Novel Yeasts Producing High Levels of Conjugated Linoleic Acid and Organic Acids in Fermented Doughs
Abstract
:1. Introduction
2. Materials and Methods
2.1. Microorganisms
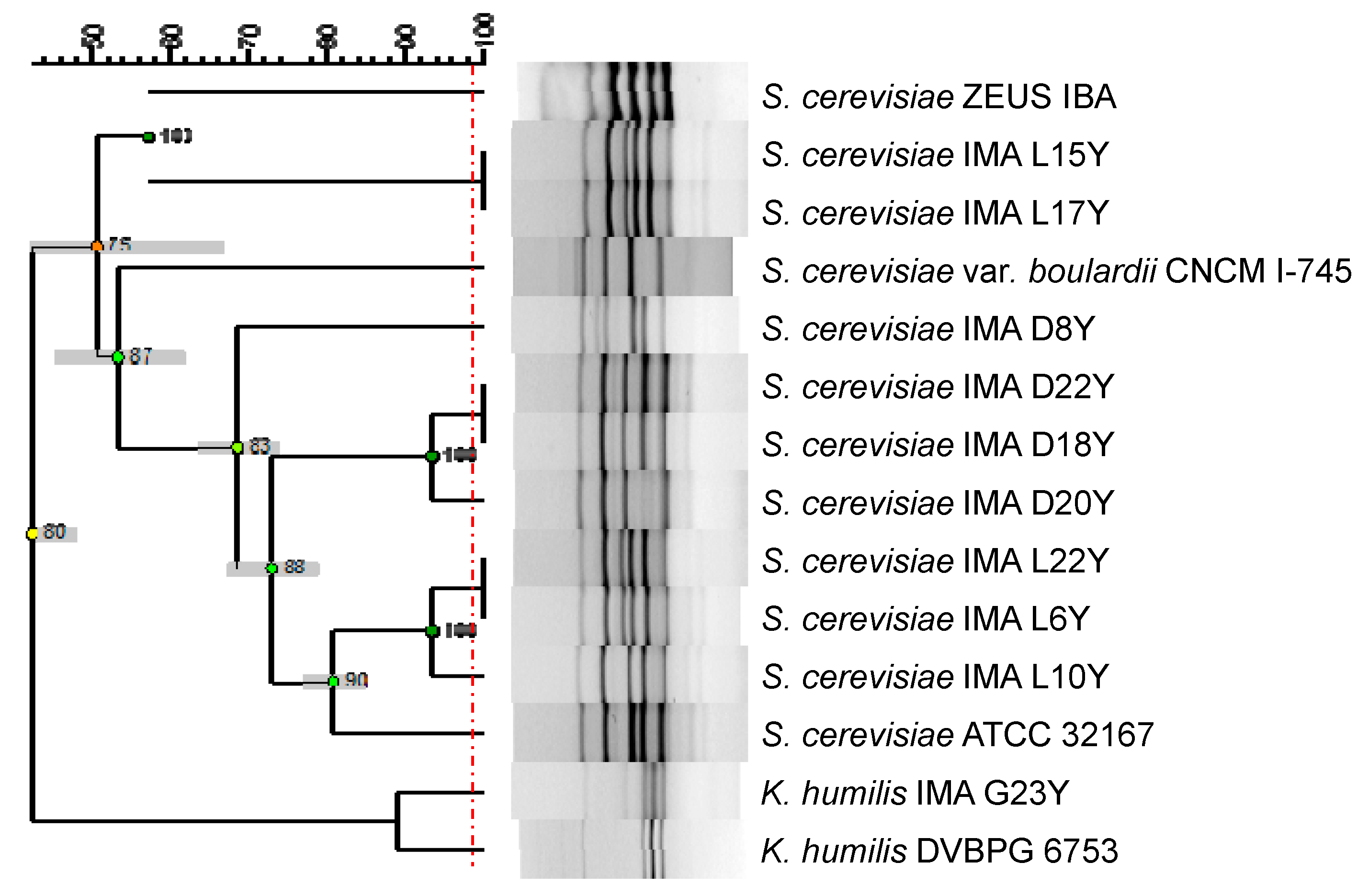
2.2. Phylogenetic Relationship among Yeast Isolates
2.3. In Vitro Production of Organic Acids
2.4. Dough Preparation
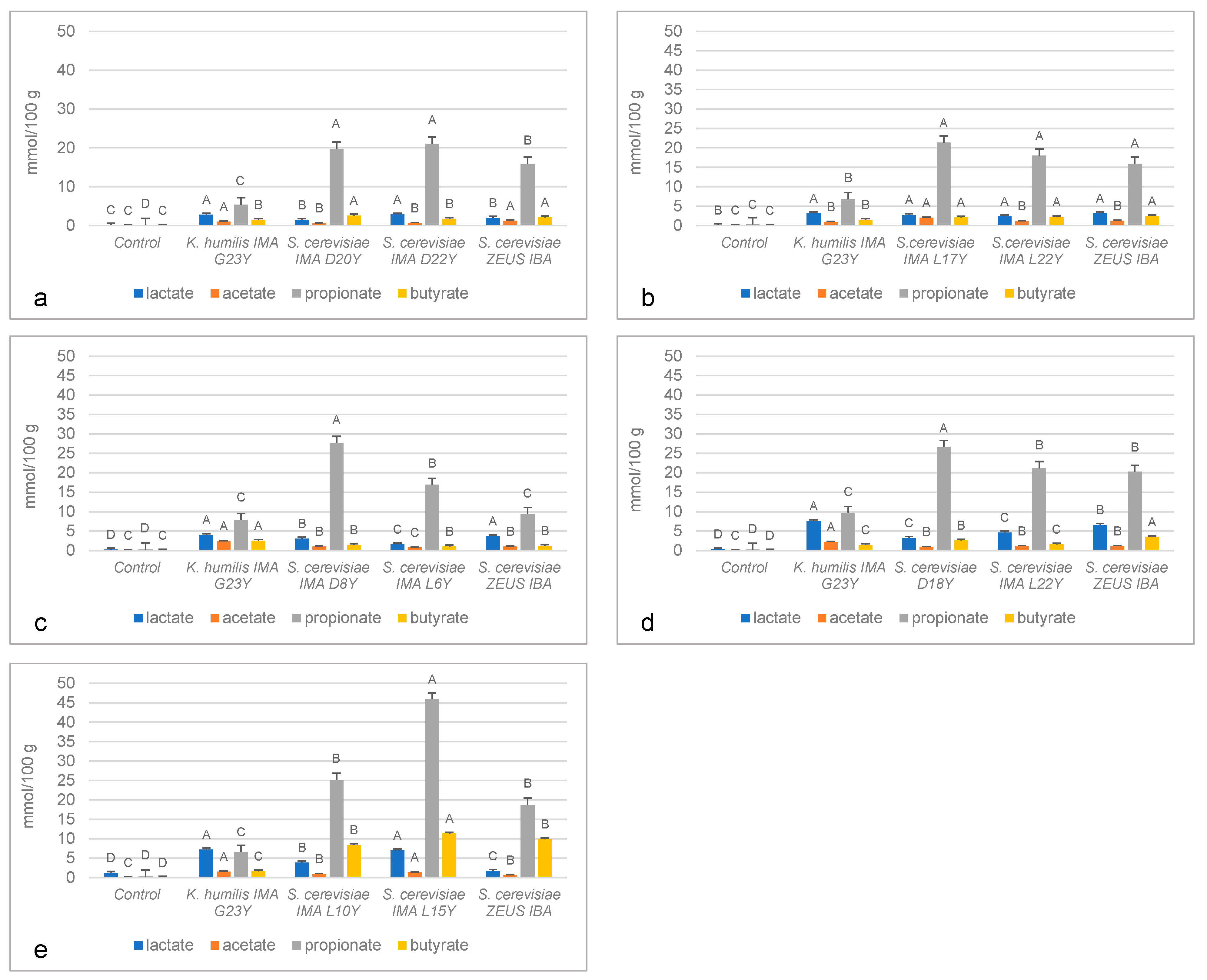
2.5. In Vivo Detection of Organic Acids
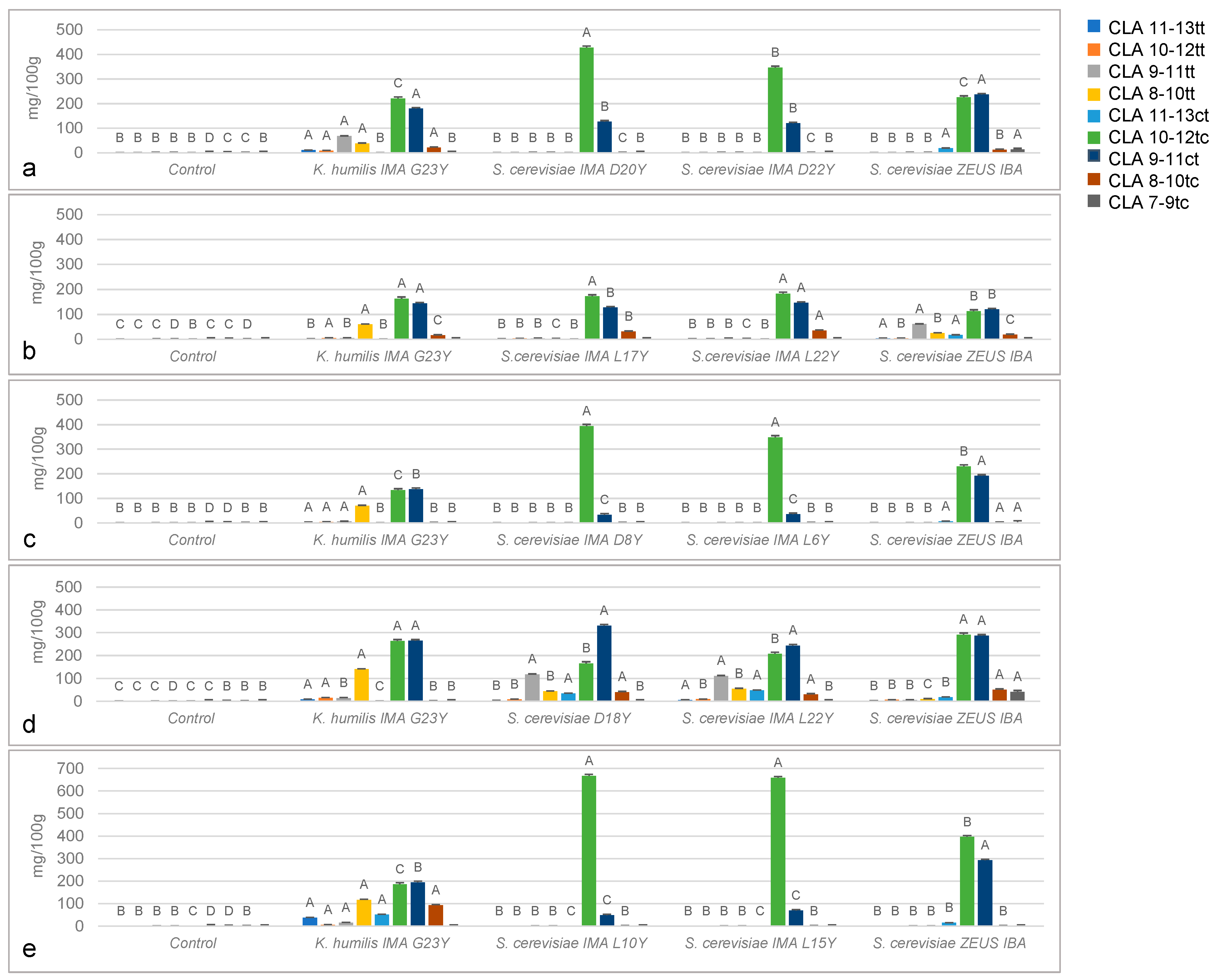
2.6. In Vivo Detection of Fatty Acid and CLA Isomers
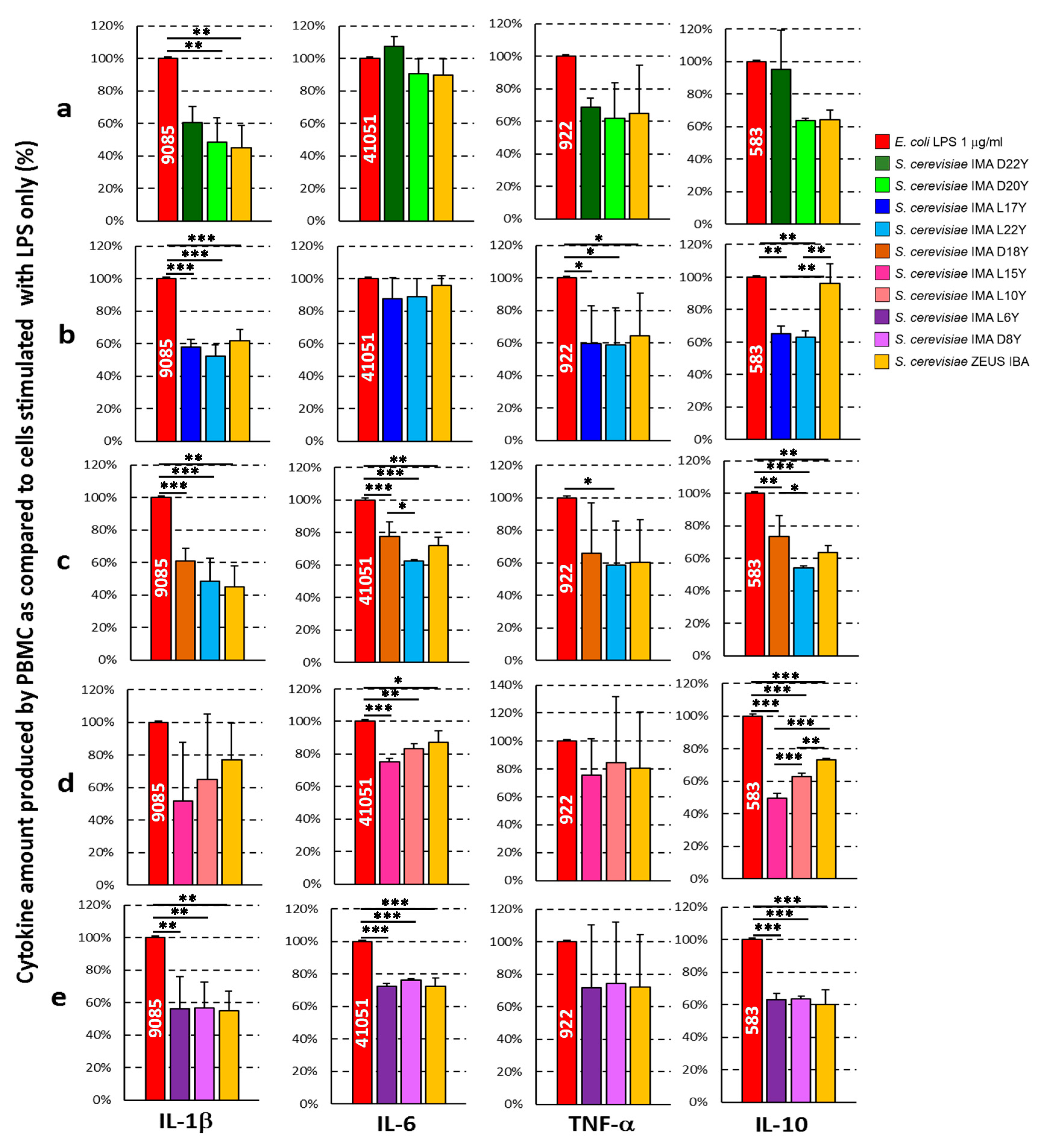
2.7. Ex Vivo Anti-Inflammatory Activity
2.8. Resistance to Simulated Gastric and Intestinal Fluids of Yeast Isolates
2.9. Statistical Analysis
3. Results
3.1. Phylogenetic Relationship among Yeast Strains
3.2. In Vitro Production of Organic Acids
3.3. In Vivo Detection of Organic Acids
3.4. In Vivo Detection of Fatty Acid and Cla Isomers
3.5. Ex Vivo Anti-Inflammatory Activity
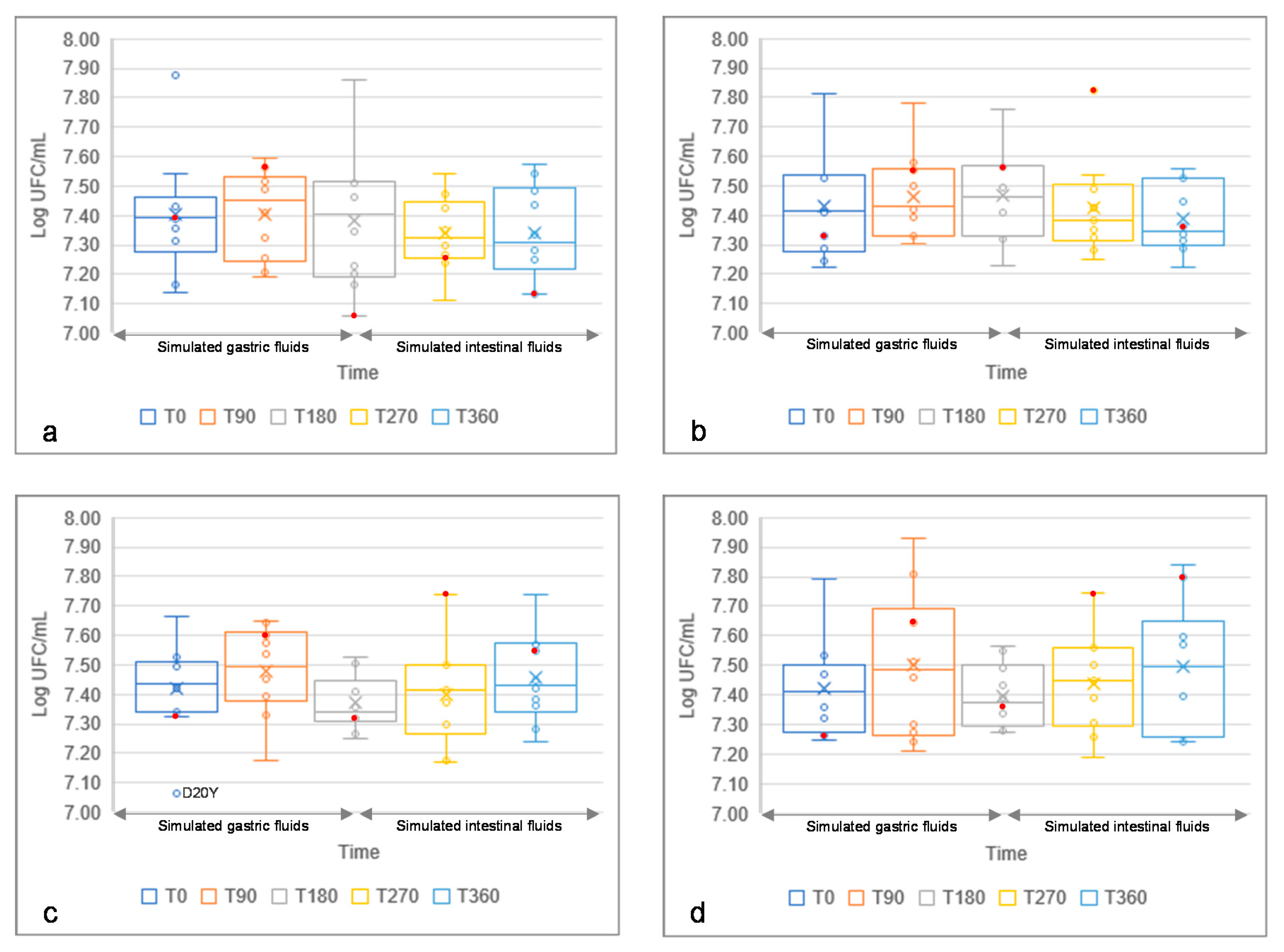
3.6. Resistance to Simulated Gastric and Intestinal Fluids of Yeast Isolates
4. Discussions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Melini, F.; Melini, V.; Luziatelli, F.; Ficca, A.G.; Ruzzi, M. Health-Promoting Components in Fermented Foods: An Up-to-Date Systematic Review. Nutrients 2019, 11, 1189. [Google Scholar] [CrossRef] [Green Version]
- Roffet-Salque, M.; Bogucki, P.I.; Pyzel, J.; Sobkowiak-Tabaka, I.; Grygiel, R.; Szmyt, M.; Evershed, R.P. Earliest evidence for cheese making in the sixth millennium BC in northern Europe. Nature 2013, 493, 522–525. [Google Scholar] [CrossRef]
- Marco, M.L.; Sanders, M.E.; Gänzle, M.; Arrieta, M.C.; Cotter, P.D.; De Vuyst, L.; Hill, C.; Holzapfel, W.; Lebeer, S.; Merenstein, D.; et al. The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on fermented foods. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 196–208. [Google Scholar] [CrossRef] [PubMed]
- Tamang, J.P.; Shin, D.-H.; Jung, S.-J.; Chae, S.-W. Functional Properties of Microorganisms in Fermented Foods. Front. Microbiol. 2016, 7, 578. [Google Scholar] [CrossRef] [Green Version]
- Arora, K.; Ameur, H.; Polo, A.; Di Cagno, R.; Rizzello, C.G.; Gobbetti, M. Thirty years of knowledge on sourdough fermentation: A systematic review. Trends Food Sci. Technol. 2021, 108, 71–83. [Google Scholar] [CrossRef]
- Annunziata, G.; Arnone, A.; Ciampaglia, R.; Tenore, G.C.; Novellino, E. Fermentation of Foods and Beverages as a Tool for Increasing Availability of Bioactive Compounds. Focus on Short-Chain Fatty Acids. Foods 2020, 9, 999. [Google Scholar] [CrossRef]
- Rai, A.K.; Jeyaram, K. Role of yeasts in food fermentation. In Yeast Diversity in Human Welfare; Satyanarayana, T., Kunze, G., Eds.; Springer: Singapore, 2017; pp. 83–113. [Google Scholar] [CrossRef]
- Pessione, A.; Bianco, G.L.; Mangiapane, E.; Cirrincione, S.; Pessione, E. Characterization of potentially probiotic lactic acid bacteria isolated from olives: Evaluation of short chain fatty acids production and analysis of the extracellular proteome. Food Res. Int. 2015, 67, 247–254. [Google Scholar] [CrossRef]
- Ren, M.; Wang, X.; Tian, C.; Li, X.; Zhang, B.; Song, X.; Zhang, J. Characterization of Organic Acids and Phenolic Compounds of Cereal Vinegars and Fruit Vinegars in China. J. Food Process. Preserv. 2017, 41, e12937. [Google Scholar] [CrossRef]
- Lorenzini, M.; Simonato, B.; Slaghenaufi, D.; Ugliano, M.; Zapparoli, G. Assessment of yeasts for apple juice fermentation and production of cider volatile compounds. LWT Food Sci. Technol. 2019, 99, 224–230. [Google Scholar] [CrossRef]
- Hu, R.-K.; Zeng, F.; Wu, L.; Wan, X.; Chen, Y.; Zhang, J.-C.; Liu, B. Fermented carrot juice attenuates type 2 diabetes by mediating gut microbiota in rats. Food Funct. 2019, 10, 2935–2946. [Google Scholar] [CrossRef]
- Jayabalan, R.; Marimuthu, S.; Swaminathan, K. Changes in content of organic acids and tea polyphenols during kombucha tea fermentation. Food Chem. 2007, 102, 392–398. [Google Scholar] [CrossRef]
- Villarreal-Soto, S.A.; Beaufort, S.; Bouajila, J.; Souchard, J.-P.; Renard, T.; Rollan, S.; Taillandier, P. Impact of fermentation conditions on the production of bioactive compounds with anticancer, anti-inflammatory and antioxidant properties in kombucha tea extracts. Process Biochem. 2019, 83, 44–54. [Google Scholar] [CrossRef] [Green Version]
- Annunziata, G.; Maisto, M.; Schisano, C.; Ciampaglia, R.; Daliu, P.; Narciso, V.; Tenore, G.C.; Novellino, E. Colon Bioaccessibility and Antioxidant Activity of White, Green and Black Tea Polyphenols Extract after In Vitro Simulated Gastrointestinal Digestion. Nutrients 2018, 10, 1711. [Google Scholar] [CrossRef] [Green Version]
- Erbaş, M.; Uslu, M.K.; Certel, M. Effects of fermentation and storage on the organic and fatty acid contents of tarhana, a Turkish fermented cereal food. J. Food Compos. Anal. 2006, 19, 294–301. [Google Scholar] [CrossRef]
- Zhang, C.; Brandt, M.J.; Schwab, C.; Gänzle, M. Propionic acid production by cofermentation of Lactobacillus buchneri and Lactobacillus diolivorans in sourdough. Food Microbiol. 2010, 27, 390–395. [Google Scholar] [CrossRef] [PubMed]
- Jayaram, V.B.; Cuyvers, S.; Verstrepen, K.; Delcour, J.; Courtin, C.M. Succinic acid in levels produced by yeast (Saccharomyces cerevisiae) during fermentation strongly impacts wheat bread dough properties. Food Chem. 2014, 151, 421–428. [Google Scholar] [CrossRef]
- Rezaei, M.N.; Aslankoohi, E.; Verstrepen, K.J.; Courtin, C.M. Contribution of the tricarboxylic acid (TCA) cycle and the glyoxylate shunt in Saccharomyces cerevisiae to succinic acid production during dough fermentation. Int. J. Food Microbiol. 2015, 204, 24–32. [Google Scholar] [CrossRef]
- Olšovská, J.; Vrzal, T.; Štěrba, K.; Slabý, M.; Kubizniaková, P.; Čejka, P. The chemical profiling of fatty acids during the brewing process. J. Sci. Food Agric. 2019, 99, 1772–1779. [Google Scholar] [CrossRef]
- Álvarez-Martín, P.; Flórez, A.B.; Hernández-Barranco, A.; Mayo, B. Interaction between dairy yeasts and lactic acid bacteria strains during milk fermentation. Food Control 2008, 19, 62–70. [Google Scholar] [CrossRef]
- Asarat, M.; Apostolopoulos, V.; Vasiljevic, T.; Donkor, O. Short-chain fatty acids produced by synbiotic mixtures in skim milk differentially regulate proliferation and cytokine production in peripheral blood mononuclear cells. Int. J. Food Sci. Nutr. 2015, 66, 755–765. [Google Scholar] [CrossRef]
- Jia, R.; Chen, H.; Chen, H.; Ding, W. Effects of fermentation with Lactobacillus rhamnosus GG on product quality and fatty acids of goat milk yogurt. J. Dairy Sci. 2016, 99, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Li, H.; Chen, Y.; Cao, Y. Method for Screening of Bacterial Strains Biosynthesizing Specific Conjugated Linoleic Acid Isomers. J. Agric. Food Chem. 2012, 60, 9705–9710. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Kim, Y.; Kim, Y.J.; Park, Y. Conjugated Linoleic Acid: Potential Health Benefits as a Functional Food Ingredient. Annu. Rev. Food Sci. Technol. 2016, 7, 221–244. [Google Scholar] [CrossRef]
- Jenkins, T.C.; Wallace, R.J.; Moate, P.; Mosley, E.E. Board-invited review: Recent advances in biohydrogenation of unsaturated fatty acids within the rumen microbial ecosystem. J. Anim. Sci. 2008, 86, 397–412. [Google Scholar] [CrossRef] [PubMed]
- Fritsche, J.; Rickert, R.; Steinhart, H.; Yurawecz, M.P.; Mossoba, M.M.; Sehat, N.; Roach, J.A.G.; Kramer, J.K.G.; Ku, Y. Conjugated linoleic acid (CLA) isomers: Formation, analysis, amounts in foods, and dietary intake. Lipid 1999, 101, 272–276. [Google Scholar] [CrossRef]
- Adamczak, M.; Bornscheuer, U.; Bednarski, W. Properties and biotechnological methods to produce lipids containing conjugated linoleic acid. Eur. J. Lipid Sci. Technol. 2008, 110, 491–504. [Google Scholar] [CrossRef]
- Mooney, D.; McCarthy, C.; Belton, O. Effects of conjugated linoleic acid isomers on monocyte, macrophage and foam cell phenotype in atherosclerosis. Prostaglandins Other Lipid Mediat. 2012, 98, 56–62. [Google Scholar] [CrossRef]
- Andrade, J.; Ascenção, K.; Gullón, P.; Henriques, S.M.S.; Pinto, J.; Rocha-Santos, T.; Freitas, A.C.; Gomes, A.M. Production of conjugated linoleic acid by food-grade bacteria: A review. Int. J. Dairy Technol. 2012, 65, 467–481. [Google Scholar] [CrossRef]
- Dahiya, D.K.; Puniya, A.K. Isolation, molecular characterization and screening of indigenous lactobacilli for their abilities to produce bioactive conjugated linoleic acid (CLA). J. Food Sci. Technol. 2017, 54, 792–801. [Google Scholar] [CrossRef] [Green Version]
- Silvetti, T.; Morandi, S.; Brasca, M. Lactic acid bacteria: A cell factory for delivering functional biomolecules in dairy products. In Microbiology in Dairy Processing; Poltronieri, P., Ed.; John Wiley & Sons Ltd.: Chicago, IL, USA; The Institute of Food Technologists: Chicago, IL, USA, 2018; pp. 251–278. [Google Scholar]
- Lin, T.Y. Influence of lactic cultures, linoleic acid and fructo-oligosaccharides on conjugated linoleic acid concentration in non-fat set yogurt. Aust. J. Dairy Technol. 2003, 58, 11–14. [Google Scholar]
- Csápo, J.; Varga-Visi, É. Conjugated linoleic acid production in fermented foods. In Advances in Fermented Foods and Beverages; Holzapfel, W., Ed.; Woodhead Publishing: Sawston, UK, 2015; pp. 75–105. [Google Scholar] [CrossRef]
- Gorissen, L.; Leroy, F.; De Vuyst, L.; De Smet, S.; Raes, K. Bacterial Production of Conjugated Linoleic and Linolenic Acid in Foods: A Technological Challenge. Crit. Rev. Food Sci. Nutr. 2015, 55, 1561–1574. [Google Scholar] [CrossRef]
- Liu, P.; Shen, S.-R.; Ruan, H.; Zhou, Q.; Ma, L.-L.; He, G.-Q. Production of conjugated linoleic acids by Lactobacillus plantarum strains isolated from naturally fermented Chinese pickles. J. Zhejiang Univ. Sci. B 2011, 12, 923–930. [Google Scholar] [CrossRef] [Green Version]
- Black, B.A.; Zannini, E.; Curtis, J.M.; Gänzle, M. Antifungal Hydroxy Fatty Acids Produced during Sourdough Fermentation: Microbial and Enzymatic Pathways, and Antifungal Activity in Bread. Appl. Environ. Microbiol. 2013, 79, 1866–1873. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ando, A.; Ogawa, J.; Sugimoto, S.; Kishino, S.; Sakuradani, E.; Yokozeki, K.; Shimizu, S. Selective production of cis-9, trans-11 isomer of conjugated linoleic acid from trans-vaccenic acid methyl ester by Delacroixia coronata. J. Appl. Microbiol. 2009, 106, 1697–1704. [Google Scholar] [CrossRef] [PubMed]
- Fadda, M.E.; Mossa, V.; Deplano, M.; Pisano, M.B.; Cosentino, S. In vitro screening of Kluyveromyces strains isolated from Fiore Sardo cheese for potential use as probiotics. LWT Food Sci. Technol. 2017, 75, 100–106. [Google Scholar] [CrossRef]
- Sambrani, R.; Abdolalizadeh, J.; Kohan, L.; Jafari, B. Recent advances in the application of probiotic yeasts, particularly Saccharomyces, as an adjuvant therapy in the management of cancer with focus on colorectal cancer. Mol. Biol. Rep. 2021, 48, 951–960. [Google Scholar] [CrossRef]
- Agarbati, A.; Canonico, L.; Marini, E.; Zannini, E.; Ciani, M.; Comitini, F. Potential Probiotic Yeasts Sourced from Natural Environmental and Spontaneous Processed Foods. Foods 2020, 9, 287. [Google Scholar] [CrossRef] [Green Version]
- Kumura, H.; Tanoue, Y.; Tsukahara, M.; Tanaka, T.; Shimazaki, K. Screening of Dairy Yeast Strains for Probiotic Applications. J. Dairy Sci. 2004, 87, 4050–4056. [Google Scholar] [CrossRef]
- Diosma, G.; Romanin, D.; Rey-Burusco, M.F.; Londero, A.; Garrote, G.L. Yeasts from kefir grains: Isolation, identification, and probiotic characterization. World J. Microbiol. Biotechnol. 2014, 30, 43–53. [Google Scholar] [CrossRef]
- Menezes, A.; Ramos, C.; Cenzi, G.; Melo, D.S.; Dias, D.R.; Schwan, R.F. Probiotic Potential, Antioxidant Activity, and Phytase Production of Indigenous Yeasts Isolated from Indigenous Fermented Foods. Probiotics Antimicrob. Proteins 2020, 12, 280–288. [Google Scholar] [CrossRef]
- Perricone, M.; Bevilacqua, A.; Corbo, M.R.; Sinigaglia, M. Technological characterization and probiotic traits of yeasts isolated from Altamura sourdough to select promising microorganisms as functional starter cultures for cereal-based products. Food Microbiol. 2014, 38, 26–35. [Google Scholar] [CrossRef]
- Ogunremi, O.; Sanni, A.; Agrawal, R. Probiotic potentials of yeasts isolated from some cereal-based Nigerian traditional fermented food products. J. Appl. Microbiol. 2015, 119, 797–808. [Google Scholar] [CrossRef] [PubMed]
- Hsu, S.A.; Chou, J.Y. Yeasts in fermented food and kefir: In Vitro characterization of probiotic traits. J. Anim. Plant Sci. 2021, 31, 567–582. [Google Scholar] [CrossRef]
- Li, S.; Zhang, Y.; Yin, P.; Zhang, K.; Liu, Y.; Gao, Y.; Li, Y.; Wang, T.; Lu, S.; Li, B. Probiotic potential of γ-aminobutyric acid (GABA)–producing yeast and its influence on the quality of cheese. J. Dairy Sci. 2021, 104, 6559–6576. [Google Scholar] [CrossRef]
- Chaves-López, C.; Serio, A.; Paparella, A.; Martuscelli, M.; Corsetti, A.; Tofalo, R.; Suzzi, G. Impact of microbial cultures on proteolysis and release of bioactive peptides in fermented milk. Food Microbiol. 2014, 42, 117–121. [Google Scholar] [CrossRef] [PubMed]
- Rai, A.K.; Pandey, A.; Sahoo, D. Biotechnological potential of yeasts in functional food industry. Trends Food Sci. Technol. 2019, 83, 129–137. [Google Scholar] [CrossRef]
- Palla, M.; Blandino, M.; Grassi, A.; Giordano, D.; Sgherri, C.; Quartacci, M.F.; Reyneri, A.; Agnolucci, M.; Giovannetti, M. Characterization and selection of functional yeast strains during sourdough fermentation of different cereal wholegrain flours. Sci. Rep. 2020, 10, 12856. [Google Scholar] [CrossRef]
- Palla, M.; Agnolucci, M.; Calzone, A.; Giovannetti, M.; Di Cagno, R.; Gobbetti, M.; Rizzello, C.G.; Pontonio, E. Exploitation of autochthonous Tuscan sourdough yeasts as potential starters. Int. J. Food Microbiol. 2019, 302, 59–68. [Google Scholar] [CrossRef]
- Palla, M.; Cristani, C.; Giovannetti, M.; Agnolucci, M. Large Genetic Intraspecific Diversity of Autochthonous Lactic Acid Bacteria and Yeasts Isolated from PDO Tuscan Bread Sourdough. Appl. Sci. 2020, 10, 1043. [Google Scholar] [CrossRef] [Green Version]
- Sandri, M.; Monego, S.D.; Conte, G.; Sgorlon, S.; Stefanon, B. Raw meat based diet influences faecal microbiome and end products of fermentation in healthy dogs. BMC Vet. Res. 2017, 13, 65. [Google Scholar] [CrossRef]
- Rodriguez-Estrada, M.; Penazzi, G.; Caboni, M.F.; Bertacco, G.; Lercker, G. Effect of different cooking methods on some lipid and protein components of hamburgers. Meat Sci. 1997, 45, 365–375. [Google Scholar] [CrossRef]
- Christie, W.W. (Ed.) Preparation of ester derivatives of fatty acids for chromatographic analysis. In Advances in Lipid Methodology; Oily Press: Dundee, UK, 1993; pp. 69–111. [Google Scholar]
- Mele, M.; Serra, A.; Pauselli, M.; Luciano, G.; Lanza, M.; Pennisi, P.; Conte, G.; Taticchi, A.; Esposto, S.; Morbidini, L. The use of stoned olive cake and rolled linseed in the diet of intensively reared lambs: Effect on the intramuscular fatty-acid composition. Animal 2014, 8, 152–162. [Google Scholar] [CrossRef] [PubMed]
- Conte, G.; Serra, A.; Casarosa, L.; Ciucci, F.; Cappucci, A.; Bulleri, E.; Corrales-Retana, L.; Buccioni, A.; Mele, M. Effect of Linseed Supplementation on Total Longissimus Muscle Lipid Composition and Shelf-Life of Beef from Young Maremmana Bulls. Front. Vet. Sci. 2019, 5, 326. [Google Scholar] [CrossRef]
- Kramer, J.K.G.; Cruz-Hernandez, C.; Deng, Z.; Zhou, J.; Jahreis, G.; Dugan, M.E.R. Analysis of conjugated linoleic acid and trans 18:1 isomers in synthetic and animal products. Am. J. Clin. Nutr. 2004, 79, 1137S–1145S. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Osborne, T.B. The Proteins of the Wheat Kernel; The Carnegie Institution of Washington: Washington, DC, USA, 1907; pp. 1–119. [Google Scholar]
- Weiss, W.; Vogelmeier, C.; Görg, A. Electrophoretic characterization of wheat grain allergens from different cultivars involved in bakers’ asthma. Electrophoresis 1993, 14, 805–816. [Google Scholar] [CrossRef]
- Esin, S.; Shigematsu, M.; Nagai, S.; Eklund, A.; Wigzell, H.; Grunewald, J. Different Percentages of Peripheral Blood γδ+ T Cells in Healthy Individuals from Different Areas of the World. Scand. J. Immunol. 1996, 43, 593–596. [Google Scholar] [CrossRef] [PubMed]
- Kaya, E.; Grassi, L.; Benedetti, A.; Maisetta, G.; Pileggi, C.; Di Luca, M.; Batoni, G.; Esin, S. In vitro Interaction of Pseudomonas aeruginosa Biofilms with Human Peripheral Blood Mononuclear Cells. Front. Cell. Infect. Microbiol. 2020, 10, 187. [Google Scholar] [CrossRef]
- Zárate, G.; Chaia, A.P.; Gonzalez, S.N.; Oliver, G. Viability and β-Galactosidase Activity of Dairy Propionibacteria Subjected to Digestion by Artificial Gastric and Intestinal Fluids. J. Food Prot. 2000, 63, 1214–1221. [Google Scholar] [CrossRef] [PubMed]
- Moslehi-Jenabian, S.; Lindegaard, L.; Jespersen, L. Beneficial Effects of Probiotic and Food Borne Yeasts on Human Health. Nutrients 2010, 2, 449–473. [Google Scholar] [CrossRef]
- Fernández, M.F.; Boris, S.; Barbes, C. Probiotic properties of human lactobacilli strains to be used in the gastrointestinal tract. J. Appl. Microbiol. 2003, 94, 449–455. [Google Scholar] [CrossRef]
- Pétel, C.; Onno, B.; Prost, C. Sourdough volatile compounds and their contribution to bread: A review. Trends Food Sci. Technol. 2017, 59, 105–123. [Google Scholar] [CrossRef]
- De Vero, L.; Iosca, G.; La China, S.; Licciardello, F.; Gullo, M.; Pulvirenti, A. Yeasts and Lactic Acid Bacteria for Panettone Production: An Assessment of Candidate Strains. Microorganisms 2021, 9, 1093. [Google Scholar] [CrossRef]
- Maljaars, P.; Peters, H.; Mela, D.; Masclee, A. Ileal brake: A sensible food target for appetite control. A review. Physiol. Behav. 2008, 95, 271–281. [Google Scholar] [CrossRef]
- Al-Lahham, S.H.; Peppelenbosch, M.P.; Roelofsen, H.; Vonk, R.J.; Venema, K. Biological effects of propionic acid in humans; metabolism, potential applications and underlying mechanisms. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2010, 1801, 1175–1183. [Google Scholar] [CrossRef]
- Zhang, D.; Tan, B. Effects of different solid-state fermentation ratios of S. cerevisiae and L. plantarum on physico-chemical properties of wheat bran and the quality of whole wheat bread. J. Sci. Food Agric. 2021, 101, 4551–4560. [Google Scholar] [CrossRef]
- Scharinger, M.; Kuntz, M.; Scharinger, A.; Teipel, J.; Kuballa, T.; Walch, S.; Lachenmeier, D. Rapid Approach to Determine Propionic and Sorbic Acid Contents in Bread and Bakery Products Using 1H NMR Spectroscopy. Foods 2021, 10, 526. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Björck, L.; Fondén, R. Production of conjugated linoleic acid by dairy starter cultures. J. Appl. Microbiol. 1998, 85, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Dilzer, A.; Park, Y. Implication of Conjugated Linoleic Acid (CLA) in Human Health. Crit. Rev. Food Sci. Nutr. 2012, 52, 488–513. [Google Scholar] [CrossRef]
- Rossol, M.; Heine, H.; Meusch, U.; Quandt, D.; Klein, C.; Sweet, M.; Hauschildt, S. LPS-induced Cytokine Production in Human Monocytes and Macrophages. Crit. Rev. Immunol. 2011, 31, 379–446. [Google Scholar] [CrossRef] [PubMed]
- Romanin, D.E.; Llopis, S.; Genoves, S.; Martorell, P.; Ramón, V.; Garrote, G.L.; Rumbo, M. Probiotic yeast Kluyveromyces marxianus CIDCA 8154 shows anti-inflammatory and anti-oxidative stress properties in in vivo models. Benef. Microbes 2016, 7, 83–93. [Google Scholar] [CrossRef]
- Aloğlu, H.Ş.; Özer, E.D.; Öner, Z. Assimilation of cholesterol and probiotic characterisation of yeast strains isolated from raw milk and fermented foods. Int. J. Dairy Technol. 2016, 69, 63–70. [Google Scholar] [CrossRef]
- Arevalo-Villena, M.; Briones-Perez, A.; Corbo, M.; Sinigaglia, M.; Bevilacqua, A. Biotechnological application of yeasts in food science: Starter cultures, probiotics and enzyme production. J. Appl. Microbiol. 2017, 123, 1360–1372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Freire, A.L.; Ramos, C.L.; Souza, P.N.C.; Cardoso, M.G.B.; Schwan, R.F. Nondairy beverage produced by controlled fermentation with potential probiotic starter cultures of lactic acid bacteria and yeast. Int. J. Food Microbiol. 2017, 248, 39–46. [Google Scholar] [CrossRef]
- Priyodip, P.; Prakash, P.Y.; Balaji, S. Phytases of Probiotic Bacteria: Characteristics and Beneficial Aspects. Indian J. Microbiol. 2017, 57, 148–154. [Google Scholar] [CrossRef] [PubMed]
| Strains a | Source of Isolation |
|---|---|
| Saccharomyces cerevisiae IMA D8Y, D18Y, D20Y, D22Y, L6Y, L10Y, L15Y, L17Y, L22Y, Kazachstania humilis IMA G23Y | Tuscan wheat sourdoughs [51] |
| Saccharomyces cerevisiae Zeus IBA | Commercial baker’s yeast of Zeus IBA srl |
| Saccharomyces cerevisiae var. boulardii CNCM I-745 | Commercial probiotics’ yeast from Codex (CODEX, Zambon Italia S.r.l., Bresso, Italy) |
| Saccharomyces cerevisiae ATCC 32167 | Unknown |
| Kazachstania humilis DBVPG 6753 | San Francisco Sourdough Bread |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palla, M.; Conte, G.; Grassi, A.; Esin, S.; Serra, A.; Mele, M.; Giovannetti, M.; Agnolucci, M. Novel Yeasts Producing High Levels of Conjugated Linoleic Acid and Organic Acids in Fermented Doughs. Foods 2021, 10, 2087. https://doi.org/10.3390/foods10092087
Palla M, Conte G, Grassi A, Esin S, Serra A, Mele M, Giovannetti M, Agnolucci M. Novel Yeasts Producing High Levels of Conjugated Linoleic Acid and Organic Acids in Fermented Doughs. Foods. 2021; 10(9):2087. https://doi.org/10.3390/foods10092087
Chicago/Turabian StylePalla, Michela, Giuseppe Conte, Arianna Grassi, Semih Esin, Andrea Serra, Marcello Mele, Manuela Giovannetti, and Monica Agnolucci. 2021. "Novel Yeasts Producing High Levels of Conjugated Linoleic Acid and Organic Acids in Fermented Doughs" Foods 10, no. 9: 2087. https://doi.org/10.3390/foods10092087
APA StylePalla, M., Conte, G., Grassi, A., Esin, S., Serra, A., Mele, M., Giovannetti, M., & Agnolucci, M. (2021). Novel Yeasts Producing High Levels of Conjugated Linoleic Acid and Organic Acids in Fermented Doughs. Foods, 10(9), 2087. https://doi.org/10.3390/foods10092087








