Investigating the Use of Ultraviolet Light Emitting Diodes (UV-LEDs) for the Inactivation of Bacteria in Powdered Food Ingredients
Abstract
1. Introduction
2. Materials and Methods
2.1. UV Devices
2.2. Bacterial Strains and Growth Conditions
2.3. Sample Preparation
2.3.1. Petri Dish Surfaces
2.3.2. Seasoning Powders
2.4. UV and UV-LED Inactivation
2.5. Bacterial Enumeration
2.6. Modelling of Bacterial Inactivation Kinetics
2.6.1. Log-Linear Model
2.6.2. Biphasic Model
2.6.3. Weibull Model
2.6.4. Geeraerd shoulder–tail Model
2.7. Statistical Analysis
3. Results
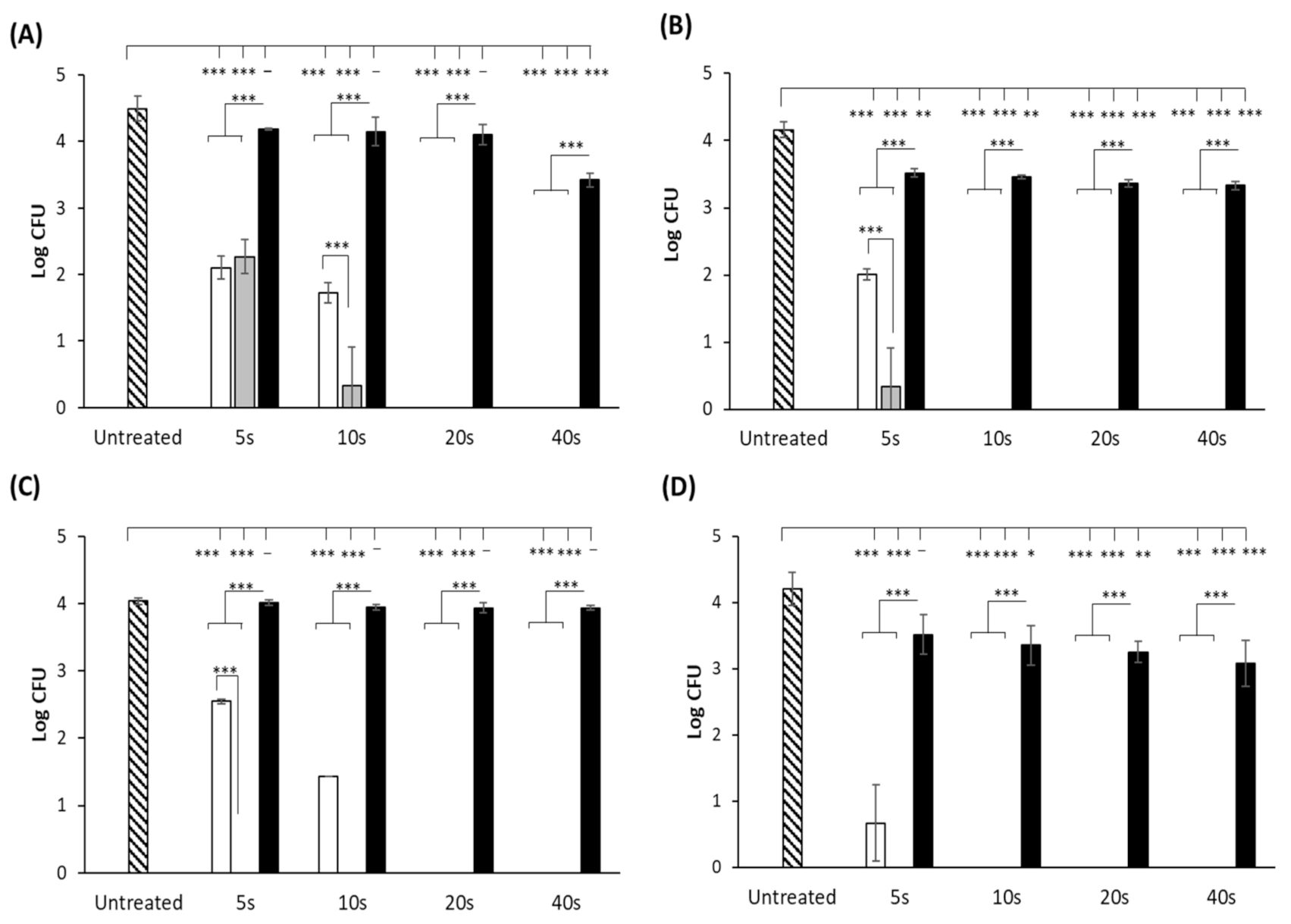
3.1. UV-LED Surface Inactivation of Microorganisms
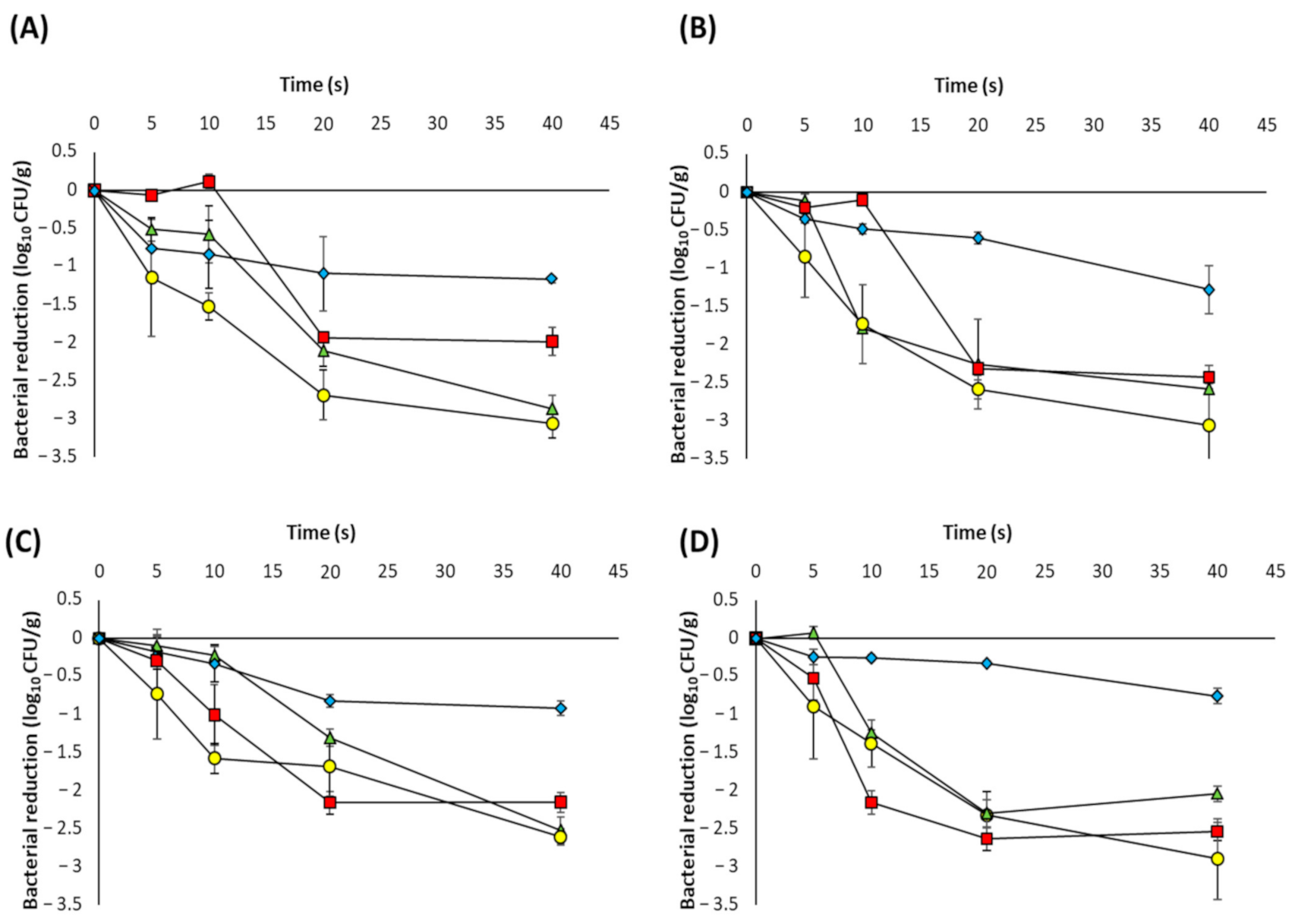
3.2. UVC-LED Inactivation of Microorganisms in Powdered Ingredients
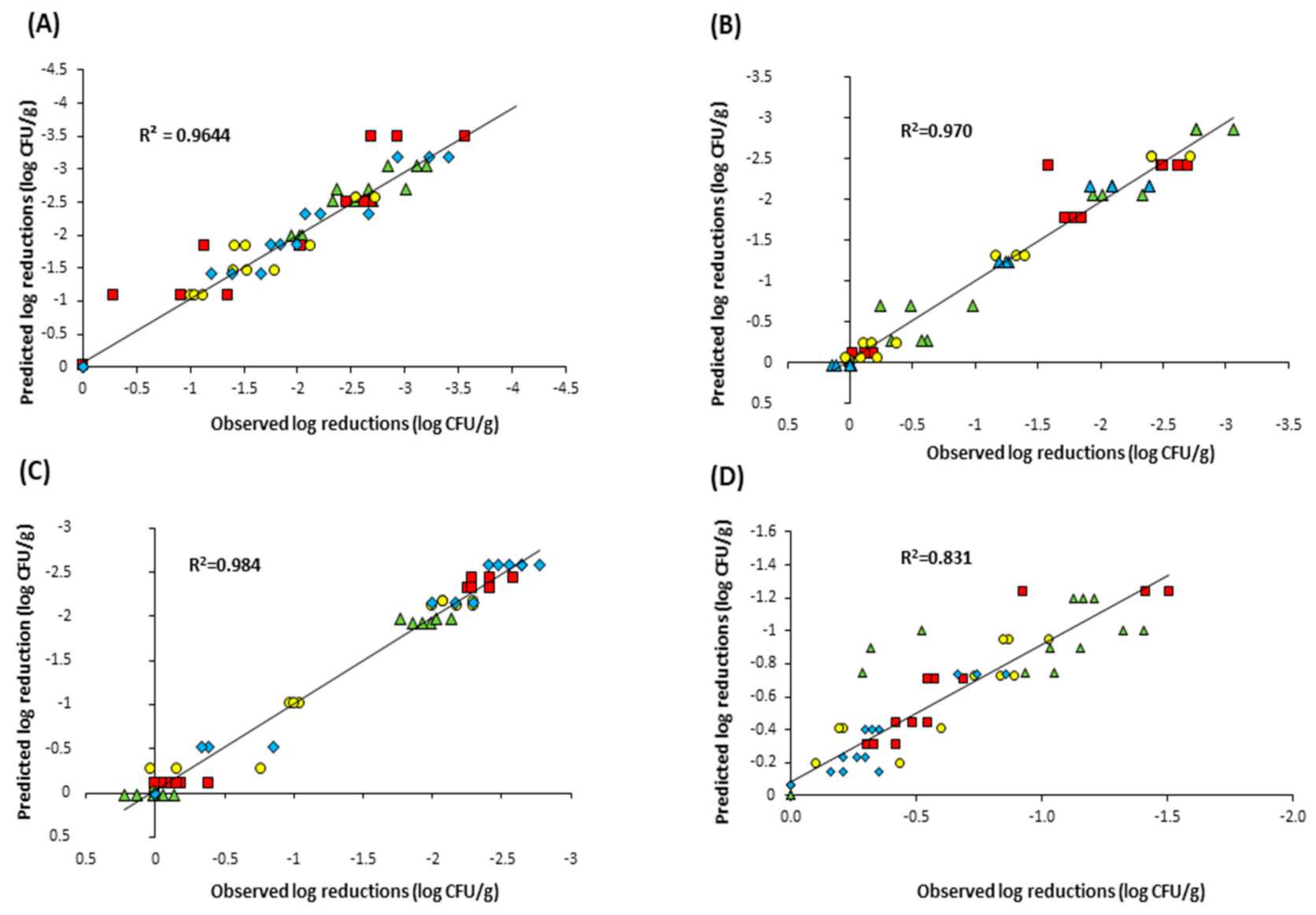
3.3. Model Fitting
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Van Doren, J.M.; Neil, K.P.; Parish, M.; Gieraltowski, L.; Gould, L.H.; Gombas, K.L. Foodborne illness outbreaks from microbial contaminants in spices, 1973–2010. Food Microbiol. 2013, 36, 456–464. [Google Scholar] [CrossRef] [PubMed]
- Moreira, P.L.; Lourencao, T.B.; Pinto, J.P.A.N.; Rall, V.L.M. Microbiological quality of spices marketed in the city of Botucatu, Sao Paulo, Brazil. J. Food Prot. 2009, 72, 421–424. [Google Scholar] [CrossRef] [PubMed]
- Sagoo, S.K.; Little, C.L.; Greenwood, M.; Mithani, V.; Grant, K.A.; McLauchlin, J.; de Pinna, E.; Threlfall, E.J. Assessment of the microbiological safety of dried spices and herbs from production and retail premises in the United Kingdom. Food Microbiol. 2009, 26, 39–43. [Google Scholar] [CrossRef] [PubMed]
- Sospedra, I.; Soriano, J.M.; Mañes, J. Assessment of the microbiological safety of dried spices and herbs commercialized in Spain. Plant. Foods Hum. Nutr. 2010, 65, 364–368. [Google Scholar] [CrossRef]
- Laroche, C.; Fine, F.; Gervais, P. Water activity affects heat resistance of microorganisms in food powders. Int. J. Food Microbiol. 2005, 97, 307–315. [Google Scholar] [CrossRef]
- Santillana Farakos, S.M.; Frank, J.F.; Schaffner, D.W. Modeling the influence of temperature, water activity and water mobility on the persistence of Salmonella in low-moisture foods. Int. J. Food Microbiol. 2013, 166, 280–293. [Google Scholar] [CrossRef]
- Rhim, J.W.; Hong, S.I. Effect of water activity and temperature on the color change of red pepper (Capsicum annuum L.) powder. Food Sci. Biotechnol. 2011, 20, 215–222. [Google Scholar] [CrossRef]
- Baechler, R.; Clerc, M.-F.; Ulrich, S.; Benet, S. Physical changes in heat-treated whole milk powder. Lait 2005, 85, 305–314. [Google Scholar] [CrossRef]
- Duncan, S.E.; Moberg, K.; Amin, K.N.; Wright, M.; Newkirk, J.J.; Ponder, M.A.; Acuff, G.R.; Dickson, J.S. Processes to preserve spice and herb quality and sensory integrity during pathogen inactivation. J. Food Sci. 2017, 82, 1208–1215. [Google Scholar] [CrossRef]
- Jung, K.; Song, B.S.; Kim, M.J.; Moon, B.G.; Go, S.M.; Kim, J.K.; Lee, Y.J.; Park, J.H. Effect of X-ray, gamma ray, and electron beam irradiation on the hygienic and physicochemical qualities of red pepper powder. LWT Food Sci. Technol. 2015, 63, 846–851. [Google Scholar] [CrossRef]
- Holck, A.; Liland, K.H.; Carlehög, M.; Heir, E. Reductions of Listeria monocytogenes on cold-smoked and raw salmon fillets by UV-C and pulsed UV light. Innov. Food Sci. Emerg. Technol. 2018, 50, 1–10. [Google Scholar] [CrossRef]
- Keklik, N.; Demirci, A.; Puri, V.; Heinemann, P. Modeling the inactivation of Salmonella Typhimurium, Listeria monocytogenes and Salmonella Enteritidis on poultry products exposed to pulsed UV light. J. Food Prot. 2012, 75, 281–288. [Google Scholar] [CrossRef]
- McLeod, A.; Hovde Liland, K.; Haugen, J.-E.; Sørheim, O.; Myhrer, K.S.; Holck, A.L. Chicken fillets subjected to UV-C and pulsed UV light: Reduction of pathogenic and spoilage bacteria, and changes in sensory quality. J. Food Saf. 2018, 38, e12421. [Google Scholar] [CrossRef]
- Unluturk, S.; Atilgan, M.R.; Baysal, A.H.; Unluturk, M.S. Modeling inactivation kinetics of liquid egg white exposed to UV-C irradiation. Int. J. Food Microbiol. 2010, 142, 341–347. [Google Scholar] [CrossRef]
- Arroyo, C.; Dorozko, A.; Gaston, E.; O’Sullivan, M.; Whyte, P.; Lyng, J.G. Light based technologies for microbial inactivation of liquids, bead surfaces and powdered infant formula. Food Microbiol. 2017, 67, 49–57. [Google Scholar] [CrossRef]
- Cheon, H.L.; Shin, J.Y.; Park, K.H.; Chung, M.S.; Kang, D.H. Inactivation of foodborne pathogens in powdered red pepper (Capsicum annuum L.) using combined UV-C irradiation and mild heat treatment. Food Control. 2015, 50, 441–445. [Google Scholar] [CrossRef]
- Condón-Abanto, S.; Condón, S.; Raso, J.; Lyng, J.G.; Álvarez, I. Inactivation of Salmonella Typhimurium and Lactobacillus plantarum by UV-C light in flour powder. Innov. Food Sci. Emerg. Technol. 2016, 35, 1–8. [Google Scholar] [CrossRef]
- Fine, F.; Gervais, P. Efficiency of pulsed UV light for microbial decontamination of food powders. J. Food Prot. 2004, 67, 787–792. [Google Scholar] [CrossRef]
- Ha, J.W.; Kang, D.H. Simultaneous near-infrared radiant heating and UV radiation for inactivating Escherichia coli O157: H7 and Salmonella enterica serovar Typhimurium in powdered red pepper (Capsicum annuum L.). Appl. Environ. Microbiol. 2013, 79, 6568–6575. [Google Scholar] [CrossRef]
- Ha, J.W.; Kang, D.H. Synergistic bactericidal effect of simultaneous near-infrared radiant heating and UV radiation against Cronobacter sakazakii in powdered infant formula. Appl. Environ. Microbiol. 2014, 80, 1858–1863. [Google Scholar] [CrossRef]
- Liu, Q.; Lu, X.; Swanson, B.G.; Rasco, B.A.; Kang, D.H. Monitoring ultraviolet (UV) radiation inactivation of Cronobacter sakazakii in dry infant formula using fourier transform infrared spectroscopy. J. Food Sci. 2012, 77, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Nicorescu, I.; Nguyen, B.; Moreau-Ferret, M.; Agoulon, A.; Chevalier, S.; Orange, N. Pulsed light inactivation of Bacillus subtilis vegetative cells in suspensions and spices. Food Control. 2013, 31, 151–157. [Google Scholar] [CrossRef]
- Dai, T.; Vrahas, M.S.; Murray, C.K.; Hamblin, M.R. Ultraviolet C irradiation: An alternative antimicrobial approach to localized infections? Expert Rev. Anti. Infect. 2012, 10, 185–195. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Kim, D.K.; Kang, D.H. Using UVC light-emitting diodes at wavelengths of 266 to 279 nanometers to inactivate foodborne pathogens and pasteurize sliced cheese. Appl. Environ. Microbiol. 2016, 82, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Clarke, S. Ultraviolet Light Disinfection in the Use of Individual Water Purification Devices; Technical Information Paper #31-006-0211; U.S. Army Public Health Command: Aberdeen Proving Ground, MD, USA, 2006; pp. 1–12.
- Hamamoto, A.; Mori, M.; Takahashi, A.; Nakano, M.; Wakikawa, N.; Akutagawa, M.; Ikehara, T.; Nakaya, Y.; Kinouchi, Y. New water disinfection system using UVA light-emitting diodes. J. Appl. Microbiol. 2007, 103, 2291–2298. [Google Scholar] [CrossRef] [PubMed]
- Hinds, L.M.; O’Donnell, C.P.; Akhter, M.; Tiwari, B.K. Principles and mechanisms of ultraviolet light emitting diode technology for food industry applications. Innov. Food Sci. Emerg. Technol. 2019, 56, 102153. [Google Scholar] [CrossRef]
- D’Souza, C.; Yuk, H.G.; Khoo, G.H.; Zhou, W. Application of light-emitting diodes in food production, postharvest preservation, and microbiological food safety. Compr. Rev. Food Sci. Food Saf. 2015, 14, 719–740. [Google Scholar] [CrossRef]
- Akgün, M.P.; Ünlütürk, S. Effects of ultraviolet light emitting diodes (LEDs) on microbial and enzyme inactivation of apple juice. Int. J. Food Microbiol. 2017, 260, 65–74. [Google Scholar] [CrossRef]
- Lian, X.; Tetsutani, K.; Katayama, M.; Nakano, M.; Mawatari, K.; Harada, N.; Hamamoto, A.; Yamato, M.; Akutagawa, M.; Kinouchi, Y.; et al. A new colored beverage disinfection system using UV-A light-emitting diodes. Biocontrol Sci. 2010, 15, 33–37. [Google Scholar] [CrossRef]
- Xiang, Q.; Fan, L.; Zhang, R.; Ma, Y.; Liu, S.; Bai, Y. Effect of UVC light-emitting diodes on apple juice: Inactivation of Zygosaccharomyces rouxii and determination of quality. Food Control. 2020, 111, 107082. [Google Scholar] [CrossRef]
- Aihara, M.; Lian, X.; Shimohata, T.; Uebanso, T.; Mawatari, K.; Harada, Y.; Akutagawa, M.; Kinouchi, Y.; Takahashi, A. Vegetable surface sterilization system using UVA light-emitting diodes. J. Med. Investig. 2014, 61, 286–290. [Google Scholar] [CrossRef]
- Fan, L.; Liu, X.; Dong, X.; Dong, S.; Xiang, Q.; Bai, Y. Effects of UVC light-emitting diodes on microbial safety and quality attributes of raw tuna fillets. LWT Food Sci. Technol. 2020, 139, 110553. [Google Scholar] [CrossRef]
- Haughton, P.N.; Grau, E.G.; Lyng, J.; Cronin, D.; Fanning, S.; Whyte, P. Susceptibility of Campylobacter to high intensity near ultraviolet/visible 395 ± 5 nm light and its effectiveness for the decontamination of raw chicken and contact surfaces. Int. J. Food Microbiol. 2012, 159, 267–273. [Google Scholar] [CrossRef]
- Du, L.; Jaya Prasad, A.; Gänzle, M.; Roopesh, M.S. Inactivation of Salmonella spp. in wheat flour by 395 nm pulsed light emitting diode (LED) treatment and the related functional and structural changes of gluten. Food Res. Int. 2020, 127, 108716. [Google Scholar] [CrossRef]
- Subedi, S.; Du, L.; Prasad, A.; Yadav, B.; Roopesh, M.S. Inactivation of Salmonella and quality changes in wheat flour after pulsed light-emitting diode (LED) treatments. Food Bioprod. Process. 2020, 121, 166–177. [Google Scholar] [CrossRef]
- Callanan, M.; Paes, M.; Iversen, C.; Kleijn, R.; Bravo Almeida, C.; Peñaloza, W.; Johnson, N.; Vuataz, G.; Michel, M. Behavior of Enterobacter pulveris in amorphous and crystalline powder matrices treated with supercritical carbon dioxide. J. Dairy Sci. 2012, 95, 6300–6306. [Google Scholar] [CrossRef]
- Bigelow, W.D.; Esty, J.R. The thermal death point in relation to time of typical thermophilic organisms. J. Infect. Dis. 1920, 27, 602–617. [Google Scholar] [CrossRef]
- Cerf, O. Tailing of survival curves of bacterial spores. J. Appl. Bacteriol. 1977, 42, 1–19. [Google Scholar] [CrossRef]
- Mafart, P.; Couvert, O.; Gaillard, S.; Leguerinel, I. On calculating sterility in thermal preservation methods: Application of the Weibull frequency distribution model. Int. J. Food Microbiol. 2002, 72, 107–113. [Google Scholar] [CrossRef]
- Geeraerd, A.H.; Herremans, C.H.; Van Impe, J.F. Structural model requirements to describe microbial inactivation during a mild heat treatment. Int. J. Food Microbiol. 2000, 59, 185–209. [Google Scholar] [CrossRef]
- Geeraerd, A.H.; Valdramidis, V.P.; Van Impe, J.F. GInaFiT, a freeware tool to assess non-log-linear microbial survivor curves. Int. J. Food Microbiol. 2005, 102, 95–105. [Google Scholar] [CrossRef]
- Fenoglio, D.; Ferrario, M.; García Carrillo, M.; Schenk, M.; Guerrero, S. Characterization of microbial inactivation in clear and turbid juices processed by short-wave ultraviolet light. J. Food Process. Preserv. 2020, 1–18. [Google Scholar] [CrossRef]
- Mutz, Y.S.; Rosario, D.K.A.; Bernardes, P.C.; Paschoalin, V.M.F.; Conte-Junior, C.A. Modeling Salmonella Typhimurium inactivation in dry-fermented sausages: Previous habituation in the food matrix undermines UV-C decontamination efficacy. Front. Microbiol. 2020, 11. [Google Scholar] [CrossRef]
- Cheng, Y.; Chen, H.; Sánchez Basurto, L.A.; Protasenko, V.V.; Bharadwaj, S.; Islam, M.; Moraru, C.I. Inactivation of Listeria and E. coli by Deep-UV LED: Effect of substrate conditions on inactivation kinetics. Sci. Rep. 2020, 10, 1–14. [Google Scholar] [CrossRef]
- Sholtes, K.A.; Lowe, K.; Walters, G.W.; Sobsey, M.D.; Linden, K.G.; Casanova, L.M. Comparison of ultraviolet light-emitting diodes and low-pressure mercury-arc lamps for disinfection of water. Environ. Technol. 2016, 37, 2183–2188. [Google Scholar] [CrossRef]
- Shin, J.Y.; Kim, S.J.; Kim, D.K.; Kang, D.H. Fundamental characteristics of deep-UV light-emitting diodes and their application to control foodborne pathogens. Appl. Environ. Microbiol. 2016, 82, 2–10. [Google Scholar] [CrossRef]
- Kim, D.K.; Kim, S.J.; Kang, D.H. Bactericidal effect of 266 to 279 nm wavelength UVC-LEDs for inactivation of Gram positive and Gram negative foodborne pathogenic bacteria and yeasts. Food Res. Int. 2017, 97, 280–287. [Google Scholar] [CrossRef] [PubMed]
- Hinds, L.M.; Charoux, C.M.G.; Akhter, M.; O’Donnell, C.P.; Tiwari, B.K. Effectiveness of a novel UV light emitting diode based technology for the microbial inactivation of Bacillus subtilis in model food systems. Food Control. 2019, 114, 106910. [Google Scholar] [CrossRef]
- Santos, A.L.; Oliveira, V.; Baptista, I.; Henriques, I.; Gomes, N.C.M.; Almeida, A.; Correia, A.; Cunha, Â. Wavelength dependence of biological damage induced by UV radiation on bacteria. Arch. Microbiol. 2013, 195, 63–74. [Google Scholar] [CrossRef]
- Wang, C.; Lu, S.; Zhang, Z. Inactivation of airborne bacteria using different UV sources: Performance modeling, energy utilization and endotoxin degradation. Sci. Total Environ. 2019, 655, 787–795. [Google Scholar] [CrossRef]
- Lui, G.Y.; Roser, D.; Corkish, R.; Ashbolt, N.J.; Stuetz, R. Point-of-use water disinfection using ultraviolet and visible light-emitting diodes. Sci. Total Environ. 2016, 553, 626–635. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A.; Gänzle, M.; Roopesh, M.S. Inactivation of Escherichia coli and Salmonella using 365 and 395 nm high intensity pulsed light emitting diodes. Foods 2019, 8, 679. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, A.A.; Nakano, H. Inactivation of Salmonella, E. coli and Listeria monocytogenes in phosphate-buffered saline and apple juice by ultraviolet and heat treatments. Food Control. 2009, 20, 443–446. [Google Scholar] [CrossRef]
- Gayán, E.; Condón, S.; Álvarez, I. Biological aspects in food preservation by ultraviolet light: A review. Food Bioprocess. Technol. 2014, 7, 1–20. [Google Scholar] [CrossRef]
- Stoops, J.; Jansen, M.; Claes, J.; Van Campenhout, L. Decontamination of powdery and granular foods using continuous wave UV radiation in a dynamic process. J. Food Eng. 2013, 119, 254–259. [Google Scholar] [CrossRef]
- Gayán, E.; Mañas, P.; Álvarez, I.; Condón, S. Mechanism of the synergistic inactivation of Escherichia coli by UV-C light at mild temperatures. Appl. Environ. Microbiol. 2013, 79, 4465–4473. [Google Scholar] [CrossRef]
- Lasagabaster, A.; Martínez de Marañón, I. Impact of process parameters on Listeria innocua inactivation kinetics by pulsed light technology. Food Bioprocess. Technol. 2013, 6, 1828–1836. [Google Scholar] [CrossRef]
- Arroyo, C.; Gayán, E.; Pagán, R.; Condón, S. UV-C inactivation of Cronobacter sakazakii. Foodborne Pathog. Dis. 2012, 9, 907–914. [Google Scholar] [CrossRef]
- Gayán, E.; Serrano, M.J.; Raso, J.; Álvarez, I.; Condón, S. Inactivation of Salmonella enterica by UV-C light alone and in combination with mild temperatures. Appl. Environ. Microbiol. 2012, 78, 8353–8361. [Google Scholar] [CrossRef]
- Gouma, M.; Gayán, E.; Raso, J.; Condón, S.; Álvarez, I. Inactivation of spoilage yeasts in apple juice by UV-C light and in combination with mild heat. Innov. Food Sci. Emerg. Technol. 2015, 32, 146–155. [Google Scholar] [CrossRef]
- Gabriel, A.A.; David, M.M.C.; Elpa, M.S.C.; Michelena, J.C.D. Decontamination of dried whole black peppercorns using ultraviolet-c irradiation. Food Microbiol. 2020, 88, 103401. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.S.; Patras, A.; Pokharel, B.; Vergne, M.J.; Sasges, M.; Begum, A.; Rakariyatham, K.; Pan, C.; Xiao, H. Effect of UV irradiation on the nutritional quality and cytotoxicity of apple juice. J. Agric. Food Chem. 2016, 64, 7812–7822. [Google Scholar] [CrossRef] [PubMed]
- Guneser, O.; Karagul Yuceer, Y. Effect of ultraviolet light on water- and fat-soluble vitamins in cow and goat milk. J. Dairy Sci. 2012, 95, 6230–6241. [Google Scholar] [CrossRef]
- Molina, B.; Sáez, M.I.; Martínez, T.F.; Guil-Guerrero, J.L.; Suárez, M.D. Effect of ultraviolet light treatment on microbial contamination, some textural and organoleptic parameters of cultured sea bass fillets (Dicentrarchus labrax). Innov. Food Sci. Emerg. Technol. 2014, 26, 205–213. [Google Scholar] [CrossRef]
- Matak, K.E.; Sumner, S.S.; Duncan, S.E.; Hovingh, E.; Worobo, R.W.; Hackney, C.R.; Pierson, M.D. Effects of ultraviolet irradiation on chemical and sensory properties of goat milk. J. Dairy Sci. 2007, 90, 3178–3186. [Google Scholar] [CrossRef]
- Ghate, V.; Kumar, A.; Zhou, W.; Yuk, H.G. Irradiance and temperature influence the bactericidal effect of 460-nanometer light-emitting diodes on Salmonella in orange juice. J. Food Prot. 2016, 79, 553–560. [Google Scholar] [CrossRef]
- Kim, M.J.; Bang, W.S.; Yuk, H.G. 405 ± 5 nm light emitting diode illumination causes photodynamic inactivation of Salmonella spp. on fresh-cut papaya without deterioration. Food Microbiol. 2017, 62, 124–132. [Google Scholar] [CrossRef]
- Kim, M.J.; Tang, C.H.; Bang, W.S.; Yuk, H.G. Antibacterial effect of 405 ± 5 nm light emitting diode illumination against Escherichia coli O157:H7, Listeria monocytogenes, and Salmonella on the surface of fresh-cut mango and its influence on fruit quality. Int. J. Food Microbiol. 2017, 244, 82–89. [Google Scholar] [CrossRef]
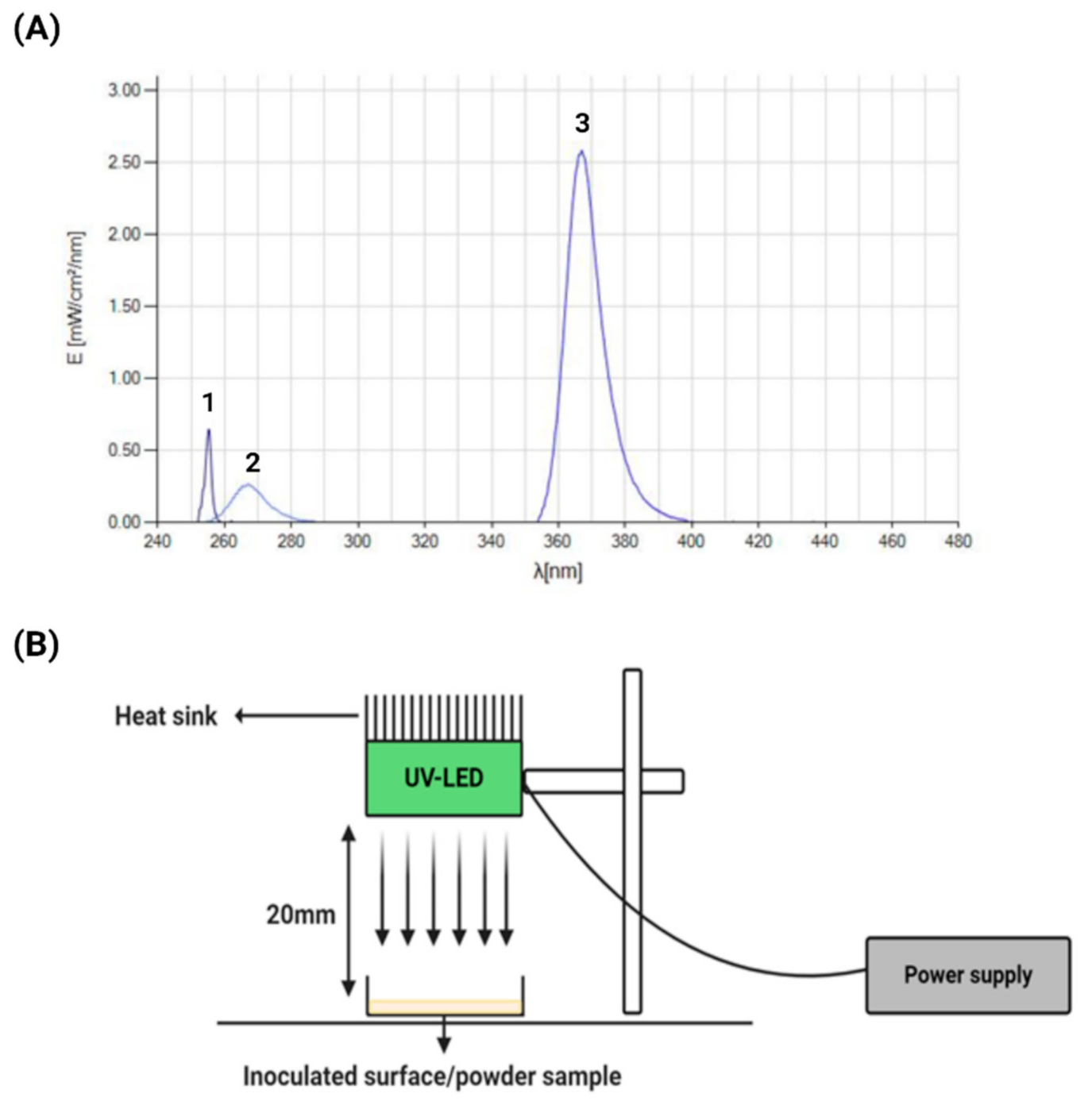
| Powder | Particle Size (µm) | aw |
|---|---|---|
| Garlic | 4.64 ± 2.58 | 0.358 |
| Onion | 24.14 ± 14.57 | 0.336 |
| Cheese and onion | 31.24 ± 14.69 | 0.371 |
| Chilli (large particles) 1 | 55.75 ± 55.28 | 0.430 |
| Chilli (fine particles) | 15.20 ± 20.99 | 0.430 |
| Time (s) | UV Radiation Dose (mJ/cm2) | ||
|---|---|---|---|
| 254 nm | 270 nm | 365 nm | |
| 5 | 20 | 16 | 1700 |
| 10 | 40 | 32 | 3400 |
| 20 | 80 | 64 | 6800 |
| 40 | 160 | 128 | 12,600 |
| Powder | Microorganism | Inactivation Model | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Log-Linear | Biphasic | Weibull | Geeraerd Shoulder–Tail | ||||||
| R2adj | RMSE | R2adj | RMSE | R2adj | RMSE | R2adj | RMSE | ||
| Garlic | L. monocytogenes | 0.902 | 0.357 | 0.928 | 0.305 | 0.906 | 0.349 | 0.9471 | 0.261 |
| E. coli | 0.684 | 0.645 | 0.857 | 0.433 | 0.766 | 0.555 | 0.938 | 0.286 | |
| B. subtilis | 0.959 | 0.205 | - | - | 0.962 | 0.197 | 0.985 | 0.121 | |
| S. Typhimurium | 0.629 | 0.627 | 0.880 | 0.357 | 0.701 | 0.563 | 0.982 | 0.137 | |
| Onion | L. monocytogenes | 0.720 | 0.533 | 0.756 | 0.497 | 0.701 | 0.550 | 0.986 | 0.121 |
| E. coli | 0.761 | 0.567 | 0.789 | 0.531 | 0.752 | 0.577 | 0.987 | 0.130 | |
| B. subtilis | 0.738 | 0.487 | 0.908 | 0.289 | 0.812 | 0.413 | 0.956 | 0.200 | |
| S. Typhimurium | 0.575 | 0.742 | 0.929 | 0.303 | 0.771 | 0.545 | 0.979 | 0.164 | |
| Cheese and onion | L. monocytogenes | 0.514 | 0.787 | 0.975 | 0.180 | 0.868 | 0.410 | - | - |
| E. coli | 0.834 | 0.514 | 0.929 | 0.336 | 0.937 | 0.316 | - | - | |
| B. subtilis | 0.789 | 0.413 | 0.942 | 0.217 | 0.935 | 0.229 | - | - | |
| S. Typhimurium | 0.788 | 0.510 | 0.967 | 0.202 | 0.968 | 0.199 | - | - | |
| Chilli | L. monocytogenes | 0.368 | 0.413 | 0.576 | 0.339 | 0.616 | 0.322 | - | - |
| E. coli | 0.876 | 0.160 | 0.879 | 0.158 | 0.874 | 0.161 | - | - | |
| B. subtilis | 0.746 | 0.199 | 0.851 | 0.152 | 0.787 | 0.182 | 0.827 | 0.164 | |
| S. Typhimurium | 0.884 | 0.088 | 0.863 | 0.096 | 0.877 | 0.091 | 0.863 | 0.096 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nyhan, L.; Przyjalgowski, M.; Lewis, L.; Begley, M.; Callanan, M. Investigating the Use of Ultraviolet Light Emitting Diodes (UV-LEDs) for the Inactivation of Bacteria in Powdered Food Ingredients. Foods 2021, 10, 797. https://doi.org/10.3390/foods10040797
Nyhan L, Przyjalgowski M, Lewis L, Begley M, Callanan M. Investigating the Use of Ultraviolet Light Emitting Diodes (UV-LEDs) for the Inactivation of Bacteria in Powdered Food Ingredients. Foods. 2021; 10(4):797. https://doi.org/10.3390/foods10040797
Chicago/Turabian StyleNyhan, Laura, Milosz Przyjalgowski, Liam Lewis, Máire Begley, and Michael Callanan. 2021. "Investigating the Use of Ultraviolet Light Emitting Diodes (UV-LEDs) for the Inactivation of Bacteria in Powdered Food Ingredients" Foods 10, no. 4: 797. https://doi.org/10.3390/foods10040797
APA StyleNyhan, L., Przyjalgowski, M., Lewis, L., Begley, M., & Callanan, M. (2021). Investigating the Use of Ultraviolet Light Emitting Diodes (UV-LEDs) for the Inactivation of Bacteria in Powdered Food Ingredients. Foods, 10(4), 797. https://doi.org/10.3390/foods10040797






