Abstract
The reaction of the bulky ligand 1,2-bis-(di-tert-butylphosphinomethyl)benzene, 1 with [Ni(DME)Cl2], 3, DME = 1,2-dimethoxyethane, at room temperature over extended periods, affords the new blue Zwitterionic complex [2-(C6H4-CH2P(H)tBu2-1-(CH2PtBu2NiCl3)], 4, which contains a phosphonium group and an anionic nickel trichloride. This complex decomposes in alcohols such as methanol and the solution turns yellow. A discussion of the possible mechanism leading to the observed product is presented. Key to this is identification of the source of the phosphonium proton, which we speculated to arise from trace water in the initial nickel complex. To prove that trace water was present in [Ni(DME)Cl2], a sample of this precursor was reacted under similar condition with anhydrous DMF alone. In addition to the known complex [Ni(DMF)6)]2+[NiCl4]2−, 5, we identified the trans-diaqua complex [Ni(Cl)2(H2O)2(DMF)2], 6, which proved the presence of trace water. Interestingly in dimethylformamide, [2-(C6H4-CH2P(H)tBu2-1-(CH2PtBu2NiCl3)] exhibits thermochromic properties: an solution that is pale blue at ambient temperature reversibly changes colour to yellow upon cooling. This behaviour is specific to DMF and is related to the solvato-chromic behaviour exhibited by related DMF–nickel complexes. A discussion of the NMR spectra of compound 4 in a range of solvents is presented. The structures of the previously prepared molybdenum complex, [1,2-(C6H4-CH2PtBu2)2Mo(CO)4] and the bis-(phosphine sulphide) of the ligand, [1,2-(C6H4-H2P(S)tBu2)2], 5, are described for structural comparative purposes.
Keywords:
metal complex; Zwitterion; nickel; thermochromic; phosphine; alpha; complexation; crystal; structure; DMF; self-assembly; synthesis 1. Introduction
The coordination chemistry of nickel (II) dichlorides with bidentate phosphines is one of the most studied areas of co-ordination chemistry. They present varying coordination geometries [1,2,3] between the square planar and tetrahedral [4,5] and are used as a vehicle to teach the differences between high- and low-spin coordination complexes [6,7]. In most cases, the colour of a metal complex is enough to determine whether the complex is low- or high-spin: a larger energy gap (low-spin) means the colour of the complex is complementary, i.e., there are blue or green wavelengths and no yellow, orange, or red. Some complexes, such as [Ni(PPh3)2Cl2], exist in both coordination modes depending on the solvent in which they were prepared [8]. In rare cases, a complex may be observed to be a Zwitterion, which is the subject of this work. The ligand we used was 1,2-bis-(di-tbutylphosphinomethyl)benzene, [9,10] 1, which is the classic so-called alpha ligand [11]. This ligand was first reported and developed by Pringle’s group, and its synthesis and use remain seminal in the field of catalysis [12]. We, and other, have extensive experience in the coordination of these ligands, particularly the ferrocene class [13,14,15]. The palladium coordinates with the phosphorus atoms of the ferrocene alpha ligand, butphos, 2, in a square planar fashion, Figure 1 [13]. The side-on views show that the ligand arms flank the palladium in a sterically crowded environment.
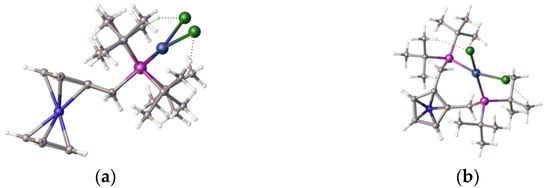
Figure 1.
(a) Side and top (b) views of the crystal structure of [{1,2-η5-C5H3(CH2PtBu2)2}Fe(η5-C5H5)PdCl2], [(butphos)PdCl2]*, showing the square planar coordination mode [13], (solvent omitted). Atom colour coding: dark blue, palladium; purple, phosphorus; pale blue, iron; green, chlorine.
As part of a model study, it was decided to initially examine the complexation reaction of nickel with a simple alpha ligand. Clearly, this is a very important research activity because the development of a nickel-based catalyst of either the alpha ligand or the butphos ligand for acrylic production would be much cheaper than the current technology. Much is known about the similar square planar coordination chemistry of ligand 1 with palladium and platinum, as these complexes are useful industrial catalysts, [16,17], but less is known about nickel [18].
2. Results and Discussion
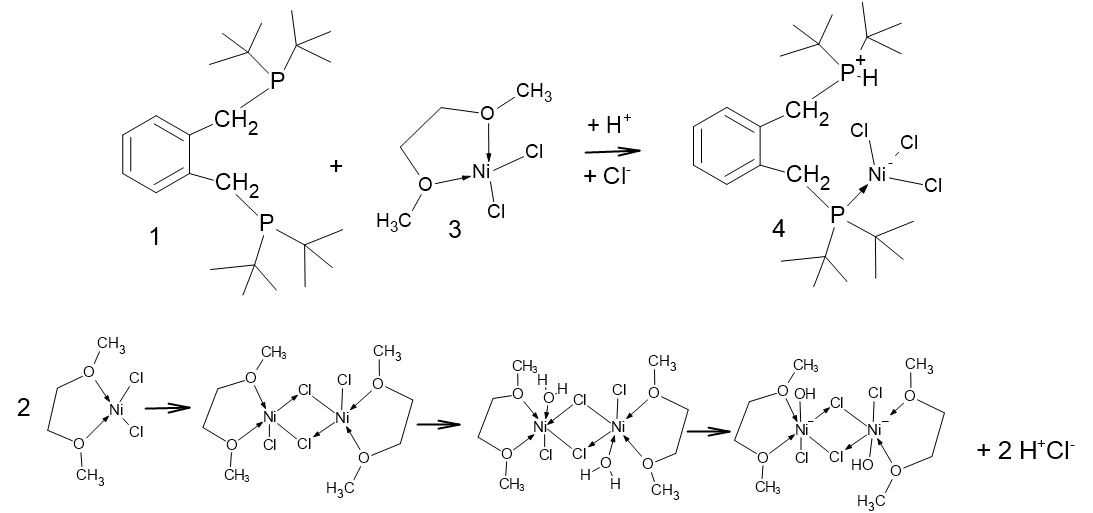
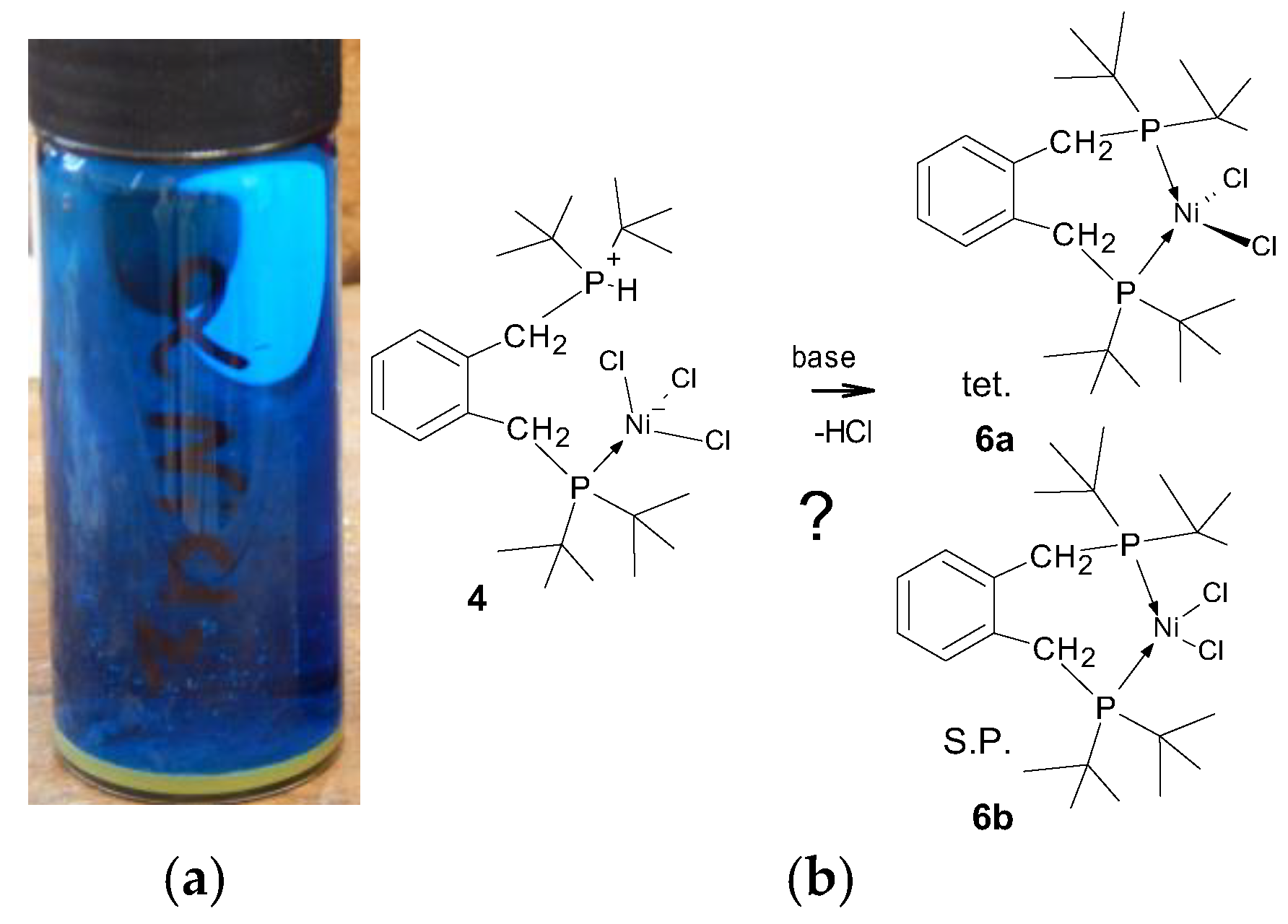
With this information in hand, the reaction of 1,2-bis-(di-tert-butylphosphinomethyl)benzene, 1 with hydrated nickel chloride in alcoholic solvents, under reflux, was attempted, but this led only to amorphous yellow powders. However, when a solution of the ligand was added to the [Ni(DME)Cl2], 3, (DME = 1,2-dimethoxyethane) complex [19] in dichloromethane, the solution developed a blue colour and, when left for several days, blue crystals began to grow. We had anticipated the formation of red crystals, which would have indicated the formation of the square planar complex, or deep blue/green crystals, which would indicate a tetrahedral complex. The pale blue crystals were collected after a minimum of 2 weeks and examined by single-crystal diffraction; they were a rare Zwitterionic compound, which accounts for their poor solubility in organic solvents. The product complex was identified as compound 4, as shown in Figure 2.
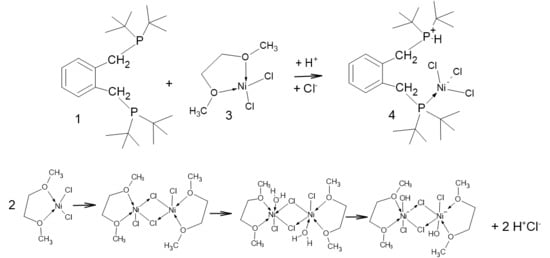
Figure 2.
Schematic showing the formation of [2-(C6H4-CH2P(H)tBu2-1-(CH2PtBu2NiCl3)], 4. Crucial to this complexation is the addition of a proton and a chloride ion (i.e., HCl). In the lower graphic, one of the many possible modes of formation of HCl is shown; the key to this is the reaction of water with [Ni(DME)Cl2], 3.
Only one phosphorus is bound to nickel, with three chloride ligands attached in a pseudo-tetrahedral coordination, as shown in Figure 3, while the other phosphorus is protonated, forming a phosphonium pendant arm.
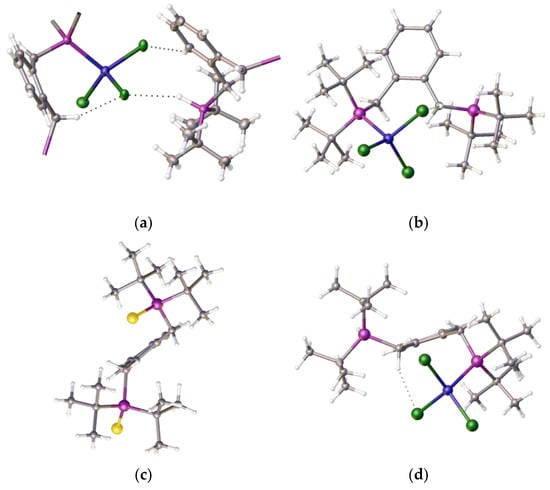
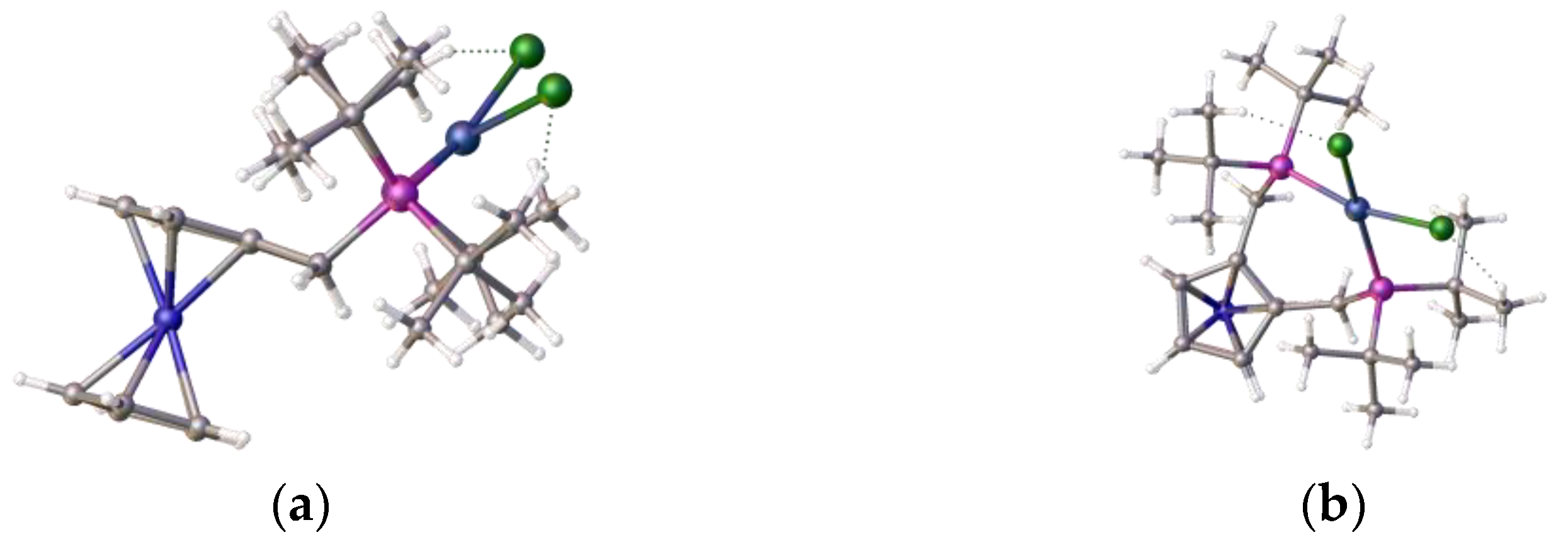
Figure 3.
Views of the crystal structure of [2-(C6H4-CH2P(H)tBu2-1-(CH2PtBu2NiCl3)], 4 regarding the benzene ring: (a) cutaway showing the P-H…Cl and C-H…Cl interaction between neighbouring molecules; (b) top view (c) comparison between the ligand, shown as disulfide, [1,2-(C6H4-H2P(S)tBu2)2], 5, with compound 4 (d) showing the pendant arms above and below the benzene ring plane. Atom colour coding: blue, nickel; purple, phosphorus; green, chlorine; yellow, sulfur.
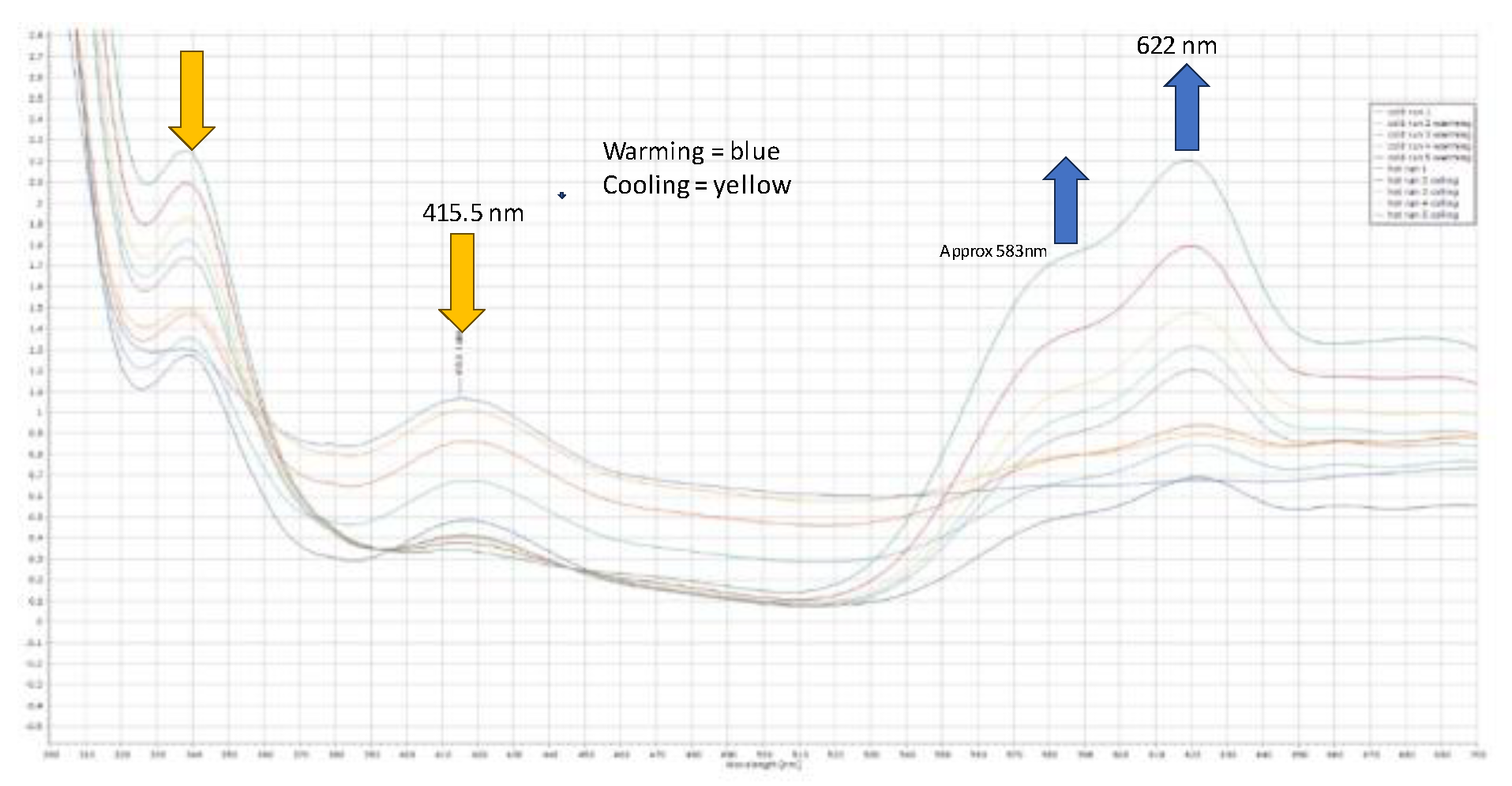
The Cl-Ni-Cl angles were 101.011(32), 120.882(32), and 109.770(29) degrees, respectively, when there were two close contacts between a chloride and proton on the phosphorus (2.7091(3) Å) and a chloride and a proton on the phenyl ring (2.794(7) Å), along with the internal close contact (Figure 3a) between a chloride and methylene proton (2.4747(9) Å). In the structure, the pendant arms of the ligand lie above and below the benzene ring; the same orientation was observed in both the free ligand and its disulfide, which is shown, for comparison, in Figure 3c. The origin of the additional proton and the additional chloride on nickel is unclear, but we propose that the chloride ion is likely to originate from the excess nickel complex and that the proton probably originates from trace water, as the starting nickel complex is highly hygroscopic. The product complex decomposes in methanol and forms a pale yellow solution/slurry. It is sparingly soluble in chloroform, dichloromethane, tetrachloroethane, and DMF, and forms a pale blue solution at room temperature. Upon cooling the solution in DMF to −20 °C, the blue colour (λmax, ~621 nm) reversibly changes to yellow (λmax, ~417 nm) (see Figure 4). It is likely that DMF reacts reversibly with the nickel centre as it is a hard Lewis base with a low pKa. Interestingly,[2-(C6H4-CH2P(H)tBu2-1-(CH2PtBu2NiCl3)] ] is a complex that crystallizes well, in a regimented fashion, in local magnetic fields, as was examined in a separate study we were involved in [20].
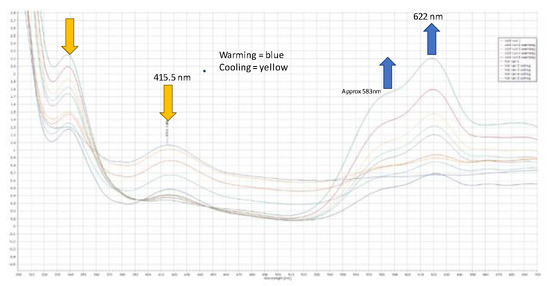
Figure 4.
Compound 4 saturated solution in dmf; U.V., V.T. spectra, warming above and below ambient temperature and showing a thermochromic nature, changing from blue to yellow.
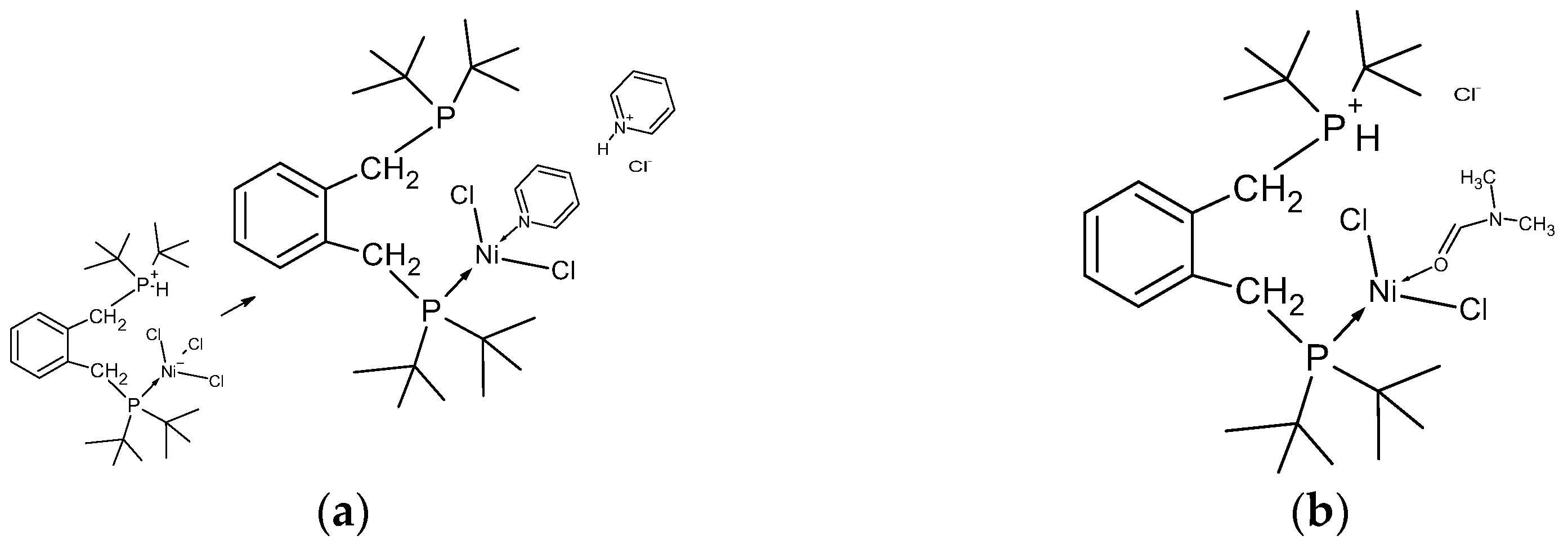
When looking at the structure, it might be imagined that compound 4 would react under basic conditions to produce either a square planar, 6a, or tetrahedral, 6b, complex, as shown in Figure 5; hence, a closer examination by NMR is warranted. Excepting the obvious steric crowding, it is not immediately clear why one of these chelate complexes does not form under the experimental conditions, since we have successfully used this synthetic method for the preparation of related nickel phosphine complexes [21].
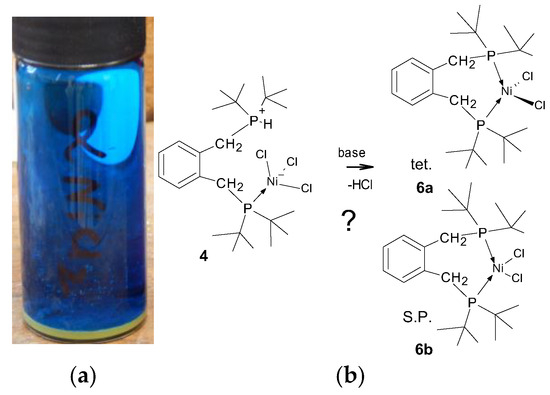
Figure 5.
(a) Mid-blue crystals of compound 4 forming on vial wall, with the yellow/ochre powder of compound 3 on the bottom; (b) schematic showing that the deprotonation of compound 4 with a base to obtain the chelate complex(es) should be possible.
2.1. Structural Comparisons
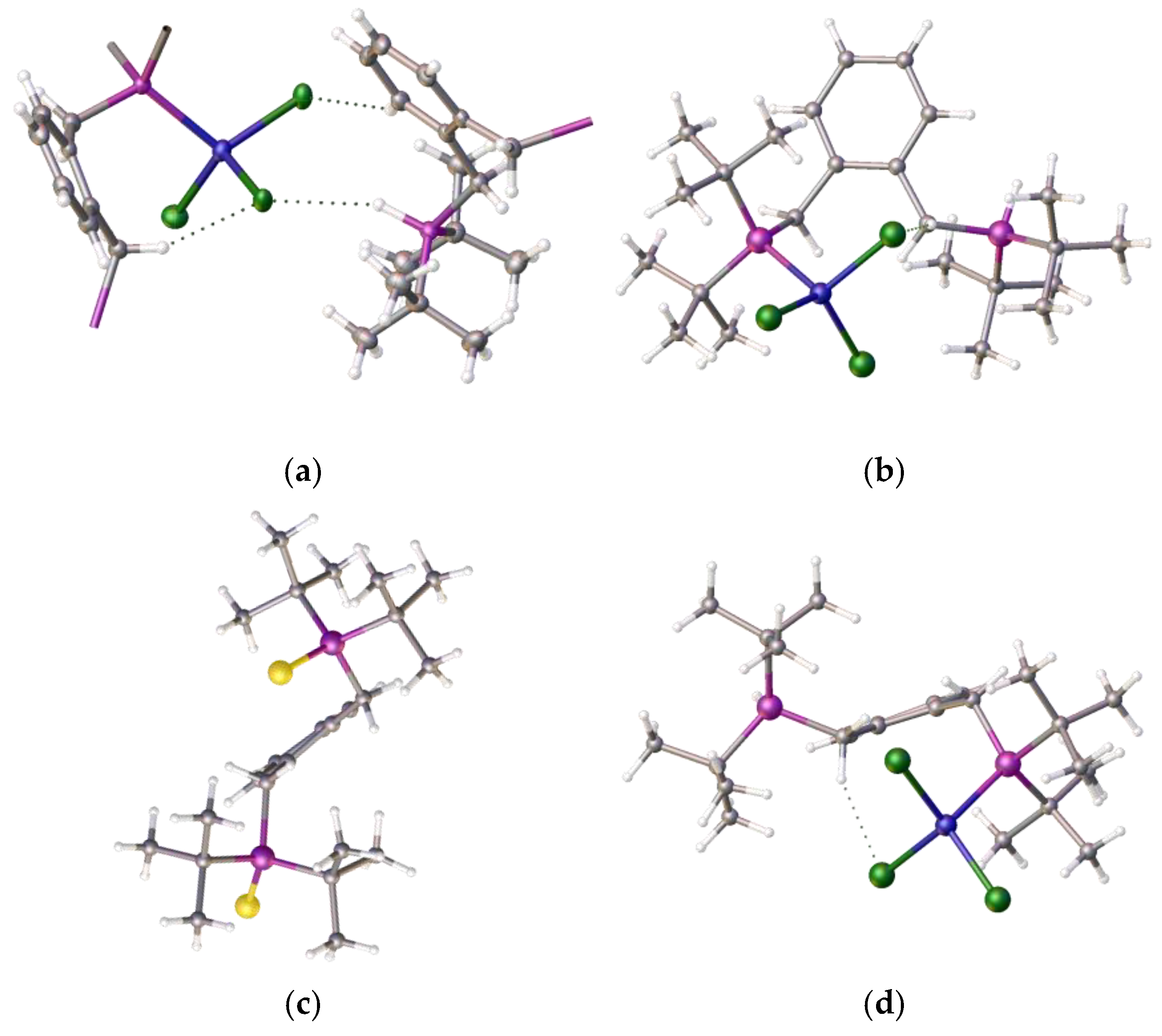
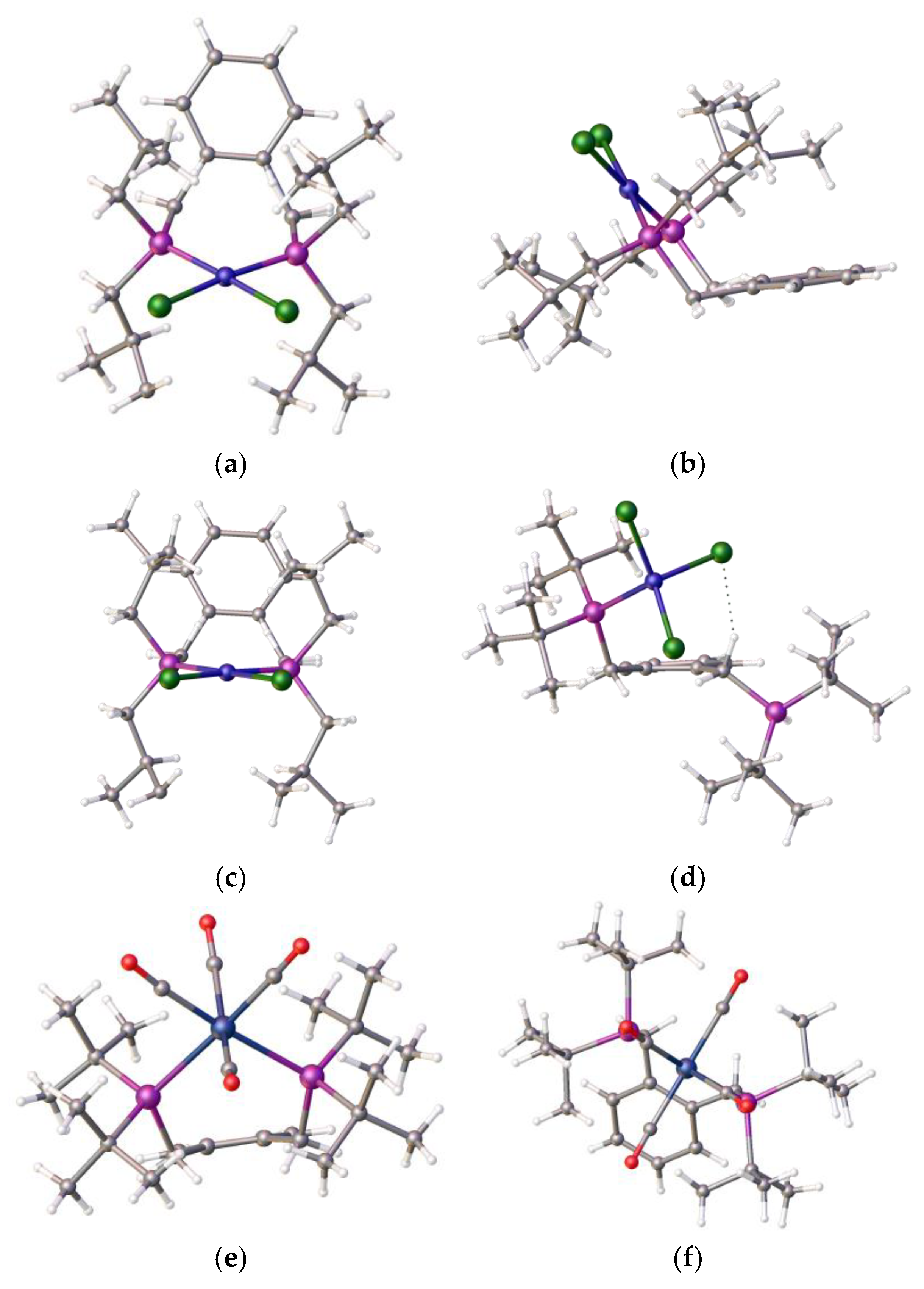
To understand the reason for the formation of compound 4 and not the chelate complex, we decided to compare the product structure with related structures within our portfolio. During these coordination studies, we obtained non-definitive evidence for the formation of the anticipated square–planar Ni–alpha complex as small red crystals that were isolated during one of our room-temperature coordination reactions; however, the initial crystallographic results indicated a disordered structure. However, we note that we were able to determine the structure of the isomeric red nickel dichloride complex, 7, of the related isomeric ligand [1,2-C6H4-(CH2PiBu2)2]. The structure is disordered but it clearly shows that the nickel adopts a square planar coordination mode, as shown in Figure 6a–c.
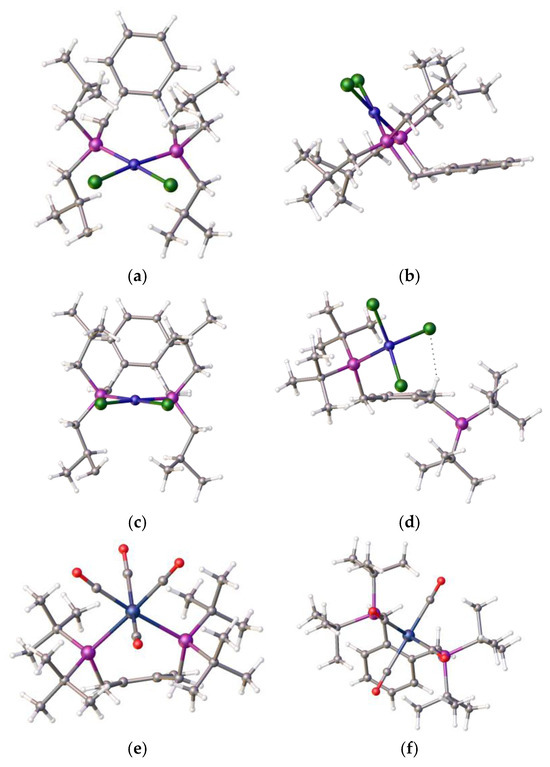
Figure 6.
(a) Top and (b) side views of the nickel dichloride complex [(1,2-C6H4-(CH2PiBu2)2NiCl2], 7, solvent omitted, prepared from ligand and compound 3. (c,d) Side-by-side comparison of compounds 7 and 4, highlighting the difference in the pendant arms. (e,f) An example of ligand 1 in chelate coordination with a tungsten tetracarbonyl moiety Atom colour coding, (a–d): blue, nickel; green, chlorine; purple, phosphorus; (e,f): blue, tungsten, purple, phosphorus; red, oxygen. (CCDC 2421627: full details in Supplementary Materials).
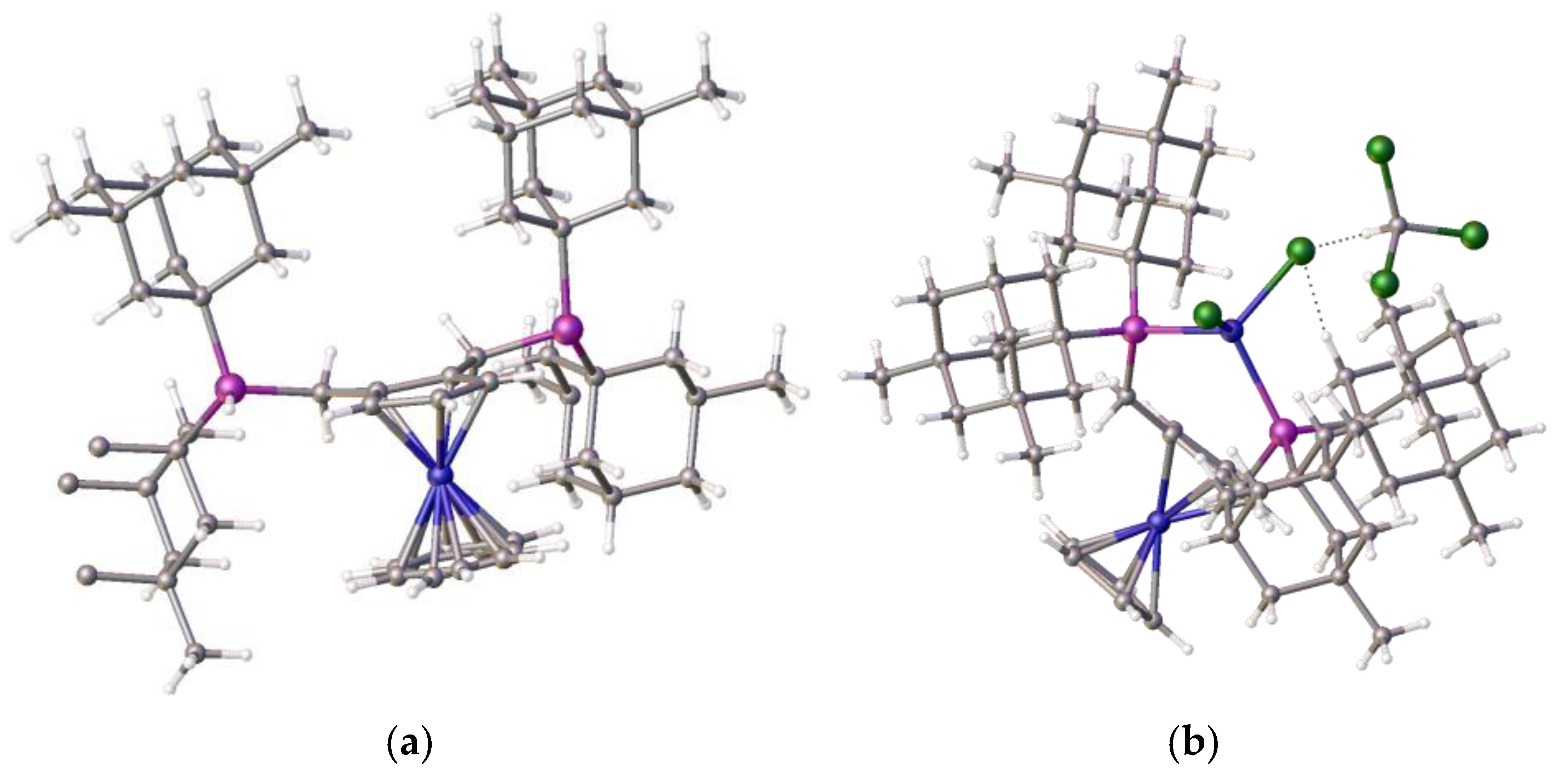
This ligand with pendant iso-butylphosphine arms is less hindered than its tert-butylphosphine isomer, which partially accounts for the difference in coordination properties, and therefore its relative ease of formation. However, this cannot be the only reason for the observed differences. An examination of the structure of the tungsten tetracarbonyl complex of ligand 1 [22], as shown in Figure 6e,f, visually highlights a possible reason for this: the bond length of the nickel–phosphine bond is shorter compared to the those of the palladium-P or tungsten-P, and consequently, the tighter chelate bond angles of the P-Ni-P prevent coordination. Interestingly, we previously observed structural disorder in the chloroform solvate of the nickel dichloride complex of the bis-1,2-[(1,3-dimethyladamantyl)phosphino]ferrocene, 8, which exhibited tetrahedral coordination compared with its palladium analogue, presumably, in part, because of the shorter bond length with the very sterically bulky ligand, as shown in Figure 7.
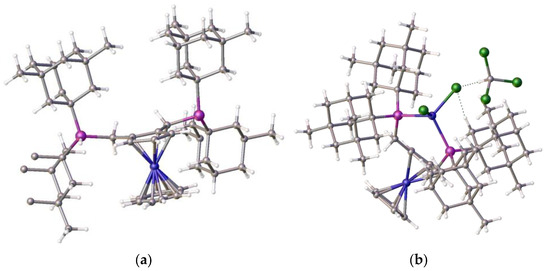
Figure 7.
(a) The 1,2-bis(dimethyladamantylphosphino)ferrocene ligand, 8 (fragments removed to show structure more clearly) and (b) its nickel dichloride complex, 9, chloroform solvate. This exhibits tetrahedral coordination, as well as the hydrogen bonding between the ligand and a nickel-chloride [13]. Atom colour coding: deep blue, nickel; purple, phosphorus; mid blue, iron; green, chlorine.
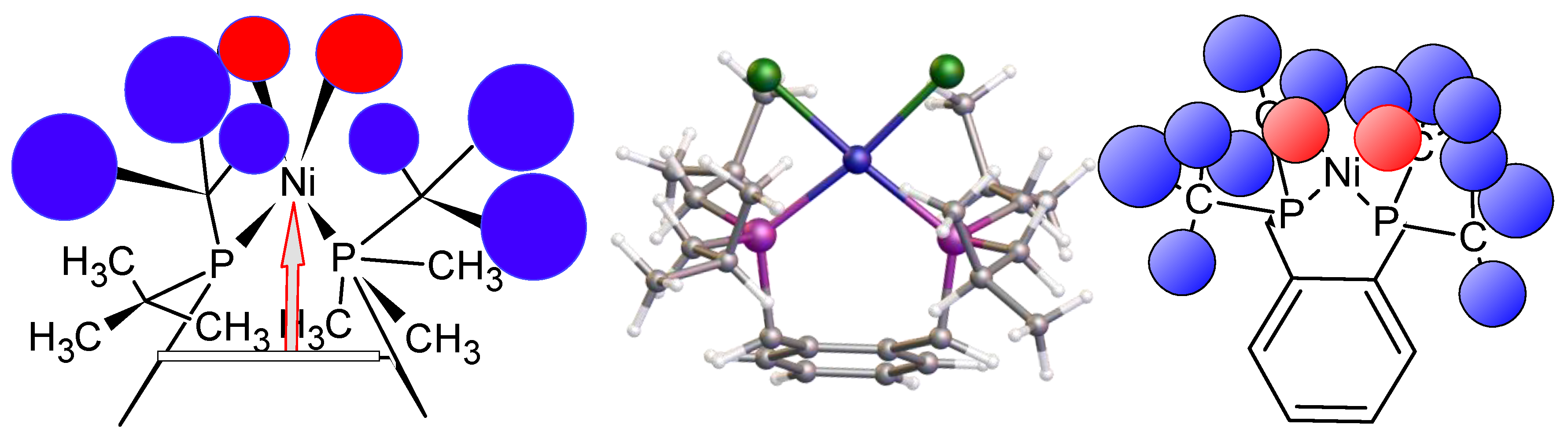
This, of course, leads to the question “why does tetrahedral coordination not occur if square planar coordination is not possible due to steric crowding?” The answer is not immediately obvious but it may simply be that the mild reaction conditions used or the shorter length of the nickel phosphine bond hinder the formation of the bidentate product. Structural comparisons between nickel and palladium complexes are complex [23,24], and clearly an extended study of a range of related ligands is warranted. Clearly, the geometric parameters around the metal are important and will affect any catalytic reaction [25], as shown in Figure 8. To highlight the importance of these structures, a recent study used a chiral variant of this ligand class in the asymmetric hydrogenation of α-substituted acrylic acids to yield chiral alpha-substituted propionic acids. In this case, the catalyst was prepared in situ using nickel acetate as a precursor [26]. The next question we need to address is the source of the trace water required in the synthesis, which is examined next.
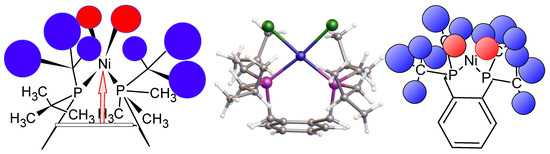
Figure 8.
(Left) Front view shows the importance of the distance of the nickel from the benzene ring in the formation of the chelate complex of 1 (distance shown as an arrow). (Middle) A partially congested structure of complex 7. (Right) The top view, of either 6a or 6b, were it to form, indicating the steric congestion present above the benzene ring, which impacts the chlorine (red spheres) environment due to its geometry.
2.2. Examination of the Reaction of DMF with in [Ni(DME)Cl2], Looking for Trace Water
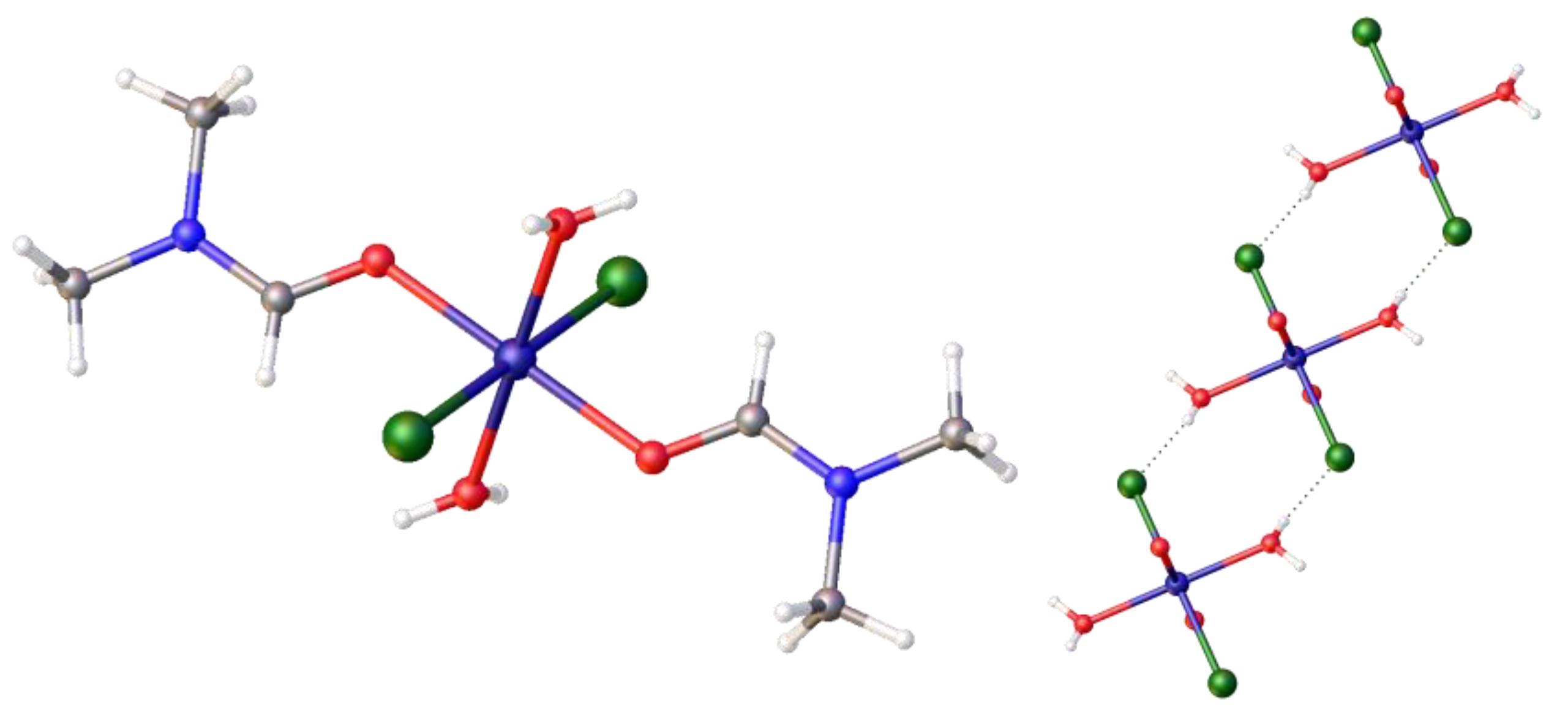
As of yet, we have not proven that there is trace water present in the precursor, [Ni(DME)Cl2], which could act as the source of the proton in the Zwitterionic complex. Therefore, we decided to examine the reaction of DMF with [Ni(DME)Cl2] alone. It is known that complex equilibria exist in DMF solutions of nickel chlorides, and thus DMF binding to nickel may occur. This will also influence the observed thermochromic properties of the product. Two complexes were formed in this room-temperature reaction using anhydrous DMF. The first was the previously reported octahedral hexadimethylformaldehydo-nickel tetrachloronickelate, [Ni (DMF)6)]2+[NiCl4]2−, 10, [27], which we crystallographically characterized (CCD Deposition Number: 2421634, full data presented in the Supplementary Materials). This confirms that DMF readily coordinates with the nickel centre (to yield [Ni (DMF)6)]2+), and it also confirms that chloro-ligands are transferred, under ambient conditions, between nickel centres (to yield [NiCl4]2−) in DMF, thus confirming the source of the additional chloride. The second complex that was identified was the all trans-di-aqua-bis-dimethylformaldehydo-dichloro nickel (II), [Ni(H2O)2(DMF)2 Cl2], 11, as shown in Figure 9, left, (2017ncs0366c; CCDC 2421635). A detailed literature search indicated this complex was published elsewhere after completion of this work, as [Ni(DMF)2(H2O)4]Cl2·2H2O [28]. This complex exists as hydrogen-bonded ribbons; cutaway sections of its packing diagram are also shown in Figure 9, right. This complex completes a series of aqua nickel complexes, as the related nickel (II), trans-[NiCl2(H2O)4]·2H2O, had been previously reported [29]. More importantly for this work, it confirms the presence of trace water in the precursor. Thus, we established the likely sources of the additional H+ and Cl−.
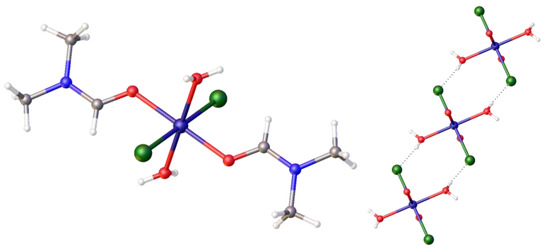
Figure 9.
The crystal structure of the diaqua- complex, trans-[Ni(Cl)2(H2O)2(DMF)2], 11, formed from [Ni(DME)Cl2], and DMF at room temperature, and its partial packing diagram showing the hydrogen bonding between molecules. Atom colour coding: deep blue, nickel; green chlorine; red, oxygen; pale blue, nitrogen.
2.3. NMR Spectroscopic Examination of Compound 4
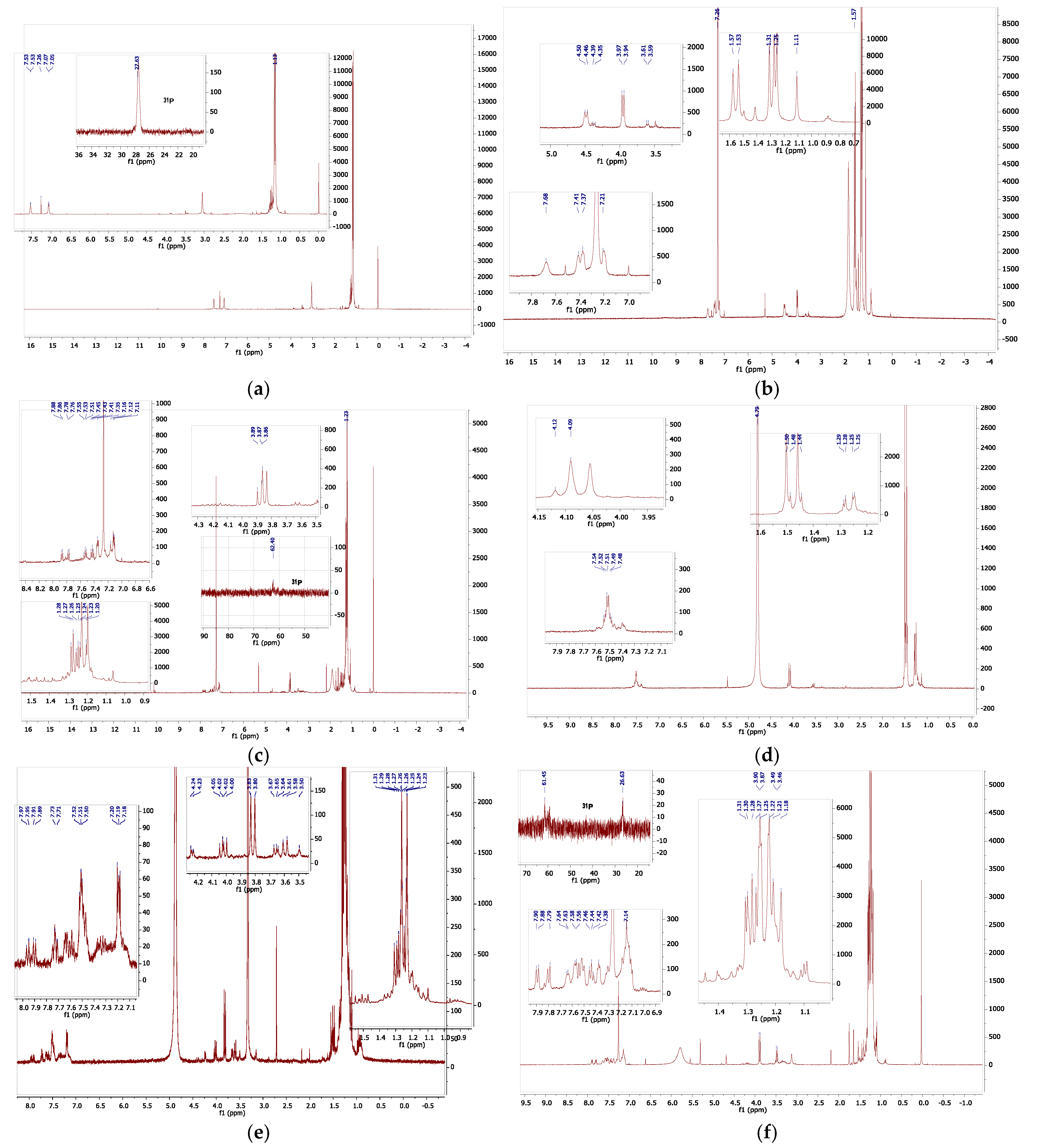
To obtain further clarification, the proton and phosphorus NMR spectra of the complex were examined in a range of solvents. In some solvents, only broad resonance was observed, which generally meant that there were equilibria involving the solvent, and in other solvents there was a clear complex decomposition; nevertheless, some useful data were obtained, which are visually summarized in Figure 10. The low solubility in all solvents studied hindered the measurements, but general conclusions may be drawn. Solvents, which themselves can act as bases or ligands, interact with compound 4 in a more complex fashion. This is not surprising as we already observed thermochromic behaviour in DMF. In all cases, there is considerable resonance overlap in the aryl region and in the alkyl–methyl regions of the spectra; however, the methylene region (approx. 2.5–4.5 ppm) is relatively clear of additional resonances and is a useful diagnostic. The protons of the -CH2− spacer in the ligand were observed at ca 3.4 ppm, while those of the mono-protonated ligand, which we generated independently (ligand 1 was treated with 1,1,2,2-tetrabromoethane, which generates HBr in situ in the presence of alcohols, amine, and phosphines [30]), were observed at ca 4.5 ppm. In chloroform-d, two groups of methylene proton were observed: the first at 3.96 ppm, which we assigned to those on the nickel-bonded arm, and the second at 4.48 ppm, (slightly broader), which we assigned to those on the protonated phosphorus arm. The tert-butyl resonances of these were observed at 1.29 and 1.55 ppm, respectively. The four aryl resonances were observed between 7.2 and 7.7 ppm. To observe the decomposition, a sample in chloroform was treated with D2O. In this case, there were two methylene resonances that overlapped at 3.86 and 3.88 ppm. In addition, there were two sets of very-low-intensity multiplets at ca 3.3 and 4.3 ppm. All methyl resonances overlapped between 1.1 and 1.3 ppm. In the aryl region, there appeared to be two groups of resonance: one set from an unsymmetrically substituted benzene ring (2 triplets, 2 doublets) and a second set of multiplet resonances at 7.12 and 7.35 ppm. In the phosphorus NMR, there were again two sets of resonances: one corresponding to that of the ligand at ca 27 ppm and one set at ca 61.5 ppm. It is clear there is partial deligation upon the addition of D2O, but new products are also formed. In neat D2O, the spectra are complex: there are at least six methylene resonances (and therefore at least three compounds) and several overlapping aryl resonances, at 7.1–7.6 ppm, while all tert-butyl resonances are overlapped. In deuterated methanol, a similar decomposition was observed. In this case, the one weak phosphorus resonance was observed at 65.4 ppm. In the case of the diagnostic methylene resonances, there was one large doublet (3.82 ppm, symmetrical compound), two smaller pairs of doublets (3.62, 4.01 ppm; 3.66, 4.04 ppm), both unsymmetrical compounds with very-low-intensity resonance (dd, 4.23 ppm). In the 31P NMR spectrum, only very-low-intensity resonances were observed; therefore, a separate, long-acquisition-time experiment was conducted.
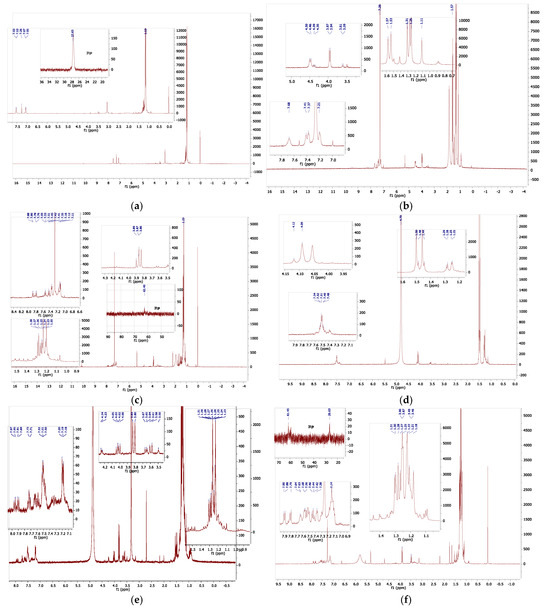
Figure 10.
(a) 1H NMR of compound 1 with 31P NMR inset (b) 1H NMR of compound 4 in CDCl3. (c) 1H NMR of compound 4 in wet (D2O) CDCl3,, with resonance expansions.(d) Compound 4 in D2O. (e) Compound 4 in methanol-d4. (f) 1H NMR of compound 4 in CDCl3 with added pyridine-d5, with 31P NMR inset. For additional information please see the Supplementary Materials section.
This led to the observation of a high-intensity singlet resonance at 66.6 ppm, a lower-intensity 1:1:1 triplet at 64.36 ppm (JP-D = 182 Hz), and several low-intensity singlets at 65.04, 64.20, 58.46, and 42.17 ppm, respectively. These were all sharp resonances, whereas a broader, yet significant, 1:1:1 triplet was observed at 38.79 ppm (JP-D = 70 Hz). These data show that significant proton/deuterium exchange is occurring in the sample. In one last sample, pyridine-d5 was added to a sample of compound 4 in chloroform-d; this was because the pyridine should deprotonate the phosphonium phosphorus. Two large methylene resonances were observed at 3.47 and 3.89 ppm, respectively, together with a series of low-intensity multiplets which centred at 3.3 and 4.2 ppm. Thus, the deprotonation leads to complex spectrum results. From these data, it may be concluded that the compound 4 reacts readily with solvents, which themselves may act as ligands. The chloro-ligands may be replaced or augmented with solvent. In DMF, it is likely that the DMF completely and reversibly solvates the nickel centre akin to the preparation of compound 11, shown earlier. Thus, the observed thermochromic behaviour in DMF is almost certainly a solvent-related phenomenon, with the chloro-ligands being displaced by DMF [31]. The first step is shown in Figure 11. Clearly, it would be interesting to examine the behaviour of similar solvents towards the nickel centre. Additional NMR spectra in additional solvents may be seen in the Supplementary Materials, as well as a comparison with the ferrocenyl alpha ligand. In conclusion, complex 4 is highly sensitive to solvent reactions, and therefore exhibits a high degree of lability. An examination of compound 4 using mass spectrometry (APCI on Orbitrap Instrument) was carried out; mass ions of the oxidized phosphine ligands were apparent; however, no parent mass ion was found. Interestingly, however, a mass ion of (M+ + 3) with the anticipated isotopic pattern was observed. Also present were higher mass ions with isotopic patterns centred at of m/z 938 and 971, respectively (these data are shown in the Supplementary Materials). Clearly, it would be interesting to further investigate the solid-state magnetic properties of this compound.
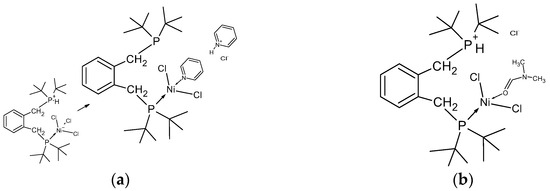
Figure 11.
(a) The interaction of pyridine with compound 4 showing the deprotonation of the phosphine and coordination of pyridine with nickel displacing a chloride. (b) the first step in the coordination of compound 4 with DMF.
3. Materials and Methods
The protocols used in this work were essentially standard coordination and crystallization at room temperature, conditions which were developed with a view to keeping the methodology as simple as possible. We could add that crystallization is one of the main methods of self-assembly. All metal complex crystallizations were carried out with the compound dissolved in a solution of reagent-grade dichloromethane with a top layer of diethyl ether added carefully. Crystals formed in all cases after the solvents co-diffused over several days. In the case of sulphide compound 4, ligand 1 (500 mg) was added to a saturated suspension of elemental sulphur (150mg, excess) in dichloromethane (10 mL). After 2 days, the solution was filtered, a top layer of diethyl ether was added of equal volume was carefully added, and the product was left to crystallize by slow diffusion over 3 days. A similar experiment conducted in an NMR tube using a filtered solution of elemental sulphur and the ligand indicated that the reaction was essentially quantitative after 2 days. All NMR experiments were conducted at 400 MHz for proton on a Bruker WH-400 instrument. Additional experimental detail is given in the Supplementary Materials.
4. Conclusions
Although the coordination of the alpha ligand with palladium and platinum is relatively straightforward, the coordination with nickel is less so: the natural propensity towards square–planar coordination may be difficult because of the steric bulk of the ligand in a relatively small sphere of coordination. However, it is interesting to observe the formation of a new Zwitterionic complex. Given that there is a readily available supply of related ligands, it will be of interest to observe the general trends in coordination chemistry when the synthetic method described here is applied. Clearly, as this nickel complex is so much cheaper to prepare than the corresponding palladium and platinum derivatives, and the palladium complex is used in such large quantities in industrial acrylic formation, it is imperative to test them using carboxy–alkylation reactions. We would encourage interested research groups to contact us to develop such work. Also, by performing the reaction in 1,1,2,2-tetrabromoethane, it may be possible to add a bromine instead of chlorine to the nickel centre, as this compound acts as a source of HBr in the presence of phosphines. At the suggestion of reviewers, it would be extremely interesting to examine the temperature-varyiant magnetic properties of compound 4 and its congeners and to expose the reactant [NiCl2DME] to D2O and then see if the deuterium is transferred into the product. We thank the reviewers for their extemely helpful positive suggestions. As the experimental and reference work was completed several years ago, a reviewer has kindly suggested some additional, more recent references relating to magnetic nickel complexes [32,33,34,35,36]. We also include some additional general background references [37,38,39], which include the recent synthesis of the related chiral ferrocene-based ligands by Erb and coworkers [39].
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/inorganics13090291/s1, Detailed NMR spectra in a range of solvents are presented. Crystallographic data for [1,2-(C6H4-CH2PtBu2)2Mo(CO)4], [1,2-(C6H4-H2P(S)tBu2)2], [2-(C6H4-CH2P(H)tBu2-1-(CH2PtBu2NiCl3)], [1,2-(C6H4-CH2PiBu2)2NiCl2], [Ni(DMF)6)]2+[NiCl4]2−, [Ni(Cl)2(H2O)2(DMF)2] are available as Supplementary Materials. CCDC 2421627 and 2421631-5, respectively, also contain the supplementary crystallographic data for this paper. These CCDC data can be obtained free of charge via http://www.ccdc.cam.ac.uk/conts/retrieving.html (or from CCDC, 12 Union Road, Cambridge, CB2 1EZ. Fax: +44 1223 336033; E-mail: deposit@ccdc.cam.ac.uk). Total 114 pages.
Author Contributions
I.R.B.: investigations, experimental work, writing, reviewing and editing, supervision, methodology, and conception. P.N.H.: crystallography, data deposition, and writing; W.C.: proofing, crystallography work, and director of crystallographic facilities; S.J.C.: Director of National Crystallographic Services and project overseer; K.M.F.: synthesis and thesis work; M.G.B.D.: early crystallography (2004); K.S.: synthetic work and experimental work. All authors have read and agreed to the published version of the manuscript.
Funding
We thank EPSRC for funding the National Crystallography Service (both Southampton. and Daresbury: grant number for synchrotron work was EP/D07746X/1). No additional external funding was used for the synthetic work.
Data Availability Statement
The original contributions presented in this study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
Acknowledgments
The authors would like to thank Mohammed Al-mashhadani, who helped us with the many thermochromic data runs. We all thank the EPSRC National Crystallographic Services for grants and facilities and the EPSRC National Mass Spectrometry Centre and their staff, based in Swansea University. Crystallographic work was carried out at the EPSRC National Crystallography Centre, based in the University of Southampton, and the Daresbury Laboratory Synchrotron Radiation Source. We thank the staff of both these institutions for their excellent and painstaking work. Additional mass spectroscopic data was also obtained in-house, and we thank the technical staff, based in Bangor. IRB thanks Patrick Murphy for his reliable support and lab provision, which enabled this work to be performed.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Nicholls, D. The Chemistry of Iron, Cobalt and Nickel: Comprehensive Inorganic Chemistry, Pergamon texts. In Inorganic Chemistry; Elsevier: Amsterdam, The Netherlands, 2013; Volume 24, 199p, ISBN 9781483146430. [Google Scholar]
- Blanchard, S.; Neese, F.; Bothe, E.; Weyhermüllerb, E.B.T.; Wieghardt, K. Square planar vs tetrahedral coordination in diamagnetic complexes of nickel(II) containing two bidentate pi-radical monoanions. Inorg. Chem. 2005, 44, 3636–3656. [Google Scholar] [CrossRef]
- Collinson, S.R.; Schröder, M. Nickel: Inorganic & Coordination Chemistry, Encyclopaedia of Inorganic Chemistry; Wiley: Hoboken, NJ, USA, 2006. [Google Scholar] [CrossRef]
- Lomjanský, D.; Rajnák, C.; Titiš, J.; Moncoľ, J.; Smolko, L.; Boča, R. Impact of tetrahedral and square planar geometry of Ni(II) complexes with (pseudo)halide ligands to magnetic properties. Inorg. Chim. Acta 2018, 483, 352–358. [Google Scholar] [CrossRef]
- Cope, J.D.; Denny, J.A.; Lamb, R.W.; McNamara, L.E.; Hammer, N.I.; Webster, C.E.; Hollis, T.K. Electrocatalytic reduction of CO2 with CCC-NHC pincer nickel complexes. J. Organometal. Chem. 2017, 845, 258–265. [Google Scholar] [CrossRef]
- Venanzi, L.M. Tetrahedral complexes of nickel (II) and the factors determining their formation. J. Inorg. Nucl. Chem. 1958, 8, 137–142. [Google Scholar] [CrossRef]
- Balakrishnan, K.P. Preparation and properties of nickel(II) complex dyes. J. Chem. Eng. Data 1980, 25, 186–187. [Google Scholar] [CrossRef]
- Garton, G.; Henn, D.E.; Powell, H.M.; Venanzi, L.M. Tetrahedral nickel(II) complexes and the factors determining their formation. Part V. The tetrahedral co-ordination of nickel in dichlorobistriphenylphosphinenickel. J. Chem. Soc 1963, 3625–3629. [Google Scholar] [CrossRef]
- Vondran, J.; Furst, M.R.L.; Eastham, G.R.; Seidensticker, T.; Cole-Hamilton, D.J. Magic of Alpha: The Chemistry of a Remarkable Bidentate Phosphine, 1,2-Bis(di-tert-butylphosphinomethyl)benzene. Chem. Rev. 2021, 121, 6610–6653. [Google Scholar] [CrossRef]
- Liu, J.; Heaton, B.T.; Iggo, J.A.; Whyman, R.; Bickley, J.F.; Steiner, A. The Mechanism of the Hydroalkoxycarbonylation of Ethene and Alkene–CO Copolymerization Catalyzed by Pd II–Diphosphine Cations. Chem. Eur. J. 2006, 12, 4417–4430. [Google Scholar] [CrossRef]
- Fanjul, T.; Eastham, G.; Floure, J.; Forrest, S.J.; Haddow, M.F.; Hamilton, A.; Pringle, P.G.; Orpen, A.G.; Waugh, M. Interplay of bite angle and cone angle effects. A comparison between o-C6H4(CH2PR2)(PR’2) and o-C6H4(CH2PR2)(CH2PR’2) as ligands for Pd-catalysed ethene hydromethoxycarbonylation. Dalton Trans. 2013, 42, 100–115. [Google Scholar] [CrossRef]
- Fanjul, T.; Eastham, G.; Fey, N.; Hamilton, A.; Orpen, A.G.; Pringle, P.G.; Waugh, M. Palladium complexes of the heterodiphosphine o-C6H4 (CH2PtBu2)(CH2PPh2) are highly selective and robust catalysts for the hydromethoxycarbonylation of ethene. Organometallics 2010, 29, 2292–2305. [Google Scholar] [CrossRef]
- Fortune, K.M.; Castel, C.; Robertson, C.M.; Horton, P.N.; Light, M.; Coles, S.J.; Waugh, M.; Clegg, W.; Harrington, R.W.; Butler, I.R. Ferrocenylmethylphosphanes and the Alpha Process for Methoxycarbonylation: The Original Story. Inorganics 2021, 9, 57. [Google Scholar] [CrossRef]
- Butler, I.R.; Baker, P.K.; Eastham, G.R.; Fortune, K.M.; Horton, P.N.; Hursthouse, M.B. Ferrocenylmethylphosphines ligands in the palladium-catalysed synthesis of methyl propionate. Inorg. Chem. Commun. 2004, 7, 1049–1052. [Google Scholar] [CrossRef]
- Dong, K.; Sang , R.; Fang, X.; Spannenberg, A.; Neumann, H.; Jackstell, R.; Beller, M. Efficient Palladium-Catalyzed Alkoxycarbonylation of Bulk Industrial Olefins Using Ferrocenyl Phosphine Ligands. Angew. Chem. Int. Ed. 2017, 56, 5267–5271. [Google Scholar] [CrossRef]
- Fanjul, T.; Eastham , G.; Haddow, M.F.; Hamilton, A.; Pringle, P.G.; Orpen, A.G.; Turner, T.P.W.; Waugh, M. Efficient and chemoselective ethene hydromethoxycarbonylation catalysts based on Pd-complexes of heterodiphosphines o-C6H4 (CH2PtBu2)(CH2PR2). Catal. Sci. Technol. 2012, 2, 937–950. [Google Scholar] [CrossRef]
- Knight, J.G.; Doherty, S.; Harriman, A.; Robins, E.G.; Berham, M.; Eastham, G.R.; Tooze, R.P.; Elsegood, M.R.J.; Champkin, P.; Clegg, W. Remarkable Differences in Catalyst Activity and Selectivity fo the Production of Methyl Propanoate versus CO−Ethylene Copolymer by a Series of Palladium Complexes of Related C4-Bridged Diphosphines. Organometallics 2000, 19, 4957–4967. [Google Scholar] [CrossRef]
- Clevenger, A.L.; Stolley, R.M.; Aderibigbe, J.; Louie, J. Trends in the Usage of Bidentate Phosphines as Ligands in Nickel Catalysis. Chem. Rev. 2020, 120, 6124–6196. [Google Scholar] [CrossRef]
- Ward, L.G.L. Anhydrous nickel(II) halides and their tetrakis(ethanol) and 1, 2-dimethoxyethane complexes. Inorg. Synth. 1971, 13, 154–164. Available online: https://onlinelibrary.wiley.co (accessed on 24 July 2025).
- Butler, I.R.; Williams, R.M.; Heeroma, A.; Horton, P.N.; Coles, S.J.; Jones, L.F. The Effect of Localized Magnetic Fields on the Spatially Controlled Crystallization of Transition Metal Complexes. Inorganics 2025, 13, 117. [Google Scholar] [CrossRef]
- Horton, P.N.; Coles, S.J.; Clegg, W.; Harrington, R.W.; Butler, I.R. A Rapid General Synthesis and the Spectroscopic Data of 2, 2′-Bis-(di-isopropylphosphino)-1, 1′-dibromoferrocene, (bpdbf), 1, 1′, 2, 2′-Tetrakis-(di-isopropylphosphino) Ferrocene, (tdipf) and Related Ligands: Taking dppf into the Future. Inorganics 2025, 13, 10. [Google Scholar] [CrossRef]
- Fortune, K. Nitrogen Donor Complexes of Molybdenum and Tungsten and New Routes to bis-1, 2 & tris-1, 2, 3 Substituted Ferrocenes. Ph.D. Thesis, Bangor University, Bangor, UK, 2004. Available online: https://research.bangor.ac.uk/portal/files/50696506/K_M_FORTUNE_PhD_2004_OCR.pdf (accessed on 24 July 2025).
- Oberhauser, W.; Bachmann, C.; Stampfl, T.; Haid, R.; Brüggeller, P. Structural differences in nickel(II) and palladium(II) complexes containing cis-1, 2-bis(diphenylphosphino)ethene or 1, 2-bis(diphenylphosphino)ethane. Polyhedron 1997, 16, 2827–2835. [Google Scholar] [CrossRef]
- Donahue, C.M.; McCollom, S.P.; Forrest, C.M.; Blake, A.V.; Bellott, B.J.; Keith, J.M.; Daly, S.R. Impact of Coordination Geometry, Bite Angle, and Trans Influence on Metal-Ligand Covalency in Phenyl-Substituted Phosphine Complexes of Ni and Pd. Inorg Chem. 2015, 54, 5646–5659. [Google Scholar] [CrossRef]
- Wu, K.; Doyle, A. Parameterization of phosphine ligands demonstrates enhancement of nickel catalysis via remote steric effects. Nat. Chem. 2017, 9, 779–784. [Google Scholar] [CrossRef]
- Li, B.; Wang, Z.; Luo, Y.; Wei, H.; Chen, J.; Liu, D.; Zhang, W. Nickel-catalyzed asymmetric hydrogenation for the preparation of α-substituted propionic acids. Nat. Commun. 2024, 15, 5482. [Google Scholar] [CrossRef]
- Hay, R.W.; Albedyhl, S.; Lightfoot, P. The crystal structure of [Ni(dmf)6][NiCl4] and comments on the hydrolysis of coordinated amides and peptides in metal complexes. Transit. Met. Chem. 1998, 23, 257–260. [Google Scholar] [CrossRef]
- Du, J.-L.; Li, L.-J. Synthesis and Crystal Structure of Nickel Complex, Ni[(DMF)4(H2O)2]·Br2. Inorg. Chem. Indian J. 2007, 2, 88–90. Available online: https://www.tsijournals.com/articles/synthesis-and-crystal-structure-of-nickel-complex-nidmf4h2o2br2.pdf (accessed on 24 July 2025).
- Bogachev, N.A.; Tsyrul’nikov, N.A.; Makarova, A.A.; Tolmachev, M.V.; Starova, L.; Yu, M.; Skripkin, Y.M.; Nikol’skiiet, A.B. Solubility of d-Element Salts in Organic and Aqueous-Organic Solvents: VII. Structure of Nickel Chloride Solvatocomplexes. Russ. J. Gen. Chem. 2019, 89, 859–864. [Google Scholar] [CrossRef]
- Butler, I.R.; Coles, S.J.; Horton, P.N. On the self-assembly of a trans-dibromo-bis-(dppfo2) iron (III), a ferrocene-ligand complex, dppfo2 = [(ƞ5-C5H4P(O)Ph2)2Fe]: Letting nature do the work. Inorg. Chem. Commun. 2018, 97, 166–170. [Google Scholar] [CrossRef]
- Pathania, A.; Bagchi, S. Slowdown of solvent structural dynamics in aqueous DMF solutions. Chem. Phys. Impact 2024, 9, 100711. [Google Scholar] [CrossRef]
- Shao, D.; Shi, L.; Liu, G.; Yue, J.; Ming, S.; Yang, X.; Zhu, J.; Ruan, Z. Metalo Hydrogen-Bonded Organic Frameworks Self-Assembled by Charge-Assisted Synthons for Ultrahigh Proton Conduction. Cryst. Growth Des. 2023, 23, 5035–5042. [Google Scholar] [CrossRef]
- Zhang, Y.-L.; Shi, L.; Liu, G.; Yang, J.; Shao, D. Hydrogen-bonded nickel(II)-organosulfonate framework: Thermal expansion behavior, proton conduction, and magnetic properties. J. Mol. Struct. 2023, 1292, 136131. [Google Scholar] [CrossRef]
- Zhou, Y.; Xiang, H.; Zhu, J.-Y.; Shi, L.; You, W.-J.; Wei, X.-Q.; Tian, Z.; Shao, D. Synthesis, structure, magnetism and proton conductivity of a cyanide-bridged NiIICoIII framework. Polyhedron 2022, 228, 116181. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, Y.L.; Zhang, Q.; Yang, S.-Y.; Wei, X.-Q.; Tian, Z.; Shao, D. Supramolecular porous frameworks of two Ni(II) coordination polymers with varying structures, porosities, and magnetic properties. Polyhedron 2022, 225, 116078. [Google Scholar] [CrossRef]
- Wei, X.-Q.; Wan, Y.; Tang, W.-J.; Dong, J.; Huang, L.; Yang, J.; Huang, X.-C.; Shao, D. Two cobalt(II) and nickel(II) sulfonate complexes constructed by mixed bipyridyl-organodisulfonate strategy: Syntheses, crystal structures, and magnetic properties. J. Mol. Struct. 2024, 1305, 137823. [Google Scholar] [CrossRef]
- Keskin, S.G.; Stanley, J.M.; Cowley, A.H. Synthesis, characterization and theoretical investigations of molybdenum carbonyl complexes with phosphorus/nitrogen/phosphorus ligand as bidentate and tridentate modes. Polyhedron 2017, 138, 206–217. [Google Scholar] [CrossRef]
- Laine, T.V.; Klinga, M.; Leskela, M. Pyridinylimine-based nickel(II) and palladium(II) complexes: Preparation, structural characterization and use as alkene polymerization catalysts. Eur. J. Inorg. Chem. 1999, 6, 959–964. [Google Scholar] [CrossRef]
- Boussandel, S.; Erb, W.; Roisnel, T.; Blot, M.; Hurvois, J.-P.; Butler, I.; Samarat, A.; Mongin, F. Hexafluoroisopropanol-promoted substitution towards the synthesis of enantiopure ferrocene phosphines. Synthesis 2025, in press. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).