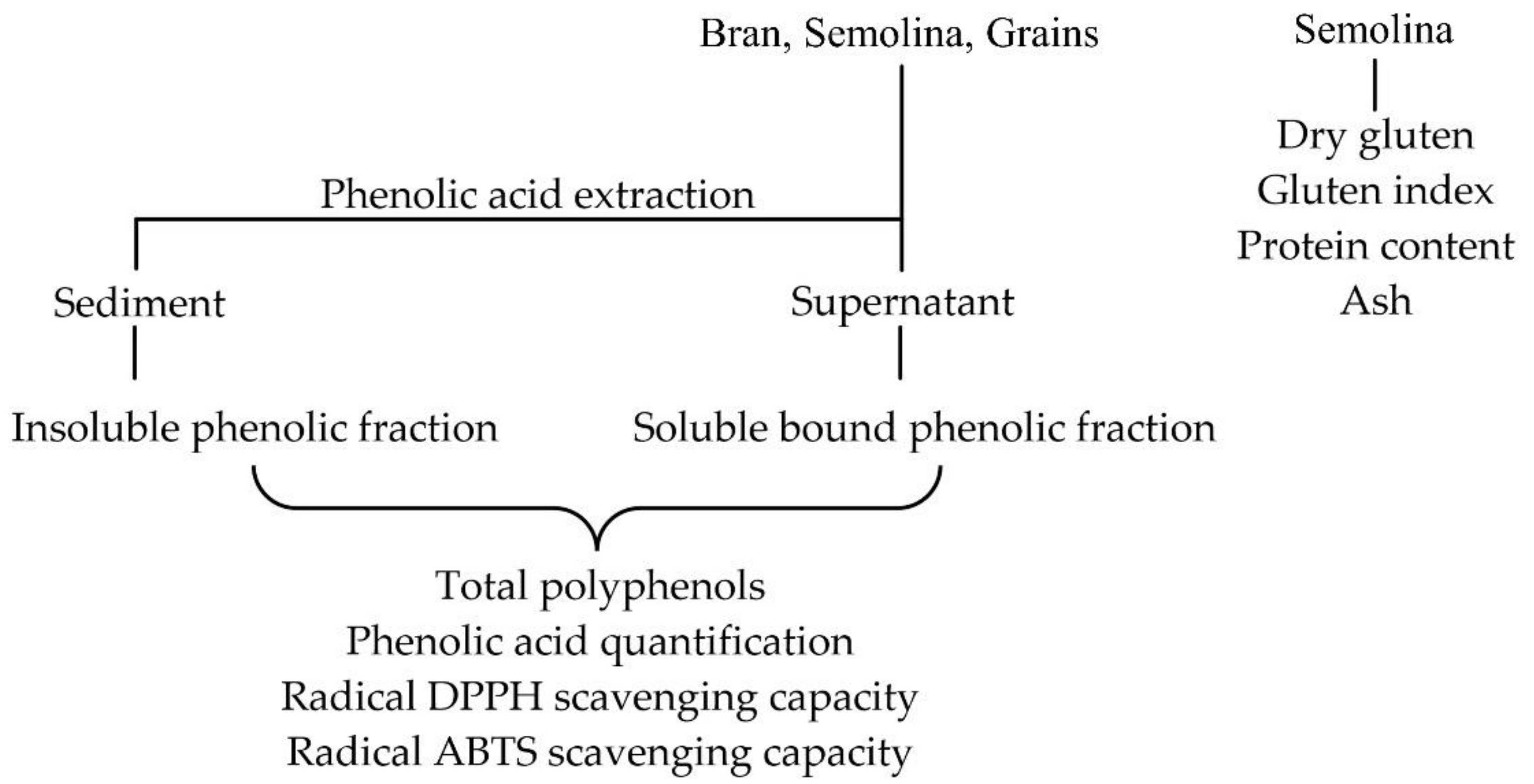
2.2. Determination of Dry Gluten
2.3. Determination of Gluten Index
2.4. Determination of Total Protein Content
2.5. Determination of Ash
2.6. Phenolic Extraction
2.7. Soluble Phenolic Fraction
2.8. Insoluble Bound Phenolic Fraction
2.9. Total Polyphenols
2.10. Phenolic Acid Quantification by HPLC
2.11. Determination of Anthocyanins Content
2.12. Determination of Proanthocyanidin Content
2.13. Radical DPPH Scavenging Capacity
2.14. Radical ABTS Scavenging Capacity
2.15. Statistical Analysis
3. Results and Discussion
3.1. Analysis of Dry Gluten, Gluten Index, Protein Content, and Ash
3.2. Polyphenol Content
3.3. Determination of Phenolic Acids
3.4. Anthocyanins and Proanthocyanidins Content
3.5. Antioxidant Activity
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hasanuzzaman, M.; Nahar, K.; Hossain, M.A. Wheat Production in Changing Environments; Spinger: Dordrecht, The Netherlands, 2019. [Google Scholar]
- Senatore, M.; Ward, T.; Cappelletti, E.; Beccari, G.; McCormick, S.; Busman, M.; Laraba, I.; O‘Donnell, K.; Prodi, A. Species diversity and mycotoxin production by members of the Fusarium tricinctum species complex associated with Fusarium head blight of wheat and barley in Italy. Int. J. Food Microbiol. 2021, 358, 109298. [Google Scholar] [CrossRef]
- Lupton, F. Wheat Vreeding: Its Scientific Basis; Spinger: Dordrecht, The Netherlands, 2014. [Google Scholar]
- Royo, C.; Nachit, M.M.; Di Fonzo, N.; Araus, J.L.; Pfeiffer, W.H.; Slafer, G.A. Durum Wheat Breeding: Current Approaches and Future Strategies, Volumes 1 and 2; Food Products Press: Binghamton, NY, USA, 2005; p. 525. [Google Scholar]
- Ficco, D.B.; Mastrangelo, A.M.; Trono, D.; Borrelli, G.M.; De Vita, P.; Fares, C.; Beleggia, R.; Platani, C.; Papa, R. The colours of durum wheat: A review. Crop Pasture Sci. 2014, 65, 1–15. [Google Scholar] [CrossRef]
- Arzani, A.; Ashraf, M. Cultivated ancient wheats (Triticum spp.): A potential source of health-beneficial food products. Comp. Rev. Food Sci. Food Safety 2017, 16, 477–488. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cooper, R. Re-discovering ancient wheat varieties as functional foods. J. Tradit. Complementary Med. 2015, 5, 138–143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dinu, M.; Whittaker, A.; Pagliai, G.; Benedettelli, S.; Sofi, F. Ancient wheat species and human health: Biochemical and clinical implications. J. Nutr. Biochem. 2018, 52, 1–9. [Google Scholar] [CrossRef]
- Shoeva, O.Y.; Gordeeva, E.I.; Khlestkina, E.K. The regulation of anthocyanin synthesis in the wheat pericarp. Molecules 2014, 19, 20266–20279. [Google Scholar] [CrossRef] [Green Version]
- Lachman, J.; Martinek, P.; Kotíková, Z.; Orsák, M.; Šulc, M. Genetics and chemistry of pigments in wheat grain–A review. J. Cereal Sci. 2017, 74, 145–154. [Google Scholar] [CrossRef]
- Brandolini, A.; Hidalgo, A.; Gabriele, S.; Heun, M. Chemical composition of wild and feral diploid wheats and their bearing on domesticated wheats. J. Cereal Sci. 2015, 63, 122–127. [Google Scholar] [CrossRef]
- Yilmaz, V.A.; Brandolini, A.; Hidalgo, A. Phenolic acids and antioxidant activity of wild, feral and domesticated diploid wheats. J. Cereal Sci. 2015, 64, 168–175. [Google Scholar] [CrossRef]
- Newton, A.C.; Akar, T.; Baresel, J.P.; Bebeli, P.J.; Bettencourt, E.; Bladenopoulos, K.V.; Czembor, J.H.; Fasoula, D.A.; Katsiotis, A.; Koutis, K. Cereal landraces for sustainable agriculture. Sustain. Agric. 2011, 2, 147–186. [Google Scholar]
- AACC. Method 38-12.02, 11th ed.; AACC International, Inc.: St. Paul, MN, USA, 2005. [Google Scholar]
- Hayes, M. Measuring protein content in food: An overview of methods. Foods 2020, 9, 1340. [Google Scholar] [CrossRef]
- Ismail, B.P. Ash content determination. In Food Analysis Laboratory Manual; Spinger: Dordrecht, Netherlands, 2017; pp. 117–119. [Google Scholar]
- Abdel-Aal, E.-S.; Hucl, P.; Sosulski, F.; Graf, R.; Gillott, C.; Pietrzak, L. Screening spring wheat for midge resistance in relation to ferulic acid content. J. Agric. Food Chem. 2001, 49, 3559–3566. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. [14] Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. In Methods Enzymol.; Elsevier: Amsterdam, The Netherlands, 1999; Volume 299, pp. 152–178. [Google Scholar]
- Bastola, K.P.; Guragain, Y.N.; Bhadriraju, V.; Vadlani, P.V. Evaluation of standards and interfering compounds in the determination of phenolics by Folin-Ciocalteu assay method for effective bioprocessing of biomass. Am. J. Anal. Chem. 2017, 8, 416–431. [Google Scholar] [CrossRef] [Green Version]
- Medina, M.B. Simple and rapid method for the analysis of phenolic compounds in beverages and grains. J. Agric. Food Chem. 2011, 59, 1565–1571. [Google Scholar] [CrossRef]
- Dellagreca, M.; Fiorentino, A.; Izzo, A.; Napoli, F.; Purcaro, R.; Zarrelli, A. Phytotoxicity of secondary metabolites from Aptenia cordifolia. Chem. Biodivers. 2007, 4, 118–128. [Google Scholar] [CrossRef]
- Hosseinian, F.S.; Li, W.; Beta, T. Measurement of anthocyanins and other phytochemicals in purple wheat. Food Chem. 2008, 109, 916–924. [Google Scholar] [CrossRef]
- Siebenhandl, S.; Grausgruber, H.; Pellegrini, N.; Del Rio, D.; Fogliano, V.; Pernice, R.; Berghofer, E. Phytochemical profile of main antioxidants in different fractions of purple and blue wheat, and black barley. J. Agric. Food Chem. 2007, 55, 8541–8547. [Google Scholar] [CrossRef]
- Sun, B.; Ricardo-da-Silva, J.M.; Spranger, I. Critical factors of vanillin assay for catechins and proanthocyanidins. J. Agric. Food Chem. 1998, 46, 4267–4274. [Google Scholar] [CrossRef]
- Lavelli, V.; Hidalgo, A.; Pompei, C.; Brandolini, A. Radical scavenging activity of einkorn (Triticum monococcum L. subsp. monococcum) wholemeal flour and its relationship to soluble phenolic and lipophilic antioxidant content. J. Cereal Sc. 2009, 49, 319–321. [Google Scholar]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Vida, G.; Szunics, L.; Veisz, O.; Bedő, Z.; Láng, L.; Árendás, T.; Bónis, P.; Rakszegi, M. Effect of genotypic, meteorological and agronomic factors on the gluten index of winter durum wheat. Euphytica 2014, 197, 61–71. [Google Scholar] [CrossRef] [Green Version]
- Ames, N.; Clarke, J.; Marchylo, B.; Dexter, J.; Woods, S. Effect of environment and genotype on durum wheat gluten strength and pasta viscoelasticity. Cereal Chem. 1999, 76, 582–586. [Google Scholar] [CrossRef]
- Oikonomou, N.; Bakalis, S.; Rahman, M.; Krokida, M. Gluten index for wheat products: Main variables in affecting the value and nonlinear regression model. Int. J. Food Prop. 2015, 18, 1–11. [Google Scholar] [CrossRef]
- Clarke, F.; Clarke, J.; Pozniak, C.; Knox, R.; McCaig, T. Protein concentration inheritance and selection in durum wheat. Can. J. Plant Sci. 2009, 89, 601–612. [Google Scholar] [CrossRef]
- Cecchini, C.; Bresciani, A.; Menesatti, P.; Pagani, M.A.; Marti, A. Assessing the rheological properties of durum wheat semolina: A Review. Foods 2021, 10, 2947. [Google Scholar] [CrossRef]
- Ames, N.; Clarke, J.; Marchylo, B.; Dexter, J.; Schlichting, L.; Woods, S. The effect of extra-strong gluten on quality parameters in durum wheat. Can. J. Plant Sci. 2003, 83, 525–532. [Google Scholar] [CrossRef]
- Peterson, C. Influence of cultivar and environment on mineral and protein concentrations of wheat flour, bran, and grain. Cereal Chem. 1986, 63, 118–186. [Google Scholar]
- Fares, C.; Troccoli, A.; Di Fonzo, N. Use of friction debranning to evaluate ash distribution in Italian durum wheat cultivars. Cereal Chem. 1996, 73, 232–234. [Google Scholar]
- Dykes, L.; Rooney, L. Phenolic compounds in cereal grains and their health benefits. Cereal Foods World 2007, 52, 105–111. [Google Scholar] [CrossRef]
- Hodzic, Z.; Pasalic, H.; Memisevic, A.; Srabovic, M.; Saletovic, M.; Poljakovic, M. The influence of total phenols content on antioxidant capacity in the whole grain extracts. Eur. J. Sci. Res. 2009, 28, 471–477. [Google Scholar]
- Salunkhe, D.; Jadhav, S.; Kadam, S.; Chavan, J.; Luh, B. Chemical, biochemical, and biological significance of polyphenols in cereals and legumes. Crit. Rev. Food Sci. Nutr. 1983, 17, 277–305. [Google Scholar] [CrossRef]
- Van Hung, P. Phenolic compounds of cereals and their antioxidant capacity. Crit. Rev. Food Sci. Nutr. 2016, 56, 25–35. [Google Scholar] [CrossRef]
- Lafiandra, D.; Masci, S.; Sissons, M.; Dornez, E.; Delcour, J.; Courtin, C.; Caboni, M.F. Kernel components of technological value. In Durum Wheat Chemistry and Technology, 2nd ed.; Sissons, M., Marchylo, B., Abecassis, J., Eds.; AACC International Inc.: St. Paul, MN, USA, 2012; pp. 85–124. [Google Scholar]
- Khoo, H.E.; Azlan, A.; Tang, S.T.; Lim, S.M. Anthocyanidins and anthocyanins: Colored pigments as food, pharmaceutical ingredients, and the potential health benefits. Food Nutr. Res. 2017, 61, 1361779. [Google Scholar] [CrossRef] [Green Version]
- Abdel-Aal, E.-S.M.; Hucl, P. Composition and stability of anthocyanins in blue-grained wheat. J. Agric. Food Chem. 2003, 51, 2174–2180. [Google Scholar] [CrossRef]
- Abdel-Aal, E.-S.M.; Young, J.C.; Rabalski, I. Anthocyanin composition in black, blue, pink, purple, and red cereal grains. J. Agric. Food Chem. 2006, 54, 4696–4704. [Google Scholar] [CrossRef]
- Hu, C.; Cai, Y.-Z.; Li, W.; Corke, H.; Kitts, D.D. Anthocyanin characterization and bioactivity assessment of a dark blue grained wheat (Triticum aestivum L. cv. Hedong Wumai) extract. Food Chem. 2007, 104, 955–961. [Google Scholar] [CrossRef]
- Biolley, J.; Jay, M. Anthocyanins in modern roses: Chemical and colorimetric features in relation to the colour range. J. Exp. Bot. 1993, 44, 1725–1734. [Google Scholar] [CrossRef]
- Fossen, T.; Cabrita, L.; Andersen, O.M. Colour and stability of pure anthocyanins influenced by pH including the alkaline region. Food Chem. 1998, 63, 435–440. [Google Scholar] [CrossRef]
- Torskangerpoll, K.; Andersen, Ø.M. Colour stability of anthocyanins in aqueous solutions at various pH values. Food Chem. 2005, 89, 427–440. [Google Scholar] [CrossRef]
- Dedio, W.; Hill, R.; Evans, L. Anthocyanins in the pericarp and coleoptiles of purple wheat. Can. J. Plant Sc. 1972, 52, 977–980. [Google Scholar] [CrossRef]
- Dedio, W.; Hill, R.; Evans, L. Anthocyanins in the pericarp and coleoptiles of purple-seeded rye. Can. J. Plant Sc. 1972, 52, 981–983. [Google Scholar] [CrossRef] [Green Version]
- Abdel-Aal, E.S.M.; Hucl, P.; Shipp, J.; Rabalski, I. Compositional differences in anthocyanins from blue-and purple-grained spring wheat grown in four environments in Central Saskatchewan. Cereal Chem. 2016, 93, 32–38. [Google Scholar] [CrossRef]
- Cammerata, A.; Laddomada, B.; Milano, F.; Camerlengo, F.; Bonarrigo, M.; Masci, S.; Sestili, F. Qualitative characterization of unrefined durum wheat air-classified fractions. Foods 2021, 10, 2817. [Google Scholar] [CrossRef]
- Hemery, Y.; Rouau, X.; Lullien-Pellerin, V.; Barron, C.; Abecassis, J. Dry processes to develop wheat fractions and products with enhanced nutritional quality. J. Cereal Sci. 2007, 46, 327–347. [Google Scholar] [CrossRef]
- Prabhasankar, P.; Haridas Rao, P. Effect of different milling methods on chemical composition of whole wheat flour. Eur. Food Res. Technol. 2001, 213, 465–469. [Google Scholar] [CrossRef]
| Parameters (%) | ‘Purple’ | ‘Red’ |
|---|---|---|
| Protein | 15.20 ± 0.10 b | 16.80 ± 0.20 a |
| Dry gluten | 12.80 ± 0.88 | 12.50 ± 0.06 |
| Ash | 1.25 ± 0.02 b | 1.35 ± 0.01 a |
| Gluten index | 37.00 ± 0.20 a | 31.00 ± 0.60 b |
| Polyphenols (µg/g DM Gallic Acid Equivalent) | Bran | Semolina | Grain | |||
|---|---|---|---|---|---|---|
| ‘Purple’ | ‘Red’ | ‘Purple’ | ‘Red’ | ‘Purple’ | ‘Red’ | |
| Soluble phenolic fraction | 112 ± 21 a | 64 ± 4 b | 31 ± 4 b | 49 ± 3 a | 103 ± 15 a | 65 ± 5 b |
| Insoluble bound phenolic fraction | 2391 ± 471 a | 1430 ± 284 b | 751 ± 105 | 849 ± 112 | 2380 ± 455 | 1840 ± 329 |
| Soluble Hpenolic Fraction (mg/kg DM) | ‘Purple’ | ‘Red’ |
|---|---|---|
| Ferulic acid | 17.9 ± 1.10 | 18.3 ± 1.05 |
| p-Hydroxybenzoic acid | 3.1 ± 0.28 | 3.3 ± 0.27 |
| Vanillic acid | 7.2 ± 0.33 | 7.4 ± 0.38 |
| p-Coumaric acid | 2.2 ± 0.32 | 2.5 ± 0.30 |
| Insoluble Bound Phenolic Fraction (mg/kg DM) | ||
| Ferulic acid | 625 ± 39.3 | 639 ± 41.4 |
| p-Hydroxybenzoic acid | 2.1 ± 0.29 | 2.3 ± 0.28 |
| Vanillic acid | 3.1 ± 0.35 | 3.4 ± 0.29 |
| p-Coumaric acid | 18.5 ± 2.35 | 19.2 ± 2.26 |
| Phenolic Components (µg/g DM Catechin Equivalent) | Bran | Semolina | Grain | |||
|---|---|---|---|---|---|---|
| ‘Purple’ | ‘Red’ | ‘Purple’ | ‘Red’ | ‘Purple’ | ‘Red’ | |
| Anthocyanins | 72.9 ± 1.1 a | 36.3 ± 0.4 b | 12.5 ± 0.8 b | 16.8 ± 0.1 a | 116.6 ± 1.9 a | 39.2 ± 0.8 b |
| Proanthocyanidins | 1530 ± 133 a | 1031 ± 33 b | 244 ± 22 b | 466 ± 111 a | 3437 ± 377 a | 1807 ± 166 b |
| Anthocyanins (µg/kg DM) | Bran | Semolina | Grain | |||
|---|---|---|---|---|---|---|
| ‘Purple’ | ‘Red’ | ‘Purple’ | ‘Red’ | ‘Purple’ | ‘Red’ | |
| Delphinidin 3-glucoside | - | - | - | - | - | 1.20 ± 0.10 |
| Delphinidin 3-rutinoside | - | - | - | - | - | 2.66 ± 0.14 |
| Cyanidin 3-glucoside | 5.09 ± 0.11 a | 1.15 ± 0.05 b | 2.05 ± 0.15 | - | 7.32 ± 0.48 a | 2.39 ± 0.03 b |
| Petunidin 3-glucoside | 1.53 ± 0.11 a | 0.57 ± 0.07 b | - | - | 1.55 ± 0.05 a | 1.29 ± 0.01 b |
| Peonidin 3-glucoside | 2.05 ± 0.05 a | 0.86 ± 0.06 b | 0.89 ± 0.05 | - | 3.35 ± 0.15 a | 1.68 ± 0.04 b |
| Malvidin 3-glucoside | - | - | - | - | - | - |
| Anthocyanidins (µg/kg DM) | Bran | Semolina | Grain | |||
|---|---|---|---|---|---|---|
| ‘Purple’ | ‘Red’ | ‘Purple’ | ‘Red’ | ‘Purple’ | ‘Red’ | |
| Delphinidin | 1.75 ± 0.05 a | 0.91 ± 0.01 b | 1.15 ± 0.05 | - | 3.15 ± 0.45 a | 0.87 ± 0.03 b |
| Cyanidin | 1.87 ± 0.03 | 1.60 ± 0.20 | 1.27 ± 0.07 a | 0.56 ± 0.14 b | 5.45 ± 0.35 a | 1.47 ± 0.03 b |
| Petunidin | - | - | - | - | 1.68 ± 0.08 a | 0.45 ± 0.05 b |
| Peonidin | - | - | - | - | 1.35 ± 0.05 | - |
| Malvidin | - | - | - | - | 1.15 ± 0.75 | - |
| Total anthocyanins (Anthocyanins + Proanthocyanidins) | 12.29 ± 0.15 a | 5.09 ± 0.37 b | 5.36 ± 0.32 a | 0.56 ± 0.14 b | 25.00 ± 0.40 a | 12.01 ± 0.09 b |
| Assay | Bran | Semolina | Grain | |||
|---|---|---|---|---|---|---|
| ‘Purple’ | ‘Red’ | ‘Purple’ | ‘Red’ | ‘Purple’ | ‘Red’ | |
| DPPH | 9.95 ± 0.30 a | 8.47 ± 0.44 b | 8.14 ± 0.04 | 8.03 ± 0.01 | 11.07 ± 0.59 a | 9.85 ± 0.01 b |
| ABTS | 1.75 ± 0.30 b | 2.70 ± 0.12 a | 1.67 ± 0.24 | 1.68 ± 0.10 | 5.29 ± 0.25 a | 4.33 ± 0.40 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ladhari, A.; Corrado, G.; Rouphael, Y.; Carella, F.; Nappo, G.R.; Di Marino, C.; De Marco, A.; Palatucci, D. Chemical, Functional, and Technological Features of Grains, Brans, and Semolina from Purple and Red Durum Wheat Landraces. Foods 2022, 11, 1545. https://doi.org/10.3390/foods11111545
Ladhari A, Corrado G, Rouphael Y, Carella F, Nappo GR, Di Marino C, De Marco A, Palatucci D. Chemical, Functional, and Technological Features of Grains, Brans, and Semolina from Purple and Red Durum Wheat Landraces. Foods. 2022; 11(11):1545. https://doi.org/10.3390/foods11111545
Chicago/Turabian StyleLadhari, Afef, Giandomenico Corrado, Youssef Rouphael, Francesca Carella, Giuseppina Rita Nappo, Cinzia Di Marino, Anna De Marco, and Domenico Palatucci. 2022. "Chemical, Functional, and Technological Features of Grains, Brans, and Semolina from Purple and Red Durum Wheat Landraces" Foods 11, no. 11: 1545. https://doi.org/10.3390/foods11111545







