Use of Meat-Bone Paste to Develop Calcium-Enriched Liver Pâté
Abstract
1. Introduction
2. Materials and Methods
2.1. Meat-Bone Paste Preparation
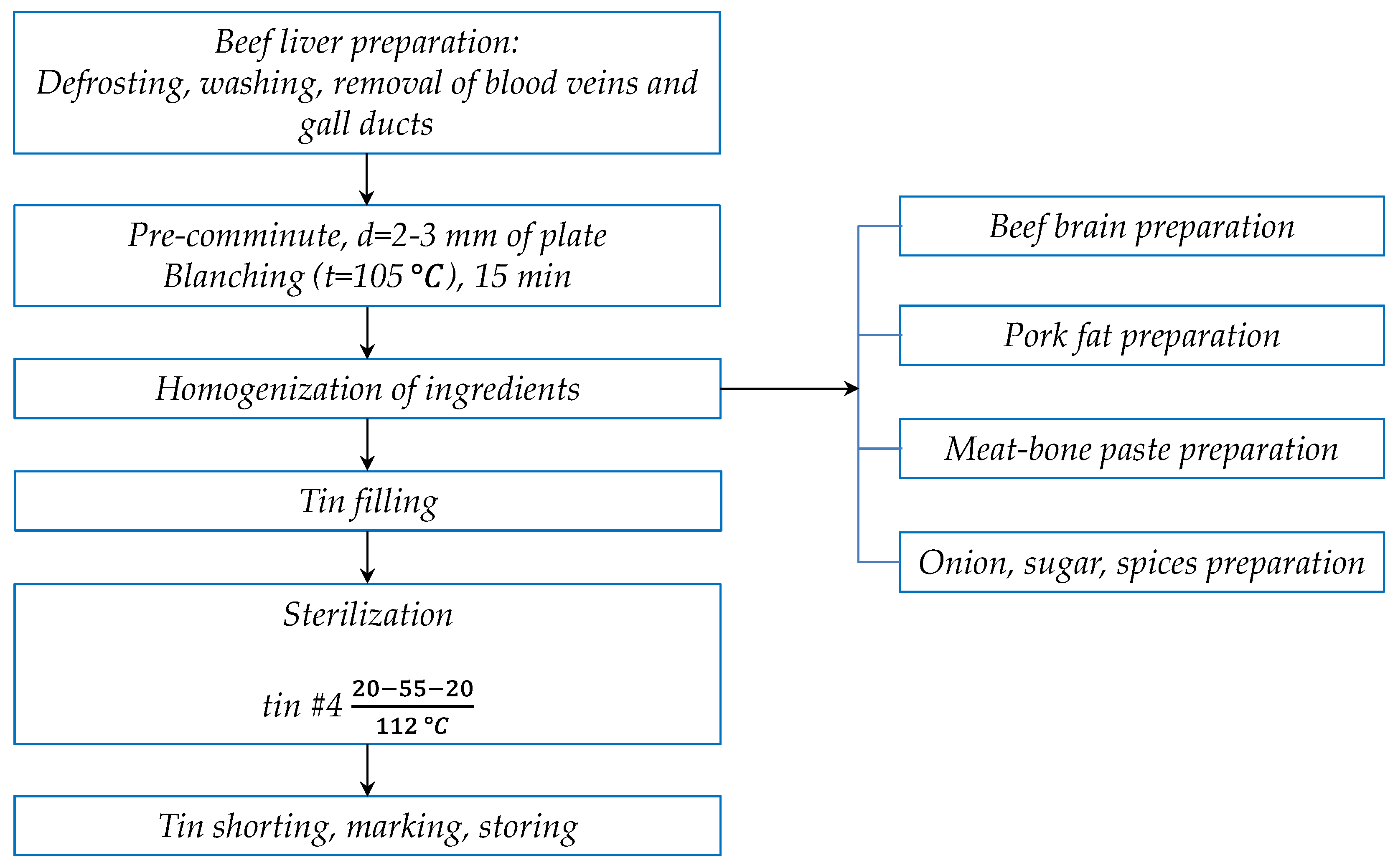
2.2. Liver Pâté Preparation
2.3. Determination of Chemical Composition
2.4. Caloric Value Calculation
2.5. Mineral Composition Determination
2.6. Amino Acid Composition Determination
2.7. Microstructure Analysis
2.8. Statistical Analysis
3. Results and Discussion
3.1. Mineral Composition of Meat-Bone Paste
3.2. Microstructure Analysis of Meat-Bone Paste
3.3. Liver Pâte Chemical and Mineral Composition
3.4. Amino Acid Composition
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hamzeh, A.; Azizieh, A.; Yazagy, S. Effect of the Fat Percentage and Liver Type in the Stability and pH Value of Locally Prepared Liver Pate. Int. Food Res. J. 2016, 23, 1131–1135. [Google Scholar]
- Xiong, G.; Wang, P.; Zheng, H.; Xu, X.; Zhu, Y.; Zhou, G. Effects of Plant Oil Combinations Substituting Pork Back-Fat Combined with Pre-Emulsification on Physicochemical, Textural, Microstructural and Sensory Properties of Spreadable Chicken Liver PÂTÉ. J. Food Qual. 2016, 39, 331–341. [Google Scholar] [CrossRef]
- Munekata, P.E.S.; Domínguez, R.; Campagnol, P.C.B.; Franco, D.; Trindade, M.A.; Lorenzo, J.M. Effect of natural antioxidants on physicochemical properties and lipid stability of pork liver pâté manufactured with healthy oils during refrigerated storage. J. Food Sci. Technol. 2017, 54, 4324–4334. [Google Scholar] [CrossRef] [PubMed]
- Domínguez, R.; Agregán, R.; Gonçalves, A.; Lorenzo, J.M. Effect of fat replacement by olive oil on the physico-chemical properties, fatty acids, cholesterol and tocopherol content of pâté. Grasas Aceites 2016, 67, e133. [Google Scholar]
- Tiensa, B.E.; Barbut, S.; Marangoni, A.G. Influence of fat structure on the mechanical properties of commercial pate products. Food Res. Int. 2017, 100, 558–565. [Google Scholar] [CrossRef] [PubMed]
- Vargas-Ramella, M.; Pateiro, M.; Barba, F.J.; Franco, D.; Campagnol, P.C.B.; Munekata, P.E.S.; Tomasevic, I.; Domínguez, R.; Lorenzo, J.M. Microencapsulation of healthier oils to enhance the physicochemical and nutritional properties of deer pâté. LWT 2020, 125, 109223. [Google Scholar] [CrossRef]
- Madruga, M.S.; Stephen Elmore, J.; Dodson, A.T.; Mottram, D.S. Volatile flavour profile of goat meat extracted by three widely used techniques. Food Chem. 2009, 115, 1081–1087. [Google Scholar] [CrossRef]
- Domínguez, R.; Purriños, L.; Pérez-Santaescolástica, C.; Pateiro, M.; Barba, F.J.; Tomasevic, I.; Campagnol, P.C.B.; Lorenzo, J.M. Characterization of volatile compounds of dry-cured meat products using HS-SPME-GC/MS technique. Food Anal. Methods 2019, 12, 1263–1284. [Google Scholar] [CrossRef]
- Domínguez, R.; Pateiro, M.; Gagaoua, M.; Barba, F.J.; Zhang, W.; Lorenzo, J.M. A comprehensive review on lipid oxidation in meat and meat products. Antioxidants 2019, 8, 429. [Google Scholar] [CrossRef]
- Lorido, L.; Estévez, M.; Ventanas, S. A novel approach to assess temporal sensory perception of muscle foods: Application of a time–intensity technique to diverse Iberian meat products. Meat Sci. 2014, 96, 385–393. [Google Scholar] [CrossRef]
- Yude, L. Comprehensive Utilization of Animal Bones. Food Sci. 2000, 2, 1–10. [Google Scholar]
- Peshuk, L.; Budnyk, N.; Halenko, O. Gerodietic Meat Products Technology Enriched with Calcium and Phosphorus. Food Environ. Saf. 2011, 10, 18–23. [Google Scholar]
- Kakimov, A.; Kabdylzhar, B.; Yessimbekov, Z.; Suychinov, A.; Baikadamova, A. Identifying patterns in the effect exerted by a cooling process and the fine grinding modes on the qualitative indicators of a meat and bone paste. East.-Eur. J. Enterp. Technol. 2020, 2, 6–12. [Google Scholar] [CrossRef]
- Regulation (EC) No 1069/2009 European Parliament and of the Council of 21 October 2009 laying down health rules as regards animal by-products and derived products not intended for human consumption and repealing Regulation (EC) No 1774/2002 (Animal by-products Regulation). Off. J. Eur. Union 2009, L300, 1–33.
- Kakimov, A.; Suychinov, A.; Mayorov, A.; Yessimbekov, Z.; Okuskhanova, E.; Kuderinova, N.; Bakiyeva, A. Meat-bone paste as an ingredient for meat batter, effect on physicochemical properties and amino acid composition. Pak. J. Nutr. 2017, 16, 797–804. [Google Scholar] [CrossRef][Green Version]
- Kakimov, A.; Kabdylzhar, B.; Suychinov, A.; Yessimbekov, Z.; Baikadamova, A.; Zolotov, A.; Zharykbasova, K. The chemical profile and the effect of temperature and storage time on the change of yield stress and pH of meat-bone paste. Eur. Asian J. Biosci. 2019, 13, 2093–2097. [Google Scholar]
- Domínguez, R.; Pateiro, M.; Munekata, P.E.S.; Campagnol, P.C.B.; Lorenzo, J.M.; Sichetti Munekata, P.E.; Bastianello Campagnol, P.C.; Lorenzo, J.M. Influence of partial pork backfat replacement by fish oil on nutritional and technological properties of liver pâté. Eur. J. Lipid Sci. Technol. 2017, 119, 1600178. [Google Scholar] [CrossRef]
- Okuskhanova, E.; Assenova, B.; Rebezov, M.; Yessimbekov, Z.; Kulushtayeva, B.; Zinina, O.; Stuart, M. Mineral composition of deer meat pate. Pak. J. Nutr. 2016, 15, 217–222. [Google Scholar] [CrossRef][Green Version]
- Bayer, N.A.; Nekludov, A.D.; Dubina, V.I.; Telyashevskaya, L.Y.; Aleshin, A.A.; Lunev, G.G.; Timoshkina, E.A.; Lamm, E.L. Method of Production of Protein Hydrolyzate from Meat and Meat-Bone Raw Material 2000. Available online: https://patents.google.com/patent/RU2160538C1/ru (accessed on 28 August 2021).
- Kutsakova, V.E.; Frolov, S.V.; Udachin, S.P.; Goryainov, S.N.; Marchenko, V.I. Method of Hydrolyzates Production from the Bone Wastes of Poultry and Fish. 2006. Available online: https://patents.google.com/patent/RU2272418C1/ru?oq=2272418 (accessed on 28 August 2021).
- Soshieeda, K.K. Method for Producing Bone-Mixed Meat Paste. Japan Patent No. 2012210197, 1 November 2012. [Google Scholar]
- Kinichi, E.; Tatsu, M. Production of Fish Bone Paste and Utilization of Fish Bone Paste. Japan Patent No. 04-190768, 9 July 1992. [Google Scholar]
- Amirkhanov, K.; Igenbayev, A.; Nurgazezova, A.; Okuskhanova, E.; Kassymov, S.; Muslimova, N.; Yessimbekov, Z. Comparative Analysis of Red and White Turkey Meat Quality. Pak. J. Nutr. 2017, 16, 412–416. [Google Scholar] [CrossRef][Green Version]
- Kyoichi, I. Japan Calcium-Rich Health Food. Japan Patent No. 03-078463, 13 October 1992. [Google Scholar]
- Shinjuro, N. Meat-like Composition and Its Preparation. Japan Patent No. 58-205472, 30 November 1983. [Google Scholar]
- GOST 12319-77. The Standard Applies to Canned Meat “Liver Pate”, Packed in Cans, Hermetically Sealed and Sterilized. 2012. Available online: https://docs.cntd.ru/document/1200021499 (accessed on 28 August 2021).
- Toldrá, F.; Group, F. Handbook of Analysis of Edible Animal By-Products, 1st ed.; Toldrá, F., Group, F., Eds.; CRC Press: Boca Raton, FL, USA, 2011; ISBN 9781439803608. [Google Scholar]
- GOST 9793-74. Method of Moisture Determination. 2010. Available online: https://docs.cntd.ru/document/901712025 (accessed on 28 August 2021).
- GOST R 51479-99. Method for Determination of Moisture Content. 2010. Available online: https://docs.cntd.ru/document/1200028186 (accessed on 28 August 2021).
- GOST 23042-86. Method of Fat Determination. 2010. Available online: https://docs.cntd.ru/document/1200021649 (accessed on 28 August 2021).
- GOST 25011-81. Method of Protein Determination. 2010. Available online: https://docs.cntd.ru/document/1200021660 (accessed on 28 August 2021).
- Rudenko, A.O.; Kartsova, L.A. Determination of the most important amino acids in complex objects of biological origin by reversed-phase HPLC with obtaining phenylthiohydantoin amino acids. Sorption Chromatogr. Process. 2010, 10, 223–230. [Google Scholar]
- Rao, M.; Gault, N.; Kennedy, S. Changes in the Ultrastructure of Beef Muscle as Influenced by Acidic Conditions Below the Ultimate pH. Food Struct. 1989, 8, 115–124. [Google Scholar]
- Kakimov, A.; Yessimbekov, Z.; Kabulov, B.; Bepeyeva, A.; Kuderinova, N.; Ibragimov, N. Studying Chemical Composition and Yield Stress of Micronized Grinded Cattle Bone Paste. Res. J. Pharm. Biol. Chem. Sci. 2016, 7, 805–8012. [Google Scholar]
- Kakimov, A.K.; Kabulov, B.B.; Yessimbekov, Z.S.; Kuderinova, N.A. Use of meat-bone paste as a protein source in meat product production. Theory Pract. Meat Process. 2016, 1, 42–50. [Google Scholar] [CrossRef]
- Kakimov, A.; Yessimbekov, Z.; Suychinov, A.; Japanov, T.; Zolotov, A. Mineral and amino acid composition of meat-bone paste. Int. Sci. Pract. Conf. WORLD Sci. 2017, 5, 5–7. [Google Scholar]
- Kakimov, A.; Suychinov, A.; Tsoy, A.; Mustambayev, N.; Ibragimov, N.; Kuderinova, N.; Mirasheva, G.; Yessimbekov, Z. Nutritive and biological value of liver and blood of various slaughtered animals. J. Pharm. Res. Int. 2018, 22, 1–5. [Google Scholar] [CrossRef]
- Sitalakshmi, R.; Kumar, S.P. Trace Elements in Health and Disease: A Review. Res. J. Pharm. Biol. Chem. Sci. 2014, 5, 450–455. [Google Scholar]
- Beto, J.A. The Role of Calcium in Human Aging. Clin. Nutr. Res. 2015, 4, 8. [Google Scholar] [CrossRef]
- Aikawa, J.K. Magnesium: It’s Biologic Significance, 1st ed.; Aikawa, J.K., Ed.; CRC Press: Boca Raton, FL, USA, 1981; ISBN 9780367226374. [Google Scholar]
- Gröber, U.; Schmidt, J.; Kisters, K. Magnesium in Prevention and Therapy. Nutrients 2015, 7, 8199–8226. [Google Scholar] [CrossRef]
- Domínguez, R.; Pateiro, M.; Pérez-Santaescolástica, C.; Munekata, P.E.S.; Lorenzo, J.M. Salt reduction strategies in meat products made from whole pieces. In Strategies for Obtaining Healthier Foods; Lorenzo, J.M., Carballo, F.J., Eds.; Nova Science Publishers: Hauppauge, NY, USA, 2017; pp. 267–289. ISBN 978-1-53612-159-9. [Google Scholar]
- Pedro, D.; Saldaña, E.; Lorenzo, J.M.; Pateiro, M.; Dominguez, R.; Dos Santos, B.A.; Cichoski, A.J.; Campagnol, P.C.B. Low-sodium dry-cured rabbit leg: A novel meat product with healthier properties. Meat Sci. 2021, 173, 108372. [Google Scholar] [CrossRef]
- Pateiro, M.; Munekata, P.E.S.; Cittadini, A.; Domínguez, R.; Lorenzo, J.M. Metallic-based salt substitutes to reduce sodium content in meat products. Curr. Opin. Food Sci. 2021, 38, 21–31. [Google Scholar] [CrossRef]
- Rebrov, V.G.; Gromova, O.A. Vitamins, Macro-and Micronutrients, 1st ed.; Rebrov, V.G., Gromova, O.A., Eds.; GEOTAR-Media: Moscow, Rusia, 2008. [Google Scholar]
- Raina, R.; Garg, G.; Sethi, S.K.; Schreiber, M.J.; Simon, J.F.; Thomas, G. Phosphorus Metabolism. J. Nephrol. Ther. 2012, 3, 1–7. [Google Scholar] [CrossRef]
- Yeung, D.L. Iron and micronutrients: Complementary food fortification. Food Nutr. Bullet 1998, 19, 159–163. [Google Scholar] [CrossRef]
- Troshina, E.A.; Platonova, N.M. Iodine metabolism and prevention of iodine deficiency diseases in children and teenagers. Issues Mod. Pediatr. 2008, 4, 71–72. [Google Scholar]
- Naismith, D.; Braschi, A. The effect of low-dose potassium supplementation on blood pressure in apparently healthy volunteers. Br. J. Nutr. 2003, 90, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Nieder, R.; Benbi, D.K.; Reichl, F.X. Macro- and Secondary Elements and Their Role in Human Health. In Soil Components and Human Health; Nieder, R., Benbi, D.K., Reichl, F.X., Eds.; Springer: Dordrecht, Switzerland, 2018; pp. 257–315. [Google Scholar]
- McCormick, F.; Nwachukwu, B.; Provencher, M. Stress fractures in runners. Clin. Sports Med. 2012, 31, 291–306. [Google Scholar] [CrossRef] [PubMed]
- Nekrasov, V.I.; Skalny, A.V.; Oak, R.M. Role of microelements in increasing the functional reserves of the human body. Bull. Russ. Mil. Med. Acad. 2006, 1, 111–113. [Google Scholar]
- Mena, I. The role of manganese in human disease. Ann. Clin. Lab. Sci. 1974, 4, 487–491. [Google Scholar] [PubMed]
- Podobed, V.M. Zinc Carnosine: A new formula for gastroprotection and zinc deficiency. Med. News 2015, 2, 17. [Google Scholar]
- Halliulina, S.V. Clinical significance of zinc deficiency in a child’s body (literature review). Bull. Mod. Clin. Med. 2013, 6, 72–78. [Google Scholar]
- Wang, W.; Gou, T.; Liu, D. Method for Processing Meat Product Rich in Mineral Substances of Biological Active Calcium and the Like. 2012. Available online: https://patents.google.com/patent/CN102669715B/en (accessed on 28 August 2021).
- Boyle, E.A.E.; Addis, P.B.; Epley, R.J. Calcium Fortified, Reduced Fat Beef Emulsion Product. J. Food Sci. 1994, 59, 928–932. [Google Scholar] [CrossRef]
- Cáceres, E.; García, M.L.; Selgas, M.D. Design of a new cooked meat sausage enriched with calcium. Meat Sci. 2006, 73, 368–377. [Google Scholar] [CrossRef] [PubMed]
- Hemung, B.-O.; Yongsawatdigul, J.; Chin, K.B.; Limphirat, W.; Siritapetawee, J. Silver Carp Bone Powder as Natural Calcium for Fish Sausage. J. Aquat. Food Prod. Technol. 2018, 27, 305–315. [Google Scholar] [CrossRef]
- Ustinova, A.V.; Timoshenko, N.V.; Lyubina, N.V.; Soldatova, N.E.; Verkhosova, A.V. Homogenized Meat Product for Child Nutrition. Russian Federation Patent 2160546, 20 December 2000. Bul. 6. [Google Scholar]
- Daengprok, W.; Garnjanagoonchorn, W.; Mine, Y. Fermented pork sausage fortified with commercial or hen eggshell calcium lactate. Meat Sci. 2002, 62, 199–204. [Google Scholar] [CrossRef]
- Mehta, N.; Sharma, B.D.; Kumar, R.R.; Kumar, P.; Malav, O.P.; Verma, A.K. Fortification of low-fat chicken meat patties with calcium, vitamin E and vitamin C. Nutr. Food Sci. 2015, 45, 688–702. [Google Scholar] [CrossRef]
- Ha, E.; Zemel, M. Functional properties of whey, whey components, and essential amino acids: Mechanisms underlying health benefits for active people (review). J. Nutr. Biochem. 2003, 14, 251–258. [Google Scholar] [CrossRef]
- Ahluwalia, V.K.; Kumar, L.S.; Kumar, S. Chemistry of Natural Product: Amino Acids, Peptides, Proteins, and Enzymes; Ahluwalia, V.K., Kumar, L.S., Kumar, S., Eds.; CRC Press: Boca Raton, FL, USA, 2006; ISBN 1420059173. [Google Scholar]
- Rennie, M.; Tipton, K. Protein and amino acid metabolism during and after exercise and the effects of nutrition. Annu. Rev. Nutr. 2000, 20, 457–483. [Google Scholar] [CrossRef]

| Ingredients | Samples | |||||
|---|---|---|---|---|---|---|
| Control | MBP-5 | MBP-10 | MBP-15 | MBP-20 | MBP-25 | |
| Meat-bone paste | 0.00 | 5.00 | 10.0 | 15.0 | 20.0 | 25.0 |
| Beef liver | 55.0 | 50.0 | 45.0 | 40.0 | 35.0 | 30.0 |
| Beef brain | 10.0 | 10.0 | 10.0 | 10.0 | 10.0 | 10.0 |
| Pork back-fat | 30.0 | 30.0 | 30.0 | 30.0 | 30.0 | 30.0 |
| Sauteed onion | 3.10 | 3.10 | 3.10 | 3.10 | 3.10 | 3.10 |
| Salt | 1.30 | 1.30 | 1.30 | 1.30 | 1.30 | 1.30 |
| White sugar | 0.40 | 0.40 | 0.40 | 0.40 | 0.40 | 0.40 |
| Spices * | 0.20 | 0.20 | 0.20 | 0.20 | 0.20 | 0.20 |
| Total | 100 | 100 | 100 | 100 | 100 | 100 |
| Time (min) | Volume of Eluent (%) | ||
|---|---|---|---|
| Eluent A | Eluent B | Eluent C | |
| 0.01 | 96.0 | 4.00 | 0.00 |
| 10.0 | 37.0 | 11.0 | 52.0 |
| 13.0 | 88.5 | 11.5 | 0.00 |
| 21.0 | 80.0 | 20.0 | 0.00 |
| 22.0 | 58.0 | 22.0 | 20.0 |
| 24.0 | 0.00 | 24.0 | 76.0 |
| 32.0 | 0.00 | 33.5 | 66.5 |
| 32.01 | 20.0 | 80.0 | 0.00 |
| 35.3 | 20.0 | 80.0 | 0.00 |
| 35.31 | 97.0 | 3.00 | 0.00 |
| 41.3 | 97.0 | 3.00 | 0.00 |
| Chemical Composition | |
|---|---|
| Moisture | 69.7 ± 1.57 |
| Protein | 13.6 ± 0.32 |
| Fat | 5.22 ± 0.14 |
| Ash | 11.4 ± 0.36 |
| Caloric value (Kcal/100 g) | 102 |
| Minerals | |
| Calcium | 3080 ± 72.7 |
| Potassium | 115 ± 1.86 |
| Magnesium | 2120 ± 43.5 |
| Sodium | 390 ± 14.8 |
| Phosphorous | 2564 ± 50.4 |
| Copper | 1.33 ± 0.04 |
| Iron | 7.30 ± 0.14 |
| Manganese | 0.10 ± 0.002 |
| Zinc | 2.15 ± 0.08 |
| Parameter 1 | Control | Pâté Reformulated with Meat-Bone Paste | p Value | ||||
|---|---|---|---|---|---|---|---|
| MBP-5 | MBP-10 | MBP-15 | MBP-20 | MBP-25 | |||
| Moisture | 56.9 ± 1.27 | 56.8 ± 1.59 | 56.7 ± 1.27 | 57.0 ± 0.93 | 56.5 ± 1.71 | 56.4 ± 1.58 | >0.50 |
| Protein | 11.3 ± 0.46 | 11.1 ± 0.21 | 10.9 ± 0.21 | 10.7 ± 0.30 | 10.5 ± 0.26 | 10.2 ± 0.23 | >0.25 |
| Fat | 27.7 ± 0.80 | 27.7 ± 0.71 | 27.8 ± 0.56 | 27.2 ± 0.98 | 28.0 ± 0.78 | 28.0 ± 0.73 | >0.50 |
| Ash | 0.92 ± 0.02 f | 1.42 ± 0.04 e | 1.92 ± 0.04 d | 2.36 ± 0.07 c | 2.92 ± 0.08 b | 3.42 ± 0.08 a | <0.05 |
| Carbohydrates | 3.25 ± 0.07 a | 2.98 ± 0.07 a | 2.72 ± 0.05 b | 2.68 ± 0.07 b | 2.19 ± 0.06 c | 1.92 ± 0.05 c | <0.05 |
| Caloric value | 307 | 306 | 305 | 299 | 302 | 301 | |
| Mineral | Control | Pâté Reformulated with Meat-Bone Paste | p Value | ||||
|---|---|---|---|---|---|---|---|
| MBP-5 | MBP-10 | MBP-15 | MBP-20 | MBP-25 | |||
| Calcium | 11.36 ± 0.19 f | 165 ± 4.21 e | 319 ± 7.81 d | 500 ± 18.7 c | 626 ± 10.9 b | 780 ± 14.1 a | <0.001 |
| Potassium | 211 ± 7.30 a | 201 ± 4.82 a | 191 ± 1.81 b | 181 ± 5.91 b | 171 ± 6.04 b,c | 161 ± 4.65 c | <0.01 |
| Magnesium | 12.6 ± 0.27 e | 17.7 ± 0.39 d | 22.7 ± 0.64 d | 25.0 ± 0.76 c | 32.7 ± 0.73 b | 37.7 ± 0.74 a | <0.001 |
| Sodium | 61.5 ± 2.08 f | 77.4 ± 1.52 e | 93.2 ± 1.76 d | 109 ± 3.17 c | 125 ± 2.50 b | 141 ± 4.39 a | <0.001 |
| Phosphorous | 275 ± 7.37 f | 382 ± 12.6 e | 489 ± 8.56 d | 596 ± 10.8 c | 703 ± 11.7 b | 810 ± 18.4 a | <0.001 |
| Copper | 6.99 ± 0.21 a | 6.42 ± 0.15 a | 5.86 ± 0.19 b | 5.29 ± 0.16 b | 4.72 ± 0.11 c | 4.16 ± 0.13 c | <0.01 |
| Iron | 4.19 ±0.11 b | 4.20 ±0.10 b | 4.32 ±0.12 b | 4.59 ±0.09 b | 4.64 ±0.16 a | 4.86 ±0.12 a | <0.01 |
| Manganese | 0.20 ± 0.01 a | 0.19 ± 0.01 a | 0.17 ± 0.01 a | 0.16 ± 0.01 b | 0.15 ± 0.01 b | 0.14 ± 0.01 b | <0.01 |
| Zinc | 3.09 ± 0.08 a | 2.95 ± 0.08 a | 2.82 ± 0.09 a | 2.68 ± 0.05 b | 2.54 ± 0.07 b | 2.40 ± 0.06 b | <0.01 |
| Mineral | Recommended 1 | Maximum 2 | Recommended for Children 3 |
|---|---|---|---|
| Calcium | 800–1200 | 2500 | 400–1200 |
| Magnesium | 300–400 | - | 55–400 |
| Potassium | 1500–2500 | - | 400–2500 |
| Sodium | 3000–5000 | - | 200–1300 |
| Phosphorous | 550–1400 | - | 300–1200 |
| Iron | 8–10 (for men) 15–20 (for women) | - | 4–18 |
| Zinc | 11–15 (for men) 10–12 (for women) | 25 | 3–12 |
| Copper | 0.9–2.3 | 5 | 0.5–1.0 |
| Manganese | 2–5 | 5 | 2 |
| Amino Acid | Control | Pâté Reformulated with Meat-Bone Paste | p Value | ||||
|---|---|---|---|---|---|---|---|
| MBP-5 | MBP-10 | MBP-15 | MBP-20 | MBP-25 | |||
| Non-essential | 6080 ± 109 | 4995 ± 89.9 | 5083 ± 91.5 | 4586 ± 82.5 | 4088 ± 73.6 | 3590 ± 64.4 | |
| Alanine | 718 ± 7.71 a | 583 ± 8.72 b | 602 ± 10.1 b | 544 ± 6.41 b | 485 ± 5.15 c | 427 ± 5.29 c | <0.001 |
| Aspartic acid | 1177 ± 24.9 a | 967 ± 18.2 b | 984 ± 11.8 b | 888 ± 14.6 c | 792 ± 10.5 d | 695 ± 8.24 e | <0.001 |
| Glycine | 702 ± 9.12 a | 583 ± 7.38 b | 586 ± 7.65 b | 528 ± 6.73 b | 469 ± 5.46 c | 411 ± 4.70 d | <0.001 |
| Glutamic acid | 1585 ± 19.6 a | 1312 ± 16.8 b | 1324 ± 19.3 b | 1193 ± 12.6 c | 1063 ± 12.3 c | 932 ± 14.7 d | <0.001 |
| Proline | 603 ± 11.2 a | 481 ± 7.30 b | 506 ± 7.15 b | 458 ± 6.51 b | 410 ± 4.36 c | 362 ± 4.62 d | <0.001 |
| Serine | 555. ± 10.5 a | 454 ± 6.55 b | 465 ± 9.40 b | 419 ± 8.36 b | 374 ± 5.66 c | 329 ± 6.72 d | <0.001 |
| Tyrosine | 483 ± 9.20 a | 405 ± 5.88 b | 402 ± 5.63 b | 362 ± 3.92 c | 322 ± 4.10 c | 281 ± 4.38 d | <0.001 |
| Cystine | 232 ± 3.50 a | 188 ± 3.37 b | 194 ± 2.57 b | 175 ± 2.43 b | 157 ± 2.49 c | 138 ± 1.55 d | <0.001 |
| Oxyproline | 25.3 ± 0.37 a | 23.0 ± 0.38 b | 20.7 ± 0.32 b | 18.4 ± 0.28 c | 16.1 ± 0.30 d | 13.8 ± 0.27 e | <0.02 |
| Essential | 6280 ± 113 | 5610 ± 100 | 6070 ± 109 | 5965 ± 107 | 5860 ± 105 | 5756 ± 103 | |
| Arginine | 741 ± 14.9 a | 622 ± 6.43 b | 617 ± 8.08 b | 555 ± 8.42 c | 493 ± 9.02 d | 431 ± 6.22 e | <0.001 |
| Histidine | 409 ± 5.03 b | 356 ± 4.26 b | 427 ± 6.67 b | 436 ± 6.87 b | 445 ± 7.82 a | 454 ± 6.57 a | <0.02 |
| Valine | 754 ± 13.1 | 686 ± 7.15 | 738 ± 112 | 730 ± 9.76 | 722 ± 7.69 | 714 ± 5.38 | <0.02 |
| Isoleucine | 588 ± 11.5 | 535 ± 8.08 | 592 ± 10.9 | 593 ± 7.15 | 595 ± 7.02 | 597 ± 6.89 | >0.50 |
| Leucine | 1149 ± 22.8 | 1042 ± 18.1 | 1128 ± 18.9 | 1118 ± 15.9 | 1107 ± 16.1 | 1097 ± 13.5 | >0.25 |
| Lysine | 969 ± 9.04 | 898 ± 16.4 | 994 ± 12.8 | 1006 ± 13.2 | 1018 ± 16.7 | 1030 ± 13.4 | >0.25 |
| Methionine | 322 ± 5.05 a | 297 ± 2.65 b | 318 ± 4.71 a | 316 ± 4.58 a | 313 ± 5.15 a | 311 ± 4.89 a | <0.01 |
| Threonine | 533 ± 6.07 a | 460 ± 5.44 b | 496 ± 8.46 a | 478 ± 6.20 b | 459 ± 8.25 b | 441 ± 6.89 b | <0.02 |
| Tryptophan | 161 ± 2.25 a | 132 ± 1.68 b | 135 ± 2.32 b | 122 ± 1.77 b | 108 ± 1.70 b | 95.2 ± 1.23 c | <0.001 |
| Phenylalanine | 654 ± 10.7 a | 583 ± 9.12 b | 627 ± 7.04 a | 613 ± 13.0 a | 599 ± 8.32 a | 586 ± 8.76 b | <0.05 |
| Total Amino Acids | 12360 ± 223 | 10605 ± 191 | 11154 ± 201 | 10551 ± 190 | 9948 ± 179 | 9345 ± 168 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yessimbekov, Z.; Kakimov, A.; Caporaso, N.; Suychinov, A.; Kabdylzhar, B.; Shariati, M.A.; Baikadamova, A.; Domínguez, R.; Lorenzo, J.M. Use of Meat-Bone Paste to Develop Calcium-Enriched Liver Pâté. Foods 2021, 10, 2042. https://doi.org/10.3390/foods10092042
Yessimbekov Z, Kakimov A, Caporaso N, Suychinov A, Kabdylzhar B, Shariati MA, Baikadamova A, Domínguez R, Lorenzo JM. Use of Meat-Bone Paste to Develop Calcium-Enriched Liver Pâté. Foods. 2021; 10(9):2042. https://doi.org/10.3390/foods10092042
Chicago/Turabian StyleYessimbekov, Zhanibek, Aitbek Kakimov, Nicola Caporaso, Anuarbek Suychinov, Baktybala Kabdylzhar, Mohammad Ali Shariati, Assemgul Baikadamova, Rubén Domínguez, and José M. Lorenzo. 2021. "Use of Meat-Bone Paste to Develop Calcium-Enriched Liver Pâté" Foods 10, no. 9: 2042. https://doi.org/10.3390/foods10092042
APA StyleYessimbekov, Z., Kakimov, A., Caporaso, N., Suychinov, A., Kabdylzhar, B., Shariati, M. A., Baikadamova, A., Domínguez, R., & Lorenzo, J. M. (2021). Use of Meat-Bone Paste to Develop Calcium-Enriched Liver Pâté. Foods, 10(9), 2042. https://doi.org/10.3390/foods10092042










