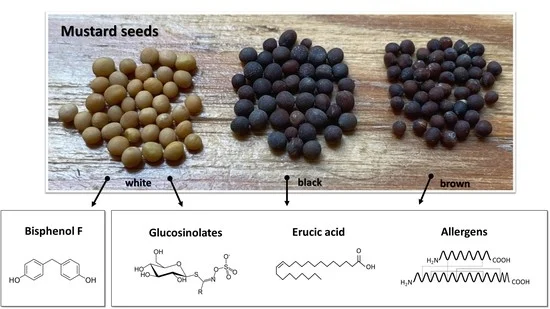
Biologically Active Compounds in Mustard Seeds: A Toxicological Perspective
Abstract
1. Introduction
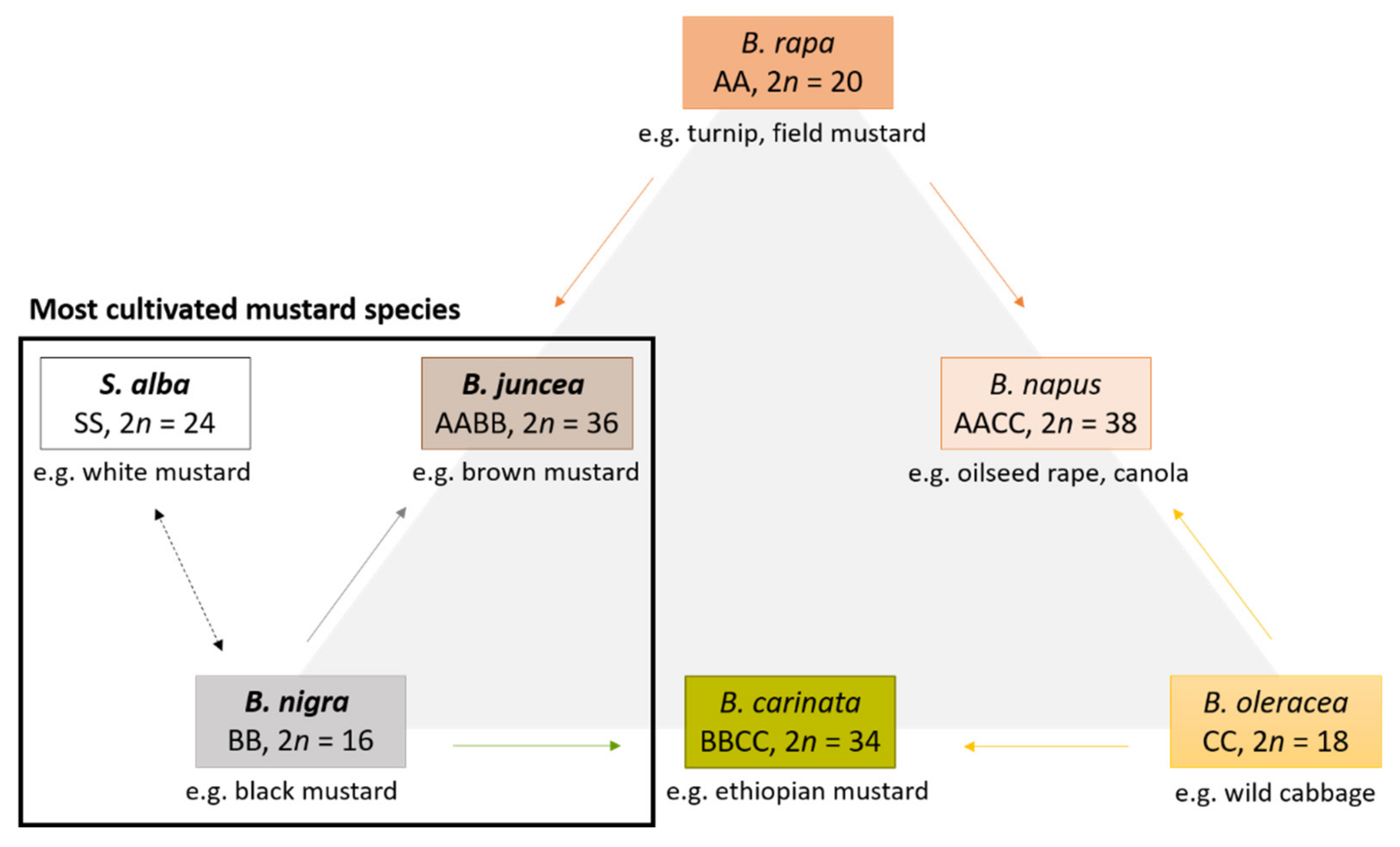
2. Biologically Active Compounds in Mustard Seeds
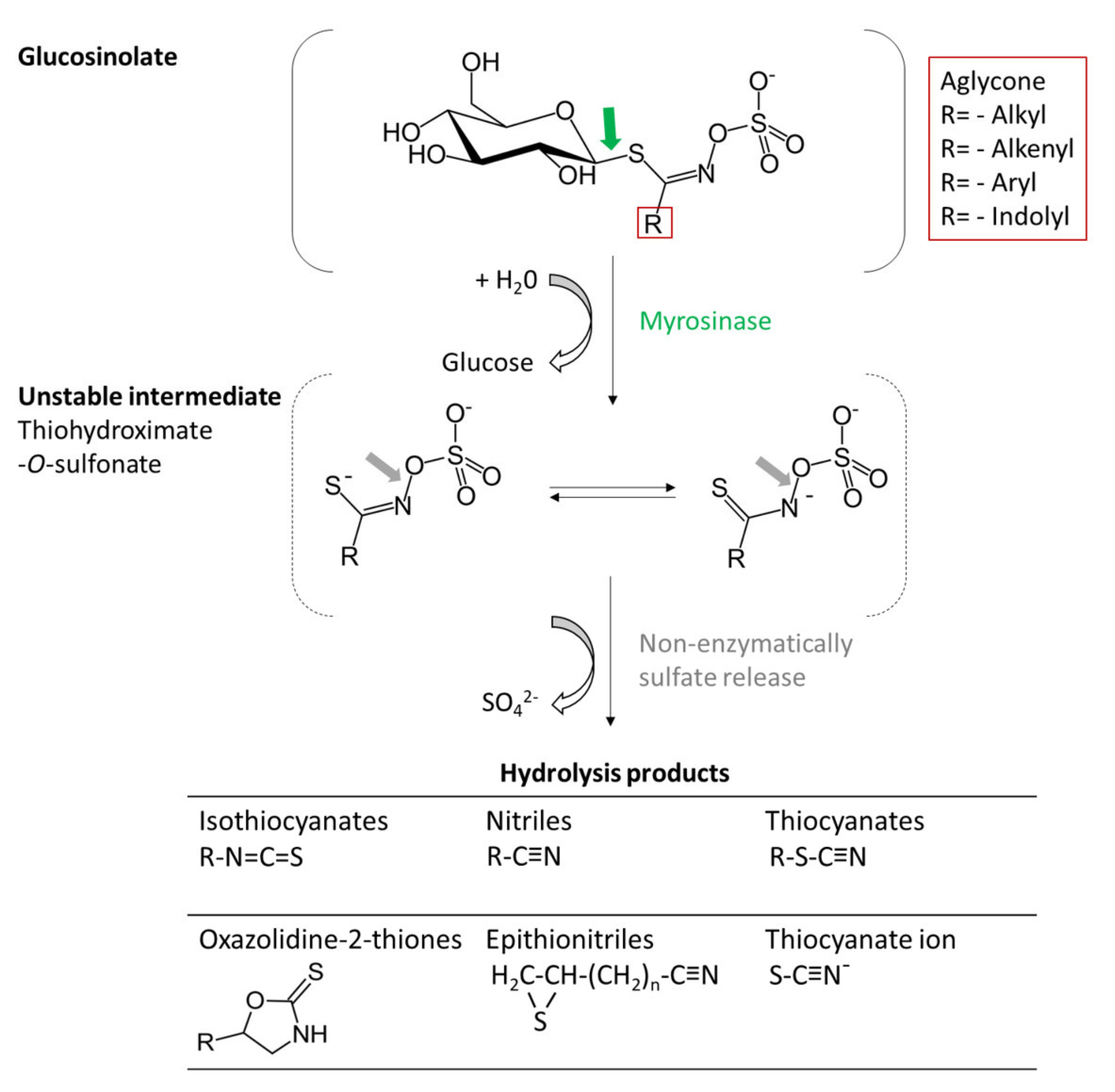
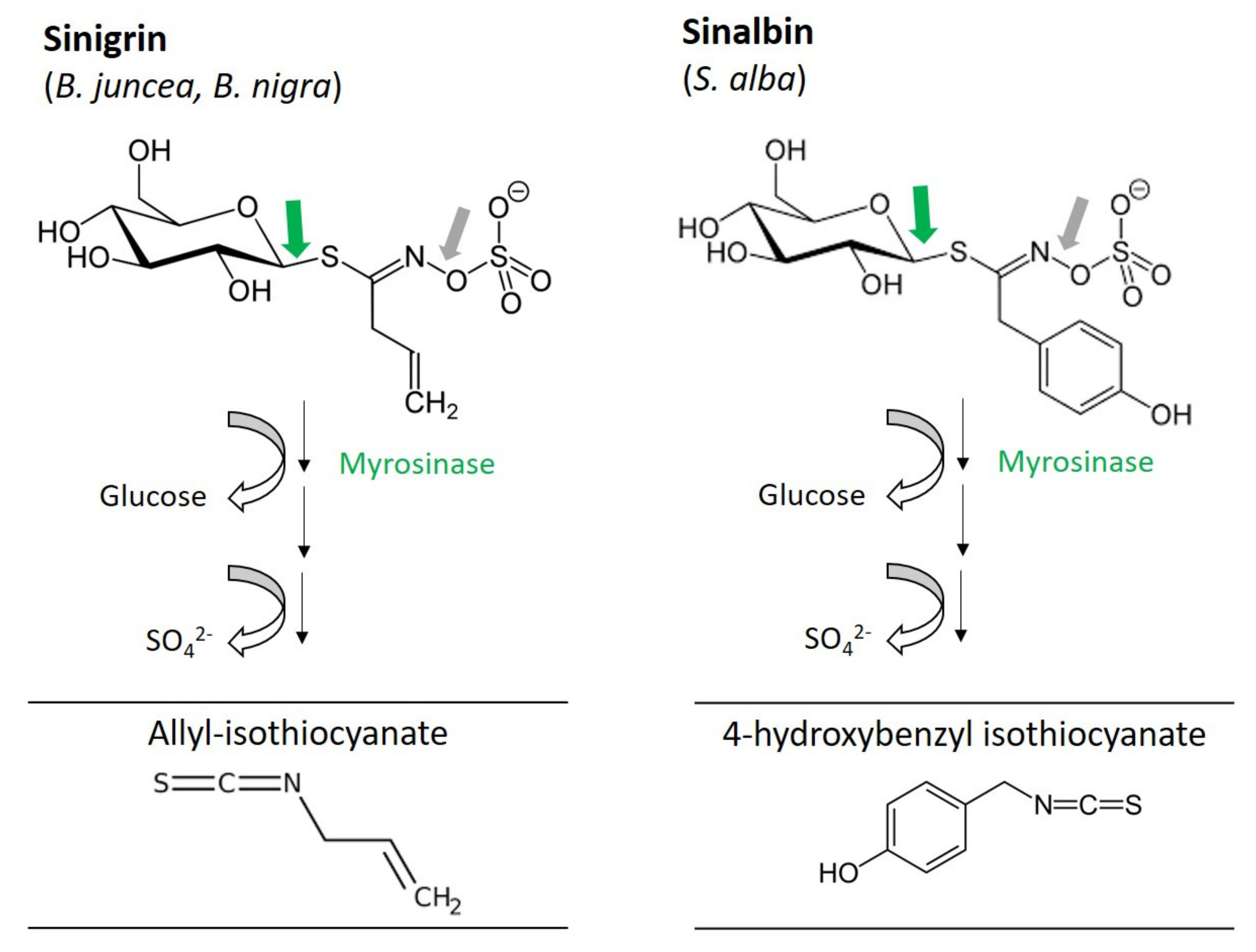
2.1. Glucosinolates
2.1.1. Toxicological and Antinutritional Effects
2.1.2. Goitrogenic Effects
2.1.3. Genotoxic and Carcinogenic Effects
2.1.4. Evaluation of Toxicological Effects
2.2. Bisphenol F
Evaluation of Possible Health Risks
2.3. Erucic Acid
2.3.1. Toxicological Effects
2.3.2. Evaluation of Toxicological Effects
2.4. Allergens
2.4.1. Clinical Indication
2.4.2. Evaluation of the Allergic Potential
3. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dixon, G. Origins and diversity of Brassica and its relatives. In Vegetable Brassicas and Related Crucifers; CABI: Wallingford, UK, 2006. [Google Scholar]
- Warwick, S.I.; Francis, A.; Al-Shehbaz, I.A. Brassicaceae: Species checklist and database on CD-Rom. Plant Syst. Evol. 2006, 259, 249–258. [Google Scholar] [CrossRef]
- Cheng, F.; Wu, J.; Wang, X. Genome triplication drove the diversification of Brassica plants. Hortic. Res. 2014, 1, 14024. [Google Scholar] [CrossRef]
- Warwick, S.I.; Black, L.D. Molecular systematics of Brassica and allied genera (Subtribe Brassicinae, Brassiceae)—Chloroplast genome and cytodeme congruence. Theor. Appl. Genet. 1991, 82, 81–92. [Google Scholar] [CrossRef]
- Xue, J.-Y.; Wang, Y.; Chen, M.; Dong, S.; Shao, Z.-Q.; Liu, Y. Maternal Inheritance of U’s Triangle and Evolutionary Process of Brassica Mitochondrial Genomes. Front. Plant Sci. 2020, 11, 805. [Google Scholar] [CrossRef]
- Vaughan, J.G. A Multidisciplinary Study of the Taxonomy and Origin of Brassica Crops. BioScience 1977, 27, 35–40. [Google Scholar] [CrossRef]
- Thomas, J.; Kuruvilla, K.M.; Hrideek, T.K. 21—Mustard. In Handbook of Herbs and Spices, 2nd ed.; Peter, K.V., Ed.; Woodhead Publishing: Cambridge, UK, 2012; pp. 388–398. [Google Scholar] [CrossRef]
- EC (European Commission). Common Catalogue of Varieties of Agricultural Plant Species—Version of 1 December 2020; European Commission. 2020. Available online: https://ec.europa.eu/food/plants/plant-reproductive-material/plant-variety-catalogues-databases-information-systems.de/ (accessed on 26 July 2021).
- Divakaran, M.; Babu, K.N. Mustard. In Encyclopedia of Food and Health; Caballero, B., Finglas, P.M., Toldrá, F., Eds.; Academic Press: Oxford, UK, 2016; pp. 9–19. [Google Scholar] [CrossRef]
- Rahman, M.; Khatun, A.; Liu, L.; Barkla, B.J. Brassicaceae Mustards: Traditional and Agronomic Uses in Australia and New Zealand. Molecules 2018, 23, 231. [Google Scholar] [CrossRef]
- Rakow, G. Species Origin and Economic Importance of Brassica. In Brassica; Pua, E.C., Douglas, C.J., Eds.; Springer: Berlin/Heidelberg, Germany, 2004; Volume 54. [Google Scholar]
- McVetty, P.B.E.; Duncan, R.W. Canola, Rapeseed, and Mustard: For Biofuels and Bioproducts. In Industrial Crops: Handbook of Plant Breeding 9; Cruz, V.M.V., Dierig, D.A., Eds.; Springer: Berlin/Heidelberg, Germany, 2015; pp. 133–156. [Google Scholar] [CrossRef]
- Government of Canada. Guidance Document Repository (GDR). The Biology of Brassica juncea (Canola/Mustard). Biology Document BIO2007-01: A Companion Document to the Directive 94-08 (Dir94-08), Assessment Criteria for Determining Environmental Safety of Plants with Novel Traits . 2007. Available online: https://inspection.canada.ca/plant-varieties/plants-with-novel-traits/applicants/directive-94-08/biology-documents/brassica-juncea/eng/1330727837568/1330727899677 (accessed on 26 July 2021).
- Tridge (Global Sourcing Hub of Food & Agriculture). Available online: https://www.tridge.com/intelligences/mustard-seed/ (accessed on 26 July 2021).
- FAOSTAT (Food and Agriculture Organization of the United Nations). Available online: http://www.fao.org/faostat/en/#search/mustard%20seed (accessed on 26 July 2021).
- Statista (Statistisches Bundesamt). ID 620, Pro-Kopf-Verbrauch von Senf in Deutschland in den Jahren 1983 bis 2019 (in Gramm). Available online: https://de.statista.com/statistik/daten/studie/620/umfrage/pro-kopf-verzehr-von-senf-in-deutschland/ (accessed on 26 July 2021).
- Santini, A.; Cicero, N. Development of Food Chemistry, Natural Products, and Nutrition Research: Targeting New Frontiers. Foods 2020, 9, 482. [Google Scholar] [CrossRef] [PubMed]
- Durazzo, A.; D’Addezio, L.; Camilli, E.; Piccinelli, R.; Turrini, A.; Marletta, L.; Marconi, S.; Lucarini, M.; Lisciani, S.; Gabrielli, P.; et al. From Plant Compounds to Botanicals and Back: A Current Snapshot. Molecules 2018, 23, 1844. [Google Scholar] [CrossRef] [PubMed]
- ReasearchAndMarkets.com. Global Mustard Market—Forecasts from 2020 to 2025; Knowledge Sourcing Intelligence LLP: Noida, India, 2020. [Google Scholar]
- Kayaçetin, F. Botanical characteristics, potential uses, and cultivation possibilities of mustards in turkey: A review. Turk. J. Bot. 2020, 44, 101–127. [Google Scholar] [CrossRef]
- Robiquet, P.J.; Bussy, A. Notice sur l’huile volatile de moutarde. J. Pharm. Sci. 1840, 26, 116–120. [Google Scholar]
- Fahey, J.W.; Zalcmann, A.T.; Talalay, P. The chemical diversity and distribution of glucosinolates and isothiocyanates among plants. Phytochemistry 2001, 56, 5–51. [Google Scholar] [CrossRef]
- Clarke, D.B. Glucosinolates, structures and analysis in food. Anal. Methods 2010, 2, 310–325. [Google Scholar] [CrossRef]
- Agerbirk, N.; Olsen, C.E. Glucosinolate structures in evolution. Phytochemistry 2012, 77, 16–45. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, D.W.; Birch, A.N.E.; Hillman, J.R. Antinutritional compounds in the Brassicaceae: Analysis, biosynthesis, chemistry and dietary effects. J. Hortic. Sci. Biotechnol. 1998, 73, 1–18. [Google Scholar] [CrossRef]
- Mithen, R.F. Glucosinolates and their degradation products. Adv. Bot. Res. 2001, 35, 213–232. [Google Scholar]
- Sikorska-Zimny, K.; Beneduce, L. The glucosinolates and their bioactive derivatives in Brassica: A review on classification, biosynthesis and content in plant tissues, fate during and after processing, effect on the human organism and interaction with the gut microbiota. Crit. Rev. Food Sci. Nutr. 2021, 61, 2544–2571. [Google Scholar] [CrossRef]
- Bhat, R.; Vyas, D. Myrosinase: Insights on structural, catalytic, regulatory, and environmental interactions. Crit. Rev. Biotechnol. 2019, 39, 508–523. [Google Scholar] [CrossRef]
- Nguyen, V.P.T.; Stewart, J.; Lopez, M.; Ioannou, I.; Allais, F. Glucosinolates: Natural Occurrence, Biosynthesis, Accessibility, Isolation, Structures, and Biological Activities. Molecules 2020, 25, 4537. [Google Scholar] [CrossRef]
- Shapiro, T.A.; Fahey, J.W.; Wade, K.L.; Stephenson, K.K.; Talalay, P. Human metabolism and excretion of cancer chemoprotective glucosinolates and isothiocyanates of cruciferous vegetables. Cancer Epidemiol. Biomark. Prev. 1998, 7, 1091–1100. [Google Scholar]
- Bones, A.M.; Rossiter, J.T. The myrosinase-glucosinolate system, its organisation and biochemistry. Physiol. Plant. 1996, 97, 194–208. [Google Scholar] [CrossRef]
- Halkier, B.A.; Gershenzon, J. Biology and Biochemistry of Glucosinolates. Annu. Rev. Plant Biol. 2006, 57, 303–333. [Google Scholar] [CrossRef]
- Bones, A.M.; Rossiter, J.T. The enzymic and chemically induced decomposition of glucosinolates. Phytochemistry 2006, 67, 1053–1067. [Google Scholar] [CrossRef] [PubMed]
- Grubb, C.D.; Abel, S. Glucosinolate metabolism and its control. Trends Plant Sci. 2006, 11, 89–100. [Google Scholar] [CrossRef]
- Agrawal, S.; Yallatikar, T.; Gurjar, P. Brassica Nigra: Ethopharmacological Review of a Routinely Used Condiment. Curr. Drug Discov. Technol. 2019, 16, 40–47. [Google Scholar] [CrossRef]
- Kuchernig, J.C.; Burow, M.; Wittstock, U. Evolution of specifier proteins in glucosinolate-containing plants. BMC Evol. Biol. 2012, 12, 127. [Google Scholar] [CrossRef]
- Kissen, R.; Bones, A.M. Nitrile-specifier proteins involved in glucosinolate hydrolysis in Arabidopsis thaliana. J. Biol. Chem. 2009, 284, 12057–12070. [Google Scholar] [CrossRef]
- Halkier, B.A.; Du, L. The biosynthesis of glucosinolates. Trends Plant Sci. 1997, 2, 425–431. [Google Scholar] [CrossRef]
- Rangkadilok, N.; Nicolas, M.; Bennett, R.; Premier, R.; Eagling, D.; Taylor, P. Developmental changes of sinigrin and glucoraphanin in three Brassica species (Brassica nigra, Brassica juncea and Brassica oleracea var. italica). Sci. Hortic. 2002, 96, 11–26. [Google Scholar] [CrossRef]
- Daxenbichler, M.E.; Spencer, G.F.; Carlson, D.G.; Rose, G.B.; Brinker, A.M.; Powell, R.G. Glucosinolate composition of seeds from 297 species of wild plants. Phytochemistry 1991, 30, 2623–2638. [Google Scholar] [CrossRef]
- Popova, I.E.; Morra, M.J. Simultaneous Quantification of Sinigrin, Sinalbin, and Anionic Glucosinolate Hydrolysis Products in Brassica juncea and Sinapis alba Seed Extracts Using Ion Chromatography. J. Agric. Food Chem. 2014, 62, 10687–10693. [Google Scholar] [CrossRef]
- Velíšek, J.; Mikulcová, R.; Míková, K.; Woldie, K.B.; Link, J.; Davídek, J. Chemometric investigation of mustard seed. Lebensm. Wiss. Und-Technol. Food Sci. Technol. 1995, 28, 620–624. [Google Scholar] [CrossRef]
- Velasco, L.; Becker, H.C. Variability for seed glucosinolates in a germplasm collection of the genus Brassica. Genet. Resour. Crop Evol. 2000, 47, 231–238. [Google Scholar] [CrossRef]
- Gupta, S.; Sangha, M.K.; Kaur, G.; Atwal, A.K.; Banga, S.; Banga, S.S. Variability for Leaf and Seed Glucosinolate Contents and Profiles in a Germplasm Collection of the Brassica juncea. Biochem. Anal. Biochem. 2012, 1. [Google Scholar] [CrossRef]
- Merah, O. Genetic Variability in Glucosinolates in Seed of Brassica juncea: Interest in Mustard Condiment. J. Chem. 2015, 2015, 606142. [Google Scholar] [CrossRef]
- Saikia, S.; Kumar, G.; Jammu, J.; Kashmir; Gyanendra, C.; Rai, K.; Rai, G.; Salgotra, R.; Rai, S.K.; Singh, M.; et al. Erucic acid and glucosinolate variability in Brassica juncea L. Int. J. Chem. Stud. 2018, 6, 1223–1226. [Google Scholar]
- McGregor, D.L. Thiocyanate ion, a hydrolysis product of glucosinolates from rape and mustard seed. Can. J. Plant Sci. 1978, 58, 795–800. [Google Scholar] [CrossRef]
- Ettlinger, M.G.; Lundeen, A.J. The structures of sinigrin and sinalbin; an enzymatic rearrangement. J. Am. Chem. Soc. 1956, 78, 4172–4173. [Google Scholar] [CrossRef]
- Ettlinger, M.G.; Lundeen, A.J. First synthesis of a mustard oil glucoside, the enzymatic lossen rearrangement. J. Am. Chem. Soc. 1957, 79, 1764–1765. [Google Scholar] [CrossRef]
- Nugrahedi, P.Y.; Verkerk, R.; Widianarko, B.; Dekker, M. A Mechanistic Perspective on Process-Induced Changes in Glucosinolate Content in Brassica Vegetables: A Review. Crit. Rev. Food Sci. Nutr. 2015, 55, 823–838. [Google Scholar] [CrossRef]
- Van Eylen, D.; Oey, I.; Hendrickx, M.; Van Loey, A. Behavior of mustard seed (Sinapis alba L.) myrosinase during temperature/pressure treatments: A case study on enzyme activity and stability. Eur. Food Res. Technol. 2008, 226, 545–553. [Google Scholar] [CrossRef]
- Srisangnam, C.; Salunkhe, K.; Dull, G. Quality of cabbage II. Physical, Chemical and biochemical modification in processing treatments to improve flavour of blanched cabbage (Brassica oleracea L.). J. Food Qual. 2007, 3, 233–250. [Google Scholar] [CrossRef]
- Prieto, M.A.; López, C.J.; Simal-Gandara, J. Chapter Six—Glucosinolates: Molecular structure, breakdown, genetic, bioavailability, properties and healthy and adverse effects. In Advances in Food and Nutrition Research; Ferreira, I.C.F.R., Barros, L., Eds.; Academic Press: Cambridge, MA, USA, 2019; Volume 90, pp. 305–350. [Google Scholar]
- de Vos, R.H.; Blijleven, W.G.H. The effect of processing conditions on glucosinolates in cruciferous vegetables. Z. Lebensm. Unters. Forsch. 1988, 187, 525–529. [Google Scholar] [CrossRef]
- Cools, K.; Terry, L.A. The effect of processing on the glucosinolate profile in mustard seed. Food Chem. 2018, 252, 343–348. [Google Scholar] [CrossRef]
- Kübler, K. Dissertation: Analyse von Glucosinolaten und Isothiocyanaten mittels Flüssigkeitschromatographie- bzw. Gaschromatographie-Massenspektrometrie; Justus-Liebig-University: Gießen, Germany, 2010; Available online: http://geb.uni-giessen.de/geb/volltexte/2010/7884/ (accessed on 9 June 2021).
- Eib, S.; Schneider, D.; Hensel, O.; Seuss-Baum, I. Relationship between mustard pungency and allyl-isothiocyanate content: A comparison of sensory and chemical evaluations. J. Food Sci. 2020, 85, 2728–2736. [Google Scholar] [CrossRef]
- EFSA (European Food Safety Authority). Safety of rapeseed powder from Brassica rapa L. and Brassica napus L. as a Novel food pursuant to Regulation (EU) 2015/2283. EFSA J. 2020, 18, e06197. [Google Scholar] [CrossRef]
- Fenwick, G.R.; Heaney, R.K. Glucosinolates and their Breakdown Products in Cruciferous Crops, Foods and Feedingstuffs. Food Chem. 1983, 11, 249–271. [Google Scholar] [CrossRef]
- van Doorn, H.E.; van der Kruk, G.C.; van Holst, G.-J.; Raaijmakers-Ruijs, N.C.M.E.; Postma, E.; Groeneweg, B.; Jongen, W.H.F. The glucosinolates sinigrin and progoitrin are important determinants for taste preference and bitterness of Brussels sprouts. J. Sci. Food Agric. 1998, 78, 30–38. [Google Scholar] [CrossRef]
- EP (European Parlament and the Council). Directive 2002/32/EC of the European Parlament and of the Council of 7 May 2002 on Undesirable Substances in Animal Feed. 2002. Available online: http://data.europa.eu/eli/dir/2002/32/2019-11-28/ (accessed on 9 June 2021).
- Mawson, R.; Heaney, R.K.; Zdunczyk, Z.; Kozlowska, H. Rapeseed meal-glucosinolates and their antinutritional effects Part 4. Goitrogenicity and internal organs abnormalities in animals. Die Nahr. 1994, 38, 178–191. [Google Scholar] [CrossRef]
- Tripathi, M.K.; Mishra, A.S. Glucosinolates in animal nutrition: A review. Anim. Feed Sci. Technol. 2007, 132, 1–27. [Google Scholar] [CrossRef]
- Langer, P.; Greer, M.A. Antithyroid Substances and Naturally Occurring Goitrogens; S. Karger AG: Basel, Switzerland, 1977. [Google Scholar]
- McDanell, R.; McLean, A.E.M.; Hanley, A.B.; Heaney, R.K.; Fenwick, G.R. Chemical and biological properties of indole glucosinolates (glucobrassicins): A review. Food Chem. Toxicol. 1988, 26, 59–70. [Google Scholar] [CrossRef]
- Kawakishi, S.; Namiki, M.; Watanabe, H.; Muramatsu, K. Studies on the Decomposition of Sinalbin. Agric. Biol. Chem. 1967, 31, 823–830. [Google Scholar] [CrossRef]
- Paunovic, D.; Solevic Knudsen, T.; Krivokapic, M.; Zlatković, B.; Antić, M. Sinalbin degradation products in mild yellow mustard paste. Hem. Ind. 2012, 66, 29–32. [Google Scholar] [CrossRef]
- Mitchell, M.L.; O’Rourke, M.E. Response of the thyroid gland to thiocyanate and thyrotropin. J. Clin. Endocrinol. Metab. 1960, 20, 47–56. [Google Scholar] [CrossRef]
- Felker, P.; Bunch, R.; Leung, A.M. Concentrations of thiocyanate and goitrin in human plasma, their precursor concentrations in brassica vegetables, and associated potential risk for hypothyroidism. Nutr. Rev. 2016, 74, 248–258. [Google Scholar] [CrossRef]
- Nishie, K.; Daxenbichler, M.E. Toxicology of glucosinolates, related compounds (nitriles, R-goitrin, isothiocyanates) and vitamin U found in cruciferae. Food Cosmet. Toxicol. 1980, 18, 159–172. [Google Scholar] [CrossRef]
- Daxenbichler, M.E.; Van Etten, C.H.; Spencer, G.F. Glucosinolates and derived products in cruciferous vegetables. Identification of organic nitriles from cabbage. J. Agric. Food Chem. 1977, 25, 121–124. [Google Scholar] [CrossRef]
- Cole, R.A. Isothiocyanates, nitriles and thiocyanates as products of autolysis of glucosinolates in Cruciferae. Phytochemistry 1976, 15, 759–762. [Google Scholar] [CrossRef]
- Choi, E.-J.; Zhang, P.; Kwon, H. Determination of goitrogenic metabolites in the serum of male wistar rat fed structurally different glucosinolates. Toxicol. Res. 2014, 30, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Dahlberg, P.A.; Bergmark, A.; Björck, L.; Bruce, A.; Hambraeus, L.; Claesson, O. Intake of thiocyanate by way of milk and its possible effect on thyroid function. Am. J. Clin. Nutr. 1984, 39, 416–420. [Google Scholar] [CrossRef] [PubMed]
- Langer, P.; Michajlovskij, N.; Sedlák, J.; Kueka, M. Studies on the antithyroid activity of naturally occurring L-5-vinyl-2-thiooxazolidone in man. Endokrinologie 1971, 57, 225–229. [Google Scholar] [PubMed]
- Kassie, F.; Knasmüller, S. Genotoxic effects of allyl isothiocyanate (AITC) and phenethyl isothiocyanate (PEITC). Chem. Biol. Interact. 2000, 127, 163–180. [Google Scholar] [CrossRef]
- Kassie, F.; Parzefall, W.; Musk, S.; Johnson, I.; Lamprecht, G.; Sontag, G.; Knasmüller, S. Genotoxic effects of crude juices from Brassica vegetables and juices and extracts from phytopharmaceutical preparations and spices of cruciferous plants origin in bacterial and mammalian cells. Chem. Biol. Interact. 1996, 102, 1–16. [Google Scholar] [CrossRef]
- Kassie, F.; Pool-Zobel, B.; Parzefall, W.; Knasmüller, S. Genotoxic effects of benzyl isothiocyanate, a natural chemopreventive agent. Mutagenesis 1999, 14, 595–604. [Google Scholar] [CrossRef]
- Baasanjav-Gerber, C.; Hollnagel, H.M.; Brauchmann, J.; Iori, R.; Glatt, H. Detection of genotoxicants in Brassicales using endogenous DNA as a surrogate target and adducts determined by 32P-postlabelling as an experimental end point. Mutagenesis 2010, 26, 407–413. [Google Scholar] [CrossRef]
- Glatt, H.; Baasanjav-Gerber, C.; Schumacher, F.; Monien, B.H.; Schreiner, M.; Frank, H.; Seidel, A.; Engst, W. 1-Methoxy-3-indolylmethyl glucosinolate; a potent genotoxicant in bacterial and mammalian cells: Mechanisms of bioactivation. Chem. Biol. Interact. 2011, 192, 81–86. [Google Scholar] [CrossRef]
- Musk, S.R.; Johnson, I.T. The clastogenic effects of isothiocyanates. Mutat. Res. 1993, 300, 111–117. [Google Scholar] [CrossRef]
- Musk, S.R.; Smith, T.K.; Johnson, I.T. On the cytotoxicity and genotoxicity of allyl and phenethyl isothiocyanates and their parent glucosinolates sinigrin and gluconasturtiin. Mutat. Res. 1995, 348, 19–23. [Google Scholar] [CrossRef]
- EFSA (European Food Safety Authority). Scientific Opinion on the safety of allyl isothiocyanate for the proposed uses as a food additive. EFSA J. 2010, 8, 1943. [Google Scholar] [CrossRef]
- NTP (National Toxicology Program). Carcinogenesis Bioassay of Allyl Isothiocyanate (CAS No. 57-06-7) in F344/N Rats and B6C3F1 Mice (Gavage Study). Natl. Toxicol. Prpgram Tech. Rep. Ser. 1982, 234, 1–142. [Google Scholar]
- Baasanjav-Gerber, C.; Monien, B.H.; Mewis, I.; Schreiner, M.; Barillari, J.; Iori, R.; Glatt, H. Identification of glucosinolate congeners able to form DNA adducts and to induce mutations upon activation by myrosinase. Mol. Nutr. Food Res. 2011, 55, 783–792. [Google Scholar] [CrossRef]
- Kołodziejski, D.; Piekarska, A.; Hanschen, F.S.; Pilipczuk, T.; Tietz, F.; Kusznierewicz, B.; Bartoszek, A. Relationship between conversion rate of glucosinolates to isothiocyanates/indoles and genotoxicity of individual parts of Brassica vegetables. Eur. Food Res. Technol. 2019, 245, 383–400. [Google Scholar] [CrossRef]
- Latté, K.P.; Appel, K.-E.; Lampen, A. Health benefits and possible risks of broccoli—An overview. Food Chem. Toxicol. 2011, 49, 3287–3309. [Google Scholar] [CrossRef]
- EFSA (European Food Safety Authority). GE 85: Consideration of miscellaneous nitrogen-containing substances evaluated by JECFA (65th meeting). EFSA J. 2008, 804, 1–30. [Google Scholar]
- JECFA (Joint FAO/WHO Expert Committee on Food Additives). Safety evaluation of certain food additives I prepared by the sixty-fifth meeting of the Joint FAO/WHO Expert Committee on Food Additives (JEFCA). WHO Food Addit. Ser. 2007, 56. Available online: https://apps.who.int/iris/handle/10665/43407 (accessed on 9 June 2021).
- Aboulfadl, M.; El-Badry, N.; Ammar, M. Nutritional and Chemical Evaluation for Two Different Varieties of Mustard Seeds. World Appl. Sci. J. 2011, 15, 1225–1233. [Google Scholar]
- Holst, B.; Williamson, G. A Critical Review of the Bioavailability of Glucosinolates and Related Compounds. Nat. Prod. Rep. 2004, 21, 425–447. [Google Scholar] [CrossRef]
- Mandrich, L.; Caputo, E. Brassicaceae-Derived Anticancer Agents: Towards a Green Approach to Beat Cancer. Nutrients 2020, 12, 868. [Google Scholar] [CrossRef]
- Verhoeven, D.T.; Goldbohm, R.A.; van Poppel, G.; Verhagen, H.; van den Brandt, P.A. Epidemiological studies on brassica vegetables and cancer risk. Cancer Epidemiol. Biomark. Prev. 1996, 5, 733–748. [Google Scholar]
- Lamy, E.; Crössmann, C.; Saeed, A.; Schreiner, P.R.; Kotke, M.; Mersch-Sundermann, V. Three structurally homologous isothiocyanates exert “Janus” characteristics in human HepG2 cells. Environ. Mol. Mutagenesis 2009, 50, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.; Danaher, L.A.; Brüschweiler, B.J.; Kass, G.E.N.; Merten, C. Naturally occurring bisphenol F in plants used in traditional medicine. Arch. Toxicol. 2019, 93, 1485–1490. [Google Scholar] [CrossRef]
- Cao, X.-L.; Kosarac, I.; Popovic, S.; Zhou, S.; Smith, D.; Dabeka, R. LC-MS/MS analysis of bisphenol S and five other bisphenols in total diet food samples. Food Addit. Contam. Part A 2019, 36, 1740–1747. [Google Scholar] [CrossRef]
- Zoller, O.; Brüschweiler, B.J.; Magnin, R.; Reinhard, H.; Rhyn, P.; Rupp, H.; Zeltner, S.; Felleisen, R. Natural occurrence of bisphenol F in mustard. Food Addit. Contam. Part A 2016, 33, 137–146. [Google Scholar] [CrossRef]
- BLV (Bundesamt für Lebensmittelsicherheit und Veterinärwesen, Schweiz) Bisphenol F in Senf—Fakten und Risikobewertung des BLV. Bericht. 2015. Available online: https://www.blv.admin.ch/dam/blv/de/dokumente/lebensmittel-und-ernaehrung/lebensmittelsicherheit/stoffe-im-fokus/bisphenol-f-senf-fakten-risikobewertung-blv.pdf.download.pdf/Bisphenol_Risikobewertung_BLV_DE.pdf/ (accessed on 9 June 2021).
- Reger, D.; Pavlovic, M.; Pietschmann-Keck, M.; Klinger, R. Bisphenol F in Senf: Aktueller Wissensstand und Nachweis mittels LC-MS/MS. J. Consum. Prot. Food Saf. 2017, 12, 131–137. [Google Scholar] [CrossRef]
- Liao, C.; Kannan, K. Concentrations and profiles of bisphenol A and other bisphenol analogues in foodstuffs from the United States and their implications for human exposure. J. Agric. Food Chem. 2013, 61, 4655–4662. [Google Scholar] [CrossRef]
- Karak, N. Vegetable Oil-Based Polymers Properties, Processing and Applications; Woodhead Publishing: Cambridge, UK, 2012. [Google Scholar] [CrossRef]
- De Kruif, C.A.; Marsman, J.W.; Venekamp, J.C.; Falke, H.E.; Noordhoek, J.; Blaauboer, B.J.; Wortelboer, H.M. Structure elucidation of acid reaction products of indole-3-carbinol: Detection in vivo and enzyme induction in vitro. Chem. Biol. Interact. 1991, 80, 303–315. [Google Scholar] [CrossRef]
- Cabaton, N.; Chagnon, M.C.; Lhuguenot, J.C.; Cravedi, J.P.; Zalko, D. Disposition and metabolic profiling of bisphenol F in pregnant and nonpregnant rats. J. Agric. Food Chem. 2006, 54, 10307–10314. [Google Scholar] [CrossRef] [PubMed]
- EFSA (European Food Safety Authority). Scientific Opinion on the risks to public health related to the presence of bisphenol A (BPA) in foodstuffs. EFSA J. 2015, 13, 3978. [Google Scholar] [CrossRef]
- EFSA (European Food Safety Authority). Scientific Opinion of the Panel on food contact materials, enzymes, flavourings and processing aids (CEF) on 24th list of substances for food contact materials. EFSA J. 2009, 1157–1163, 1–27. [Google Scholar]
- Rochester, J.R.; Bolden, A.L. Bisphenol S and F: A Systematic Review and Comparison of the Hormonal Activity of Bisphenol A Substitutes. Environ. Health Perspect. 2015, 123, 643–650. [Google Scholar] [CrossRef]
- Dietrich, D.R.; Hengstler, J.G. From bisphenol A to bisphenol F and a ban of mustard due to chronic low-dose exposures? Arch. Toxicol. 2016, 90, 489–491. [Google Scholar] [CrossRef] [PubMed]
- Gramec Skledar, D.; Peterlin Mašič, L. Bisphenol A and its analogs: Do their metabolites have endocrine activity? Environ. Toxicol. Pharmacol. 2016, 47, 182–199. [Google Scholar] [CrossRef] [PubMed]
- EFSA (European Food Safety Authority). In Proceedings of the Minutes of the 54th Meeting of the Working Group on BPA Evaluation, 12–13 July 2021. Available online: https://www.efsa.europa.eu/sites/default/files/wgs/food-ingredients-and-packaging/wg-BPA-re-evaluation-m.pdf (accessed on 26 July 2021).
- BfR (Bundesinstitut für Risikobewertung). Bisphenol F in Mustard: Adverse Effects on Health Due to the Measured BPF Concentrations Are Unlikely. Opinion No. 044/2015. Available online: www.bfr.bund.de.BfR (accessed on 17 June 2021).
- Liu, J.; Wattar, N.; Field, C.J.; Dinu, I.; Dewey, D.; Martin, J.W. Exposure and dietary sources of bisphenol A (BPA) and BPA-alternatives among mothers in the APrON cohort study. Environ. Int. 2018, 119, 319–326. [Google Scholar] [CrossRef]
- Lehmler, H.-J.; Liu, B.; Gadogbe, M.; Bao, W. Exposure to Bisphenol A, Bisphenol F, and Bisphenol S in U.S. Adults and Children: The National Health and Nutrition Examination Survey 2013–2014. Am. Chem. Soc. (AMS) Omega 2018, 3, 6523–6532. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, J.; Bhadauria, V.P.S.; Singh, V.; Singh, M.; Singh, K.; Kumar, A. Quality characteristics and their interrelationships in Indian rapeseed-mustard (Brassica sp.) varieties. Indian J. Agric. Sci. 2007, 77, 616–620. [Google Scholar]
- Wendlinger, C.; Hammann, S.; Vetter, W. Various concentrations of erucic acid in mustard oil and mustard. Food Chem. 2014, 153, 393–397. [Google Scholar] [CrossRef]
- Mortuza, M.; Dutta, P. Erucic acid content in some rapeseed/mustard cultivars developed in Bangladesh. J. Sci. Food Agric. 2005, 86, 135–139. [Google Scholar] [CrossRef]
- Mandal, S.; Yadav, S.; Singh, R.; Begum, G.; Suneja, P.; Singh, M. Correlation studies on oil content and fatty acid profile of some Cruciferous species. Genet. Resour. Crop Evol. 2002, 49, 551–556. [Google Scholar] [CrossRef]
- Vetter, W.; Darwisch, V.; Lehnert, K. Erucic acid in Brassicaceae and salmon—An evaluation of the new proposed limits of erucic acid in food. NFS J. 2020, 19, 9–15. [Google Scholar] [CrossRef]
- Mahmoud, K.; Tahoun, A.H.; Ghazy, I.; Amin, E.A. Oil and erucic acid. Contents of five Brassica species grown for four successive years in Egypt. In Proceedings of the 10th International Rapeseed Congress, Canberra, Australia, 26–29 September 1999. [Google Scholar]
- Chowdhury, K.; Banu, L.A.; Khan, S.; Latif, A. Studies on the Fatty Acid Composition of Edible Oil. Bangladesh J. Sci. Ind. Res. 2007, 42, 311–316. [Google Scholar] [CrossRef]
- Rahman, D.M.H.; Sarwar, M.; Rahman, M.; Raza, M.; Rouf, S.; Rahman, M. Determination of Erucic acid content in traditional and commercial mustard oils of Bangladesh by Gas- Liquid Chromatography. Adv. Biochem. 2014, 2, 9–13. [Google Scholar]
- Dorni, C.; Sharma, P.; Saikia, G.; Longvah, T. Fatty acid profile of edible oils and fats consumed in India. Food Chem. 2018, 238, 9–15. [Google Scholar] [CrossRef]
- Konuskan, D.B.; Arslan, M.; Oksuz, A. Physicochemical properties of cold pressed sunflower, peanut, rapeseed, mustard and olive oils grown in the Eastern Mediterranean region. Saudi J. Biol. Sci. 2019, 26, 340–344. [Google Scholar] [CrossRef] [PubMed]
- Kok, W.-M.; Mainal, A.; Chuah, C.-H.; Cheng, S.-F. Content of Erucic Acid in Edible Oils and Mustard by Quantitative 13C NMR. Eur. J. Lipid Sci. Technol. 2018, 120, 1700230. [Google Scholar] [CrossRef]
- Sawicka, B.; Kotiuk, E.; Kiełtyka-Dadasiewicz, A.; Krochmal-Marczak, B. Fatty Acids Composition of Mustard Oil from Two Cultivars and Physico-chemical Characteristics of the Seeds. J. Oleo Sci. 2020, 69, 207–217. [Google Scholar] [CrossRef] [PubMed]
- BLS (Bundeslebensmittelschlüssel). Version 3.02. Max-Rubner Institut (MRI) 2005–2021, Standort Karlsruhe. Available online: https://blsdb.de/ (accessed on 17 June 2021).
- Burton, W.; Salisbury, P.; Potts, D. The potential of canola quality Brassica juncea as an oilseed crop for Australia. In Proceedings of the 13th Australian Research Assembly on Brassicas, Tamworth, NSW, Australia, 8–12 September 2003. [Google Scholar]
- EFSA (European Food Safety Authority). Erucic acid in feed and food. EFSA J. 2016, 14, e04593. [Google Scholar] [CrossRef]
- Osmundsen, H.; Bremer, J. Comparative biochemistry of beta-oxidation. An investigation into the abilities of isolated heart mitochondria of various animal species to oxidize long-chain fatty acids, including the C22:1 monoenes. Biochem. J. 1978, 174, 379–386. [Google Scholar] [CrossRef]
- Buddecke, E.; Filipović, I.; Wortberg, B.; Seher, A. Wirkungsmechanismus langkettiger Monoenfettsäuren im Energiestoffwechsel des Herzens. Fette Seifen Anstrichm. 1976, 78, 196–200. [Google Scholar] [CrossRef]
- Chien, K.R.; Bellary, A.; Nicar, M.; Mukherjee, A.; Buja, L.M. Induction of a reversible cardiac lipidosis by a dietary long-chain fatty acid (erucic acid). Relationship to lipid accumulation in border zones of myocardial infarcts. Am. J. Pathol. 1983, 112, 68–77. [Google Scholar]
- Kramer, J.K.; Farnworth, E.R.; Johnston, K.M.; Wolynetz, M.S.; Modler, H.W.; Sauer, F.D. Myocardial changes in newborn piglets fed sow milk or milk replacer diets containing different levels of erucic acid. Lipids 1990, 25, 729–737. [Google Scholar] [CrossRef]
- Kramer, J.K.G.; Sauer, F.D.; Wolynetz, M.S.; Farnworth, E.R.; Johnston, K.M. Effects of dietary saturated fat on erucic acid induced myocardial lipidosis in rats. Lipids 1992, 27, 619–623. [Google Scholar] [CrossRef]
- Beare-Rogers, J.L. Docosenoic acids in dietary fats. Prog. Chem. Fats Other Lipids 1977, 15, 29–56. [Google Scholar] [CrossRef]
- Clouet, P.; Bézard, J. Comparative oxidation of erucic and oleic acids by mitochondria isolated from heart auricle of living man. C. R. L’académie Sci. 1979, 288, 1683–1686. [Google Scholar]
- Imamura, F.; Lemaitre, R.N.; King, I.B.; Song, X.; Steffen, L.M.; Folsom, A.R.; Siscovick, D.S.; Mozaffarian, D. Long-chain monounsaturated Fatty acids and incidence of congestive heart failure in 2 prospective cohorts. Circulation 2013, 127, 1512–1521. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, C.; Matthan, N.R.; Lichtenstein, A.H.; Gaziano, J.M.; Djoussé, L. Red blood cell MUFAs and risk of coronary artery disease in the Physicians’ Health Study. Am. J. Clin. Nutr. 2013, 98, 749–754. [Google Scholar] [CrossRef]
- Schulze, P.C. Myocardial lipid accumulation and lipotoxicity in heart failure. J Lipid Res 2009, 50, 2137–2138. [Google Scholar] [CrossRef]
- FSANZ (Australia New Zealand Food Standards Code). Maximum Levels of Contaminants and Natural Toxicants. 2016. Available online: https://www.legislation.gov.au/Details/F2016C00167 (accessed on 17 June 2021).
- FDA (U.S. Food and Drug Administration). Import Alert 26-04 “Detention without Physical Examination of Expressed Mustard Oil”. 2016. Available online: https://www.accessdata.fda.gov/cms_ia/ind._26.html (accessed on 17 June 2021).
- Rastogi, T.; Reddy, K.S.; Vaz, M.; Spiegelman, D.; Prabhakaran, D.; Willett, W.C.; Stampfer, M.J.; Ascherio, A. Diet and risk of ischemic heart disease in India. Am. J. Clin. Nutr. 2004, 79, 582–592. [Google Scholar] [CrossRef]
- Wanasundara, J.P. Proteins of Brassicaceae oilseeds and their potential as a plant protein source. Crit. Rev. Food Science Nutr. 2011, 51, 635–677. [Google Scholar] [CrossRef]
- Wanasundara, J.P.D.; Abeysekara, S.J.; McIntosh, T.C.; Falk, K.C. Solubility Differences of Major Storage Proteins of Brassicaceae Oilseeds. J. Am. Oil Chem. Soc. 2012, 89, 869–881. [Google Scholar] [CrossRef]
- Menéndez-Arias, L.; Moneo, I.; Domínguez, J.; Rodríguez, R. Primary structure of the major allergen of yellow mustard (Sinapis alba L.) seed, Sin a I. Eur. J. Biochem. 1988, 177, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Menéndez-Arias, L.; Domínguez, J.; Moneo, I.; Rodríguez, R. Epitope mapping of the major allergen from yellow mustard seeds, Sin a I. Mol. Immunol. 1990, 27, 143–150. [Google Scholar] [CrossRef]
- Gonzalez de la Peña, M.A.; Menéndez-Arias, L.; Monsalve, R.I.; Rodríguez, R. Isolation and Characterization of a Major Allergen from Oriental Mustard Seeds, Bra j I. Int. Arch. Allergy Immunol. 1991, 96, 263–270. [Google Scholar] [CrossRef]
- WHO/IUIS Allergen Nomenclature Sub-Committee. Available online: http://www.allergen.org/ (accessed on 26 July 2021).
- Moreno, F.J.; Clemente, A. 2S Albumin Storage Proteins: What Makes them Food Allergens? Open Biochem. J. 2008, 2, 16–28. [Google Scholar] [CrossRef]
- Morisset, M.; Moneret-Vautrin, D.-A.; Maadi, F.; Frémont, S.; Guénard, L.; Croizier, A.; Kanny, G. Prospective study of mustard allergy: First study with double-blind placebo-controlled food challenge trials (24 cases). Allergy 2003, 58, 295–299. [Google Scholar] [CrossRef] [PubMed]
- Sirvent, S.; Palomares, O.; Vereda, A.; Villalba, M.; Cuesta-Herranz, J.; Rodríguez, R. nsLTP and profilin are allergens in mustard seeds: Cloning, sequencing and recombinant production of Sin a 3 and Sin a 4. Clin. Exp. Allergy 2009, 39, 1929–1936. [Google Scholar] [CrossRef] [PubMed]
- L’Hocine, L.; Pitre, M.; Achouri, A. Detection and identification of allergens from Canadian mustard varieties of Sinapis alba and Brassica juncea. Biomolecules 2019, 9, 489. [Google Scholar] [CrossRef] [PubMed]
- Perera, S.P.; McIntosh, T.C.; Wanasundara, J.P.D. Structural Properties of Cruciferin and Napin of Brassica napus (Canola) Show Distinct Responses to Changes in pH and Temperature. Plants 2016, 5, 36. [Google Scholar] [CrossRef] [PubMed]
- EFSA (European Food Safety Authority). Opinion of the Scientific Panel on Dietetic products, nutrition and allergies [NDA] on a request from the Commission relating to the evaluation of allergenic foods for labelling purposes. EFSA J. 2004, 2, 32. [Google Scholar] [CrossRef]
- Maruyama, T.; Iizuka, H.; Tobisawa, Y.; Shiba, T.; Matsuda, T.; Kurohane, K.; Imai, Y. Influence of Local Treatments with Capsaicin or Allyl Isothiocyanate in the Sensitization Phase of a Fluorescein-Isothiocyanate-Induced Contact Sensitivity Model. Int. Arch. Allergy Immunol. 2007, 143, 144–154. [Google Scholar] [CrossRef] [PubMed]
- Pasricha, J.S.; Gupta, R.; Gupta, S.K. Contact Hwersensitaity to Mustard Khal and Mustard Oil. Indian J. Dermatol. Venereol. Leprol. 1985, 51, 108–110. [Google Scholar]
- EFSA (European Food Safety Authority). Scientific Opinion on the evaluation of allergenic foods and food ingredients for labelling purposes. EFSA J. 2014, 12, 3894. [Google Scholar]
- Rancé, F.; Dutau, G.; Abbal, M. Mustard allergy in children. Allergy 2000, 55, 496–500. [Google Scholar] [CrossRef]
- Rancé, F.; Abbal, M.; Dutau, G. Mustard allergy in children. Pediatr. Pulmonol. 2001, 23, 44–45. [Google Scholar] [CrossRef] [PubMed]
- Rancé, F. Mustard allergy as a new food allergy. Allergy 2003, 58, 287–288. [Google Scholar] [CrossRef]
- Sirvent, S.; Palomares, O.; Cuesta-Herranz, J.; Villalba, M.; Rodríguez, R. Analysis of the structural and immunological stability of 2S albumin, nonspecific lipid transfer protein, and profilin allergens from mustard seeds. J. Agric. Food Chem. 2012, 60, 6011–6018. [Google Scholar] [CrossRef]
- Vereda, A.; Sirvent, S.; Villalba, M.; Rodríguez, R.; Cuesta-Herranz, J.; Palomares, O. Improvement of mustard (Sinapis alba) allergy diagnosis and management by linking clinical features and component-resolved approaches. J. Allergy Clin. Immunol. 2011, 127, 1304–1307. [Google Scholar] [CrossRef]
- Figueroa, J.; Blanco, C.; Dumpiérrez, A.G.; Almeida, L.; Ortega, N.; Castillo, R.; Navarro, L.; Pérez, E.; Gallego, M.D.; Carrillo, T. Mustard allergy confirmed by double-blind placebo-controlled food challenges: Clinical features and cross-reactivity with mugwort pollen and plant-derived foods. Allergy 2005, 60, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Kulka, M.; Sheen, C.H.; Tancowny, B.P.; Grammer, L.C.; Schleimer, R.P. Neuropeptides activate human mast cell degranulation and chemokine production. Immunology 2008, 123, 398–410. [Google Scholar] [CrossRef]
- FARRP (Food Allergy Research and Resource Program). Food Allergens—International Regulatory Chart. Available online: https://farrp.unl.edu/IRChart/ (accessed on 26 July 2021).
- Monsalve, R.; Villalba, M.; Rodriguez, R. Allergy to mustard seeds: The importance of 2S albumins as food allergens. Internet Symp. Food Allerg. 2001, 3, 57–69. [Google Scholar]
- Sharma, A.; Verma, A.K.; Gupta, R.K.; Neelabh; Dwivedi, P.D. A Comprehensive Review on Mustard-Induced Allergy and Implications for Human Health. Clin. Rev. Allergy Immunol. 2019, 57, 39–54. [Google Scholar] [CrossRef]
- Pałgan, K.; Żbikowska-Gotz, M.; Bartuzi, Z. Dangerous anaphylactic reaction to mustard. Arch. Med. Sci. 2018, 14, 477–479. [Google Scholar] [CrossRef]
- Moneret-Vautrin, A. Epidemiology of food allergies and relative prevalence of trophallergens. Cah. Nutr. Diet. 2001, 36, 247–252. [Google Scholar]
- BfR (Bundesinstitut für Risikobewertung). ‘VITAL 3.0’: New and updated proposals for reference doses of food allergens. BfR Opin. 2020. Available online: https://www.bfr.bund.de/cm/349/vital-30-new-and-updated-proposals-for-reference-doses-of-food-allergens.pdf/ (accessed on 26 July 2021). [CrossRef]
| Mustard Species | Common Names | Glucosinolates Identified | Systematic Name |
|---|---|---|---|
| Trivial Name | (a-Glycone = R Side Chain) | ||
| S. alba | white, yellow mustard | Gluconapin a | 3-Butenyl |
| Progoitrin a | 2R-2-Hydroxy-3-butenyl | ||
| Glucobrassicanapin a | Pent-4-enyl | ||
| # (Gluco-)Sinalbin b | 4-Hydroxybenzyl | ||
| Glucotropaeolin b | Benzyl | ||
| Gluconasturtiin b | 2-Phenylethyl | ||
| Glucoerucin c | 4-Methylthiobutyl | ||
| Glucoibe(rve)rin c | 3-Methylthiopropyl | ||
| Glucoiberin c | 3-Methylsulphinylpropyl | ||
| 2-Methylpropyl Isobutyl d | |||
| Glucobrassicin e | 3-Indolylmethyl | ||
| Neoglucobrassicin e | N-Methoxy-3-indolylmethyl | ||
| B. nigra | wlack, shortpod mustard, moutarde noire | # (Gluco-)Sinigrin a | 2-Propenyl |
| B. juncea | brown, indian, asiatic, chinese, sarepta mustard | # (Gluco-)Sinigrin a | 2-Propenyl |
| # Gluconapin a | 3-Butenyl | ||
| Progoitrin a | 2R-2-Hydroxy-3-butenyl | ||
| Epiprogoitrin a | 2S-2-Hydroxy-3-butenyl | ||
| Glucosinalbin b | 4-Hydroxybenzyl |
| Mustard Species | Total Content of Glucosinolates | Content of Predominant Glucosinolates | Reference |
|---|---|---|---|
| S. alba | Sinalbin (seed): 250 µmol/g sinalbin | [23] | |
| Sinalbin (seed): 165 ± 3 µmol/g Sinigrin (seed): 155 ± 4 μmol/g | [41] | ||
| Seed: 45.4–61.9 g/kg FW | Sinalbin (seed): 16.6–46.2 g/kg FW Progoitrin (seed): 0.8–1.3 g/kg FW Glucoibervirin/Glucobrassicin/4-hydroxglucobrassicin (seed): <0.2 g/kg FW | [42] | |
| B. nigra | Seed: 207.2–229.0 µmol/g FW | Sinigrin (seed): 202–226.7 µmol/g FW | [43] |
| Seed: 26–62.4 g/kg FW | Sinigrin (seed): 24.5–61.2 g/kg FW Gluconapin/Progoitrin/Glucoibervirin (seed): <0.3 g/kg FW Sinalbin/4-hydroxglucobrassicin (seed): <0.9 g/kg FW | [42] | |
| B. juncea | Intact seed: 15.7–127.6 µmol/g DW Leaf: 4.3–129.9 µmol/g DW | Gluconapin: (leaf): 0.6–70.1 µmol/g DW (seed): 2.3–90.8 µmol/g Sinalbin (leaf): 0.1–64.9 µmol/g DW (seed): 0.2–35.2 µmol/g Epiprogoitrin (leaf): 0.7–16.4 µmol/g DW | [44] |
| Seed: 94.4–169.2 µmol/g FW | Sinigrin (seed): 43.79–145.5 µmol/g FW Gluconapin (seed): 0–110.58 µmol/g FW | [43] | |
| Seed: 68–153 μmol/g FW | Sinigrin (seed): 0–134.2 μmol/g FW Gluconapin: 68–153 µmol/g FW | [45] | |
| Defatted seed meal: 58.83–132.26 μmol/g | [46] | ||
| Oil free seed meal: 123.8–152.1 µmol/g | [47] | ||
| Sinigrin (seed): 155 ± 4 µmol/g | [41] | ||
| Seed: 18.1–61.8 g/kg FW | Sinigrin (seed): 16.6–46.2 g/kg FW Gluconapin/Progoitrin/Glucoibervirin/Glucoerucin/ Sinalbin/Gluconasturiin (seed): <1 g/kg FW 4-hydroxyglucobrassicin (seed): 0.6–1.6 g/kg FW | [42] |
| Food Items | Bisphenol F Content (in mg/kg) | Reference |
|---|---|---|
| Condiments | 2.4 | [96] |
| Commercial table mustard (n = 61) | mean 1.84 (max 8.4) | [97,98] |
| thereof mild mustards (n = 19) | mean 3.2 | |
| Commercial table mustard | [99] | |
| Medium hot mustard (n = 39) | <5 (n = 37); >9 (n = 2) | |
| Sweet mustard (n = 7) | <3 | |
| Hot mustard (n = 10) | <3 | |
| Quince mustard (n = 1) | <1 | |
| Self-made mustard products | ||
| Mustard flour | n.d. | |
| Table mustard from B. nigra | n.d. | |
| Table mustard from B. juncea | n.d. | |
| Table mustard from S. alba | 0.18–0.94 | |
| Mustard dressing | 1.13 | [100] |
| Type | Origin | Amount of Erucic Acid (of Total FA) | Reference | |
|---|---|---|---|---|
| Mustard seed | B. juncea mustard (54 varieties) | India | 35.7–51.4% | [113] |
| B. juncea varieties | Bangladesh | 43.7–48.6% | [115] | |
| B. juncea (51 collections) | India | 22.5–53.4% | [116] | |
| B. nigra (3 collections) | 35.9–40.1% | |||
| S. alba (4 collections) | 39.1–47.2% | |||
| B. nigra (n = 5) | Germany | 20–40% | [117] | |
| B. juncea (n = 5) | ||||
| S. alba (n = 5) | ||||
| B. juncea (n = 12) | India | 39.4–51.8% | [46] | |
| B. juncea (n = 3) | 0.9–1.6% | |||
| B. nigra (n = 5) | Egypt | 33.3–45/32–42.9 %a | [118] | |
| B. juncea (n = 5) | 21.9–40/24.2–42.7 %a | |||
| Mustard seed oil | Mustard oil (n = 3) | Australia | 43.8–45.8% | [114] |
| Mustard oil (n = 6) | Germany | 0.3–50.8% | ||
| S. alba mustard oil | Egypt | 37.90% | [90] | |
| B. juncea mustard oil | 23.90% | |||
| Mustard oil | India | [119] | ||
| Variety 1 (n = 3) | 0.9–4.7% | |||
| Variety 2 (n = 2) | 14.7–34.1% | |||
| Mustard oil | Bangladesh | 41.80% | [120] | |
| Mustard oil (Erucic free) | 20.10% | |||
| Mustard oil (n = 59) | India | 48.5–54.02% | [121] | |
| Mustard oil | Turkey | 11.40% | [122] | |
| Mustard oil | Malaysia | 18.50% | [123] | |
| S. alba mustard oil | Poland | [124] | ||
| Cultivar 1 | 22.20% | |||
| Cultivar 2 (Erucic free) | 3.80% | |||
| Other mustard products | Mustard samples (n = 15) | Germany | 14–33% | [114] |
| Mustard sauces (n = 5) | <5% | |||
| Mustard condiment | Germany | 1.6 g/100 g condiment b | [125] | |
| Mustard seeds | 11.5 g/100 g seeds c | |||
| Mustard sauces | Malaysia | 33.10% | [123] | |
| Mustard sauces | 32.20% | |||
| Mustard powder | 25.50% | |||
| Mustard Species | List of Allergens | Type | Source a |
|---|---|---|---|
| S. alba | Sin a 1 | 2S albumin | http://www.allergen.org |
| Sin a 2 | 11S globulin (legumin-like) seed storage protein | http://www.allergen.org | |
| Sin a 3 | Non-specific lipid transfer protein type 1 (ns-LTP) | http://www.allergen.org | |
| Sin a 4 | Profilin | http://www.allergen.org | |
| B. nigra | Bra j 1 | 2S albumin seed storage protein | http://www.uniprot.org * |
| B. juncea | Bra j 1 | 2S albumin seed storage protein | http://www.allergen.org |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lietzow, J. Biologically Active Compounds in Mustard Seeds: A Toxicological Perspective. Foods 2021, 10, 2089. https://doi.org/10.3390/foods10092089
Lietzow J. Biologically Active Compounds in Mustard Seeds: A Toxicological Perspective. Foods. 2021; 10(9):2089. https://doi.org/10.3390/foods10092089
Chicago/Turabian StyleLietzow, Julika. 2021. "Biologically Active Compounds in Mustard Seeds: A Toxicological Perspective" Foods 10, no. 9: 2089. https://doi.org/10.3390/foods10092089
APA StyleLietzow, J. (2021). Biologically Active Compounds in Mustard Seeds: A Toxicological Perspective. Foods, 10(9), 2089. https://doi.org/10.3390/foods10092089