Effects of Vacuum Frying on the Preparation of Ready-to-Heat Batter-Fried and Sauced Chub Mackerel (Scomber japonicus)
Abstract
1. Introduction
2. Materials and Methods
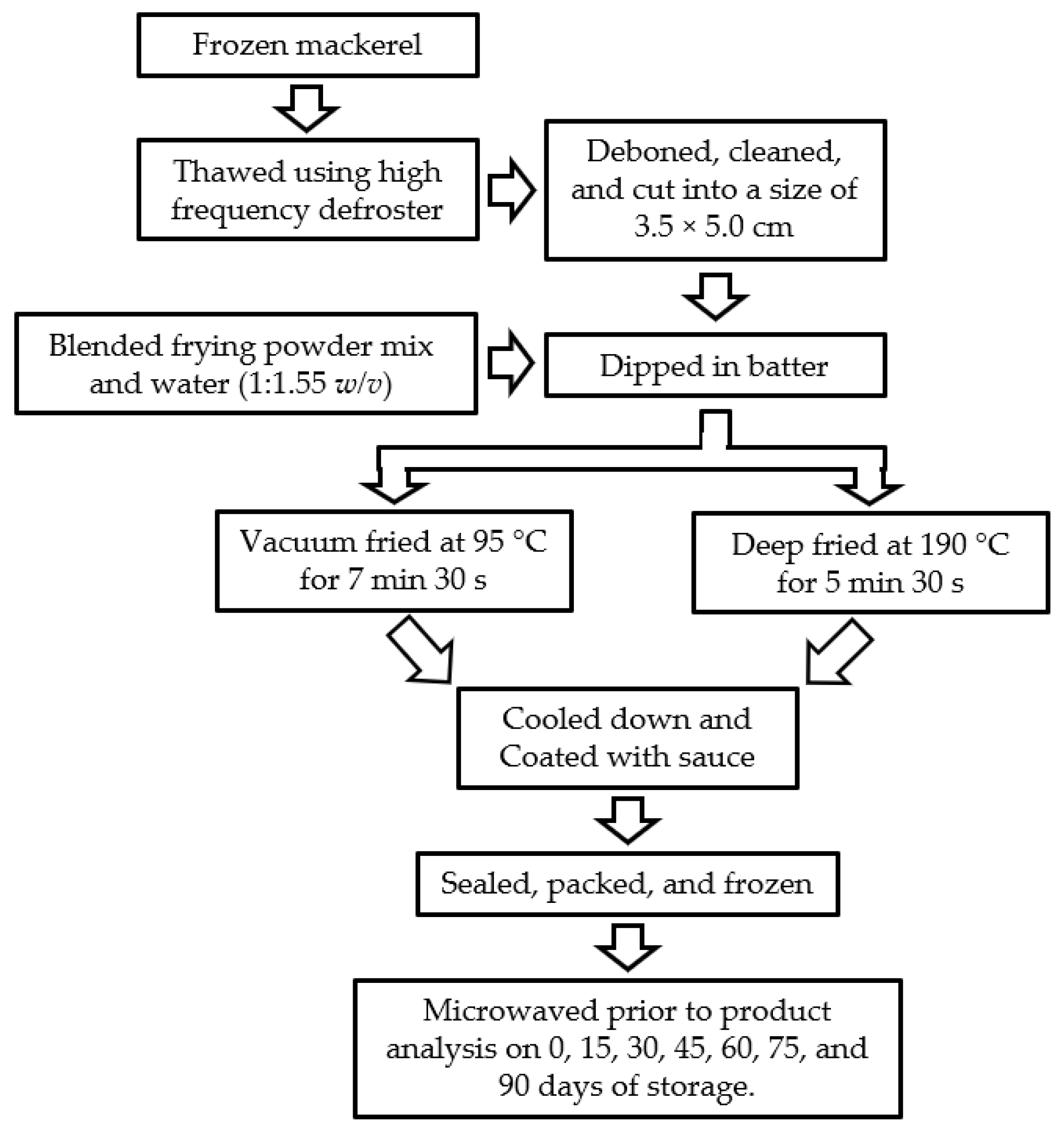
2.1. Preparation of Mackerel Sample
2.2. Preparation of Batter
2.3. Optimization of Frying Conditions
2.4. Analysis of Sensory Properties
2.5. Analysis of Chemical Properties
2.6. Analysis of Microbial Properties
2.7. Analysis of Nutritional Composition
2.8. Prediction of Shelf Life
2.9. Statistical Analysis
3. Results and Discussion
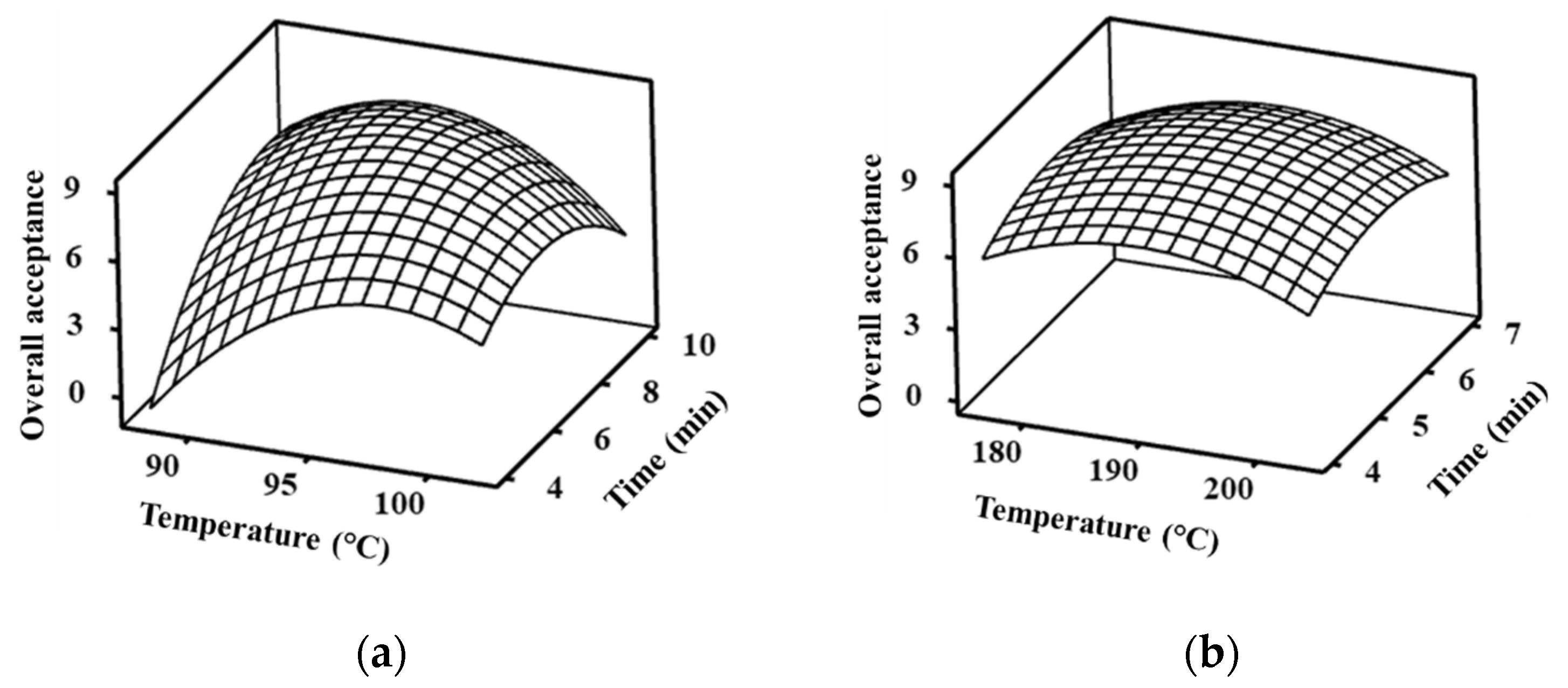
3.1. Optimum Conditions of Vacuum and Deep Frying Treatments
3.2. Sensory Properties of Processed Chub Mackerel
3.3. Chemical and Microbial Properties of Raw and Processed Chub Mackerel
3.4. Nutritional Composition of Raw and Processed Chub Mackerel
3.5. Shelf Life Prediction of Vacuum Batter-Fried Chub Mackerel with Sauce
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lee, D.; Son, S.; Kim, W.; Park, J.; Joo, H.; Lee, S. Spatio-Temporal Variability of the Habitat Suitability Index for Chub Mackerel (Scomber japonicus) in the East/Japan Sea and the South Sea of South Korea. Remote Sens. 2018, 10, 938. [Google Scholar] [CrossRef]
- Shulgina, L.V.; Davletshina, T.A.; Pavlovsky, A.M.; Solodova, E.A.; Pavel, K.G. Composition of lipids and fatty acids in muscle tissue of chub mackerel Scomber japonicus. Izvestiya Tinro 2019, 196, 193–203. [Google Scholar] [CrossRef]
- Bae, J.H.; Yoon, S.H.; Lim, S.Y. A comparison of the biochemical characteristics of different anatomical regions of chub (Scomber japonicus) and blue mackerel (Scomber australasicus) muscles. Korean J. Fish. Aquat. Sci. 2010, 43, 6–11. [Google Scholar]
- Rana, M.M.; Mohibbullah, M.; Won, N.E.; Baten, M.A.; Sohn, J.H.; Kim, J.-S.; Choi, J.-S. Improved Hot Smoke Processing of Chub Mackerel (Scomber japonicus) Promotes Sensorial, Physicochemical and Microbiological Characteristics. Appl. Sci. 2021, 11, 2629. [Google Scholar] [CrossRef]
- Tirtawijaya, G.; Kim, S.R.; Cho, W.H.; Sohn, J.H.; Kim, J.-S.; Choi, J.-S. Development of a Home Meal Replacement Product Containing Braised Mackerel (Scomber japonicus) with Radish (Raphanus sativus). Foods 2021, 10, 1135. [Google Scholar] [CrossRef] [PubMed]
- Vieira, E.F.; Soares, C.; Machado, S.; Oliva-Teles, M.T.; Correia, M.; João Ramalhosa, M.; Carvalho, A.; Domingues, V.F.; Antunes, F.; Morais, S.; et al. Development of New Canned Chub Mackerel Products Incorporating Edible Seaweeds—Influence on the Minerals and Trace Elements Composition. Molecules 2020, 25, 1133. [Google Scholar] [CrossRef]
- Oduro, F.A.; Choi, N.D.; Ryu, H.S. Effects of cooking conditions on the protein quality of chub Mackerel Scomber japonicus. Fish. Aquat. Sci. 2011, 14, 257–265. [Google Scholar] [CrossRef]
- Bordin, K.; Kunitake, M.T.; Aracava, K.K.; Trindade, C.S.F. Changes in food caused by deep fat frying—A review. Arch. Latinoam. Nutr. 2013, 63, 5–13. [Google Scholar] [PubMed]
- Hosseini, H.; Ghorbani, M.; Meshginfar, N.; Mahoonak, A.S. A Review on Frying: Procedure, Fat, Deterioration Progress and Health Hazards. J. Am. Oil Chem’ Soc. 2016, 93, 445–466. [Google Scholar] [CrossRef]
- Pourshamsian, K.; Ghomi, M.R.; Nikoo, M. Fatty Acid and Proximate Composition of Farmed Great Sturgeon (Huso huso) Affected by Thawing Methods, Frying Oils and Chill Storage. Adv. Stud. Biol. 2012, 4, 67–76. [Google Scholar]
- Chaula, D.; Laswai, H.; Chove, B.; Dalsgaard, A.; Mdegela, R.; Hyldig, G. Fatty Acid Profiles and Lipid Oxidation Status of Sun Dried, Deep Fried, and Smoked Sardine (Rastrineobola argentea) from Lake Victoria, Tanzania. J. Aquat. Food Prod. Technol. 2019, 28, 165–176. [Google Scholar] [CrossRef]
- Diamante, L.M.; Shi, S.; Hellmann, A.; Busch, J. Vacuum frying foods: Products, process and optimization. Int. Food Res. J. 2015, 22, 15–22. [Google Scholar]
- Shyu, S.L.; Hau, L.B.; Hwang, L.S. Effect of vacuum frying on the oxidative stability of oils. J. Am. Oil Chem’ Soc. 1998, 75, 1393–1398. [Google Scholar] [CrossRef]
- Albertos, I.; Martin-Diana, A.B.; Jaime, I.; Diez, A.M.; Rico, D. Protective role of vacuum vs. atmospheric frying on PUFA balance and lipid oxidation. Innov. Food Sci. Emerg. 2016, 36, 336–342. [Google Scholar] [CrossRef]
- Yamsaengsung, R.; Yaeed, S.; Ophithakorn, T. Vacuum frying of fish tofu and effect on oil quality usage life. J. Food Process. Eng. 2017, 40, e12587. [Google Scholar] [CrossRef]
- Pan, G.; Ji, H.; Liu, S.; He, X. Vacuum frying of breaded shrimps. LWT Food Sci. Technol. 2015, 62, 734–739. [Google Scholar] [CrossRef]
- Castillo, P.M.M.; Díaz, L.T.; Díaz, S.T.; Correa, D.A.; Gómez, R.J.M. Mass Transfer during Atmospheric and Vacuum Frying of Chorizo. Int. J. Food Sci. 2021, 2021, 9142412. [Google Scholar] [CrossRef]
- Nurdiani, R.; Prihanto, A.A.; Jatmiko, Y.D.; Muyasyaroh, H.; Azizah, M.N.; Priyatmoko, F.; Qodim, A.F. Effect of vacuum frying on the proximate quality of crispy catfish (Clarias gariepinus). Proc. ICoFMR 2020, 1, 17–20. [Google Scholar]
- Li, M.; Wang, W.; Fang, W.; Li, Y. Inhibitory effects of chitosan coating combined with organic acids on Listeria monocytogenes in refrigerated ready-to-eat shrimps. J. Food Prot. 2013, 76, 1377–1383. [Google Scholar] [CrossRef] [PubMed]
- Tirtawijaya, G.; Park, Y.; Won, N.E.; Kim, H.; An, J.H.; Jeon, J.H.; Park, S.M.; Yoon, S.J.; Sohn, J.H.; Kim, J.S.; et al. Effect of steaming and hot smoking treatment combination on the quality characteristics of hagfish (Myxine glutinosa). J. Food Process. Preserv. 2020, 44, e14694. [Google Scholar] [CrossRef]
- Association of Official Analytical Chemist. Official Methods of Analysis; Horwitz, W., Ed.; AOAC International: Washington, DC, USA, 2000. [Google Scholar]
- Czech, A.; Grela, E.R.; Ognik, K. Effect of frying on nutrients content and fatty acid composition of muscles of selected freezing seafoods. J. Food Nutr. Res. 2015, 3, 9–14. [Google Scholar] [CrossRef]
- Lee, D.H.; Kang, D.M.; Park, S.K.; Jeong, M.C.; Kang, M.G.; Jo, D.M.; Lee, J.H.; Lee, D.E.; Sim, Y.A.; Jeong, G.J.; et al. Shelf-life extension of Raw Oyster Crassostrea gigas by depuration process. Korean J. Fish. Aquat. Sci. 2020, 53, 842–850. [Google Scholar] [CrossRef]
- Abraha, B.; Admassu, H.; Mahmud, A.; Tsighe, N.; Shui, X.W.; Fang, Y. Effect of processing methods on nutritional and physico-chemical composition of fish: A review. MOJ Food Process. Technol. 2018, 6, 376–382. [Google Scholar] [CrossRef]
- Negara, B.F.S.P.; Lee, M.-J.; Tirtawijaya, G.; Cho, W.-H.; Sohn, J.-H.; Kim, J.-S.; Choi, J.-S. Application of Deep, Vacuum, and Air Frying Methods to Fry Chub Mackerel (Scomber japonicus). Processes 2021, 9, 1225. [Google Scholar] [CrossRef]
- Vongsawasdi, P.; Nopharatana, M.; Srisuwatchree, W.; Pasukcharoenying, S.; Wongkitcharoen, N. Using modified starch to decrease the oil absorption in fried battered chicken. Asian J. Food Agro-Ind. 2008, 1, 174–183. [Google Scholar]
- Da Silva, P.F.; Moreira, R.G. Vacuum frying of high-quality fruit and vegetable-based snacks. LWT Food Sci. Technol. 2008, 41, 1758–1767. [Google Scholar] [CrossRef]
- Shyu, S.L.; Hwang, L.S. Effects of processing conditions on the quality of vacuum fried apple chips. Food Res. Int. 2001, 34, 133–142. [Google Scholar] [CrossRef]
- Oke, E.K.; Idowu, M.A.; Sobukola, O.P.; Adeyeye, S.A.O.; Akinsola, A.O. Frying of Food: A Critical Review. J. Culin. Sci. Technol. 2017, 16, 107–127. [Google Scholar] [CrossRef]
- Babu, U.S.J.; Rao, B.M.; Khasim, D.I.; Nair, K.G.R. Biochemical and Microbiological quality of formic acid silage and Lactobacillus fermented silage. Fish. Technol. 2005, 42, 163–170. [Google Scholar]
- Li, H.; Chen, Q.; Zhao, J.; Wu, M. Nondestructive detection of total volatile basic nitrogen (TVB-N) content in pork meat by integrating hyperspectral imaging and colorimetric sensor combined with a nonlinear data fusion. LWT Food Sci. Technol. 2015, 63, 268–274. [Google Scholar] [CrossRef]
- Wu, T.H.; Bechtel, P.J. Ammonia, Dimethylamine, Trimethylamine, and Trimethylamine Oxide from raw and processed fish by-products. J. Aquat. Food Prod. Technol. 2008, 17, 27–38. [Google Scholar] [CrossRef]
- Jiang, Q.; Han, J.; Gao, P.; Yu, L.; Xu, Y.; Xia, W. Effect of heating temperature and duration on the texture and protein composition of Bighead Carp (Aristichthys nobilis) muscle. Int. J. Food Prop. 2018, 21, 2110–2120. [Google Scholar] [CrossRef]
- Sohn, M.; Ho, C.T. Ammonia Generation during Thermal Degradation of Amino Acids. J. Agric. Food Chem. 1995, 43, 3001–3003. [Google Scholar] [CrossRef]
- Domínguez, R.; Gómez, M.; Fonseca, S.; Lorenzo, J.M. Influence of thermal treatment on formation of volatile compounds, cooking loss and lipid oxidation in foal meat. LWT Food Sci. Technol. 2014, 58, 439–445. [Google Scholar] [CrossRef]
- Halamickova, A.; Malota, L. Muscle thiobarbituric acid reactive substance of the Atlantic herring (Clupea harengus) in marinades collected in the market network. Acta Vet. Brno 2010, 79, 329–333. [Google Scholar] [CrossRef][Green Version]
- Ruiz-Capillas, C.; Moral, A. Correlation between biochemical and sensory quality indices in hake stored in ice. Food Res. Int. 2001, 34, 441–447. [Google Scholar] [CrossRef]
- Tavares, S.W.P.; Dong, S.; Jin, W.; Yang, Y.; Han, K.; Zha, F.; Zhao, Y.; Zeng, M. Effect of different cooking conditions on the profiles of Maillard reaction products and nutrient composition of hairtail (Thichiurus lepturus) fillets. Food Res. Int. 2018, 103, 390–397. [Google Scholar] [CrossRef]
- Hu, L.; Ren, S.; Shen, Q.; Chen, J.; Ye, X.; Ling, J. Proteomic study of the effect of different cooking methods on protein oxidation in fish fillets. R. Soc. Chem. 2017, 7, 27496–27505. [Google Scholar] [CrossRef]
- Okolie, N.P.; Okugbo, O.T. A comparative study of malondialdehyde contents of some meat and fish samples processed by different methods. J. Pharm. Sci. Innov. 2013, 2, 26–29. [Google Scholar] [CrossRef]
- Rosnes, J.T.; Skåra, T.; Skipnes, D. Recent Advances in Minimal Heat Processing of Fish: Effects on Microbiological Activity and Safety. Food Bioprocess. Technol. 2011, 4, 833–848. [Google Scholar] [CrossRef]
- Yildiz, P.O. Effect of essential oils and packaging on hot smoked rainbow trout during storage. J. Food Process. Preserv. 2015, 39, 806–815. [Google Scholar] [CrossRef]
- International Commission of Microbiological Specification for Food—ICMSF. Microorganisms in Food 2. Sampling for Microbiological Analysis: Principles and Specific Applications, 2nd ed.; University of Toronto Press: Toronto, ON, Canada, 1986. [Google Scholar]
- Moon, S.K.; Kang, J.Y.; Kim, I.S.; Jeong, B.Y. Changes in proximate composition and lipid components in chub mackerel Scomber japonicus and Japanese jack mackerel Trachurus japonicus with various cooking methods. Korean J. Fish. Aquat. Sci. 2013, 46, 708–716. [Google Scholar] [CrossRef]
- Mbassi, J.E.G.; Tsafack, A.L.S.; Maboune, A.S.; Eyenga, E.F.; Sophie, N.E.; Bongse, K.P. Quality evaluation of local Cameroonian mackerel (Scomber scombrus) processed by different methods. Int. J. Food Sci. Nutr. 2019, 4, 162–167. [Google Scholar]
- Rahman, M.M.; Zamri, M.; Fadilla, N. Effects of deep frying on proximate composition and micronutrient of Indian mackerel (Rastrelliger kanagurta), eel (Monopterus albus), and cockle (Anadara granosa). Pak. J. Biol. Sci. 2012, 15, 589–594. [Google Scholar] [CrossRef][Green Version]
- Andrés-Bello, A.; García-Segovia, P.; Martínez-Monzó, J. Vacuum frying process of gilthead sea bream (Sparus aurata) fillets. Innov. Food Sci. Emerg. Technol. 2010, 11, 630–636. [Google Scholar] [CrossRef]
- Smida, M.A.B.; Bolje, A.; Ouerhani, A.; Barhoumi, M.; Mejri, H.; El Cafsi, M.; Fehri-Bedoui, R. Effects of Drying on the Biochemical Composition of Atherina boyeri from the Tunisian Coast. Food Nutr. Sci. 2014, 5, 1399–1407. [Google Scholar] [CrossRef]
- Erkan, N.; Ozden, O.; Selcuk, A. Effect of Frying, Grilling, and Steaming on Amino Acid Composition of Marine Fishes. J. Med. Food 2010, 13, 1524–1531. [Google Scholar] [CrossRef] [PubMed]
- Oluwaniyi, O.O.; Dosumu, O.O.; Awolola, G.V. Effect of local processing methods (boiling, frying and roasting) on the amino acid composition of four marine fishes commonly consumed in Nigeria. Food Chem. 2010, 123, 1000–1006. [Google Scholar] [CrossRef]
- Nurjanah, N.; Nurilmala, M.; Hidayat, T.; Ginanjar, T.M.G. Amino acid and taurine changes of Indian Mackarel due to frying process. Int. J. Chem. Biomole Sci. 2015, 1, 163–166. [Google Scholar]
- Gaurat, P.V.; Koli, J.M.; Bhosale, B.P.; Mulye, V.B.; Sonavane, A.E.; Shingare, P.E.; Swami, S.B.; Gitte, M.J. Effect of different cooking methods on physicochemical and nutritional properties of catla (Catla catla). J. Exp. Zool. 2020, 23, 847–853. [Google Scholar]
- Sobral, M.M.; Cunha, S.C.; Faria, M.A.; Ferreira, I.M.P.L.V.O. Domestic cooking of muscle foods: Impact on composition of nutrients and contaminants. Compr. Rev. Food Sci. Food Saf. 2018, 17, 309–333. [Google Scholar] [CrossRef] [PubMed]
- Savva, S.C.; Kafatos, A. Vegetable oils: Dietary importance. Encyclo. Food Health 2016, 365–372. [Google Scholar] [CrossRef]
- Farahmandfar, R.; Asnaashari, M.; Sayyad, R. Comparison antioxidant activity ofTarom Mahalirice bran extracted from different extraction methods and its effect on canola oil stabilization. Iran. J. Food Sci. Technol. 2015, 52, 6385–6394. [Google Scholar] [CrossRef] [PubMed]
- Kazuo, M. Prevention of fish oil oxidation. J. Oleo Sci. 2019, 68, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Gebauer, S.; Psota, T.; Harris, W.; Kris-Etherton, P. Fatty acid dietary recommendations and food sources to achieve essential and cardiovascular benefits. Am. J. Clin. Nutr. 2006, 83, 1526–1535. [Google Scholar] [CrossRef] [PubMed]
- Moussa, E.R.W.H.; Shereen, A.N.; Manal, A.; Mehanni, A.H.E.; Rasha, A.E. Nutritional value and fatty acid composition of household cooking on fish fatty acids profile using atherogenicity and thrombogenicity indices. J. Food Chem. Nutr. 2014, 2, 27–41. [Google Scholar]
- Zula, A.T.; Desta, D.T. Fatty acid-related health lipid index of raw and fried nile tilapia (Oreochromis niloticus) fish muscle. J. Food Qual. 2021, 2021, 6676528. [Google Scholar] [CrossRef]
- Simopoulos, A.P. The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed. Pharmacother. 2002, 56, 365–379. [Google Scholar] [CrossRef]
- van Boekel, M.A.J.S. Kinetic modeling of food quality: A critical review. Compr. Rev. Food Sci. Food Saf. 2008, 7, 144–158. [Google Scholar] [CrossRef]
| Ingredients | (%) |
|---|---|
| Corn syrup | 40.0 |
| Sugar | 6.0 |
| Tomato ketchup | 10.0 |
| Chili powder | 1.2 |
| Spicy chili powder | 2.5 |
| Chopped garlic | 4.0 |
| Purified water | 3.5 |
| Soy sauce | 7.0 |
| Apple juice | 3.0 |
| Oligosaccharide | 12.0 |
| Starch | 0.2 |
| Oyster sauce | 3.0 |
| Apple cider vinegar | 1.0 |
| Mayonnaise | 0.7 |
| Brewed vinegar | 1.0 |
| Cooking wine | 1.8 |
| Lemon juice | 2.0 |
| Monosodium glutamate | 0.4 |
| Pepper powder | 0.4 |
| Citric acid | 0.3 |
| Frying Method | Std. Order | Frying Condition | OA (Score) | VBN (mg%) | TBARS (mg MDA/kg) | |
|---|---|---|---|---|---|---|
| Temperature (°C) | Time (min) | |||||
| VBF | 1 | 90 | 5.0 | 4.13 | 10.53 | 1.40 |
| 2 | 100 | 5.0 | 6.52 | 11.94 | 1.35 | |
| 3 | 90 | 9.0 | 7.25 | 12.99 | 1.43 | |
| 4 | 100 | 9.0 | 6.03 | 11.58 | 1.52 | |
| 5 | 88 | 7.0 | 4.02 | 12.29 | 1.27 | |
| 6 | 102 | 7.0 | 5.90 | 11.58 | 1.73 | |
| 7 | 95 | 4.0 | 4.51 | 13.69 | 1.28 | |
| 8 | 95 | 10.0 | 7.25 | 11.23 | 1.74 | |
| 9 | 95 | 7.0 | 8.39 | 11.94 | 1.43 | |
| 10 | 95 | 7.0 | 8.70 | 11.58 | 1.41 | |
| 11 | 95 | 7.0 | 8.51 | 12.29 | 1.17 | |
| DBF | 1 | 180 | 4.5 | 7.42 | 12.64 | 1.59 |
| 2 | 200 | 4.5 | 7.26 | 12.99 | 1.40 | |
| 3 | 180 | 6.5 | 6.70 | 13.34 | 1.66 | |
| 4 | 200 | 6.5 | 6.60 | 12.29 | 1.71 | |
| 5 | 176 | 5.5 | 7.09 | 13.34 | 1.31 | |
| 6 | 204 | 5.5 | 6.98 | 13.69 | 1.76 | |
| 7 | 190 | 4.0 | 7.39 | 14.39 | 1.38 | |
| 8 | 190 | 7.0 | 7.56 | 12.64 | 1.64 | |
| 9 | 190 | 5.5 | 8.38 | 12.29 | 1.32 | |
| 10 | 190 | 5.5 | 8.20 | 12.99 | 1.57 | |
| 11 | 190 | 5.5 | 8.13 | 11.94 | 1.59 | |
| Method | Variable | p-Value | |||
|---|---|---|---|---|---|
| Model | Linear | Square | 2-Way Interaction | ||
| VBF | OA | 0.001 | 0.000 | 0.000 | 0.008 |
| VBN | 0.707 | 0.485 | 0.846 | 0.200 | |
| TBARS | 0.311 | 0.591 | 0.509 | 0.673 | |
| DBF | OA | 0.037 | 0.009 | 0.023 | 0.251 |
| VBN | 0.109 | 0.149 | 0.076 | 0.110 | |
| TBARS | 0.930 | 0.814 | 0.975 | 0.649 | |
| Response | Predictive Model | R2 | Lack of Fit (p-Value) |
|---|---|---|---|
| VBF | −693.3 + 13.71X1 + 12.79X2 − 0.06831X12 − 0.2731X22 − 0.0903X1X2 | 97.00% | 0.081 |
| DBF | −250.6 + 2.604X1 + 4.37X2 − 0.00687X12 − 0.394X22 | 80.55% | 0.539 |
| Sensory Property | VBF | VBFS | DBF | DBFS |
|---|---|---|---|---|
| Appearance | 8.68 ± 0.18 a | 9.00 ± 0.00 a | 8.20 ± 0.20 b | 9.00 ± 0.00 a |
| Aroma | 8.63 ± 0.19 ab | 8.77 ± 0.15 a | 8.17 ± 0.15 b | 8.73 ± 0.12 a |
| Texture | 8.52 ± 0.45 a | 8.80 ± 0.12 a | 8.48 ± 0.08 a | 8.67 ± 0.09 a |
| Taste | 8.70 ± 0.20 a | 8.90 ± 0.10 a | 8.15 ± 0.13 b | 8.73 ± 0.15 a |
| Overall Acceptance | 8.63 ± 0.17 a | 8.92 ± 0.08 a | 8.25 ± 0.03 b | 8.85 ± 0.03 a |
| Parameters | Raw | VBF | VBFS | DBF | DBFS |
|---|---|---|---|---|---|
| VBN (mg%) | 7.84 ± 0.42 a | 11.94 ± 0.20 b | 11.13 ± 0.48 b | 12.40 ± 0.31 b | 11.74 ± 0.17 b |
| TBARS (mg MDA/kg) | 1.02 ± 0.29 a | 1.34 ± 0.08 b | 1.28 ± 0.04 ab | 1.49 ± 0.09 b | 1.38 ± 0.02 b |
| TBC (log CFU/g) | 3.20 ± 0.02 a | 2.72 ± 0.10 b | 2.19 ± 0.05 c | 2.77 ± 0.05 b | 2.14 ± 0.07 c |
| Composition | Raw | VBF | VBFS | DBF | DBFS |
|---|---|---|---|---|---|
| Calories (kcal) | 196.99 | 280.67 | 355.86 | 265.48 | 348.14 |
| Sodium (mg) | 67.99 | 0.23 | 318.64 | 0.17 | 333.31 |
| Carbohydrate (g) | 0.03 | 7.01 | 31.52 | 6.26 | 31.04 |
| Sugars (g) | 0.00 | 0.06 | 21.87 | 0.00 | 20.50 |
| Dietary fiber (g) | 1.96 | 5.89 | 1.49 | 4.84 | 1.68 |
| Crude lipid (g) | 13.59 | 28.09 | 19.19 | 21.88 | 21.40 |
| Crude protein (g) | 15.44 | 36.40 | 17.53 | 27.35 | 12.59 |
| Iron (mg) | 1.15 | 2.65 | 1.04 | 1.16 | 0.91 |
| Potassium (g) | 0.36 | 0.55 | 0.23 | 0.26 | 0.16 |
| Calcium (g) | 0.09 | 0.12 | 0.06 | 0.11 | 0.05 |
| Moisture (%) | 69.36 | 24.02 | 40.96 | 39.54 | 53.43 |
| Amino Acid | Raw | VBF | VBFS | DBF | DBFS |
|---|---|---|---|---|---|
| Essential | 4.29 | 16.44 | 7.27 | 8.09 | 3.55 |
| Threonine | 0.59 | 1.63 | 0.82 | 0.83 | 0.40 |
| Valine | 0.65 | 2.02 | 0.76 | 0.99 | 0.38 |
| Isoleucine | 0.44 | 1.69 | 0.65 | 0.82 | 0.31 |
| Leucine | 0.80 | 2.97 | 1.41 | 1.46 | 0.71 |
| Phenylalanine | 0.50 | 1.45 | 0.67 | 0.73 | 0.36 |
| Lysine | 0.86 | 3.37 | 1.56 | 1.64 | 0.71 |
| Methionine | 0.12 | 1.35 | 0.57 | 0.58 | 0.26 |
| Tryptophan | 0.14 | 0.24 | 0.15 | 0.15 | 0.07 |
| Histidine | 0.19 | 1.72 | 0.68 | 0.89 | 0.35 |
| Non-essential | 6.15 | 19.48 | 9.74 | 9.99 | 5.27 |
| Arginine | 0.55 | 2.15 | 1.02 | 1.06 | 0.50 |
| Aspartic acid | 1.12 | 3.48 | 1.69 | 1.74 | 0.81 |
| Serine | 0.65 | 1.37 | 0.77 | 0.71 | 0.40 |
| Glutamic acid | 1.40 | 5.33 | 2.83 | 2.79 | 1.68 |
| Proline | 0.56 | 1.55 | 0.76 | 0.89 | 0.49 |
| Glycine | 0.64 | 2.02 | 0.90 | 1.03 | 0.50 |
| Alanine | 0.62 | 2.27 | 1.10 | 1.12 | 0.55 |
| Tyrosine | 0.25 | 0.82 | 0.44 | 0.40 | 0.21 |
| Cystine | 0.36 | 0.49 | 0.23 | 0.25 | 0.13 |
| Total | 10.44 | 35.92 | 17.01 | 18.08 | 8.82 |
| Item | Raw | VBF | VBFS | DBF | DBFS |
|---|---|---|---|---|---|
| Lauric acid (%) | 0.06 | 0.02 | 0.03 | 0.03 | 0.02 |
| Myristic acid (%) | 4.31 | 1.33 | 2.16 | 2.33 | 1.45 |
| Pentadecanoic acid (%) | 0.54 | 0.21 | 0.32 | 0.36 | 0.20 |
| Palmitic acid (%) | 20.44 | 9.31 | 15.59 | 15.87 | 18.50 |
| Magaric acid (%) | 0.63 | 0.29 | 0.34 | 0.50 | 0.32 |
| Stearic acid (%) | 5.67 | 3.02 | 2.85 | 4.89 | 4.16 |
| Arachidic acid (%) | 0.43 | 0.58 | 0.26 | 0.49 | 0.21 |
| Heneicosylic acid (%) | 0.21 | 0.08 | nd | 0.13 | nd |
| Behenic acid (%) | nd | nd | 0.14 | nd | 0.11 |
| Lignoceric acid (%) | 1.68 | 0.48 | 0.31 | 0.83 | 0.49 |
| Myristoleic acid (%) | 0.22 | 0.07 | 0.04 | 0.12 | 0.03 |
| Pentadecenoic acid (%) | 0.11 | 0.04 | 0.10 | 0.06 | 0.11 |
| Palmitoleic acid (%) | 3.36 | 1.19 | 1.62 | 1.93 | 1.12 |
| Magaoleic acid (%) | 0.6 | 0.23 | 0.13 | 0.49 | 0.24 |
| Oleic acid (%) | 22.94 | 30.73 | 27.72 | 25.21 | 23.77 |
| Linoleic acid (%) | 5.22 | 31.20 | 32.64 | 30.07 | 36.09 |
| γ-Linolenic acid (%) | 0.1 | 0.05 | nd | 0.05 | nd |
| Linolenic acid (%) | 3.22 | 6.09 | 5.22 | 3.36 | 3.72 |
| Eicosenoic acid (%) | 4.57 | 1.98 | 2.11 | 1.45 | 1.78 |
| Eicosadienoic acid (%) | 0.28 | 0.13 | 0.04 | 0.14 | 0.08 |
| Arachidonic acid (%) | 0.94 | 0.31 | 0.20 | 0.59 | 0.35 |
| Erucic acid (%) | 4.34 | 1.47 | 0.71 | 1.28 | 0.58 |
| Nervonic acid (%) | 0.57 | 0.27 | 0.16 | 0.32 | 0.18 |
| Docosapentaenoic acid (%) | nd | nd | 0.33 | nd | 0.30 |
| DHA (%) | 12.32 | 6.85 | 4.75 | 5.95 | 4.29 |
| EPA (%) | 7.25 | 4.07 | 2.23 | 3.55 | 1.90 |
| ∑SFA (%) | 33.97 | 15.32 | 22.00 | 25.43 | 25.46 |
| ∑PUFA (%) | 24.33 | 31.77 | 30.45 | 43.71 | 46.73 |
| ∑MUFA (%) | 41.71 | 52.91 | 47.55 | 30.86 | 27.81 |
| ∑ω3 (%) | 20.79 | 17.01 | 12.53 | 12.86 | 10.21 |
| ∑ω6 (%) | 3.54 | 14.76 | 17.92 | 30.85 | 36.52 |
| ω6/ω3 | 0.17 | 0.87 | 1.43 | 2.40 | 3.58 |
| TI | 0.33 | 0.16 | 0.29 | 0.33 | 0.38 |
| AI | 0.57 | 0.17 | 0.31 | 0.34 | 0.33 |
| h/H | 2.10 | 7.45 | 4.12 | 3.78 | 3.53 |
| Temperature (°C) | Day | OA (Score) | VBN (mg%) | TBARS (mg MDA/kg) | TBC (log CFU/g) |
|---|---|---|---|---|---|
| −13 | 0 | 8.92 ± 0.08 a | 11.13 ± 0.47 a | 1.28 ± 0.05 a | 2.19 ± 0.05a |
| 15 | 8.83 ± 0.04 ab | 11.58 ± 0.20 a | 1.41 ± 0.12 a | 2.69 ± 0.01 b | |
| 30 | 8.68 ± 0.05 abc | 11.82 ± 1.22 a | 1.68 ± 0.45 a | 2.73 ± 0.03 b | |
| 45 | 8.50 ± 0.03 abc | 12.05 ± 0.96 a | 1.83 ± 0.31 a | 2.76 ± 0.02 b | |
| 60 | 8.44 ± 0.00 bc | 12.40 ± 0.47 a | 2.08 ± 0.56 a | 2.78 ± 0.03 b | |
| 75 | 8.27 ± 0.06 c | 13.11 ± 0.62 a | 2.13 ± 0.21 a | 2.85 ± 0.02 b | |
| −18 | 0 | 8.92 ± 0.08 a | 11.13 ± 0.47 a | 1.28 ± 0.05 a | 2.19 ± 0.05 a |
| 15 | 8.95 ± 0.04 a | 11.59 ± 0.20 a | 1.47 ± 0.28 a | 2.81 ± 0.02 b | |
| 30 | 8.63 ± 0.10 abc | 11.70 ± 0.12 a | 1.64 ± 0.27 a | 2.74 ± 0.01 b | |
| 45 | 8.67 ± 0.06 abc | 11.82 ± 0.77 a | 1.81 ± 0.26 a | 2.76 ± 0.04 b | |
| 60 | 8.39 ± 0.08 bc | 12.17 ± 0.23 a | 2.03 ± 0.21 a | 2.81 ± 0.03 b | |
| 75 | 8.30 ± 0.05 c | 12.29 ± 0.35 a | 2.08 ± 0.10 a | 2.86 ± 0.03 b | |
| −23 | 0 | 8.92 ± 0.08 a | 11.13 ± 0.47 a | 1.28 ± 0.05 a | 2.19 ± 0.05 a |
| 15 | 8.83 ± 0.04 ab | 11.59 ± 0.41 a | 1.47 ± 0.48 a | 2.77 ± 0.04 b | |
| 30 | 8.67 ± 0.08 abc | 11.94 ± 0.20 a | 1.69 ± 0.09 a | 2.75 ± 0.03 b | |
| 45 | 8.54 ± 0.24 abc | 11.94 ± 0.20 a | 1.81 ± 0.07 a | 2.71 ± 0.11 b | |
| 60 | 8.29 ± 0.08 c | 12.05 ± 0.59 a | 1.97 ± 0.32 a | 2.75 ± 0.04 b | |
| 75 | 8.25 ± 0.07 c | 12.17 ± 0.31 a | 1.97 ± 0.14 a | 2.77 ± 0.05 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tirtawijaya, G.; Lee, M.-J.; Negara, B.F.S.P.; Cho, W.-H.; Sohn, J.-H.; Kim, J.-S.; Choi, J.-S. Effects of Vacuum Frying on the Preparation of Ready-to-Heat Batter-Fried and Sauced Chub Mackerel (Scomber japonicus). Foods 2021, 10, 1962. https://doi.org/10.3390/foods10081962
Tirtawijaya G, Lee M-J, Negara BFSP, Cho W-H, Sohn J-H, Kim J-S, Choi J-S. Effects of Vacuum Frying on the Preparation of Ready-to-Heat Batter-Fried and Sauced Chub Mackerel (Scomber japonicus). Foods. 2021; 10(8):1962. https://doi.org/10.3390/foods10081962
Chicago/Turabian StyleTirtawijaya, Gabriel, Mi-Jeong Lee, Bertoka Fajar Surya Perwira Negara, Woo-Hee Cho, Jae-Hak Sohn, Jin-Soo Kim, and Jae-Suk Choi. 2021. "Effects of Vacuum Frying on the Preparation of Ready-to-Heat Batter-Fried and Sauced Chub Mackerel (Scomber japonicus)" Foods 10, no. 8: 1962. https://doi.org/10.3390/foods10081962
APA StyleTirtawijaya, G., Lee, M.-J., Negara, B. F. S. P., Cho, W.-H., Sohn, J.-H., Kim, J.-S., & Choi, J.-S. (2021). Effects of Vacuum Frying on the Preparation of Ready-to-Heat Batter-Fried and Sauced Chub Mackerel (Scomber japonicus). Foods, 10(8), 1962. https://doi.org/10.3390/foods10081962







