Sensory Characteristics of Male Impala (Aepyceros melampus) Meat, Produced under Varying Production Systems and Nutrition
Abstract
1. Introduction
2. Materials and Methods
2.1. Culling, Sample Collection and Chemical Analyses
2.2. Fatty Acid Analysis
2.3. Descriptive Sensory and Shear Force Analyses
2.4. Statistical Analyses
3. Results
3.1. Physical Meat Quality
3.2. Fatty Acids
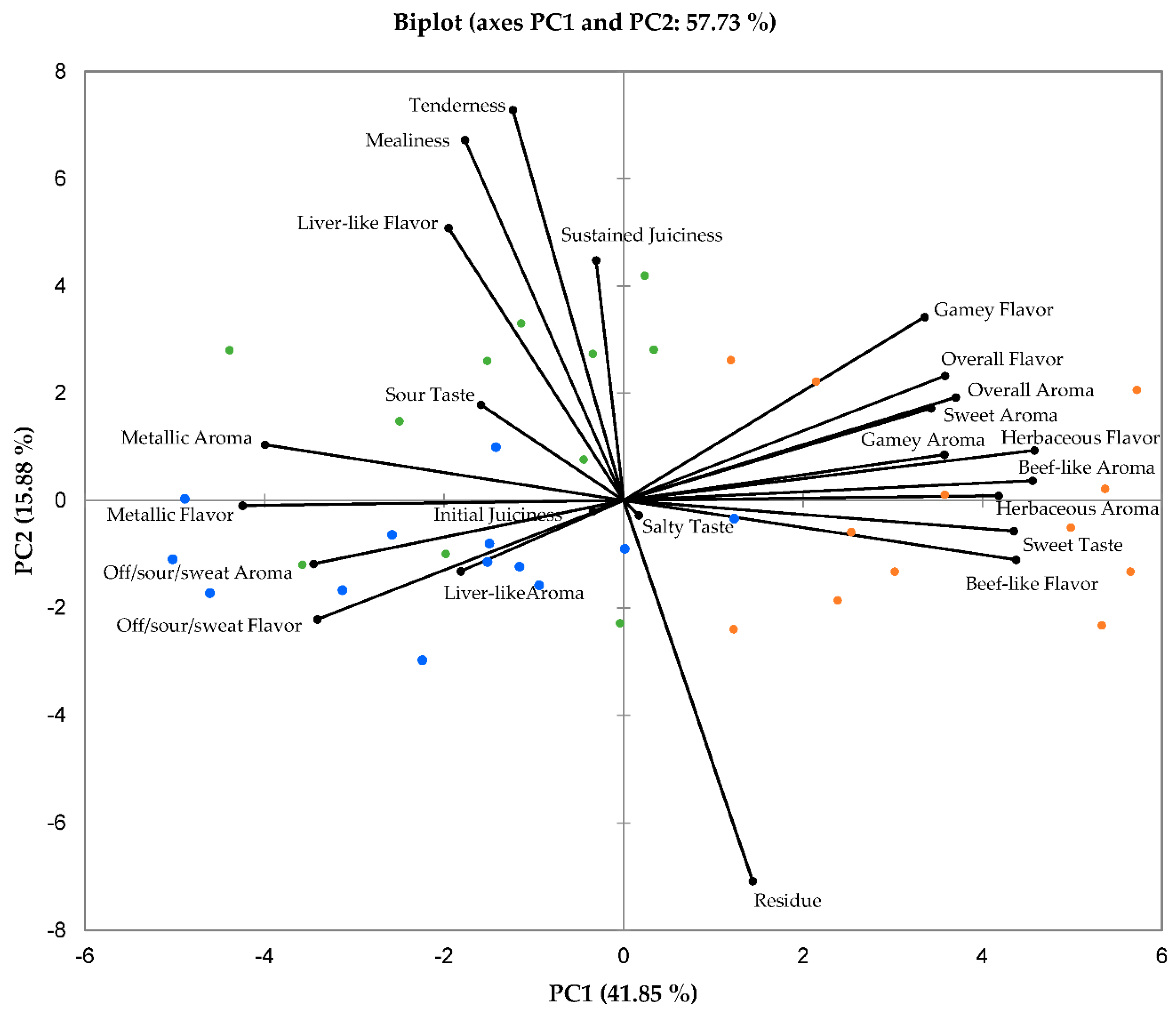
3.3. Descriptory Sensory Characteristics
3.4. Correlations between Physical, Chemical, and Sensory Meat Characteristics
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Taylor, A.; Lindsey, P.; Davies-Mostert, H. An Assessment of the Economic, Social and Conservation Value of the Wildlife Ranching Industry and Its Potential to Support the Green Economy in South Africa; The Endangered Wildlife Trust: Johannesburg, South Africa, 2016; Available online: http://www.sagreenfund.org.za/wordpress/wpcontent/uploads/2016/04/EWT-RESEARCH-REPORT.pdf (accessed on 30 December 2020).
- Child, B. Private conservation in southern Africa: Practice and emerging principles. In Evolution and Innovation in Wildlife Conservation; Suich, H., Child, B., Eds.; Routledge, Taylor & Francis Group: Abingdon, UK, 2009. [Google Scholar]
- Bothma, J.; du, P.; Suich, H.; Spenceley, A. Extensive wildlife production on private land in South Africa. In Evolution and Innovation in Wildlife Conservation; Suich, H., Child, B., Eds.; Routledge, Taylor & Francis Group: Abingdon, UK, 2009. [Google Scholar]
- Snijders, D. Wild property and its boundaries—On wildlife policy and rural consequences in South Africa. J. Peasant Stud. 2012, 39, 503–520. [Google Scholar] [CrossRef]
- Taylor, W.A.; Lindsey, P.A.; Nicholson, S.K.; Relton, C.; Davies-Mostert, H.T. Jobs, game meat and profits: The benefits of wildlife ranching on marginal lands in South Africa. Biol. Conserv. 2020, 245, 108561. [Google Scholar] [CrossRef]
- Lindsey, P.A.; Barnes, J.I.; Nyirenda, V.R.; Pumfrett, B.; Tambling, C.J.; Taylor, W.A.; Rolfes, M.T.S. The Zambian wildlife ranching industry: Scale, associated benefits, and limitations affecting its development. PLoS ONE 2013, 8, e81761. [Google Scholar] [CrossRef]
- National Agricultural Marketing Council (NAMC). Report on the Investigation to Identify Problems for Sustainable Growth and Development in South African Wildlife Ranching. 2006. Available online: https://www.namc.co.za/wp-content/uploads/2017/09/Report-on-Wildlife-ranching.pdf (accessed on 8 February 2021).
- Department of Environmental Affairs. South Africa Protected Areas Database. Vector Digital Data. 2019. Available online: http://egis.environment.gov.za (accessed on 8 February 2021).
- Macdonald, D.W.; Jacobsen, K.; Burnham, D.; Johnson, P.J.; Loveridge, A.J. Cecil: A moment or a movement? Analysis of media coverage of the death of a lion, Panthera leo. Animals 2016, 6, 26. [Google Scholar] [CrossRef]
- Van der Merwe, M. Investigating the Concept of a Game Meat Scheme to Promote Safe Game Meat on the South African Market. Ph.D. Thesis, Department of Environmental Health, Tshwane University of Technology, Pretoria, South Africa, 2012. Available online: http://tutvital.tut.ac.za:8080/vital/access/manager/Repository/tut:1540 (accessed on 30 December 2020).
- Hoffman, L.C.; Muller, M.; Schutte, D.W.; Crafford, K. The retail of South African game meat: Current trade and marketing trends. S. Afr. J. Wildl. Res. 2004, 34, 123. [Google Scholar]
- Bothma, J.P.; Sartorius Von Bach, H.J.; Cloete, P.C. Economics of the wildlife industry in South Africa. In Game Ranch Management; Bothma, J.P., Toit, J.G., Eds.; Van Schaik Publishers: Pretoria, South Africa, 2016. [Google Scholar]
- Shepstone, C. Nutrition in Game Animals. In The New Game Rancher; Oberem, P., Ed.; Briza Publications: Pretoria, South Africa, 2016; pp. 77–90. [Google Scholar]
- Furstenburg, D. Impala (Aepyceros melampus). In The New Game Rancher; Oberem, P., Ed.; Briza Publications: Pretoria, South Africa, 2016; pp. 217–225. [Google Scholar]
- McCrindle, C.M.E.; Siegmund-Schultze, M.; Heeb, A.W.; Zárate, A.V.; Ramrajh, S. Improving food security and safety through use of edible by-products from wild game. J. Arid Land Stud. 2013, 15, 1245–1257. [Google Scholar] [CrossRef]
- Fairall, N. Production parameters of the impala, Aepyceros melampus. S. Afr. J. Anim. Sci. 1983, 13, 176–179. Available online: https://www.sasas.co.za/journals/production-parameters-of-the-impalaaepyceros-melampus/ (accessed on 28 December 2020).
- Needham, T.; Engels, R.A.; Bureš, D.; Kotrba, R.; Van Rensburg, B.J.; Hoffman, L.C. Carcass Yields and Physiochemical Meat Quality of Semi-extensive and Intensively Farmed Impala (Aepyceros melampus). Foods 2020, 9, 418. [Google Scholar] [CrossRef]
- Needham, T.; Engels, R.A.; Hoffman, L.C. Physical Changes during Post-Mortem Ageing of High-Value Impala (Aepyceros Melampus) Steaks. Appl. Sci. 2020, 10, 4485. [Google Scholar] [CrossRef]
- Listrat, A.; Lebret, B.; Louveau, I.; Astruc, T.; Bonnet, M.; Lefaucheur, L.; Picard, B.; Bugeon, J. How muscle structure and composition influence meat and flesh quality. Sci. World J. 2016, 2016, 1–14. [Google Scholar] [CrossRef]
- Williamson, C.S.; Foster, R.K.; Stanner, S.A.; Buttriss, J.L. Red meat in the diet. Nutr. Bull. 2005, 30, 323–355. [Google Scholar] [CrossRef]
- Wood, J.D.; Enser, M.; Richardson, R.I.; Whittington, F.M. Fatty acids in meat and meat products. In Fatty Acids in Foods and Their Health Implications; Chow, C.K., Ed.; CRC Press: New York, NY, USA, 2008. [Google Scholar]
- World Health Organisation. Diet, Nutrition and the Prevention of Chronic Diseases; Report of a Joint WHO/FAO Expert Consultation; WHO Technical Report Series 916; WHO: Geneva, Switzerland, 2003. [Google Scholar]
- Hoffman, L.C.; Wiklund, E. Game and venison—Meat for the modern consumer. Meat Sci. 2006, 74, 197–208. [Google Scholar] [CrossRef]
- Calkins, C.R.; Hodgen, J.M. A fresh look at meat flavor. Meat Sci. 2007, 77, 63–80. [Google Scholar] [CrossRef]
- Neethling, J.; Hoffman, L.C.; Muller, M. Factors influencing the flavour of game meat: A review. Meat Sci. 2016, 113, 139–153. [Google Scholar] [CrossRef]
- Hoffman, L.C.; Kritzinger, B.; Ferreira, A.V. The effects of region and gender on the fatty acid, amino acid, mineral, myoglobin and collagen contents of impala (Aepyceros melampus) meat. Meat Sci. 2005, 69, 551–558. [Google Scholar] [CrossRef] [PubMed]
- Duckett, S.K.; Kuber, P.S. Genetic and nutritional effects on lamb flavor. J. Anim. Sci. 2001, 79, E249–E254. [Google Scholar] [CrossRef]
- Nuernberg, K.; Dannenberger, D.; Nuernberg, G.; Ender, K.; Voigt, J.; Scollan, N.D.; Wood, J.D.; Nute, G.R.; Richardson, R.I. Effect of a grass-based and a concentrate feeding system on meat quality characteristics and fatty acid composition of longissimus muscle in different cattle breeds. Livest. Prod. Sci. 2005, 94, 137–147. [Google Scholar] [CrossRef]
- Mucina, L.; Rutherford, M.C. The Vegetation of South Africa, Lesotho and Swaziland; South African National Biodiversity Institute: Pretoria, South Africa, 2006. [Google Scholar]
- Dahlan, I.; Norfarizan Hanoon, N.A. Chemical composition, palatability and physical characteristics of venison from farmed deer. Anim. Sci. J. 2008, 79, 498–503. [Google Scholar] [CrossRef]
- Association of Official Analytical Chemist International. Official Method of Analysis, 17th ed.; Association of Official Analytical Chemists Inc.: Arlington, TX, USA, 2002. [Google Scholar]
- Lee, C.M.; Trevino, B.; Chaiyawat, M. A simple and rapid solvent extraction method for determining total lipids in fish tissue. J. AOAC Int. 1996, 79, 487–492. [Google Scholar] [CrossRef] [PubMed]
- American Meat Science Association. Research Guidelines for Cookery, Sensory Evaluation, and Instrumental Tenderness Measurements of Meat; National Livestock and Meat Board: Chicago, IL, USA, 2015. [Google Scholar]
- Leygonie, C.; Britz, T.J.; Hoffman, L.C. Impact of freezing and thawing on the quality of meat: Review. Meat Sci. 2012, 91, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Shange, N.; Gouws, P.; Homan, L.C. Changes in pH, color and the microbiology of black wildebeest (Connochaetes gnou) longissimus thoracis et lumborum (LTL) muscle with normal and high (DFD) muscle pH. Meat Sci. 2019, 147, 13–19. [Google Scholar] [CrossRef]
- Troy, D.J.; Kerry, J.P. Consumer perception and the role of science in the meat industry. Meat Sci. 2010, 86, 214–226. [Google Scholar] [CrossRef]
- Wood, J.D.; Richardson, R.I.; Nute, G.R.; Fisher, A.V.; Campo, M.M.; Kasapidou, E.; Sheard, P.R.; Enser, M. Effects of fatty acids on meat quality: A review. Meat Sci. 2003, 66, 21–32. [Google Scholar] [CrossRef]
- Wiklund, E.; Johansson, L.; Malmfors, G. Sensory meat quality, ultimate pH values, blood parameters and carcass characteristics in reindeer (Rangifer tarandus tarandus L.) grazed on natural pastures or fed a commercial feed mixture. Food Qual. Prefer. 2003, 14, 573–581. [Google Scholar] [CrossRef]
- Oltra, O.R.; Farmer, L.J.; Gordon, A.W.; Moss, B.W.; Birnie, J.; Devlin, D.J.; Tolland, E.L.C.; Tollerton, I.J.; Beattie, A.M.; Kennedy, J.T.; et al. Identification of sensory attributes, instrumental and chemical measurements important for consumer acceptability of grilled lamb Longissimus lumborum. Meat Sci. 2015, 100, 97–109. [Google Scholar] [CrossRef]
- Gkarane, V.; Brunton, N.P.; Allen, P.; Gravador, R.S.; Claffey, N.A.; Diskin, M.G.; Fahey, A.G.; Farmer, L.J.; Moloney, A.P.; Alcalde, M.J.; et al. Effect of finishing diet and duration on the sensory quality and volatile profile of lamb meat. Food Res. Int. 2019, 115, 54–64. [Google Scholar]
- Furnols, M.F.; Realini, C.E.; Guerrero, L.; Oliver, M.A.; Sanudo, C.; Campo, M.M.; Nute, G.R.; Cañeque, V.; Alvarez, I.; San Julián, R.; et al. Acceptability of lamb fed on pasture, concentrate or combinations of both systems by European consumers. Meat Sci. 2009, 81, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Howes, N.L.; Bekhit, A.E.; Burritt, D.J.; Campbell, A.W. Opportunities and implications of pasture-based lamb fattening to enhance the long-chain fatty acid composition in meat. Compr. Rev. Food Sci. F. 2015, 14, 22–36. [Google Scholar] [CrossRef]
- Wiklund, E.; Stevenson-Barry, J.; Cummings, T. Sensory meat quality in red deer (Cervus elaphus) grazed on pasture or fed a commercial feed mixture. In Proceedings of the 46th ICoMST, Buenos Aires, Argentina, August 2000. [Google Scholar]
- Volpelli, L.A.; Valusso, R.; Morgante, M.; Pittia, P.; Piasentier, E. Meat quality in male fallow deer (Dama dama): Effects of age and supplementary feeding. Meat Sci. 2003, 65, 555–562. [Google Scholar] [CrossRef]
- Ponnampalam, E.; Sinclair, A.J.; Egan, A.R.; Ferrier, G.R.; Leury, B.J. Dietary manipulation of muscle long-chain omega-3 and omega-6 fatty acids and sensory properties of lamb meat. Meat Sci. 2002, 60, 125–132. [Google Scholar] [CrossRef]
- Erasmus, S.W.; Muller, M.; Alewijn, M.; Koot, A.H.; Van Ruth, S.M.; Hoffman, L.C. Proton-transfer reaction mass spectrometry (PTR-MS) for the authentication of regionally unique South African lamb. Food Chem. 2017, 233, 331–342. [Google Scholar]
| Feed Ingredient | Inclusion (%) |
|---|---|
| Lucerne meal | 38.7 |
| Maize | 16.1 |
| Molasses meal | 12.9 |
| Sunflower oilcake meal | 9.7 |
| Eragrostis hay | 8.1 |
| Melagac rumen bypass fat | 1.6 |
| Vitamins and mineral premix (Game Production Macropack) | 12.9 |
| Sample | Reference Purpose | Internal Cooking Temperature | Scale |
|---|---|---|---|
| Beef fillet | Beef-like aroma and flavor, initial and sustained juiciness, tenderness, residue, mealiness | 71 °C | 0 = low intensity; 100 = high intensity |
| Beef ox liver | Liver-like aroma and flavor | No probe | |
| Beef rib-eye | Initial juiciness, sustained juiciness, toughness, residue, mealiness | 72 °C | |
| Ostrich fillet | Metallic aroma and flavor | 71 °C |
| Sensory Attribute | Description of Attributes | Scale |
|---|---|---|
| Aroma and Flavor | ||
| Overall intensity a | Intensity of aroma in the first few sniffs and the intensity of all flavors. | 0 = low intensity; 100 = high intensity |
| Gamey a | Aroma/flavor associated with meat from wild animal species. | |
| Beef-like a | Aroma/flavor associated with cooked beef fillet. | |
| Metallic a | Aroma/flavor associated with raw meat/blood-like aroma/flavor. | |
| Liver-like a | Aroma/flavor associated with pan-fried beef liver. | |
| Herbaceous a | Aroma/flavor associated with earthy, Fynbos-like herbs. | |
| Off/sour/sweat-like a | Aroma/flavor associated with an off/sour/sweat-like characteristic of meat. | |
| Sweet-associated aroma | Aroma associated with the browning of meat (Maillard reaction). | |
| Sweet-associated taste | Taste associated with a sucrose solution. | |
| Salty taste | Taste associated with sodium ions. | |
| Sour taste | Taste associated with a citric acid solution. | |
| Texture | ||
| Initial juiciness | Amount of fluid extruded when pressed perpendicular to fibres, between the thumb and forefinger. | 0 = dry; 100 = extremely juicy |
| Sustained juiciness | Amount of moisture perceived during mastication (after five chews). | 0 = dry; 100 = extremely juicy |
| Tenderness | Impression of tenderness after mastication (after five chews). | 0 = tough; 100 = extremely tender |
| Residue | Residual tissue remaining after mastication (after 10 chews). | 0 = none; 100 = abundant |
| Mealiness | Disintegration of muscle fibers into small particles (within first few chews). | 0 = none; 100 = abundant |
| Parameter | Production System | SEM | p-Value | ||
|---|---|---|---|---|---|
| Intensive | Semi-Extensive | Extensive | |||
| Chemical composition (g/100 g meat) | |||||
| Moisture | 75.3 b | 75.7 a | 74.7 c | 0.10 | <0.001 |
| Crude Protein | 22.7 b | 22.0 c | 23.4 a | 0.12 | <0.001 |
| Intramuscular fat | 2.0 a | 1.8 b | 1.5 c | 0.05 | <0.001 |
| Ash | 1.21 | 1.19 | 1.18 | 0.01 | 0.062 |
| Physical meat quality | |||||
| pH 24 h post-mortem | 5.8 b | 5.6 c | 6.2 a | 0.05 | <0.001 |
| Thaw loss (%) | 9.9 a | 10.1 a | 4.0 b | 0.50 | <0.001 |
| Cooking loss (%) | 29.9 | 29.3 | 30.5 | 2.09 | 0.921 |
| Shear force (N) | 52.5 | 37.2 | 52.3 | 5.13 | 0.068 |
| Parameter | Production System | SEM | p-Value | ||
|---|---|---|---|---|---|
| Intensive | Semi-Extensive | Extensive | |||
| Total fatty acids (mg/g meat) | 19.7 | 17.8 | 17.6 | 0.88 | 0.181 |
| Fatty Acids (% of total FAMEs): | |||||
| C6:0 (Hexanoic) | 2.5 | 2.3 | 2.3 | 0.07 | 0.061 |
| C8:0 (Caprylic) | 2.5 | 2.5 | 2.3 | 0.09 | 0.416 |
| C10:0 (Capic) | 2.6 | 2.5 | 2.4 | 0.11 | 0.634 |
| C12:0 (Lauric) | 2.7 a | 2.8 a | 2.4 b | 0.12 | 0.043 |
| C14:0 (Myristic) | 3.0 a | 3.0 a | 2.7 b | 0.10 | 0.027 |
| C15:0 (Pentadecyclic) | 1.1 | 1.1 | 1.0 | 0.13 | 0.722 |
| C16:0 (Palmitic) | 10.0 b | 11.8 a | 9.4 b | 0.48 | 0.003 |
| C18:0 (Stearic) | 12.3 | 12.4 | 12.3 | 0.35 | 0.967 |
| C20:0 (Arachidic) | 1.2 | 1.2 | 1.1 | 0.04 | 0.127 |
| C22:0 (Behenic) | 1.9 | 2.1 | 2.3 | 0.12 | 0.089 |
| C24:0 (Lignoceric) | 2.9 a | 2.5 b | 2.5 b | 0.10 | 0.009 |
| Total SFA (% of total FAMEs) | 42.7 ab | 44.2 a | 40.7 b | 0.82 | 0.016 |
| C14:1n9c (Myristoleic) | 1.1 | 1.1 | 1.00 | 0.04 | 0.146 |
| C15:1n9t (Cis-10-pentadecenoic) | 1.7 b | 1.8 b | 2.6 a | 0.27 | 0.048 |
| C16:1n7 (Palmitoleic) | 1.7 | 1.8 | 1.6 | 0.06 | 0.154 |
| C17:1 (Heptadecenoic) | 1.7 | 1.6 | 1.5 | 0.09 | 0.237 |
| C18:1n9 (Oleic) | 5.0 | 6.0 | 5.8 | 0.44 | 0.307 |
| C20:1n9 (Gondoic) | 1.0 | 1.1 | 1.0 | 0.04 | 0.444 |
| Total MUFA (% of total FAMEs) | 12.3 | 13.3 | 13.5 | 0.56 | 0.269 |
| C18:2n6 (Linoleic) | 7.5 | 7.7 | 7.8 | 0.22 | 0.687 |
| C18:3n6 (Gamma-linolenic) | 4.5 | 4.4 | 4.2 | 0.15 | 0.202 |
| C18:3n3 (Alpha-linolenic) | 1.5 b | 2.6 a | 3.0 a | 0.14 | < 0.001 |
| C20:2n6 (Eicosadienoic) | 4 | 2.6 | 3.8 | 0.73 | 0.325 |
| C20:3n6 (Dihomo-gamma-linolenic) | 2.0 | 2.0 | 1.9 | 0.07 | 0.555 |
| C20:3n3 (Eicosatrienoic) | 10.0 a | 8.3 b | 7.8 b | 0.39 | 0.001 |
| C20:5n3 (Eicosapentaienoic) | 2.4 b | 2.9 ab | 4.6 a | 0.61 | 0.045 |
| C22:2n6 (Docosadienoic) | 4.5 | 3.5 | 4.2 | 0.41 | 0.239 |
| C22:6n3 (Docosahexaenoic) | 7.5 | 7.5 | 7.5 | 0.24 | 0.989 |
| Total PUFA (% of total FAMEs) | 45 | 42.5 | 45.8 | 1.28 | 0.176 |
| PUFA:SFA ratio | 1.1 | 1.0 | 1.1 | 0.05 | 0.098 |
| n6 PUFA | 23.7 | 21.2 | 23 | 0.99 | 0.201 |
| n3 PUFA | 21.4 | 21.3 | 22.9 | 0.56 | 0.102 |
| n6:n3 PUFA ratio | 1.1 | 1.0 | 1.0 | 0.05 | 0.144 |
| Sensory Characteristic | Production System | SEM | p-Value | ||
|---|---|---|---|---|---|
| Intensive | Semi-Extensive | Extensive | |||
| Aroma | |||||
| Overall aroma intensity | 65.1 b | 66.3 b | 69.1 a | 0.49 | <0.001 |
| Gamey aroma | 54.7 b | 56.1 b | 58.5 a | 0.59 | <0.001 |
| Beef-like aroma | 37.2 b | 38.5 b | 42.4 a | 0.53 | <0.001 |
| Metallic aroma | 6.3 a | 6.0 a | 2.4 b | 0.56 | <0.001 |
| Liver-like aroma | 1.8 | 2.2 | 1.5 | 0.35 | 0.461 |
| Herbaceous aroma | 6.8 b | 8.0 b | 13.2 a | 0.52 | <0.001 |
| Off, sour, sweat-like aroma | 5.5 a | 3.6 ab | 2.4 b | 0.69 | 0.014 |
| Sweet-associated aroma | 8.4 b | 9.5 b | 11.5 a | 0.42 | <0.001 |
| Flavor | |||||
| Overall flavor intensity | 62.9 b | 64.2 ab | 65.7 a | 0.61 | 0.008 |
| Gamey flavor | 54.0 b | 55.9 a | 56.7 a | 0.42 | <0.001 |
| Beef-like flavor | 39.4 b | 38.5 b | 45.0 a | 0.66 | <0.001 |
| Metallic flavor | 8.4 a | 8.4 a | 3.3 b | 0.51 | <0.001 |
| Liver-like flavor | 1.2 b | 2.2 a | 0.6 b | 0.30 | 0.002 |
| Herbaceous flavor | 7.1 b | 8.2 b | 12.1 a | 0.48 | <0.001 |
| Off, sour, sweat-like flavor | 1.3 a | 0.9 a | 0.2 b | 0.24 | 0.012 |
| Sweet-associated taste | 10.5 b | 10.2 b | 12.6 a | 0.32 | <0.001 |
| Salty taste | 9.1 | 9.1 | 9.1 | 0.01 | 1.000 |
| Sour taste | 4.2 ab | 4.5 a | 3.7 b | 0.22 | 0.041 |
| Texture | |||||
| Initial juiciness | 39.4 | 39.3 | 39.7 | 0.98 | 0.950 |
| Sustained juiciness | 46 | 45.7 | 46.5 | 0.83 | 0.772 |
| Tenderness | 59.9 b | 66.9 a | 59.7 b | 1.90 | 0.016 |
| Residue | 11.3 a | 6.9 b | 12.2 a | 1.31 | 0.015 |
| Mealiness | 6.0 b | 9.9 a | 4.7 b | 0.85 | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Needham, T.; Engels, R.A.; Hoffman, L.C. Sensory Characteristics of Male Impala (Aepyceros melampus) Meat, Produced under Varying Production Systems and Nutrition. Foods 2021, 10, 619. https://doi.org/10.3390/foods10030619
Needham T, Engels RA, Hoffman LC. Sensory Characteristics of Male Impala (Aepyceros melampus) Meat, Produced under Varying Production Systems and Nutrition. Foods. 2021; 10(3):619. https://doi.org/10.3390/foods10030619
Chicago/Turabian StyleNeedham, Tersia, Retha A. Engels, and Louwrens C. Hoffman. 2021. "Sensory Characteristics of Male Impala (Aepyceros melampus) Meat, Produced under Varying Production Systems and Nutrition" Foods 10, no. 3: 619. https://doi.org/10.3390/foods10030619
APA StyleNeedham, T., Engels, R. A., & Hoffman, L. C. (2021). Sensory Characteristics of Male Impala (Aepyceros melampus) Meat, Produced under Varying Production Systems and Nutrition. Foods, 10(3), 619. https://doi.org/10.3390/foods10030619







