A Critical Review of Disinfection Processes to Control SARS-CoV-2 Transmission in the Food Industry
Abstract
1. Introduction
2. Disinfectants, Concentrations and Contact Times to Inactivate SARS-CoV-2 in the Food Industry
2.1. SARS-CoV-2 Stability on Surfaces
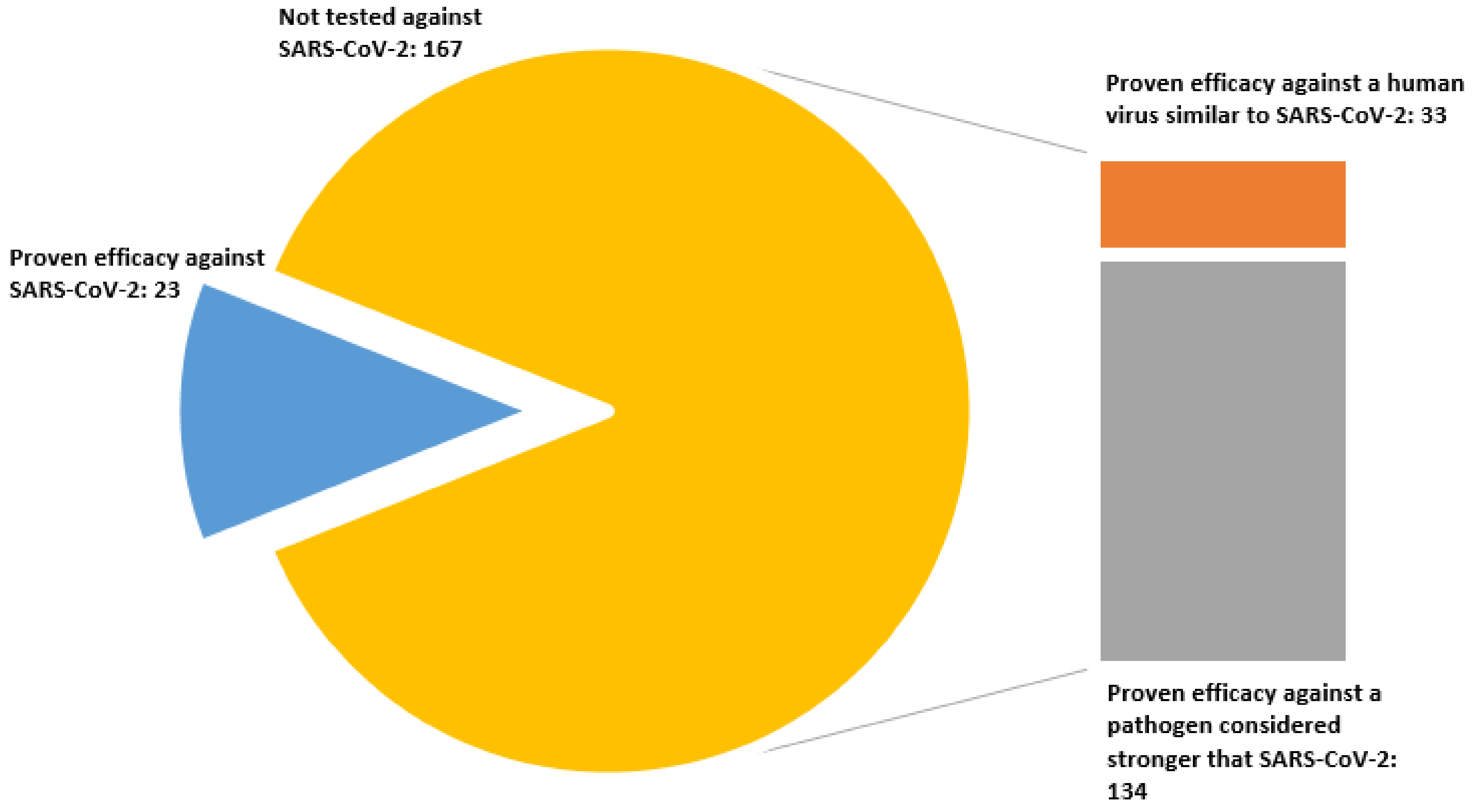
2.2. Approved Substances for SARS-CoV-2 Disinfection
- (a)
- Demonstrate efficacy against SARS-CoV-2.
- (b)
- Demonstrate efficacy against a pathogen that is harder to inactivate than SARS-CoV-2.
- (c)
- Demonstrate efficacy against another type of human coronavirus similar to SARS-CoV-2.
2.3. Efficacy of Authorised Disinfectants
2.3.1. Disinfectants Tested against SARS-CoV-2 or Similar Coronaviruses
2.3.2. Disinfectants Tested against Viruses Different from Coronavirus
3. Potential of SARS-CoV-2 Disinfectants to Inactivate Other Pathogens or Spoilers in the Food Industry
4. Disinfection under the Paradigm of the “One-Health” Approach
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Codex Alimentarius. COVID-19 | CODEXALIMENTARIUS FAO-WHO. Available online: http://www.fao.org/fao-who-codexalimentarius/thematic-areas/COVID-19/en/ (accessed on 31 July 2020).
- EFSA. Coronavirus: No Evidence that Food is a Source or Transmission Route. Available online: https://www.efsa.europa.eu/en/news/coronavirus-no-evidence-food-source-or-transmission-route (accessed on 29 July 2020).
- WHO. COVID-19 and Food Safety: Guidance for Food Businesses: Interim Guidance; WHO: Geneva, Switzerland, 2020. [Google Scholar] [CrossRef]
- Boone, S.A.; Gerba, C.P. Significance of fomites in the spread of respiratory and enteric viral disease. Appl. Environ. Microbiol. 2007, 73, 1687–1696. [Google Scholar] [CrossRef]
- Otter, J.A.; Yezli, S.; Salkeld, J.A.; French, G.L. Evidence that contaminated surfaces contribute to the transmission of hospital pathogens and an overview of strategies to address contaminated surfaces in hospital settings. Am. J. Infect. Control 2013, 41, S6–S11. [Google Scholar] [CrossRef]
- Olaimat, A.N.; Shahbaz, H.M.; Fatima, N.; Munir, S.; Holley, R.A. Food Safety During and After the Era of COVID-19 Pandemic. Front. Microbiol. 2020, 11, 1854. [Google Scholar] [CrossRef]
- Ceylan, Z.; Meral, R.; Cetinkaya, T. Relevance of SARS-CoV-2 in food safety and food hygiene: Potential preventive measures, suggestions and nanotechnological approaches. VirusDisease 2020, 31, 154–160. [Google Scholar] [CrossRef]
- Zuber, S.; Brüssow, H. COVID 19: Challenges for virologists in the food industry. Microb. Biotechnol. 2020, 13, 1689–1701. [Google Scholar] [CrossRef]
- Roy, A.; Parida, S.P.; Bhatia, V. Role of disinfection and hand hygiene: A COVID-19 perspective. Int. J. Community Med. Public Health 2020, 7, 2845. [Google Scholar] [CrossRef]
- Han, J.; Zhang, X.; He, S.; Jia, P. Can the coronavirus disease be transmitted from food? A review of evidence, risks, policies and knowledge gaps. Environ. Chem. Lett. 2020. [Google Scholar] [CrossRef]
- Kramer, A.; Schwebke, I.; Kampf, G. How long do nosocomial pathogens persist on inanimate surfaces? A systematic review. BMC Infect. Dis. 2006, 6, 130. [Google Scholar] [CrossRef]
- Holah, J.T. Cleaning and disinfection practices in food processing. In Hygiene in Food Processing: Principles and Practice: Second Edition; Elsevier Inc.: New York, NY, USA, 2013; pp. 259–304. [Google Scholar] [CrossRef]
- Kakurinov, V. Food Safety Assurance Systems: Cleaning and Disinfection. In Encyclopedia of Food Safety; Elsevier: New York, NY, USA, 2014; Volume 4, pp. 211–225. [Google Scholar] [CrossRef]
- McDonnell, G.; Russell, A.D. Antiseptics and disinfectants: Activity, action, and resistance. Clin. Microbiol. Rev. 1999, 12, 147–179. [Google Scholar] [CrossRef]
- Chin, A.W.H.; Chu, J.T.S.; Perera, M.R.A.; Hui, K.P.Y.; Yen, H.L.; Chan, M.C.W.; Peiris, M.; Poon, L.L.M. Stability of SARS-CoV-2 in different environmental conditions. Lancet Microbe 2020, 1, e10. [Google Scholar] [CrossRef]
- Spaulding, E.H. Chemical disinfection and antisepsis in the hospital. J. Hosp. Res. 1972, 9, 5–31. [Google Scholar]
- McDonnell, G.; Burke, P. Disinfection: Is it time to reconsider Spaulding? J. Hosp. Infect. 2011, 78, 163–170. [Google Scholar] [CrossRef]
- Goldman, E. Exaggerated risk of transmission of COVID-19 by fomites. Lancet Infect. Dis. 2020, 20, 892–893. [Google Scholar] [CrossRef]
- Mondelli, M.U.; Colaneri, M.; Seminari, E.M.; Baldanti, F.; Bruno, R. Low risk of SARS-CoV-2 transmission by fomites in real-life conditions. Lancet Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Quevedo-León, R.; Bastías-Montes, J.M.; Espinoza-Tellez, T.; Ronceros, B.; Balic, I.; Muñoz, O. Inactivation of Coronaviruses in food industry: The use of inorganic and organic disinfectants, ozone, and UV radiation. Sci. Agropecu. 2020, 11, 257–266. [Google Scholar] [CrossRef]
- Dyal, J.W.; Grant, M.P.; Broadwater, K.; Bjork, A.; Waltenburg, M.A.; Gibbins, J.D.; Hale, C.; Silver, M.; Fischer, M.; Steinberg, J.; et al. COVID-19 among Workers in Meat and Poultry Processing Facilities—19 States, April 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 557–561. [Google Scholar] [CrossRef]
- Waltenburg, M.A.; Victoroff, T.; Rose, C.E.; Butterfield, M.; Jervis, R.H.; Fedak, K.M.; Gabel, J.A.; Feldpausch, A.; Dunne, E.M.; Austin, C.; et al. Update: COVID-19 among Workers in Meat and Poultry Processing Facilities—United States, April–May 2020. MMWR. Morb. Mortal. Wkly. Rep. 2020, 69, 887–892. [Google Scholar] [CrossRef]
- Stephenson, J. COVID-19 Outbreaks Among Food Production Workers May Intensify Pandemic’s Disproportionate Effects on People of Color. JAMA Health Forum 2020, 1, e200783. [Google Scholar] [CrossRef]
- Murray, A.K. The Novel Coronavirus COVID-19 Outbreak: Global Implications for Antimicrobial Resistance. Front. Microbiol. 2020, 11, 1020. [Google Scholar] [CrossRef]
- Duda-Chodak, A.; Lukasiewicz, M.; Ziȩć, G.; Florkiewicz, A.; Filipiak-Florkiewicz, A. Covid-19 pandemic and food: Present knowledge, risks, consumers fears and safety. Trends Food Sci. Technol. 2020. [Google Scholar] [CrossRef]
- Aboubakr, H.A.; Sharafeldin, T.A.; Goyal, S.M. Stability of SARS-CoV-2 and other coronaviruses in the environment and on common touch surfaces and the influence of climatic conditions: A review. Transbound. Emerg. Dis. 2020. [Google Scholar] [CrossRef]
- Van Doremalen, N.; Bushmaker, T.; Morris, D.H.; Holbrook, M.G.; Gamble, A.; Williamson, B.N.; Tamin, A.; Harcourt, J.L.; Thornburg, N.J.; Gerber, S.I.; et al. Aerosol and Surface Stability of SARS-CoV-2 as Compared with SARS-CoV-1. N. Engl. J. Med. 2020, 382, 1564–1567. [Google Scholar] [CrossRef]
- Duan, S.M.; Zhao, X.S.; Wen, R.F.; Huang, J.J.; Pi, G.H.; Zhang, S.X.; Han, J.; Bi, S.L.; Ruan, L.; Dong, X.P.; et al. Stability of SARS coronavirus in human specimens and environment and its sensitivity to heating and UV irradiation. Biomed. Environ. Sci. 2003, 3, 246–255. [Google Scholar]
- Warnes, S.L.; Little, Z.R.; Keevil, C.W. Human Coronavirus 229E Remains Infectious on Common Touch Surface Materials. mBio 2015, 6. [Google Scholar] [CrossRef]
- Lai, M.Y.Y.; Cheng, P.K.C.; Lim, W.W.L. Survival of Severe Acute Respiratory Syndrome Coronavirus. Clin. Infect. Dis. 2005, 41, e67–e71. [Google Scholar] [CrossRef]
- Sizun, J.; Yu, M.W.; Talbot, P.J. Survival of human coronaviruses 229E and OC43 in suspension and after drying on surfaces: A possible source of hospital-acquired infections. J. Hosp. Infect. 2000, 46, 55–60. [Google Scholar] [CrossRef]
- Skåra, T.; Rosnes, J.T. Emerging Methods and Principles in Food Contact Surface Decontamination/Prevention. In Innovation and Future Trends in Food Manufacturing and Supply Chain Technologies; Elsevier Inc.: New York, NY, USA, 2016; pp. 151–172. [Google Scholar] [CrossRef]
- Holah, J.T.; Thorpe, R.H. Cleanability in relation to bacterial retention on unused and abraded domestic sink materials. J. Appl. Bacteriol. 1990, 69, 599–608. [Google Scholar] [CrossRef]
- Verran, J. Testing surface cleanability in food processing. In Handbook of Hygiene Control in the Food Industry; Elsevier Inc.: New York, NY, USA, 2005; pp. 556–571. [Google Scholar] [CrossRef]
- Hirneisen, K.A.; Markland, S.M.; Kniel, K.E. Ozone inactivation of norovirus surrogates on fresh produce. J. Food Prot. 2011, 74, 836–839. [Google Scholar] [CrossRef]
- Mullis, L.; Saif, L.J.; Zhang, Y.; Zhang, X.; Azevedo, M.S. Stability of bovine coronavirus on lettuce surfaces under household refrigeration conditions. Food Microbiol. 2012, 30, 180–186. [Google Scholar] [CrossRef]
- Biryukov, J.; Boydston, J.A.; Dunning, R.A.; Yeager, J.J.; Wood, S.; Reese, A.L.; Ferris, A.; Miller, D.; Weaver, W.; Zeitouni, N.E.; et al. Increasing temperature and relative humidity accelerates inactivation of SARS-COV-2 on surfaces. mSphere 2020, 5. [Google Scholar] [CrossRef]
- Rabenau, H.F.; Cinatl, J.; Morgenstern, B.; Bauer, G.; Preiser, W.; Doerr, H.W. Stability and inactivation of SARS coronavirus. Med. Microbiol. Immunol. 2005, 194, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Van Doremalen, N.; Bushmaker, T.; Munster, V.J. Stability of middle east respiratory syndrome coronavirus (MERS-CoV) under different environmental conditions. Eurosurveillance 2013, 18, 20590. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.H.; Peiris, J.S.M.; Lam, S.Y.; Poon, L.L.M.; Yuen, K.Y.; Seto, W.H. The Effects of Temperature and Relative Humidity on the Viability of the SARS Coronavirus. Adv. Virol. 2011, 2011, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Codex Alimentarius Commission. Food Hygiene. Basic Texts; Food & Agriculture Organization: Rome, Italy, 2009. [Google Scholar]
- ECHA. COVID-19 Lists of Disinfectant Active Substances and Products—Datasets. Available online: https://data.europa.eu/euodp/en/data/dataset/biocidal-products-lists-of-disinfectant-active-substances-and-products (accessed on 14 October 2020).
- Ministerio de Sanidad. Viricidal Product Authorised in Spain. Available online: https://echa.europa.eu/documents/10162/29202019/spain_disinfection_products_pt1-pt2_art89_en.pdf/aba3d2dc-67d1-ddeb-dc77-2e778ef918c0 (accessed on 13 August 2020).
- Ctgb (Board for the Authorisation of Plant Protection Products and Biocides in The Netherlands). Vrijstelling Desinfectiemiddelen Oppervlakken COVID-19 2020’ | Besluit | College Voor de Toelating van Gewasbeschermingsmiddelen en Biociden. Available online: https://www.ctgb.nl/documenten/besluiten/2020/03/31/desinfectiemiddelen-oppervlakken-covid-19 (accessed on 14 October 2020).
- ECRI. Disinfectant Concentrations, Contact Times, and Use Settings for EPA’s List of Products Effective against SARS-CoV-2, the Cause of COVID-19; ECRI: Plymouth, PA, USA, 2020. [Google Scholar]
- EPA (United States Enviromental Protection Agency). List N Tool: COVID-19 Disinfectants | US EPA. Available online: https://cfpub.epa.gov/giwiz/disinfectants/index.cfm (accessed on 14 October 2020).
- Australian Government Department of Health Therapeutic Goods Administration. Disinfectants for Use against COVID-19 in the ARTG for Legal Supply in Australia. Available online: https://www.tga.gov.au/disinfectants-use-against-covid-19-artg-legal-supply-australia (accessed on 3 November 2020).
- Government of Canada. Hard-Surface Disinfectants and Hand Sanitizers (COVID-19): List of Disinfectants with Evidence for Use against COVID-19. Available online: https://www.canada.ca/en/health-canada/services/drugs-health-products/disinfectants/covid-19/list.html#tbl1 (accessed on 16 November 2020).
- Drug Product Database: Access the Database—Canada.ca. Available online: https://www.canada.ca/en/health-canada/services/drugs-health-products/drug-products/drug-product-database.html (accessed on 3 November 2020).
- Zeitler, B.; Rapp, I. Surface-dried viruses can resist glucoprotamin-based disinfection. Appl. Environ. Microbiol. 2014, 80, 7169–7175. [Google Scholar] [CrossRef]
- Kindermann, J.; Karbiener, M.; Leydold, S.M.; Knotzer, S.; Modrof, J.; Kreil, T.R. Virus disinfection for biotechnology applications: Different effectiveness on surface versus in suspension. Biologicals 2020, 64, 1–9. [Google Scholar] [CrossRef]
- Bidra, A.S.; Pelletier, J.S.; Westover, J.B.; Frank, S.; Brown, S.M.; Tessema, B. Comparison of In Vitro Inactivation of SARS CoV-2 with Hydrogen Peroxide and Povidone-Iodine Oral Antiseptic Rinses. J. Prosthodont. 2020, 29, 599–603. [Google Scholar] [CrossRef]
- Marks, M.I. Variables influencing the in vitro susceptibilities of herpes simplex viruses to antiviral drugs. Antimicrob. Agents Chemother. 1974, 6, 34–38. [Google Scholar] [CrossRef]
- Hayden, F.G.; Douglas, R.G.; Simons, R.; Vanvoris, P.; Betts, R.B.; Hayden, F.G.; Christman, W.A.; Douglas, R.G. Enhancement of Activity Against Influenza Viruses by Combinations of Antiviral Agents. Antimicrob. Agents Chemother. 1980, 18, 536–541. [Google Scholar] [CrossRef]
- Bruce Burlington, D.; Meiklejohn, G.; Mostowt, S.R. Anti-Influenza A Virus Activity of Amantadine Hydrochloride and Rimantadine Hydrochloride in Ferret Tracheal Ciliated Epithelium. Antimicrob. Agents Chemother. 1982, 21, 794–799. [Google Scholar] [CrossRef]
- Ijaz, M.K.; Whitehead, K.; Srinivasan, V.; McKinney, J.; Rubino, J.R.; Ripley, M.; Jones, C.; Nims, R.W.; Charlesworth, B. Microbicidal actives with virucidal efficacy against SARS-CoV-2. Am. J. Infect. Control 2020, 48, 972–973. [Google Scholar] [CrossRef]
- Rabenau, H.F.; Schwebke, I.; Blümel, J.; Eggers, M.; Glebe, D.; Rapp, I.; Sauerbrei, A.; Steinmann, E.; Steinmann, J.; Willkommen, H.; et al. Guideline for testing chemical disinfectants regarding their virucidal activity within the field of human medicine: As of December 1st, 2014 Prepared by the German Association for the Control of Virus Diseases (DVV) and the Robert Koch Institute (RKI). Bundesgesundheitsblatt Gesundheitsforschung Gesundheitsschutz 2020. [Google Scholar] [CrossRef] [PubMed]
- Saknimit, M.; Inatsuki, I.; Sugiyama, Y.; Yagami, K. Virucidal efficacy of physico-chemical treatments against coronaviruses and parvoviruses of laboratory animals. Jikken Dobutsu. Exp. Anim. 1988, 37, 341–345. [Google Scholar] [CrossRef]
- Wood, A.; Payne, D. The action of three antiseptics/disinfectants against enveloped and non-enveloped viruses. J. Hosp. Infect. 1998, 38, 283–295. [Google Scholar] [CrossRef]
- Sattar, S.A.; Springthorpe, V.S.; Karim, Y.; Loro, P. Chemical disinfection of non-porous inanimate surfaces experimentally contaminated with four human pathogenic viruses. Epidemiol. Infect. 1989, 102, 493–505. [Google Scholar] [CrossRef] [PubMed]
- Eterpi, M.; McDonnell, G.; Thomas, V. Virucidal Activity of Disinfectants against Parvoviruses and Reference Viruses. Appl. Biosaf. 2010, 15, 165–171. [Google Scholar] [CrossRef]
- Omidbakhsh, N.; Sattar, S.A. Broad-spectrum microbicidal activity, toxicologic assessment, and materials compatibility of a new generation of accelerated hydrogen peroxide-based environmental surface disinfectant. Am. J. Infect. Control 2006, 34, 251–257. [Google Scholar] [CrossRef]
- Goyal, S.M.; Chander, Y.; Yezli, S.; Otter, J.A. Evaluating the virucidal efficacy of hydrogen peroxide vapour. J. Hosp. Infect. 2014, 86, 255–259. [Google Scholar] [CrossRef]
- Kratzel, A.; Todt, D.; V’kovski, P.; Steiner, S.; Gultom, M.; Thao, T.T.N.; Ebert, N.; Holwerda, M.; Steinmann, J.; Niemeyer, D.; et al. Inactivation of Severe Acute Respiratory Syndrome Coronavirus 2 by WHO-Recommended Hand Rub Formulations and Alcohols. Emerg. Infect. Dis. 2020, 26, 1592–1595. [Google Scholar] [CrossRef]
- Welch, S.R.; Davies, K.A.; Buczkowski, H.; Hettiarachchi, N.; Green, N.; Arnold, U.; Jones, M.; Hannah, M.J.; Evans, R.; Burton, C.; et al. Inactivation analysis of SARS-CoV-2 by specimen transport media, nucleic acid extraction reagents, detergents and fixatives. J. Clin. Microbiol. 2020. [Google Scholar] [CrossRef]
- Rabenau, H.; Kampf, G.; Cinatl, J.; Doerr, H. Efficacy of various disinfectants against SARS coronavirus. J. Hosp. Infect. 2005, 61, 107–111. [Google Scholar] [CrossRef]
- Pratelli, A. Canine coronavirus inactivation with physical and chemical agents. Vet. J. 2008, 117, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Hulkower, R.L.; Casanova, L.M.; Rutala, W.A.; Weber, D.J.; Sobsey, M.D. Inactivation of surrogate coronaviruses on hard surfaces by health care germicides. Am. J. Infect. Control 2011, 39, 401–407. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, A.; Kaleta, E.F. Evaluation of virucidal activity of three commercial disinfectants and formic acid using bovine enterovirus type 1 (ECBO virus), mammalian orthoreovirus type 1 and bovine adenovirus type 1. Vet. J. 2003, 166, 67–78. [Google Scholar] [CrossRef]
- Heinzel, M.; Kyas, A.; Weide, M.; Breves, R.; Bockmühl, D.P. Evaluation of the virucidal performance of domestic laundry procedures. Int. J. Hyg. Environ. Health 2010, 213, 334–337. [Google Scholar] [CrossRef] [PubMed]
- Poli, G.; Biondi, P.A.; Uberti, F.; Ponti, W.; Balsari, A.; Cantoni, C. Virucidal activity of organic acids. Food Chem. 1979, 4, 251–258. [Google Scholar] [CrossRef]
- Lai, W.L.; Chuang, H.S.; Lee, M.H.; Wei, C.L.; Lin, C.F.; Tsai, Y.C. Inhibition of herpes simplex virus type 1 by thymol-related monoterpenoids. Planta Med. 2012, 78, 1636–1638. [Google Scholar] [CrossRef]
- Sharifi-Rad, J.; Salehi, B.; Baghalpour, N.; Kobarfard, F.; Sharifi-Rad, M.; Mohammadizade, M. Antiviral activity of monoterpenes thymol, carvacrol and p-cymene against herpes simplex virus in vitro. Int. Pharm. Acta 2018, 1, 73. [Google Scholar] [CrossRef]
- Codex Alimentarius Commission. Guidelines on the Application of General Principles of Food Hygiene to the Control of Viruses in Food; Codex Alimentarius Commission: Rome, Italy, 2012. [Google Scholar]
- Kampf, G. Antiseptic Stewardship: Biocide Resistance and Clinical Implications; Springer International Publishing: Cham, Switzerland, 2018; pp. 1–694. [Google Scholar] [CrossRef]
- Cosentino, S.; Tuberoso, C.I.; Pisano, B.; Satta, M.; Mascia, V.; Arzedi, E.; Palmas, F. In-vitro antimicrobial activity and chemical composition of Sardinian Thymus essential oils. Lett. Appl. Microbiol. 1999, 29, 130–135. [Google Scholar] [CrossRef]
- Walsh, S.E.; Maillard, J.Y.; Russell, A.D.; Catrenich, C.E.; Charbonneau, D.L.; Bartolo, R.G. Development of bacterial resistance to several biocides and effects on antibiotic susceptibility. J. Hosp. Infect. 2003, 55, 98–107. [Google Scholar] [CrossRef]
- Noma, Y.; Asakawa, Y. Biotransformation of monoterpenoids. In Comprehensive Natural Products II: Chemistry and Biology; Elsevier Ltd.: New York, NY, USA, 2010; Volume 3, pp. 669–801. [Google Scholar] [CrossRef]
- ECHA. Biocidal Products Committee (BPC) Opinion on the Application for Approval of the Active Substance: PHMB (1415; 4.7) Polyhexamethylene Biguanide Hydrochloride with a Mean Number-Average Molecular Weight (Mn) of 1415 and a Mean Polydispersity (PDI) of 4.7). Available online: https://echa.europa.eu/fr/addressing-chemicals-of-concern/biocidal-products- (accessed on 7 September 2020).
- Bonin, L.; Vitry, V.; Olivier, M.G.; Bertolucci-Coelho, L. Covid-19: Effect of disinfection on corrosion of surfaces. Corros. Eng. Sci. Technol. 2020, 55, 693–695. [Google Scholar] [CrossRef]
- Nabi, G.; Wang, Y.; Hao, Y.; Khan, S.; Wu, Y.; Li, D. Massive use of disinfectants against COVID-19 poses potential risks to urban wildlife. Environ. Res. 2020, 188, 109916. [Google Scholar] [CrossRef] [PubMed]
- Chang, A.; Schnall, A.H.; Law, R.; Bronstein, A.C.; Marraffa, J.M.; Spiller, H.A.; Hays, H.L.; Funk, A.R.; Mercurio-Zappala, M.; Calello, D.P.; et al. Cleaning and Disinfectant Chemical Exposures and Temporal Associations with COVID-19—National Poison Data System, United States, 1 January 2020–31 March 2020. MMWR. Morb. Mortal. Wkly. Rep. 2020, 69, 496–498. [Google Scholar] [CrossRef] [PubMed]
- Takeda, Y.; Uchiumi, H.; Matsuda, S.; Ogawa, H. Acidic electrolyzed water potently inactivates SARS-CoV-2 depending on the amount of free available chlorine contacting with the virus. Biochem. Biophys. Res. Commun. 2020, 530, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Chu, W.; Fang, C.; Deng, Y.; Xu, Z. Intensified Disinfection Amid COVID-19 Pandemic Poses Potential Risks to Water Quality and Safety. Environ. Sci. Technol. 2020. [Google Scholar] [CrossRef]
- Ölmez, H.; Kretzschmar, U. Potential alternative disinfection methods for organic fresh-cut industry for minimizing water consumption and environmental impact. LWT Food Sci. Technol. 2009, 42, 686–693. [Google Scholar] [CrossRef]
- Zhang, C.; Cui, F.; Zeng, G.M.; Jiang, M.; Yang, Z.Z.; Yu, Z.G.; Zhu, M.Y.; Shen, L.Q. Quaternary ammonium compounds (QACs): A review on occurrence, fate and toxicity in the environment. Sci. Total Environ. 2015, 518, 352–362. [Google Scholar] [CrossRef]
- Melin, V.E.; Potineni, H.; Hunt, P.; Griswold, J.; Siems, B.; Werre, S.R.; Hrubec, T.C. Exposure to common quaternary ammonium disinfectants decreases fertility in mice. Reprod. Toxicol. 2014, 50, 163–170. [Google Scholar] [CrossRef]
- WHO. Silver as a Drinking-Water Disinfectant. Available online: http://apps.who.int/bookorders (accessed on 16 September 2020).
- Mijnendonckx, K.; Leys, N.; Mahillon, J.; Silver, S.; Van Houdt, R. Antimicrobial silver: Uses, toxicity and potential for resistance. BioMetals 2013, 26, 609–621. [Google Scholar] [CrossRef]
- Condell, O.; Iversen, C.; Cooney, S.; Power, K.A.; Walsh, C.; Burgess, C.; Fanning, S. Efficacy of biocides used in the modern food industry to control Salmonella enterica, and links between biocide tolerance and resistance to clinically relevant antimicrobial compounds. Appl. Environ. Microbiol. 2012, 78, 3087–3097. [Google Scholar] [CrossRef]
- Capita, R.; Alonso-Calleja, C. Antibiotic-Resistant Bacteria: A Challenge for the Food Industry. Crit. Rev. Food Sci. Nutr. 2013, 53, 11–48. [Google Scholar] [CrossRef]
- Thorrold, C.A.; Letsoalo, M.E.; Dusé, A.G.; Marais, E. Efflux pump activity in fluoroquinolone and tetracycline resistant Salmonella and E. coli implicated in reduced susceptibility to household antimicrobial cleaning agents. Int. J. Food Microbiol. 2007, 113, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Abdel Malek, S.M.; Badran, Y.R. Pseudomonas aeruginosa PAO1 Adapted to 2-Phenoxyethanol Shows Cross-Resistance to Dissimilar Biocides and Increased Susceptibility to Antibiotics. Folia Microbiol. 2010, 55, 588–592. [Google Scholar] [CrossRef] [PubMed]
- Nomura, K.; Ogawa, M.; Miyamoto, H.; Muratani, T.; Taniguchi, H. Antibiotic susceptibility of glutaraldehyde-tolerant Mycobacterium chelonae from bronchoscope washing machines. Am. J. Infect. Control 2004, 32, 185–188. [Google Scholar] [CrossRef] [PubMed]
- Vikram, A.; Bomberger, J.M.; Bibby, K.J. Efflux as a glutaraldehyde resistance mechanism in Pseudomonas fluorescens and Pseudomonas aeruginosa biofilms. Antimicrob. Agents Chemother. 2015, 59, 3433–3440. [Google Scholar] [CrossRef]
- Percival, S.L.; Salisbury, A.M.; Chen, R. Silver, biofilms and wounds: Resistance revisited. Crit. Rev. Microbiol. 2019, 45, 223–237. [Google Scholar] [CrossRef]
- Langsrud, S.; Sundheim, G.; Borgmann-Strahsen, R. Intrinsic and acquired resistance to quaternary ammonium compounds in food-related Pseudomonas spp. J. Appl. Microbiol. 2003, 95, 874–882. [Google Scholar] [CrossRef]
- García, M.; Cabo, M. Optimization of E. coli inactivation by benzalkonium chloride reveals the importance of quantifying the inoculum effect on chemical disinfection. Front. Microbiol. 2018, 9. [Google Scholar] [CrossRef]
- Kampf, G. Adaptive microbial response to low-level benzalkonium chloride exposure. J. Hosp. Infect. 2018, 100, e1–e22. [Google Scholar] [CrossRef]
- Zou, L.; Meng, J.; McDermott, P.F.; Wang, F.; Yang, Q.; Cao, G.; Hoffmann, M.; Zhao, S. Presence of disinfectant resistance genes in Escherichia coli isolated from retail meats in the USA. J. Antimicrob. Chemother. 2014, 69, 2644–2649. [Google Scholar] [CrossRef]
- Zhang, A.; He, X.; Meng, Y.; Guo, L.; Long, M.; Yu, H.; Li, B.; Fan, L.; Liu, S.; Wang, H.; et al. Antibiotic and Disinfectant Resistance of Escherichia coli Isolated from Retail Meats in Sichuan, China. Microb. Drug Resist. 2016, 22, 80–87. [Google Scholar] [CrossRef]
- Gadea, R.; Fernández Fuentes, M.A.; Pérez Pulido, R.; Gálvez, A.; Ortega, E. Effects of exposure to quaternary-ammonium-based biocides on antimicrobial susceptibility and tolerance to physical stresses in bacteria from organic foods. Food Microbiol. 2017, 63, 58–71. [Google Scholar] [CrossRef] [PubMed]
- Deng, W.; Quan, Y.; Yang, S.; Guo, L.; Zhang, X.; Liu, S.; Chen, S.; Zhou, K.; He, L.; Li, B.; et al. Antibiotic Resistance in Salmonella from Retail Foods of Animal Origin and Its Association with Disinfectant and Heavy Metal Resistance. Microb. Drug Resist. 2018, 24, 782–791. [Google Scholar] [CrossRef] [PubMed]
- Bigliardi, P.L.; Alsagoff, S.A.L.; El-Kafrawi, H.Y.; Pyon, J.K.; Wa, C.T.C.; Villa, M.A. Povidone iodine in wound healing: A review of current concepts and practices. Int. J. Surg. 2017, 44, 260–268. [Google Scholar] [CrossRef] [PubMed]
| Virus | Surface/Fomite | Inoculum | TCDI50 | T (C) | RH (%) | Stability | Ref. |
|---|---|---|---|---|---|---|---|
| hCoV 229E | Aluminium | 10 L | 5.5 × 105 | 21 | 55–70 | >8 h | [31] |
| Brasses >70% Cu | 20 L | 1.4 × 103 (a) | 21 | 30–40 | ≤40 min | [29] | |
| Brasses ≥70% Cu | 1 L | 1.4 × 103 (a) | 21 | 30–40 | ≤5 min | [29] | |
| Ceramic tile | 20 L | 1.4 × 103 (a) | 21 | 30–40 | >120 h | [29] | |
| Copper | 1 L | 1.4 × 103 (a) | 21 | 30–40 | ≤5 min | [29] | |
| Copper-nickel ≥79% Cu | 20 L | 1.4 × 103 (a) | 21 | 30–40 | ≤50 min | [29] | |
| Copper-nickel 70% Cu | 20 L | 1.4 × 103 (a) | 21 | 30–40 | ≤2 h | [29] | |
| Cotton gauze sponges | 10 L | 5.5 × 105 (a) | 21 | 55–70 | ≤6 h | [31] | |
| Glass | 20 L | 1.4 × 103 (a) | 21 | 30–40 | >120 h | [29] | |
| Latex gloves | 10 L | 5.5 × 105 | 21 | 55–70 | ≤6 h | [31] | |
| Plastic (PVC) | 20 L | 1.4 × 103 (a) | 21 | 30–40 | >120 h | [29] | |
| Plastic (PE) | 500 L | 107 | 21–25 | ND | >72 h | [38] | |
| Plastic (PTFE) | 20 L | 1.4 × 103 (a) | 21 | 30–40 | >120 h | [29] | |
| Silicon rubber | 20 L | 1.4 × 103 (a) | 21 | 30–40 | ≤120 h | [29] | |
| Steel | 20 L | 1.4 × 103 (a) | 21 | 30–40 | >120 h | [29] | |
| hCoV OC43 | Aluminium | 10 L | 5.5 × 105 | 21 | 55–70 | ≤3 h | [31] |
| Cotton gauze sponges | 10 L | 5.5 × 105 | 21 | 55–70 | ≤1 h | [31] | |
| Latex gloves | 10 L | 5.5 × 105 | 21 | 55–70 | ≤1 h | [31] | |
| MERS-CoV EMC/2012 | Plastic (undefined) | 100 L | 106 | 20 | 40 | ≤72 h | [39] |
| Plastic (undefined) | 100 L | 106 | 30 | 30 | ≤48 h | [39] | |
| Plastic (undefined) | 100 L | 106 | 30 | 80 | ≤24 h | [39] | |
| Steel | 100 L | 106 | 20 | 40 | ≤72 h | [39] | |
| Steel | 100 L | 106 | 30 | 30 | ≤48 h | [39] | |
| Steel | 100 L | 106 | 30 | 80 | ≤24 h | [39] | |
| SARS-CoV-1 HKU39849 | Plastic (Undefined) | 10 L | 107 | 22–25 | 80 | >120 h | [40] |
| SARS-CoV-1 P9 | Ceramic tile | 300 L | 106 | 20 | ND | ≤96 h | [28] |
| Glass | 300 L | 106 | 20 | ND | ≤120 h | [28] | |
| Metal (undefined) | 300 L | 106 | 20 | ND | >120 h | [28] | |
| Paper (filter paper) | 300 L | 106 | 20 | ND | >120 h | [28] | |
| Paper (press paper) | 300 L | 106 | 20 | ND | ≤120 h | [28] | |
| Plastic (undefined) | 300 L | 106 | 20 | ND | ≤120 h | [28] | |
| Wood | 300 L | 106 | 20 | ND | ≤96 h | [28] | |
| SARS-CoV-1 GVU6109 | Cotton gown | 5 L | 104 | 37 | ND | ≤5 min | [30] |
| Cotton gown | 5 L | 105 | 37 | ND | ≤1 h | [30] | |
| Cotton gown | 5 L | 106 | 37 | ND | ≤24 h | [30] | |
| Paper (request form) | 5 L | 104 | 37 | ND | ≤5 min | [30] | |
| Paper (request form) | 5 L | 105 | 37 | ND | ≤3 h | [30] | |
| Paper (request form) | 5 L | 106 | 37 | ND | ≤24 h | [30] | |
| Plastic disposable gown | 5 L | 104 | 37 | ND | ≤1 h | [30] | |
| Plastic disposable gown | 5 L | 105 | 37 | ND | ≤24 h | [30] | |
| Plastic disposable gown | 5 L | 106 | 37 | ND | ≤48 h | [30] | |
| SARS-CoV-1 FFM1 | Plastic (PE) | 500 L | 107 | 21–25 | ND | ≤216 h | [38] |
| SARS-CoV-1 Tor2 | Capboard | 50 L | 107 | RC (c) | RC (c) | ≤8 h | [27] |
| Copper | 50 L | 107 | RC (c) | RC (c) | ≤8 h | [27] | |
| Plastic (PP) | 50 L | 107 | RC (c) | RC (c) | ≤72 h | [27] | |
| Steel | 50 L | 107 | RC (c) | RC (c) | ≤72 h | [27] | |
| SARS-CoV-2 WA1-2020 | Capboard | 50 L | 107 | RC (c) | RC (c) | ≤24 h | [27] |
| Copper | 50 L | 107 | RC (c) | RC (c) | ≤4 h | [27] | |
| Plastic (PP) | 50 L | 107 | RC (c) | RC (c) | ≤72 h | [27] | |
| Steel | 50 L | 107 | RC (c) | RC (c) | ≤72 h | [27] | |
| SARS-CoV-2 Unknow strain | Paper (tissue paper) | 5 L | 107–108 | 22 | 65 | ≤3 h | [15] |
| Paper (undefined) | 5 L | 107–108 | 22 | 65 | ≤3 h | [15] | |
| Wood | 5 L | 107–108 | 22 | 65 | ≤3 h | [15] | |
| Cloth | 5 L | 107–108 | 22 | 65 | ≤48 h | [15] | |
| Glass | 5 L | 107–108 | 22 | 65 | ≤96 h | [15] | |
| Paper (banknote) | 5 L | 107–108 | 22 | 65 | ≤96 h | [15] | |
| Steel | 5 L | 107–108 | 22 | 65 | ≤168 h | [15] | |
| Plastic (undefined) | 5 L | 107–108 | 22 | 65 | ≤168 h | [15] | |
| Mask (inner layer) | 5 L | 107–108 | 22 | 65 | ≤168 h | [15] | |
| Mask (outter layer) | 5 L | 107–108 | 22 | 65 | >168 h | [15] |
| SARS-2-CoV Status | Food Industry Status | Concentration (%) in CP | ||||
|---|---|---|---|---|---|---|
| ECHA | EPA | ECHA | EPA | EU | USA | |
| Alcohols | ||||||
| Ethanol | AP | AP | UR | AP | 65.00–75.00 | 7.50–68.61 |
| 1-propanol | AP | NE | AP | AP | 17.00–49.00 | b |
| 2-propanol | AP | AP | AP | AP | 9.99 | 12.25–63.25 |
| Aldehides | ||||||
| Glutaraldehyde | AP | AP | AP | AP | 2.50–12.00 | 7.00 |
| Glyoxal | AP | NE | AP | AP | 6.00 | b |
| Amines | ||||||
| Ampholyt 20 * | AP | NE | AP | NE | a | b |
| Glucoprotamin | AP | NE | AP | AP | a | b |
| N-(3-aminopropyl)- N-dodecylpropane-1,3-diamine | AP | AP | AP | AP | 0.1–2.80 | 7.50–68.61 |
| Biguanides | ||||||
| Polyhexamethylene biguanide hydrochloride | AP | AP | AP | AP | a | 0.050–0.089 |
| Chlorine based compounds | ||||||
| Calcium hypochlorite | AP | NE | AP | AP | a | b |
| Chlorine dioxide | UR | AP | UR | AP | a | 0.20–5.00 |
| Hypochlorous acid | UR | AP | UR | AP | 0.017 | 0.017–0.046 |
| Sodium hypochlorite | AP | AP | AP | AP | 2.60–13.00 | 0.086–8.60 |
| Sodium chlorite | UR | AP | UR | AP | a | 0.50–30.50 |
| Sodium dichloroisocyanurate | UR | AP | UR | AP | 81.00 | 7.00–48.21 |
| Tetrachlorodecaoxide complex | UR | NE | UR | NE | a | b |
| Tosylchloramide sodium | UR | NE | UR | AP | a | b |
| Trichloroisocyanuric acid | UR | NE | UR | AP | a | b |
| Iodine and iodophors | ||||||
| Iodine | AP | NE | AP | AP | a | b |
| Povidone-iodine | AP | NE | AP | AP | a | b |
| Isothiazolinones | ||||||
| Mixture of CMIT/MIT ** | AP | NE | AP | AP | a | b |
| Organic acids | ||||||
| Citric acid | AP | AP | AP | AP | a | 0.60–6.00 |
| Formic acid | UR | NE | UR | AP | a | b |
| Glycolic acid | UR | AP | UR | AP | a | 11.19 |
| Lactic acid | AP | AP | AP | AP | 0.42–1.75 | 0.16–34.10 |
| Performic acid | UR | NE | UR | AP | a | b |
| Peroxides and derivates | ||||||
| Hydrogen peroxide | AP | AP | AP | AP | 0.20–35.00 | 0.30–27.50 |
| Peracetic acid | AP | AP | AP | AP | 0.05–5.00 | 0.05–15.00 |
| Peroxyoctanoic acid | UR | AP | UR | AP | a | 0.63 |
| Potassium peroxymonosulfate | NE | AP | UR | AP | 49.70 | 21.41 |
| Sodium carbonate peroxyhydrate | NE | AP | NE | AP | a | 12.10–29.75 |
| Phenolic compounds | ||||||
| 2-Phenylphenol | UR | AP | UR | AP | a | 0.026–10.50 |
| 2-Benzyl-4-chlorophenol | NE | AP | NE | AP | a | 0.023–3.03 |
| 4-tert-amylphenol | NE | AP | NE | AP | a | 5.27–7.66 |
| 5-chloro-2-(4-chlorophenoxy) phenol | AP | NE | AP | NE | a | b |
| Biphenyl-2-ol | AP | AP | AP | AP | a | 0.06 |
| Salicylic acid | UR | NE | UR | AP | a | b |
| Thymol | NE | AP | NE | AP | a | 0.092–0.23 |
| Quaternary ammonium compounds | ||||||
| Benzalkonium chloride | AP | AP | UR | AP | 0.008–24.00 | 0.015–26.00 |
| Benzalkonium saccharinate | AP | AP | UR | AP | a | 0.10–0.20 |
| Benzethonium chloride | NE | AP | NE | AP | a | 0.28 |
| Didecyldimethylammonium chloride | AP | AP | UR | AP | 0.20–7.20 | 0.003–21.05 |
| Didecylmethylpoly(oxethyl) ammonium propionate | AP | NE | UR | NE | a | b |
| Didecylmethylammonium carbonate/bicarbonate | NE | AP | NE | AP | a | 0.0369–1.38 |
| Silver and derivates | ||||||
| Silver | AP | AP | UR | AP | 0.004 | 0.003–0.01 |
| Silver nitrate | AP | AP | UR | AP | a | 0.016 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pedreira, A.; Taşkın, Y.; García, M.R. A Critical Review of Disinfection Processes to Control SARS-CoV-2 Transmission in the Food Industry. Foods 2021, 10, 283. https://doi.org/10.3390/foods10020283
Pedreira A, Taşkın Y, García MR. A Critical Review of Disinfection Processes to Control SARS-CoV-2 Transmission in the Food Industry. Foods. 2021; 10(2):283. https://doi.org/10.3390/foods10020283
Chicago/Turabian StylePedreira, Adrián, Yeşim Taşkın, and Míriam R. García. 2021. "A Critical Review of Disinfection Processes to Control SARS-CoV-2 Transmission in the Food Industry" Foods 10, no. 2: 283. https://doi.org/10.3390/foods10020283
APA StylePedreira, A., Taşkın, Y., & García, M. R. (2021). A Critical Review of Disinfection Processes to Control SARS-CoV-2 Transmission in the Food Industry. Foods, 10(2), 283. https://doi.org/10.3390/foods10020283






