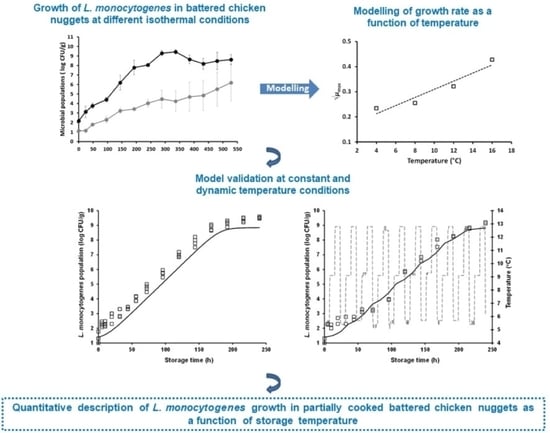
Growth of Listeria monocytogenes in Partially Cooked Battered Chicken Nuggets as a Function of Storage Temperature
Abstract
1. Introduction
2. Materials and Methods
2.1. Listeria monocytogenes Strains and Inoculum Preparation
2.2. Inoculation of Chicken Nuggets and Storage Conditions
2.3. Microbiological Analyses
2.4. Modelling of L. monocytogenes Growth
3. Results and Discussion
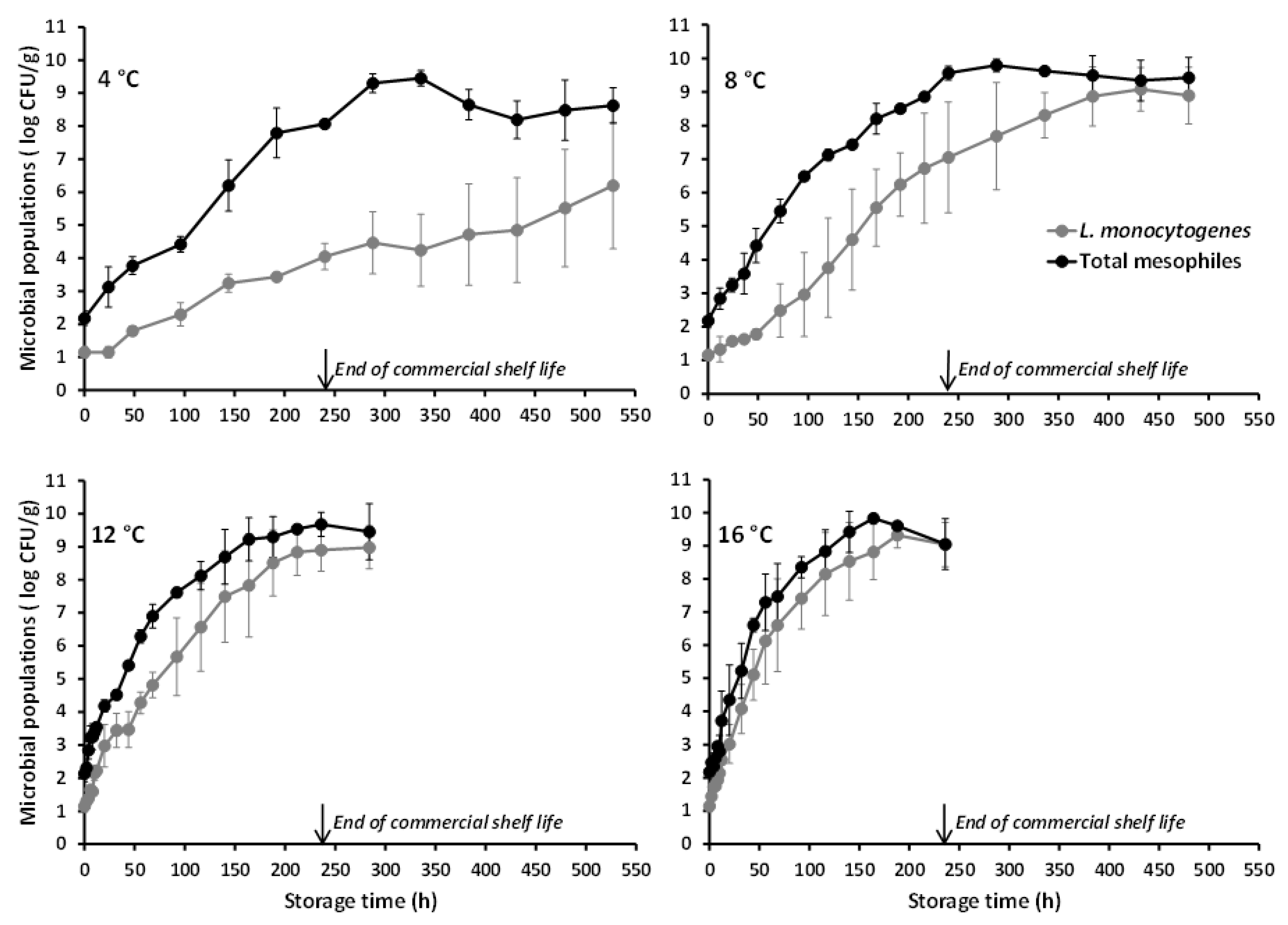
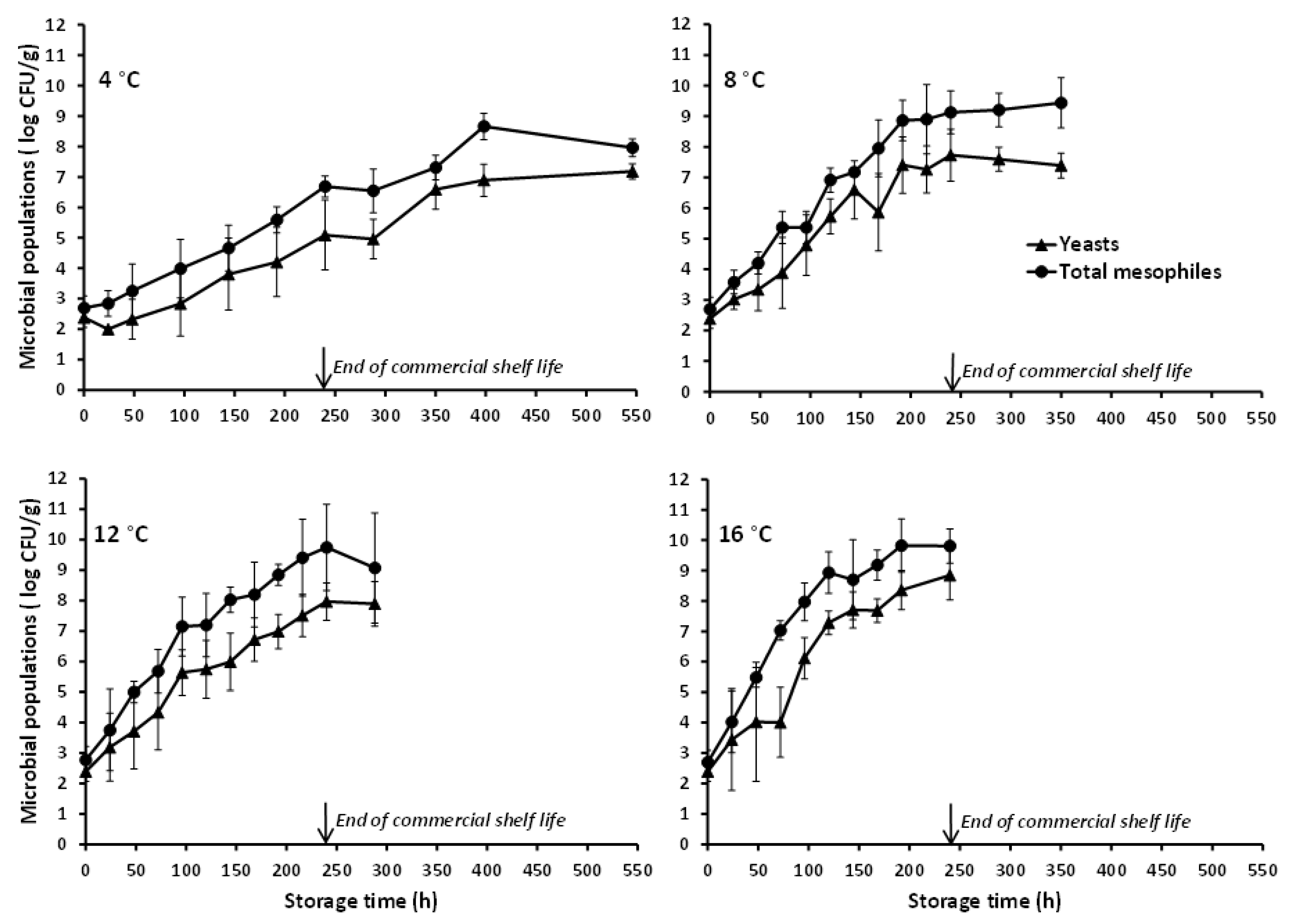
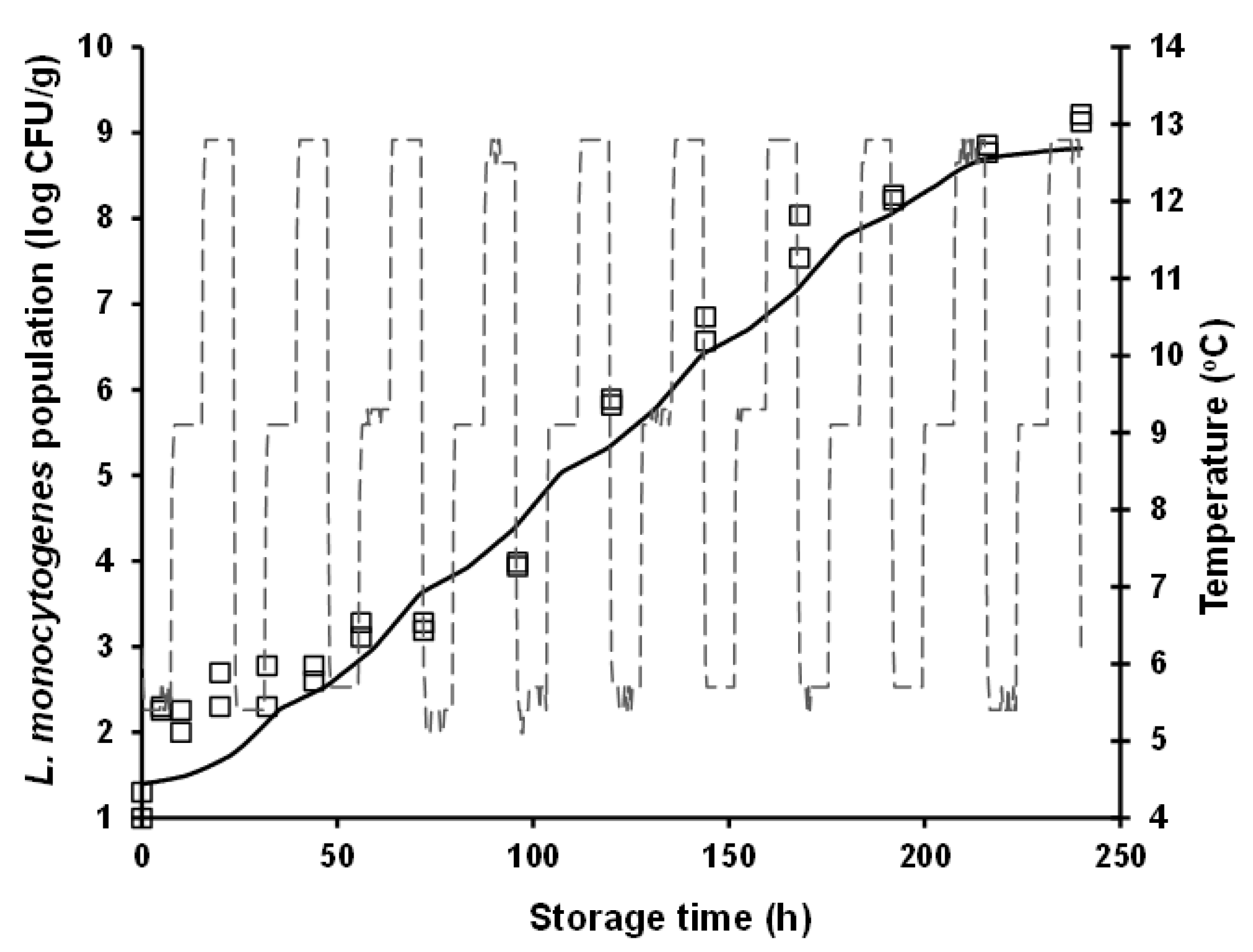
3.1. Microbial Growth Data
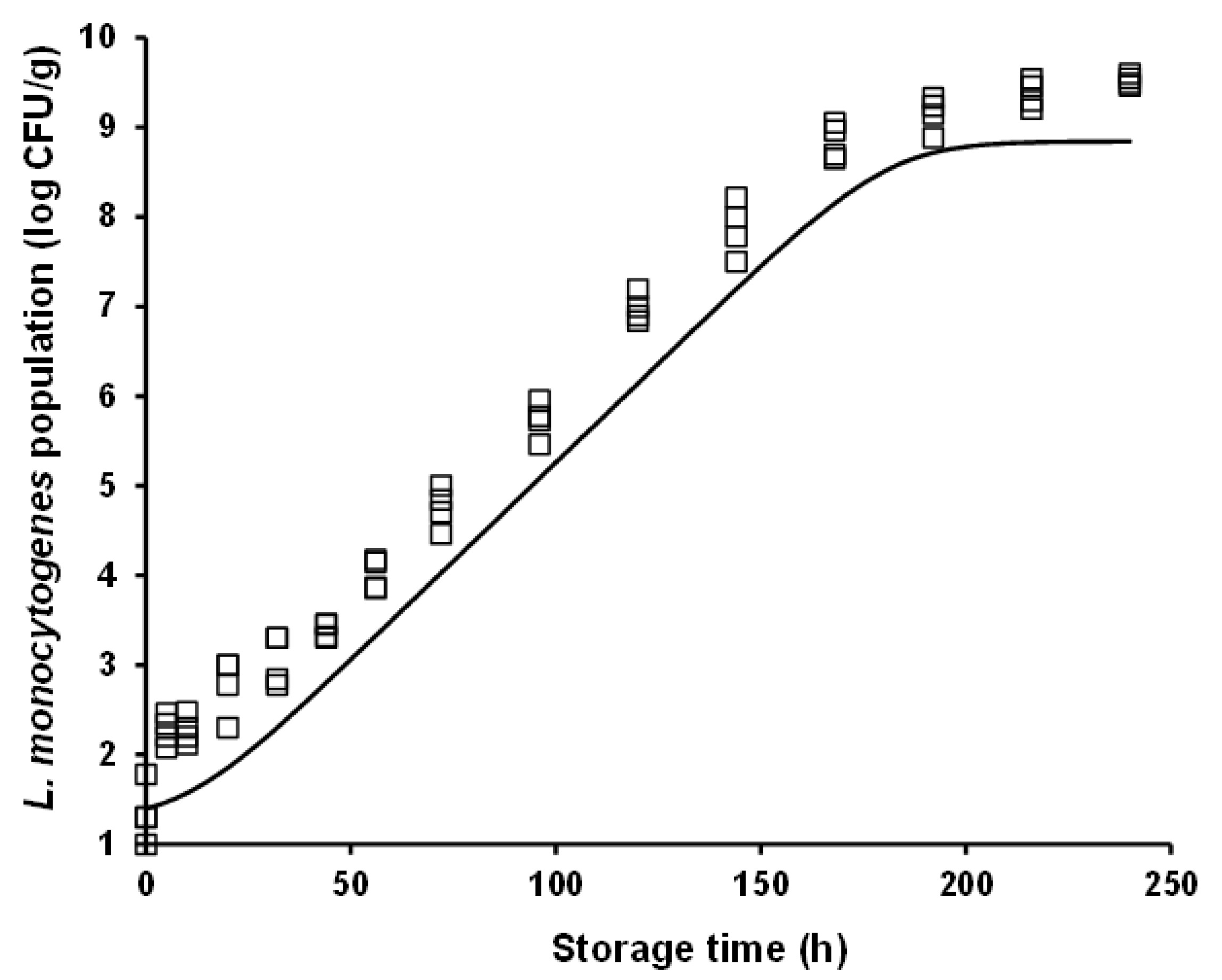
3.2. Modelling of L. monocytogenes Growth Rate
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- European Food Safety Authority-European Centre for Disease Prevention and Control (EFSA-ECDC). The European Union One Health 2018 Zoonoses Report. EFSA J. 2019, 17, e05926. [Google Scholar] [CrossRef]
- Ross, T.; Rasmussen, S.; Fazil, A.; Paoli, G.; Sumner, J. Quantitative risk assessment of Listeria monocytogenes in ready-to-eat meats in Australia. Int. J. Food Microbiol. 2009, 131, 128–137. [Google Scholar] [CrossRef]
- United States Department of Agriculture-Food Safety and Inspection Service (USDA-FSIS). FSIS Comparative Risk Assessment for Listeria monocytogenes in Ready-to-Eat Meat and Poultry Deli Meats Report. 2010. Available online: https://www.fsis.usda.gov/shared/PDF/Comparative_RA_Lm_Report_May2010.pdf (accessed on 28 January 2021).
- Alonso-Hernando, A.; Prieto, M.; García-Fernández, C.; Alonso-Calleja, C.; Capita, R. Increase over time in the prevalence of multiple antibiotic resistance among isolates of Listeria monocytogenes from poultry in Spain. Food Control 2012, 23, 37–41. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). Analysis of the baseline survey on the prevalence of Listeria monocytogenes in certain ready-to-eat foods in the EU, 2010–2011 Part A: Listeria monocytogenes prevalence estimates. EFSA J. 2013, 11, 3241. [Google Scholar] [CrossRef]
- Wang, G.; Qian, W.; Zhang, X.; Wang, H.; Ye, K.; Bai, Y.; Zhou, G. Prevalence, genetic diversity and antimicrobial resistance of Listeria monocytogenes isolated from ready-to-eat meat products in Nanjing, China. Food Control 2015, 50, 202–208. [Google Scholar] [CrossRef]
- Kurpas, M.; Wieczorek, K.; Osek, J. Ready-to-eat meat products as a source of Listeria monocytogenes. J. Vet. Res. 2018, 61, 49–55. [Google Scholar] [CrossRef]
- Liu, Y.; Sun, W.; Sun, T.; Gorris, L.G.M.; Wang, X.; Liu, B.; Dong, Q. The prevalence of Listeria monocytogenes in meat products in China: A systematic literature review and novel meta-analysis approach. Int. J. Food Microbiol. 2020, 312, 108358. [Google Scholar] [CrossRef] [PubMed]
- Lianou, A.; Geornaras, I.; Kendall, P.A.; Scanga, J.A.; Sofos, J.N. Behavior of Listeria monocytogenes at 7 °C in commercial turkey breast, with or without antimicrobials, after simulated contamination for manufacturing, retail and consumer settings. Food Microbiol. 2007, 24, 433–443. [Google Scholar] [CrossRef] [PubMed]
- Uyttendaele, M.; Busschaert, P.; Valero, A.; Geeraerd, A.H.; Vermeulen, A.; Jacxsens, L.; Goh, K.K.; De Loy, A.; Van Impe, J.F.; Devlieghere, F. Prevalence and challenge tests of Listeria monocytogenes in Belgian produced and retailed mayonnaise-based deli-salads, cooked meat products and smoked fish between 2005 and 2007. Int. J. Food Microbiol. 2009, 133, 94–104. [Google Scholar] [CrossRef]
- Mejlholm, O.; Gunvig, A.; Borggaard, C.; Blom-Hanssen, J.; Mellefont, L.; Ross, T.; Leroi, F.; Else, T.; Visser, D.; Dalgaard, P. Predicting growth rates and growth boundary of Listeria monocytogenes—An international validation study with focus on processed and ready-to-eat meat and seafood. Int. J. Food Microbiol. 2010, 141, 137–150. [Google Scholar] [CrossRef]
- Khalid, T.; Hdaifeh, A.; Federighi, M.; Cummins, E.; Boué, G.; Guillou, S.; Tesson, V. Review of quantitative microbial risk assessment in poultry meat: The central position of consumer behavior. Foods 2020, 9, 1661. [Google Scholar] [CrossRef]
- Foerster, C.; Figueroa, G.; Evers, E. Risk assessment of Listeria monocytogenes in poultry and beef. Br. Food J. 2015, 117, 779–792. [Google Scholar] [CrossRef]
- Buchanan, R.L.; Gorris, L.G.M.; Hayman, M.M.; Jackson, T.C.; Whiting, R.C. A review of Listeria monocytogenes: An update on outbreaks, virulence, dose-response, ecology, and risk assessments. Food Control 2017, 75, 1–13. [Google Scholar] [CrossRef]
- Rothrock, M.J., Jr.; Davis, M.L.; Locatelli, A.; Bodie, A.; McIntosh, T.G.; Donaldson, J.R.; Ricke, S.C. Listeria occurrence in poultry flocks: Detection and potential implications. Front. Vet. Sci. 2017, 4, 125. [Google Scholar] [CrossRef] [PubMed]
- Baquero, F.; Lanza, V.F.; Duval, M.; Coque, T.M. Ecogenetics of antibiotic resistance in Listeria monocytogenes. Mol. Microbiol. 2020, 113, 570–579. [Google Scholar] [CrossRef]
- Iannetti, L.; Schirone, M.; Neri, D.; Visciano, P.; Acciari, V.A.; Centorotola, G.; Mangieri, M.S.; Torresi, M.; Santarelli, G.A.; Di Marzio, V.; et al. Listeria monocytogenes in poultry: Detection and strain characterization along an integrated production chain in Italy. Food Microbiol. 2020, 91, 103533. [Google Scholar] [CrossRef]
- Jamshidi, A.; Zeinali, T. Significance and characteristics of Listeria monocytogenes in poultry products. Int. J. Food Sci. 2019, 7835253. [Google Scholar] [CrossRef]
- Lytou, A.E.; Renieri, C.T.; Doulgeraki, A.I.; Nychas, G.-J.E.; Panagou, E.Z. Assessment of the microbiological quality and safety of marinated chicken products from Greek retail outlets. Int. J. Food Microbiol. 2020, 320, 108506. [Google Scholar] [CrossRef]
- Lundén, J.M.; Autio, T.J.; Sjöberg, A.-M.; Korkeala, H.J. Persistent and nonpersistent Listeria monocytogenes contamination in meat and poultry processing plants. J. Food Prot. 2003, 66, 2062–2069. [Google Scholar] [CrossRef]
- Moura, G.F.; Tomborelli, P.M.; Carvalho, R.C.T.; Sigarini, C.O.; Carvalho, F.T.; Vieira, B.S.; Figueiredo, E.E.S. Listeria monocytogenes and other species as persistent contaminants in the processing of chicken meat. J. Appl. Poult. Res. 2019, 28, 470–478. [Google Scholar] [CrossRef]
- DeNoon, D.J. Listeria Forces Recalls of Ready-to-Eat Chicken Meat. 2011. Available online: https://www.medicinenet.com/script/main/art.asp?articlekey=147524 (accessed on 28 January 2021).
- United States Department of Agriculture-Food Safety and Inspection Service (USDA-FSIS). New Jersey Firm Recalls Breaded Chicken Product for Possible Listeria contamination. 2014. Available online: https://www.fsis.usda.gov/wps/portal/fsis/topics/recalls-and-public-health-alerts/recall-case-archive/archive/2014/recall-055-2014-release (accessed on 28 January 2021).
- Murphy, R.Y.; Duncan, L.K.; Johnson, E.R.; Davis, M.D.; Marcy, J.A. Thermal inactivation of Salmonella Senftenberg and Listeria innocua in beef/turkey blended patties cooked via fryer and/or air convection oven. J. Food Sci. 2002, 67, 1879–1885. [Google Scholar] [CrossRef]
- Line, J.E.; Harrison, M.A. Listeria monocytogenes inactivation in turkey rolls and battered chicken nuggets subjected to simulated commercial cooking. J. Food Sci. 1992, 57, 787–793. [Google Scholar] [CrossRef]
- Osaili, T.M.; Griffis, C.L.; Martin, E.M.; Beard, B.L.; Keener, A.E.; Marcy, J.A. Thermal inactivation of Escherichia coli O157:H7, Salmonella, and Listeria monocytogenes in breaded pork patties. J. Food Sci. 2007, 72, M56–M61. [Google Scholar] [CrossRef] [PubMed]
- McLauchlin, J.; Aird, H.; Amar, C.; Barker, C.; Dallman, T.; Elviss, N.; Jørgensen, F.; Willis, C. Listeria monocytogenes in cooked chicken: Detection of an outbreak in the United Kingdom (2016 to 2017) and analysis of L. monocytogenes from unrelated monitoring of foods (2013 to 2017). J. Food Prot. 2020, 83, 2041–2052. [Google Scholar] [CrossRef]
- Kenny, B.; Hall, R.; Cameron, S. Consumer attitudes and behaviours-key risk factors in an outbreak of Salmonella Typhimurium phage type 12 infection sourced to chicken nuggets. Aust. N. Z. J. Public Health 1999, 23, 164–167. [Google Scholar] [CrossRef]
- MacDougall, L.; Fyfe, M.; McIntyre, L.; Paccagnella, A.; Cordner, K.; Kerr, A.; Aaramini, J. Frozen chicken nuggets and strips—A Newly identified risk factor for Salmonella Heidelberg infection in British Columbia, Canada. J. Food Prot. 2004, 67, 1111–1115. [Google Scholar] [CrossRef] [PubMed]
- Currie, A.; MacDougall, L.; Aramini, J.; Gaulin, C.; Ahmed, R.; Isaacs, S. Frozen chicken nuggets and strips and eggs are leading risk factors for Salmonella Heidelberg infections in Canada. Epidemiol. Infect. 2005, 133, 809–816. [Google Scholar] [CrossRef]
- Smith, K.E.; Medus, C.; Meyer, S.D.; Boxrud, D.J.; Leano, F.; Hedberg, C.W.; Elfering, K.; Braymen, C.; Bender, J.B.; Danila, R.N. Outbreaks of salmonellosis in Minnesota (1998 through 2006) associated with frozen, microwaveable, breaded, stuffed chicken products. J. Food Prot. 2008, 71, 2153–2160. [Google Scholar] [CrossRef] [PubMed]
- Murray, K.; Tremblay, C.; Rghei, A.; Warriner, K. Challenges and options for enhancing Salmonella control in partially cooked breaded poultry products. Curr. Opin. Food Sci. 2018, 20, 44–50. [Google Scholar] [CrossRef]
- Roccato, A.; Uyttendaele, M.; Cibin, V.; Barrucci, F.; Cappa, C.; Zavagnin, P.; Longo, A.; Ricci, A. Survival of Salmonella Typhimurium in poultry-based meat preparations during grilling, frying and baking. Int. J. Food Microbiol. 2015, 197, 1–8. [Google Scholar] [CrossRef]
- Koutsoumanis, K.; Pavlis, K.; Nychas, G.-J.E.; Xanthiakos, K. Probabilistic model for Listeria monocytogenes growth during distribution, retail storage, and domestic storage of pasteurized milk. Appl. Environ. Microbiol. 2010, 76, 2181–2191. [Google Scholar] [CrossRef] [PubMed]
- Mercier, S.; Villeneuve, S.; Mondor, M.; Uysal, I. Time–temperature management along the food cold chain: A review of recent developments. Comp. Rev. Food Sci. Food Saf. 2017, 16, 647–667. [Google Scholar] [CrossRef] [PubMed]
- Dominguez, S.A.; Schaffner, D.W. Survival of Salmonella in processed chicken products during frozen storage. J. Food Prot. 2009, 72, 2088–2092. [Google Scholar] [CrossRef] [PubMed]
- Moschonas, G.; Geornaras, I.; Stopforth, J.D.; Woerner, D.R.; Belk, K.E.; Smith, G.C.; Sofos, J.N. Effect of product dimensions and surface browning method on Salmonella contamination in frozen, surface-browned, breaded chicken products treated with antimicrobials. J. Food Sci. 2015, 80, M2815–M2821. [Google Scholar] [CrossRef] [PubMed]
- Marshall, D.L.; Liese-Lehigh, P.L.; Wells, J.H.; Farr, A.J. Comparative growth of Listeria monocytogenes and Pseudomonas fluorescens on precooked chicken nuggets stored under modified atmospheres. J. Food Prot. 1991, 54, 841–843. [Google Scholar] [CrossRef]
- Thayer, D.W.; Boyd, G.; Kim, A.; Fox, J.B., Jr.; Farrell, H.M., Jr. Fate of gamma-irradiated Listeria monocytogenes during refrigerated storage on raw or cooked turkey breast meat. J. Food Prot. 1998, 61, 979–987. [Google Scholar] [CrossRef] [PubMed]
- Chorianopoulos, N.G.; Giaouris, E.D.; Skandamis, P.N.; Haroutounian, S.A.; Nychas, G.-J.E. Disinfectant test against monoculture and mixed-culture biofilms composed of technological, spoilage and pathogenic bacteria: Bactericidal effect of essential oil and hydrosol of Satureja thymbra and comparison with standard acid-base sanitizers. J. Appl. Microbiol. 2008, 104, 1586–1596. [Google Scholar] [CrossRef] [PubMed]
- Cocolin, L.; Stella, S.; Nappi, R.; Bozzetta, E.; Cantoni, C.; Comi, G. Analysis of PCR-based methods for characterization of Listeria monocytogenes strains isolated from different sources. Int. J. Food Microbiol. 2005, 103, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Ordóñez, A.; Leong, D.; Hickey, B.; Beaufort, A.; Jordan, K. The challenge of challenge testing to monitor Listeria monocytogenes growth on ready-to-eat foods in Europe by following the European Commission (2014) Technical Guidance document. Food Res. Int. 2015, 75, 233–243. [Google Scholar] [CrossRef]
- NACMCF (National Advisory Committee on Microbiological Criteria for Foods). Parameters for determining inoculated pack/challenge study protocols. J. Food Prot. 2010, 140–202. [Google Scholar] [CrossRef]
- Scott, V.N.; Swanson, K.M.J.; Freier, T.A.; Pruett, W.P., Jr.; Sveum, W.H.; Hall, P.A.; Smoot, L.A.; Brown, D.G. Guidelines for conducting Listeria monocytogenes challenge testing of foods. Food Prot. Trends 2005, 25, 818–825. [Google Scholar]
- Lianou, A.; Moschonas, G.; Nychas, G.-J.E.; Panagou, E.Z. Growth of Listeria monocytogenes in pasteurized vanilla cream pudding as affected by storage temperature and the presence of cinnamon extract. Food Res. Int. 2018, 106, 1114–1122. [Google Scholar] [CrossRef]
- Baranyi, J.; Roberts, T.A. A dynamic approach to predicting bacterial growth in food. Int. J. Food Microbiol. 1994, 23, 277–294. [Google Scholar] [CrossRef]
- Ratkowsky, D.A.; Olley, J.; McMeekin, T.A.; Ball, A. Relationship between temperature and growth rate of bacterial cultures. J. Bacteriol. 1982, 149, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Baranyi, J.; Roberts, T.A.; McClure, P. A non-autonomous differential equation to model bacterial growth. Food Microbiol. 1993, 10, 43–59. [Google Scholar] [CrossRef]
- Xanthiakos, K.; Simos, D.; Angelidis, A.S.; Nychas, G.-J.E.; Koutsoumanis, K. Dynamic modeling of Listeria monocytogenes growth in pasteurised milk. J. Appl. Microbiol. 2006, 100, 1289–1298. [Google Scholar] [CrossRef]
- Ross, T. Indices for performance evaluation of predictive models in food microbiology. J. Appl. Bacteriol. 1996, 81, 501–508. [Google Scholar] [CrossRef] [PubMed]
- Pouillot, R.; Hoelzer, K.; Chen, Y.; Dennis, S.B. Listeria monocytogenes dose response revisited—Incorporating adjustments for variability in strain virulence and host susceptibility. Risk Anal. 2015, 35, 90–108. [Google Scholar] [CrossRef]
- Rouger, A.; Tresse, O.; Zagorec, M. Bacterial contaminants of poultry meat: Sources, species, and dynamics. Microorganisms 2017, 5, 50. [Google Scholar] [CrossRef] [PubMed]
- Ismail, S.A.S.; Deak, T.; El-Rahman, H.A.A.; Yassien, M.A.M.; Beuchat, L.R. Presence and changes in populations of yeasts on raw and processed poultry products stored at refrigeration temperature. Int. J. Food Microbiol. 2000, 62, 113–121. [Google Scholar] [CrossRef]
- Khanipour, A.A.; Jorjani, S.; Soltani, M. Chemical, sensory and microbial quality changes of breaded kilka (Clupeonella cultriventris) with tempura batter in production stage and during frozen storage. Int. Food Res. J. 2014, 21, 2421–2430. [Google Scholar]
- Tsigarida, E.; Skandamis, P.; Nychas, G.-J.E. Behaviour of Listeria monocytogenes and autochthonous flora on meat stored under aerobic, vacuum and modified atmosphere packaging conditions with or without the presence of oregano essential oil at 5 °C. J. Appl. Microbiol. 2000, 89, 901–909. [Google Scholar] [CrossRef]
- Ross, T.; Dalgaard, P. Secondary models. In Modelling Microbial Responses in Food; McKellar, R.B., Lu, X., Eds.; CRC Press: Boca Raton, FL, USA, 2004; pp. 63–150. [Google Scholar]
- Gnanou Besse, N.; Audinet, N.; Barre, L.; Cauquil, A.; Cornu, M.; Colin, P. Effect of the inoculum size on Listeria monocytogenes growth in structured media. Int. J. Food Microbiol. 2006, 110, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Dupont, C.; Augustin, J.-C. Influence of stress on single-cell lag time and growth probability for Listeria monocytogenes in half Fraser broth. Appl. Environ. Microbiol. 2009, 75, 3069–3076. [Google Scholar] [CrossRef] [PubMed]
- Koutsoumanis, K.P.; Lianou, A. Stochasticity in colonial growth dynamics of individual bacterial cells. Appl. Environ. Microbiol. 2013, 79, 2294–2301. [Google Scholar] [CrossRef] [PubMed]
- Augustin, J.-C.; Kalmokoff, M.; Ells, T.; Favret, S.; Desreumaux, J.; Brasseur, E.D.; Gnanou Besse, N. Modeling the behavior of Listeria monocytogenes during enrichment in half Fraser broth; impact of pooling and the duration of enrichment on the detection of L. monocytogenes in food. Food Microbiol. 2016, 60, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Mellefont, L.A.; McMeekin, T.A.; Ross, T. Performance evaluation of a model describing the effects of temperature, water activity, pH and lactic acid concentration on the growth of Escherichia coli. Int. J. Food Microbiol. 2003, 82, 45–58. [Google Scholar] [CrossRef]
- Nyati, H. Survival characteristics and the applicability of predictive mathematical modelling to Listeria monocytogenes growth in sous vide products. Int. J. Food Microbiol. 2000, 56, 123–132. [Google Scholar] [CrossRef]
- Augustin, J.-C.; Zuliani, V.; Cornu, M.; Guillier, L. Growth rate and growth probability of Listeria monocytogenes in dairy, meat and seafood products in suboptimal conditions. J. Appl. Microbiol. 2005, 99, 1019–1042. [Google Scholar] [CrossRef] [PubMed]
- Lerasle, M.; Guillou, S.; Simonin, H.; Anthoine, V.; Chéret, R.; Federighi, M.; Membré, J.-M. Assessment of Salmonella and Listeria monocytogenes level in ready-to-cook poultry meat: Effect of various high pressure treatments and potassium lactate concentrations. Int. J. Food Microbiol. 2014, 186, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.-R.; Park, S.Y.; Ha, S.-D. Combined effects of chlorine and thiamine dilauryl sulfate on reduction of Listeria monocytogenes in chicken breast and development of predictive growth models. Poult. Sci. 2014, 93, 1503–1510. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.-R.; Kang, I.; Oh, M.H.; Ha, S.-D. Inhibitory effect of chlorine and ultraviolet radiation on growth of Listeria monocytogenes in chicken breast and development of predictive growth models. Poult. Sci. 2014, 93, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Guillard, V.; Couvert, O.; Stahl, V.; Hanin, A.; Denis, C.; Huchet, V.; Chaix, E.; Loriot, C.; Vincelot, T.; Thuault, D. Validation of a predictive model coupling gas transfer and microbial growth in fresh food packed under modified atmosphere. Food Microbiol. 2016, 58, 43–55. [Google Scholar] [CrossRef] [PubMed]
| Temperature (°C) | Growth Kinetic Parameter 1 | Adjusted R2 (min–max) 2 | ||
|---|---|---|---|---|
| λ (h) | μmax (h−1) | yend (log CFU/g) | ||
| 4 | 21.80 ± 5.59 | 0.055 ± 0.005 | − 3 | 0.962–0.985 |
| 8 | 17.56 ± 1.26 | 0.065 ± 0.020 | 8.93 ± 0.55 | 0.974–0.992 |
| 12 | – 3 | 0.103 ± 0.130 | 8.95 ± 0.56 | 0.981–0.992 |
| 16 | – 3 | 0.183 ± 0.055 | 8.65 ± 0.82 | 0.979–0.990 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lianou, A.; Raftopoulou, O.; Spyrelli, E.; Nychas, G.-J.E. Growth of Listeria monocytogenes in Partially Cooked Battered Chicken Nuggets as a Function of Storage Temperature. Foods 2021, 10, 533. https://doi.org/10.3390/foods10030533
Lianou A, Raftopoulou O, Spyrelli E, Nychas G-JE. Growth of Listeria monocytogenes in Partially Cooked Battered Chicken Nuggets as a Function of Storage Temperature. Foods. 2021; 10(3):533. https://doi.org/10.3390/foods10030533
Chicago/Turabian StyleLianou, Alexandra, Ourania Raftopoulou, Evgenia Spyrelli, and George-John E. Nychas. 2021. "Growth of Listeria monocytogenes in Partially Cooked Battered Chicken Nuggets as a Function of Storage Temperature" Foods 10, no. 3: 533. https://doi.org/10.3390/foods10030533
APA StyleLianou, A., Raftopoulou, O., Spyrelli, E., & Nychas, G.-J. E. (2021). Growth of Listeria monocytogenes in Partially Cooked Battered Chicken Nuggets as a Function of Storage Temperature. Foods, 10(3), 533. https://doi.org/10.3390/foods10030533