Spectrophotometric Determination of the Aggregation Activity of Platelets in Platelet-Rich Plasma for Better Quality Control
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of P-PRP and Platelet Suspension
2.2. Spectrophotometric Determination of Platelet Aggregation
2.3. Statistical Analysis
3. Results
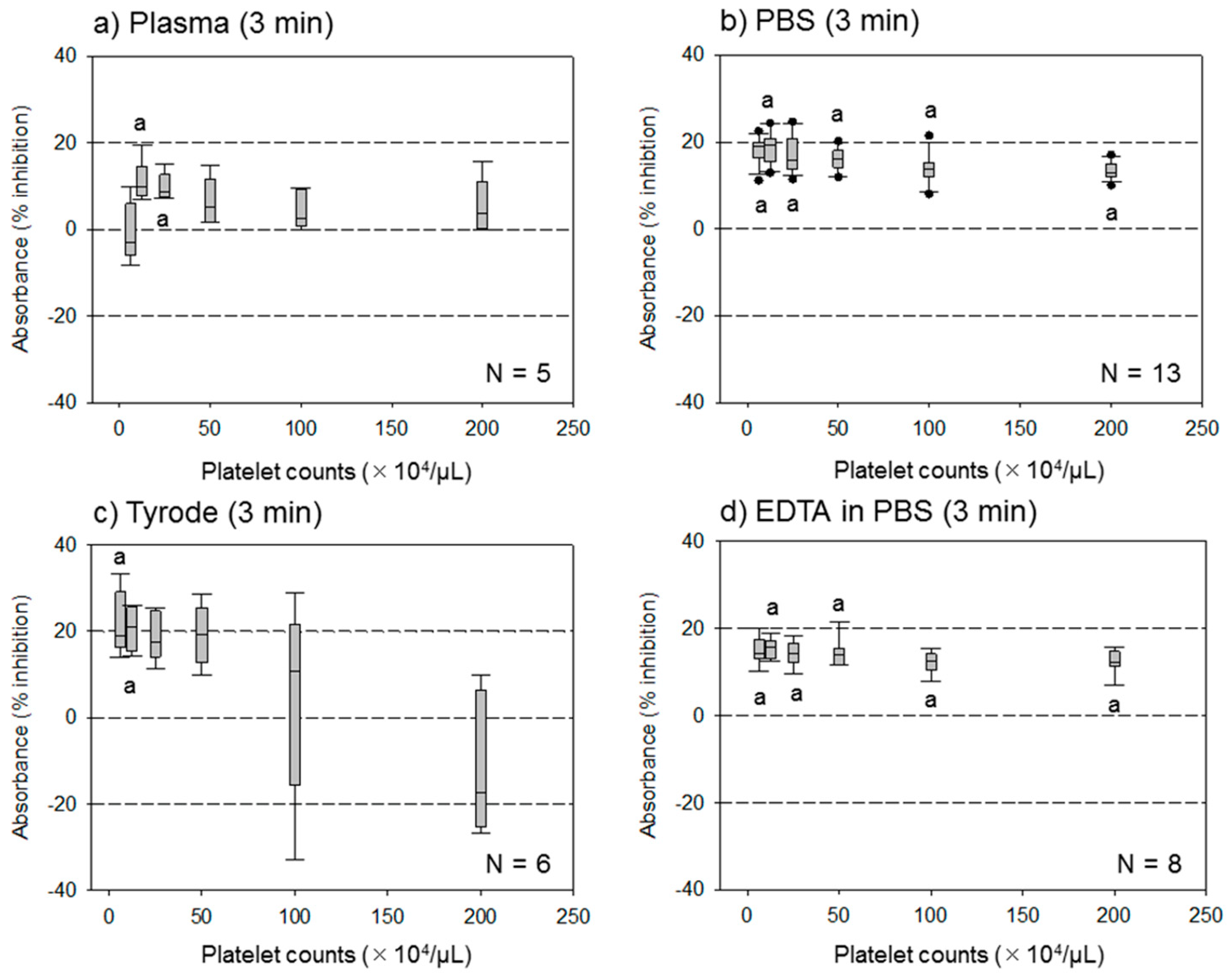
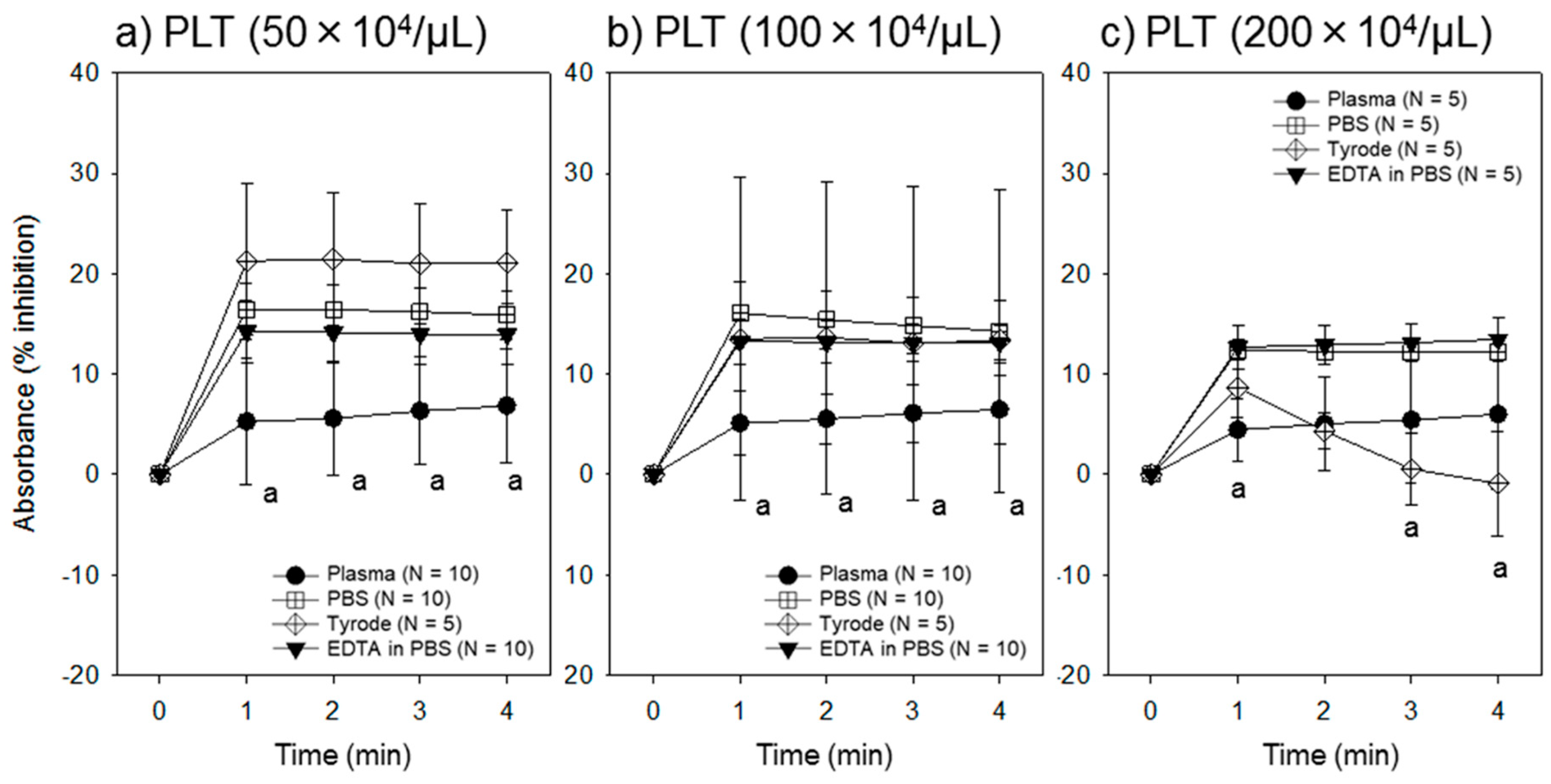
3.1. Effect of Different Assay Buffer Solutions, Time Points, and Platelet Densities on the Assay System
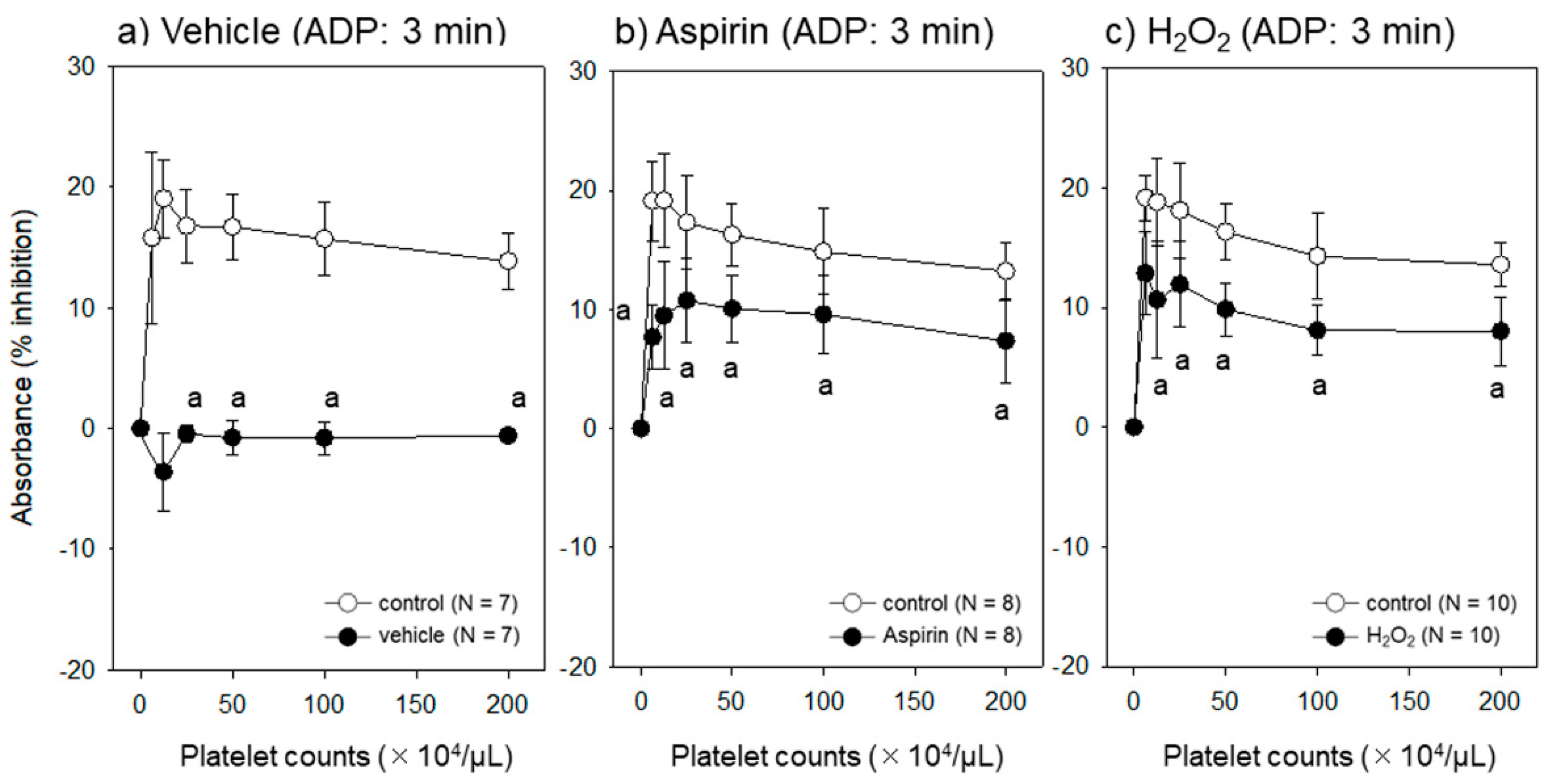
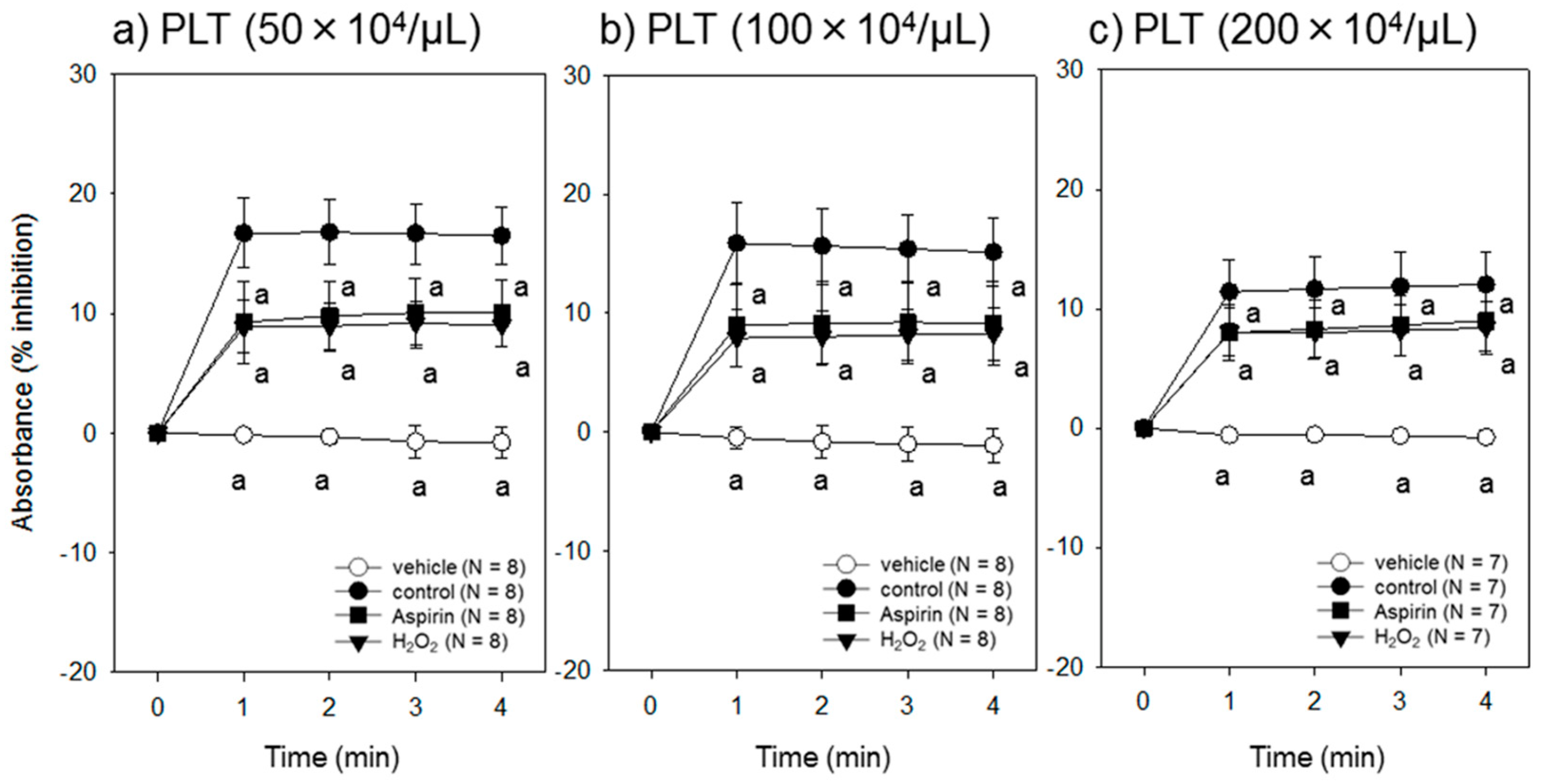
3.2. Effect of Different Platelet Conditions on the Assay System
4. Discussion
4.1. Optimal Assay Buffer Solutions
4.2. Optimal Range of Platelet Density
4.3. Optimal End Point
4.4. Comparisons with Suppressed or Injured Platelets
4.5. Limitations of This Study
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| PRP | Platelet-rich plasma |
| P-PRP | Pure platelet-rich plasma |
| ACD-A | Acid-citrate-dextrose formula A |
| ADP | Adenosine diphosphate |
| PGE1 | Prostaglandin E1 |
| PBS | Phosphate buffered saline |
| PCR | Polymerase chain reaction |
| H2O2 | Hydrogen peroxide |
| EDTA | Ethylenediaminetetraacetic acid |
| CBMP | Cell-based medicinal product |
| HIV | Human immunodeficiency virus |
| HBV | Hepatitis B virus |
| HCV | Hepatitis C virus |
References
- Marx, R.E.; Carlson, E.R.; Eichstaedt, R.M.; Schimmele, S.R.; Strauss, J.E.; Georgeff, K.R. Platelet-rich plasma: Growth factor enhancement for bone grafts. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 1998, 85, 638–646. [Google Scholar] [CrossRef]
- Kawase, T.; Takahashi, A.; Watanabe, T.; Tsujino, T. Proposal for point-of-care testing of platelet-rich plasma quality. Int. J. Growth Factors Stem Cells Dent. 2019, 2, 13–17. [Google Scholar] [CrossRef]
- Kawase, T.; Okuda, K. Comprehensive Quality Control of the Regenerative Therapy Using Platelet Concentrates: The Current Situation and Prospects in Japan. BioMed Res. Int. 2018, 2018, 6389157. [Google Scholar] [CrossRef]
- Anitua, E.; Zalduendo, M.; Troya, M.; Padilla, S.; Orive, G. Leukocyte inclusion within a platelet rich plasma-derived fibrin scaffold stimulates a more pro-inflammatory environment and alters fibrin properties. PLoS ONE 2015, 10, e0121713. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Yin, W.; Zhang, Y.; Qi, X.; Chen, Y.; Xie, X.; Zhang, C. Comparative evaluation of leukocyte- and platelet-rich plasma and pure platelet-rich plasma for cartilage regeneration. Sci. Rep. 2017, 7, 43301. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, Y.; Suzuki, M.; Tsukioka, T.; Isobe, K.; Tsujino, T.; Watanabe, T.; Watanabe, T.; Okudera, H.; Nakata, K.; Tanaka, T.; et al. Spectrophotometric determination of platelet counts in platelet-rich plasma. Int. J. Implant Dent. 2018, 4, 29. [Google Scholar] [CrossRef]
- Bednar, B.; Condra, C.; Gould, R.J.; Connolly, T.M. Platelet aggregation monitored in a 96 well microplate reader is useful for evaluation of platelet agonists and antagonists. Thromb. Res. 1995, 77, 453–463. [Google Scholar] [CrossRef]
- Chan, M.V.; Armstrong, P.C.; Warner, T.D. 96-well plate-based aggregometry. Platelets 2018, 29, 650–655. [Google Scholar] [CrossRef]
- Krause, S.; Scholz, T.; Temmler, U.; Losche, W. Monitoring the effects of platelet glycoprotein IIb/IIIa antagonists with a microtiter plate method for detection of platelet aggregation. Platelets 2001, 12, 423–430. [Google Scholar] [CrossRef]
- Fratantoni, J.C.; Poindexter, B.J. Measuring platelet aggregation with microplate reader. A new technical approach to platelet aggregation studies. Am. J. Clin. Pathol. 1990, 94, 613–617. [Google Scholar] [CrossRef]
- Isobe, K.; Suzuki, M.; Watanabe, T.; Kitamura, Y.; Suzuki, T.; Kawabata, H.; Nakamura, M.; Okudera, T.; Okudera, H.; Uematsu, K.; et al. Platelet-rich fibrin prepared from stored whole-blood samples. Int. J. Implant Dent. 2017, 3, 6. [Google Scholar] [CrossRef] [PubMed]
- Kawabata, H.; Isobe, K.; Watanabe, T.; Okudera, T.; Nakamura, M.; Suzuki, M.; Ryu, J.; Kitamura, Y.; Okudera, H.; Okuda, K.; et al. Quality Assessment of Platelet-Rich Fibrin-Like Matrix Prepared from Whole Blood Samples after Extended Storage. Biomedicines 2017, 5, 57. [Google Scholar] [CrossRef]
- Takahashi, A.; Takahashi, S.; Tsujino, T.; Isobe, K.; Watanabe, T.; Kitamura, Y.; Watanabe, T.; Nakata, K.; Kawase, T. Platelet adhesion on commercially pure titanium plates in vitro I: Effects of plasma components and involvement of the von Willebrand factor and fibronectin. Int. J. Implant Dent. 2019, 5, 5. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Isobe, K.; Suzuki, T.; Kawabata, H.; Nakamura, M.; Tsukioka, T.; Okudera, T.; Okudera, H.; Uematsu, K.; Okuda, K.; et al. An Evaluation of the Accuracy of the Subtraction Method Used for Determining Platelet Counts in Advanced Platelet-Rich Fibrin and Concentrated Growth Factor Preparations. Dent. J. 2017, 5, 7. [Google Scholar] [CrossRef] [PubMed]
- Practical-Haemostasis.com. A Practical Guide to Laboratory Haemostasis. Volume 2019. 2013. Available online: http://www.practical-haemostasis.com/Platelets/platelet_function_testing_lta.html (accessed on 16 April 2019).
- Kitamura, Y.; Isobe, K.; Kawabata, H.; Tsujino, T.; Watanabe, T.; Nakamura, M.; Toyoda, T.; Okudera, H.; Okuda, K.; Nakata, K.; et al. Quantitative evaluation of morphological changes in activated platelets in vitro using digital holographic microscopy. Micron 2018, 113, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Toyoda, T.; Isobe, K.; Tsujino, T.; Koyata, Y.; Ohyagi, F.; Watanabe, T.; Nakamura, M.; Kitamura, Y.; Okudera, H.; Nakata, K.; et al. Direct activation of platelets by addition of CaCl2 leads coagulation of platelet-rich plasma. Int. J. Implant Dent. 2018, 4, 23. [Google Scholar] [CrossRef]
- Meyers, K.M.; Lindner, C.; Katz, J.; Grant, B. Phenylbutazone inhibition of equine platelet function. Am. J. Vet. Res. 1979, 40, 265–270. [Google Scholar] [PubMed]
- Belisario, M.A.; Tafuri, S.; Di Domenico, C.; Squillacioti, C.; Della Morte, R.; Lucisano, A.; Staiano, N. H2O2 activity on platelet adhesion to fibrinogen and protein tyrosine phosphorylation. Biochim. Biophys. Acta 2000, 1495, 183–193. [Google Scholar] [CrossRef]
- Del Principe, D.; Menichelli, A.; De Matteis, W.; Di Corpo, M.L.; Di Giulio, S.; Finazzi-Agro, A. Hydrogen peroxide has a role in the aggregation of human platelets. FEBS Lett. 1985, 185, 142–146. [Google Scholar] [CrossRef]
- Krotz, F.; Sohn, H.Y.; Pohl, U. Reactive oxygen species: Players in the platelet game. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 1988–1996. [Google Scholar] [CrossRef]
- Chandrashekar, V. Does platelet count in platelet-rich plasma influence slope, maximal amplitude and lag phase in healthy individuals? Results of light transmission aggregometry. Platelets 2015, 26, 699–701. [Google Scholar] [CrossRef] [PubMed]
- Lindkvist, M.; Fernberg, U.; Ljungberg, L.U.; Falker, K.; Fernstrom, M.; Hurtig-Wennlof, A.; Grenegard, M. Individual variations in platelet reactivity towards ADP, epinephrine, collagen and nitric oxide, and the association to arterial function in young, healthy adults. Thromb. Res. 2019, 174, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Backman, J.D.; Yerges-Armstrong, L.M.; Horenstein, R.B.; Newcomer, S.; Shaub, S.; Morrisey, M.; Donnelly, P.; Drolet, M.; Tanner, K.; Pavlovich, M.A.; et al. Prospective Evaluation of Genetic Variation in Platelet Endothelial Aggregation Receptor 1 Reveals Aspirin-Dependent Effects on Platelet Aggregation Pathways. Clin. Transl. Sci. 2017, 10, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Edelstein, L.C.; Simon, L.M.; Lindsay, C.R.; Kong, X.; Teruel-Montoya, R.; Tourdot, B.E.; Chen, E.S.; Ma, L.; Coughlin, S.; Nieman, M.; et al. Common variants in the human platelet PAR4 thrombin receptor alter platelet function and differ by race. Blood 2014, 124, 3450–3458. [Google Scholar] [CrossRef] [PubMed]
- Fontana, P.; Dupont, A.; Gandrille, S.; Bachelot-Loza, C.; Reny, J.L.; Aiach, M.; Gaussem, P. Adenosine diphosphate-induced platelet aggregation is associated with P2Y12 gene sequence variations in healthy subjects. Circulation 2003, 108, 989–995. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsujino, T.; Isobe, K.; Kawabata, H.; Aizawa, H.; Yamaguchi, S.; Kitamura, Y.; Masuki, H.; Watanabe, T.; Okudera, H.; Nakata, K.; et al. Spectrophotometric Determination of the Aggregation Activity of Platelets in Platelet-Rich Plasma for Better Quality Control. Dent. J. 2019, 7, 61. https://doi.org/10.3390/dj7020061
Tsujino T, Isobe K, Kawabata H, Aizawa H, Yamaguchi S, Kitamura Y, Masuki H, Watanabe T, Okudera H, Nakata K, et al. Spectrophotometric Determination of the Aggregation Activity of Platelets in Platelet-Rich Plasma for Better Quality Control. Dentistry Journal. 2019; 7(2):61. https://doi.org/10.3390/dj7020061
Chicago/Turabian StyleTsujino, Tetsuhiro, Kazushige Isobe, Hideo Kawabata, Hachidai Aizawa, Sadahiro Yamaguchi, Yutaka Kitamura, Hideo Masuki, Taisuke Watanabe, Hajime Okudera, Koh Nakata, and et al. 2019. "Spectrophotometric Determination of the Aggregation Activity of Platelets in Platelet-Rich Plasma for Better Quality Control" Dentistry Journal 7, no. 2: 61. https://doi.org/10.3390/dj7020061
APA StyleTsujino, T., Isobe, K., Kawabata, H., Aizawa, H., Yamaguchi, S., Kitamura, Y., Masuki, H., Watanabe, T., Okudera, H., Nakata, K., & Kawase, T. (2019). Spectrophotometric Determination of the Aggregation Activity of Platelets in Platelet-Rich Plasma for Better Quality Control. Dentistry Journal, 7(2), 61. https://doi.org/10.3390/dj7020061





