Native Species Facing Climate Changes: Response of Calafate Berries to Low Temperature and UV Radiation
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Chemical Reagents
2.3. Meteorological Data Information
2.4. Sample Preparation
2.5. Physical and Chemical Parameters
2.6. Identification and Quantification of Anthocyanins, Flavonols and Other Phenolic Compounds
2.7. Antioxidant Capacity Determination
2.8. Statistical Analysis
3. Results and Discussion
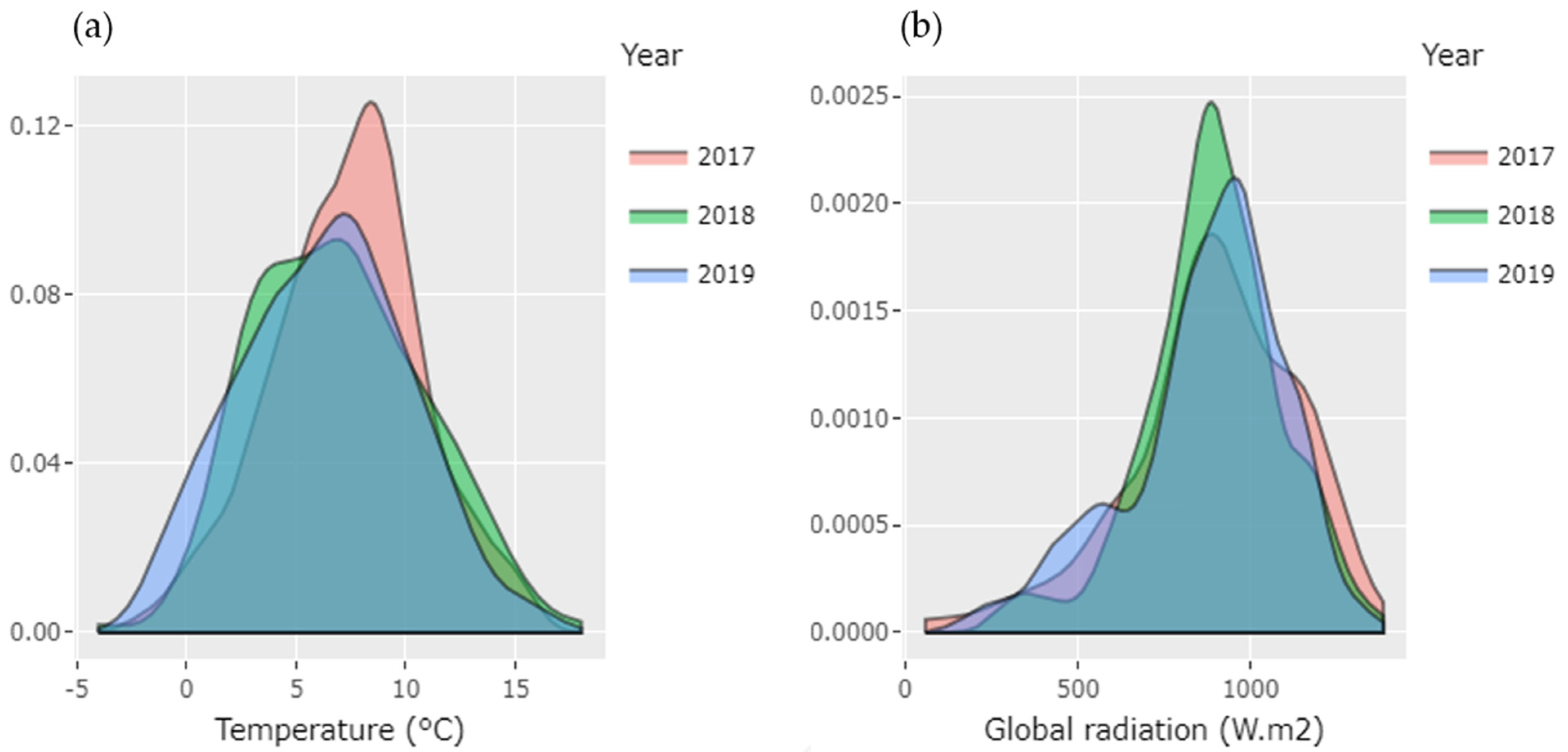
3.1. Variation in Environmental Conditions
3.2. Physicochemical Parameters
3.3. Analysis of CIEL*a*b* Values
3.4. Comparison of Calafate Anthocyanin Profile and Other Phenolic Compounds
Antioxidant Capacity in Calafate Berries
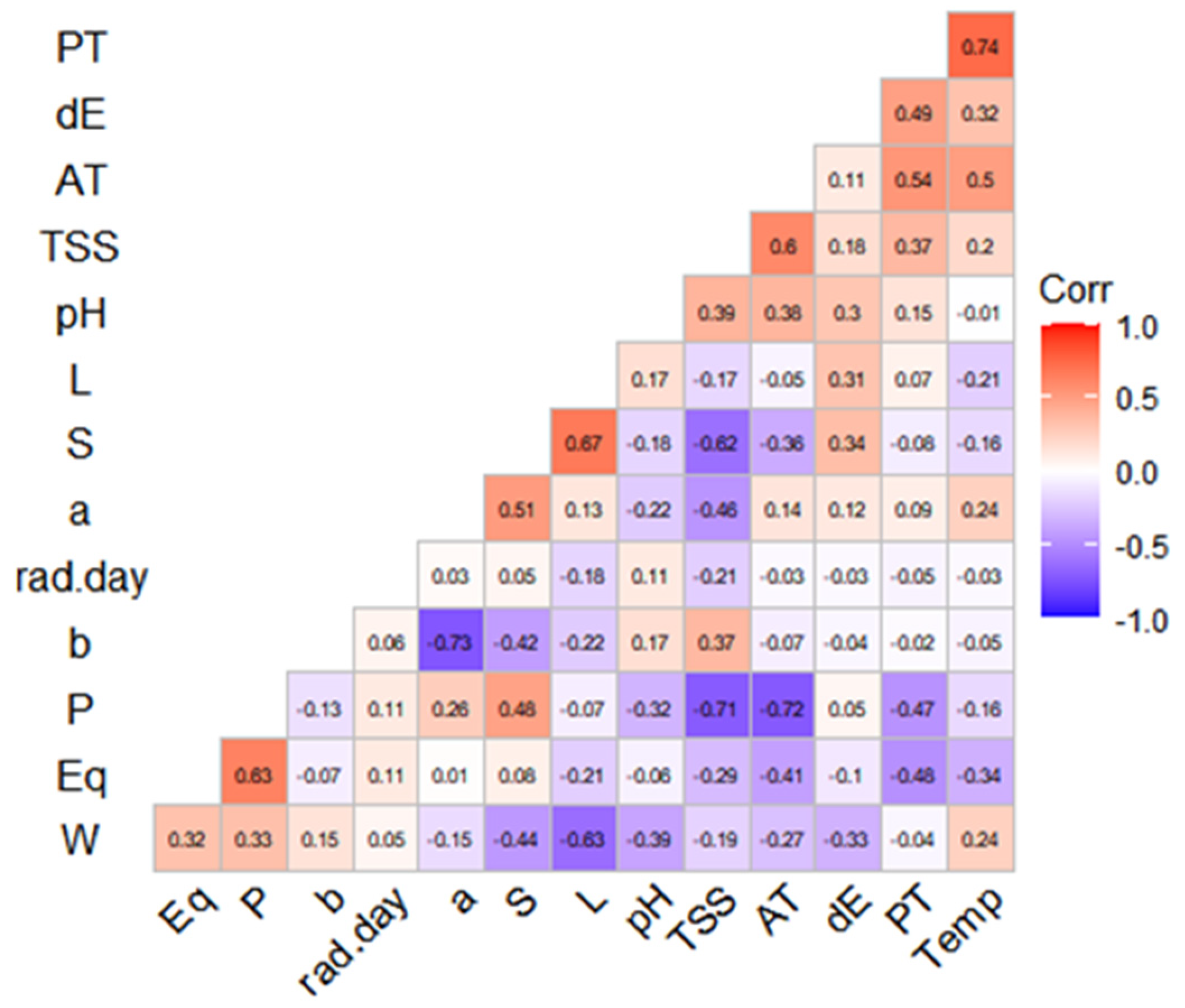
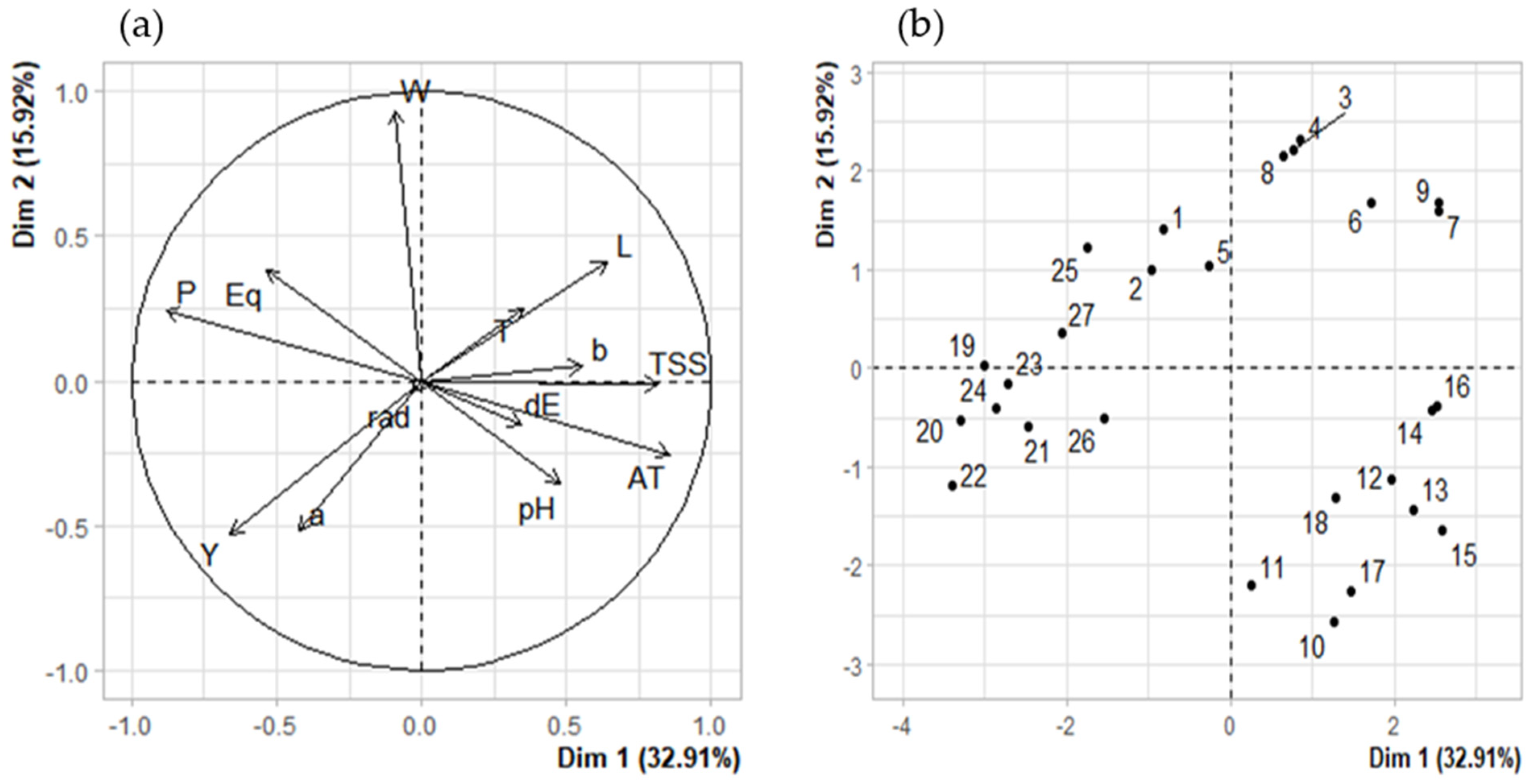
3.5. Correlations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Khoo, H.E.; Azlan, A.; Tang, S.T.; Lim, S.M. Anthocyanidins and anthocyanins: Colored pigments as food, pharmaceutical ingredients, and the potential health benefits. Food Nutr. Res. 2017, 61, 1361779. [Google Scholar] [CrossRef] [PubMed]
- Mariangel, E.; Reyes-Diaz, M.; Lobos, W.; Bensch, E.; Schalchli, H.; Ibarra, P. The antioxidant properties of calafate (Berberis microphylla) fruits from four different locations in southern Chile. Ciencia Invest. Agrar. 2013, 40, 161–170. [Google Scholar] [CrossRef]
- Manosalva, L.; Mutis, A.; Urzúa, A.; Fajardo, V.; Quiroz, A. Antibacterial Activity of Alkaloid Fractions from Berberis microphylla G. Forst and Study of Synergism with Ampicillin and Cephalothin. Molecules 2016, 21, 76. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Selamoglu, Z.; Sener, B.; Kilic, M.; Jugran, A.K.; De Tommasi, N.; Sinisgalli, C.; Milella, L.; Rajkovic, J.; Morais-Braga, M.F.B.; et al. Berberis Plants-Drifting from Farm to Food Applications, Phytotherapy, and Phytopharmacology. Foods 2019, 8, 522. [Google Scholar] [CrossRef] [PubMed]
- Araya, M. Estudio Preliminar de La Composición Química y El Valor Nutricional de Frutos Regionales de Interés Econó-mico y Sociocultural de Magallanes; Trabajo de Titulación Para La Obtención de Ingeniero En Química y Medio Ambiente Uni-versidad de Magallanes; Universidad de Magallanes: Punta Arenas, Chile, 2010. [Google Scholar]
- Ruiz, A.; Hermosín-Gutiérrez, I.; Mardones, C.; Vergara, C.; Herlitz, E.; Vega, M.; Dorau, C.; Winterhalter, P.; Von Baer, D. Polyphenols and Antioxidant Activity of Calafate (Berberis microphylla) Fruits and Other Native Berries from Southern Chile. J. Agric. Food Chem. 2010, 58, 6081–6089. [Google Scholar] [CrossRef] [PubMed]
- Radice, S.; Arena, M.E. Environmental effect on the leaf morphology and anatomy of Berberis microphylla G. Forst. Int. J. Plant Biol. 2015, 6. [Google Scholar] [CrossRef]
- Arena, M.E.; Pastur, G.M.; Lencinas, M.V.; Soler, R.; Bustamante, G. Changes in the leaf nutrient and pigment contents of Berberis microphylla G. Forst. in relation to irradiance and fertilization. Heliyon 2020, 6, e03264. [Google Scholar] [CrossRef]
- Rodoni, L.M.; Feuring, V.; Zaro, M.J.; Sozzi, G.O.; Vicente, A.R.; Arena, M.E. Ethylene Responses and Quality of Antioxi-dant-Rich Stored Barberry Fruit (Berberis Microphylla). Sci. Hortic. 2014, 179, 233–238. [Google Scholar] [CrossRef]
- Yan, X.; Yan, J.; Pan, S.; Yuan, F. Changes of the Aroma Composition and Other Quality Traits of Blueberry ‘Garden Blue’ during the Cold Storage and Subsequent Shelf Life. Foods 2020, 9, 1223. [Google Scholar] [CrossRef]
- De Oliveira, A.F.; Mercenaro, L.; Del Caro, A.; Pretti, L.; Nieddu, G. Distinctive Anthocyanin Accumulation Responses to Temperature and Natural UV Radiation of Two Field-Grown Vitis vinifera L. Cultivars. Molecules 2015, 20, 2061–2080. [Google Scholar] [CrossRef]
- Ferreira, S.S.; Silva, P.; Silva, A.M.; Nunes, F.M. Effect of harvesting year and elderberry cultivar on the chemical composition and potential bioactivity: A three-year study. Food Chem. 2020, 302, 125366. [Google Scholar] [CrossRef] [PubMed]
- Pino, M.T.; Pérez, R.; Vergara, C.; Zamora, O.; Dominguez, E. Michay: Berry Nativo de Amplia Distribución Con Metabo-litos de Interés Para La Industria de Alimentos. Inf. INIA La Platina 2019, 39, 1–4. [Google Scholar]
- Silva, F. Flora agropecuaria de Aysén. Servicio de Agricultura y Ganadería; Museo Nacional de Historia Natural: Coyhaique, Chile, 2013. [Google Scholar]
- Hepp, K.; Stolpe, N.B. Caracterización y Propiedades de Los Suelos de La Patagonia Occidental (Aysén); Boletin; INIA-Instituto de Investigaciones Agropecuarias: Coyhaique, Aysén, 2014. [Google Scholar]
- Salinas, S.; Gómez, N.; Riquelme Espergue, F.; Acuña Aroca, B.; Díaz, V. Manual de Productos Forestales No Madereros (PFNM). Proyecto Manejo Sustentable de La Tierra Comuna de Coyhaique; Corporación Nacional Forestal: Santiago, Chile, 2019. [Google Scholar]
- Magrin, G.O.; Marengo, J.A.; Boulanger, J.-P.; Buckeridge, M.S.; Castellanos, E.; Poveda, G.; Scarano, F.R.; Barros, V.; Field, C.; Dokken, D. Central and south america. In Impacts, Adaptation, and Vulnerability; Part B: Regional Aspects. Contribution of Working Group II to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2014; pp. 1499–1566. [Google Scholar]
- Bais, A.F.; McKenzie, R.L.; Bernhard, G.; Aucamp, P.J.; Ilyas, M.; Madronich, S.; Tourpali, K. Ozone depletion and climate change: Impacts on UV radiation. Photochem. Photobiol. Sci. 2014, 14, 19–52. [Google Scholar] [CrossRef] [PubMed]
- Arribillaga, G.P. Domesticación del Calafate (Berberis buxifolia L.) Para Fines Agroindustriales; INIA: Coyhaique, Chile, 2001; p. 144. [Google Scholar]
- López, M.D.; Baenas, N.; Retamal-Salgado, J.; Zapata, N.; Moreno, D.A. Underutilized Native Biobío Berries: Opportunities for Foods and Trade. Nat. Prod. Commun. 2018, 13, 1934578–1801301226. [Google Scholar] [CrossRef]
- Romero Román, M.; Noriega Vásquez, F.; Farías Villagra, M.; Belchi, L.; Jara Zapata, P.; Vera Flores, B. Nuevas Fuentes de Antioxidantes Naturales: Caracterización de Compuestos Bioactivos En Cinco Frutos Nativos de Chile. Perfiles 2019, 22, 34–41. [Google Scholar]
- Cesa, S.; Carradori, S.; Bellagamba, G.; Locatelli, M.; Casadei, M.A.; Masci, A.; Paolicelli, P. Evaluation of processing effects on anthocyanin content and colour modifications of blueberry (Vaccinium spp.) extracts: Comparison between HPLC-DAD and CIELAB analyses. Food Chem. 2017, 232, 114–123. [Google Scholar] [CrossRef]
- Salar, F.J.; Agulló, V.; García-Viguera, C.; Domínguez-Perles, R. Stevia vs. Sucrose: Influence on the Phytochemical Content of a Citrus–Maqui Beverage—A Shelf Life Study. Foods 2020, 9, 219. [Google Scholar] [CrossRef]
- Agulló, V.; Villaño, D.; García-Viguera, C.; Domínguez-Perles, R. Anthocyanin Metabolites in Human Urine after the Intake of New Functional Beverages. Molecules 2020, 25, 371. [Google Scholar] [CrossRef]
- Bondet, V.; Brand-Williams, W.; Berset, C. Kinetics and Mechanisms of Antioxidant Activity using the DPPH.Free Radical Method. LWT 1997, 30, 609–615. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef]
- RStudio Team. RStudio: Integrated Development for R.; RStudio, PBC: Boston, MA, USA, 2020. [Google Scholar]
- Wickham, H. Ggplot2. Wiley Interdiscip. Rev. Comput. Stat. 2011, 3, 180–185. [Google Scholar] [CrossRef]
- Del-Castillo-Alonso, M.Á.; Castagna, A.; Csepregi, K.; Hideg, É.; Jakab, G.; Jansen, M.A.; Jug, T.; Llorens, L.; Mátai, A.; Mar-tínez-Lüscher, J. Environmental Factors Correlated with the Metabolite Profile of Vitis Vinifera Cv. Pinot Noir Berry Skins along a European Latitudinal Gradient. J. Agric. Food chem. 2016, 64, 8722–8734. [Google Scholar] [CrossRef] [PubMed]
- Cardenas, A. Identificación e Incidencia de Puccinia Meyeri-Albertii P. Magn. en Calafate (Berberis microphylla G. Forst.) y Michay (Berberis darwinii Hook.) en el Llano Central de la Región de La Araucanía; Universidad de la Frontera: Temuco, Chile, 2012. [Google Scholar]
- Saavedra, J.; Pino, M.T.; Zamora, O.; Ojeda, A.; Leod, C.M.; Aguila, K. Análisis de diversidad genética del calafate en Maga-llanes. Inf. INIA Kampenaike 2017, 1–4. [Google Scholar]
- Hykkerud, A.L.; Uleberg, E.; Hansen, E.; Vervoort, M.; Mølmann, J.; Martinussen, I. Seasonal and Yearly Variation of Total Polyphenols, Total Anthocyanins and Ellagic Acid in Different Clones of Cloudberries (Rubus Chamaemorus L.). Angew. Bot. 2018, 91, 96–102. [Google Scholar]
- Gentilucci, M. Grapevine Prediction of End of Flowering Date; Springer: Berlin/Heidelberg, Germany, 2018; pp. 1231–1233. [Google Scholar]
- Nunes, N.; Leite, A.; Castro, C. Phenology, Reproductive Biology and Growing Degree Days of the Grapevine ‘Isabel’(Vitis Labrusca, Vitaceae) Cultivated in Northeastern Brazil. Braz. J. Biol. 2016, 76, 975–982. [Google Scholar] [CrossRef] [PubMed]
- Portal de Servicios Climáticos—Dirección Meteorológica de Chile. Available online: https://climatologia.meteochile.gob.cl/application/mensuales/climatMensualDatosDiarios/450004/2019/3 (accessed on 14 July 2020).
- Pellicciotti, F.; Ragettli, S.; Carenzo, M.; McPhee, J. Changes of glaciers in the Andes of Chile and priorities for future work. Sci. Total Environ. 2014, 493, 1197–1210. [Google Scholar] [CrossRef] [PubMed]
- Di Vittori, L.; Mazzoni, L.; Battino, M.; Mezzetti, B. Pre-harvest factors influencing the quality of berries. Sci. Hortic. 2018, 233, 310–322. [Google Scholar] [CrossRef]
- Anuar, N.; Taha, R.; Mahmad, N.; Mohajer, S.; Musa, S.A.N.C.; Abidin, Z.H. Correlation of colour, antioxidant capacity and phytochemical diversity of imported saffron by principal components analysis. Pigment. Resin Technol. 2017, 46, 107–114. [Google Scholar] [CrossRef]
- Chamorro, M.F.; Reiner, G.; Theoduloz, C.; Ladio, A.H.; Schmeda-Hirschmann, G.; Gómez-Alonso, S.; Jiménez-Aspee, F. Polyphenol Composition and (Bio)Activity of Berberis Species and Wild Strawberry from the Argentinean Patagonia. Molecules 2019, 24, 3331. [Google Scholar] [CrossRef]
- Brito, A.; Areche, C.; Sepulveda, B.; Kennelly, E.J.; Simirgiotis, M.J. Anthocyanin Characterization, Total Phenolic Quantification and Antioxidant Features of Some Chilean Edible Berry Extracts. Molecules 2014, 19, 10936–10955. [Google Scholar] [CrossRef]
- Guan, L.; Dai, Z.; Wu, B.-H.; Wu, J.; Merlin, I.; Hilbert, G.; Renaud, C.; Gomès, E.; Edwards, E.; Li, S.-H. Anthocyanin Bio-synthesis is Differentially Regulated by Light in the Skin and Flesh of White-Fleshed and Teinturier Grape Berries. Planta 2016, 243, 23–41. [Google Scholar] [CrossRef] [PubMed]
| Sample | Weight (g) | Equatorial Diameter (mm) | Polar Diameter (mm) | pH | Soluble Solids (°Brix) | Acidity (%) |
|---|---|---|---|---|---|---|
| Calafate 2017 | 0.60 ± 0.02 c | 9.9 ± 0.04 ab | 9.0 ± 0.06 a | 3.61 ± 0.03 ab | 27.2 ± 0.10 b | 2.0 ± 0.07 a |
| Calafate 2018 | 0.50 ± 0.01 a | 8.5 ± 0.38 a | 7.8 ± 0.14 a | 3.66 ± 0.01 a | 26.8 ± 0.02 b | 1.0 ± 0.03 b |
| Calafate 2019 | 0.54 ± 0.06 b | 10.3 ± 0.16 b | 8.4 ± 0.50 c | 3.7 ± 0.01 b | 22.7 ± 0.37 a | 0.9 ± 0.03 b |
| Year/Color Parameter | L* | a* | b* | c* | H° | ΔE |
|---|---|---|---|---|---|---|
| 2017 | 29.47 ± 7.5 a | 0.61 ± 0.02 a | −0.08 ± 0.21 a | 1.59 ± 0.14 a | 7.71 ± 0.11 a | 2.35 ± 0.05 a |
| 2018 | 35.12 ± 6.20 b | 1.05 ± 0.18 a | −0.88 ± 0.07 ab | 2.48 ± 0.43 ab | 39.79 ± 0.71 b | 4.41 ± 0.59 b |
| 2019 | 33.78 ± 5.43 b | 1.98 ± 0.08 b | −2.21 ± 0.16 b | 3.36 ± 0.08 b | 47.98 ± 0.49 c | 3.74 ± 0.54 a,b |
| Compounds | 2017 | 2018 | 2019 |
|---|---|---|---|
| Delphinidin 3-glucoside | 10.68 b | 11.60 a | 9.78 a,b |
| Cyanidin 3-glucoside | 0.20 a | 0.42 b | 0.25 a,b |
| Petunidin 3-glucoside | 3.08 b | 3.38 b | 1.73 a |
| Petunidin 3-rutinoside | 4.17 b | 4.44 b | 2.47 a |
| Peonidin 3-glucoside | 0.03 a,b | 0.02 b | 0.33 a |
| Malvidin 3-glucoside | 0.35 c | 0.03 a | 0.14 b |
| Malvidin 3-rutinoside | 0.10 b | 0.12 c | 0.07 a |
| Sum of anthocyanins | 18.62 b | 20.0 a | 14.77 c |
| Sum of flavanols and other phenolic compounds | 3.42 b | 2.69 c | 3.59 a |
| Sum of phenolic compounds | 22.04 b | 22.69 a | 18.36 c |
| Year | FRAP | DPPH● |
|---|---|---|
| 2017 | 4.07 ± 0.03 a | 41.55 ± 0.96 b |
| 2018 | 3.53 ± 0.07 a | 36.47 ± 0.94 a |
| 2019 | 4.12 ± 0.02 a | 35.44 ± 0.11 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Romero-Román, M.E.; Schoebitz, M.; Bastías, R.M.; Fernández, P.S.; García-Viguera, C.; López-Belchi, M.D. Native Species Facing Climate Changes: Response of Calafate Berries to Low Temperature and UV Radiation. Foods 2021, 10, 196. https://doi.org/10.3390/foods10010196
Romero-Román ME, Schoebitz M, Bastías RM, Fernández PS, García-Viguera C, López-Belchi MD. Native Species Facing Climate Changes: Response of Calafate Berries to Low Temperature and UV Radiation. Foods. 2021; 10(1):196. https://doi.org/10.3390/foods10010196
Chicago/Turabian StyleRomero-Román, María Eugenia, Mauricio Schoebitz, Richard M. Bastías, Pablo S. Fernández, Cristina García-Viguera, and María Dolores López-Belchi. 2021. "Native Species Facing Climate Changes: Response of Calafate Berries to Low Temperature and UV Radiation" Foods 10, no. 1: 196. https://doi.org/10.3390/foods10010196
APA StyleRomero-Román, M. E., Schoebitz, M., Bastías, R. M., Fernández, P. S., García-Viguera, C., & López-Belchi, M. D. (2021). Native Species Facing Climate Changes: Response of Calafate Berries to Low Temperature and UV Radiation. Foods, 10(1), 196. https://doi.org/10.3390/foods10010196