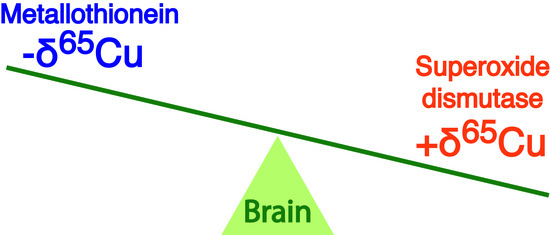
Copper Isotope Compositions of Superoxide Dismutase and Metallothionein from Post-Mortem Human Frontal Cortex
Abstract
:1. Introduction
2. Results and Discussion
3. Materials and Methods
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Roberts, B.R.; Bush, T.M.; Masters, A.I.; Ryan, C.L.; Duce, J.A. The role of metallobiology and amyloid-β peptides in Alzheimer’s disease. J. Neurochem. 2012, 120, 149–166. [Google Scholar] [CrossRef] [PubMed]
- Johnson, C.M.; Beard, B.L. Biogeochemical cycling of iron isotopes. Science 2005, 309, 1025–1027. [Google Scholar] [CrossRef] [PubMed]
- Anbar, A. Iron stable isotopes: Beyond biosignatures. Earth Planet. Sci. Lett. 2004, 217, 223–236. [Google Scholar] [CrossRef]
- Zhu, X.K.; Guo, Y.; Williams, R.J.P.; O’Nions, R.K.; Matthews, A.; Belshaw, N.S.; Canters, G.W.; de Waal, E.C.; Weser, U.; Burgess, B.K.; et al. Mass fractionation processes of transition metal isotopes. Earth Planet. Sci. Lett. 2002, 200, 47–62. [Google Scholar] [CrossRef]
- Cadiou, J.L.; Pichat, S.; Bondanese, V.P.; Soulard, A.; Fujii, T.; Albarède, F.; Oger, P. Copper transporters are responsible for copper isotopic fractionation in eukaryotic cells. Sci. Rep. 2017, 7, 44533. [Google Scholar] [CrossRef] [PubMed]
- Aramendia, M.; Rello, L.; Resano, M.; Vanhaecke, F. Isotopic analysis of Cu in serum samples for diagnosis of Wilson’s disease: A pilot study. J. Anal. At. Spectrom. 2013, 28, 675–681. [Google Scholar] [CrossRef]
- Balter, V.; Nogueira da Costa, A.; Bondanese, V.P.; Jaouen, K.; Lamboux, A.; Sangrajrang, S.; Vincent, N.; Fourel, F.; Telouk, P.; Gigou, M.; et al. Natural variations of copper and sulfur stable isotopes in blood of hepatocellular carcinoma patients. Proc. Nat. Acad. Sci. USA 2015, 112, 982–985. [Google Scholar] [CrossRef] [Green Version]
- Costa-Rodriguez, M.; Anoushkina, Y.; Lauwens, S.; Van Vlierberghe, H.; Delanghe, J.; Vanhaecke, F. Isotopic analysis of Cu in blood serum by multi-collector ICP-mass spectrometry: A new approach for the diagnosis and prognosis of liver cirrhosis? Metallomics 2015, 7, 491–498. [Google Scholar] [CrossRef]
- Larner, F.; Woodley, L.N.; Shousha, S.; Moyes, A.; Humphreys-Williams, E.; Strekopytov, S.; Halliday, A.N.; Rehkämper, M.; Coombes, R.C. Zinc isotopic compositions of breast cancer tissue. Metallomics 2015, 7, 112–117. [Google Scholar] [CrossRef] [Green Version]
- Telouk, P.; Puisieux, A.; Fujii, T.; Balter, V.; Bondanese, V.P.; Morel, A.-P.; Clapisson, G.; Lamboux, A.; Albarède, F. Copper isotope effect in serum of cancer patients. A pilot study. Metallomics 2015, 7, 299–308. [Google Scholar] [CrossRef]
- Lauwens, S.; Costas-Rodrigues, M.; Van Vlierberghe, H.; Vanhaecke, F. Cu isotopic signature in blood serum of liver transplant patients: A follow up study. Sci. Rep. 2016, 6, 30683. [Google Scholar] [CrossRef] [PubMed]
- Albarède, F.; Telouk, P.; Lamboux, A.; Jaouen, K.; Balter, V. Isotopic evidence of unaccounted for Fe and Cu erythropoietic pathways. Metallomics 2011, 3, 926–933. [Google Scholar] [CrossRef] [PubMed]
- Jaouen, K.; Gibert, M.; Lamboux, A.; Telouk, P.; Fourel, F.; Albarède, F.; Alekseev, A.N.; Crubezy, E.; Balter, V. Is aging recorded in blood Cu and Zn isotope compositions? Metallomics 2013, 5, 1016–1024. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Heghe, L.; Engstrom, E.; Rodushkin, I.; Cloquet, C.; Vanhaecke, F. Isotopic analysis of the metabolically relevant transition metals Cu, Fe and Zn in human blood from vegetarians and omnivores using multi-collector ICP-mass spectrometry. J. Anal. At. Spectrom. 2012, 27, 1327–1334. [Google Scholar] [CrossRef]
- Van Heghe, L.; Deltombe, O.; Delanghe, J.; Depypere, H.; Vanhaecke, F. The influence of menstrual blood loss and age on the isotopic composition of Cu, Fe and Zn in human whole blood. J. Anal. At. Spectrom. 2014, 29, 478–482. [Google Scholar] [CrossRef]
- Fujii, T.; Moynier, F.; Telouk, P.; Fourel, F.; Abe, M. Experimental and theoretical investigation of isotope fractionation of zinc between aqua, chloro and macrocyclic complexes. J. Phys. Chem. A 2010, 114, 2543–2552. [Google Scholar] [CrossRef] [PubMed]
- Fujii, T.; Moynier, F.; Abe, M.; Nemoto, K.; Albarède, F. Copper isotope fractionation between aqueous compounds relevant to low temperature geochemistry and biology. Geochim. Cosmochim. Acta 2013, 110, 29–44. [Google Scholar] [CrossRef] [Green Version]
- Moynier, F.; Fujii, T.; Shaw, A.S.; Le Borgne, M. Heterogeneous distribution of natural zinc isotopes in mice. Metallomics 2013, 5, 693–699. [Google Scholar] [CrossRef]
- Navarette, J.U.; Borrock, D.M.; Viverous, M.; Ellzey, J.T. Copper isotope fractionation during surface adsorption and intracellular incorporation by bacteria. Geochim. Cosmochim. Acta 2011, 75, 784–799. [Google Scholar] [CrossRef] [Green Version]
- Pokrovsky, O.S.; Viers, J.; Emnova, E.; Kompantseva, E.I.; Freydier, R. Copper isotope fractionation during during its interaction with soil and aquatic microorganisms and metal oxy(hydr)oxides: Possible structural control. Geochim. Cosmochim. Acta 2008, 72, 1742–1757. [Google Scholar] [CrossRef]
- Weiss, D.J.; Mason, T.F.D.; Zhao, F.J.; Kirk, G.J.D.; Coles, B.J.; Horstwood, M.S.A. Isotopic discrimination of zinc in higher plants. New Phytol. 2004, 165, 703–710. [Google Scholar] [CrossRef] [PubMed]
- Tennant, A.; Rauk, A.; Wieser, M.E. Computational modelling of the redistribution of copper isotopes by proteins in the liver. Metallomics 2017, 9, 1809–1819. [Google Scholar] [CrossRef] [PubMed]
- Krizkova, S.; Kepinska, M.; Emri, G.; Rodrigo, M.A.M.; Tmejova, K.; Nerudova, D.; Kizek, R.; Adam, V. Microarray analysis of metallothioneins in human diseases—A review. J. Pharm. Biomed. Anal. 2016, 117, 464–473. [Google Scholar] [CrossRef] [PubMed]
- Fukai, T.; Ushio-Fukai, M. Superoxide dismutases: Role in redox signalling, vascular function and diseases. Antioxid. Redox Signal. 2001, 15, 1583–1606. [Google Scholar] [CrossRef] [PubMed]
- Rakhit, R.; Chakrabartty, R.A. Structure, folding, and misfolding of Cu, Zn superoxide dismutase in amyotrophic lateral sclerosis. Biochim. Biophys. Acta 2006, 1762, 1025–1037. [Google Scholar] [CrossRef]
- Ebadi, M.; Brown-Borg, H.; El Refaey, H.; Singh, B.B.; Garrett, S.; Shavali, S.; Sharma, S.K. Metallothionein-mediated neuroprotection in genetically engineered mouse models of Parkinson’s disease. Mol. Brain Res. 2005, 134, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Uchida, Y.; Takio, K.; Titani, K.; Ihara, Y.; Tomonaga, M. The growth inhibitory factor that is deficient in the Alzheimer’s disease brain is a 68 amino acid metallothionein-like protein. Neuron 1991, 7, 337–347. [Google Scholar] [CrossRef]
- Roberts, B.R.; Lim, N.K.H.; McAllum, E.J.; Donnelly, P.S.; Hare, D.J.; Doble, P.A.; Turner, B.J.; Price, K.A.; Lim, S.C.; Paterson, B.M.; et al. Oral treatment with Cu(II)(atsm) increases mutant SOD1 in vivo but protects motor neurons and improves the phenotype of a transgenic mouse model of amyotrophic lateral sclerosis. J. Neurosci. 2014, 34, 8021–8031. [Google Scholar] [CrossRef]
- Williams, J.R.; Trias, E.; Beilby, P.R.; Lopez, N.I.; Labut, E.M.; Bradford, C.S.; Roberts, B.R.; McAllum, E.J.; Crouch, P.J.; Rhoads, T.W.; et al. Copper delivery to the CNS by CuATSM effectively treats motor neuron disease in SOD(G39A) mice co-expressing the Copper-Chaperone-for-SOD. Neurobiol. Dis. 2016, 89, 1–9. [Google Scholar] [CrossRef]
- Chasapis, C.T.; Luotsidou, A.C.; Spiliopoulou, C.A.; Stefanidou, M.E. Zinc and human health: An update. Arch. Toxicol. 2012, 86, 521–534. [Google Scholar] [CrossRef]
- Tainer, J.A.; Getzoff, E.D.; Richardson, J.S.; Richardson, D.C. Structure and mechanism of copper, zinc superoxide dismutase. Nature 1983, 306, 284–287. [Google Scholar] [CrossRef] [PubMed]
- Buchl, A.; Hawkesworth, C.J.; Vala Ragnarsdottir, K.; Brown, D.R. Repartitioning of Cu and Zn isotopes by modified protein expression. Geochem. Trans. 2008, 9, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Harrington, C.F.; Vidler, D.S.; Watts, M.J.; Hall, J.F. Potential for using isotopically altered metalloproteins in species-specific isotope dilution analysis of proteins by HPLC coupled to inductively coupled plasma mass spectrometry. Anal. Chem. 2005, 77, 4034–4041. [Google Scholar] [CrossRef] [PubMed]
- Westergard, L.; Christensen, H.M.; Harris, D.A. The cellular prion protein (PrPc): Its physiological function and role in disease. Biochim. Biophys. Acta 2007, 1772, 629–644. [Google Scholar] [CrossRef] [PubMed]
- Larner, F.; Rehkämper, M.; Coles, B.J.; Kreissig, K.; Weiss, D.J.; Sampson, B.; Unsworth, C.; Strekpytov, S. A new separation procedure for Cu prior to stable isotope analysis by MC-ICP-MS. J. Anal. At. Spectrom. 2011, 26, 1627–1632. [Google Scholar] [CrossRef]
- Moeller, K.; Schoenberg, R.; Pedersen, R.-B.; Weiss, D.; Dong, S. Calibration of the new certified reference materials ERM-AE633 and ERM-AE647 for copper and IRMM-3702 for zinc isotope amount ratio determinations. Geostand. Geoanal. Res. 2012, 36, 177–199. [Google Scholar] [CrossRef]
- Hare, D.J.; Doecke, J.D.; Faux, N.G.; Rembach, A.; Volitakis, I.; Fowler, C.J.; Grimm, R.; Doble, P.A.; Cherny, R.A.; Masters, C.L.; et al. Decreased plasma iron in Alzheimer’s disease is due to transferrin desaturation. ACS Chem. Neurosci. 2015, 6, 398–402. [Google Scholar] [CrossRef]
- Rembach, A.; Hare, D.J.; Doecke, J.D.; Burnham, S.C.; Volitakis, I.; Fowler, C.J.; Cherny, R.A.; McLean, C.; Grimm, R.; Martins, R.; et al. Decreased serum zinc is an effect of ageing and not Alzheimer’s disease. Metallomics 2014, 6, 1216–1219. [Google Scholar] [CrossRef]
- Krężel, A.; Maret, W. The functions of metamorphic metallothioneins in zinc and copper metabolism. Int. J. Mol. Sci. 2017, 18, 1237. [Google Scholar] [CrossRef]
- Roberts, B.R.; Hare, D.J.; McLean, C.A.; Conquest, A.; Lind, M.; Li, Q.X.; Bush, A.I.; Masters, C.L.; Morganti-Kossmann, M.C.; Frugier, T. Traumatic brain injury induces elevation of Co in the human brain. Metallomics 2015, 7, 66–70. [Google Scholar] [CrossRef] [Green Version]
- Lothian, A.; Roberts, B.R. Standards for quantitative metalloproteomic analysis using size exclusion ICP-MS. J. Vis. Exp. 2016, 110, 53737. [Google Scholar] [CrossRef] [PubMed]
- Larner, F.; Dogra, Y.; Dybowska, A.; Fabrega, J.; Stolpe, B.; Bridgestock, L.J.; Goodhead, R.; Weiss, D.J.; Moger, J.; Lead, J.R.; et al. Tracing bioavailability of ZnO nanoparticles using stable isotope labeling. Environ. Sci. Technol. 2012, 46, 12137–12145. [Google Scholar] [CrossRef] [PubMed]
- Larner, F.; Sampson, B.; Rehkämper, M.; Weiss, D.J.; Dainty, J.R.; O’Riordan, S.; Panetta, T.; Bain, P.G. High precision isotope measurements reveal poor control of copper metabolism in Parkinsonism. Metallomics 2013, 5, 125–132. [Google Scholar] [CrossRef] [PubMed]
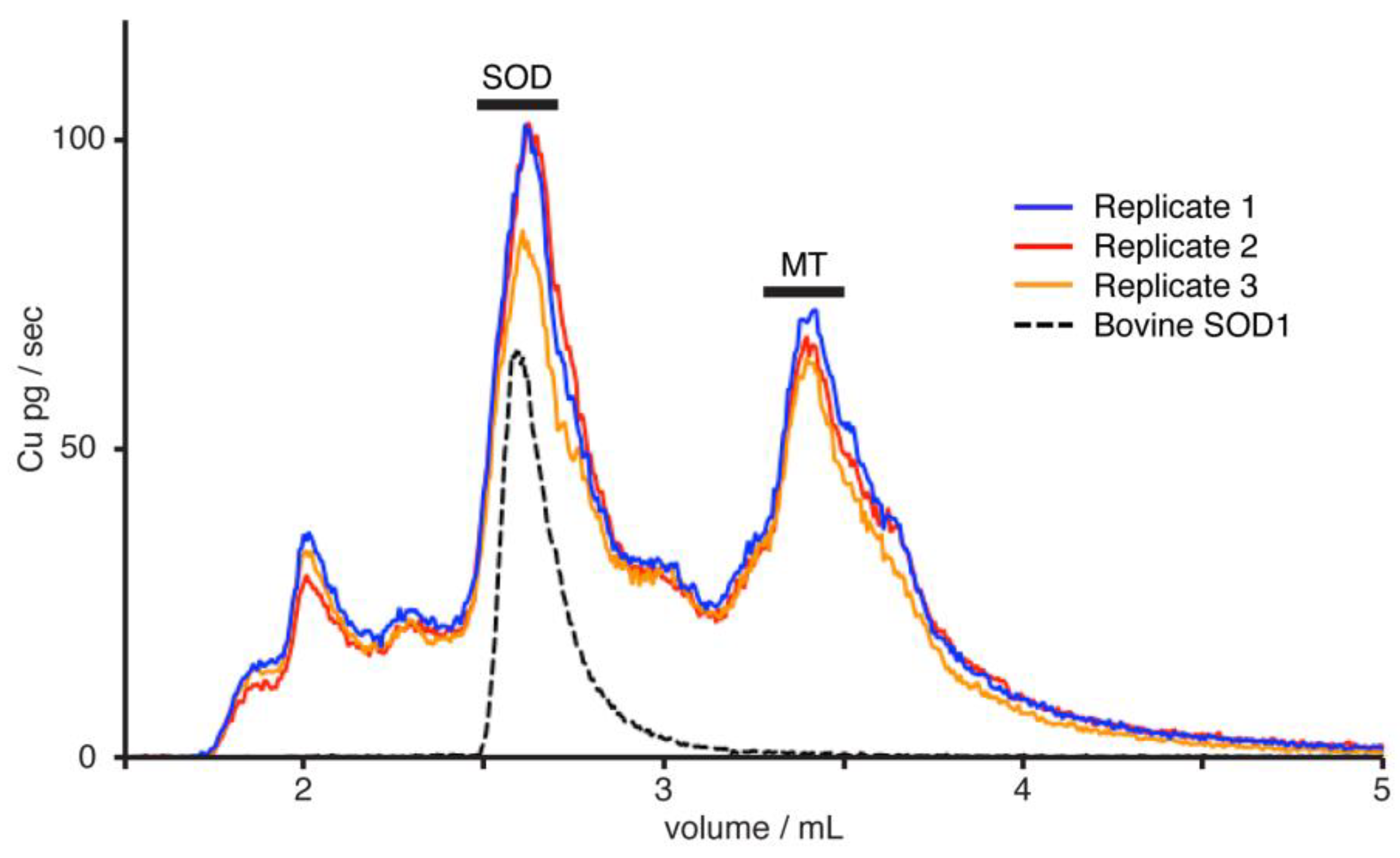

| Sample | Sex | Total CuSOD (ng) | δ65CuN (‰) 4 | Total CuMT (ng) | δ65CuS (‰) 4 | Δ65CuN–S (‰) 4 | Source |
|---|---|---|---|---|---|---|---|
| AD1 1,2 | Male | 180 | 0.34 | 43 | −0.25 | 0.59 | this study 5 |
| AD2 1 | Male | 86 | 0.36 | 30 | −0.08 | 0.44 | 5 |
| AD3 1 | Female | 40 | −0.33 | 27 | −0.39 | 0.06 | 5 |
| CON1 | Male | 83 | 0.37 | 26 | −0.42 | 0.79 | 5 |
| CON2 3 | Female | 48 | 0.31 | 47 | 0.14 | 0.17 | 5 |
| YeastSOD–MT | 0.53 | [4] | |||||
| RBCs–serum | Female | 0.46 | −0.24 | 0.60 | [12] | ||
| RBCs–serum | Male | 0.67 | −0.28 | 0.95 | [12] |
| Sample | Total Cu (ng) | δ65Cu (‰) | Δ65Cu (‰) |
|---|---|---|---|
| Bulk Brain | 635 | −0.09 | |
| Metallothionein | |||
| Replicate 1 | 43 | −0.17 | |
| Replicate 2 | 37 | −0.21 | |
| Replicate 3 | 45 | −0.37 | |
| Mean (SD) | −0.25 (0.11) | ||
| Superoxide dismutase | |||
| Replicate 1 | 168 | 0.50 | |
| Replicate 2 | 130 | 0.17 | |
| Replicate 3 | 166 | 0.34 | |
| Mean (SD) | 0.34 ** (0.17) | ||
| SOD–MT Pair 1 | 0.67 | ||
| SOD–MT Pair 2 | 0.38 | ||
| SOD–MT Pair 3 | 0.73 | ||
| Mean (SD) | 0.59 (0.17) |
| Sample Code | Disease State of Brain | Sex | Age | PMI 1 |
|---|---|---|---|---|
| AD1 2 | Alzheimer’s disease | Male | 70 | |
| AD2 2 | Alzheimer’s disease | Male | 75.4 | 18.5 |
| AD3 2 | Alzheimer’s disease | Female | 76.8 | 31.0 |
| CON1 | Healthy | Male | 76.9 | 49.0 |
| CON2 3 | Healthy | Female | 76.8 3 | 40.3 3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Larner, F.; McLean, C.A.; Halliday, A.N.; Roberts, B.R. Copper Isotope Compositions of Superoxide Dismutase and Metallothionein from Post-Mortem Human Frontal Cortex. Inorganics 2019, 7, 86. https://doi.org/10.3390/inorganics7070086
Larner F, McLean CA, Halliday AN, Roberts BR. Copper Isotope Compositions of Superoxide Dismutase and Metallothionein from Post-Mortem Human Frontal Cortex. Inorganics. 2019; 7(7):86. https://doi.org/10.3390/inorganics7070086
Chicago/Turabian StyleLarner, Fiona, Catriona A. McLean, Alex N. Halliday, and Blaine R. Roberts. 2019. "Copper Isotope Compositions of Superoxide Dismutase and Metallothionein from Post-Mortem Human Frontal Cortex" Inorganics 7, no. 7: 86. https://doi.org/10.3390/inorganics7070086