Physicochemical and Sensory Assessments in Spain and United States of PGI-Certified Ternera de Navarra vs. Certified Angus Beef
Abstract
:1. Introduction
2. Materials and Methods
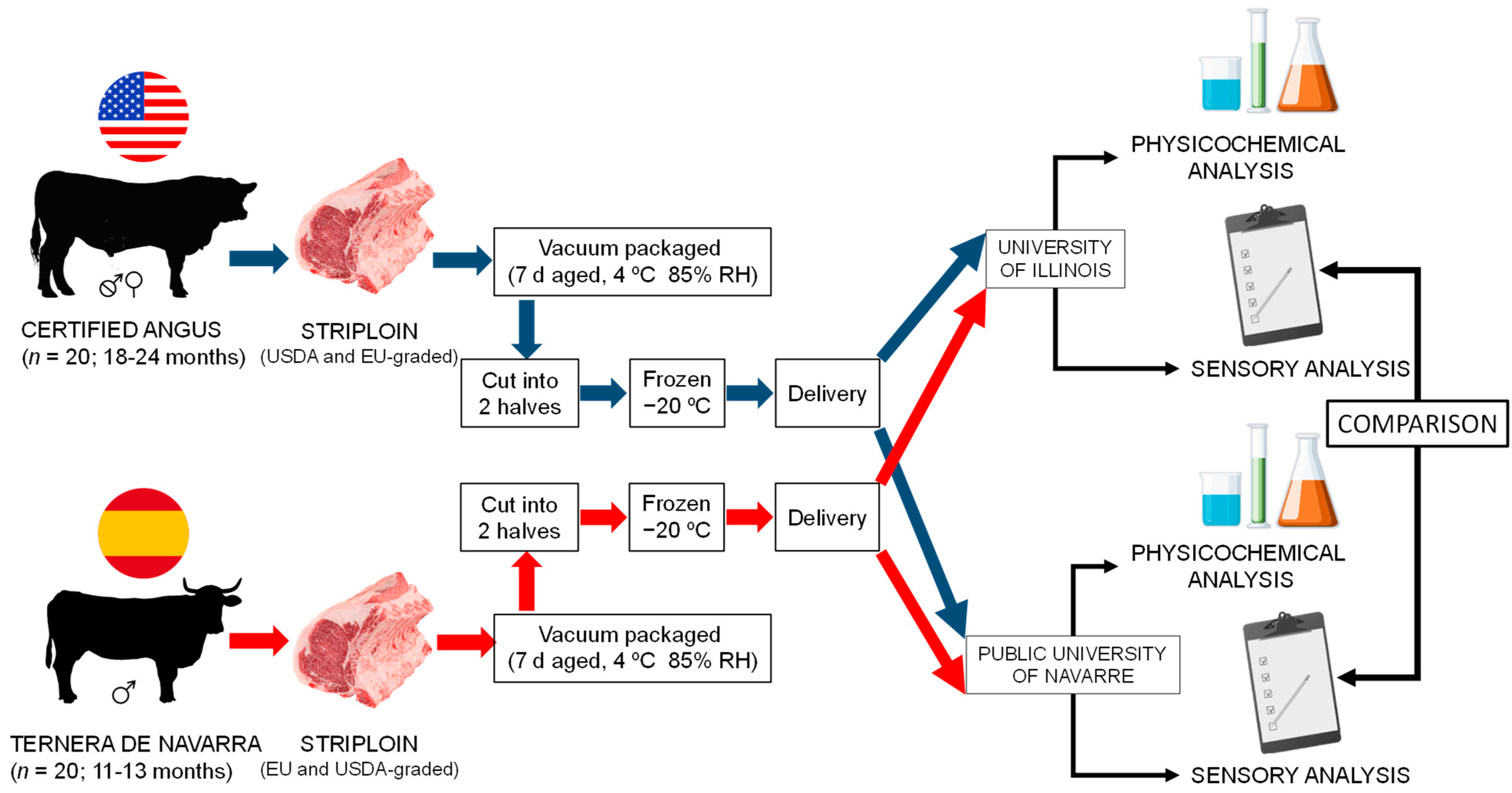
2.1. Animals Handling and Sampling
2.2. Marbling Score and Longissimus Dorsi Area
2.3. Warner Bratzler Shear Force
2.4. Proximate Analysis
2.5. Total Collagen Quantification
2.6. Soluble Collagen Quantification
2.7. Sensory Assessment
2.8. Statistical Analysis
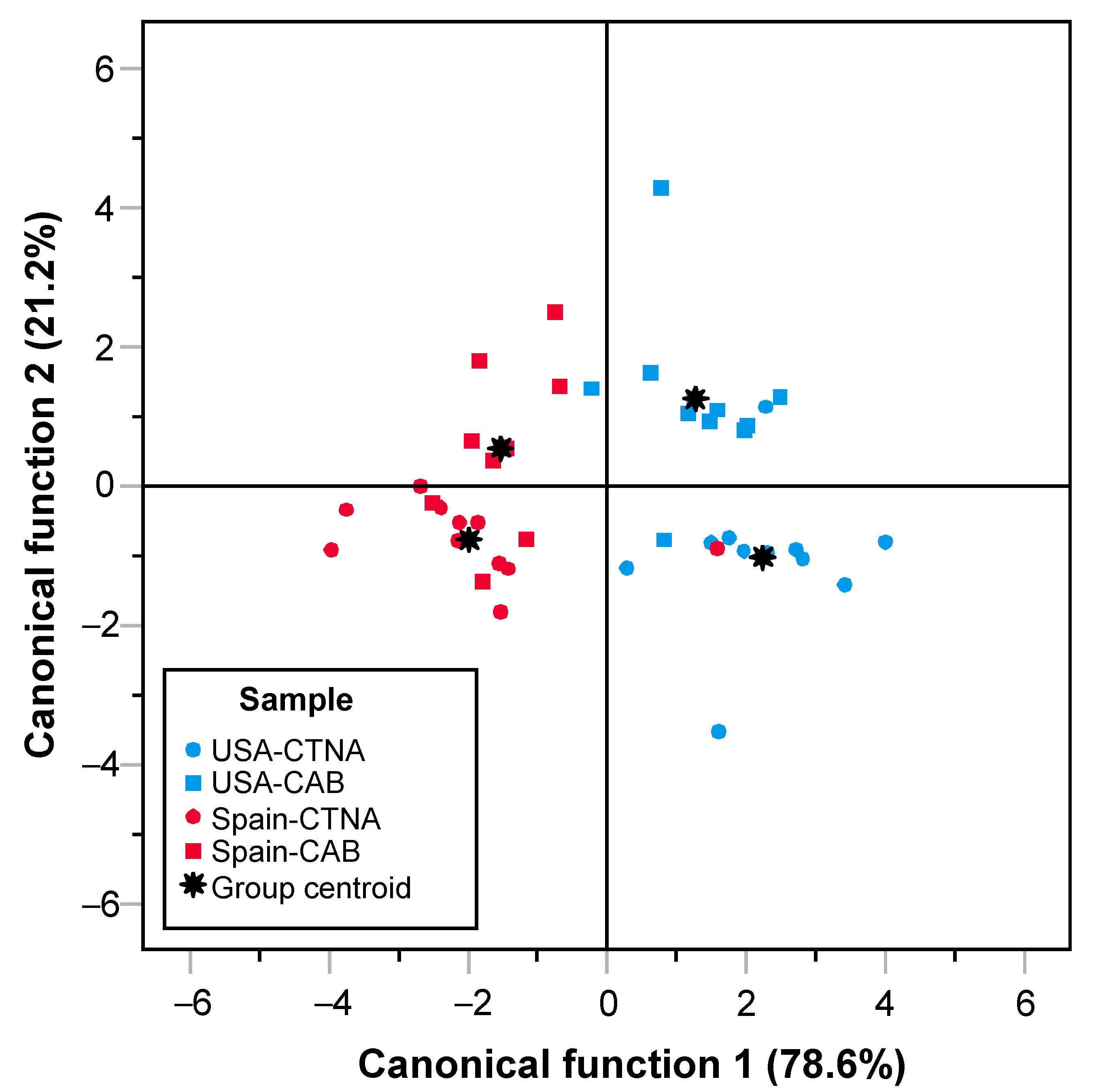
3. Results and Discussion
3.1. Marbling Score and REA
3.2. Warner–Bratzler Shear Force
3.3. Proximate Analysis
3.4. Total and Soluble Collagen
3.5. Sensory Assessment
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Whitnall, T.; Pitts, N. Global trends in meat consumption. Agric. Commod. 2019, 9, 96–99. [Google Scholar]
- OECD/FAO. OECD/FAO Meat. In OECD-FAO Agricultural Outlook 2020–2029; OECD Publishing: Rome, Italy, 2020; pp. 162–173. [Google Scholar]
- Henchion, M.M.; McCarthy, M.; Resconi, V.C. Beef quality attributes: A systematic review of consumer perspectives. Meat Sci. 2017, 128, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Petrescu, D.C.; Vermeir, I.; Petrescu-Mag, R.M. Consumer understanding of food quality, healthiness, and environmental impact: A cross-national perspective. Int. J. Environ. Res. Public. Health 2020, 17, 169. [Google Scholar] [CrossRef] [Green Version]
- Morales, R.; Aguiar, A.P.S.; Subiabre, I.; Realini, C.E. Beef acceptability and consumer expectations associated with production systems and marbling. Food Qual. Prefer. 2013, 29, 166–173. [Google Scholar] [CrossRef]
- Lee, B.; Yoon, S.; Choi, Y.M. Comparison of marbling fleck characteristics between beef marbling grades and its effect on sensory quality characteristics in high-marbled Hanwoo steer. Meat Sci. 2019, 152, 109–115. [Google Scholar] [CrossRef]
- Barjolle, D.; Lehmann, B.; Chappuis, J.-M.; Dufour, M. Protected Designation of Origin and Institutions (France, Spain and Italy); European Association of Agricultural Economists: Parma, Italy, 1997; pp. 483–502. [Google Scholar]
- Ministerio de Agricultura, Pesca y Alimentación. Orden de 26 de diciembre de 2001 por la que se ratifica el Reglamento de la Indicación Geográfica Protegida “Ternera de Navarra o Nafarroako Aratxea” y de su Consejo Regulador. Bol. Of. Estado 2002, 13, 1755–1763. [Google Scholar]
- Berian, M.J.; Sanchez, M.; Carr, T.R. A comparison of consumer sensory acceptance, purchase intention, and willingness to pay for high quality United States and Spanish beef under different information scenarios. J. Anim. Sci. 2009, 87, 3392–3402. [Google Scholar] [CrossRef]
- Lizaso, G.; Beriain, M.J.; Horcada, A.; Chasco, J.; Purroy, A. Effect of intended purpose (dairy/beef production) on beef quality. Can. J. Anim. Sci. 2011, 91, 97–102. [Google Scholar] [CrossRef] [Green Version]
- USDA Certified Angus Beef. G-1 specification. In Standards and Specifications Division; US Department of Agriculture: Washington, DC, USA, 2020. [Google Scholar]
- Nelson, J.L.; Dolezal, H.G.; Ray, F.K.; Morgan, J.B. Characterization of certified angus beef steaks from the round, loin, and chuck. J. Anim. Sci. 2004, 82, 1437–1444. [Google Scholar] [CrossRef]
- Ministerio de Agricultura, Pesca y Alimentación. I.G.P. Ternera de Navarra/Nafarroako Aratxea. Available online: https://www.mapa.gob.es/es/alimentacion/temas/calidad-diferenciada/dop-igp/carnes/IGP_Ternera_Navarra.aspx (accessed on 18 June 2021).
- García-González, R.; García Serrano, A.; Revilla, R. Comparación del regimen alimentario de vacas pardo alpinas y pirenaicas en un puerto del Pirineo occidental. In Proceedings of the Actas XXXII Reunion Científica de la S.E.E.P. Sociedad Española para el Estudio de los Pastos: Pamplona, Spain, 1992; pp. 299–305. [Google Scholar]
- USDA Carcass Beef Grades and Standards; US Department of Agriculture: Washington, DC, USA, 2017; p. 17.
- Frank, D.; Ball, A.; Hughes, J.; Krishnamurthy, R.; Piyasiri, U.; Stark, J.; Watkins, P.; Warner, R. Sensory and flavor chemistry characteristics of Australian beef: Influence of intramuscular fat, feed, and breed. J. Agric. Food Chem. 2016, 64, 4299–4311. [Google Scholar] [CrossRef]
- Joo, S.-T.; Hwang, Y.-H.; Frank, D. Characteristics of Hanwoo cattle and health implications of consuming highly marbled Hanwoo beef. Meat Sci. 2017, 132, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Sitz, B.M.; Calkins, C.R.; Feuz, D.M.; Umberger, W.J.; Eskridge, K.M. Consumer sensory acceptance and value of domestic, Canadian, and Australian grass-fed beef steaks. J. Anim. Sci. 2005, 83, 2863–2868. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Banović, M.; Grunert, K.G.; Barreira, M.M.; Fontes, M.A. Beef quality perception at the point of purchase: A study from Portugal. Food Qual. Prefer. 2009, 20, 335–342. [Google Scholar] [CrossRef]
- Killinger, K.M.; Calkins, C.R.; Umberger, W.J.; Feuz, D.M.; Eskridge, K.M. A comparison of consumer sensory acceptance and value of domestic beef steaks and steaks from a branded, Argentine beef program. J. Anim. Sci. 2004, 82, 3302–3307. [Google Scholar] [CrossRef] [PubMed]
- Ellies-Oury, M.-P.; Hocquette, J.-F.; Chriki, S.; Conanec, A.; Farmer, L.; Chavent, M.; Saracco, J. Various statistical approaches to assess and predict carcass and meat quality traits. Foods 2020, 9, 525. [Google Scholar] [CrossRef]
- European Commission. Council Regulation (EC) No 1099/2009 of 24 September 2009 on the protection of animals at the time of killing. Off. J. Eur. Union 2009, L 303, 1–30. [Google Scholar]
- Tyson Foods Inc. Angus Beef. In Tyson Fresh Meats; Tyson Foods Inc.: Dakona Dunes, SD, USA, 2021. [Google Scholar]
- European Commission. Commission Regulation (EC) No 1249/2008 of 10 December 2008 laying down detailed rules on the implementation of the Community scales for the classification of beef, pig and sheep carcasses and the reporting of prices thereof. Off. J. Eur. Union 2008, L 337, 1–35. [Google Scholar]
- Novaković, S.; Tomašević, I. A comparison between Warner-Bratzler shear force measurement and texture profile analysis of meat and meat products: A review. IOP Conf. Ser. Earth Environ. Sci. 2017, 85, 012063. [Google Scholar] [CrossRef] [Green Version]
- ISO. ISO 1442:1997 Meat and Meat Products—Determination of Moisture Content (Reference Method); ISO: Geneva, Switzerland, 1997. [Google Scholar]
- AOAC Fat (crude) in meat and meat products (Official method of analysis, no. 991.36). In Official Methods of Analysis of AOAC International; AOAC International: Gaithersburg, MD, USA, 2007.
- AOAC Hydroxyproline in meat and meat products (Official method of analysis, no 990.26). In Official Methods of Analysis of AOAC International; AOAC International: Gaithersburg, MD, USA, 1993.
- Hill, F. The solubility of intramuscular collagen in meat animals of various ages. J. Food Sci. 1966, 31, 161–166. [Google Scholar] [CrossRef]
- Bergman, I.; Loxley, R. Two improved and simplified methods for the spectrophotometric determination of hydroxyproline. Anal. Chem. 1963, 35, 1961–1965. [Google Scholar] [CrossRef]
- Cross, H.R.; Carpenter, Z.L.; Smith, G.C. Effects of intramuscular collagen and elastin on bovine muscle tenderness. J. Food Sci. 1973, 38, 998–1003. [Google Scholar] [CrossRef]
- AMSA Research Guidelines for Cookery, Sensory Evaluation, and Instrumental Tenderness Measurements of Meat, 2nd ed.; American Meat Science Association: Champaign, IL, USA, 2015.
- Gorraiz, C.; Beriain, M.J.; Chasco, J.; Iraizoz, M. Descriptive analysis of meat from young ruminants in mediterranean systems. J. Sens. Stud. 2000, 15, 137–150. [Google Scholar] [CrossRef]
- Liu, J.; Chriki, S.; Ellies-Oury, M.-P.; Legrand, I.; Pogorzelski, G.; Wierzbicki, J.; Farmer, L.; Troy, D.; Polkinghorne, R.; Hocquette, J.-F. European conformation and fat scores of bovine carcasses are not good indicators of marbling. Meat Sci. 2020, 170, 108233. [Google Scholar] [CrossRef] [PubMed]
- Albertí, P.; Sañudo, C.; Lahoz, F.; Olleta, J.; Tena, R.; Jaime, S.; Panea, B.; Campo, M.; Pardos, J. Producción y rendimiento carnicero de siete razas bovinas españolas faenadas a distintos pesos. Inf. Téc. DGA 2001, 101, 1–16. [Google Scholar]
- Stewart, S.M.; Lauridsen, T.; Toft, H.; Pethick, D.W.; Gardner, G.E.; McGilchrist, P.; Christensen, M. Objective grading of eye muscle area, intramuscular fat and marbling in Australian beef and lamb. Meat Sci. 2020, 108358. [Google Scholar] [CrossRef]
- Panea, B.; Olleta, J.L.; Sañudo, C.; del Mar Campo, M.; Oliver, M.A.; Gispert, M.; Serra, X.; Renand, G.; del Carmen Oliván, M.; Jabet, S.; et al. Effects of breed-production system on collagen, textural, and sensory traits of 10 European beef cattle breeds. J. Texture Stud. 2018, 49, 528–535. [Google Scholar] [CrossRef] [PubMed]
- López-Pedrouso, M.; Rodríguez-Vázquez, R.; Purriños, L.; Oliván, M.; García-Torres, S.; Sentandreu, M.Á.; Lorenzo, J.M.; Zapata, C.; Franco, D. Sensory and physicochemical analysis of meat from bovine breeds in different livestock production systems, pre-slaughter handling conditions, and ageing time. Foods 2020, 9, 176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thompson, J.M. The effects of marbling on flavour and juiciness scores of cooked beef, after adjusting to a constant tenderness. Aust. J. Exp. Agric. 2004, 44, 645–652. [Google Scholar] [CrossRef]
- Thompson, J.M.; Polkinghorne, R.; Hwang, I.H.; Gee, A.M.; Cho, S.H.; Park, B.Y.; Lee, J.M.; Thompson, J.M.; Polkinghorne, R.; Hwang, I.H.; et al. Beef quality grades as determined by Korean and Australian consumers. Aust. J. Exp. Agric. 2008, 48, 1380–1386. [Google Scholar] [CrossRef] [Green Version]
- Sañudo, C.; Campo, M.; Lasalle, P.; Olleta, J.; Panea, B. Calidad instrumental de la carne de bovino de siete razas españolas. Arch. Zootec. 1998, 47, 397–402. [Google Scholar]
- Wheeler, T.L.; Cundiff, L.V.; Koch, R.M. Effect of marbling degree on beef palatability in Bos taurus and Bos indicus cattle. J. Anim. Sci. 1994, 72, 3145–3151. [Google Scholar] [CrossRef]
- Matthews, K.R. Saturated fat reduction in butchered meat. In Reducing Saturated Fats in Foods; Talbot, G., Ed.; Woodhead Publishing Series in Food Science, Technology and Nutrition; Woodhead Publishing: Cambridge, UK, 2011; pp. 195–209. ISBN 978-1-84569-740-2. [Google Scholar]
- Corbin, C.H.; O’Quinn, T.G.; Garmyn, A.J.; Legako, J.F.; Hunt, M.R.; Dinh, T.T.N.; Rathmann, R.J.; Brooks, J.C.; Miller, M.F. Sensory evaluation of tender beef strip loin steaks of varying marbling levels and quality treatments. Meat Sci. 2015, 100, 24–31. [Google Scholar] [CrossRef]
- Frank, D.; Kaczmarska, K.; Paterson, J.; Piyasiri, U.; Warner, R. Effect of marbling on volatile generation, oral breakdown and in mouth flavor release of grilled beef. Meat Sci. 2017, 133, 61–68. [Google Scholar] [CrossRef]
- Legako, J.F.; Dinh, T.T.N.; Miller, M.F.; Adhikari, K.; Brooks, J.C. Consumer palatability scores, sensory descriptive attributes, and volatile compounds of grilled beef steaks from three USDA Quality Grades. Meat Sci. 2016, 112, 77–85. [Google Scholar] [CrossRef]
- Arshad, M.S.; Sohaib, M.; Ahmad, R.S.; Nadeem, M.T.; Imran, A.; Arshad, M.U.; Kwon, J.-H.; Amjad, Z. Ruminant meat flavor influenced by different factors with special reference to fatty acids. Lipids Health Dis. 2018, 17, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Miller, R.; A, T.; Kerry, J.; Ledward, D. Factors Affecting the Quality of Raw Meat; Woodhead Publishing: Cambridge, UK, 2002; pp. 27–63. ISBN 978-1-85573-583-5. [Google Scholar]
- Jenschke, B.E.; Hodgen, J.M.; Calkins, C.R. Fatty acids and minerals affect the liver-like off-flavor in cooked beef. Neb. Beef Cattle Rep. 2007, 85, 84–85. [Google Scholar]
- Dransfield, E.; Rhodes, D.N.; Nute, G.R.; Roberts, T.A.; Boccard, R.; Touraille, C.; Buchter, L.; Hood, D.E.; Joseph, R.L.; Schon, I.; et al. Eating quality of European beef assessed at five research institutes. Meat Sci. 1982, 6, 163–184. [Google Scholar] [CrossRef]
- Dransfield, E.; Nute, G.R.; Roberts, T.A.; Boccard, R.; Touraille, C.; Buchter, L.; Casteels, M.; Cosentino, E.; Hood, D.E.; Joseph, R.L.; et al. Beef quality assessed at European research centres. Meat Sci. 1984, 10, 1–20. [Google Scholar] [CrossRef]
| Attribute | CTNA | CAB | SEM | p-Value | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mean | Min | Max | SD | Mean | Min | Max | SD | |||
| Marbling 1,3 | 307.5 Traces07 | 230.0 Devoid30 | 410.0 Slight10 | 45 | 837.5 Slightly abundant73 | 730.0 Moderate30 | 1090.0 Abundant90 | 86 | 15.80 | <0.0001 |
| Marbling 2,4 | 102.5 Traces07 | 30.0 Practically devoid30 | 210.0 Slight10 | 50 | 739.0 Slightly abundant75 | 620.0 Moderate30 | 920.0 Abundant90 | 93 | 16.49 | <0.0001 |
| REA (cm2) 1,3 | 104.47 | 83.87 | 136.77 | 14.09 | 77.89 | 66.32 | 98.64 | 7.62 | 2.53 | <0.0001 |
| REA (cm2) 2,4 | 141.96 | 106.54 | 181.41 | 20.79 | 78.36 | 69.51 | 99.02 | 7.35 | 3.48 | <0.0001 |
| Attribute | CTNA Striploins | CAB Striploins | SEM | p-Value | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mean | Min | Max | SD | Mean | Min | Max | SD | |||
| Fat (%) 2 | 0.85 | 0.39 | 1.33 | 0.25 | 5.84 | 3.12 | 8.92 | 1.70 | 0.27 | <0.0001 |
| Moisture(%) 1 | 75.43 | 72.92 | 76.34 | 0.82 | 65.41 | 58.02 | 67.91 | 2.51 | 0.41 | <0.0001 |
| Total Collagen(mg/g) 1 | 2.52 | 1.45 | 3.31 | 0.48 | 2.69 | 2.09 | 3.33 | 0.40 | 0.10 | 0.2485 |
| Soluble Collagen (mg/g) 2 | 0.41 | 0.27 | 0.78 | 0.15 | 0.43 | 0.24 | 0.71 | 0.11 | 0.29 | 0.6488 |
| Shear Force (kg) 2 | 5.90 | 2.20 | 8.25 | 1.54 | 3.86 | 2.84 | 5.46 | 0.55 | 0.25 | <0.0001 |
| Attribute | CTNA Striploins | CAB Striploins | SEM | p-Value | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mean | Min | Max | SD | Mean | Min | Max | SD | |||
| Tenderness 1 | 8.14 | 5.70 | 10.10 | 1.36 | 9.92 | 7.63 | 11.90 | 1.26 | 0.29 | <0.0001 |
| Juiciness 1 | 9.20 | 6.80 | 10.77 | 1.12 | 10.06 | 8.12 | 11.78 | 0.87 | 0.22 | 0.0099 |
| Beef flavor 1 | 6.20 | 4.33 | 7.93 | 0.88 | 7.99 | 6.55 | 10.47 | 1.01 | 0.21 | <0.0001 |
| Off-flavor 1 | 0.88 | 0.17 | 1.75 | 0.50 | 0.32 | 0.07 | 0.78 | 0.22 | 0.09 | <0.0001 |
| Tenderness 2 | 8.72 | 5.46 | 9.72 | 0.86 | 9.86 | 7.77 | 11.90 | 0.96 | 0.22 | <0.0001 |
| Juiciness 2 | 6.05 | 3.66 | 9.19 | 1.34 | 7.39 | 5.69 | 9.48 | 1.19 | 0.28 | 0.0018 |
| Beef flavor 2 | 6.25 | 3.77 | 7.69 | 1.00 | 7.28 | 5.90 | 8.70 | 0.77 | 0.18 | 0.0004 |
| Liver-like flavor 2 | 3.77 | 2.63 | 5.04 | 0.75 | 3.23 | 2.12 | 5.38 | 0.79 | 0.17 | 0.0353 |
| Fat flavor 2 | 2.83 | 1.77 | 3.66 | 0.57 | 4.27 | 2.80 | 6.86 | 1.01 | 0.18 | <0.0001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beriain, M.J.; Murillo-Arbizu, M.T.; Insausti, K.; Ibañez, F.C.; Cord, C.L.; Carr, T.R. Physicochemical and Sensory Assessments in Spain and United States of PGI-Certified Ternera de Navarra vs. Certified Angus Beef. Foods 2021, 10, 1474. https://doi.org/10.3390/foods10071474
Beriain MJ, Murillo-Arbizu MT, Insausti K, Ibañez FC, Cord CL, Carr TR. Physicochemical and Sensory Assessments in Spain and United States of PGI-Certified Ternera de Navarra vs. Certified Angus Beef. Foods. 2021; 10(7):1474. https://doi.org/10.3390/foods10071474
Chicago/Turabian StyleBeriain, María José, María T. Murillo-Arbizu, Kizkitza Insausti, Francisco C. Ibañez, Christine Leick Cord, and Tom R. Carr. 2021. "Physicochemical and Sensory Assessments in Spain and United States of PGI-Certified Ternera de Navarra vs. Certified Angus Beef" Foods 10, no. 7: 1474. https://doi.org/10.3390/foods10071474
APA StyleBeriain, M. J., Murillo-Arbizu, M. T., Insausti, K., Ibañez, F. C., Cord, C. L., & Carr, T. R. (2021). Physicochemical and Sensory Assessments in Spain and United States of PGI-Certified Ternera de Navarra vs. Certified Angus Beef. Foods, 10(7), 1474. https://doi.org/10.3390/foods10071474







