Oregano Oil, Epsilon-Polylysine and Citric Acid Assisted Inactivation of Salmonella in Two Kinds of Tahini during Thermal Treatment and Storage
Abstract
1. Introduction
2. Materials and Methods
2.1. Salmonella Strains and Culture Preparation
2.2. Sample Preparation
2.3. Thermal Treatment of S. montevideo CICC21588 in Undiluted or Diluted Tahini
2.4. Oxford Cup Assay of Antimicrobials
2.5. Control of S. montevideo CICC21588 in Undiluted or Diluted Tahini by Antimicrobials
2.6. Bacterial Enumeration
2.7. Modeling of Thermal Inactivation Kinetics
2.8. Statistical Analysis
3. Results and Discussion
3.1. Thermal Inactivation of S. montevideo CICC21588 in Undiluted or Diluted Tahini
3.2. The Sublethal Injured Cells in Thermal Treatments in Undiluted and Diluted Tahini
3.3. Inhibitory Activity against Salmonella of EOs, ε-PL, Tea Polyphenol or Organic Acid
3.4. Effect of Oregano Oil, ε-PL and CA on the Viability of S. Montevideo CICC21588 in Undiluted and Diluted Tahini
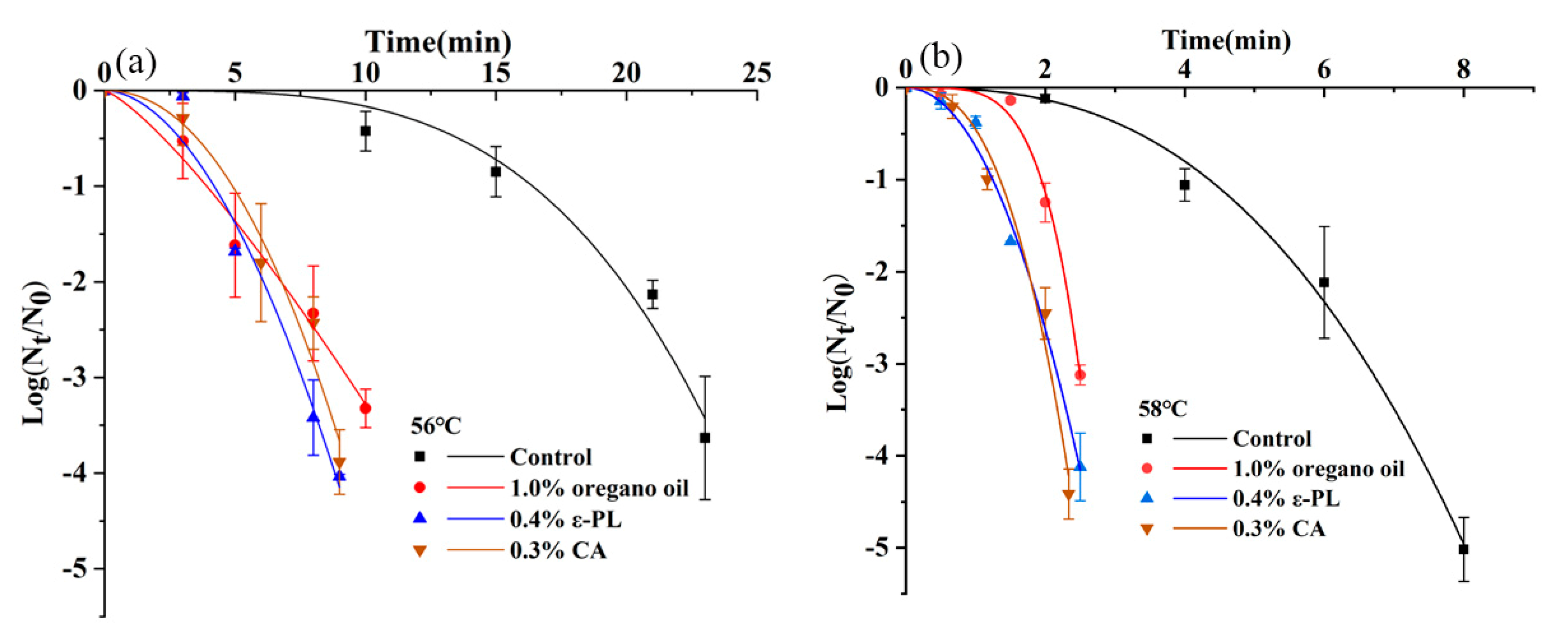
3.5. Synergistic Effects of Thermal Inactivation and Antimicrobials in Undiluted or Diluted Tahini
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- El-Adawy, T.A.; Mansour, E.H. Nutritional and physicochemical evaluations of tahina (sesame butter) prepared from heat-treated sesame seeds. J. Sci. Food Agric. 2000, 80, 2005–2011. [Google Scholar] [CrossRef]
- Al-Nabulsi, A.A.; Osaili, T.M.; Olaimat, A.N.; Almasri, W.E.; Ayyash, M.; Al-Holy, M.A.; Jaradat, Z.W.; Obaid, R.S.; Holley, R.A. Inactivation of Salmonella spp. in tahini using plant essential oil extracts. Food Microbiol. 2020, 86, 103338. [Google Scholar] [CrossRef]
- Olaimat, A.N.; Al-Holy, M.A.; Abu Ghoush, M.; Al-Nabulsi, A.A.; Holley, R.A. Control of Salmonella enterica and Listeria monocytogenes in hummus using allyl isothiocyanate. Int. J. Food Microbiol. 2018, 278, 73–80. [Google Scholar] [CrossRef]
- Osaili, T.M.; Al-Nabulsi, A.A.; Aljaafreh, T.F.; Olaimat, A.N. Use of gamma radiation to inactivate stressed Salmonella spp.; Escherichia coli O157:H7 and Listeria monocytogenes in tahini halva. LWT Food Sci. Technol. 2018, 98, 438–443. [Google Scholar] [CrossRef]
- Salazar, J.K.; Natarajan, V.; Stewart, D.; Fay, M.; Gonsalves, L.J.; Mhetras, T.; Sule, C.; Tortorello, M.L. Listeria monocytogenes growth kinetics in refrigerated ready-to-eat dips and dip components. PLoS ONE 2020, 15, e0235472. [Google Scholar] [CrossRef] [PubMed]
- Ren, X.; Lou, G.; Shen, Q. Effect of high-pressure thermal sterilization on quality attributes of compound sesame paste. J. Chin. Inst. Food Sci. Tech. 2016, 16, 140–148. (In Chinese) [Google Scholar] [CrossRef]
- Brockmann, S.O.; Piechotowski, I.; Kimmig, P. Salmonella in sesame seed products. J. Food Prot. 2004, 67, 178–180. [Google Scholar] [CrossRef]
- Unicomb, L.E.; Simmons, G.; Merritt, T.; Gregory, J.; Nicol, C.; Jelfs, P.; Kirk, M.; Tan, A.; Thomson, R.; Adamopoulos, J.; et al. Sesame seed products contaminated with Salmonella: Three outbreaks associated with tahini. Epidemiol. Infect. 2005, 133, 1065–1072. [Google Scholar] [CrossRef] [PubMed]
- Blackwell, R.; Blaylock, M.; Merid, S.; DaviesCole, J.; Gibson, A.; Herdman, D.; Sudler, R.; Lee, H.; Corvese, K.; Levine, S. Multistate outbreak of Salmonella serotype Bovismorbificans infections associated with hummus and tahini—United States, 2011. Morb. Mortal. Wkly. Rep. 2012, 61, 944–947. [Google Scholar]
- Paine, S.; Thornley, C.; Wilson, M.; Dufour, M.; Sexton, K.; Miller, J.; King, G.; Bell, S.; Bandaranayake, D.; Mackereth, G. An outbreak of multiple serotypes of Salmonella in New Zealand linked to consumption of contaminated tahini imported from Turkey. Foodborne Pathog. Dis. 2014, 11, 887–892. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Multistate outbreak of Salmonella montevideo and Salmonella mbandaka Infections Linked to Tahini Sesame Paste (Final Update). 2013. Available online: https://www.cdc.gov/salmonella/montevideo-tahini-05-13/index.html (accessed on 15 February 2021).
- Meinen, A.; Simon, S.; Banerji, S.; Szabo, I.; Malorny, B.; Borowiak, M.; Hadziabdic, S.; Becker, N.; Luber, P.; Lohr, D.; et al. Salmonellosis outbreak with novel Salmonella enterica subspecies enterica serotype (11: z41: E, n, z15) attributable to sesame products in five European countries, 2016 to 2017. Eurosurveillance 2019, 24, 6–14. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Outbreak of Salmonella Infections Linked to Tahini from Achdut Ltd. 2018. Available online: https://www.cdc.gov/salmonella/concord-11-18/index.html (accessed on 15 February 2021).
- Centers for Disease Control and Prevention (CDC). Outbreak of Salmonella Infections Linked to Karawan Brand Tahini. 2019. Available online: https://www.cdc.gov/salmonella/concord-05-19/index.html (accessed on 15 February 2021).
- Olaimat, A.N.; Osaili, T.M.; Al-Holy, M.A.; Al-Nabulsi, A.A.; Obaid, R.S.; Alaboudi, A.R.; Ayyash, M.; Holley, R. Microbial safety of oily, low water activity food products: A review. Food Microbiol. 2020, 92, 103571. [Google Scholar] [CrossRef]
- Torlak, E.; Sert, D.; Serin, P. Fate of Salmonella during sesame seeds roasting and storage of tahini. Int. J. Food Microbiol. 2013, 163, 214–217. [Google Scholar] [CrossRef]
- Zhang, Y.J.N.; Keller, S.E.; Grasso-Kelley, E.M. Fate of Salmonella throughout production and refrigerated storage of tahini. J. Food Prot. 2017, 80, 940–946. [Google Scholar] [CrossRef] [PubMed]
- Aviles, B.; Klotz, C.; Smith, T.; Williams, R.; Ponder, M. Survival of Salmonella enterica serotype Tennessee during simulated gastric passage is improved by low water activity and high fat content. J. Food Prot. 2013, 76, 333–337. [Google Scholar] [CrossRef] [PubMed]
- Bhargava, K.; Conti, D.S.; da Rocha, S.R.P.; Zhang, Y.F. Application of an oregano oil nanoemulsion to the control of foodborne bacteria on fresh lettuce. Food Microbiol. 2015, 47, 69–73. [Google Scholar] [CrossRef]
- Chang, S.S.; Lu, W.Y.W.; Park, S.H.; Kang, D.H. Control of foodborne pathogens on ready-to-eat roast beef slurry by epsilon-polylysine. Int. J. Food Microbiol. 2010, 141, 236–241. [Google Scholar] [CrossRef]
- Mortazavi, N.; Aliakbarlu, J. Antibacterial effects of ultrasound, cinnamon essential oil, and their combination against Listeria monocytogenes and Salmonella Typhimurium in milk. J. Food Sci. 2019, 84, 3700–3706. [Google Scholar] [CrossRef] [PubMed]
- Ha, J.W.; Kang, D.H. Combining lactic acid spray with NIR radiant heating to inactivate Salmonella enterica Serovar Enteritidis on almond and pine nut kernels. Appl. Environ. Microbiol. 2015, 81, 4517–4524. [Google Scholar] [CrossRef]
- Zhang, H.C.; Tikekar, R.V.; Ding, Q.; Gilbert, A.R.; Wimsatt, S.T. Inactivation of foodborne pathogens by the synergistic combinations of food processing technologies and food-grade compounds. Compr. Rev. Food. Sci. Food Saf. 2020, 19, 2110–2138. [Google Scholar] [CrossRef]
- Kou, X.X.; Li, R.; Hou, L.X.; Huang, Z.; Ling, B.; Wang, S.J. Performance of a heating block system designed for studying the heat resistance of bacteria in foods. Sci. Rep. 2016, 6, 1–12. [Google Scholar] [CrossRef]
- Bian, X.; Evivie, S.E.; Muhammad, Z.; Luo, G.W.; Liang, H.Z.; Wang, N.N.; Huo, G.C. In vitro assessment of the antimicrobial potentials of Lactobacillus helveticus strains isolated from traditional cheese in Sinkiang China against food-borne pathogens. Food. Funct. 2016, 7, 789–797. [Google Scholar] [CrossRef]
- Wang, X.; Devlieghere, F.; Geeraerd, A.; Uyttendaele, M. Thermal inactivation and sublethal injury kinetics of Salmonella enterica and Listeria monocytogenes in broth versus agar surface. Int. J. Food Microbiol. 2017, 243, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Sanz-Puig, M.; Santos-Carvalho, L.; Cunha, L.M.; Pina-Perez, M.C.; Martinez, A.; Rodrigo, D. Effect of pulsed electric fields (PEF) combined with natural antimicrobial by-products against S. typhimurium. Innov. Food Sci. Emerg. Technol. 2016, 37, 322–328. [Google Scholar] [CrossRef]
- Espina, L.; Somolinos, M.; Ouazzou, A.A.; Condon, S.; Garcia-Gonzalo, D.; Pagan, R. Inactivation of Escherichia coli O157:H7 in fruit juices by combined treatments of citrus fruit essential oils and heat. Int. J. Food Microbiol. 2012, 159, 9–16. [Google Scholar] [CrossRef] [PubMed]
- He, Y.S.; Li, Y.; Salazar, J.K.; Yang, J.Y.; Tortorello, M.L.; Zhang, W. Increased water activity reduces the thermal resistance of Salmonella enterica in peanut butter. Appl. Environ. Microbiol. 2013, 79, 4763–4767. [Google Scholar] [CrossRef] [PubMed]
- Krapf, T.; Gantenbein-Demarchi, C. Thermal inactivation of Salmonella spp. during conching. LWT-Food Sci. Technol. 2010, 43, 720–723. [Google Scholar] [CrossRef]
- Gautam, B.; Govindan, B.N.; Gänzle, M.; Roopesh, M.S. Influence of water activity on the heat resistance of Salmonella enterica in selected low-moisture foods. Int. J. Food Microbiol. 2020, 334, 108813. [Google Scholar] [CrossRef]
- Li, C.C.; Huang, L.H.; Chen, J.Q. Comparative study of thermal inactivation kinetics of Salmonella spp. in peanut butter and peanut butter spread. Food Control 2014, 45, 143–149. [Google Scholar] [CrossRef]
- Ban, C.; Lee, D.H.; Jo, Y.; Bae, H.; Seong, H.; Kim, S.O.; Lim, S.; Choi, Y.J. Use of superheated steam to inactivate Salmonella enterica serovars Typhimurium and Enteritidis contamination on black peppercorns, pecans, and almonds. J. Food Eng. 2018, 222, 284–291. [Google Scholar] [CrossRef]
- Gurtler, J.B.; Juneja, V.K.; Jones, D.R.; Purohit, A. Thermal inactivation kinetics of three heat-resistant Salmonella strains in whole liquid egg. J. Food Prot. 2019, 82, 1465–1471. [Google Scholar] [CrossRef]
- Espina, L.; Condon, S.; Pagan, R.; Garcia-Gonzalo, D. Synergistic effect of orange essential oil or (+)-limonene with heat treatments to inactivate Escherichia coli O157:H7 in orange juice at lower intensities while maintaining hedonic acceptability. Food Bioprocess Technol. 2014, 7, 471–481. [Google Scholar] [CrossRef]
- Maria Cava-Roda, R.; Taboada, A.; Palop, A.; Lopez-Gomez, A.; Marin-Iniesta, F. Heat resistance of Listeria monocytogenes in semi-skim milk supplemented with vanillin. Int. J. Food Microbiol. 2012, 157, 314–318. [Google Scholar] [CrossRef]
- Lee, S.Y.; Oh, S.W.; Ghung, H.J.; Reyes-De-Corchuera, J.I.; Powers, J.R.; Kang, D.H. Reduction of Salmonella enterica Serovar Enteritidis on the surface of raw shelled almonds by exposure to steam. J. Food Prot. 2006, 69, 591–595. [Google Scholar] [CrossRef] [PubMed]
- Mani-Lopez, E.; Garcia, H.S.; Lopez-Malo, A. Organic acids as antimicrobials to control Salmonella in meat and poultry products. Food Res. Int. 2012, 45, 713–721. [Google Scholar] [CrossRef]
- Farakos, S.M.S.; Pouillot, R.; Keller, S.E. Salmonella survival kinetics on pecans, hazelnuts, and pine nuts at various water activities and temperatures. J. Food Prot. 2017, 80, 879–885. [Google Scholar] [CrossRef] [PubMed]
- Park, E.J.; Oh, S.W.; Kang, D.H. Fate of Salmonella Tennessee in peanut butter at 4 and 22 °C. J. Food Sci. 2008, 73, M82–M86. [Google Scholar] [CrossRef] [PubMed]
- Al-Nabulsi, A.A.; Olaimat, A.N.; Osaili, T.M.; Shaker, R.R.; Elabedeen, N.Z.; Jaradat, Z.W.; Abushelaibi, A.; Holley, R.A. Use of acetic and citric acids to control Salmonella Typhimurium in tahini (sesame paste). Food Microbiol. 2014, 42, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Golden, D.A.; Critzer, F.J.; Davidson, P.M. Antimicrobial activity of cinnamaldehyde, carvacrol, and lauric arginate against Salmonella Tennessee in a glycerol-sucrose model and peanut paste at different fat concentrations. J. Food Prot. 2015, 78, 1488–1495. [Google Scholar] [CrossRef]
- Smith-Palmer, A.; Stewart, J.; Fyfe, L. The potential application of plant essential oils as natural food preservatives in soft cheese. Food Microbiol. 2001, 18, 463–470. [Google Scholar] [CrossRef]
- Gutierrez, J.; Barry-Ryan, C.; Bourke, R. The antimicrobial efficacy of plant essential oil combinations and interactions with food ingredients. Int. J. Food Microbiol. 2008, 124, 91–97. [Google Scholar] [CrossRef]
- Min, K.-J.; Kwon, K.-Y.; Yoon, K.-S. Effect of various antimicrobials on the growth kinetics of foodborne pathogens in ready-to-eat, pyeonyuk (cooked and pressed pork). Food Sci. Biotechnol. 2010, 19, 99–106. [Google Scholar] [CrossRef]
- Geornaras, I.; Sofos, J.N. Activity of epsilon-polylysine against Escherichia coli O157: H7, Salmonella Typhimurium, and Listeria monocytogenes. J. Food Sci. 2005, 70, M404–M408. [Google Scholar] [CrossRef]
- Geornaras, I.; Yoon, Y.; Belk, K.E.; Smith, G.C.; Sofos, J.N. Antimcrobial activity of epsilon-polylyrsine against Escherichia coli O157:H7, Salmonella Typhimurium, and Listeria manocytogenes in various food extracts. J. Food Sci. 2007, 72, M330–M334. [Google Scholar] [CrossRef]
- Osaili, T.M.; Al-Nabulsi, A.A.; Jaradat, Z.; Shaker, R.R.; Alomari, D.Z.; Al-Dabbas, M.M.; Alaboudi, A.R.; Al-Natour, M.Q.; Holley, R.A. Survival and growth of Salmonella Typhimurium, Escherichia coli O157: H7 and Staphylococcus aureus in eggplant dip during storage. Int. J. Food Microbiol. 2015, 198, 37–42. [Google Scholar] [CrossRef]
- Al-Rousan, W.M.; Olaimat, A.N.; Osaili, T.M.; Al-Nabulsi, A.A.; Ajo, R.Y.; Holley, R.A. Use of acetic and citric acids to inhibit Escherichia coli O157:H7, Salmonella Typhimurium and Staphylococcus aureus in tabbouleh salad. Food Microbiol. 2018, 73, 61–66. [Google Scholar] [CrossRef]
- Kang, J.H.; Song, K.B. Inactivation of pre-existing bacteria and foodborne pathogens on perilla leaves using a combined treatment with an organic acid and a surfactant. Hortic. Environ. Biotechnol. 2015, 56, 195–199. [Google Scholar] [CrossRef]
- Park, S.H.; Choi, M.R.; Park, J.W.; Park, K.H.; Chung, M.S.; Ryu, S.; Kang, D.H. Use of organic acids to inactivate Escherichia coli O157:H7, Salmonella Typhimurium, and Listeria monocytogenes on organic fresh apples and lettuce. J. Food Sci. 2011, 76, M293–M298. [Google Scholar] [CrossRef]
- Borges, T.J.; Moretti, L.K.; Silva, M.M.N.; Tondo, E.C.; Pereira, K.S. Salmonella sensitivity to sodium hypochlorite and citric acid in washing water of lettuce residues. J. Food Saf. 2020, 40, e12748. [Google Scholar] [CrossRef]
- Rao, J.J.; Chen, B.C.; McClement, D.J. Improving the efficacy of essential oils as antimicrobials in foods: Mechanisms of action. Ann. Rev. Food Sci. Technol. 2019, 10, 365–387. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.W.; Shi, C.; Liu, Z.J.; Pan, F.G.; Meng, R.Z.; Bu, X.J.; Xing, H.Q.; Deng, Y.H.; Guo, N.; Yu, L. Antibacterial activity and mode of action of epsilon-polylysine against Escherichia coli O157: H7. J. Med. Microbiol. 2018, 67, 838–845. [Google Scholar] [CrossRef] [PubMed]
- Mohan, A.; Purohit, A.S. Anti-Salmonella activity of pyruvic and succinic acid in combination with oregano essential oil. Food Control 2020, 110, 106960. [Google Scholar] [CrossRef]
- Kwon, S.A.; Song, W.J.; Kang, D.H. Combination effect of saturated or superheated steam and lactic acid on the inactivation of Escherichia coli O157:H7, Salmonella typhimurium and Listeria monocytogenes on cantaloupe surfaces. Food Microbiol. 2019, 82, 342–348. [Google Scholar] [CrossRef] [PubMed]
- Dogruyol, H.; Mol, S.; Cosansu, S. Increased thermal sensitivity of Listeria monocytogenes in sous-vide salmon by oregano essential oil and citric acid. Food Microbiol. 2020, 90, 103496. [Google Scholar] [CrossRef] [PubMed]

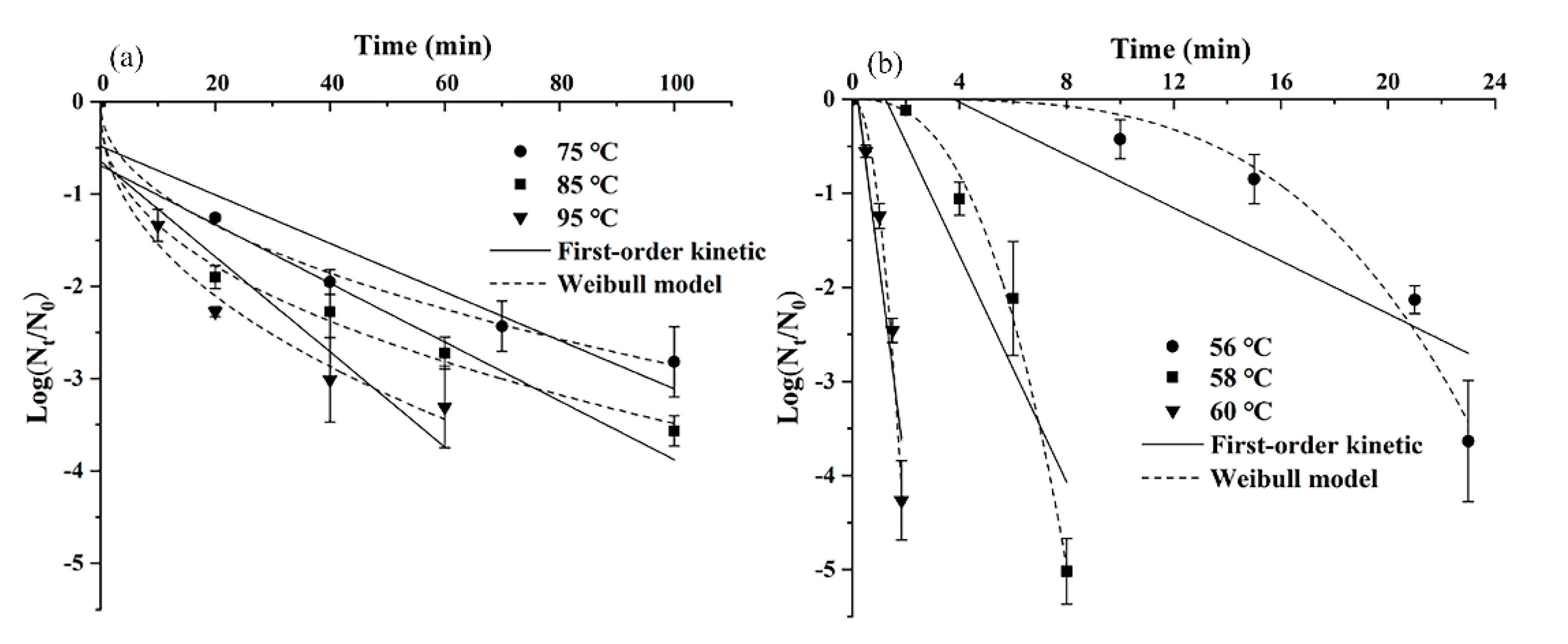
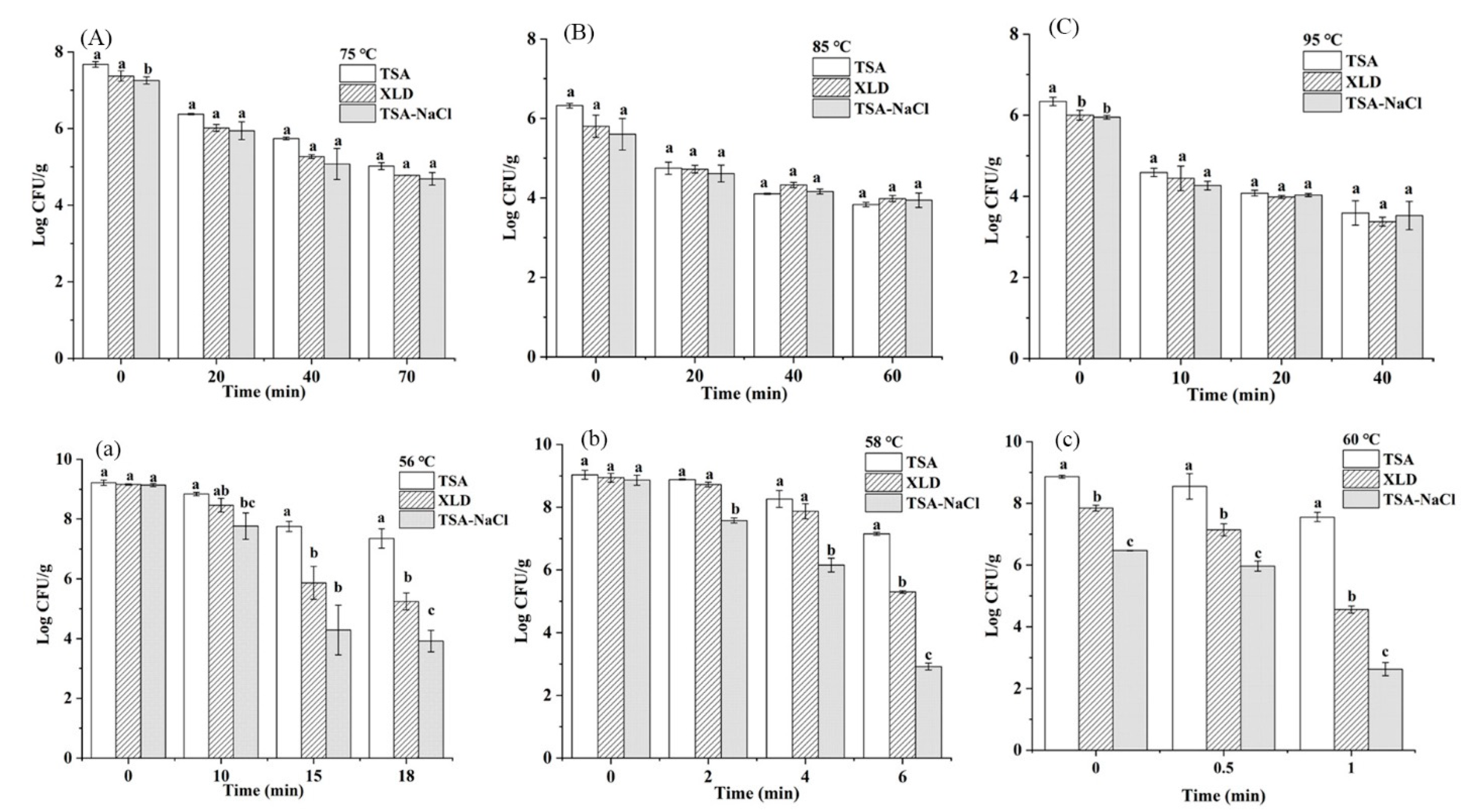

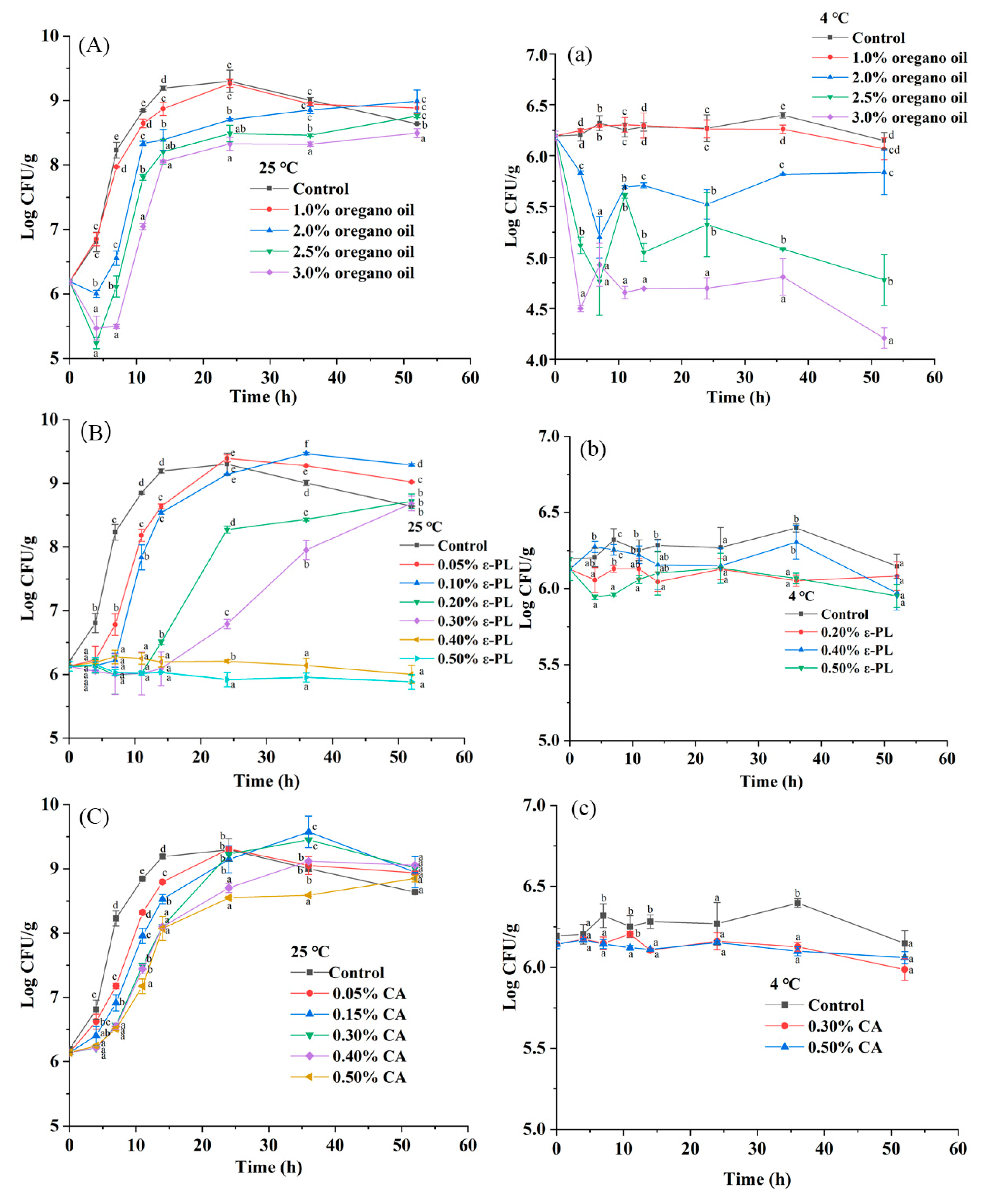
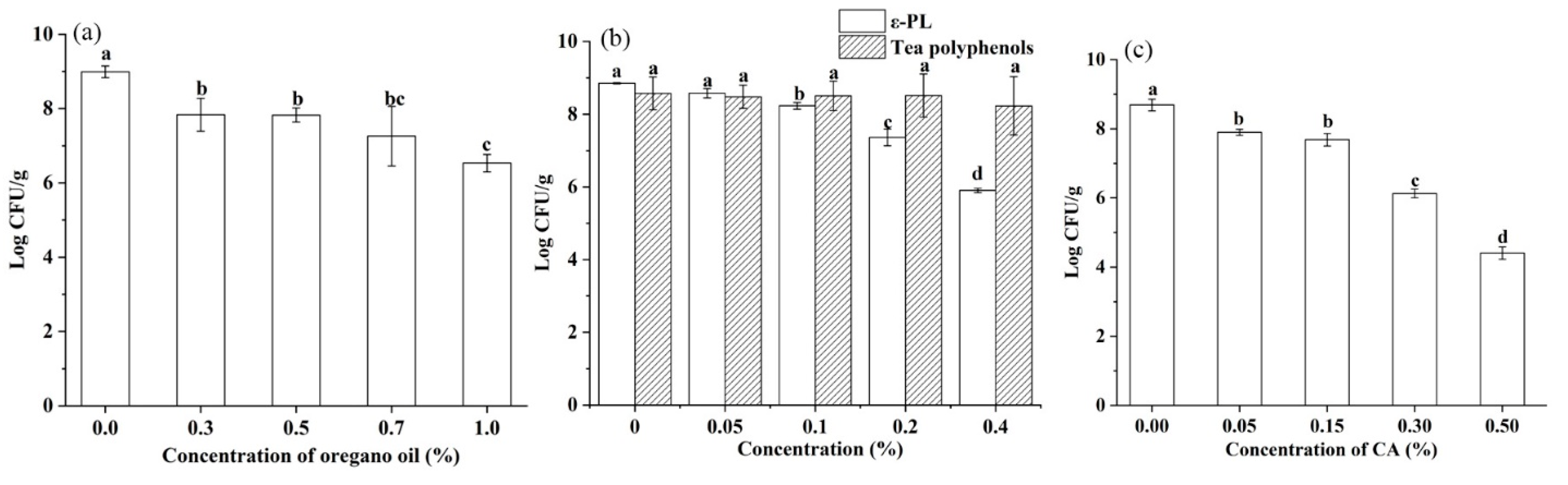
| Samples | Moisture Content (%) | aw | Temperature (°C) | First-Order Model | Weibull Model | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| D (min) | R2 | RMSE | δ | p | R2 | RMSE | ||||
| Undiluted tahini | 0.629 ± 0.001 | 0.256 ± 0.002 | 75 | 38.43 ± 4.62 | 0.885 | 0.435 | 10.65 ± 1.18 | 0.47 ± 0.07 | 0.996 | 0.078 |
| 85 | 31.43 ± 1.41 | 0.856 | 0.581 | 5.21 ± 1.71 | 0.42 ± 0.04 | 0.994 | 0.114 | |||
| 95 | 19.66 ± 2.74 | 0.851 | 0.601 | 3.84 ± 0.67 | 0.45 ± 0.05 | 0.985 | 0.190 | |||
| Diluted tahini | 50.240 ± 1.290 | 1.000 ± 0.000 | 56 | 7.17 ± 0.76 | 0.775 | 0.810 | 16.52 ± 1.42 | 3.91 ± 1.21 | 0.973 | 0.279 |
| 58 | 1.67 ± 0.17 | 0.853 | 0.913 | 4.38 ± 0.52 | 2.70 ± 0.48 | 0.993 | 0.195 | |||
| 60 | 0.46 ± 0.04 | 0.906 | 0.602 | 0.92 ± 0.08 | 2.09 ± 0.39 | 0.985 | 0.237 | |||
| Foodborne Pathogens | Essential Oils (EOs) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Oregano | Cinnamon | Thyme | Tea tree | Eucalyptus | Peppermint | Garlic | Turmeric | Control | |
| S. montevideo CICC21588 | 23.42 ± 1.02 a | 22.58 ± 0.92 a | 16.28± 0.66 b | 12.50 ± 0.41 c | 8.75 ± 0.35 d | 8.00 ± 0.00 d | 8.00 ± 0.00 d | 8.00 ± 0.00 d | 8.00 ± 0.00 d |
| S. Typhimurium CMCC50115 | 24.00 ± 0.41 a | 22.00 ± 0.41 b | 20.63 ± 0.48 c | 14.38 ± 0.48 e | 15.25 ± 0.65 d | 8.83 ± 0.29 g | 9.76 ± 0.35 f | 8.00 ± 0.00 h | 8.00 ± 0.00 h |
| S. Mbandaka NYS5-8 | 21.38 ± 0.48 a | 19.88 ± 0.75 b | 17.38 ± 0.48 c | 13.00 ± 0.00 d | 11.00 ± 0.00 e | 8.00 ± 0.00 g | 9.25 ± 0.35 f | 8.00 ± 0.00 g | 8.00 ± 0.00 g |
| S. enteritidis R8-1-1 | 22.38 ± 0.48 a | 22.75 ± 0.65 a | 16.00 ± 0.41 b | 14.25 ± 0.29 c | 11.00 ± 0.00 d | 8.00 ± 0.00 e | 8.00 ± 0.00 e | 8.00 ± 0.00 e | 8.00 ± 0.00 e |
| Foodborne Pathogens | Control | ε-PL | Tea Polyphenol | Citric Acid | Lactic Acid | ||||
|---|---|---|---|---|---|---|---|---|---|
| 10% | 5% | 10% | 5% | 10% | 5% | 10% | 5% | ||
| S. montevideo CICC21588 | 8.00 ± 0.00 f | 23.50 ± 0.71 c | 20.50 ± 0.71 d | 11.38 ± 0.75 e | 8.00 ± 0.00 f | 27.61 ± 0.48 a | 23.13 ± 0.85 c | 26.08 ± 0.38 b | 21.00 ± 0.41 d |
| S. Typhimurium CMCC50115 | 8.00 ± 0.00 f | 17.50 ± 0.50 d | 12.75 ± 0.65 e | 18.50 ± 0.71 d | 13.33 ± 0.58 e | 25.75 ± 1.19 a | 21.38 ± 0.48 c | 24.63 ± 0.48 b | 20.38 ± 0.48 c |
| S. Mbandaka NYS5-8 | 8.00 ± 0.00 h | 16.25 ± 0.35 e | 11.50 ± 0.71 g | 14.75 ± 0.35 f | 11.75 ± 0.35 g | 26.50 ± 0.50 a | 22.25 ± 0.50 c | 24.00 ± 0.41 b | 17.75 ± 0.29 d |
| S. enteritidis R8-1-1 | 8.00 ± 0.00 f | 18.13 ± 0.63 c | 15.00 ± 0.41 d | 13.00 ± 1.47 e | 8.00 ± 0.00 f | 27.70 ± 0.68 a | 24.09 ± 0.33 b | 27.20 ± 0.53 a | 23.68 ± 0.35 b |
| Antimicrobials | Day 0 | 25 °C | 4 °C | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 (52 h) | 3 | 7 | 1 | 2 (52 h) | 3 | 7 | |||
| Control | 0% | 6.19 ± 0.05 a | 9.30 ± 0.17 a | 8.64 ± 0.01 cd | 8.70 ± 0.14 abc | DE | 6.27±0.13 a | 6.15 ± 0.08 a | 6.14 ± 0.08 a | 6.22 ± 0.19 a |
| Oregano oil | 1% | 6.19 ± 0.05 a | 9.26 ± 0.06 a | 8.88 ± 0.08 ab | 8.54 ± 0.04 bc | 8.48 ± 0.19 c | 6.26 ± 0.09 a | 6.07 ± 0.11 a | 6.06 ± 0.05 ab | 5.83 ± 0.05 ab |
| 2% | 6.19 ± 0.05 a | 8.70 ± 0.02 b | 8.99 ± 0.17 a | 8.93 ± 0.04 ab | 8.44 ± 0.06 c | 5.52 ± 0.14 b | 5.84 ± 0.22 b | 5.99 ± 0.02 ab | 5.50 ± 0.40 b | |
| 3% | 6.19 ± 0.05 a | 8.33 ± 0.10 c | 8.49 ± 0.07 d | 8.67 ± 0.36 abc | 8.74 ± 0.04 b | 4.70 ± 0.10 c | 4.21 ± 0.10 c | 4.18 ± 0.32 c | 2.63 ± 0.46 e | |
| ε-PL | 0.2% | 6.13 ± 0.08 a | 8.27 ± 0.06 c | 8.72 ± 0.12 bc | 8.95 ± 0.13 a | 9.36 ± 0.004 a | 6.13 ± 0.07 a | 6.08 ± 0.10 a | 6.04 ± 0.03 ab | 6.02 ± 0.11 a |
| 0.4% | 6.13 ± 0.08 a | 6.21 ± 0.02 d | 6.00 ± 0.14 e | 7.48 ± 0.25 d | 9.29 ± 0.16 a | 6.15 ± 0.12 a | 5.97 ± 0.11 ab | 5.99 ± 0.02 ab | 4.58 ± 0.17 c | |
| 0.5% | 6.13 ± 0.08 a | 5.92 ± 0.11 e | 5.88 ± 0.11 e | 6.16 ± 0.06 e | 9.31 ± 0.04 a | 6.13 ± 0.10 a | 5.95 ± 0.08 ab | 5.88 ± 0.06 b | 3.68 ± 0.59 d | |
| CA | 0.3% | 6.14 ± 0.03 a | 9.23 ± 0.03 a | 9.02 ± 0.06 a | 8.44 ± 0.07 c | DE | 6.16 ± 0.05 a | 5.99 ± 0.07 ab | 6.14 ± 0.11 a | 6.03 ± 0.11 a |
| 0.5% | 6.14 ± 0.03 a | 8.55 ± 0.03 b | 8.85 ± 0.05 abc | 8.57 ± 0.04 abc | DE | 6.15 ± 0.01 a | 6.06 ± 0.04 a | 6.12 ± 0.10 a | 6.04 ± 0.15 a | |
| Temperature (°C) | Weibull Model | ||||
|---|---|---|---|---|---|
| δ | p | R2 | RMSE | ||
| Diluted tahini | 56 | 16.52 ± 1.42 a | 3.91 ± 1.21 | 0.973 | 0.279 |
| 58 | 4.38 ± 0.52 b | 2.70 ± 0.48 | 0.993 | 0.195 | |
| Diluted tahini+1% oregano oil | 56 | 3.98 ± 0.64 b | 1.30 ± 0.21 | 0.983 | 0.201 |
| 58 | 1.94 ± 0.06 c | 4.57 ± 0.39 | 0.994 | 0.123 | |
| Diluted tahini+0.4% ε-PL | 56 | 4.20 ± 0.13 b | 1.87 ± 0.02 | 0.976 | 0.333 |
| 58 | 1.25 ± 0.03 c | 2.04 ± 0.19 | 0.991 | 0.194 | |
| Diluted tahini+0.3% CA | 56 | 4.92 ± 1.13 b | 2.24 ± 0.73 | 0.971 | 0.313 |
| 58 | 1.35 ± 0.11 c | 2.66 ± 0.30 | 0.980 | 0.299 | |
| Samples | aw | 75 °C + Holding Time (min) | Weibull Model | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Population Reduction (Log CFU/g) | |||||||||
| 20 | 40 | 70 | 100 | δ | p | R2 | RMSE | ||
| Undiluted tahini | 0.256 ± 0.002 | 1.26 ± 0.04 a | 1.95 ± 0.13 a | 2.43 ± 0.27 a | 2.82± 0.38 ab | 10.65 ± 1.18 a | 0.47 ± 0.07 | 0.996 | 0.078 |
| Undiluted tahini+2% oregano oil | 1.13 ± 0.01 a | 1.79 ± 0.02 a | 2.36 ± 0.18 a | 2.52 ± 0.21 a | 12.48 ± 1.46 a | 0.53 ± 0.03 | 0.986 | 0.028 | |
| Undiluted tahini | 0.283 ± 0.012 | 1.58 ± 0.02 b | 1.68 ± 0.10 a | 2.84 ± 0.12 ab | 3.16 ± 0.10 bc | 10.29 ± 0.35 a | 0.51 ± 0.01 | 0.965 | 0.266 |
| Undiluted tahini+0.3% CA | 1.82 ± 0.16 c | 2.22 ± 0.02 ab | 2.86 ± 0.09 abc | 3.32 ± 0.06 bc | 4.72 ± 1.26 b | 0.39 ± 0.03 | 0.998 | 0.059 | |
| Undiluted tahini | 0.335 ± 0.004 | 1.81 ± 0.01 c | 2.66 ± 0.56 bc | 3.37 ± 0.21 c | 3.54 ± 0.33 c | 3.81 ± 0.96 b | 0.40 ± 0.01 | 0.991 | 0.160 |
| Undiluted tahini+0.4% ε-PL | 2.33 ± 0.14 d | 2.84 ± 0.19 c | 3.23 ± 0.28 bc | 3.48 ± 0.07 c | 0.61 ± 0.09 c | 0.23 ± 0.03 | 0.999 | 0.016 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, Y.; Guan, X.; Lin, B.; Li, R.; Wang, S. Oregano Oil, Epsilon-Polylysine and Citric Acid Assisted Inactivation of Salmonella in Two Kinds of Tahini during Thermal Treatment and Storage. Foods 2021, 10, 1272. https://doi.org/10.3390/foods10061272
Xu Y, Guan X, Lin B, Li R, Wang S. Oregano Oil, Epsilon-Polylysine and Citric Acid Assisted Inactivation of Salmonella in Two Kinds of Tahini during Thermal Treatment and Storage. Foods. 2021; 10(6):1272. https://doi.org/10.3390/foods10061272
Chicago/Turabian StyleXu, Yuanmei, Xiangyu Guan, Biying Lin, Rui Li, and Shaojin Wang. 2021. "Oregano Oil, Epsilon-Polylysine and Citric Acid Assisted Inactivation of Salmonella in Two Kinds of Tahini during Thermal Treatment and Storage" Foods 10, no. 6: 1272. https://doi.org/10.3390/foods10061272
APA StyleXu, Y., Guan, X., Lin, B., Li, R., & Wang, S. (2021). Oregano Oil, Epsilon-Polylysine and Citric Acid Assisted Inactivation of Salmonella in Two Kinds of Tahini during Thermal Treatment and Storage. Foods, 10(6), 1272. https://doi.org/10.3390/foods10061272






