Impact of Cold Storage on Bioactive Compounds and Their Stability of 36 Organically Grown Beetroot Genotypes
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Plant Materials and Sample Preparation
2.3. Total Dry Matter Content
2.4. Total Phenolic Content (TPC)
2.5. Betalain Content
2.6. Nitrate Content Determination
2.7. Total Soluble Sugar Content
2.8. Statistical Analysis
3. Results and Discussion
3.1. Total Dry Matter Content
3.2. Total Phenolic Content
3.3. Betalain Content
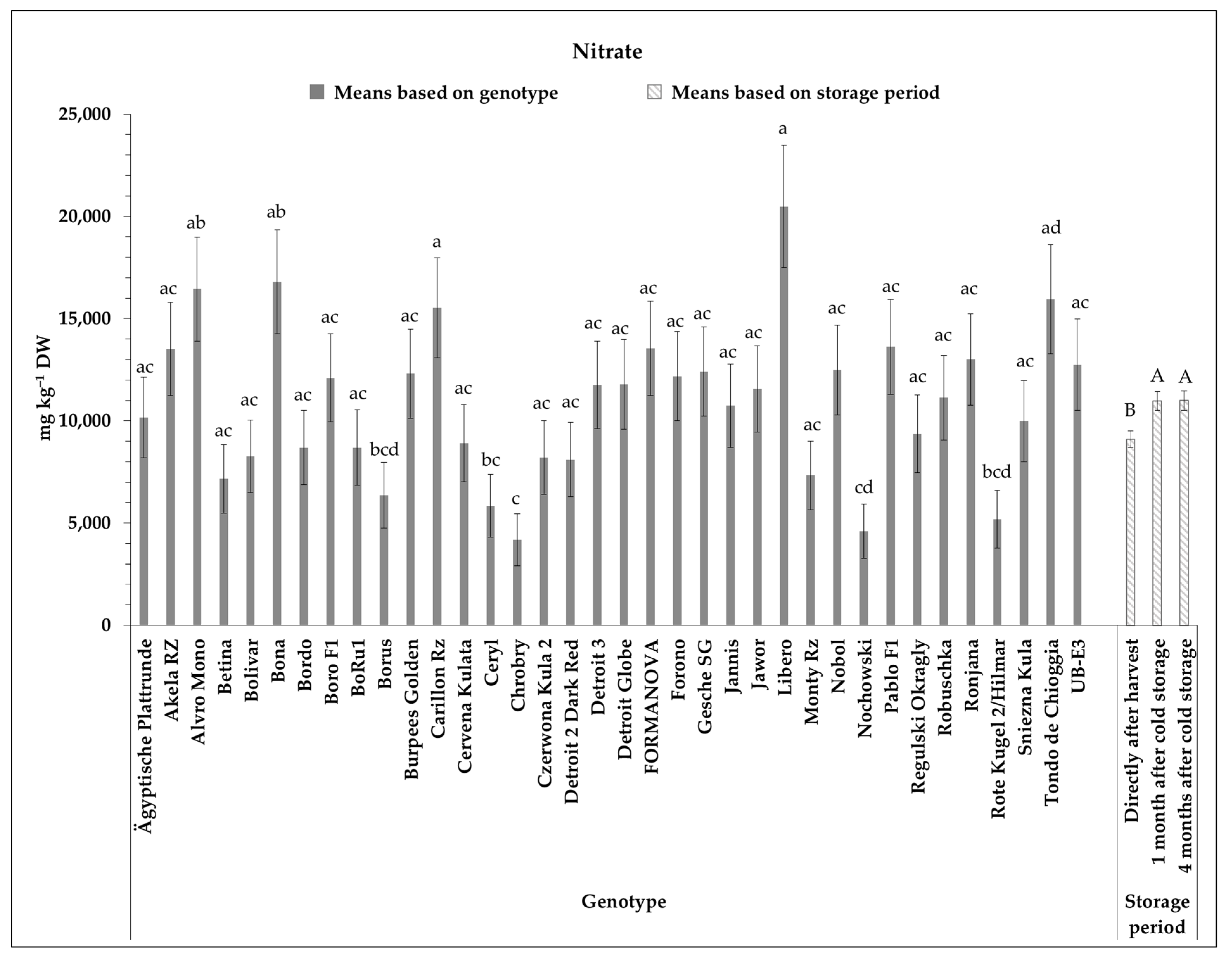
3.4. Nitrate Content
3.5. Total Soluble Sugar Content
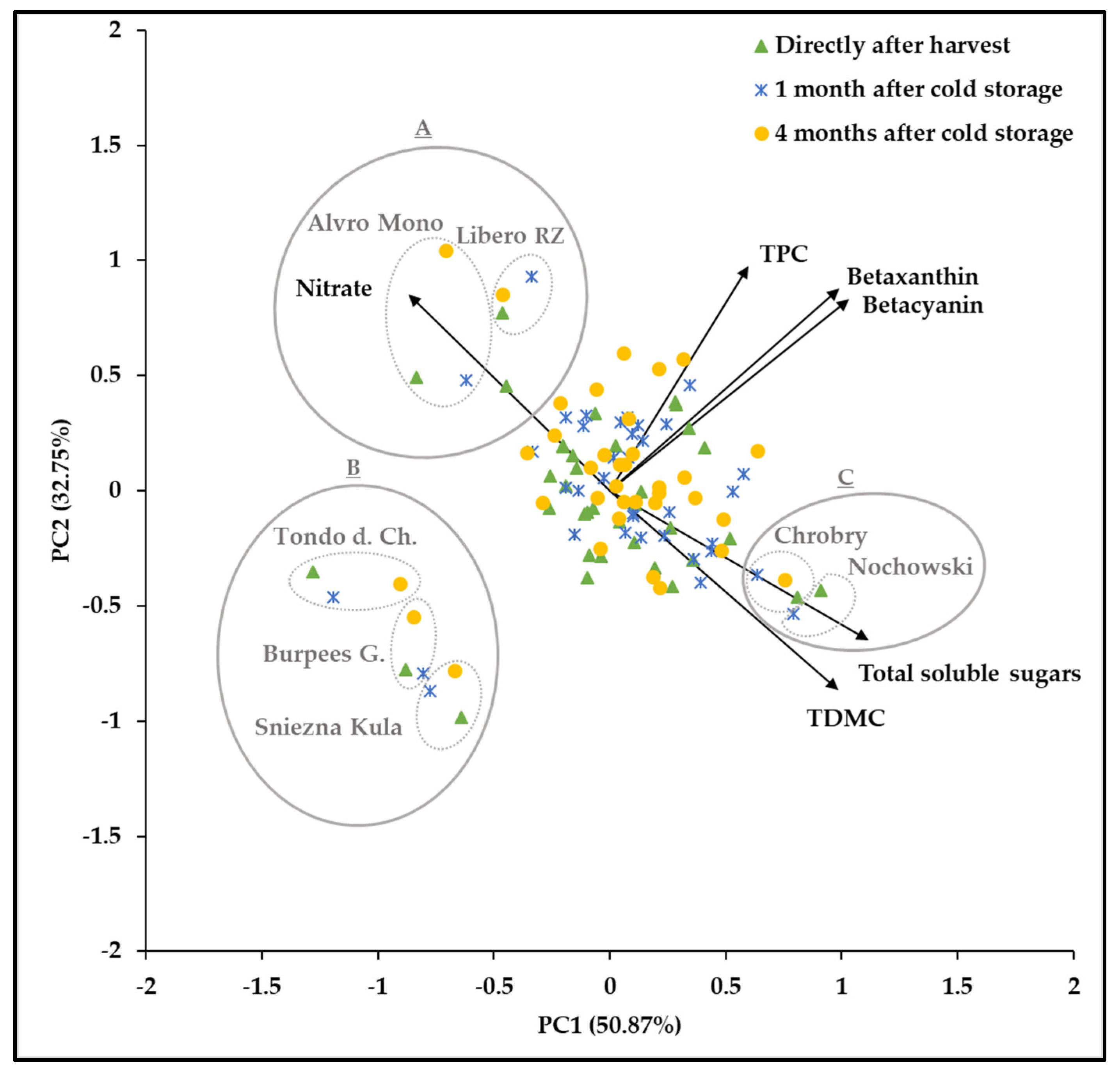
3.6. Principal Component Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wruss, J.; Waldenberger, G.; Huemer, S.; Uygun, P.; Lanzerstorfer, P.; Müller, U.; Höglinger, O.; Weghuber, J. Compositional Characteristics of Commercial Beetroot Products and Beetroot Juice Prepared from Seven Beetroot Varieties Grown in Upper Austria. J. Food Compos. Anal. 2015, 42, 46–55. [Google Scholar] [CrossRef]
- Viskelis, J.; Nevidomskis, S.; Bobinas, C.; Urbonaviciene, D.; Bobinaite, R.; Karkleliene, R.; Viskelis, P. Evaluation of Beetroot Quality during Various Storage Conditions. In Proceedings of the FOODBALT 2019 13th Baltic Conference on Food Science and Technology “Food, Nutrition, Well-being”, Jelgava, Latvia, 2–3 May 2019; pp. 170–175. [Google Scholar] [CrossRef]
- Clifford, T.; Howatson, G.; West, D.J.; Stevenson, E.J. The Potential Benefits of Red Beetroot Supplementation in Health and Disease. Nutrients 2015, 7, 2801–2822. [Google Scholar] [CrossRef] [PubMed]
- Harsh, C.; Milind Parle, K.S. Beetroot: A Health Promoting Functional Food. Inven. Rapid Nutraceuticals 2016, 1, 1–5. [Google Scholar]
- Vasconcellos, J.; Conte-Junior, C.; Silva, D.; Pierucci, A.P.; Paschoalin, V.; Alvares, T.S. Comparison of Total Antioxidant Potential, and Total Phenolic, Nitrate, Sugar, and Organic Acid Contents in Beetroot Juice, Chips, Powder, and Cooked Beetroot. Food Sci. Biotechnol. 2016, 25, 79–84. [Google Scholar] [CrossRef]
- Kujala, T.S.; Vienola, M.S.; Klika, K.D.; Loponen, J.M.; Pihlaja, K. Betalain and Phenolic Compositions of Four Beetroot (Beta Vulgaris) Cultivars. Eur. Food Res. Technol. 2002, 214, 505–510. [Google Scholar] [CrossRef]
- Lombardelli, C.; Benucci, I.; Mazzocchi, C.; Esti, M. A Novel Process for the Recovery of Betalains from Unsold Red Beets by Low-Temperature Enzyme-Assisted Extraction. Foods 2021, 10, 236. [Google Scholar] [CrossRef]
- Martins, N.; Roriz, C.L.; Morales, P.; Barros, L.; Ferreira, I.C.F.R. Coloring Attributes of Betalains: A Key Emphasis on Stability and Future Applications. Food Funct. 2017, 8, 1357–1372. [Google Scholar] [CrossRef] [PubMed]
- Strack, D.; Vogt, T.; Schliemann, W. Recent Advances in Betalain Research. Phytochemistry 2003, 62, 247–269. [Google Scholar] [CrossRef]
- Georgiev, V.G.; Weber, J.; Kneschke, E.M.; Denev, P.N.; Bley, T.; Pavlov, A.I. Antioxidant Activity and Phenolic Content of Betalain Extracts from Intact Plants and Hairy Root Cultures of the Red Beetroot Beta Vulgaris Cv. Detroit Dark Red. Plant Foods Hum. Nutr. 2010, 65, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Skalicky, M.; Kubes, J.; Shokoofeh, H.; Tahjib-Ul-Arif, M.; Vachova, P.; Hejnak, V. Betacyanins and Betaxanthins in Cultivated Varieties of Beta Vulgaris L. Compared to Weed Beets. Molecules 2020, 25, 5395. [Google Scholar] [CrossRef]
- Gasztonyi, M.N.; Daood, H.; Hájos, M.T.; Biacs, P. Comparison of Red Beet (Beta Vulgaris Var Conditiva) Varieties on the Basis of Their Pigment Components. J. Sci. Food Agric. 2001, 81, 932–933. [Google Scholar] [CrossRef]
- Mohammed, A.N.; Ishwarya, S.P.; Nisha, P. Nanoemulsion Versus Microemulsion Systems for the Encapsulation of Beetroot Extract: Comparison of Physicochemical Characteristics and Betalain Stability. Food Bioprocess Technol. 2021, 14, 133–150. [Google Scholar] [CrossRef]
- Azeredo, H.M.C. Betalains: Properties, Sources, Applications, and Stability—A Review. Int. J. Food Sci. Technol. 2009, 44, 2365–2376. [Google Scholar] [CrossRef]
- Stintzing, F.C.; Carle, R. Functional Properties of Anthocyanins and Betalains in Plants, Food, and in Human Nutrition. Trends Food Sci. Technol. 2004, 15, 19–38. [Google Scholar] [CrossRef]
- Gengatharan, A.; Dykes, G.A.; Choo, W.S. Betalains: Natural Plant Pigments with Potential Application in Functional Foods. LWT 2015, 64, 645–649. [Google Scholar] [CrossRef]
- Delgado-Vargas, F.; Jiménez, A.R.; Paredes-López, O.; Francis, F.J. Natural Pigments: Carotenoids, Anthocyanins, and Betalains—Characteristics, Biosynthesis, Processing, and Stability. Crit. Rev. Food Sci. Nutr. 2000, 40, 173–289. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.; Hathan, B.S.; Devi, C.; Memorial, L.; Longowal, S. Chemical Composition, Functional Properties and Processing of Beetroot—A Review. Int. J. Sci. Eng. Res. 2014, 5, 679–684. [Google Scholar]
- Blekkenhorst, L.C.; Bondonno, N.P.; Liu, A.H.; Ward, N.C.; Prince, R.L.; Lewis, J.R.; Devine, A.; Croft, K.D.; Hodgson, J.M.; Bondonno, C.P. Nitrate, the Oral Microbiome, and Cardiovascular Health: A Systematic Literature Review of Human and Animal Studies. Am. J. Clin. Nutr. 2018, 107, 504–522. [Google Scholar] [CrossRef] [PubMed]
- Rocchetti, G.; Tomas, M.; Zhang, L.; Zengin, G.; Lucini, L.; Capanoglu, E. Red Beet (Beta Vulgaris) and Amaranth (Amaranthus Sp.) Microgreens: Effect of Storage and in Vitro Gastrointestinal Digestion on the Untargeted Metabolomic Profile. Food Chem. 2020, 332, 127415. [Google Scholar] [CrossRef] [PubMed]
- Andac, M.; Eren, H.; Coldur, F. Determination of Nitrate in Leafy Vegetables by Flow Injection Analysis with Potentiometric Detection. J. Food Drug Anal. 2011, 19, 457–462. [Google Scholar] [CrossRef]
- Babateen, A.M.; Fornelli, G.; Donini, L.M.; Mathers, J.C.; Siervo, M. Assessment of Dietary Nitrate Intake in Humans: A Systematic Review. Am. J. Clin. Nutr. 2018, 108, 878–888. [Google Scholar] [CrossRef] [PubMed]
- Lundberg, J.O.; Carlstörm, M.; Larsen, F.J.; Weitzberg, E. Roles of Dietary Inorganic Nitrate in Cardiovascular Health and Disease. Cardiovasc. Res. 2011, 89, 525–532. [Google Scholar] [CrossRef] [PubMed]
- Baião, D.d.S.; da Silva, D.V.T.; Paschoalin, V.M.F. Beetroot, a Remarkable Vegetable: Its Nitrate and Phytochemical Contents Can Be Adjusted in Novel Formulations to Benefit Health and Support Cardiovascular Disease Therapies. Antioxidants 2020, 9, 960. [Google Scholar] [CrossRef]
- Chhikara, N.; Kushwaha, K.; Sharma, P.; Gat, Y.; Panghal, A. Bioactive Compounds of Beetroot and Utilization in Food Processing Industry: A Critical Review. Food Chem. 2019, 272, 192–200. [Google Scholar] [CrossRef]
- Fan, H.; Du, C.; Xu, Y.; Wu, X. Exogenous Nitric Oxide Improves Chilling Tolerance of Chinese Cabbage Seedlings by Affecting Antioxidant Enzymes in Leaves. Hortic. Environ. Biotechnol. 2014, 55, 159–165. [Google Scholar] [CrossRef]
- Fancy, N.N.; Bahlmann, A.K.; Loake, G.J. Nitric Oxide Function in Plant Abiotic Stress. Plant Cell Environ. 2017, 40, 462–472. [Google Scholar] [CrossRef] [PubMed]
- Sohag, A.A.M.; Tahjib-Ul-Arif, M.; Afrin, S.; Khan, M.K.; Hannan, M.A.; Skalicky, M.; Mortuza, M.G.; Brestic, M.; Hossain, M.A.; Murata, Y. Insights into Nitric Oxide-Mediated Water Balance, Antioxidant Defence and Mineral Homeostasis in Rice (Oryza Sativa L.) under Chilling Stress. Nitric. Oxide Biol. Chem. 2020, 100–101, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Bavec, M.; Turinek, M.; Grobelnik-Mlakar, S.; Slatnar, A.; Bavec, F. Influence of Industrial and Alternative Farming Systems on Contents of Sugars, Organic Acids, Total Phenolic Content, and the Antioxidant Activity of Red Beet (Beta Vulgaris L. Ssp. Vulgaris Rote Kugel). J. Agric. Food Chem. 2010, 58, 11825–11831. [Google Scholar] [CrossRef]
- Cicia, G.; Del Giudice, T.; Scarpa, R. Consumers’ Perception of Quality in Organic Food: A Random Utility Model under Preference Heterogeneity and Choice Correlation from Rank-Orderings. Br. Food J. 2002, 104, 200–213. [Google Scholar] [CrossRef]
- Carrillo, C.; Wilches-Pérez, D.; Hallmann, E.; Kazimierczak, R.; Rembiałkowska, E. Organic versus Conventional Beetroot. Bioactive Compounds and Antioxidant Properties. LWT 2019, 116, 108552. [Google Scholar] [CrossRef]
- Corleto, K.A.; Singh, J.; Jayaprakasha, G.K.; Patil, B.S. Storage Stability of Dietary Nitrate and Phenolic Compounds in Beetroot (Beta Vulgaris) and Arugula (Eruca Sativa) Juices. J. Food Sci. 2018, 83, 1237–1248. [Google Scholar] [CrossRef] [PubMed]
- Lal Basediya, A.; Samuel, D.V.K.; Beera, V. Evaporative Cooling System for Storage of Fruits and Vegetables—A Review. J. Food Sci. Technol. 2013, 50, 429–442. [Google Scholar] [CrossRef] [PubMed]
- Gopala Rao, C. Engineering for Storage of Fruits and Vegetables: Cold Storage, Controlled Atmosphere Storage, Modified Atmosphere Storage; Academic Press: Cambridge, MA, USA, 2015. [Google Scholar] [CrossRef]
- Lepse, L.; Viskelis, P.; Lepsis, J.; Bimsteine, G. Influence of Controlled Atmosphere on the Carrot Storage Quality. ISHS Acta Hortic. 2012, 1033, 59–64. [Google Scholar] [CrossRef]
- El-Ramady, H.R.; Domokos-Szabolcsy, É.; Abdalla, N.A.; Taha, H.S.; Fári, M. Postharvest Management of Fruits and Vegetables Storage. In Sustainable Agriculture Reviews; Lichtfouse, E., Ed.; Springer: Cham, Switzerland, 2015; Volume 15, pp. 65–152. [Google Scholar] [CrossRef]
- Yasaminshirazi, K.; Hartung, J.; Groenen, R.; Heinze, T.; Fleck, M.; Zikeli, S.; Graeff-Hoenninger, S. Agronomic Performance of Different Open-Pollinated Beetroot Genotypes Grown Under Organic Farming Conditions. Agronomy 2020, 10, 812. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R. Analysis of Total Phenols and Other Oxidation Substrates and Antioxidants by Means of Folin-Ciocalteu Reagent. Methods Enzym. 1999, 299, 152–178. [Google Scholar] [CrossRef]
- Koubaier, H.B.H.; Snoussi, A.; Essaidi, I.; Chaabouni, M.M.; Thonart, P.; Bouzouita, N. Betalain and Phenolic Compositions, Antioxidant Activity of Tunisian Red Beet (Beta Vulgaris L. Conditiva) Roots and Stems Extracts. Int. J. Food Prop. 2014, 17, 1934–1945. [Google Scholar] [CrossRef]
- Sawicki, T.; Bączek, N.; Wiczkowski, W. Betalain Profile, Content and Antioxidant Capacity of Red Beetroot Dependent on the Genotype and Root Part. J. Funct. Foods 2016, 27, 249–261. [Google Scholar] [CrossRef]
- Yasaminshirazi, K.; Hartung, J.; Fleck, M.; Graeff-Hoenninger, S. Bioactive Compounds and Total Sugar Contents of Different Open-Pollinated Beetroot Genotypes Grown Organically. Molecules 2020, 25, 4884. [Google Scholar] [CrossRef]
- Piepho, H.P. An Algorithm for a Letter-Based Representation of All-Pairwise Comparisons. J. Comput. Graph. Stat. 2004, 13, 456–466. [Google Scholar] [CrossRef]
- Kosson, R.; Elkner, K.; Szafirowska, A. Quality of Fresh and Processed Red Beet from Organic and Conventionl Cultivation. Veg. Crop. Res. Bull. 2011, 75, 125–132. [Google Scholar] [CrossRef]
- Hagen, S.F.; Borge, G.I.A.; Solhaug, K.A.; Bengtsson, G.B. Effect of Cold Storage and Harvest Date on Bioactive Compounds in Curly Kale (Brassica oleracea L. Var. Acephala). Postharvest Biol. Technol. 2009, 51, 36–42. [Google Scholar] [CrossRef]
- Gawęda, M. The Effect of Storage Conditions on Red Beetroot Quality. Veg. Crop. Res. Bull. 2006, 65, 85–94. [Google Scholar]
- Jakopic, J.; Veberic, R.; Slatnar, A. Changes in Quality Parameters in Rutabaga (Brassica Napus Var. Napobrassica) Roots during Long Term Storage. LWT 2021, 147, 111587. [Google Scholar] [CrossRef]
- Romo-Pérez, M.L.; Weinert, C.H.; Häußler, M.; Egert, B.; Frechen, M.A.; Trierweiler, B.; Kulling, S.E.; Zörb, C. Metabolite Profiling of Onion Landraces and the Cold Storage Effect. Plant Physiol. Biochem. 2020, 146, 428–437. [Google Scholar] [CrossRef]
- Lattanzio, V.; Lattanzio, V.M.; Cardinali, A. Role of Phenolics in the Resistance Mechanisms of Plants against Fungal Pathogens and Insects. Phytochemistry 2006, 661, 23–67. [Google Scholar]
- Kujala, T.S.; Loponen, J.M.; Klika, K.D.; Pihlaja, K. Phenolics and Betacyanins in Red Beetroot (Beta Vulgaris) Root: Distribution and Effect of Cold Storage on the Content of Total Phenolics and Three Individual Compounds. J. Agric. Food Chem. 2000, 48, 5338–5342. [Google Scholar] [CrossRef] [PubMed]
- Serrano, M.; Martinez-romero, D.; Guill, F. Maintenance of Broccoli Quality and Functional Properties during Cold Storage as Affected by Modified Atmosphere Packaging. Postharvest Biol. Technol. 2006, 39, 61–68. [Google Scholar] [CrossRef]
- Sayyari, M.; Valero, D.; Babalar, M.; Kalantari, S.; Zapata, P.J.; Serrano, M. Prestorage Oxalic Acid Treatment Maintained Visual Quality, Bioactive Compounds, and Antioxidant Potential of Pomegranate after Long-Term Storage at 2 °C. J. Agric. Food Chem. 2010, 58, 6804–6808. [Google Scholar] [CrossRef]
- Külen, O.; Stushnoff, C.; Holm, D.G. Effect of Cold Storage on Total Phenolics Content, Antioxidant Activity and Vitamin C Level of Selected Potato Clones. J. Sci. Food Agric. 2013, 93, 2437–2444. [Google Scholar] [CrossRef] [PubMed]
- Maity, T.; Bawa, A.S.; Raju, P.S. Optimization and Quality Assessment of Ready-to-Eat Intermediate Moisture Compressed Beetroot Bar. J. Food Sci. Technol. 2016, 53, 3233–3243. [Google Scholar] [CrossRef]
- Ismail, A.; Marjan, Z.M.; Foong, C.W. Total Antioxidant Activity and Phenolic Content in Selected Vegetables. Food Chem. 2004, 87, 581–586. [Google Scholar] [CrossRef]
- Cejudo-Bastante, M.J.; Hurtado, N.; Delgado, A.; Heredia, F.J. Impact of PH and Temperature on the Colour and Betalain Content of Colombian Yellow Pitaya Peel (Selenicereus Megalanthus). J. Food Sci. Technol. 2016, 53, 2405–2413. [Google Scholar] [CrossRef]
- Pandey, G.; Pandey, V.; Pandey, P.R.; Thomas, G. Effect of Extraction Solvent Temperature on Betalain Content, Phenolic Content, Antioxidant Activity and Stability of Beetroot (Beta Vulgaris L.) Powder under Different Storage Conditions. Plant Arch. 2018, 18, 1623–1627. [Google Scholar]
- Yong, Y.Y.; Dykes, G.; Lee, S.M.; Choo, W.S. Effect of Refrigerated Storage on Betacyanin Composition, Antibacterial Activity of Red Pitahaya (Hylocereus Polyrhizus) and Cytotoxicity Evaluation of Betacyanin Rich Extract on Normal Human Cell Lines. LWT Food Sci. Technol. 2018, 91, 491–497. [Google Scholar] [CrossRef]
- Obenland, D.; Cantwell, M.; Lobo, R.; Collin, S.; Sievert, J.; Arpaia, M.L. Impact of Storage Conditions and Variety on Quality Attributes and Aroma Volatiles of Pitahaya (Hylocereus Spp.). Sci. Hortic. (Amst.) 2016, 199, 15–22. [Google Scholar] [CrossRef]
- Chung, J.C.; Chou, S.S.; Hwang, D.F. Changes in Nitrate and Nitrite Content of Four Vegetables during Storage at Refrigerated and Ambient Temperatures. Food Addit. Contam. 2004, 21, 317–322. [Google Scholar] [CrossRef]
- Felczynski, K.; Elkner, K. Effect of Long-Term Organic and Mineral Fertilization on the Yield and Quality of Red Beet (Beta Vulgaris L.). Veg. Crop. Res. Bull. 2008, 68, 111–125. [Google Scholar] [CrossRef]
- Grzebelus, D.; Baranski, R. Identification of Accessions Showing Low Nitrate Accumulation in a Germplasm Collection of Garden Beet. Acta Hortic. 2001, 563, 253–257. [Google Scholar] [CrossRef]
- Wang, X.; Cai, X.; Xu, C.; Wang, S.; Dai, S.; Wang, Q. Nitrate Accumulation and Expression Patterns of Genes Involved in Nitrate Transport and Assimilation in Spinach. Molecules 2018, 23, 2231. [Google Scholar] [CrossRef]
- Pal, S.; Bhattacharya, A.; Konar, A.; Mazumdar, D.; Das, A.K. Chemical Composition of Potato at Harvest and after Cold Storage. Int. J. Veg. Sci. 2008, 14, 162–176. [Google Scholar] [CrossRef]
- Barboni, T.; Cannac, M.; Chiaramonti, N. Effect of Cold Storage and Ozone Treatment on Physicochemical Parameters, Soluble Sugars and Organic Acids in Actinidia Deliciosa. Food Chem. 2010, 121, 946–951. [Google Scholar] [CrossRef]
- Galani, J.H.Y.; Patel, J.S.; Patel, N.J.; Talati, J.G. Storage of Fruits and Vegetables in Refrigerator Increases Their Phenolic Acids but Decreases the Total Phenolics, Anthocyanins and Vitamin C with Subsequent Loss of Their Antioxidant Capacity. Antioxidants 2017, 6, 59. [Google Scholar] [CrossRef] [PubMed]
- Anjana, S.U.; Iqbal, M. Nitrate Accumulation in Plants, Factors Affecting the Process, and Human Health Implications. A Review. Agron. Sustain. Dev. 2007, 27, 45–57. [Google Scholar] [CrossRef]
- Kugler, F.; Graneis, S.; Stintzing, F.C.; Carle, R. Studies on Betaxanthin Profiles of Vegetables and Fruits from the Chenopodiaceae and Cactaceae. Z. Nat. Sect. C J. Biosci. 2007, 62, 311–318. [Google Scholar] [CrossRef] [PubMed]
- Vulić, J.; Čanadanović-Brunet, J.; Ćetković, G.; Tumbas, V.; Djilas, S.; Četojević-Simin, D.; Čanadanović, V. Antioxidant and Cell Growth Activities of Beet Root Pomace Extracts. J. Funct. Foods 2012, 4, 670–678. [Google Scholar] [CrossRef]
| Genotype | Beet Color | Shape | Seed Origin |
|---|---|---|---|
| Ägyptische Plattrunde (Ä. P.) | red | flat-spherical | Sativa (DE) |
| Akela Rijk Zwaan (RZ) | red | spherical | Rijk Zwaan (NL) |
| Alvro Mono | red | spherical | Vitalis (US) |
| Betina | red | spherical | Moravo Seeds (CZ) |
| Bolivar | red | spherical | Hild (DE) |
| Bona | red | spherical | Moravo Seeds (CZ) |
| Bordo | red | spherical | Seklos (LT) |
| Boro F1 | red | spherical | Bejo (DE) |
| BoRu1 | red | spherical | Kultursaat e.V. (DE) |
| Borus | red | spherical | Spójnia (PL) |
| Burpees Golden (Burpees G.) | yellow | spherical | Bingenheimer S. AG (DE) |
| Carillon RZ | red | cylindrical | Rijk Zwaan (NL) |
| Cervena Kulata (Cervena K.) | red | spherical | Moravo Seeds (CZ) |
| Ceryl | red | spherical | Spójnia (PL) |
| Chrobry | red | spherical | Spójnia (PL) |
| Czerwona Kula 2 (Czerwona K. 2) | red | spherical | Spójnia (PL) |
| Detroit 2 Dark Red (Detroit 2 D. R.) | red | spherical | Samen Schenker (DE) |
| Detroit 3 | red | spherical | Caillard (FR) |
| Detroit Globe (Detroit G.) | red | spherical | King Seed (UK) |
| Formanova | red | cylindrical | Sativa (DE) |
| Forono | red | cylindrical | Bingenheimer S. AG (DE) |
| Gesche SG | red | spherical | Christiansens Biolandhof (DE) |
| Jannis | red | spherical | Bingenheimer S. AG (DE) |
| Jawor | red | spherical | Snówidza (PL) |
| Libero RZ | red | spherical | Rijk Zwaan (NL) |
| Monty RZ F1 | red | spherical | Rijk Zwaan (NL) |
| Nobol | red | spherical | Vilmorin (PL) |
| Nochowski | red | spherical | Spójnia (PL) |
| Pablo F1 | red | spherical | Bejo (DE) |
| Regulski Okragly (Regulski O.) | red | spherical | Pnos (PL) |
| Robuschka | red | spherical | Bingenheimer S. AG (DE) |
| Ronjana | red | spherical | Bingenheimer S. AG (DE) |
| Hilmar | red | spherical | Hild (DE) |
| Sniezna Kula | white | spherical | Torseed (PL) |
| Tondo de Chioggia (Tondo d. Ch.) | red-white | spherical | Bingenheimer S. AG (DE) |
| UB-E3 | red | spherical | U.Behrendt (DE) |
| Effect | Total Dry Matter Content | Total Phenolic Compounds | Betaxanthin | Betacyanin | Nitrate | Total Soluble Sugars |
|---|---|---|---|---|---|---|
| Genotype | <0.0001 | <0.0001 | <0.0001 | <0.0001 | 0.0005 | <0.0001 |
| Storage period | 0.0005 | <0.0001 | <0.0001 | <0.0001 | <0.0001 | n.s. 1 |
| Genotype × Storage period | n.s. | n.s. | <0.0001 | <0.0001 | n.s. | 0.0121 |
| Year | n.s. | <0.0001 | 0.0001 | <0.0001 | n.s. | 0.0008 |
| Storage period × Year | 0.0009 | 0.0001 | n.s. | n.s. | n.s. | <0.0001 |
| (A) Means (Medians) Based on Genotype | |||
|---|---|---|---|
| Genotype | TDMC (%) | Total Phenolic Content (mg GAE 100 g−1 DW) | |
| Ä. P. | 14.77 bcde ± 0.55 | 417.20 ac ± 27.18 | |
| Akela RZ | 14.81 bcde ± 0.55 | 413.30 ac ± 26.94 | |
| Alvro Mono | 11.49 e ± 0.55 | 612.39 a ± 40.58 | |
| Betina | 16.94 ac ± 0.56 | 413.71 ac ± 27.41 | |
| Bolivar | 14.85 ad ± 0.55 | 364.91 cdef ± 23.79 | |
| Bona | 14.78 bcde ± 0.55 | 405.26 ac ± 26.41 | |
| Bordo | 16.79 ac ± 0.55 | 449.00 ac ± 29.26 | |
| Boro F1 | 13.84 bcde ± 0.55 | 414.61 ac ± 27.03 | |
| BoRu1 | 14.85 ad ± 0.55 | 417.89 ac ± 27.25 | |
| Borus | 16.39 ab ± 0.56 | 376.35 bcde ± 25.94 | |
| Burpees G. | 14.87 ad ± 0.55 | 201.45 i ± 13.13 | |
| Carillon RZ | 13.81 bcde ± 0.55 | 432.35 ac ± 28.19 | |
| Cervena K. | 16.15 ab ± 0.56 | 465.96 ac ± 31.57 | |
| Ceryl | 16.25 ab ± 0.55 | 426.45 ac ± 27.80 | |
| Chrobry | 18.14 a ± 0.55 | 449.16 ac ± 29.26 | |
| Czerwona K. 2 | 15.92 ab ± 0.55 | 355.25 cdefg ± 24.04 | |
| Detroit 2 D. R. | 15.64 ad ± 0.56 | 418.26 ac ± 28.83 | |
| Detroit 3 | 14.30 bcde ± 0.55 | 403.59 ac ± 26.73 | |
| Detroit G. | 14.89 ad ± 0.56 | 350.31 cdefg ± 24.15 | |
| Formanova | 14.46 bcde ± 0.55 | 397.19 bcd ± 26.31 | |
| Forono | 15.21 ad ± 0.55 | 561.74 ab ± 37.37 | |
| Gesche SG | 15.25 ad ± 0.55 | 484.20 ac ± 31.56 | |
| Jannis | 14.68 bcde ± 0.55 | 414.71 ac ± 27.02 | |
| Jawor | 15.17 ad ± 0.55 | 430.75 ac ± 28.07 | |
| Libero RZ | 12.31 de ± 0.56 | 408.49 ac ± 29.38 | |
| Monty RZ F1 | 15.65 ad ± 0.55 | 519.91 ad ± 33.89 | |
| Nobol | 14.20 bcde ± 0.55 | 456.04 ac ± 29.74 | |
| Nochowski | 18.15 a ± 0.55 | 486.33 ac ± 31.69 | |
| Pablo F1 | 14.58 bcde ± 0.55 | 440.53 ac ± 29.33 | |
| Regulski O. | 15.89 ab ± 0.55 | 381.10 bcde ± 24.84 | |
| Robuschka | 15.57 ad ± 0.55 | 322.93 cefgh ± 21.04 | |
| Ronjana | 15.39 ad ± 0.55 | 508.49 ad ± 33.16 | |
| Hilmar | 15.01 ad ± 0.55 | 420.64 ac ± 27.41 | |
| Sniezna Kula | 15.65 ad ± 0.55 | 242.55 fi ± 16.07 | |
| Tondo d. Ch. | 13.37 bde ± 0.58 | 241.16 ei ± 20.35 | |
| UB-E3 | 16.06 ab ± 0.55 | 393.07 bcd ± 26.04 | |
| (B) Means (Medians) Based on Storage Period | |||
| Storage Period | |||
| Year 1 | Directly after harvest | 15.19 abA ± 0.14 | 294.55 cB ± 8.57 |
| 1 month after cold storage | 15.63 aA ± 0.14 | 341.18 bA ± 9.93 | |
| 4 months after cold storage | 14.95 bA ± 0.14 | 437.83 aA ± 12.78 | |
| Year 2 | Directly after harvest | 14.51 bA ± 0.21 | 395.91 aA ± 12.59 |
| 1 month after cold storage | 15.34 aA ± 0.21 | 379.09 aA ± 12.17 | |
| 4 months after cold storage | 15.46 aA ± 0.22 | 425.18 aA ± 14.47 | |
| Genotype | Betaxanthin (mg g−1 DW) | Betacyanin (mg g−1 DW) | ||||
|---|---|---|---|---|---|---|
| Directly after Harvest | 1 Month after Cold Storage | 4 Months after Cold Storage | Directly after Harvest | 1 Month after Cold Storage | 4 Months after Cold Storage | |
| Ä. P. | 4.41 aA ± 0.84 | 4.62 abA ± 0.88 | 4.40 abA ± 0.84 | 6.12 abcA ± 1.10 | 6.17 aA ± 1.11 | 5.42 aA ± 0.97 |
| Akela RZ | 4.44 aA ± 0.85 | 5.70 aA ± 1.09 | 5.04 abA ± 0.96 | 6.65 abA ± 1.19 | 8.02 aA ± 1.44 | 6.77 aA ± 1.22 |
| Alvro Mono | 2.69 aA ± 0.51 | 3.08 adA ± 0.59 | 3.05 bcdA ± 0.60 | 3.61 abcdA ± 0.65 | 4.13 aA ± 0.74 | 4.28 aA ± 0.79 |
| Betina | 4.31 aA ± 0.82 | 4.73 abA ± 0.90 | 5.13 abA ± 1.00 | 5.47 abcdA ± 0.98 | 7.20 aA ± 1.29 | 6.83 aA ± 1.26 |
| Bolivar | 4.65 aA ± 0.89 | 4.55 abA ± 0.87 | 5.28 abA ± 1.01 | 6.30 abcA ± 1.13 | 5.93 aA ± 1.07 | 6.83 aA ± 1.23 |
| Bona | 4.07 aA ± 0.78 | 4.76 abA ± 0.91 | 3.71 beA ± 0.71 | 5.79 abcA ± 1.04 | 6.39 aA ± 1.15 | 4.94 aA ± 0.89 |
| Bordo | 5.03 aA ± 0.96 | 4.48 abA ± 0.86 | 5.23 abA ± 1.00 | 6.99 abA ± 1.25 | 7.05 aA ± 1.27 | 7.28 aA ± 1.31 |
| Boro F1 | 4.75 aA ± 0.91 | 4.88 aA ± 0.93 | 4.58 abA ± 0.87 | 6.50 abA ± 1.17 | 6.17 aA ± 1.11 | 5.74 aA ± 1.03 |
| BoRu1 | 5.38 aA ± 1.03 | 5.88 aA ± 1.12 | 4.99 abA ± 0.95 | 7.23 aA ± 1.30 | 7.56 aA ± 1.36 | 6.34 aA ± 1.14 |
| Borus | 4.10 aA ± 0.78 | 4.63 abA ± 0.88 | 4.28 bcA ± 0.88 | 5.75 abcA ± 1.03 | 6.40 aA ± 1.15 | 5.63 aA ± 1.10 |
| Burpees G. | 0.20 bB ± 0.04 | 1.15 dA ± 0.22 | 1.03 deA ± 0.20 | 0.43 egA ± 0.08 | 0.09 bB ± 0.02 | 0.11 bB ± 0.02 |
| Carillon RZ | 4.83 aA ± 0.92 | 5.17 aA ± 0.99 | 4.72 abA ± 0.90 | 6.65 abA ± 1.20 | 7.47 aA ± 1.34 | 6.51 aA ± 1.17 |
| Cervena K. | 4.11 aA ± 0.79 | 4.49 abA ± 0.86 | 4.54 abA ± 0.90 | 5.75 abcA ± 1.03 | 6.30 aA ± 1.13 | 5.93 aA ± 1.11 |
| Ceryl | 5.57 aA ± 1.06 | 6.40 aA ± 1.22 | 5.48 abA ± 1.05 | 7.91 aA ± 1.42 | 8.55 aA ± 1.54 | 7.22 aA ± 1.30 |
| Chrobry | 5.93 aA ± 1.13 | 4.58 abA ± 0.88 | 4.71 abA ± 0.90 | 7.84 aA ± 1.41 | 7.69 aA ± 1.38 | 7.73 aA ± 1.39 |
| Czerwona K. 2 | 4.51 aA ± 0.88 | 4.41 abcA ± 0.84 | 3.91 bcA ± 0.76 | 6.67 abA ± 1.22 | 6.17 aA ± 1.11 | 5.48 aA ± 1.01 |
| Detroit 2 D. R. | 4.67 aA ± 0.89 | 4.40 abcA ± 0.84 | 4.63 abA ± 0.95 | 6.65 abA ± 1.19 | 6.22 aA ± 1.12 | 6.20 aA ± 1.21 |
| Detroit 3 | 5.46 aA ± 1.04 | 4.58 abA ± 0.87 | 5.41 abA ± 1.05 | 7.73 aA ± 1.39 | 6.15 aA ± 1.10 | 6.72 aA ± 1.24 |
| Detroit G. | 4.67 aA ± 0.89 | 4.08 adA ± 0.78 | 4.45 bcA ± 0.90 | 7.02 abA ± 1.26 | 5.59 aA ± 1.00 | 5.63 aA ± 1.10 |
| Formanova | 3.63 aA ± 0.69 | 3.54 adA ± 0.69 | 3.28 bcdA± 0.63 | 5.01 abcdA ± 0.90 | 5.08 aA ± 0.94 | 5.00 aA ± 0.90 |
| Forono | 5.76 aA ± 1.10 | 6.39 aA ± 1.25 | 6.70 bA ± 1.28 | 8.39 aA ± 1.51 | 7.99 aA ± 1.46 | 8.97 aA ± 1.61 |
| Gesche SG | 6.60 aA ± 1.26 | 5.74 aA ± 1.10 | 5.38 abA ± 1.03 | 7.79 aA ± 1.40 | 7.73 aA ± 1.42 | 7.11 aA ± 1.28 |
| Jannis | 4.17 aA ± 0.80 | 4.81 abA ± 0.92 | 4.90 abA ± 0.94 | 5.85 abcA ± 1.05 | 6.49 aA ± 1.16 | 6.17 aA ± 1.11 |
| Jawor | 4.24 aA ± 0.81 | 4.23 adA ± 0.81 | 3.85 bcA ± 0.74 | 5.86 abcA ± 1.05 | 6.18 aA ± 1.11 | 5.84 aA± 1.05 |
| Libero RZ | 6.25 aA ± 1.19 | 6.73 aA ± 1.38 | 5.24 abA ± 1.07 | 8.63 aA ± 1.55 | 8.70 aA ± 1.70 | 6.51 aA ± 1.27 |
| Monty RZ F1 | 5.87 aA ± 1.12 | 5.40 aA ± 1.03 | 5.29 abA ± 1.01 | 8.70 aA ± 1.56 | 8.20 aA ± 1.47 | 7.32 aA ± 1.32 |
| Nobol | 5.72 aA ± 1.09 | 5.38 aA ± 1.03 | 5.83 abA ± 1.11 | 8.18 aA ± 1.47 | 7.64 aA ± 1.37 | 8.16 aA ± 1.47 |
| Nochowski | 6.08 aA ± 1.16 | 5.05 aA ± 0.97 | 5.95 abA ± 1.14 | 7.77 aA ± 1.39 | 7.57 aA ± 1.36 | 8.49 aA ± 1.53 |
| Pablo F1 | 4.35 aA ± 0.83 | 5.00 aA ± 0.96 | 4.72 abA ± 0.92 | 6.13 abcA ± 1.10 | 6.36 aA ± 1.14 | 5.71 aA ± 1.04 |
| Regulski O. | 3.91 aA ± 0.75 | 4.96 aA ± 0.95 | 4.48 abA ± 0.86 | 5.93 abcA ± 1.06 | 6.88 aA ± 1.24 | 6.23 aA ± 1.12 |
| Robuschka | 3.85 aA ± 0.74 | 4.60 abA ± 0.88 | 5.34 abA ± 1.02 | 5.50 abcA ± 0.99 | 6.22 aA ± 1.12 | 6.70 aA ± 1.20 |
| Ronjana | 6.16 aA ± 1.18 | 4.68 abA ± 0.89 | 6.08 abA ± 1.16 | 8.07 aA ± 1.45 | 7.42 aA ± 1.33 | 9.05 aA ± 1.63 |
| Hilmar | 4.42 aA ± 0.84 | 4.73 abA ± 0.90 | 5.05 abA ± 0.97 | 5.89 abcA ± 1.06 | 6.54 aA ± 1.17 | 6.63 aA ± 1.19 |
| Sniezna Kula | 0.13 bA ± 0.03 | 0.18 eA ± 0.05 | 0.16 gA ± 0.05 | 0.12 gA ± 0.03 | 0.17 bA ± 0.04 | 0.15 bA ± 0.04 |
| Tondo d. Ch. | 0.22 bA ± 0.04 | 0.26 eA ± 0.05 | 0.19 fgA ± 0.05 | 0.30 fgA ± 0.05 | 0.25 bA ± 0.05 | 0.20 bA ± 0.05 |
| UB-E3 | 3.40 aA ± 0.65 | 4.21 adA ± 0.80 | 3.50 bcdA ± 0.68 | 5.31 abcdA ± 0.95 | 5.75 aA ± 1.03 | 4.29 aA ± 0.79 |
| Genotype | Total Soluble Sugars (°Bx) | ||
|---|---|---|---|
| Directly after Harvest | 1 Month after Cold Storage | 4 Months after Cold Storage | |
| Ä. P. | 11.08 adA ± 0.67 | 11.77 adA ± 0.67 | 12.31 bcA ± 0.67 |
| Akela RZ | 11.26 adA ± 0.67 | 11.58 adA ± 0.67 | 12.10 bcA ± 0.67 |
| Alvro Mono | 8.55 dA ± 0.67 | 9.49 dA ± 0.67 | 8.41 cA ± 0.71 |
| Betina | 13.40 abA ± 0.67 | 13.55 adA ± 0.67 | 13.74 abA ± 0.71 |
| Bolivar | 11.14 adA ± 0.67 | 9.53 cdA ± 0.67 | 12.37 bcA ± 0.67 |
| Bona | 11.18 adA ± 0.67 | 11.14 adA ± 0.67 | 10.88 bcA ± 0.67 |
| Bordo | 12.04 adA ± 0.67 | 13.50 adA ± 0.67 | 12.47 bcA ± 0.67 |
| Boro F1 | 10.99 bcdA ± 0.67 | 11.13 adA ± 0.67 | 11.16 bcA ± 0.67 |
| BoRu1 | 11.79 adA ± 0.67 | 10.60 adA ± 0.67 | 11.53 bcA ± 0.67 |
| Borus | 12.40 adA ± 0.67 | 13.53 adA ± 0.67 | 13.38 abA ± 0.76 |
| Burpees G. | 10.66 bcdA ± 0.67 | 10.59 adA ± 0.67 | 10.29 acA ± 0.67 |
| Carillon RZ | 9.76 bdA ± 0.67 | 11.87 adA ± 0.67 | 11.15 bcA ± 0.67 |
| Cervena K. | 13.85 abA ± 0.67 | 13.89 abcA ± 0.67 | 11.88 bcA ± 0.77 |
| Ceryl | 13.47 abA ± 0.67 | 12.97 adA ± 0.67 | 13.70 abA ± 0.71 |
| Chrobry | 14.40 acA ± 0.67 | 13.96 abA ± 0.67 | 14.92 bA ± 0.67 |
| Czerwona K. 2 | 12.21 adA ± 0.67 | 12.14 adA ± 0.67 | 13.44 abA ± 0.71 |
| Detroit 2 D. R. | 12.28 adA ± 0.67 | 12.91 adA ± 0.67 | 11.29 bcA ± 0.84 |
| Detroit 3 | 10.98 bcdA ± 0.67 | 11.34 adA ± 0.67 | 11.22 bcA ± 0.71 |
| Detroit G. | 10.53 bcdA ± 0.67 | 11.47 adA ± 0.67 | 11.72 bcA ± 0.76 |
| Formanova | 11.15 adA ± 0.67 | 11.11 adjA ± 0.67 | 11.34 bcA ± 0.67 |
| Forono | 11.75 adA ± 0.67 | 12.05 adA ± 0.67 | 11.87 bcA ± 0.67 |
| Gesche SG | 13.07 abeA ± 0.67 | 12.63 adA ± 0.67 | 11.80 bcA ± 0.67 |
| Jannis | 11.19 adA ± 0.67 | 11.72 adA ± 0.67 | 11.87 bcA ± 0.67 |
| Jawor | 11.71 adA ± 0.67 | 12.22 adA ± 0.67 | 12.93 abA ± 0.67 |
| Libero RZ | 8.97 deA ± 0.67 | 9.64 bdA ± 0.67 | 9.60 acA ± 0.76 |
| Monty RZ F1 | 11.36 adA ± 0.67 | 13.17 adA ± 0.67 | 12.90 bcA ± 0.71 |
| Nobol | 10.61 bcdA ± 0.67 | 11.53 adA ± 0.67 | 11.17 bcA ± 0.67 |
| Nochowski | 15.43 aA ± 0.67 | 14.88 aA ± 0.67 | 12.20 bcA ± 0.67 |
| Pablo F1 | 10.81 bcdA ± 0.67 | 11.11 adA ± 0.67 | 11.61 bcA ± 0.81 |
| Regulski O. | 11.76 adA ± 0.67 | 12.26 adA ± 0.67 | 11.99 bcA ± 0.67 |
| Robuschka | 11.86 adA ± 0.67 | 12.82 adA ± 0.67 | 11.99 bcA ± 0.67 |
| Ronjana | 12.71 adA ± 0.67 | 11.71 adA ± 0.67 | 11.73 bcA ± 0.67 |
| Hilmar | 11.43 adA ± 0.67 | 12.71 adA ± 0.67 | 11.75 bcA ± 0.67 |
| Sniezna Kula | 11.60 adA ± 0.67 | 10.74 adA ± 0.67 | 11.50 bcA ± 0.71 |
| Tondo d. Ch. | 8.77 deA ± 0.68 | 9.71 bdA ± 0.71 | 10.07 bcA ± 0.84 |
| UB-E3 | 12.70 adA ± 0.67 | 12.58 adA ± 0.67 | 13.00 abA ± 0.67 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yasaminshirazi, K.; Hartung, J.; Fleck, M.; Graeff-Hönninger, S. Impact of Cold Storage on Bioactive Compounds and Their Stability of 36 Organically Grown Beetroot Genotypes. Foods 2021, 10, 1281. https://doi.org/10.3390/foods10061281
Yasaminshirazi K, Hartung J, Fleck M, Graeff-Hönninger S. Impact of Cold Storage on Bioactive Compounds and Their Stability of 36 Organically Grown Beetroot Genotypes. Foods. 2021; 10(6):1281. https://doi.org/10.3390/foods10061281
Chicago/Turabian StyleYasaminshirazi, Khadijeh, Jens Hartung, Michael Fleck, and Simone Graeff-Hönninger. 2021. "Impact of Cold Storage on Bioactive Compounds and Their Stability of 36 Organically Grown Beetroot Genotypes" Foods 10, no. 6: 1281. https://doi.org/10.3390/foods10061281
APA StyleYasaminshirazi, K., Hartung, J., Fleck, M., & Graeff-Hönninger, S. (2021). Impact of Cold Storage on Bioactive Compounds and Their Stability of 36 Organically Grown Beetroot Genotypes. Foods, 10(6), 1281. https://doi.org/10.3390/foods10061281






