Veterinary Drug Residues in Animal-Derived Foods: Sample Preparation and Analytical Methods
Abstract
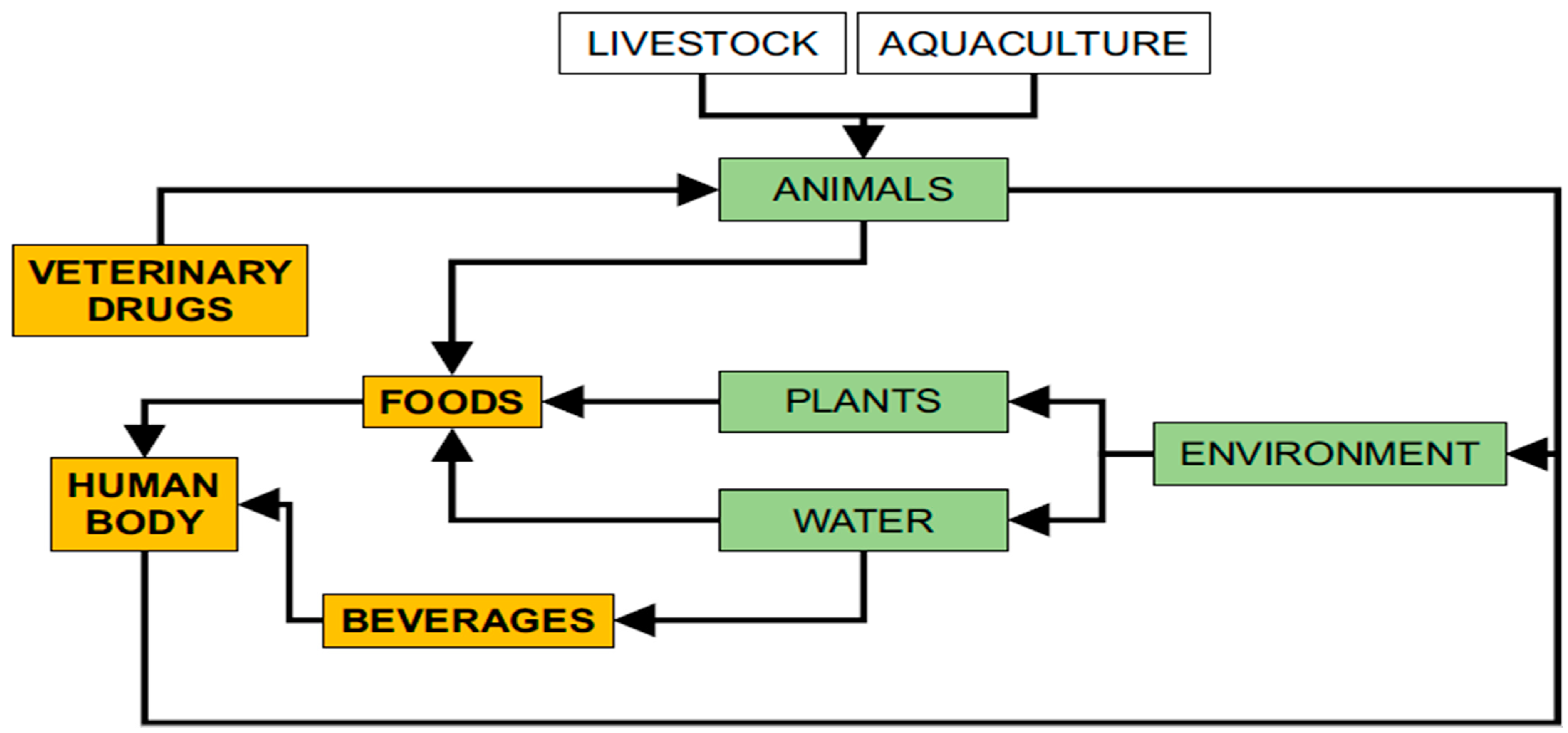
1. Introduction
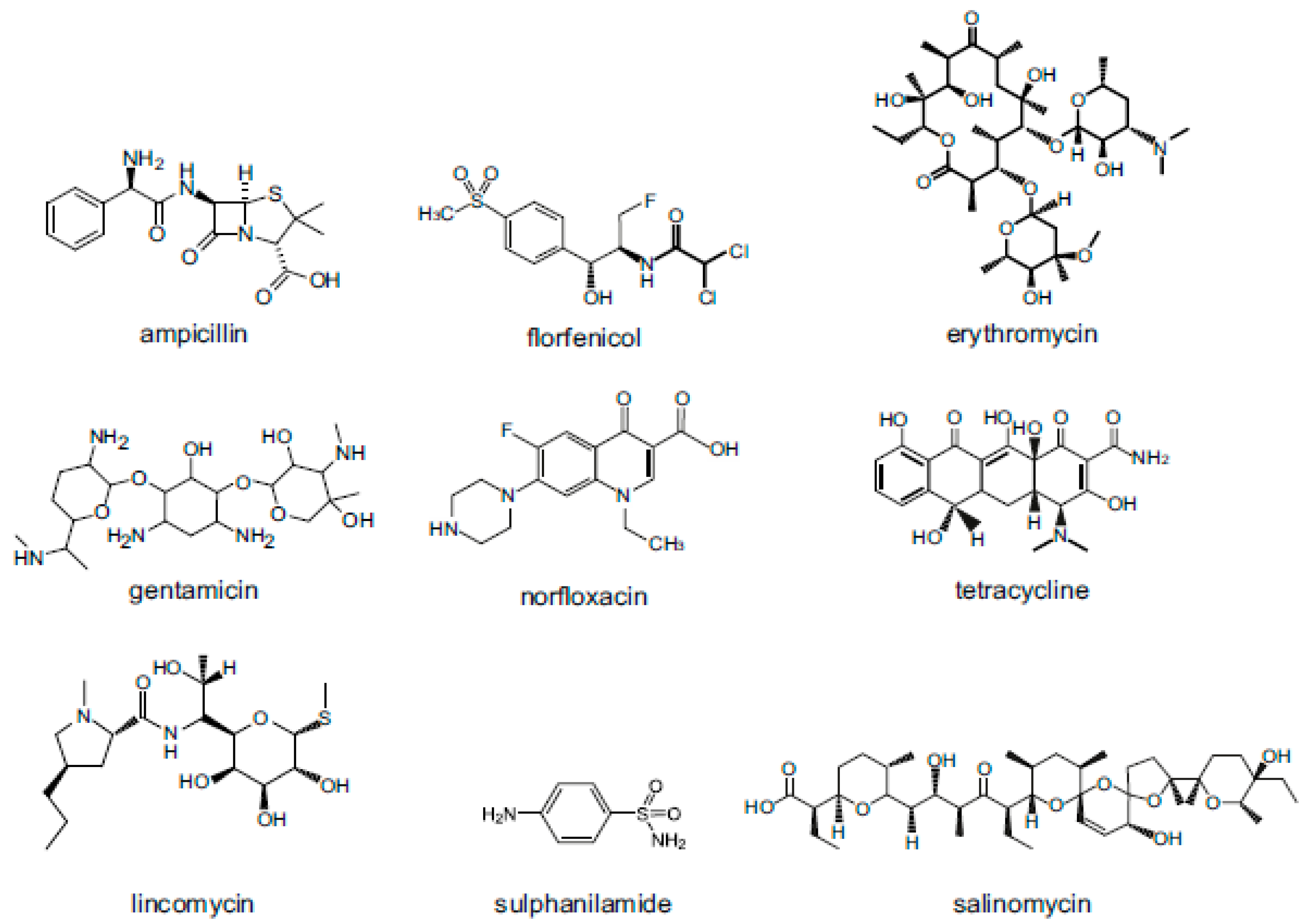
2. Veterinary Drugs: Classification, Use and Physicochemical Characteristics
3. Extraction Methods
3.1. Liquid-Liquid Extraction (LLE)
3.2. Solid-Phase Extraction (SPE)
3.3. Accelerated Solvent Extraction (ASE)
3.4. Quick, Easy, Cheap, Effective, Rugged and Safe (QuEChERS) Extraction
3.5. Matrix Solid-Phase Dispersion (MSPD) Extraction
3.6. Other Extraction Methods
4. Analytical Methods for Detection
4.1. Liquid Chromatography (LC)
4.2. Gas Chromatography (GC)
4.3. Enzyme-Linked Immunosorbent Assay (ELISA)
4.4. Capillary Electrophoresis (CE)
4.5. Micellar Electrokinetic Capillary Chromatography (MEKC)
5. Advanced Methods for Detection
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tufa, T.B. Veterinary drug residues in food-animal products: Its risk factors and potential effects on public health. J. Vet. Sci. Technol. 2015, 7, 1–7. [Google Scholar] [CrossRef]
- Gehring, R.; Baynes, R.E.; Riviere, J.E. Application of risk assessment and management principles to the extralabel use of drugs in food-producing animals. J. Vet. Pharmacol. Ther. 2006, 29, 5–14. [Google Scholar] [CrossRef]
- Boobis, A.; Cerniglia, C.; Chicoine, A.; Fattori, V.; Lipp, M.; Reuss, R.; Verger, P.; Tritscher, A. Characterizing chronic and acute health risks of residues of veterinary drugs in food: Latest methodological developments by the joint FAO/WHO expert committee on food additives. Crit. Rev. Toxicol. 2017, 47, 885–899. [Google Scholar] [CrossRef]
- Bartikova, H.; Skálová, L.; Stuchlíková, L.; Vokřál, I.; Vanek, T.; Podlipná, R. Xenobiotic-metabolizing enzymes in plants and their role in uptake and biotransformation of veterinary drugs in the environment. Drug Metab. Rev. 2015, 47, 374–387. [Google Scholar] [CrossRef]
- The European Medicines Agency. Commission Regulation (EU) No. 37/2010 of 22 December 2009 on Pharmacologically Active Substances and their Classification Regarding Maximum Residue Limits in Foodstuffs of Animal Origin; The European Medicines Agency: Amsterdam, The Netherlands, 2010. [Google Scholar]
- US Food and Drug Administration. CFR-Code of Federal Regulations Title 21 Part 556 Tolerances for Residue of New Animal Drugs in Food; US Food and Drug Administration: Rockville, MD, USA, 2014.
- Ministry of Agriculture of the People’s Republic of China. Maxium Residue Level of Veterinary Drugs in Food of Animal Origin; Notice No. 235 (Appendix 4); Ministry of Agriculture of the People’s Republic of China: Beijing, China, 2002.
- Kirchhelle, C. Pharming animals: A global history of antibiotics in food production (1935–2017). Palgrave Commun. 2018, 4, 96. [Google Scholar] [CrossRef]
- Li, R.; Lin, Z.J.; Yang, J.Y.; Xu, Z.L.; Wang, H.; Lei, H.T.; Sun, Y.M.; Shen, Y.D. An indirect competitive enzyme-linked immunosorbent assay for simultaneous determination of florfenicol and thiamphenicol in animal meat and urine. Chin. J. Anal. Chem. 2018, 46, 1321–1328. [Google Scholar] [CrossRef]
- Kowalski, P.; Oledzka, I.; Lamparczyk, H. Capillary electrophoresis in analysis of veterinary drugs. J. Pharm. Biomed. Anal. 2003, 32, 937–947. [Google Scholar] [CrossRef]
- Wang, B.; Zhao, X.; Xie, X.; Xie, K.; Zhang, G.; Zhang, T.; Liu, X. Development of an accelerated solvent extraction approach for quantitative analysis of chloramphenicol, thiamphenicol, florfenicol, and florfenicol amine in poultry eggs. Food Anal. Methods 2019, 12, 1705–1714. [Google Scholar] [CrossRef]
- Wang, B.; Wang, Y.; Xie, X.; Diao, Z.; Xie, K.; Zhang, G.; Zhang, T.; Dai, G. Quantitative analysis of spectinomycin and lincomycin in poultry eggs by accelerated solvent extraction coupled with gas chromatography tandem mass spectrometry. Foods 2020, 9, 651. [Google Scholar] [CrossRef] [PubMed]
- Jank, L.; Martins, M.T.; Arsand, J.B.; Motta, T.M.C.; Hoff, R.B.; Barreto, F.; Pizzolato, T.M. High-throughput method for macrolides and lincosamides antibiotics residues analysis in milk and muscle using a simple liquid-liquid extraction technique and liquid chromatography-electrospray-tandem mass spectrometry analysis (LC-MS/MS). Talanta 2015, 144, 686–695. [Google Scholar] [CrossRef]
- Płotka-Wasylka, J.; Szczepańska, N.; De La Guardia, M.; Namieśnik, J. Miniaturized solid-phase extraction techniques. TrAC Trends Anal. Chem. 2015, 73, 19–38. [Google Scholar] [CrossRef]
- Richter, B.E.; Raynie, D. Accelerated solvent extraction (ASE) and high-temperature water extraction. Compr. Sampl. Sample Prep. 2012, 2, 105–115. [Google Scholar] [CrossRef]
- Liu, H.Y.; Lin, S.L.; Fuh, M.R. Determination of chloramphenicol, thiamphenicol and florfenicol in milk and honey using modified QuEChERS extraction coupled with polymeric monolith-based capillary liquid chromatography tandem mass spectrometry. Talanta 2016, 150, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.D.; Wu, P.G.; Jiang, W.; Ma, B.J. Determination of chloramphenicol, thiamphenicol, and florfenicol in fish muscle by matrix solid-phase dispersion extraction (MSPD) and ultra-high pressure liquid chromatography tandem mass spectrometry. Food Control. 2015, 52, 34–38. [Google Scholar] [CrossRef]
- Chen, D.; Yu, J.; Tao, Y.; Pan, Y.; Xie, S.; Huang, L.; Peng, D.; Wang, X.; Wang, Y.; Liu, Z.; et al. Qualitative screening of veterinary anti-microbial agents in tissues, milk, and eggs of food-producing animals using liquid chromatography coupled with tandem mass spectrometry. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2016, 1017–1018, 82–88. [Google Scholar] [CrossRef]
- Yang, F.; Zhang, W.; Gu, C.; Xu, J.; Zhou, K. Separation of seven residues of fluoroquinolones from fish samples using solid phase microextraction and Electrophoresis and their detection by Electrochemiluminescence method. Int. J. Electrochem. Sci. 2020, 15, 9300–9312. [Google Scholar] [CrossRef]
- Samsidar, A.; Siddiquee, S.; Shaarani, S.M. A review of extraction, analytical and advanced methods for determination of pesticides in environment and foodstuffs. Trends Food Sci. Technol. 2018, 71, 188–201. [Google Scholar] [CrossRef]
- Majdinasab, M.; Yaqub, M.; Rahim, A.; Catanante, G.; Hayat, A.; Marty, J.L. An overview on recent progress in electrochemical biosensors for antimicrobial drug residues in animal-derived food. Sensors 2017, 17, 1947. [Google Scholar] [CrossRef]
- JECFA. Residue Evaluation of Certain Veterinary Drugs (Seventy-Fifth Report of the Joint FAO/WHO Expert Committee on Food Additives); FAO JECFA Monographs 12; Food and Agriculture Organization of the United Nations: Rome, Italy, 2012. [Google Scholar]
- Blumenthal, K.G.; Peter, J.G.; Trubiano, J.A.; Phillips, E.J. Antibiotic allergy. Lancet 2019, 393, 183–198. [Google Scholar] [CrossRef]
- Graziani, C.; Busani, L.; Dionisi, A.M.; Lucarelli, C.; Owczarek, S.; Ricci, A.; Mancin, M.; Caprioli, A.; Luzzi, I. Antimicrobial resistance in Salmonella enterica serovar Typhimurium from human and animal sources in Italy. Vet. Microbiol. 2008, 128, 414–418. [Google Scholar] [CrossRef] [PubMed]
- Hanekamp, J.C.; Bast, A. Antibiotics exposure and health risks: Chloramphenicol. Environ. Toxicol. Pharmacol. 2015, 39, 213–220. [Google Scholar] [CrossRef]
- Kaiser, G. Protein Synthesis Inhibitors: Macrolides Mechanism of Action Animation. In Classification of Agents Pharmamotion; The Community College of Baltimore County: Baltimore County, MD, USA, 2009. [Google Scholar]
- Chen, J.; Ying, G.G.; Deng, W.J. Antibiotic residues in food: Extraction, analysis, and human health concerns. J. Agric. Food Chem. 2019, 67, 7569–7586. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, A.; Dos Santos, C.; Cimbro, M.; Grassi, G.G. Comparative antimicrobial activity and post-antibiotic effect of azithromycin, clarithromycin and roxithromycin against some respiratory pathogens. Int. J. Antimicrob. Agents 1996, 7, 181–186. [Google Scholar] [CrossRef]
- Zhang, M.Q.; Chen, B.; Zhang, J.P.; Chen, N.; Liu, C.Z.; Hu, C.Q. Liver toxicity of macrolide antibiotics in zebrafish. Toxicology 2020, 441, 152501. [Google Scholar] [CrossRef] [PubMed]
- Mingeot-Leclercq, M.P.; Glupczynski, Y.; Tulkens, P.M. Aminoglycosides: Activity and resistance. Antimicrob. Agents Chemother. 1999, 43, 727–737. [Google Scholar] [CrossRef] [PubMed]
- Dowling, P.M. Miscellaneous antimicrobials: Ionophores, nitrofurans, nitroimidazoles, rifamycins, oxazolidones, and others. In Antimicrobial Therapy in Veterinary Medicine; Giguere, S., Prescott, J.F., Baggot, J.D., Walker, R.D., Dowling, P.M., Eds.; Blackwell Publishing: Ames, IA, USA, 2006; pp. 285–300. [Google Scholar]
- Petersen, L.; Rogers, C. Aminoglycoside-induced hearing deficits—A review of cochlear ototoxicity. S. Afr. Fam. Pract. 2015, 57, 77–82. [Google Scholar] [CrossRef]
- Black, F.O.; Pesznecker, S.; Stallings, V. Permanent gentamicin vestibulotoxicity. Otol. Neurotol. 2004, 25, 559–569. [Google Scholar] [CrossRef]
- Blagburn, B.L.; Lindsay, D.S. Ectoparasiticides. In Veterinary Pharmacology and Therapeutics; Adams, H.R., Ed.; Iowa State University Press: Ames, IA, USA, 2001; pp. 1017–1039. [Google Scholar]
- Martinez, M.; Mcdermott, P.; Walker, R. Pharmacology of the fluoroquinolones: A perspective for the use in domestic animals. Vet. J. 2006, 172, 10–28. [Google Scholar] [CrossRef] [PubMed]
- Andersson, M.I.; Macgowan, A.P. Development of the quinolones. J. Antimicrob. Chemother. 2003, 51 (Suppl. 1), 1–11. [Google Scholar] [CrossRef]
- Heeb, S.; Fletcher, M.P.; Chhabra, S.R.; Diggle, S.P.; Williams, P.; Camara, M. Quinolones: From antibiotics to autoinducers. FEMS Microbiol. Rev. 2011, 35, 247–274. [Google Scholar] [CrossRef]
- De Sarro, A.; De Sarro, G. Adverse reactions to fluoroquinolones. An overview on mechanistic aspects. Curr. Med. Chem. 2001, 8, 371–384. [Google Scholar] [CrossRef] [PubMed]
- Chopra, I.; Roberts, M. Tetracycline antibiotics: Mode of action, applications, molecular biology, and epidemiology of bacterial resistance. Microbiol. Mol. Biol. Rev. 2001, 65, 232–260. [Google Scholar] [CrossRef] [PubMed]
- Bishop, Y. The Veterinary Formulary; Pharmaceutical Press: London, UK, 2001. [Google Scholar]
- Chopra, I.; Hawkey, P.M.; Hinton, M. Tetracyclines, molecular and clinical aspects. J. Antimicrob. Chemother. 1992, 29, 245–277. [Google Scholar] [CrossRef] [PubMed]
- Amacher, D.E.; Martin, B.A. Tetracycline-induced steatosis in primary canine hepatocyte cultures. Fundam. Appl. Toxicol. 1997, 40, 256–263. [Google Scholar] [CrossRef] [PubMed]
- Ekwall, B.; Acosta, D. In vitro comparative toxicity of selected drugs and chemicals in HeLa cells, Chang liver cells, and rat hepatocytes. Drug Chem. Toxicol. 1982, 5, 219–231. [Google Scholar] [CrossRef] [PubMed]
- Maffioli, S.I. A chemist’s survey of different antibiotic classes. In Antibiotics: Targets, Mechanisms and Resistance; Gualerzi, C.O., Brandi, L., Fabbretti, A., Pon, C.L., Eds.; Wiley-VCH: Weinheim, Germany, 2014; pp. 1–22. [Google Scholar]
- Tenson, T.; Lovmar, M.; Ehrenberg, M. The mechanism of action of macrolides, lincosamides and streptogramin B reveals the nascent peptide exit path in the ribosome. J. Mol. Biol. 2003, 330, 1005–1014. [Google Scholar] [CrossRef]
- Spizek, J.; Rezanka, T. Lincosamides: Chemical structure, biosynthesis, mechanism of action, resistance, and applications. Biochem. Pharmacol. 2017, 133, 20–28. [Google Scholar] [CrossRef] [PubMed]
- FAO/WHO. Toxicological Evaluation of Certain Veterinary Drug Residues in Food (Series 45); Joint FAO/WHO Expert Committee on Food Additives (JEFCA); World Health Organization: Geneva, Switzerland, 2000. [Google Scholar]
- Henry, R.J. The mode of action of sulfonamides. Bacteriol. Rev. 1943, 7, 175–262. [Google Scholar] [CrossRef]
- Madigan, M.T.; Martinko, J.M.; Bender, K.S.; Buckley, D.H.; Stahl, D.A. Brock Biology of Microorganisms; Pearson Education Limited: Boston, MA, USA, 2012. [Google Scholar]
- Slatore, C.G.; Tilles, S.A. Sulfonamide hypersensitivity. Immunol. Allergy Clin. 2004, 24, 477–490. [Google Scholar] [CrossRef]
- Knowles, S.; Shapiro, L.; Shear, N.H. Should celecoxib be contraindicated in patients who are allergic to sulfonamides? Revisiting the meaning of ’sulfa’ allergy. Drug Saf. 2001, 24, 239–247. [Google Scholar] [CrossRef]
- Goetting, V.; Lee, K.A.; Tell, L.A. Pharmacokinetics of veterinary drugs in laying hens and residues in eggs: A review of the literature. J. Vet. Pharmacol. Ther. 2011, 34, 521–556. [Google Scholar] [CrossRef]
- Edwards, D.I. Nitroimidazole drugs--action and resistance mechanisms. I. Mechanisms of action. J. Antimicrob. Chemother. 1993, 31, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Botsoglou, N.A.; Fletouris, D.J. Drug Residues in Food; Marcel Dekker: New York, NY, USA, 2001. [Google Scholar]
- Tang, Y.Y.; Lu, H.F.; Lin, H.Y.; Shih, Y.C.; Hwang, D.F. Multiclass analysis of 23 veterinary drugs in milk by ultraperformance liquid chromatography-electrospray tandem mass spectrometry. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2012, 881–882, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.W.C.; Lam, C.H. Development of a 15-class multiresidue method for analyzing 78 hydrophilic and hydrophobic veterinary drugs in milk, egg and meat by liquid chromatography-tandem mass spectrometry. Anal. Methods 2015, 7, 6764–6776. [Google Scholar] [CrossRef]
- Biselli, S.; Schwalb, U.; Meyer, A.; Hartig, L. A multi-class, multi-analyte method for routine analysis of 84 veterinary drugs in chicken muscle using simple extraction and LC-MS/MS. Food Addit. Contam. Part. A 2013, 30, 921–939. [Google Scholar] [CrossRef]
- Dasenaki, M.E.; Thomaidis, N.S. Multi-residue determination of 115 veterinary drugs and pharmaceutical residues in milk powder, butter, fish tissue and eggs using liquid chromatography-tandem mass spectrometry. Anal. Chim. Acta 2015, 880, 103–121. [Google Scholar] [CrossRef]
- Danezis, G.P.; Anagnostopoulos, C.J.; Liapis, K.; Koupparis, M.A. Multi-residue analysis of pesticides, plant hormones, veterinary drugs and mycotoxins using HILIC chromatography—MS/MS in various food matrices. Anal. Chim. Acta 2016, 942, 121–138. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Wang, B.; Pang, M.; Zhao, X.; Xie, K.; Zhang, Y.; Wang, Y.; Guo, Y.; Liu, C.; Bu, X.; et al. Quantitative analysis of chloramphenicol, thiamphenicol, florfenicol and florfenicol amine in eggs via liquid chromatography-electrospray ionization tandem mass spectrometry. Food Chem. 2018, 269, 542–548. [Google Scholar] [CrossRef]
- Wang, B.; Liu, J.; Zhao, X.; Xie, K.; Diao, Z.; Zhang, G.; Zhang, T.; Dai, G. Determination of eight coccidiostats in eggs by liquid–liquid extraction–solid-phase extraction and liquid chromatography–tandem mass spectrometry. Molecules 2020, 25, 987. [Google Scholar] [CrossRef]
- Kaufmann, A.; Butcher, P.; Maden, K.; Walker, S.; Widmer, M. Development of an improved high resolution mass spectrometry based multi-residue method for veterinary drugs in various food matrices. Anal. Chim. Acta 2011, 700, 86–94. [Google Scholar] [CrossRef]
- Dasenaki, M.E.; Bletsou, A.A.; Koulis, G.A.; Thomaidis, N.S. Qualitative multiresidue screening method for 143 veterinary drugs and pharmaceuticals in milk and fish tissue using liquid chromatography quadrupole-time-of-flight mass spectrometry. J. Agric. Food Chem. 2015, 63, 4493–4508. [Google Scholar] [CrossRef] [PubMed]
- Turnipseed, S.B.; Storey, J.M.; Lohne, J.J.; Andersen, W.C.; Burger, R.; Johnson, A.S.; Madson, M.R. Wide-scope screening method for multiclass veterinary drug residues in fish, shrimp, and eel using liquid chromatography-quadrupole high-resolution mass spectrometry. J. Agric. Food Chem. 2017, 65, 7252–7267. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Peng, T.; Zhu, A.; He, J.; Chang, Q.; Hu, X.; Chen, H.; Fan, C.; Jiang, W.; Chen, M.; et al. Multi-residue analysis of veterinary drugs, pesticides and mycotoxins in dairy products by liquid chromatography-tandem mass spectrometry using low-temperature cleanup and solid phase extraction. J. Chromatogr. B 2015, 1002, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Dasenaki, M.E.; Michali, C.S.; Thomaidis, N.S. Analysis of 76 veterinary pharmaceuticals from 13 classes including aminoglycosides in bovine muscle by hydrophilic interaction liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2016, 1452, 67–80. [Google Scholar] [CrossRef]
- Han, R.W.; Zheng, N.; Yu, Z.N.; Wang, J.; Xu, X.M.; Qu, X.Y.; Li, S.L.; Zhang, Y.D.; Wang, J.Q. Simultaneous determination of 38 veterinary antibiotic residues in raw milk by UPLC-MS/MS. Food Chem. 2015, 181, 119–126. [Google Scholar] [CrossRef]
- Piatkowska, M.; Jedziniak, P.; Zmudzki, J. Multiresidue method for the simultaneous determination of veterinary medicinal products, feed additives and illegal dyes in eggs using liquid chromatography-tandem mass spectrometry. Food Chem. 2016, 197, 571–580. [Google Scholar] [CrossRef]
- Richter, B.E.; Jones, B.A.; Ezzell, J.L.; Porter, N.L.; Avdalovic, N.; Pohl, C. Accelerated solvent extraction: A technique for sample preparation. Anal. Chem. 1996, 68, 1033–1039. [Google Scholar] [CrossRef]
- USEPA. Pressurised Fluid Extraction. In Test Methods for Evaluating Solid Waste; Method 3545; USEPA: Washington, DC, USA, 1995. [Google Scholar]
- Tao, Y.; Yu, G.; Chen, D.; Pan, Y.; Liu, Z.; Wei, H.; Peng, D.; Huang, L.; Wang, Y.; Yuan, Z. Determination of 17 macrolide antibiotics and avermectins residues in meat with accelerated solvent extraction by liquid chromatography-tandem mass spectrometry. J. Chromatogr. B 2012, 897, 64–71. [Google Scholar] [CrossRef]
- Yu, H.; Tao, Y.; Chen, D.; Wang, Y.; Yuan, Z. Development of an HPLC–UV method for the simultaneous determination of tetracyclines in muscle and liver of porcine, chicken and bovine with accelerated solvent extraction. Food Chem. 2011, 124, 1131–1138. [Google Scholar] [CrossRef]
- Anastassiades, M.; Lehotay, S.J.; Štajnbaher, D.; Schenck, F.J. Fast and easy multiresidue method employing acetonitrile extraction/partitioning and “dispersive solid-phase extraction” for the determination of pesticide residues in produce. J. AOAC Int. 2003, 86, 412–431. [Google Scholar] [CrossRef]
- Xu, X.; Xu, X.; Han, M.; Qiu, S.; Hou, X. Development of a modified QuEChERS method based on magnetic multiwalled carbon nanotubes for the simultaneous determination of veterinary drugs, pesticides and mycotoxins in eggs by UPLC-MS/MS. Food Chem. 2019, 276, 419–426. [Google Scholar] [CrossRef]
- Arias, J.L.O.; Schneider, A.; Batista-Andrade, J.A.; Vieira, A.A.; Caldas, S.S.; Primel, E.G. Chitosan from shrimp shells: A renewable sorbent applied to the clean-up step of the QuEChERS method in order to determine multi-residues of veterinary drugs in different types of milk. Food Chem. 2018, 240, 1243–1253. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, X.; Li, X.; Zhang, J.; Cao, Y.; Su, M.; Shi, Z.; Sun, H. Rapid screening and quantification of multi-class multi-residue veterinary drugs in royal jelly by ultra performance liquid chromatography coupled to quadrupole time-of-flight mass spectrometry. Food Control. 2016, 60, 667–676. [Google Scholar] [CrossRef]
- Shin, D.; Kang, H.S.; Jeong, J.; Kim, J.; Choe, W.J.; Lee, K.S.; Rhee, G.S. Multi-residue determination of veterinary drugs in fishery products using liquid chromatography-tandem mass spectrometry. Food Anal. Methods 2018, 11, 1815–1831. [Google Scholar] [CrossRef]
- Freitas, S.K.B.; Paim, A.P.S.; Silva, P.T.D.S. Development of a LC-IT-TOF MS procedure to quantify veterinary drug residues in milk employing a QuEChERS approach. Food Anal. Methods 2013, 7, 39–46. [Google Scholar] [CrossRef]
- Villar-Pulido, M.; Gilbert-Lopez, B.; Garcia-Reyes, J.F.; Martos, N.R.; Molina-Diaz, A. Multiclass detection and quantitation of antibiotics and veterinary drugs in shrimps by fast liquid chromatography time-of-flight mass spectrometry. Talanta 2011, 85, 1419–1427. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.; Fan, C.L.; Chang, Q.Y.; Bu, M.N.; Zhao, Z.Y.; Wang, W.; Pang, G.F. Simultaneous determination of multi-class veterinary drug residues in different muscle tissues by modified QuEChERS combined with HPLC-MS/MS. Anal. Methods 2014, 6, 6285. [Google Scholar] [CrossRef]
- Nakajima, T.; Nagano, C.; Sasamoto, T.; Hayashi, H.; Kanda, M.; Kanai, S.; Takeba, K.; Matsushima, Y.; Takano, I. Development and validation of rapid analysis method for multi-class veterinary drugs in livestock products by LC-MS/MS. J. Food Hyg. Soc. Jpn. 2012, 53, 243–253. [Google Scholar] [CrossRef]
- Barker, S.A.; Long, A.R.; Short, C.R. Isolation of drug residues from tissues by solid phase dispersion. J. Chromatogr. A 1989, 475, 353–361. [Google Scholar] [CrossRef]
- Pérez, R.A.; Albero, B.; Tadeo, J.L. Book chapter: 19—Matrix solid phase dispersion. In A Volume in Handbooks in Separation Science; Elsevier: Madrid, Spain, 2020; pp. 531–549. [Google Scholar] [CrossRef]
- Wang, G.N.; Zhang, L.; Song, Y.P.; Liu, J.X.; Wang, J.P. Application of molecularly imprinted polymer based matrix solid phase dispersion for determination of fluoroquinolones, tetracyclines and sulfonamides in meat. J. Chromatogr. B 2017, 1065–1066, 104–111. [Google Scholar] [CrossRef]
- Shen, Q.; Jin, R.; Xue, J.; Lu, Y.; Dai, Z. Analysis of trace levels of sulfonamides in fish tissue using micro-scale pipette tip-matrix solid-phase dispersion and fast liquid chromatography tandem mass spectrometry. Food Chem. 2016, 194, 508–515. [Google Scholar] [CrossRef]
- Tao, Y.; Zhu, F.; Chen, D.; Wei, H.; Pan, Y.; Wang, X.; Liu, Z.; Huang, L.; Wang, Y.; Yuan, Z. Evaluation of matrix solid-phase dispersion (MSPD) extraction for multi-fenicols determination in shrimp and fish by liquid chromatography-electrospray ionisation tandem mass spectrometry. Food Chem. 2014, 150, 500–506. [Google Scholar] [CrossRef]
- Da Silva, M.C.; Orlando, R.M.; Faria, A.F. Electrical field assisted matrix solid phase dispersion as a powerful tool to improve the extraction efficiency and clean-up of fluoroquinolones in bovine milk. J. Chromatogr. A 2016, 1461, 27–34. [Google Scholar] [CrossRef]
- Mu, G.; Liu, H.; Xu, L.; Tian, L.; Luan, F. Matrix solid-phase dispersion extraction and capillary electrophoresis determination of tetracycline residues in milk. Food Anal. Methods 2011, 5, 148–153. [Google Scholar] [CrossRef]
- Huang, Z.; Pan, X.D.; Huang, B.F.; Xu, J.J.; Wang, M.L.; Ren, Y.P. Determination of 15 β-lactam antibiotics in pork muscle by matrix solid-phase dispersion extraction (MSPD) and ultra-high pressure liquid chromatography tandem mass spectrometry. Food Control. 2016, 66, 145–150. [Google Scholar] [CrossRef]
- Capriotti, A.L.; Cavaliere, C.; Laganà, A.; Piovesana, S.; Samperi, R. Recent trends in matrix solid-phase dispersion. TrAC Trends Anal. Chem. 2013, 43, 53–66. [Google Scholar] [CrossRef]
- Capriotti, A.L.; Cavaliere, C.; Foglia, P.; Samperi, R.; Stampachiacchiere, S.; Ventura, S.; Laganà, A. Recent advances and developments in matrix solid-phase dispersion. TrAC Trends Anal. Chem. 2015, 71, 186–193. [Google Scholar] [CrossRef]
- Chico, J.; Rubies, A.; Centrich, F.; Companyo, R.; Prat, M.D.; Granados, M. Use of gel permeation chromatography for clean-up in the analysis of coccidiostats in eggs by liquid chromatography-tandem mass spectrometry. Anal. Bioanal. Chem. 2013, 405, 4777–4786. [Google Scholar] [CrossRef]
- Aguilera-Luiz, M.M.; Romero-Gonzalez, R.; Plaza-Bolanos, P.; Vidal, J.L.; Frenich, A.G. Rapid and semiautomated method for the analysis of veterinary drug residues in honey based on turbulent-flow liquid chromatography coupled to ultrahigh-performance liquid chromatography-Orbitrap mass spectrometry (TFC-UHPLC-Orbitrap-MS). J. Agric. Food Chem. 2013, 61, 829–839. [Google Scholar] [CrossRef] [PubMed]
- Samanidou, V.; Galanopoulos, L.D.; Kabir, A.; Furton, K.G. Fast extraction of amphenicols residues from raw milk using novel fabric phase sorptive extraction followed by high-performance liquid chromatography-diode array detection. Anal. Chim. Acta 2015, 855, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Lu, L.; He, J.; Zhao, T. Preparation of hydrophilic molecularly imprinted solid-phase microextraction fiber for the selective removal and extraction of trace tetracyclines residues in animal derived foods. J. Sep. Sci. 2020, 43, 2172–2179. [Google Scholar] [CrossRef] [PubMed]
- Charitonos, S.; Samanidou, V.F.; Papadoyannis, I. Development of an HPLC-DAD method for the determination of five sulfonamides in shrimps and validation according to the European decision 657/2002/EC. Food Anal. Methods 2017, 10, 2011–2017. [Google Scholar] [CrossRef]
- Tajabadi, F.; Ghambarian, M.; Yamini, Y.; Yazdanfar, N. Combination of hollow fiber liquid phase microextraction followed by HPLC-DAD and multivariate curve resolution to determine antibacterial residues in foods of animal origin. Talanta 2016, 160, 400–409. [Google Scholar] [CrossRef] [PubMed]
- Mookantsa, S.O.; Dube, S.; Nindi, M.M. Development and application of a dispersive liquid-liquid microextraction method for the determination of tetracyclines in beef by liquid chromatography mass spectrometry. Talanta 2016, 148, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Mor, F.; Kocasari, F.S.; Ozdemir, G.; Oz, B. Determination of sulphonamide residues in cattle meats by the Charm-II system and validation with high performance liquid chromatography with fluorescence detection. Food Chem. 2012, 134, 1645–1649. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Xie, X.; Zhao, X.; Xie, K.; Diao, Z.; Zhang, G.; Zhang, T.; Dai, G. Development of an accelerated solvent extraction-ultra-performance liquid chromatography-fluorescence detection method for quantitative analysis of thiamphenicol, florfenicol and florfenicol amine in poultry eggs. Molecules 2019, 24, 1830. [Google Scholar] [CrossRef]
- Yu, H.; Mu, H.; Hu, Y.M. Determination of fluoroquinolones, sulfonamides, and tetracyclines multiresidues simultaneously in porcine tissue by MSPD and HPLC-DAD. J. Pharm. Anal. 2012, 2, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Evaggelopoulou, E.N.; Samanidou, V.F. Development and validation of an HPLC method for the determination of six penicillin and three amphenicol antibiotics in gilthead seabream (Sparus Aurata) tissue according to the European Union Decision 2002/657/EC. Food Chem. 2013, 136, 1322–1329. [Google Scholar] [CrossRef]
- Kim, B.J.; Ham, H.S.; Lee, J.J.; Cheong, N.Y.; Myung, S.W. Determination of coccidiostats (Amprolium and Decoquinate) in cattle and chicken’s muscle using high performance liquid chromatography. Bull. Korean Chem. Soc. 2012, 33, 559–563. [Google Scholar] [CrossRef]
- Chitescu, C.L.; Nicolau, A.I.; Csuma, A.; Moisoiu, C. Simultaneous analysis of four sulfonamides in chicken muscle tissue by HPLC. Food Addit. Contam. A 2011, 28, 1013–1020. [Google Scholar] [CrossRef] [PubMed]
- Negarian, M.; Mohammadinejad, A.; Mohajeri, S.A. Preparation, evaluation and application of core-shell molecularly imprinted particles as the sorbent in solid-phase extraction and analysis of lincomycin residue in pasteurized milk. Food Chem. 2019, 288, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, F.A.; Nasr, J.J.M. Direct determination of ampicillin and amoxicillin residues in food samples after aqueous SDS extraction by micellar liquid chromatography with UV detection. Anal. Methods 2014, 6, 1523–1529. [Google Scholar] [CrossRef]
- Lv, Y.K.; Zhang, J.Q.; Guo, Z.Y.; Zhang, W.; Sun, H.W. Determination of tetracyclines residues in egg, milk, and milk powder by online coupling of a precolumn packed with molecular imprinted hybrid composite materials to RP-HPLC-UV. J. Liq. Chromatogr. Relat. Technol. 2014, 38, 1–7. [Google Scholar] [CrossRef]
- Zhou, Y.; Liu, H.; Li, J.; Sun, Z.; Cai, T.; Wang, X.; Zhao, S.; Gong, B. Restricted access magnetic imprinted microspheres for directly selective extraction of tetracycline veterinary drugs from complex samples. J. Chromatogr. A 2020, 1613, 460684. [Google Scholar] [CrossRef]
- Karimi, M.; Aboufazeli, F.; Zhad, H.R.L.Z.; Sadeghi, O.; Najafi, E. Determination of sulfonamides in chicken meat by magnetic molecularly imprinted polymer coupled to HPLC-UV. Food Anal. Methods 2013, 7, 73–80. [Google Scholar] [CrossRef]
- Hui, W.; Li, Q.; Ma, H.; Wu, M.; Feng, K.; Zhu, H.; Yang, P.; Li, J.; Chen, C.; Yan, K. Rapid screening for 15 sulfonamide residues in foods of animal origin by high-performance liquid chromatography–UV method. J. Chromatogr. Sci. 2018, 56, 636–643. [Google Scholar] [CrossRef]
- Liu, Q.; Li, J.; Song, X.; Zhang, M.; Li, E.; Gao, F.; He, L. Simultaneous determination of aminoglycoside antibiotics in feeds using high performance liquid chromatography with evaporative light scattering detection. RSC Adv. 2017, 7, 1251–1259. [Google Scholar] [CrossRef]
- Marazuela, M.D. Determination of veterinary drug residues in foods by liquid chromatography-mass spectrometry: Basic and cutting-edge applications. In Liquid Chromatography; Fanali, S., Haddad, P.R., Poole, C.F., Riekkola, M.L., Eds.; Elsevier: Madrid, Spain, 2017; pp. 539–570. [Google Scholar]
- Delatour, T.; Racault, L.; Bessaire, T.; Desmarchelier, A. Screening of veterinary drug residues in food by LC-MS/MS. Background and challenges. Food Addit. Contam. A 2018, 35, 632–645. [Google Scholar] [CrossRef]
- Xu, Y.; Ding, J.; Chen, H.; Zhao, Q.; Hou, J.; Yan, J.; Wang, H.; Ding, L.; Ren, N. Fast determination of sulfonamides from egg samples using magnetic multiwalled carbon nanotubes as adsorbents followed by liquid chromatography-tandem mass spectrometry. Food Chem. 2013, 140, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Pang, M.; Xie, X.; Zhao, M.; Xie, K.; Zhang, Y.; Zhao, X.; Wang, Y.J.; Wang, R.; Wu, H.; et al. Quantitative analysis of amoxicillin, amoxicillin major metabolites, and ampicillin in chicken tissues via ultra-performance liquid chromatography-electrospray ionization tandem mass spectrometry. Food Anal. Methods 2017, 10, 3292–3305. [Google Scholar] [CrossRef]
- Anumol, T.; Lehotay, S.J.; Stevens, J.; Zweigenbaum, J. Comparison of veterinary drug residue results in animal tissues by ultrahigh-performance liquid chromatography coupled to triple quadrupole or quadrupole-time-of-flight tandem mass spectrometry after different sample preparation methods, including use of a commercial lipid removal product. Anal. Bioanal. Chem. 2017, 409, 2639–2653. [Google Scholar] [CrossRef]
- Tao, Y.; Chen, D.; Yu, G.; Yu, H.; Pan, Y.; Wang, Y.; Huang, L.; Yuan, Z. Simultaneous determination of lincomycin and spectinomycin residues in animal tissues by gas chromatography-nitrogen phosphorus detection and gas chromatography-mass spectrometry with accelerated solvent extraction. Food Addit. Contam. A 2011, 28, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Caban, M.; Haliński, Ł.; Kumirska, J.; Stepnowski, P. Gas chromatography applied to the analysis of drug and veterinary drug residues in food, environmental, and biological samples. In Determination of Target Xenobiotics and Unknown Compound Residues in Food, Environmental, and Biological Samples; Sherma, J., Tuzimski, T., Eds.; CRC Press: Boca Raton, FL, USA, 2018; pp. 133–166. [Google Scholar]
- Wang, B.; Pang, M.; Xie, X.; Xie, K.; Zhang, Y.; Cui, L.; Zhao, X.; Wang, Y.; Shi, H.; Guo, Y.; et al. Quantification of piperazine in chicken and pig tissues by gas chromatography-electron ionization tandem mass spectrometry employing pre-column derivatization with acetic anhydride. J. Chromatogr. A 2017, 1519, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Leung, D.; Chow, W.; Chang, J.; Wong, J.W. Development and validation of a multiclass method for analysis of veterinary drug residues in milk using ultrahigh performance liquid chromatography electrospray ionization quadrupole orbitrap mass spectrometry. J. Agric. Food Chem. 2015, 63, 9175–9187. [Google Scholar] [CrossRef]
- Song, X.; Zhou, T.; Li, J.; Su, Y.; Xie, J.; He, L. Determination of macrolide antibiotics residues in pork using molecularly imprinted dispersive solid-phase extraction coupled with LC-MS/MS. J. Sep. Sci. 2018, 41, 1138–1148. [Google Scholar] [CrossRef]
- Tao, Y.; Chen, D.; Yu, H.; Huang, L.; Liu, Z.; Cao, X.; Yan, C.; Pan, Y.; Liu, Z.; Yuan, Z. Simultaneous determination of 15 aminoglycoside(s) residues in animal derived foods by automated solid-phase extraction and liquid chromatography-tandem mass spectrometry. Food Chem. 2012, 135, 676–683. [Google Scholar] [CrossRef] [PubMed]
- Stoilova, N.; Surleva, A.; Stoev, G. Determination of quinolonones in food of animal origin by liquid chromatography coupled with fluorescence and mass spectrometric detection. Acta Chromatogr. 2014, 26, 599–614. [Google Scholar] [CrossRef]
- Juan, C.; Moltó, J.C.; Mañes, J.; Font, G. Determination of macrolide and lincosamide antibiotics by pressurised liquid extraction and liquid chromatography-tandem mass spectrometry in meat and milk. Food Control. 2010, 21, 1703–1709. [Google Scholar] [CrossRef]
- Shen, J.; Xia, X.; Jiang, H.; Li, C.; Li, J.; Li, X.; Ding, S. Determination of chloramphenicol, thiamphenicol, florfenicol, and florfenicol amine in poultry and porcine muscle and liver by gas chromatography-negative chemical ionization mass spectrometry. J. Chromatogr. B 2009, 877, 1523–1529. [Google Scholar] [CrossRef]
- Yikilmaz, Y.; Filazi, A. Detection of florfenicol residues in salmon trout via GC–MS. Food Anal. Methods 2014, 8, 1027–1033. [Google Scholar] [CrossRef]
- Xue, L.C.; Cai, Q.R.; Zheng, X.; Liu, L.; Ling, Y.H.; Li, Z.; Peng, Y.F.; Chen, J.; Cai, D.L.; Cai, J.; et al. Determination of 9 hydroxy veterinary drug residues in fish by QuEChERS-GPC-GC/MS. J. Chin. Mass Spectrom. Soc. 2017, 38, 655–663. [Google Scholar] [CrossRef]
- Zhao, X.; Wang, B.; Xie, K.; Liu, Y.; Zhang, Y.; Wang, Y.; Guo, Y.; Bu, X.; Liu, C.; Zhang, G.; et al. Determination of dinitolmide and its metabolite 3-ANOT in chicken tissues via ASE-SPE-GC–MS/MS. J. Food Compos. Anal. 2018, 71, 94–103. [Google Scholar] [CrossRef]
- Zhao, X.; Wang, B.; Xie, K.; Liu, Y.; Zhang, Y.; Wang, Y.; Liu, C.; Guo, Y.; Bu, X.; Zhang, G.; et al. Development of an ASE-GC-MS/MS method for detecting dinitolmide and its metabolite 3-ANOT in eggs. J. Mass Spectrom. 2018, 53, 976–985. [Google Scholar] [CrossRef]
- An, L.; Wang, Y.; Pan, Y.; Tao, Y.; Chen, D.; Liu, Z.; Yang, W.; Peng, D.; Yuan, Z. Development and validation of a sensitive indirect competitive enzyme-linked immunosorbent assay for the screening of florfenicol and thiamphenicol in edible animal tissue and feed. Food Anal. Methods 2016, 9, 2434–2443. [Google Scholar] [CrossRef]
- Cao, S.; Song, S.; Liu, L.; Kong, N.; Kuang, H.; Xu, C. Comparison of an enzyme-linked immunosorbent assay with an immunochromatographic assay for detection of lincomycin in milk and honey. Immunol. Investig. 2015, 44, 438–450. [Google Scholar] [CrossRef]
- Wang, Z.; Mi, T.; Beier, R.C.; Zhang, H.; Sheng, Y.; Shi, W.; Zhang, S.; Shen, J. Hapten synthesis, monoclonal antibody production and development of a competitive indirect enzyme-linked immunosorbent assay for erythromycin in milk. Food Chem. 2015, 171, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Tian, W.; Zhang, X.; Song, M.; Jiang, H.; Ding, S.; Shen, J.; Li, J. An enzyme-linked immunosorbent assay to detect salinomycin residues based on immunomagnetic bead clean-up. Food Anal. Methods 2017, 10, 3042–3051. [Google Scholar] [CrossRef]
- Zhou, Q.; Peng, D.; Wang, Y.; Pan, Y.; Wan, D.; Zhang, X.; Yuan, Z. A novel hapten and monoclonal-based enzyme-linked immunosorbent assay for sulfonamides in edible animal tissues. Food Chem. 2014, 154, 52–62. [Google Scholar] [CrossRef] [PubMed]
- Bahmani, K.; Shahbazi, Y.; Nikousefat, Z. Monitoring and risk assessment of tetracycline residues in foods of animal origin. Food Sci. Biotechnol. 2020, 29, 441–448. [Google Scholar] [CrossRef]
- Jiang, W.; Wang, Z.; Beier, R.C.; Jiang, H.; Wu, Y.; Shen, J. Simultaneous determination of 13 fluoroquinolone and 22 sulfonamide residues in milk by a dual-colorimetric enzyme-linked immunosorbent assay. Anal. Chem. 2013, 85, 1995–1999. [Google Scholar] [CrossRef] [PubMed]
- Solomun, B.; Bilandzic, N.; Varenina, I.; Scortichini, G. Validation of an enzyme-linked immunosorbent assay for qualitative screening of neomycin in muscle, liver, kidney, eggs and milk. Food Addit. Contam. A 2011, 28, 11–18. [Google Scholar] [CrossRef]
- Jiao, S.N.; Liu, J.; Zhang, Y.F.; Zhao, G.X.; Wang, J.P. Preparation of a bi-hapten antigen and the broad-specific antibody for simultaneous immunoassay of penicillins and tetracyclines in milk. Food Agric. Immunol. 2012, 23, 273–287. [Google Scholar] [CrossRef]
- Wei, D.; Meng, H.; Zeng, K.; Huang, Z. Visual dual dot immunoassay for the simultaneous detection of kanamycin and streptomycin in milk. Anal. Methods 2019, 11, 70–77. [Google Scholar] [CrossRef]
- European Commission Commission decision 2002/657/EC of 12 august 2002 implementing council directive 96/23/EC concerning the performance of analytical methods and the interpretation of results. Off. J. Eur. Commun. 2002, 50, 8–36.
- El-Attug, M.N.; Adams, E.; Hoogmartens, J.; Van Schepdael, A. Capacitively coupled contactless conductivity detection as an alternative detection mode in CE for the analysis of kanamycin sulphate and its related substances. J. Sep. Sci. 2011, 34, 2448–2454. [Google Scholar] [CrossRef] [PubMed]
- Donegatti, T.A.; Lobato, A.; Duek, E.A.R.; Goncalves, L.M.; Alves Pereira, E. Derivatization-free determination of aminoglycosides by CZE-UV in pharmaceutical formulations. Electrophoresis 2020, 41, 1576–1583. [Google Scholar] [CrossRef]
- Li, Z.; Li, Y.; Qi, M.; Zhong, S.; Wang, W.; Wang, A.J.; Chen, J. Graphene-Fe3 O4 as a magnetic solid-phase extraction sorbent coupled to capillary electrophoresis for the determination of sulfonamides in milk. J. Sep. Sci. 2016, 39, 3818–3826. [Google Scholar] [CrossRef]
- Shuo, Z.; Hao-Tian, W.; Ke, L.I.; Jing, Z.; Xia-Yan, W.; Guang-Sheng, G.U.O. Fast determination of residual sulfonamides in milk by in-tube solid-phase microextraction coupled with capillary electrophoresis-laser induced fluorescence. Chin. J. Anal. Chem. 2018, 46, e1810–e1816. [Google Scholar] [CrossRef]
- Liu, Y.M.; Yang, Y.; Li, J.; Peng, L.F.; Mei, L. A validated method for the sensitive determination of macrolide antibiotics by capillary electrophoresis with electrochemiluminescence detection and its applications. Curr. Anal. Chem. 2011, 7, 325–332. [Google Scholar] [CrossRef]
- Ibarra, I.S.; Rodriguez, J.A.; Paez-Hernandez, M.E.; Santos, E.M.; Miranda, J.M. Determination of quinolones in milk samples using a combination of magnetic solid-phase extraction and capillary electrophoresis. Electrophoresis 2012, 33, 2041–2048. [Google Scholar] [CrossRef]
- Moreno-Gonzalez, D.; Lara, F.J.; Gamiz-Gracia, L.; Garcia-Campana, A.M. Molecularly imprinted polymer as in-line concentrator in capillary electrophoresis coupled with mass spectrometry for the determination of quinolones in bovine milk samples. J. Chromatogr. A 2014, 1360, 1–8. [Google Scholar] [CrossRef]
- Moreno-Gonzalez, D.; Lara, F.J.; Jurgovska, N.; Gamiz-Gracia, L.; Garcia-Campana, A.M. Determination of aminoglycosides in honey by capillary electrophoresis tandem mass spectrometry and extraction with molecularly imprinted polymers. Anal. Chim. Acta 2015, 891, 321–328. [Google Scholar] [CrossRef]
- Dai, T.; Duan, J.; Li, X.; Xu, X.; Shi, H.; Kang, W. Determination of sulfonamide residues in food by capillary zone electrophoresis with on-line chemiluminescence detection based on an Ag(III) complex. Int. J. Mol. Sci. 2017, 18, 1286. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Ma, S.; Zhu, K.; Wang, M.; Li, J.; Arabi, M.; Liu, H.; Li, Y.; Chen, L. Simultaneous enrichment/determination of six sulfonamides in animal husbandry products and environmental waters by pressure-assisted electrokinetic injection coupled with capillary zone electrophoresis. J. Food Compos. Anal. 2020, 88, 103462. [Google Scholar] [CrossRef]
- Hu, X.; Yang, J.; Chen, C.; Khan, H.; Guo, Y.; Yang, L. Capillary electrophoresis-integrated immobilized enzyme microreactor utilizing single-step in-situ penicillinase-mediated alginate hydrogelation: Application for enzyme assays of penicillinase. Talanta 2018, 189, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Islas, G.; Rodriguez, J.A.; Perez-Silva, I.; Miranda, J.M.; Ibarra, I.S. Solid-phase extraction and large-volume sample stacking-capillary electrophoresis for determination of tetracycline residues in milk. J. Anal. Methods Chem. 2018, 2018, 5394527. [Google Scholar] [CrossRef]
- Wang, N.; Su, M.; Liang, S.; Sun, H. Sensitive residue analysis of quinolones and sulfonamides in aquatic product by capillary zone electrophoresis using large-volume sample stacking with polarity switching combined with accelerated solvent extraction. Food Anal. Methods 2015, 9, 1020–1028. [Google Scholar] [CrossRef]
- Ferreira, T.A.; Flores-Aguilar, J.F.; Santos, E.M.; Rodriguez, J.A.; Ibarra, I.S. New poly(ionic liquid) based fiber for determination of oxytetracycline in milk samples by application of SPME-CE technique. Molecules 2019, 24, 430. [Google Scholar] [CrossRef]
- Li, M.H.; He, W.S.; Zhang, L.J.; Duan, C.L. Analysis of penicillin and its β-lactamase hydrolysis products in milk using capillary zone electrophoresis. Anal. Methods 2015, 7, 4602–4607. [Google Scholar] [CrossRef]
- Moreno-Gonzalez, D.; Hamed, A.M.; Gilbert-Lopez, B.; Gamiz-Gracia, L.; Garcia-Campana, A.M. Evaluation of a multiresidue capillary electrophoresis-quadrupole-time-of-flight mass spectrometry method for the determination of antibiotics in milk samples. J. Chromatogr. A 2017, 1510, 100–107. [Google Scholar] [CrossRef]
- Lorenzetti, A.S.; Lista, A.G.; Domini, C.E. Reverse ultrasound-assisted emulsification-microextraction of macrolides from chicken fat followed by electrophoretic determination. LWT 2019, 113, 108334. [Google Scholar] [CrossRef]
- Chen, B.; Du, Y. Evaluation of the enantioseparation capability of the novel chiral selector clindamycin phosphate towards basic drugs by micellar electrokinetic chromatography. J. Chromatogr. A 2010, 1217, 1806–1812. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Zhong, Z.; Xia, Z.; Yang, F.; Mu, X. Separation of fluoroquinolones by MEKC modified with hydrophobic ionic liquid as a modifier. Chromatographia 2011, 75, 65–70. [Google Scholar] [CrossRef]
- Kowalski, P.; Konieczna, L.; Olędzka, I.; Plenis, A.; Bączek, T. Development and validation of electromigration technique for the determination of lincomycin and clindamycin residues in poultry tissues. Food Anal. Methods 2013, 7, 276–282. [Google Scholar] [CrossRef]
- Belal, F.; El-Razeq, S.A.; Fouad, M.; Zayed, S.; Fouad, F. Simultaneous determination of five coccidiostats in veterinary powders, feed premixes, and baby food by micellar electrokinetic chromatography: Application to chicken tissues and liver. Food Anal. Methods 2018, 11, 3531–3541. [Google Scholar] [CrossRef]
- Springer, V.H.; Lista, A.G. Micellar nanotubes dispersed electrokinetic chromatography for the simultaneous determination of antibiotics in bovine milk. Electrophoresis 2012, 33, 2049–2055. [Google Scholar] [CrossRef]
- Kowalski, P.; Plenis, A.; Oledzka, I.; Konieczna, L. Optimization and validation of the micellar electrokinetic capillary chromatographic method for simultaneous determination of sulfonamide and amphenicol-type drugs in poultry tissue. J. Pharm. Biomed. Anal. 2011, 54, 160–167. [Google Scholar] [CrossRef]
- Shao, Y.X.; Chen, G.H.; Fang, R.; Zhang, L.; Yi, L.X.; Meng, H.L. Analysis of six beta-lactam residues in milk and egg by micellar electrokinetic chromatography with large-volume sample stacking and polarity switching. J. Agric. Food Chem. 2016, 64, 3456–3461. [Google Scholar] [CrossRef] [PubMed]
- Ge, L.; Liu, Q.; Hao, N.; Kun, W. Recent developments of photoelectrochemical biosensors for food analysis. J. Mater. Chem. B 2019, 7, 7283–7300. [Google Scholar] [CrossRef] [PubMed]
- Cervera-Chiner, L.; Jiménez, Y.; Montoya, Á.; Juan-Borrás, M.; Pascual, N.; Arnau, A.; Escriche, I. High fundamental frequency quartz crystal microbalance (HFF-QCMD) immunosensor for detection of sulfathiazole in honey. Food Control. 2020, 115, 107296. [Google Scholar] [CrossRef]
- Majdinasab, M.; Mitsubayashi, K.; Marty, J.L. Optical and electrochemical sensors and biosensors for the detection of quinolones. Trends Biotechnol. 2019, 37, 898–915. [Google Scholar] [CrossRef] [PubMed]
- Jalalian, S.H.; Karimabadi, N.; Ramezani, M.; Abnous, K.; Taghdisi, S.M. Electrochemical and optical aptamer-based sensors for detection of tetracyclines. Trends Food Sci. Technol. 2018, 73, 45–57. [Google Scholar] [CrossRef]
- Sadeghi, A.S.; Ansari, N.; Ramezani, M.; Abnous, K.; Mohsenzadeh, M.; Taghdisi, S.M.; Alibolandi, M. Optical and electrochemical aptasensors for the detection of amphenicols. Biosens. Bioelectron. 2018, 118, 137–152. [Google Scholar] [CrossRef] [PubMed]
- Kharewal, T.; Verma, N.; Gahlaut, A.; Hooda, V. Biosensors for penicillin quantification: A comprehensive review. Biotechnol. Lett. 2020, 42, 1829–1846. [Google Scholar] [CrossRef] [PubMed]
- Mehlhorn, A.; Rahimi, P.; Joseph, Y. Aptamer-based biosensors for antibiotic detection: A review. Biosensors 2018, 8, 54. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.P.; Zhang, Y.C.; Zhang, X.; Shen, L. Green preparation of chlorine-doped graphene and its application in electrochemical sensor for chloramphenicol detection. SN Appl. Sci. 2019, 1, 157. [Google Scholar] [CrossRef]
- Benvidi, A.; Yazdanparast, S.; Rezaeinasab, M.; Tezerjani, M.D.; Abbasi, S. Designing and fabrication of a novel sensitive electrochemical aptasensor based on poly (L-glutamic acid)/MWCNTs modified glassy carbon electrode for determination of tetracycline. J. Electroanal. Chem. 2018, 808, 311–320. [Google Scholar] [CrossRef]
- Shi, X.; Ren, X.; Jing, N.; Zhang, J. Electrochemical determination of ampicillin based on an electropolymerized poly(o-phenylenediamine)/gold nanoparticle/single-walled carbon nanotube modified glassy carbon electrode. Anal. Lett. 2020, 53, 2854–2867. [Google Scholar] [CrossRef]
- Zhao, J.; Guo, W.; Pei, M.; Ding, F. GR–Fe3O4NPs and PEDOT–AuNPs composite based electrochemical aptasensor for the sensitive detection of penicillin. Anal. Methods 2016, 8, 4391–4397. [Google Scholar] [CrossRef]
- Li, Z.; Liu, C.; Sarpong, V.; Gu, Z. Multisegment nanowire/nanoparticle hybrid arrays as electrochemical biosensors for simultaneous detection of antibiotics. Biosens. Bioelectron. 2019, 126, 632–639. [Google Scholar] [CrossRef]
- Nie, R.; Xu, X.; Chen, Y.; Yang, L. Optical fiber-mediated immunosensor with a tunable detection range for multiplexed analysis of veterinary drug residues. ACS Sens. 2019, 4, 1864–1872. [Google Scholar] [CrossRef]
- Ebarvia, B.S.; Ubando, I.E.; Sevilla, F.B. Biomimetic piezoelectric quartz crystal sensor with chloramphenicol-imprinted polymer sensing layer. Talanta 2015, 144, 1260–1265. [Google Scholar] [CrossRef] [PubMed]
- Roushani, M.; Rahmati, Z.; Hoseini, S.J.; Fath, R.H. Impedimetric ultrasensitive detection of chloramphenicol based on aptamer MIP using a glassy carbon electrode modified by 3-ampy-RGO and silver nanoparticle. Colloids Surf. B Biointerfaces 2019, 183, 110451. [Google Scholar] [CrossRef] [PubMed]
- Caglayan, M.O. Aptamer-based ellipsometric sensor for ultrasensitive determination of aminoglycoside group antibiotics from dairy products. J. Sci. Food Agric. 2020, 100, 3386–3393. [Google Scholar] [CrossRef]
- Ismail, F.; Adeloju, S.B.; Moline, A.N. Fabrication of a single layer and bilayer potentiometric biosensors for penicillin by galvanostatic entrapment of penicillinase into polypyrrole films. Electroanalysis 2014, 26, 2607–2618. [Google Scholar] [CrossRef]

| Animal-Derived Food | Cartridge Type | Extraction Recovery (%) | LOD (μg/kg or μg/L) | Ref. |
|---|---|---|---|---|
| Eggs | CNWBOND LC-C18 (6 mL/150 mg) | 71.7–102.7 | 0.16–0.52 | [61] |
| Animal tissue, fish and honey | EVOLUTE ABN (3 mL/200 mg) | 50.0–120.0 | ≥1.0 | [62] |
| Milk and fish tissue | OASIS HLB (3 mL/60 mg) | – | 15.0–200 | [63] |
| Fish, shrimp and eel | OASIS PRIME HLB (6 mL/200 mg) | 70.0–120.0 | 0.15–100 | [64] |
| Dairy products | OASIS HLB (6 mL/200 mg) | 67.3–106.9 | 0.006–0.3 | [65] |
| Bovine muscle | OASIS HLB (6 mL/200 mg) | 37.4–106.0 | – | [66] |
| Milk | OASIS HLB (3 mL/60 mg) | 68.0–118.0 | 0.01–5 | [67] |
| Eggs | Hybrid SPE (1 mL/30 mg) | 75.0–108.0 | – | [68] |
| Extraction Method | Pros | Cons |
|---|---|---|
| LLE | Simple, reliable, and widely applicable | Consumption of organic reagents and time consuming |
| SPE | Less time consuming than LLE Good purification effect and reproducibility | High cost of SPE cartridges Requires pre-treatment and toxic organic solvent |
| ASE | Low consumption of organic reagents Time saving Batch processing of samples Automated, fast, and convenient | High temperature and pressure, operation requires professional training |
| QuEChERS | Flexible and effective Simple instrumentation Low reagent consumption Wide scope of acidic and basic analytes | Low enrichment factors Low recovery of polar analytes |
| MSPD | Simple, efficient, and fast Low reagent consumption Wide scope of molecular structures and polar analytes | Relatively high degree of crushed samples |
| Class of Veterinary Drugs | Animal-Derived Food | Sample Preparation Method | Ref. |
|---|---|---|---|
| MACs (12), LAs (2) and other contaminants (9) | Milk | LLE: 2 mL fresh milk sample + 15 mL ACN | [55] |
| MACs (10), Qs (15), TCs (5), SAs (27) and other contaminants (27) | Chicken muscle | LLE: 2 g sample + 5 mL EDTA-succinate + 10 mL ACN + 2 g sodium chloride | [57] |
| PCNs (2), APs (3), MACs (6), Qs (11), TCs (4), LAs (1), SAs (18), COCs (8) and other contaminants (62) | Milk powder, butter, fish tissue and eggs | LLE: 1 g sample + 2 mL 0.1% EDTA in H2O with 0.1% formic acid + 2 mL ACN + 2 mL MeOH | [58] |
| APs (4) | Eggs | LLE: 5 g sample + 1 mL ACN:water (30:70, v/v) + 20 mL ethyl acetate:ACN:ammonium hydroxide (49:49:2, v/v) | [60] |
| COCs (8) | Eggs | SPE: 2 g sample + 2 mL ultrapure water + 16 mL ACN: ethyl acetate (60:40, v/v):acetic acid (98:2, v/v) + CNWBOND C18 150 mg, elution 15 mL ethyl acetate | [61] |
| PCNs (7), APs (2), MACs (5), Qs (10), TCs (5), LAs (1), SAs (19), COCs (13) and other contaminants (81) | Milk and fish tissue | LLE: 1 g fish tissue sample + 2 mL 0.1% EDTA in H2O with 0.1% formic acid + 2 mL ACN + 2 mL MeOH SPE: 2 mL milk sample + 16 mL 5% trichloroacetic acid (TCA) in H2O:ACN (3:1, v/v) + 15% ammonia hydroxide (NH3·H2O) + Oasis HLB 60 mg, elution 6 mL MeOH | [63] |
| PCNs (6), MACs (6), AGs (6), SAs (14), COCs (12) and other contaminants (32) | Bovine muscle | SPE: 5 g sample + 10 mL ACN + 20 mL extraction solvent (consisting of 10 mM ammonium acetate, 0.4 mM EDTA, 1% NaCl and 2% TCA in H2O) + Oasis HLB 200 mg, elution 1 mL 10% formic acid and 3 mL ACN | [66] |
| MACs (3), Qs (8), TCs (4), LAs (1), SAs (8) and other contaminants (14) | Milk | SPE: 1 mL sample + 0.5 mL water + 3 mL ACN + 3 mL 0.1 mol/L phosphate buffer solution (PBS) + Oasis HLB 60 mg, elution 3 mL ACN:water (1:1, v/v) | [67] |
| AGs (1) and LAs (1) | Poultry eggs | ASE: 2 g sample + 4 g diatomaceous earth + 0.01 M KH2PO4 solution (a total solvent rinse of 50%), two cycles + 2 mL 0.2 M sodium dodecyl sulphonate (SDS) solution + Oasis PRiME HLB 60 mg, elution 6 mL MeOH | [12] |
| MACs (17) and other contaminants (1) | Swine and bovine tissues (muscle, kidney and liver) | ASE: 2 g sample + 12 g EDTA-treated sand + ACN: MeOH (1:1, v/v) (a total solvent rinse of 60%), two cycles + 5 mL MeOH | [71] |
| TCs (7) | Porcine, chicken and bovine (muscle and liver) | ASE: 2 g sample + 5 g EDTA-treated sand + ACN and 1 mM TCA (pH 4.0) (a total solvent rinse of 50%), two cycles | [72] |
| APs (4) | Poultry eggs | ASE: 5 g sample + 4 g diatomaceous earth + MeOH:NH3·H2O:ultrapure water (97:2:1, v/v) (a total solvent rinse of 40%), one cycle + 1 mL ACN + 10 mL hexane saturated with ACN + 5 mL ACN:water (4:6, v/v) | [11] |
| PCNs (2), APs (1), MACs (2), SAs (4) and other contaminants (5) | Milk | QuEChERS: 10 g sample + 100 μL acetic acid + 10 mL ACN + 4 g MgSO4 + 50 mg chitosan + 150 mg MgSO4 | [75] |
| MACs (7), Qs (18), TCs (4), LAs (2), SAs (19) and other contaminants (40) | Royal jelly | QuEChERS: 1 g sample + 5 mL mixed solution of 0.1 M citric acid and 0.2 M Na2HPO4 (8:5, v/v, pH 4) + 20 mL 5% acetic acid in ACN + 2 g NaCl +2 g Na2SO4 + 200 mg NH2 sorbents | [76] |
| PCNs (2), APs (4), MACs (6), FQs (9), TCs (4), SAs (16) and other contaminants (9) | Flatfish, eel and shrimp | QuEChERS: 2 g sample + 1 mL 0.1 M EDTA in 50 mM ammonium acetate buffer solution (pH 4.0) + 9 mL 2 mM ammonium formate in water:ACN (1:4, v/v) + 250 mg PSA + 250 mg C18 sorbents | [77] |
| MACs (6), Qs (13), SAs (18) and other contaminants (18) | Porcine, bovine and ovine muscle | QuEChERS: 4 g sample + 16 mL 5% acetic acid in ACN + 2 g NaCl + 4 g Na2SO4 + 400 mg C18 sorbents | [80] |
| FQs (8), TCs (4) and SAs (8) | Pork | MSPD: 0.2 g sample + 0.15 g MMIP + 50 mg MMIP + 1 mL MeOH + 1 mL water + 3 mL MeOH:water (2:8, v/v) + 4 mL MeOH:acetic acid (9:1, v/v) | [84] |
| SAs (14) | Fish tissue | MSPD: 0.01 g sample + 0.02 g HLB + 2 mL ACN + 0.2 mL MeOH:water: NH3·H2O (50:49:1, v/v/v) | [85] |
| TCs (3) | Milk | MSPD: milk sample:sorbents (1:4, m/m) + 6 mL hexane + 6 mL 0.1 M citric acid aqueous solution:MeOH (1:9, v/v) | [88] |
| APs (3) | Fish muscle | MSPD: 2 g sample + 3 g C18 sorbents + 8 mL hexane + 10 mL ACN:water (1:1, v/v) + 6 mL ethyl acetate | [17] |
| APs (4), MACs (18), Qs (21), TCs (7), LAs (3), SAs (24) and other contaminants (43) | Edible muscles, eggs and milk | UAE: 2 g sample + 10 mL ACN:water (9:1, v/v) + 10 min UAE + 5 mL water + Oasis HLB 500 mg, elution 5 mL formic acid:MeOH (5:95, v/v) and 5 mL ethyl acetate | [18] |
| COCs (9) | Eggs | GPC: 2 g sample + 5 g anhydrous sodium sulfate + 10 mL ethyl acetate + online gel permeation chromatographic cleanup | [92] |
| PCNs (8), MACs (5), AGs (1), Qs (7), TCs (4), SAs (6) and other contaminants (9) | Honey | TFC: 1 g sample + 1 mL 0.1 M Na2EDTA (pH 4) + Millex-GN nylon filter (0.20 μm) + online sample extraction by TFC procedure | [93] |
| APs (3) | Milk | FPSE: FPSE media in 1 mL Cameo (1:1, v/v) + 0.5 g sample, kept for 30 min + remove the FPSE media from the extraction via and insert it into backextraction containing 0.5 mL MeOH for 10 min | [94] |
| TCs (2) | Chicken, fish and milk | SPME: 5 mL or 5 g sample + 20 mL Na2EDTA-McIlvaine extract buffer + a homemade SPME device, elution 2 mL ACN:formic acid (2:1, v/v) | [95] |
| SAs (5) | Shrimp | SLE: 0.5 g sample + 3 mL MeOH:ACN (50:50 v/v) + 0.5 mL MeOH:0.1% acetic acid aqueous solution (40:60 v/v) + the supernatant was transferred to the falcon tube + 0.5 mL MeOH:0.1% acetic acid aqueous solution (40:60 v/v) | [96] |
| TCs (4) and Qs (5) | Lamb and chicken tissues, fish, honey, and milk | LPME: 5 g lamb and chicken tissues and fish samples + 15 mL ACN + 5 g sodium sulfate + 19 aqueous solution mL (pH 12.0) 5 g honey sample + 5 mL 2 mol/L HCl + 10 mL NaOH solution (2 mol/L) 20 mL milk sample + 10 mL 0.5 mol/L K3[Fe(CN)6]·3H2O solution + 10 mL 2 mol/L Zn(CH3COO)2·2H2O | [97] |
| TCs (6) | Beef | DLLME: 1 g sample + 6 mL water:ACN (5:1, v/v) + 300 mg magnesium sulfate anhydrous + 150 mg sodium chloride + 50 mg trisodium citrate dehydrate + sodium hydroxide solution and formic acid, adjust to pH 7 + 1 mL methanol + 200 μL dichloromethane + 100 μL water | [98] |
| Class of Veterinary Drugs | Animal-Derived Food | Sample Preparation Method | Detection Method | Recovery (%) | RSD (%) | LOD (μg/kg or μg/L) | LOQ (μg/kg or μg/L) | Ref. |
|---|---|---|---|---|---|---|---|---|
| SAs (7) | Cattle meats | LLE | HPLC–FLD | 44.6–81.0 | 2.7–4.9 | 8–15 | 13–25 | [99] |
| APs (3) | Poultry eggs | ASE | UPLC–FLD | 80.1–98.6 | 1.2–4.3 | 1.8–4.9 | 4.3–11.7 | [100] |
| APs (3) | Milk | FPSE | HPLC–DAD | 92.3–106.0 | 1.0–10.7 | – | – | [94] |
| SAs (5) | Shrimp | SLE | HPLC–DAD | 90.2–109.0 | 1.5–14.4 | 15 | 50 | [96] |
| TCs (4) and Qs (5) | Lamb and chicken tissues, fish, honey, and milk | LPME | HPLC–DAD | 25.5–82.6 | 3.4–10.7 | 0.5–20 | 1.25–50 | [97] |
| FQs (2), TCs (1) and SAs (2) | Porcine tissues | MSPD | HPLC–DAD | 80.6–99.2 | 0.3–6.1 | 2–10 | 7–34 | [101] |
| PCNs (6) and APs (3) | Gilthead seabream tissues | SPE | HPLC–DAD | 95.6–104.0 | 0.3–6.7 | 11.0–20.4 | 33.2–61.7 | [102] |
| COCs (2) | Cattle and chicken muscle | SPE | HPLC–UVD | 78.5–107.1 | 2.2–10.9 | 40–130 | 130–420 | [103] |
| SAs (4) | Chicken muscle | LLE | HPLC–UVD | 70.0–84.0 | 8.0–13.0 | 0.14–6.53 | 0.42–19.6 | [104] |
| LAs (1) | Milk | SPE | HPLC–UVD | 80.0–89.0 | 0.8–4.7 | 20 | 80 | [105] |
| PCNs (2) | Eggs, chicken and bovine tissues | LLE | HPLC–UVD | 95.5–102.3 | 0.4–1.2 | 500–1300 | 1700–4500 | [106] |
| TCs (3) | Eggs, milk and milk powder | SPE | HPLC–UVD | 85.3–98.3 | 1.9–5.3 | 0.76–1.13 | 2.53–3.77 | [107] |
| TCs (4) | Milk and eggs | SPE | HPLC–UVD | 84.2–98.6 | 1.4–5.9 | 1.03–2.67 | 3.46–8.97 | [108] |
| SAs (4) | Chicken meat | SPE | HPLC–UVD | 92.0–106.0 | 3.8–6.7 | 0.5–150 | – | [109] |
| SAs (15) | Milk, pork, beef and mutton tissues | LLE | HPLC–UVD | 81.5–95.3 | 0.8–7.4 | 6.5–11.0 | – | [110] |
| MACs (7), Qs (18), TCs (4), LAs (2), SAs (19) and other contaminants (40) | Royal jelly | QuEChERS | UPLC–QTOF– MS | 70.2–120.1 | 1.8–9.9 | 0.06–6.0 | 0.21–20 | [76] |
| PCNs (2), APs (1) and TCs (2) | Milk | QuEChERS | LC–TOF–MS | 83.0–92.0 | 1.1–8.8 | 0.0075–1.92 | 0.025–6.39 | [78] |
| PCNs (8), MACs (5), AGs (1), Qs (7), TCs (4), SAs (6) and other contaminants (9) | Honey | TFC | UPLC– Orbitrap–MS | 68.0–121.0 | 1.0–25.0 | 0.1–50 | 5–50 | [93] |
| SAs (7) and other contaminants (6) | Shrimp | QuEChERS | LC–TOF–MS | 58.0–133.0 | 4.7–14.9 | 0.06–7 | – | [79] |
| PCNs (7), APs (2), MACs (8), AGs (15), FQs (17), TCs (5), SAs (26) and other contaminants (45) | Milk | QuEChERS or SLE or SPE | UPLC– Orbitrap–MS | 12.4–146.2 | 0.9–54.8 | ≤1.0 | ≤ 5.0 | [120] |
| SAs (7) | Eggs | SPE | LC–MS/MS | 73.8–96.2 | 2.9–8.3 | 1.4–2.8 | 4.7–9.2 | [114] |
| PCNs (4) | Chicken tissues | LLE | UPLC–MS/MS | 84.1–108.1 | 1.3–16.4 | 0.01–1.36 | 0.05–5.44 | [115] |
| APs (4) | Poultry eggs | ASE | LC–MS/MS | 88.3–107.0 | 1.5–3.9 | 0.04–0.5 | 0.1–1.5 | [11] |
| COCs (8) | Eggs | SPE | LC–MS/MS | 71.7–102.7 | 2.6–15.3 | 0.16–0.52 | 0.81–1.73 | [61] |
| MACs (7) | Pork | SPE | LC–MS/MS | 68.6–95.5 | 0.5–7.6 | 0.2–0.5 | 0.5–2.0 | [121] |
| AGs (15) | Pig, chicken and cattle tissues, milk, and eggs | SPE | LC–MS/MS | 71.4–93.9 | 2.0–13.0 | 5 –10 | 5 –20 | [122] |
| Qs (9) | Ovine, chicken and porcine tissues, eggs, milk and fish | SPE | HPLC–FLD LC–MS/MS | 50.0–128.0 | <30.0 | – | – | [123] |
| TCs (6) | Beef | DLLME | LC–MS/MS | 80.0–105.0 | 2.0–7.0 | 2.2–3.6 | 7.4–11.5 | [98] |
| APs (3) | Fish muscle | MSPD | UPLC–MS/MS | 84.2–99.8 | 5.6–11.4 | 0.02–0.06 | 0.11–0.16 | [17] |
| MACs (5) and LAs (2) | Meat and milk | ASE | LC–MS/MS | 70.0–93.0 | 2.7–11.3 | 3–10 | 10–30 | [124] |
| AGs (1) and LAs (1) | Animal tissues | ASE | GC–NPD | 73.0–99.0 | <17.0 | 8.1–12.1 | 16.4–21.4 | [117] |
| AGs (1) and LAs (1) | Animal tissues | ASE | GC–MS | 70.0–93.0 | <21.0 | 1.9–3.1 | 4.1–5.6 | [117] |
| APs (4) | Poultry and porcine tissues | SPE | GC–MS | 78.5–105.5 | 6.4–16.8 | 0.1–0.5 | 0.25–2 | [125] |
| APs (2) | Fish tissues | SPE | GC–MS | 84.1–100.9 | 1.3–2. 7 | 1.64–9.3 | 4.9–29.4 | [126] |
| APs (1) and other contaminants (8) | Fish | QuEChERS–GPC | GC–MS | 63.5–90.2 | 3.6–15.4 | 0.3–1.0 | – | [127] |
| AGs (1) and LAs (1) | Poultry eggs | ASE–SPE | GC–MS/MS | 80.0–95.7 | 1.0–3.4 | 2.3–4.3 | 5.6–9.5 | [12] |
| COCs (1) | Chicken and pig tissues | ASE–SPE | GC–MS/MS | 77.5–96.3 | 1.6–6.6 | 1.4–1.6 | 4.8–5.2 | [119] |
| COCs (2) | Chicken tissues | ASE–SPE | GC–MS/MS | 82.0–94.3 | 1.7–5.4 | 0.8–2.5 | 2.7–8.0 | [128] |
| COCs (2) | Eggs | ASE | GC–MS/MS | 82.7–87.5 | 1.7–4.6 | 0.8–2.8 | 3.0–10.0 | [129] |
| Veterinary Drug | Class | Animal-Derived Food | Sample Preparation Method | Detection Method | Recovery (%) | RSD (%) | LOD (μg/kg or μg/L) | LOQ (μg/kg or μg/L) | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| FF and TAP | APs | Animal tissues | LLE | ic-ELISA | 80.6–105.5 | 3.5–14.1 | 0.07–0.14 | – | [130] |
| LIN | LAs | Milk and honey | LLE | ic-ELISA | 84.6–115.6 | 1.7–25.4 | 2.1 | – | [131] |
| ERY | MACs | Milk | LLE | ic-ELISA | 76.9–85.7 | 5.1–11.3 | 0.3 | – | [132] |
| SAL | COCs | Chicken tissues | LLE | ic-ELISA | 85.7–99.3 | 1.6–6.6 | 18–22 | – | [133] |
| 20 SAs | SAs | Animal tissues | LLE | ic-ELISA | 70.6–121.0 | 0.8–24.1 | 1.5–22.3 | – | [134] |
| 4 TCs | TCs | Animal meat and milk | LLE | ELISA | 71.9–100.0 | <10.0 | 3.7–9 | 9–27 | [135] |
| 13 FQs and 22 SAs | FQs and SAs | Milk | MSPD | DC-ELISA | 67.0–105.0 | 4.8–16.4 | 2.4–5.8 | – | [136] |
| NEO | AGs | Animal tissues, eggs and milk | LLE | ELISA | 65.8–122.8 | 5.9–28.6 | 5.7–29.3 | 11.4–59.7 | [137] |
| 6 PCNs and 4 TCs | PCNs and TCs | Milk | LLE | ic-ELISA | 80.8–99.4 | 3.0–12.7 | 0.4–3.7 | – | [138] |
| KAN and STR | AGs | Milk | LLE | dot-ELISA | 84.2–124.5 | 4.5–12.4 | 0.09–1.37 | 0.38–38.66 | [139] |
| 4 SAs | SAs | Milk | MSPE | CE–UVD | 62.7–104.8 | 3.9–10.2 | 0.89–2.31 | – | [143] |
| SDZ, SMR and SMZ | SAs | Milk | SPME | CE–LIF | 91.1–94.6 | 0.9–1.1 | 0.25‒0.47 | 0.78‒1.54 | [144] |
| AZI, TIL, ACE and ROX | MACs | Egg | LLE | CE–ECL | 89.3–107.5 | 1.3–5.6 | 1.3‒70 nmol/L | 93–2100 nmol/L | [145] |
| 7 Qs | Qs | Milk | MSPE | CE–DAD | 74.0–98.0 | 1.0–9.9 | 9‒12 | – | [146] |
| 8 Qs | Qs | Milk | MISPE | CE–MS/MS | 70.0–102.3 | 3.0–12.0 | 1.0–1.4 | 3.2–4.7 | [147] |
| 9 AGs | AGs | Honey | MISPE | CE–MS/MS | 88.2–99.8 | 2.4–6.8 | 0.4–28.5 | 1.4–94.8 | [148] |
| SDD, SDZ and STZ | SAs | Milk, pork and chicken meat | SPE | CE–CL | 79.5–112.4 | 2.1–2.8 | 0.65–3.14 | – | [149] |
| 6 SAs | SAs | Milk, pork and egg | LLE | PAEKI–CZE | 89.0–113.0 | 1.6–8.4 | 1.8–63.8 | 6.1–182.6 | [150] |
| PCN | PCNs | Pork | LLE | CE–IMERs | 96.3–110.8 | 1.5–3.1 | – | – | [151] |
| CT, DT, OT and TC | TCs | Milk | SPE | LVSS-CE | – | 1.7–9.7 | 18.6–23.83 | – | [152] |
| 4 Qs and 3 SAs | Qs and SAs | Aquatic product | ASE | LVSS-CE | 84.3–95.7 | 1.1–4.7 | 13–35 | 40–100 | [153] |
| OT | TCs | Milk | SPME | CE–DAD | 89.9 | 2.25 | 70 | – | [154] |
| PCN G and PCN acid | PCNs | Milk | LLE | CZE | 89.2–96.8 | 3.1–7.3 | 10–500 | 40–1700 | [155] |
| 8 TCs and 7 Qs | TCs and Qs | Milk | LLE | CZE–QTOF-MS | 72.6–105.8 | 2.1–10.5 | 0.5–2.9 | 1.6–9.7 | [156] |
| TIL and TYL | MACs | Chicken fat | RUSAEME | CE–DAD | 73.0–117.0 | 0.7–12.4 | 5.2–18.9 | 17.4–55.0 | [157] |
| LIN and CLI | LAs | Poultry tissues | SPE | MEKC–UVD | 97.5–109.5 | 3.9–11.7 | 13.2–18.5 | 44.2–61.5 | [160] |
| 5 COCs | COCs | Chicken Tissues | LLE | MEKC–DAD | 97.0–99.4 | 0.8–1.8 | 65–172 | 183–493 | [161] |
| CIP, ENR, CAP and FF | FQs and APs | Milk | SPE | MEKC–DAD | 80.0–109.0 | 0.1–4.8 | 6.8–13.9 | – | [162] |
| 7 SAs and 3 APs | SAs and APs | Poultry tissues | SPE | MEKC–UVD | 86.4–109.4 | 3.1–14.9 | 1.3–7.8 | 4.5–26.1 | [163] |
| 6 PCNs | PCNs | Milk and egg | LLE | LVSS-MEKC–UVD | 79.3–103.6 | 2.0–5.2 | 0.16–0.26 | 2 | [164] |
| Veterinary Drug | Class | Animal-Derived Food | Detection Method | Recovery (%) | LOD (μM or μg/L) | Ref. |
|---|---|---|---|---|---|---|
| CAP | APs | Milk | Electrochemical sensor | 102.4–103.5 | 1 | [172] |
| TC | TCs | Honey | Electrochemical aptasensor | 94.0–95.0 | 3.7 × 10−11 | [173] |
| AMP | PCNs | Milk | Electrochemical biosensor | 95.0–98.1 | 1.0×10−3 | [174] |
| PCN | PCNs | Milk | Electrochemical aptasensor | 96.0–105.4 | 0.057 | [175] |
| PCN and TC | PCNs and TCs | Chicken and beef | Electrochemical biosensor | – | 10.5–15.2 | [176] |
| STZ | SAs | Honey | Piezoelectric immunosensor | 100.0–113.0 | 0.1 μg/kg | [166] |
| CAP, SDZ and NEO | APs, SAs and AGs | Milk | Optical fiber-mediated immunosensor | 85.0−109.4 | 0.00286–30 | [177] |
| CAP | APs | Porcine muscle, honey, milk and prawn | MIP biosensor | 87.0−103.0 | 7 × 10−5 | [178] |
| CAP | APs | Milk | MIP biosensor | 96.0−105.0 | 3×10−7 | [179] |
| NEO and KAN | AGs | Dairy products | Ellipsometric sensor | 96.8−106.3 | 0.048–0.22 | [180] |
| PCN | PCNs | Milk | Single layer and bilayer potentiometric biosensors | 102.0−124.0 | 0.3 | [181] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, B.; Xie, K.; Lee, K. Veterinary Drug Residues in Animal-Derived Foods: Sample Preparation and Analytical Methods. Foods 2021, 10, 555. https://doi.org/10.3390/foods10030555
Wang B, Xie K, Lee K. Veterinary Drug Residues in Animal-Derived Foods: Sample Preparation and Analytical Methods. Foods. 2021; 10(3):555. https://doi.org/10.3390/foods10030555
Chicago/Turabian StyleWang, Bo, Kaizhou Xie, and Kiho Lee. 2021. "Veterinary Drug Residues in Animal-Derived Foods: Sample Preparation and Analytical Methods" Foods 10, no. 3: 555. https://doi.org/10.3390/foods10030555
APA StyleWang, B., Xie, K., & Lee, K. (2021). Veterinary Drug Residues in Animal-Derived Foods: Sample Preparation and Analytical Methods. Foods, 10(3), 555. https://doi.org/10.3390/foods10030555