Role of Silver Nanoparticles in Wound Healing: Mechanisms, Efficacy, and Clinical Applications
Abstract
1. Introduction: Wounds and the Wound Healing Process
2. Effects of AgNPs on Wound Healing with Mechanistic Insights
2.1. Antimicrobial and Anti-Biofilm Activities
2.1.1. Oxidative Dissolution of AgNPs in Biological Media and the Effect of Some Important Physicochemical Characteristics of AgNPs on the Rate of Silver Ions Release and Antibacterial Activity
2.1.2. Disruption of Bacterial Cell Membrane
2.1.3. Effects of AgNPs on Bacterial Proteins
2.1.4. Generation of Reactive Oxygen Species
2.1.5. Interaction with DNA
2.1.6. Antibiofilm Activity
2.1.7. Comparison of the Antibacterial Activity of Antibiotics and AgNPs
2.2. The Role of Silver Nanoparticles in Modulating Inflammation and Promoting Wound Healing
2.2.1. Anti-Inflammatory Activity
2.2.2. Effects on Cell Proliferation and Migration
2.2.3. Pro-Angiogenic Effects
2.2.4. Effects on ECM Remodelling Phase
3. AgNPs-Based Nanocomposite Materials for Wound Care
3.1. AgNPs-Loaded Hydrogels
3.2. AgNPs-Loaded Fiber-Based Composites
3.3. Sponge and Foam Composites Incorporating AgNPs
3.4. Film and Membrane Composites Loaded with AgNPs
3.5. Smart Responsive Wound Dressings Incorporating AgNPs
- On-demand antimicrobial release: Cargo is liberated only under infection-like cues, aligning with the wound’s dynamic microenvironment.
- Greater antibacterial potency with less silver: Localized, pulsed release—augmented by NIR photothermal effects—enhances killing (including biofilms) while minimizing total silver exposure and host–cell cytotoxicity.
- Improved selectivity and safety: Gating release by pathophysiological cues (acid/alkaline shifts, oxidative stress, temperature) confines activity to the wound site and limits off-target/systemic effects.
- Adaptability to heterogeneous wounds: Multiple triggers address spatial and temporal variability (surface alkalinity vs. acidic biofilm pockets; fluctuating ROS), sustaining efficacy as conditions evolve.
- Synergistic mechanisms: Combining chemical cues (pH/ROS) with physical inputs (NIR heat) accelerates bacterial eradication, dampens inflammation, and may shorten time-to-closure.
- Fewer dressing changes and better adherence: Sustained, stimulus-triggered cargo delivery can extend wear time and simplify care.
- Theragnostic potential: Some platforms pair triggers with sensing (e.g., pH-induced colour change), enabling real-time monitoring alongside therapy.
4. In Vivo Fate and Safety Profile of AgNP-Based Nanocomposites in Wound Therapeutics
4.1. Pharmacokinetics of Topical AgNPs Formulations
4.2. Toxicity of Topically Administered AgNPs
5. Clinical Translation Challenges for Silver-Based Nanoformulations in Wound Care: A Decade of Clinical Evidence
- (1)
- Formulation variability & standardization. AgNP size, shape, surface coating, loading, and release kinetics differ widely across products, making direct comparisons and dosing recommendations difficult.
- (2)
- Dose, exposure, and pharmacokinetics/pharmacodynamics (PK/PD) in wounds. Local Ag+ flux depends on exudate volume, pH, proteins, and biofilm presence, meaning that identical dressings can deliver markedly different exposures. Robust local PK/PD models linking silver release to antibacterial efficacy and host safety are still lacking.
- (3)
- Safety window in real patients. Silver can be cytotoxic to keratinocytes/fibroblasts at high local concentrations. Systemic absorption increases with large total burn surface area (TBSA), prolonged application, and barrier disruption, raising concerns about argyria, hepatic/renal toxicity, and paediatric safety. Routine therapeutic monitoring (e.g., serum Ag) remains unstandardized.
- (4)
- Efficacy against biofilms in vivo. Strong in vitro antibacterial effects often fail to translate clinically. Biofilm architecture, wound matrix binding, and ion scavenging (by chloride or thiol groups) can diminish efficacy. Demonstrating biofilm clearance and healing benefit in randomized controlled trials (RCTs) remains challenging.
- (5)
- Comparative effectiveness & endpoints. Clinical trials frequently involve heterogeneous wounds and inconsistent outcome measures (e.g., bioburden reduction vs. time-to-healing vs. pain or odor control). Well-powered, blinded RCTs with clinically meaningful endpoints and head-to-head comparisons against standard care are needed.
- (6)
- Resistance, tolerance & microbiome impact. Although classical resistance to silver is uncommon, tolerance mechanisms (efflux, sequestration, biofilm protection) and co-selection with antibiotic resistance are concerns. The influence of AgNPs on the skin and wound microbiome remains poorly understood.
- (7)
- Regulatory pathway clarity. Consistent requirements for nano-specific characterization, risk assessment, and post-market surveillance are still evolving across jurisdictions. Silver dressings straddle device/drug/combination categories, since their mechanism of action may be partly physical (covering the wound, absorbing exudate, maintaining moisture) and partly pharmacological (antibacterial). This overlap creates regulatory grey zones, complicating product approval and standardization.
- (8)
- Manufacturing, quality control, and reproducibility. Scale-up must tightly control particle size distribution, surface chemistry, endotoxin levels, residual reagents, and batch-to-batch consistency. Protein corona formation during storage or use can alter biological behaviour.
- (9)
- Assay interference & measurement. Silver (in both nanoparticulate and ionic forms) can interfere with colorimetric and fluorescence assays, complicating cytotoxicity, ROS, and microbiological testing. Reliable speciation methods, such as Single Particle Inductively Coupled Plasma Mass Spectrometry (sp-ICP-MS), can distinguish metallic Ag0 nanoparticles, dissolved Ag+ ions, and protein-bound complexes, quantifying each form—essential for understanding toxicokinetics, release behaviour, and biological safety. However, such methods are not yet routine in clinical laboratories.
- (10)
- Integration with standard wound care. Compatibility with debridement, negative pressure, enzymatic agents, and adjunct antimicrobials requires clearly defined protocols to prevent over-treatment or neutralization of effect.
- (11)
- Long-term outcomes & scarring. Evidence remains limited regarding how nanosilver influences re-epithelialization quality, scarring, and tissue function over longer healing periods, beyond early bioburden reduction.
- (12)
- Cost-effectiveness & stewardship. Premium products must show value vs. simpler dressings. Antimicrobial stewardship principles should guide when to start/stop silver-based treatment. Clinicians should use clinical indicators (not habit or routine) to decide when to apply (infection or high-risk indicators) and when to discontinue silver dressings (the wound is clean and granulating), minimizing unnecessary exposure.
- (13)
- Environmental & occupational considerations. Silver release into wastewater and potential aerosolization during handling (e.g., sprays, powders, or cutting dry foams) pose ecotoxicological and occupational safety concerns that institutions must carefully evaluate.
| Dressings | Clinical Method Summary | Quantitative Results | Year (Ref.) |
|---|---|---|---|
| Aquacel Ag | The study was designed to evaluate the systemic absorption of silver in patients (criteria: silver levels > 0.5 μg/mL) with chronic inflammatory wounds and its association with silver toxicity. The study was a longitudinal, observational, multicenter, open-label pilot study using 40 elderly (patients mostly female, average age 74.3 years). | Dressing changed every 2 days between the initial day and day 28 of the treatment period. Mean wound surface area reduction was 22.8% (p = 0.041), along with a decrease in the fibrin percentage (beneficial for wound healing) between day 0 and day 28. Half the patients showed increased silver levels. There was no argyria or systemic toxicity. Elimination of silver from the body was slow and could lead to cumulative toxicity, especially in elderly patients. The study recommends against long-term silver dressing use. | 2018 [198] |
| Aquacel® Ag+ Extra™ (All patients previously managed with traditional silver (26%), iodine (23%) or polyhexamethylene biguanide (PHMB) (11%) containing products or systemic antibiotics (12%)) | The study recruited 65 patients with wounds ranging in duration from 1 week to 20 years (median duration: 12 months). 47 cases (72%) had stagnant wounds, and 15 cases (23%) had deteriorating wounds, while 3 wounds were not recorded; observations were made for 1–11 weeks. Participants also had clinical signs of infection or critical colonization. | Observations were as follows: 17% of wounds healed, 62% of wounds showed improvement, 14% of wounds remained the same, and 8% of wounds deteriorated. Moderate exudate (52% n = 24) and high exudate (37% n = 34) levels before treatment led to low (31%, n = 20) and moderate (43% n = 28) levels, respectively, after treatment. Biofilms were observed in 49% and slough in 42% of wounds. After applying the dressing, the wound bed tissue was 63% granulated. Healthy wound bed tissues increased from 33% to 67% after treatment. Necrotic, slough biofilm reduced from 92% to 40% following treatment. Peri-wound skin health improved in 67% of cases. | 2020 [216] |
| Acticoat™ Flex 7 (nano-Ag) with dressings without nano Ag | Retrospective study: 330 patients and 2242 patients in the control group in community centers with various types of wounds, including pressure injuries, diabetic foot ulcers, and venous leg ulcers (used Bates–Jensen Wound Assessment Tool). | Sustained silver release over 7 days. The mean time between dressing changes was 3.98 days vs. 1.87 days in control (p < 0.01), reducing nurse visits. The mean healing time for wounds treated with Acticoat 7 was significantly shorter (10.46 weeks) compared to wounds with a control dressing (25.49 weeks). Only 0.9% of patients treated with Acticoat 7 dressing developed a systemic infection, compared to 3% in the comparative group. Potential for bias and no control for confounding variables, e.g., concurrent treatments. | 2021 [220] |
| Biatain® Ag Non-Adhesive Foam versus silver sulfadiazine | 60 adult patients diagnosed with type 2 diabetes mellitus with diabetic foot ulcers (DFU) measuring at least 1 cm2 were recruited. Treatment Group: Biatain® Ag Non-Adhesive Foam dressing applied at least every two days (38 patients). Control Group: 1% SSD cream applied once or twice per day (22 patients) A 4-week study, where debridement was performed during weekly visits, if necessary. | Enterococcus faecalis and Staphylococcus aureus were isolated from the wound culture in both groups. The proportion of the wound healed at week 4 in the SSD group was 27.00 ± 4.95%, while Biatain was 76.43 ± 7.41% (p < 0.0001). Silver foam facilitated wound closure faster than SSD in the patient population with HbA1c > 7% (59.94 ± 8.00% vs. 14.21 ± 3.72%, p = 0.027) and in patients with positive microbial isolates in their wound culture (60.87 ± 4.06% vs. 37.50 ± 5.89%, p = 0.020). | 2021 [221] |
| Biatain alginate Ag versus gauze (some with iodoform) | 40 patients in observation and 40 patients in the control group. Debridement and Biatain Alginate Ag were applied to the wounds. Dressing changed every 1 to 3 days. Assessment at 7, 14 days, and 1 month after treatment. The study observed the frequency of dressing changes, granulation tissue growth, wound formation, and healing time. | Pain score (VAS) was significantly different between the Bitain and the control group (p < 0.05). Better outcomes in wound scar healing were observed as compared to the control group (p < 0.05). Enhanced granulation tissue growth was significantly higher in the observation vs. the control. Bacterial load was significantly lower than in the control group. | 2022 [218] |
| Aquacel Ag+ versus Sorbact dressing (Cutimed Sorbact, Essity, retains exudate, no release of any antimicrobials) | Retrospective Patient Chart Audit with 350 patient charts: 200 with Aquacel Ag+ and 150 with Sorbact. Data analyzed separately for Germany and the US (DFU and venous leg ulcers). | Unclear why specific dressings were chosen for specific patients. Germany: Wound percent reduction and wound closure comparable; greater proportion of Sorbact users needed surgery (0 vs. 11%, p = 0.039). US: Wounds were worsening before the use of Aquacel (49% vs. 34%, p = 0.01), regression analysis suggests that it was 3.53 times more likely to have wound healed in Aquacel cohort (p = 0.033). | 2023 [222] |
| Acticoat versus Standard of Care (SoC) | Prospective, open-labeled, randomized, placebo-controlled trial for acute diabetes-related foot ulcers, with 63 patients with Acticoat and 55 with SoC. The primary endpoint was the proportion of ulcers healed at 12 weeks. | Observation of ulcers healed at 12 weeks: 75% in the control group and 69% in the silver group (p = 0.49). No significant difference in complete ulcer healing (p = 0.53), osteomyelitis, need for amputation or antibiotic treatment between the silver and control groups. | 2023 [226] |
| Acticoat™ vs. Aquacel Ag | A single-blind, randomized controlled study in a Pediatric Emergency Department, included 89 children with superficial or mid-dermal burns (<10% TBSA),who were randomized to receive either the Acticoat™ (n = 45) or Aquacel® Ag (n = 44) dressings. | No significant difference between the groups in terms of percentage epithelialization by day 10, with Acticoat™ showing 93 ± 14% and Aquacel® Ag showing 94 ± 17% (p = 0.89). No significant difference in infection and escalation of care. Aquacel® Ag dressings (59) required significantly fewer dressing changes compared to Acticoat (102) (p = 0.03) | 2016 [227] |
| Procellera™ + SoC versus SoC (moleskin and Tegaderm) | A prospective randomized controlled two-arm Clinical Study for blister management. The study involved 80 Ranger recruits as participants in a 14-day study. | No significant difference in wound healing rates between the SoC group and the SoC + Procellera group (p = 0.528). No significant difference in pain management between the SoC and SoC + Procellera groups. | 2017 [228] |
| Mepilex A vs. Suprathel (DL-polylactic Acid membrane) | A prospective randomized controlled trial comparing the outpatient treatment of pediatric and adult partial-thickness burns. 29 adults and 33 pediatric patients (almost equally split between two dressings). TBSA: 1–29% in Meiplex Ag and 1–20% in Suprathel group. | The median time to complete reepithelialization was 12 days for both groups (p = 0.75). Suprathel reported better overall scar quality, and Mepilex Ag increased the stiffness of burned skin at 1 month post-burn. Patients experienced less pain with Suprathel (only for the first 5 days, p = 0.03). | 2018 [229] |
| Silverlon vs. SSD or mafenide acetate (considered topical antimicrobials) | A 10-year retrospective analysis on a total of 987 combat burn casualties, with 184 patients in Group 1 (Silverlon) and 803 in Group 2 (topical antimicrobial); 49% of the cohort had third-degree burns. | The incidence of wound infection was 5.4% in Group 1 and 9.5% in Group 2 (p = 0.08), the overall mortality rate did not differ significantly between the groups (8% in Group 1). The incidence of bacteremia was 4.3% in Group 1 and 5.5% in Group 2, showing no significant difference (p = 1.0). The application of topical antimicrobials was painful. | 2018 [230] |
| Acticoat Flex 3 vs. 1% SSD | A randomized, single-center, single-blind trial involving 100 adults aged 18–65 with second-degree burns. | Reepithelization: Acticoat: 48% (24/50 patients), SSD: 52% (26/50 patients) (p = 0.56). Number of dressing changes: Acticoat fewer than SSD (p < 0.001) | 2022 [231] |
| Procellera™ versus SoC | A single-center prospective, randomized controlled clinical trial with 38 patients with dermal burn/traumatic wounds. Procellera dressing compared with SOC: silver nylon, SSD ointment, bacitracin, xeroform, 5% sulfamylon solution, and Manuka honey, observations at 7-day. | In 52% of the Procellera-treated wounds, little to no biofilm could be detected by scanning electron microscopy compared to only 24% of SoC-treated wounds; Procellera lowered the increase in biofilm versus SoC (p < 0.05). | 2024 [223] |
6. Conclusions and Future Perspective
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AgNPs | Silver nanoparticles |
| ROS | Reactive oxygen species |
| RNS | Reactive nitrogen species |
| VEGF | Vascular endothelial growth factor |
| bFGF | Basic fibroblast growth factor |
| ECM | Extracellular matrix |
| MMP | Matrix metalloproteinase |
| TIMP | Tissue inhibitor of metalloproteinase |
| HIF-1 | Hypoxia-inducible factor-1 |
| NO | Nitric oxide |
| iNOS | Inducible nitric oxide synthase |
| NF-κB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| MAPK | Mitogen-activated protein kinase |
| PI3K | Phosphatidylinositol 3-kinase |
| Akt | Protein kinase B |
| SOD | Superoxide dismutase |
| CAT | Catalase |
| IHC | Immunohistochemistry |
| SSD | Silver sulfadiazine |
| CAM | Chorioallantoic membrane |
| QC | Quality control |
References
- Mihai, M.M.; Dima, M.B.; Dima, B.; Holban, A.M. Nanomaterials for Wound Healing and Infection Control. Materials 2019, 12, 2176. [Google Scholar] [CrossRef]
- Morton, L.M.; Phillips, T.J. Wound healing and treating wounds: Differential diagnosis and evaluation of chronic wounds. J. Am. Acad. Dermatol. 2016, 74, 589–605. [Google Scholar] [CrossRef]
- Percival, N.J. Classification of Wounds and their Management. Surg. Oxf. Int. Ed. 2002, 20, 114–117. [Google Scholar] [CrossRef]
- Lazarus, G.S.; Cooper, D.M.; Knighton, D.R.; Margolis, D.J.; Pecoraro, R.E.; Rodeheaver, G.; Robson, M.C. Definitions and guidelines for assessment of wounds and evaluation of healing. Arch. Dermatol. 1994, 130, 489–493. [Google Scholar] [CrossRef] [PubMed]
- Chhabra, S.; Chhabra, N.; Kaur, A.; Gupta, N. Wound Healing Concepts in Clinical Practice of OMFS. J. Maxillofac. Oral Surg. 2017, 16, 403–423. [Google Scholar] [CrossRef]
- Diegelmann, R.F.; Evans, M.C. Wound healing: An overview of acute, fibrotic and delayed healing. Front. Biosci. 2004, 9, 283–289. [Google Scholar] [CrossRef]
- Young, A.; McNaught, C.-E. The physiology of wound healing. Surgery 2011, 29, 475–479. [Google Scholar] [CrossRef]
- Greaves, N.S.; Ashcroft, K.J.; Baguneid, M.; Bayat, A. Current understanding of molecular and cellular mechanisms in fibroplasia and angiogenesis during acute wound healing. J. Dermatol. Sci. 2013, 72, 206–217. [Google Scholar] [CrossRef]
- Monika, P.; Chandraprabha, M.N.; Rangarajan, A.; Waiker, P.V.; Chidambara Murthy, K.N. Challenges in Healing Wound: Role of Complementary and Alternative Medicine. Front. Nutr. 2021, 8, 791899. [Google Scholar] [CrossRef]
- Whittam, A.J.; Maan, Z.N.; Duscher, D.; Wong, V.W.; Barrera, J.A.; Januszyk, M.; Gurtner, G.C. Challenges and Opportunities in Drug Delivery for Wound Healing. Adv. Wound Care 2016, 5, 79–88. [Google Scholar] [CrossRef]
- Frykberg, R.G.; Banks, J. Challenges in the Treatment of Chronic Wounds. Adv. Wound Care 2015, 4, 560–582. [Google Scholar] [CrossRef]
- Tarnuzzer, R.W.; Schultz, G.S. Biochemical analysis of acute and chronic wound environments. Wound Repair Regen. 1996, 4, 321–325. [Google Scholar] [CrossRef]
- Sutcliffe, J.E.S.; Thrasivoulou, C.; Serena, T.E.; Madden, L.; Richards, T.; Phillips, A.R.J.; Becker, D.L. Changes in the extracellular matrix surrounding human chronic wounds revealed by 2-photon imaging. Int. Wound J. 2017, 14, 1225–1236. [Google Scholar] [CrossRef]
- Landén, N.X.; Li, D.; Ståhle, M. Transition from inflammation to proliferation: A critical step during wound healing. Cell. Mol. Life Sci. 2016, 73, 3861–3885. [Google Scholar] [CrossRef] [PubMed]
- Hong, W.X.; Hu, M.S.; Esquivel, M.; Liang, G.Y.; Rennert, R.C.; McArdle, A.; Paik, K.J.; Duscher, D.; Gurtner, G.C.; Lorenz, H.P.; et al. The Role of Hypoxia-Inducible Factor in Wound Healing. Adv. Wound Care 2014, 3, 390–399. [Google Scholar] [CrossRef] [PubMed]
- Hunt, M.; Torres, M.; Bachar-Wikstrom, E.; Wikstrom, J.D. Cellular and molecular roles of reactive oxygen species in wound healing. Commun. Biol. 2024, 7, 1534. [Google Scholar] [CrossRef] [PubMed]
- Ukaegbu, K.; Allen, E.; Svoboda, K.K.H. Reactive Oxygen Species and Antioxidants in Wound Healing: Mechanisms and Therapeutic Potential. Int. Wound J. 2025, 22, e70330. [Google Scholar] [CrossRef]
- Cano Sanchez, M.; Lancel, S.; Boulanger, E.; Neviere, R. Targeting Oxidative Stress and Mitochondrial Dysfunction in the Treatment of Impaired Wound Healing: A Systematic Review. Antioxidants 2018, 7, 98. [Google Scholar] [CrossRef]
- Tan, M.L.L.; Chin, J.S.; Madden, L.; Becker, D.L. Challenges faced in developing an ideal chronic wound model. Expert Opin. Drug Discov. 2023, 18, 99–114. [Google Scholar] [CrossRef]
- Caley, M.P.; Martins, V.L.; O’Toole, E.A. Metalloproteinases and Wound Healing. Adv. Wound Care 2015, 4, 225–234. [Google Scholar] [CrossRef]
- Tyavambiza, C.; Meyer, M.; Meyer, S. Cellular and Molecular Events of Wound Healing and the Potential of Silver Based Nanoformulations as Wound Healing Agents. Bioengineering 2022, 9, 712. [Google Scholar] [CrossRef]
- Naganthran, A.; Verasoundarapandian, G.; Khalid, F.E.; Masarudin, M.J.; Zulkharnain, A.; Nawawi, N.M.; Karim, M.; Che Abdullah, C.A.; Ahmad, S.A. Synthesis, Characterization and Biomedical Application of Silver Nanoparticles. Materials 2022, 15, 427. [Google Scholar] [CrossRef]
- Kaya, M.; Akdaşçi, E.; Eker, F.; Bechelany, M.; Karav, S. Recent Advances of Silver Nanoparticles in Wound Healing: Evaluation of In Vivo and In Vitro Studies. Int. J. Mol. Sci. 2025, 26, 9889. [Google Scholar] [CrossRef]
- Verma, R.K.; Nagar, V.; Sharma, A.; Mavry, B.; Kumari, P.; Lohar, S.; Singhal, A.; Prajapati, M.K.; Singh, A.; Awasthi, K.K.; et al. Green Synthesized Nanoparticles Targeting Antimicrobial Activities. Biointerface Res. Appl. Chem. 2023, 13, 469. [Google Scholar] [CrossRef]
- El Shanshoury, A.E.R.R.; Sabae, S.Z.; El Shouny, W.A.; Elsaied, H.E.; Badr, H.M.; Abo-Shady, A.M. Biomimetic Synthesis of Silver Nanoparticles Using New Aquatic Species of Bacillus, Alcaligenes, and Paenibacillus and their Potential Antibiofilm Activity against Biofilm-Forming Escherichia coli. Lett. Appl. NanoBioSci. 2023, 12, 127. [Google Scholar] [CrossRef]
- Abdussalam-Mohammed, W.; Abraheem, M.S.; Ettarhouni, Z.O.; Dakhil, O.O.; Mezoughi, A.B. Novel Compatible Silver Nanoparticles Functionalized by Vitamin C and its Derivatives: Characterization and their Antibacterial Activity against Escherichia coli and Staphylococcus aureus. Biointerface Res. Appl. Chem. 2023, 13, 590. [Google Scholar] [CrossRef]
- Narayanaswamy, S.; Bhaskar, R.; Jayadevappa, R.K.K.; Ramachandran, S.K.M.; Prasad, M.A. A Comprehensive Review on the Antimicrobial and Photocatalytic Properties of Green Synthesized Silver Nanoparticles. Lett. Appl. NanoBioSci. 2023, 12, 140. [Google Scholar] [CrossRef]
- Abbaszadegan, A.; Ghahramani, Y.; Gholami, A.; Hemmateenejad, B.; Dorostkar, S.; Nabavizadeh, M.; Sharghi, H. The Effect of Charge at the Surface of Silver Nanoparticles on Antimicrobial Activity against Gram-Positive and Gram-Negative Bacteria: A Preliminary Study. J. Nanomater. 2015, 2015, 720654. [Google Scholar] [CrossRef]
- Godoy-Gallardo, M.; Eckhard, U.; Delgado, L.M.; de Roo Puente, Y.J.D.; Hoyos-Nogués, M.; Gil, F.J.; Perez, R.A. Antibacterial approaches in tissue engineering using metal ions and nanoparticles: From mechanisms to applications. Bioact. Mater. 2021, 6, 4470–4490. [Google Scholar] [CrossRef]
- Sadoq, B.E.; Britel, M.R.; Bouajaj, A.; Maâlej, R.; Abid, M.; Douiri, H.; Touhami, F.; Maurady, A.; Touhami, A. A Review on Antibacterial Activity of Nanoparticles. Biointerface Res. Appl. Chem. 2023, 13, 405. [Google Scholar] [CrossRef]
- Zhang, Q.; Hu, Y.; Masterson, C.M.; Jang, W.; Xiao, Z.; Bohloul, A.; Garcia-Rojas, D.; Puppala, H.L.; Bennett, G.; Colvin, V.L. When function is biological: Discerning how silver nanoparticle structure dictates antimicrobial activity. iScience 2022, 25, 104475. [Google Scholar] [CrossRef]
- Lok, C.N.; Ho, C.M.; Chen, R.; He, Q.Y.; Yu, W.Y.; Sun, H.; Tam, P.K.; Chiu, J.F.; Che, C.M. Silver nanoparticles: Partial oxidation and antibacterial activities. J. Biol. Inorg. Chem. 2007, 12, 527–534. [Google Scholar] [CrossRef]
- Xiu, Z.M.; Zhang, Q.B.; Puppala, H.L.; Colvin, V.L.; Alvarez, P.J. Negligible particle-specific antibacterial activity of silver nanoparticles. Nano Lett. 2012, 12, 4271–4275. [Google Scholar] [CrossRef]
- Le Ouay, B.; Stellacci, F. Antibacterial activity of silver nanoparticles: A surface science insight. Nano Today 2015, 10, 339–354. [Google Scholar] [CrossRef]
- Pareek, V.; Gupta, R.; Panwar, J. Do physico-chemical properties of silver nanoparticles decide their interaction with biological media and bactericidal action? A review. Mater. Sci. Eng. C 2018, 90, 739–749. [Google Scholar] [CrossRef]
- AshaRani, P.V.; Low Kah Mun, G.; Hande, M.P.; Valiyaveettil, S. Cytotoxicity and genotoxicity of silver nanoparticles in human cells. ACS Nano 2009, 3, 279–290. [Google Scholar] [CrossRef]
- Ho, C.M.; Yau, S.K.; Lok, C.N.; So, M.H.; Che, C.M. Oxidative dissolution of silver nanoparticles by biologically relevant oxidants: A kinetic and mechanistic study. Chem. Asian J. 2010, 5, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Menichetti, A.; Mavridi-Printezi, A.; Mordini, D.; Montalti, M. Effect of Size, Shape and Surface Functionalization on the Antibacterial Activity of Silver Nanoparticles. J. Funct. Biomater. 2023, 14, 244. [Google Scholar] [CrossRef]
- Raza, M.A.; Kanwal, Z.; Rauf, A.; Sabri, A.N.; Riaz, S.; Naseem, S. Size- and Shape-Dependent Antibacterial Studies of Silver Nanoparticles Synthesized by Wet Chemical Routes. Nanomaterials 2016, 6, 74. [Google Scholar] [CrossRef]
- Wu, Y.; Yang, Y.; Zhang, Z.; Wang, Z.; Zhao, Y.; Sun, L. A facile method to prepare size-tunable silver nanoparticles and its antibacterial mechanism. Adv. Powder Technol. 2018, 29, 407–415. [Google Scholar] [CrossRef]
- Skandalis, N.; Dimopoulou, A.; Georgopoulou, A.; Gallios, N.; Papadopoulos, D.; Tsipas, D.; Theologidis, I.; Michailidis, N.; Chatzinikolaidou, M. The Effect of Silver Nanoparticles Size, Produced Using Plant Extract from Arbutus unedo, on Their Antibacterial Efficacy. Nanomaterials 2017, 7, 178. [Google Scholar] [CrossRef]
- Balu, S.K.; Andra, S.; Damiri, F.; Sivaramalingam, A.; Sudandaradoss, M.V.; Kumarasamy, K.; Bhakthavachalam, K.; Ali, F.; Kundu, M.K.; Rahman, M.H.; et al. Size-Dependent Antibacterial, Antidiabetic, and Toxicity of Silver Nanoparticles Synthesized Using Solvent Extraction of Rosa indica L. Petals. Pharmaceuticals 2022, 15, 689. [Google Scholar] [CrossRef]
- Hileuskaya, K.; Ladutska, A.; Kulikouskaya, V.; Kraskouski, A.; Novik, G.; Kozerozhets, I.; Kozlovskiy, A.; Agabekov, V. ‘Green’ approach for obtaining stable pectin-capped silver nanoparticles: Physico-chemical characterization and antibacterial activity. Colloids Surf. A Physicochem. Eng. Asp. 2020, 585, 124141. [Google Scholar] [CrossRef]
- Ji, H.; Zhou, S.; Fu, Y.; Wang, Y.; Mi, J.; Lu, T.; Wang, X.; Lü, C. Size-controllable preparation and antibacterial mechanism of thermo-responsive copolymer-stabilized silver nanoparticles with high antimicrobial activity. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 110, 110735. [Google Scholar] [CrossRef]
- Haidari, H.; Bright, R.; Kopecki, Z.; Zilm, P.S.; Garg, S.; Cowin, A.J.; Vasilev, K.; Goswami, N. Polycationic Silver Nanoclusters Comprising Nanoreservoirs of Ag(+) Ions with High Antimicrobial and Antibiofilm Activity. ACS Appl. Mater. Interfaces 2022, 14, 390–403. [Google Scholar] [CrossRef] [PubMed]
- Kittler, S.; Greulich, C.; Diendorf, J.; Köller, M.; Epple, M. Toxicity of Silver Nanoparticles Increases during Storage Because of Slow Dissolution under Release of Silver Ions. Chem. Mater. 2010, 22, 4548–4554. [Google Scholar] [CrossRef]
- Lomelí-Rosales, D.A.; Zamudio-Ojeda, A.; Cortes-Llamas, S.A.; Velázquez-Juárez, G. One-step synthesis of gold and silver non-spherical nanoparticles mediated by Eosin Methylene Blue agar. Sci. Rep. 2019, 9, 19327. [Google Scholar] [CrossRef]
- Titkov, A.I.; Logutenko, O.A.; Bulina, N.V.; Yukhin, Y.M.; Lyakhov, N.Z. Synthesis of nonspherical nanoparticles by reducing silver neodecanoate extract with benzyl alcohol. Theor. Found. Chem. Eng. 2017, 51, 557–562. [Google Scholar] [CrossRef]
- Hong, X.; Wen, J.; Xiong, X.; Hu, Y. Shape effect on the antibacterial activity of silver nanoparticles synthesized via a microwave-assisted method. Environ. Sci. Pollut. Res. Int. 2016, 23, 4489–4497. [Google Scholar] [CrossRef]
- Goyal, D.; Kaur, G.; Tewari, R.; Kumar, R. Correlation of edge truncation with antibacterial activity of plate-like anisotropic silver nanoparticles. Environ. Sci. Pollut. Res. Int. 2017, 24, 20429–20437. [Google Scholar] [CrossRef]
- Seyedpour, S.F.; Arabi Shamsabadi, A.; Khoshhal Salestan, S.; Dadashi Firouzjaei, M.; Sharifian Gh, M.; Rahimpour, A.; Akbari Afkhami, F.; Shirzad Kebria, M.R.; Elliott, M.A.; Tiraferri, A.; et al. Tailoring the Biocidal Activity of Novel Silver-Based Metal Azolate Frameworks. ACS Sustain. Chem. Eng. 2020, 8, 7588–7599. [Google Scholar] [CrossRef]
- Shaikh, S.; Nazam, N.; Rizvi, S.M.D.; Ahmad, K.; Baig, M.H.; Lee, E.J.; Choi, I. Mechanistic Insights into the Antimicrobial Actions of Metallic Nanoparticles and Their Implications for Multidrug Resistance. Int. J. Mol. Sci. 2019, 20, 2468. [Google Scholar] [CrossRef]
- Armentano, I.; Arciola, C.R.; Fortunati, E.; Ferrari, D.; Mattioli, S.; Amoroso, C.F.; Rizzo, J.; Kenny, J.M.; Imbriani, M.; Visai, L. The interaction of bacteria with engineered nanostructured polymeric materials: A review. Sci. World J. 2014, 2014, 410423. [Google Scholar] [CrossRef]
- Jiang, W.; Kim, B.Y.S.; Rutka, J.T.; Chan, W.C.W. Nanoparticle-mediated cellular response is size-dependent. Nat. Nanotechnol. 2008, 3, 145–150. [Google Scholar] [CrossRef]
- Ramalingam, B.; Parandhaman, T.; Das, S.K. Antibacterial Effects of Biosynthesized Silver Nanoparticles on Surface Ultrastructure and Nanomechanical Properties of Gram-Negative Bacteria viz. Escherichia coli and Pseudomonas aeruginosa. ACS Appl. Mater. Interfaces 2016, 8, 4963–4976. [Google Scholar] [CrossRef] [PubMed]
- Buszewski, B.; Railean-Plugaru, V.; Pomastowski, P.; Rafińska, K.; Szultka-Mlynska, M.; Golinska, P.; Wypij, M.; Laskowski, D.; Dahm, H. Antimicrobial activity of biosilver nanoparticles produced by a novel Streptacidiphilus durhamensis strain. J. Microbiol. Immunol. Infect. 2018, 51, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Biao, L.; Tan, S.; Wang, Y.; Guo, X.; Fu, Y.; Xu, F.; Zu, Y.; Liu, Z. Synthesis, characterization and antibacterial study on the chitosan-functionalized Ag nanoparticles. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 76, 73–80. [Google Scholar] [CrossRef]
- Huma, Z.E.; Gupta, A.; Javed, I.; Das, R.; Hussain, S.Z.; Mumtaz, S.; Hussain, I.; Rotello, V.M. Cationic Silver Nanoclusters as Potent Antimicrobials against Multidrug-Resistant Bacteria. ACS Omega 2018, 3, 16721–16727. [Google Scholar] [CrossRef]
- Zhao, R.; Lv, M.; Li, Y.; Sun, M.; Kong, W.; Wang, L.; Song, S.; Fan, C.; Jia, L.; Qiu, S.; et al. Stable Nanocomposite Based on PEGylated and Silver Nanoparticles Loaded Graphene Oxide for Long-Term Antibacterial Activity. ACS Appl. Mater. Interfaces 2017, 9, 15328–15341. [Google Scholar] [CrossRef] [PubMed]
- Alsammarraie, F.K.; Wang, W.; Zhou, P.; Mustapha, A.; Lin, M. Green synthesis of silver nanoparticles using turmeric extracts and investigation of their antibacterial activities. Colloids Surf. B Biointerfaces 2018, 171, 398–405. [Google Scholar] [CrossRef]
- Nakamura, S.; Ando, N.; Sato, M.; Ishihara, M. Ultraviolet Irradiation Enhances the Microbicidal Activity of Silver Nanoparticles by Hydroxyl Radicals. Int. J. Mol. Sci. 2020, 21, 3204. [Google Scholar] [CrossRef]
- Quinteros, M.A.; Cano Aristizábal, V.; Dalmasso, P.R.; Paraje, M.G.; Páez, P.L. Oxidative stress generation of silver nanoparticles in three bacterial genera and its relationship with the antimicrobial activity. Toxicol. Vitr. 2016, 36, 216–223. [Google Scholar] [CrossRef] [PubMed]
- Dąbrowska-Bouta, B.; Sulkowski, G.; Strużyński, W.; Strużyńska, L. Prolonged Exposure to Silver Nanoparticles Results in Oxidative Stress in Cerebral Myelin. Neurotox. Res. 2019, 35, 495–504. [Google Scholar] [CrossRef]
- Nallathamby, P.D.; Lee, K.J.; Desai, T.; Xu, X.H. Study of the multidrug membrane transporter of single living Pseudomonas aeruginosa cells using size-dependent plasmonic nanoparticle optical probes. Biochemistry 2010, 49, 5942–5953. [Google Scholar] [CrossRef]
- Halder, S.; Yadav, K.K.; Sarkar, R.; Mukherjee, S.; Saha, P.; Haldar, S.; Karmakar, S.; Sen, T. Alteration of Zeta potential and membrane permeability in bacteria: A study with cationic agents. Springerplus 2015, 4, 672. [Google Scholar] [CrossRef]
- Holt, K.B.; Bard, A.J. Interaction of silver(I) ions with the respiratory chain of Escherichia coli: An electrochemical and scanning electrochemical microscopy study of the antimicrobial mechanism of micromolar Ag+. Biochemistry 2005, 44, 13214–13223. [Google Scholar] [CrossRef]
- Kaur, P.; Vadehra, D.V. Mechanism of resistance to silver ions in Klebsiella pneumoniae. Antimicrob. Agents Chemother. 1986, 29, 165–167. [Google Scholar] [CrossRef] [PubMed]
- Gordon, O.; Vig Slenters, T.; Brunetto, P.S.; Villaruz, A.E.; Sturdevant, D.E.; Otto, M.; Landmann, R.; Fromm, K.M. Silver coordination polymers for prevention of implant infection: Thiol interaction, impact on respiratory chain enzymes, and hydroxyl radical induction. Antimicrob. Agents Chemother. 2010, 54, 4208–4218. [Google Scholar] [CrossRef]
- Slavin, Y.N.; Asnis, J.; Häfeli, U.O.; Bach, H. Metal nanoparticles: Understanding the mechanisms behind antibacterial activity. J. Nanobiotechnol. 2017, 15, 65. [Google Scholar] [CrossRef]
- Nikaido, H. Porins and specific diffusion channels in bacterial outer membranes. J. Biol. Chem. 1994, 269, 3905–3908. [Google Scholar] [CrossRef]
- Pugsley, A.P.; Schnaitman, C.A. Outer membrane proteins of Escherichia coli. VII. Evidence that bacteriophage-directed protein 2 functions as a pore. J. Bacteriol. 1978, 133, 1181–1189. [Google Scholar] [CrossRef]
- Franci, G.; Falanga, A.; Galdiero, S.; Palomba, L.; Rai, M.; Morelli, G.; Galdiero, M. Silver nanoparticles as potential antibacterial agents. Molecules 2015, 20, 8856–8874. [Google Scholar] [CrossRef]
- Li, X.Z.; Nikaido, H.; Williams, K.E. Silver-resistant mutants of Escherichia coli display active efflux of Ag+ and are deficient in porins. J. Bacteriol. 1997, 179, 6127–6132. [Google Scholar] [CrossRef]
- Radzig, M.A.; Nadtochenko, V.A.; Koksharova, O.A.; Kiwi, J.; Lipasova, V.A.; Khmel, I.A. Antibacterial effects of silver nanoparticles on gram-negative bacteria: Influence on the growth and biofilms formation, mechanisms of action. Colloids Surf. B Biointerfaces 2013, 102, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Girma, A. Alternative mechanisms of action of metallic nanoparticles to mitigate the global spread of antibiotic-resistant bacteria. Cell Surf. 2023, 10, 100112. [Google Scholar] [CrossRef]
- Mikhailova, E.O. Silver Nanoparticles: Mechanism of Action and Probable Bio-Application. J. Funct. Biomater. 2020, 11, 84. [Google Scholar] [CrossRef]
- Mikhailova, E.O. Green Silver Nanoparticles: An Antibacterial Mechanism. Antibiotics 2025, 14, 5. [Google Scholar] [CrossRef]
- Rai, M.K.; Deshmukh, S.D.; Ingle, A.P.; Gade, A.K. Silver nanoparticles: The powerful nanoweapon against multidrug-resistant bacteria. J. Appl. Microbiol. 2012, 112, 841–852. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, M.; Hara, K.; Kudo, J. Bactericidal actions of a silver ion solution on Escherichia coli, studied by energy-filtering transmission electron microscopy and proteomic analysis. Appl. Environ. Microbiol. 2005, 71, 7589–7593. [Google Scholar] [CrossRef]
- Modi, S.K.; Gaur, S.; Sengupta, M.; Singh, M.S. Mechanistic insights into nanoparticle surface-bacterial membrane interactions in overcoming antibiotic resistance. Front. Microbiol. 2023, 14, 1135579. [Google Scholar] [CrossRef]
- Behdad, R.; Pargol, M.; Mirzaie, A.; Karizi, S.Z.; Noorbazargan, H.; Akbarzadeh, I. Efflux pump inhibitory activity of biologically synthesized silver nanoparticles against multidrug-resistant Acinetobacter baumannii clinical isolates. J. Basic Microbiol. 2020, 60, 494–507. [Google Scholar] [CrossRef]
- Abdolhosseini, M.; Zamani, H.; Salehzadeh, A. Synergistic antimicrobial potential of ciprofloxacin with silver nanoparticles conjugated to thiosemicarbazide against ciprofloxacin resistant Pseudomonas aeruginosa by attenuation of MexA-B efflux pump genes. Biologia 2019, 74, 1191–1196. [Google Scholar] [CrossRef]
- Madhi, M.; Hasani, A.; Mojarrad, J.S.; Rezaee, M.A.; Zarrini, G.; Davaran, S.; Alizadeh, E.; Sheikhalizadeh, V. Impact of chitosan and silver nanoparticles laden with antibiotics on multidrug-resistant Pseudomonas aeruginosa and Acinetobacter baumannii. Arch. Clin. Infect. Dis. 2020, 15, e100195. [Google Scholar] [CrossRef]
- Sadat Shandiz, S.A.; Montazeri, A.; Abdolhosseini, M.; Hadad Shahrestani, S.; Hedayati, M.; Moradi-Shoeili, Z.; Salehzadeh, A. Functionalization of Ag Nanoparticles by Glutamic Acid and Conjugation of Ag@Glu by Thiosemicarbazide Enhances the Apoptosis of Human Breast Cancer MCF-7 Cells. J. Clust. Sci. 2018, 29, 1107–1114. [Google Scholar] [CrossRef]
- Dedon, P.C.; Plastaras, J.P.; Rouzer, C.A.; Marnett, L.J. Indirect mutagenesis by oxidative DNA damage: Formation of the pyrimidopurinone adduct of deoxyguanosine by base propenal. Proc. Natl. Acad. Sci. USA 1998, 95, 11113–11116. [Google Scholar] [CrossRef]
- Juan, C.A.; Pérez de la Lastra, J.M.; Plou, F.J.; Pérez-Lebeña, E. The Chemistry of Reactive Oxygen Species (ROS) Revisited: Outlining Their Role in Biological Macromolecules (DNA, Lipids and Proteins) and Induced Pathologies. Int. J. Mol. Sci. 2021, 22, 4642. [Google Scholar] [CrossRef]
- Valavanidis, A.; Vlachogianni, T.; Fiotakis, C. 8-hydroxy-2′ -deoxyguanosine (8-OHdG): A critical biomarker of oxidative stress and carcinogenesis. J. Environ. Sci. Health C Environ. Carcinog. Ecotoxicol. Rev. 2009, 27, 120–139. [Google Scholar] [CrossRef]
- Rodríguez-García, A.; García-Vicente, R.; Morales, M.L.; Ortiz-Ruiz, A.; Martínez-López, J.; Linares, M. Protein Carbonylation and Lipid Peroxidation in Hematological Malignancies. Antioxidants 2020, 9, 1212. [Google Scholar] [CrossRef] [PubMed]
- Qian, H.; Zhu, K.; Lu, H.; Lavoie, M.; Chen, S.; Zhou, Z.; Deng, Z.; Chen, J.; Fu, Z. Contrasting silver nanoparticle toxicity and detoxification strategies in Microcystis aeruginosa and Chlorella vulgaris: New insights from proteomic and physiological analyses. Sci. Total Environ. 2016, 572, 1213–1221. [Google Scholar] [CrossRef]
- Nallanthighal, S.; Chan, C.; Murray, T.M.; Mosier, A.P.; Cady, N.C.; Reliene, R. Differential effects of silver nanoparticles on DNA damage and DNA repair gene expression in Ogg1-deficient and wild type mice. Nanotoxicology 2017, 11, 996–1011. [Google Scholar] [CrossRef]
- Xu, L.; Wang, Y.Y.; Huang, J.; Chen, C.Y.; Wang, Z.X.; Xie, H. Silver nanoparticles: Synthesis, medical applications and biosafety. Theranostics 2020, 10, 8996–9031. [Google Scholar] [CrossRef] [PubMed]
- Feng, Q.L.; Wu, J.; Chen, G.Q.; Cui, F.Z.; Kim, T.N.; Kim, J.O. A mechanistic study of the antibacterial effect of silver ions on Escherichia coli and Staphylococcus aureus. J. Biomed. Mater. Res. 2000, 52, 662–668. [Google Scholar] [CrossRef]
- Ishida, T. Anticancer activities of silver ions in cancer and tumor cells and DNA damages by Ag+-DNA base-pairs reactions. MOJ Tumor Res. 2017, 1, 8–16. [Google Scholar]
- Balaure, P.C.; Grumezescu, A.M. Recent Advances in Surface Nanoengineering for Biofilm Prevention and Control. Part II: Active, Combined Active and Passive, and Smart Bacteria-Responsive Antibiofilm Nanocoatings. Nanomaterials 2020, 10, 1527. [Google Scholar] [CrossRef]
- Agarwal, H.; Gurnani, B.; Pippal, B.; Jain, N. Capturing the micro-communities: Insights into biogenesis and architecture of bacterial biofilms. BBA Adv. 2025, 7, 100133. [Google Scholar] [CrossRef]
- Seebach, E.; Elschner, T.; Kraus, F.V.; Souto-Carneiro, M.; Kubatzky, K.F. Bacterial and Metabolic Factors of Staphylococcal Planktonic and Biofilm Environments Differentially Regulate Macrophage Immune Activation. Inflammation 2023, 46, 1512–1530. [Google Scholar] [CrossRef] [PubMed]
- Rather, M.A.; Gupta, K.; Mandal, M. Microbial biofilm: Formation, architecture, antibiotic resistance, and control strategies. Braz. J. Microbiol. 2021, 52, 1701–1718. [Google Scholar] [CrossRef]
- Tian, H.; Liao, Q.; Liu, M.; Hou, J.; Zhang, Y.; Liu, J. Antibacterial activity of silver nanoparticles target sara through srna-teg49, a key mediator of hfq, in Staphylococcus aureus. Int. J. Clin. Exp. Med. 2015, 8, 5794–5799. [Google Scholar]
- Joshi, A.S.; Singh, P.; Mijakovic, I. Interactions of Gold and Silver Nanoparticles with Bacterial Biofilms: Molecular Interactions behind Inhibition and Resistance. Int. J. Mol. Sci. 2020, 21, 7658. [Google Scholar] [CrossRef]
- Fröhlich, K.S.; Gottesman, S. Small Regulatory RNAs in the Enterobacterial Response to Envelope Damage and Oxidative Stress. In Regulating with RNA in Bacteria and Archaea; Storz, G., Papenfort, K., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2018; pp. 211–228. [Google Scholar]
- Ren, J.; Nong, N.T.; Lam Vo, P.N.; Lee, H.M.; Na, D. Rational Design of High-Efficiency Synthetic Small Regulatory RNAs and Their Application in Robust Genetic Circuit Performance Through Tight Control of Leaky Gene Expression. ACS Synth. Biol. 2024, 13, 3256–3267. [Google Scholar] [CrossRef]
- González-Fernández, S.; Blanco-Agudín, N.; Rodríguez, D.; Fernández-Vega, I.; Merayo-Lloves, J.; Quirós, L.M. Silver Nanoparticles: A Versatile Tool Against Infectious and Non-Infectious Diseases. Antibiotics 2025, 14, 289. [Google Scholar] [CrossRef]
- Cress, B.F.; Englaender, J.A.; He, W.; Kasper, D.; Linhardt, R.J.; Koffas, M.A. Masquerading microbial pathogens: Capsular polysaccharides mimic host-tissue molecules. FEMS Microbiol. Rev. 2014, 38, 660–697. [Google Scholar] [CrossRef]
- Kohanski, M.A.; Dwyer, D.J.; Collins, J.J. How antibiotics kill bacteria: From targets to networks. Nat. Rev. Microbiol. 2010, 8, 423–435. [Google Scholar] [CrossRef]
- Guan, J.; Wang, J.; Zhang, X.; Chi, J.; Ma, Z.; Zhang, X. Silver nanoparticles with multimodal biological activities integrated into advanced material platforms for chronic wound management. Nanoscale 2025, 17, 18409–18445. [Google Scholar] [CrossRef] [PubMed]
- Carvalho-Silva, J.M.; Reis, A.C.D. Anti-inflammatory action of silver nanoparticles in vivo: Systematic review and meta-analysis. Heliyon 2024, 10, e34564. [Google Scholar] [CrossRef]
- Jalil, K.; Ahmad, S.; Islam, N.u.; Muhammad, S.; Jalil, Q.; Ali, A. Excellent antibacterial and anti-inflammatory efficacy of amoxicillin by AgNPs and their conjugates synthesized using Micromeria biflora crude flavonoid extracts. Heliyon 2024, 10, e36752. [Google Scholar] [CrossRef]
- Moldovan, B.; David, L.; Vulcu, A.; Olenic, L.; Perde-Schrepler, M.; Fischer-Fodor, E.; Baldea, I.; Clichici, S.; Filip, G.A. In vitro and in vivo anti-inflammatory properties of green synthesized silver nanoparticles using Viburnum opulus L. fruits extract. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 79, 720–727. [Google Scholar] [CrossRef]
- Singh, P.; Ahn, S.; Kang, J.P.; Veronika, S.; Huo, Y.; Singh, H.; Chokkaligam, M.; El-Agamy Farh, M.; Aceituno, V.C.; Kim, Y.J.; et al. In vitro anti-inflammatory activity of spherical silver nanoparticles and monodisperse hexagonal gold nanoparticles by fruit extract of Prunus serrulata: A green synthetic approach. Artif. Cells Nanomed. Biotechnol. 2018, 46, 2022–2032. [Google Scholar] [CrossRef] [PubMed]
- Crisan, D.; Scharffetter-Kochanek, K.; Crisan, M.; Schatz, S.; Hainzl, A.; Olenic, L.; Filip, A.; Schneider, L.A.; Sindrilaru, A. Topical silver and gold nanoparticles complexed with Cornus mas suppress inflammation in human psoriasis plaques by inhibiting NF-κB activity. Exp. Dermatol. 2018, 27, 1166–1169. [Google Scholar] [CrossRef] [PubMed]
- You, C.; Zhu, Z.; Wang, S.; Wang, X.; Han, C.; Shao, H. Nanosilver alleviates foreign body reaction and facilitates wound repair by regulating macrophage polarization. J. Zhejiang Univ. Sci. B 2023, 24, 510–523. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Guan, M.; Ren, R.; Gao, C.; Cheng, H.; Li, Y.; Gao, B.; Wei, Y.; Fu, J.; Sun, J. Improved immunoregulation of ultra-low-dose silver nanoparticle-loaded TiO2 nanotubes via M2 macrophage polarization by regulating GLUT1 and autophagy. Int. J. Nanomed. 2020, 15, 2011–2026. [Google Scholar] [CrossRef]
- Du, J.; Liu, X.; Wong, C.W.Y.; Lok, C.N.; Yang, Z.; Yuan, Z.; Wong, K.K.Y. Silver nanoparticles promote osteogenic differentiation of mouse embryonic fibroblasts in vitro. Am. J. Stem Cells 2023, 12, 51–59. [Google Scholar] [PubMed]
- Xu, Y.; Zheng, B.; He, J.; Cui, Z.; Liu, Y. Silver nanoparticles promote osteogenic differentiation of human periodontal ligament fibroblasts by regulating the RhoA-TAZ axis. Cell Biol. Int. 2019, 43, 910–920. [Google Scholar] [CrossRef] [PubMed]
- Long, L.Y.; Hu, C.; Liu, W.; Wu, C.; Lu, L.; Yang, L.; Wang, Y.B. Microfibrillated cellulose-enhanced carboxymethyl chitosan/oxidized starch sponge for chronic diabetic wound repair. Mater. Sci. Eng. C Mater. Biol. Appl. 2022, 135, 112669. [Google Scholar] [CrossRef] [PubMed]
- Gaikwad, S.; Birla, S.; Ingle, A.P.; Gade, A.; Ingle, P.; Golińska, P.; Rai, M. Superior in vivo Wound-Healing Activity of Mycosynthesized Silver Nanogel on Different Wound Models in Rat. Front. Microbiol. 2022, 13, 881404. [Google Scholar] [CrossRef]
- Liu, X.; Lee, P.Y.; Ho, C.M.; Lui, V.C.; Chen, Y.; Che, C.M.; Tam, P.K.; Wong, K.K. Silver nanoparticles mediate differential responses in keratinocytes and fibroblasts during skin wound healing. ChemMedChem 2010, 5, 468–475. [Google Scholar] [CrossRef]
- Zhang, S.; He, T.; Zhao, F.; Tan, Q.; Li, D.; Wang, Q.; Xiao, Y.; Zhang, X. Development of a multifunctional nano-hydroxyapatite platform (nHEA) for advanced treatment of severely infected full-thickness skin wounds. Acta Biomater. 2024, 181, 440–452. [Google Scholar] [CrossRef]
- Mensah, R.A.; Trotta, F.; Briggs, E.; Sharifulden, N.S.; Silva, L.V.B.; Keskin-Erdogan, Z.; Diop, S.; Kureshi, A.K.; Chau, D.Y.S. A Sustainable, Green-Processed, Ag-Nanoparticle-Incorporated Eggshell-Derived Biomaterial for Wound-Healing Applications. J. Funct. Biomater. 2023, 14, 450. [Google Scholar] [CrossRef]
- Sabarees, G.; Velmurugan, V.; Tamilarasi, G.P.; Alagarsamy, V.; Raja Solomon, V. Recent Advances in Silver Nanoparticles Containing Nanofibers for Chronic Wound Management. Polymers 2022, 14, 3994. [Google Scholar] [CrossRef]
- Krishnan, N.; Velramar, B.; Ramatchandirin, B.; Abraham, G.C.; Duraisamy, N.; Pandiyan, R.; Velu, R.K. Effect of biogenic silver nanocubes on matrix metalloproteinases 2 and 9 expressions in hyperglycemic skin injury and its impact in early wound healing in streptozotocin-induced diabetic mice. Mater. Sci. Eng. C Mater. Biol. Appl. 2018, 91, 146–152. [Google Scholar] [CrossRef]
- Seo, S.B.; Dananjaya, S.H.S.; Nikapitiya, C.; Park, B.K.; Gooneratne, R.; Kim, T.Y.; Lee, J.; Kim, C.H.; De Zoysa, M. Silver nanoparticles enhance wound healing in zebrafish (Danio rerio). Fish. Shellfish Immunol. 2017, 68, 536–545. [Google Scholar] [CrossRef]
- Xue, H.; Zhu, C.; Wang, Y.; Gu, Q.; Shao, Y.; Jin, A.; Zhang, X.; Lei, L.; Li, Y. Stimulus-responsive cellulose hydrogels in biomedical applications and challenges. Mater. Today Bio 2025, 32, 101814. [Google Scholar] [CrossRef]
- Raho, R.; Nguyen, N.Y.; Zhang, N.; Jiang, W.; Sannino, A.; Liu, H.; Pollini, M.; Paladini, F. Photo-assisted green synthesis of silver doped silk fibroin/carboxymethyl cellulose nanocomposite hydrogels for biomedical applications. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 107, 110219. [Google Scholar] [CrossRef]
- Satheeshkumar, E.; Yang, J. Analyte-induced photoreduction method for visual and colorimetric detection of tyrosine. Anal. Chim. Acta 2015, 879, 111–117. [Google Scholar] [CrossRef]
- Maddinedi, S.B.; Mandal, B.K.; Anna, K.K. Tyrosine assisted size controlled synthesis of silver nanoparticles and their catalytic, in-vitro cytotoxicity evaluation. Environ. Toxicol. Pharmacol. 2017, 51, 23–29. [Google Scholar] [CrossRef]
- Ruffo, M.; Parisi, O.I.; Dattilo, M.; Patitucci, F.; Malivindi, R.; Pezzi, V.; Tzanov, T.; Puoci, F. Synthesis and evaluation of wound healing properties of hydro-diab hydrogel loaded with green-synthetized AGNPS: In vitro and in ex vivo studies. Drug Deliv. Transl. Res. 2022, 12, 1881–1894. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Yao, J.; Huo, S.; Xu, C.; Yang, R.; Tao, D.; Fang, B.; Ma, G.; Zhu, Z.; Zhang, Y.; et al. Designing injectable dermal matrix hydrogel combined with silver nanoparticles for methicillin-resistant Staphylococcus aureus infected wounds healing. Nano Converg. 2024, 11, 41. [Google Scholar] [CrossRef] [PubMed]
- Sharifiaghdam, M.; Shaabani, E.; Faridi-Majidi, R.; De Smedt, S.C.; Braeckmans, K.; Fraire, J.C. Macrophages as a therapeutic target to promote diabetic wound healing. Mol. Ther. 2022, 30, 2891–2908. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Jiang, T.; Liu, S.; He, Y.; Yang, G.; Nie, J.; Wang, F.; Yang, X.; Chen, Z.; Lu, C. AgNPs loaded adenine-modified chitosan composite POSS-PEG hybrid hydrogel with enhanced antibacterial and cell proliferation properties for promotion of infected wound healing. Int. J. Biol. Macromol. 2024, 267, 131575. [Google Scholar] [CrossRef]
- Li, C.; Jiang, T.; Zhou, C.; Jiang, A.; Lu, C.; Yang, G.; Nie, J.; Wang, F.; Yang, X.; Chen, Z. Injectable self-healing chitosan-based POSS-PEG hybrid hydrogel as wound dressing to promote diabetic wound healing. Carbohydr. Polym. 2023, 299, 120198. [Google Scholar] [CrossRef]
- Iranpour Mobarakeh, A.; Shahmoradi Ramsheh, A.; Khanshan, A.; Aghaei, S.; Mirbagheri, M.S.; Esmaeili, J. Fabrication and evaluation of a bi-layered electrospun PCL/PVA patch for wound healing: Release of vitamins and silver nanoparticle. Heliyon 2024, 10, e33178. [Google Scholar] [CrossRef]
- Bayram Sarıipek, F. Biopolymeric nanofibrous scaffolds of poly(3-hydroxybuthyrate)/chitosan loaded with biogenic silver nanoparticle synthesized using curcumin and their antibacterial activities. Int. J. Biol. Macromol. 2024, 256, 128330. [Google Scholar] [CrossRef]
- Liu, W.; Thomopoulos, S.; Xia, Y. Electrospun nanofibers for regenerative medicine. Adv. Healthc. Mater. 2012, 1, 10–25. [Google Scholar] [CrossRef]
- Chen, S.; Liu, B.; Carlson, M.A.; Gombart, A.F.; Reilly, D.A.; Xie, J. Recent advances in electrospun nanofibers for wound healing. Nanomedicine 2017, 12, 1335–1352. [Google Scholar] [CrossRef]
- Zhang, S.; Yang, W.; Gong, W.; Lu, Y.; Yu, D.G.; Liu, P. Recent progress of electrospun nanofibers as burning dressings. RSC Adv. 2024, 14, 14374–14391. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Chen, L.; Ma, A.; Bai, X.; Zeng, Y.; Liu, D.; Liu, B.; Zhang, W.; Tang, S. Recent advances in coaxial electrospun nanofibers for wound healing. Mater. Today Bio 2024, 29, 101309. [Google Scholar] [CrossRef]
- Lucian, B.I.; Cheregi, C.D.; Sebastian, H.M.; Ruxandra-Florina, B.; Maghiar, L.; Ilarie, B.; Anca, H.; Sachelarie, L.; Mircea-Ioan, S. Electrospun Nanofibers in Wound Healing: Real-World Evaluation of Spincare™ Technology. Bioengineering 2025, 12, 500. [Google Scholar] [CrossRef]
- Yadav, P.D.; Londhe, P.V.; Chavan, S.S.; Mohite, D.D.; Firame, G.B.; Kadam, S.S.; Patil, M.J.; Ansari, M.I. Electrospun composite nanofibers for wound healing: Synthesis, characterization, and clinical potential of biopolymer-based materials. Discov. Mater. 2024, 4, 99. [Google Scholar] [CrossRef]
- Ambekar, R.S.; Kandasubramanian, B. Advancements in nanofibers for wound dressing: A review. Eur. Polym. J. 2019, 117, 304–336. [Google Scholar] [CrossRef]
- Liu, C.; Zhu, Y.; Lun, X.; Sheng, H.; Yan, A. Effects of wound dressing based on the combination of silver@curcumin nanoparticles and electrospun chitosan nanofibers on wound healing. Bioengineered 2022, 13, 4328–4339. [Google Scholar] [CrossRef]
- Yang, J.; Wang, K.; Yu, D.G.; Yang, Y.; Bligh, S.W.A.; Williams, G.R. Electrospun Janus nanofibers loaded with a drug and inorganic nanoparticles as an effective antibacterial wound dressing. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 111, 110805. [Google Scholar] [CrossRef]
- Kim, J.; Abu Al-Rub, R.K.; Han, S.M. High-resilience conductive PVA+AgNW/PDMS nanocomposite via directional freeze-drying. Extrem. Mech. Lett. 2024, 68, 102132. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, Z.; Xu, L.; Wang, H.; Peng, S.; Zheng, L.; Yang, Z.; Wu, L.; Miao, J.-T. Preparation of silver-plated carbon nanotubes/carbon fiber hybrid fibers by combining freeze-drying deposition with a sizing process to enhance the mechanical properties of carbon fiber composites. Compos. Part A Appl. Sci. Manuf. 2021, 146, 106421. [Google Scholar] [CrossRef]
- Lu, B.; Lu, F.; Zou, Y.; Liu, J.; Rong, B.; Li, Z.; Dai, F.; Wu, D.; Lan, G. In situ reduction of silver nanoparticles by chitosan-l-glutamic acid/hyaluronic acid: Enhancing antimicrobial and wound-healing activity. Carbohydr. Polym. 2017, 173, 556–565. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Zhao, X.; Li, M.; Yan, L.; Lu, Y.; Jiang, C.; Liu, Y.; Pan, Z.; Shi, J. Antibacterial and wound healing-promoting effect of sponge-like chitosan-loaded silver nanoparticles biosynthesized by iturin. Int. J. Biol. Macromol. 2021, 181, 1183–1195. [Google Scholar] [CrossRef]
- Zhao, X.; Wang, K.; Ai, C.; Yan, L.; Jiang, C.; Shi, J. Improvement of antifungal and antibacterial activities of food packages using silver nanoparticles synthesized by iturin A. Food Packag. Shelf Life 2021, 28, 100669. [Google Scholar] [CrossRef]
- Liang, D.; Lu, Z.; Yang, H.; Gao, J.; Chen, R. Novel Asymmetric Wettable AgNPs/Chitosan Wound Dressing: In Vitro and In Vivo Evaluation. ACS Appl. Mater. Interfaces 2016, 8, 3958–3968. [Google Scholar] [CrossRef]
- Dong, Q.; Liang, X.; Chen, F.; Ke, M.; Yang, X.; Ai, J.; Cheng, Q.; Zhou, Y.; Chen, Y. Injectable shape memory hydroxyethyl cellulose/soy protein isolate based composite sponge with antibacterial property for rapid noncompressible hemorrhage and prevention of wound infection. Int. J. Biol. Macromol. 2022, 217, 367–380. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, Y.; Du, Q.; Zhang, Q.; Lv, X.; Yang, Q.; Chang, P.R.; Anderson, D.P.; He, M.; Chen, Y. Shape memory histocompatible and biodegradable sponges for subcutaneous defect filling and repair: Greatly reducing surgical incision. J. Mater. Chem. B 2019, 7, 5848–5860. [Google Scholar] [CrossRef]
- Zhao, Y.; He, M.; Jin, H.; Zhao, L.; Du, Q.; Deng, H.; Tian, W.; Li, Y.; Lv, X.; Chen, Y. Construction of highly biocompatible hydroxyethyl cellulose/soy protein isolate composite sponges for tissue engineering. Chem. Eng. J. 2018, 341, 402–413. [Google Scholar] [CrossRef]
- Ottaviano, L.; Buoso, S.; Zamboni, R.; Sotgiu, G.; Posati, T. Natural Protein Films from Textile Waste for Wound Healing and Wound Dressing Applications. J. Funct. Biomater. 2025, 16, 20. [Google Scholar] [CrossRef]
- Benkhira, I.; Zermane, F.; Cheknane, B.; Trache, D.; Brosse, N.; Paolone, A.; Chader, H.; Sobhi, W. Preparation and characterization of amidated pectin-gelatin-oxidized tannic acid hydrogel films supplemented with in-situ reduced silver nanoparticles for wound-dressing applications. Int. J. Biol. Macromol. 2024, 277, 134158. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Pang, L.; Duan, J.; Huang, N.; Chen, X.; Huang, W.; Liu, Y.; Fu, C.; Zhang, C.; Tu, H.; et al. Eco-friendly antibacterial electrospinning nanofibrous film containing nano-silver green-synthesized by natural glycoprotein for infected wound healing. J. Colloid Interface Sci. 2025, 683, 256–268. [Google Scholar] [CrossRef] [PubMed]
- Pang, L.; Liao, Q.; Zou, L.; Zhang, C.; Nie, X.; Yi, Z.; Fu, C.; Zhang, J. Two glycoproteins from medicinal insect Periplaneta americana (L.) promote diabetic wound healing via macrophage polarization modulation. Int. J. Biol. Macromol. 2022, 209, 2130–2141. [Google Scholar] [CrossRef] [PubMed]
- Gollapudi, K.K.; Dutta, S.D.; Adnan, M.; Taylor, M.L.; Reddy, K.V.N.S.; Alle, M.; Huang, X. Dialdehyde cellulose nanofibrils/polyquaternium stabilized ultra-fine silver nanoparticles for synergistic antibacterial therapy. Int. J. Biol. Macromol. 2024, 280, 135971. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.; Dou, Z.; Zhang, Y.; Li, F.; Xing, B.; Hu, Z.; Li, X.; Liu, Z.; Yang, W.; Liu, Z. Smart Responsive and Controlled-Release Hydrogels for Chronic Wound Treatment. Pharmaceutics 2023, 15, 2735. [Google Scholar] [CrossRef]
- Fauzian, F.; Garmana, A.N.; Mauludin, R. Applications of Nanotechnology-Based Drug Delivery System for Delivering Natural Products into Acute and Chronic Wounds: A Review. Biointerface Res. Appl. Chem. 2023, 13, 426. [Google Scholar] [CrossRef]
- Hu, C.; Long, L.; Cao, J.; Zhang, S.; Wang, Y. Dual-crosslinked mussel-inspired smart hydrogels with enhanced antibacterial and angiogenic properties for chronic infected diabetic wound treatment via pH-responsive quick cargo release. Chem. Eng. J. 2021, 411, 128564. [Google Scholar] [CrossRef]
- Hu, C.; Zhang, F.; Kong, Q.; Lu, Y.; Zhang, B.; Wu, C.; Luo, R.; Wang, Y. Synergistic Chemical and Photodynamic Antimicrobial Therapy for Enhanced Wound Healing Mediated by Multifunctional Light-Responsive Nanoparticles. Biomacromolecules 2019, 20, 4581–4592. [Google Scholar] [CrossRef]
- Della Vecchia, N.F.; Avolio, R.; Alfè, M.; Errico, M.E.; Napolitano, A.; d’Ischia, M. Building-Block Diversity in Polydopamine Underpins a Multifunctional Eumelanin-Type Platform Tunable Through a Quinone Control Point. Adv. Funct. Mater. 2013, 23, 1331–1340. [Google Scholar] [CrossRef]
- Burzio, L.A.; Waite, J.H. Cross-linking in adhesive quinoproteins: Studies with model decapeptides. Biochemistry 2000, 39, 11147–11153. [Google Scholar] [CrossRef]
- Haidari, H.; Kopecki, Z.; Sutton, A.T.; Garg, S.; Cowin, A.J.; Vasilev, K. pH-Responsive “Smart” Hydrogel for Controlled Delivery of Silver Nanoparticles to Infected Wounds. Antibiotics 2021, 10, 49. [Google Scholar] [CrossRef]
- Qi, X.; Huang, Y.; You, S.; Xiang, Y.; Cai, E.; Mao, R.; Pan, W.; Tong, X.; Dong, W.; Ye, F.; et al. Engineering Robust Ag-Decorated Polydopamine Nano-Photothermal Platforms to Combat Bacterial Infection and Prompt Wound Healing. Adv. Sci. 2022, 9, e2106015. [Google Scholar] [CrossRef]
- Abdali, Z.; Yeganeh, H.; Solouk, A.; Gharibi, R.; Sorayya, M. Thermoresponsive antimicrobial wound dressings via simultaneous thiol-ene polymerization and in situ generation of silver nanoparticles. RSC Adv. 2015, 5, 66024–66036. [Google Scholar] [CrossRef]
- Wu, M.; Liu, M.; Feng, G.; Jia, R.; Chen, R.; Li, Y.; Yan, G.; Qiu, Z.J. NIR-triggered temperature-sensitive polydopamine nanosystem with rapid phase transition for on-demand release to enhance antibacterial and antibiofilm performance. Colloids Surf. B Biointerfaces 2025, 255, 114901. [Google Scholar] [CrossRef]
- Chen, Y.; Xing, Y.; Han, J.; Liu, S.; Xiang, X.; Shen, J.; Du, X.; Ma, X. Multifunctional MMP9-responsive silicasomes-GelMA hydrogels with bacteria-targeting capability and tissue restoration function for chronic wound infection. Chem. Eng. J. 2023, 475, 146246. [Google Scholar] [CrossRef]
- Michalicha, A.; Belcarz, A.; Giannakoudakis, D.A.; Staniszewska, M.; Barczak, M. Designing Composite Stimuli-Responsive Hydrogels for Wound Healing Applications: The State-of-the-Art and Recent Discoveries. Materials 2024, 17, 278. [Google Scholar] [CrossRef]
- Wang, X.; Huang, J.; Zhao, J.; Peng, L.; Qi, L.; Wu, T.; He, J.; Gao, Y.; Zhou, Y. Dual stimuli-responsive and self-healing hyaluronic acid-based nanocomposite hydrogels for promoting wound healing of methicillin-resistant Staphylococcus aureus infection. Int. J. Biol. Macromol. 2025, 320, 145829. [Google Scholar] [CrossRef] [PubMed]
- Srikhao, N.; Theerakulpisut, S.; Chindaprasirt, P.; Okhawilai, M.; Narain, R.; Kasemsiri, P. Green synthesis of nano silver-embedded carboxymethyl starch waste/poly vinyl alcohol hydrogel with photothermal sterilization and pH-responsive behavior. Int. J. Biol. Macromol. 2023, 242, 125118. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Yang, X.; Liu, W.; Xi, G.; Wang, M.; Liang, B.; Ma, Z.; Feng, Y.; Chen, H.; Shi, C. Tannic Acid Cross-linked Polysaccharide-Based Multifunctional Hemostatic Microparticles for the Regulation of Rapid Wound Healing. Macromol. Biosci. 2018, 18, e1800209. [Google Scholar] [CrossRef]
- Orlowski, P.; Zmigrodzka, M.; Tomaszewska, E.; Ranoszek-Soliwoda, K.; Czupryn, M.; Antos-Bielska, M.; Szemraj, J.; Celichowski, G.; Grobelny, J.; Krzyzowska, M. Tannic acid-modified silver nanoparticles for wound healing: The importance of size. Int. J. Nanomed. 2018, 13, 991–1007. [Google Scholar] [CrossRef]
- Cao, C.; Yang, N.; Zhao, Y.; Yang, D.; Hu, Y.; Yang, D.; Song, X.; Wang, W.; Dong, X. Biodegradable hydrogel with thermo-response and hemostatic effect for photothermal enhanced anti-infective therapy. Nano Today 2021, 39, 101165. [Google Scholar] [CrossRef]
- Liu, Y.; Li, F.; Guo, Z.; Xiao, Y.; Zhang, Y.; Sun, X.; Zhe, T.; Cao, Y.; Wang, L.; Lu, Q.; et al. Silver nanoparticle-embedded hydrogel as a photothermal platform for combating bacterial infections. Chem. Eng. J. 2020, 382, 122990. [Google Scholar] [CrossRef]
- Melo, R.M.; Albuquerque, G.M.; Monte, J.P.; Pereira, G.A.L.; Pereira, G. Recent Advances in the Application of Silver Nanoparticles for Enhancing Phototherapy Outcomes. Pharmaceuticals 2025, 18, 970. [Google Scholar] [CrossRef]
- Du, T.; Xiao, Z.; Cao, J.; Wei, L.; Li, C.; Jiao, J.; Song, Z.; Liu, J.; Du, X.; Wang, S. NIR-activated multi-hit therapeutic Ag(2)S quantum dot-based hydrogel for healing of bacteria-infected wounds. Acta Biomater. 2022, 145, 88–105. [Google Scholar] [CrossRef]
- Shi, W.; Kong, Y.; Su, Y.; Kuss, M.A.; Jiang, X.; Li, X.; Xie, J.; Duan, B. Tannic acid-inspired, self-healing, and dual stimuli responsive dynamic hydrogel with potent antibacterial and anti-oxidative properties. J. Mater. Chem. B 2021, 9, 7182–7195. [Google Scholar] [CrossRef]
- Terriac, L.; Helesbeux, J.-J.; Maugars, Y.; Guicheux, J.; Tibbitt, M.W.; Delplace, V. Boronate Ester Hydrogels for Biomedical Applications: Challenges and Opportunities. Chem. Mater. 2024, 36, 6674–6695. [Google Scholar] [CrossRef]
- Deng, C.C.; Brooks, W.L.A.; Abboud, K.A.; Sumerlin, B.S. Boronic Acid-Based Hydrogels Undergo Self-Healing at Neutral and Acidic pH. ACS Macro Lett. 2015, 4, 220–224. [Google Scholar] [CrossRef] [PubMed]
- Haidari, H.; Vasilev, K.; Cowin, A.J.; Kopecki, Z. Bacteria-Activated Dual pH- and Temperature-Responsive Hydrogel for Targeted Elimination of Infection and Improved Wound Healing. ACS Appl. Mater. Interfaces 2022, 14, 51744–51762. [Google Scholar] [CrossRef] [PubMed]
- Khadem, E.; Kharaziha, M.; Salehi, S. Colorimetric pH-responsive and hemostatic hydrogel-based bioadhesives containing functionalized silver nanoparticles. Mater. Today Bio 2023, 20, 100650. [Google Scholar] [CrossRef]
- Khadem, E.; Kharaziha, M. Red cabbage anthocyanin- functionalized tannic acid-silver nanoparticles with pH sensitivity and antibacterial properties. Mater. Chem. Phys. 2022, 291, 126689. [Google Scholar] [CrossRef]
- Edwards-Jones, V. Silver nanoparticles: An overview of scientific toxicity and safety data and introduction of a new dressing, Venus Ag. Wounds UK 2022, 18, 22. [Google Scholar]
- Kumar, S.S.D.; Rajendran, N.K.; Houreld, N.N.; Abrahamse, H. Recent advances on silver nanoparticle and biopolymer-based biomaterials for wound healing applications. Int. J. Biol. Macromol. 2018, 115, 165–175. [Google Scholar] [CrossRef]
- Xu, M.; Qi, Y.; Liu, G.; Song, Y.; Jiang, X.; Du, B. Size-Dependent In Vivo Transport of Nanoparticles: Implications for Delivery, Targeting, and Clearance. ACS Nano 2023, 17, 20825–20849. [Google Scholar] [CrossRef] [PubMed]
- Panja, P.; Jana, N.R. Arginine-Terminated Nanoparticles of <10 nm Size for Direct Membrane Penetration and Protein Delivery for Straight Access to Cytosol and Nucleus. J. Phys. Chem. Lett. 2020, 11, 2363–2368. [Google Scholar] [CrossRef] [PubMed]
- Walker, M.; Parsons, D. The biological fate of silver ions following the use of silver-containing wound care products—A review. Int. Wound J. 2014, 11, 496–504. [Google Scholar] [CrossRef]
- Schleh, C.; Semmler-Behnke, M.; Lipka, J.; Wenk, A.; Hirn, S.; Schäffler, M.; Schmid, G.; Simon, U.; Kreyling, W.G. Size and surface charge of gold nanoparticles determine absorption across intestinal barriers and accumulation in secondary target organs after oral administration. Nanotoxicology 2012, 6, 36–46. [Google Scholar] [CrossRef]
- Trickler, W.J.; Lantz, S.M.; Murdock, R.C.; Schrand, A.M.; Robinson, B.L.; Newport, G.D.; Schlager, J.J.; Oldenburg, S.J.; Paule, M.G.; Slikker, W., Jr.; et al. Silver nanoparticle induced blood-brain barrier inflammation and increased permeability in primary rat brain microvessel endothelial cells. Toxicol. Sci. 2010, 118, 160–170. [Google Scholar] [CrossRef]
- Pfurtscheller, K.; Petnehazy, T.; Goessler, W.; Bubalo, V.; Kamolz, L.P.; Trop, M. Transdermal uptake and organ distribution of silver from two different wound dressings in rats after a burn trauma. Wound Repair Regen. 2014, 22, 654–659. [Google Scholar] [CrossRef]
- Hadrup, N.; Sharma, A.K.; Loeschner, K. Toxicity of silver ions, metallic silver, and silver nanoparticle materials after in vivo dermal and mucosal surface exposure: A review. Regul. Toxicol. Pharmacol. 2018, 98, 257–267. [Google Scholar] [CrossRef]
- Moiemen, N.S.; Shale, E.; Drysdale, K.J.; Smith, G.; Wilson, Y.T.; Papini, R. Acticoat dressings and major burns: Systemic silver absorption. Burns 2011, 37, 27–35. [Google Scholar] [CrossRef]
- Vlachou, E.; Chipp, E.; Shale, E.; Wilson, Y.T.; Papini, R.; Moiemen, N.S. The safety of nanocrystalline silver dressings on burns: A study of systemic silver absorption. Burns 2007, 33, 979–985. [Google Scholar] [CrossRef] [PubMed]
- Trop, M.; Novak, M.; Rodl, S.; Hellbom, B.; Kroell, W.; Goessler, W. Silver-coated dressing acticoat caused raised liver enzymes and argyria-like symptoms in burn patient. J. Trauma 2006, 60, 648–652. [Google Scholar] [CrossRef]
- Mimura, E.C.M.; Favoreto, J.P.M.; Favero, M.E.; Bonifacio, K.L.; Peixe, T.S.; Morita, A.A.; Barbosa, D.S.; Yabe, M.J.S.; Carrilho, A.J.F. Silver serum levels in burned patients treated with silver sulfadiazine and its toxicity on inflammatory cells. Burns 2020, 46, 1120–1127. [Google Scholar] [CrossRef] [PubMed]
- Maitre, S.; Jaber, K.; Perrot, J.-L.; Guy, C.; Cambazard, F. Élévation des taux sériques et urinaires d’argent au cours d’un traitement topique par sulfadiazine argentique (Flammazine®). In Proceedings of the Annales de Dermatologie et de Vénéréologie, Paris, France, 30 June 2002; pp. 217–219. [Google Scholar]
- Chaby, G.; Viseux, V.; Poulain, J.F.; De Cagny, B.; Denoeux, J.P.; Lok, C. Insuffisance rénale aiguë après application topique de sulfadiazine argentique. Ann. Dermatol. Vénéréologie 2005, 132, 891–893. [Google Scholar] [CrossRef] [PubMed]
- Brouillard, C.; Bursztejn, A.C.; Latarche, C.; Cuny, J.F.; Truchetet, F.; Goullé, J.P.; Schmutz, J.L. Silver absorption and toxicity evaluation of silver wound dressings in 40 patients with chronic wounds. J. Eur. Acad. Dermatol. Venereol. 2018, 32, 2295–2299. [Google Scholar] [CrossRef]
- Nadworny, P.L.; Landry, B.K.; Wang, J.; Tredget, E.E.; Burrell, R.E. Does nanocrystalline silver have a transferable effect? Wound Repair Regen. 2010, 18, 254–265. [Google Scholar] [CrossRef]
- Liu, J.; Wang, Z.; Liu, F.D.; Kane, A.B.; Hurt, R.H. Chemical Transformations of Nanosilver in Biological Environments. ACS Nano 2012, 6, 9887–9899. [Google Scholar] [CrossRef]
- Miclăuş, T.; Beer, C.; Chevallier, J.; Scavenius, C.; Bochenkov, V.E.; Enghild, J.J.; Sutherland, D.S. Dynamic protein coronas revealed as a modulator of silver nanoparticle sulphidation in vitro. Nat. Commun. 2016, 7, 11770. [Google Scholar] [CrossRef]
- Chen, X.; Jhee, K.H.; Kruger, W.D. Production of the neuromodulator H2S by cystathionine beta-synthase via the condensation of cysteine and homocysteine. J. Biol. Chem. 2004, 279, 52082–52086. [Google Scholar] [CrossRef] [PubMed]
- Omorou, M.; Liu, N.; Huang, Y.; Al-Ward, H.; Gao, M.; Mu, C.; Zhang, L.; Hui, X. Cystathionine beta-Synthase in hypoxia and ischemia/reperfusion: A current overview. Arch. Biochem. Biophys. 2022, 718, 109149. [Google Scholar] [CrossRef] [PubMed]
- Massi, D.; Santucci, M. Human generalized argyria: A submicroscopic and X-ray spectroscopic study. Ultrastruct. Pathol. 1998, 22, 47–53. [Google Scholar] [CrossRef]
- Payne, C.M.; Bladin, C.; Colchester, A.C.; Bland, J.; Lapworth, R.; Lane, D. Argyria from excessive use of topical silver sulphadiazine. Lancet 1992, 340, 126. [Google Scholar] [CrossRef]
- Van de Voorde, K.; Nijsten, T.; Schelfhout, K.; Moorkens, G.; Lambert, J. Long-term use of silver containing nose-drops resulting in systemic argyria. Acta Clin. Belg. 2005, 60, 33–35. [Google Scholar] [CrossRef]
- Cameron, S.J.; Hosseinian, F.; Willmore, W.G. A Current Overview of the Biological and Cellular Effects of Nanosilver. Int. J. Mol. Sci. 2018, 19, 2030. [Google Scholar] [CrossRef]
- Piao, M.J.; Kang, K.A.; Fernando, P.; Herath, H.; Hyun, J.W. Silver nanoparticle-induced cell damage via impaired mtROS-JNK/MnSOD signaling pathway. Toxicol. Mech. Methods 2024, 34, 803–812. [Google Scholar] [CrossRef]
- Coombs, C.J.; Wan, A.T.; Masterton, J.P.; Conyers, R.A.; Pedersen, J.; Chia, Y.T. Do burn patients have a silver lining? Burns 1992, 18, 179–184. [Google Scholar] [CrossRef]
- McCague, A.; Joe, V.C. A Case of Argyria and Acute Leukopenia Associated with the Use of an Antimicrobial Soft Silicone Foam Dressing. J. Burn Care Res. 2016, 37, e493–e496. [Google Scholar] [CrossRef]
- Aktepe, N.; Kocyigit, A.; Yukselten, Y.; Taskin, A.; Keskin, C.; Celik, H. Increased DNA damage and oxidative stress among silver jewelry workers. Biol. Trace Elem. Res. 2015, 164, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Rybka, M.; Mazurek, Ł.; Konop, M. Beneficial Effect of Wound Dressings Containing Silver and Silver Nanoparticles in Wound Healing—From Experimental Studies to Clinical Practice. Life 2023, 13, 69. [Google Scholar] [CrossRef]
- Shrestha, S.; Wang, B.; Dutta, P.K. Commercial Silver-Based Dressings: In Vitro and Clinical Studies in Treatment of Chronic and Burn Wounds. Antibiotics 2024, 13, 910. [Google Scholar] [CrossRef]
- Dias, M.; Zhang, R.; Lammers, T.; Pallares, R.M. Clinical translation and landscape of silver nanoparticles. Drug Deliv. Transl. Res. 2025, 15, 789–797. [Google Scholar] [CrossRef]
- Meekul, J.; Chotirosniramit, A.; Himakalasa, W.; Orrapin, S.; Wongthanee, A.; Pongtam, O.; Kulprachakarn, K.; Rerkasem, K. A Randomized Controlled Trial on the Outcome in Comparing an Alginate Silver Dressing with a Conventional Treatment of a Necrotizing Fasciitis Wound. Int. J. Low. Extrem. Wounds 2017, 16, 108–113. [Google Scholar] [CrossRef]
- Metcalf, D.G.; Bowler, P.G. Clinical impact of an anti-biofilm Hydrofiber dressing in hard-to-heal wounds previously managed with traditional antimicrobial products and systemic antibiotics. Burns Trauma 2020, 8, tkaa004. [Google Scholar] [CrossRef]
- Bowler, P.G.; Parsons, D. Combatting wound biofilm and recalcitrance with a novel anti-biofilm Hydrofiber® wound dressing. Wound Med. 2016, 14, 6–11. [Google Scholar] [CrossRef]
- Wang, R.; Guo, Y.; Li, B.; Zheng, J.; Tang, Z.; Shu, M. Application Effect of Silver-Containing Dressings in the Repair of Chronic Refractory Wounds. Evid. Based Complement. Altern. Med. 2022, 2022, 3616923. [Google Scholar] [CrossRef]
- Zhang, D.; Yao, D.; Ma, R.; Nan, S.; Lv, Y.; Zhu, Y.; Zhu, S. Effect of Silver Nanoparticles with Thermoplastic Polyurethane on Postoperative Rehabilitation of Diabetic Patients with Open Fracture of Lower Extremities. Front. Surg. 2022, 9, 954155. [Google Scholar] [CrossRef] [PubMed]
- Hurd, T.; Woodmansey, E.J.; Watkins, H.M.A. A retrospective review of the use of a nanocrystalline silver dressing in the management of open chronic wounds in the community. Int. Wound J. 2021, 18, 753–762. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.C.; Lee, H.C.; Chen, C.L.; Kuo, M.C.; Ramachandran, S.; Chen, R.F.; Kuo, Y.R. The Effects of Silver-Releasing Foam Dressings on Diabetic Foot Ulcer Healing. J. Clin. Med. 2021, 10, 1495. [Google Scholar] [CrossRef]
- Dissemond, J.; Aare, K.; Ozer, K.; Gandhi, D.; Ryan, J.L.; DeKoven, M. Aquacel Ag Advantage/Ag+ Extra and Cutimed Sorbact in the management of hard-to-heal wounds: A cohort study. J. Wound Care 2023, 32, 624–633. [Google Scholar] [CrossRef]
- Chan, R.K.; Nuutila, K.; Mathew-Steiner, S.S.; Diaz, V.; Anselmo, K.; Batchinsky, M.; Carlsson, A.; Ghosh, N.; Sen, C.K.; Roy, S. A Prospective, Randomized, Controlled Study to Evaluate the Effectiveness of a Fabric-Based Wireless Electroceutical Dressing Compared to Standard-of-Care Treatment Against Acute Trauma and Burn Wound Biofilm Infection. Adv. Wound Care 2024, 13, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Sen, C.K.; Mathew-Steiner, S.S.; Das, A.; Sundaresan, V.B.; Roy, S. Electroceutical Management of Bacterial Biofilms and Surgical Infection. Antioxid. Redox Signal. 2020, 33, 713–724. [Google Scholar] [CrossRef]
- Rada, B.; Leto, T.L. Pyocyanin effects on respiratory epithelium: Relevance in Pseudomonas aeruginosa airway infections. Trends Microbiol. 2013, 21, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Lafontaine, N.; Jolley, J.; Kyi, M.; King, S.; Iacobaccio, L.; Staunton, E.; Wilson, B.; Seymour, C.; Rogasch, S.; Wraight, P. Prospective randomised placebo-controlled trial assessing the efficacy of silver dressings to enhance healing of acute diabetes-related foot ulcers. Diabetologia 2023, 66, 768–776. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.; Dalziel, S.R.; Herd, E.; Johnson, K.; Wong She, R.; Shepherd, M. A Randomized Controlled Study of Silver-Based Burns Dressing in a Pediatric Emergency Department. J. Burn Care Res. 2016, 37, e340–e347. [Google Scholar] [CrossRef]
- Housler, G.J.; Cross, S.; Marcel, V.; Kennedy, D.O.; Husband, M.; Register, A.; Roberts, T.; Grubbs, S.; Dudewicz, D.; Setka, N.; et al. A Prospective Randomized Controlled Two-Arm Clinical Study Evaluating the Efficacy of a Bioelectric Dressing System for Blister Management in US Army Ranger Recruits. J. Spec. Oper. Med. 2017, 17, 49–58. [Google Scholar] [CrossRef]
- Hundeshagen, G.; Collins, V.N.; Wurzer, P.; Sherman, W.; Voigt, C.D.; Cambiaso-Daniel, J.; Nunez Lopez, O.; Sheaffer, J.; Herndon, D.N.; Finnerty, C.C.; et al. A Prospective, Randomized, Controlled Trial Comparing the Outpatient Treatment of Pediatric and Adult Partial-Thickness Burns with Suprathel or Mepilex Ag. J. Burn Care Res. 2018, 39, 261–267. [Google Scholar] [CrossRef]
- Aurora, A.; Beasy, A.; Rizzo, J.A.; Chung, K.K. The Use of a Silver-Nylon Dressing During Evacuation of Military Burn Casualties. J. Burn Care Res. 2018, 39, 593–597. [Google Scholar] [CrossRef]
- Moreira, S.S.; Camargo, M.C.; Caetano, R.; Alves, M.R.; Itria, A.; Pereira, T.V.; Lopes, L.C. Efficacy and costs of nanocrystalline silver dressings versus 1% silver sulfadiazine dressings to treat burns in adults in the outpatient setting: A randomized clinical trial. Burns 2022, 48, 568–576. [Google Scholar] [CrossRef]
- Jiang, X.; Khan, S.; Dykes, A.; Stulz, E.; Zhang, X. Biogenic Synthesis of Silver Nanoparticles and Their Diverse Biomedical Applications. Molecules 2025, 30, 3104. [Google Scholar] [CrossRef]
- Veličković, V.M.; Macmillan, T.; Lones, E.; Arlouskaya, Y.; Prieto, P.A.; Webb, N.; Crompton, A.; Munro, I.; Carvalho, V.F.; Attila, S.; et al. Systematic review and quality assessment of clinical and economic evidence for superabsorbent wound dressings in a population with chronic ulcers. Int. Wound J. 2024, 21, e14750. [Google Scholar] [CrossRef]
- Gould, L.J.; Liu, J.; Wan, R.; Carter, M.J.; Dotson, M.; Driver, V.R. Evidence supporting wound care end points relevant to clinical practice and patients’ lives. Part 3: The Patient Survey. Wound Repair Regen. 2021, 29, 60–69. [Google Scholar] [CrossRef]
- Rawal, M.; Singh, A.; Amiji, M.M. Quality-by-Design Concepts to Improve Nanotechnology-Based Drug Development. Pharm. Res. 2019, 36, 153. [Google Scholar] [CrossRef]
- Lin, C.; Huang, X.; Xue, Y.; Jiang, S.; Chen, C.; Liu, Y.; Chen, K. Advances in medical devices using nanomaterials and nanotechnology: Innovation and regulatory science. Bioact. Mater. 2025, 48, 353–369. [Google Scholar] [CrossRef]
- Taleuzzaman, M.; Jahangir, M.A.; Chauhan, S.; Kala, C.; Beg, S. Chapter 19—Good laboratory practice and current good manufacturing practice requirements in the development of cancer nanomedicines. In Nanoformulation Strategies for Cancer Treatment; Beg, S., Rahman, M., Choudhry, H., Souto, E.B., Ahmad, F.J., Eds.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 341–352. [Google Scholar]
- Vo, D.-K.; Trinh, K.T.L. Advances in Wearable Biosensors for Wound Healing and Infection Monitoring. Biosensors 2025, 15, 139. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Li, Z.; Xiao, J.; Nie, C. Conformable electrochemical devices for closed-loop wound management. Front. Bioeng. Biotechnol. 2023, 11, 1331567. [Google Scholar] [CrossRef] [PubMed]
- Ferdous, Z.; Nemmar, A. Health Impact of Silver Nanoparticles: A Review of the Biodistribution and Toxicity Following Various Routes of Exposure. Int. J. Mol. Sci. 2020, 21, 2375. [Google Scholar] [CrossRef]
- Huang, Y.; Chen, R.; Chen, Y.; Lü, X. Investigation of molecular mechanisms in silver nanoparticle-induced cytotoxicity from gene to metabolite level. Sci. Rep. 2025, 15, 26923. [Google Scholar] [CrossRef]
- Gao, X.; Li, R.; Yourick, J.J.; Sprando, R.L. Transcriptomic and proteomic responses of silver nanoparticles in hepatocyte-like cells derived from human induced pluripotent stem cells. Toxicol. Vitr. 2022, 79, 105274. [Google Scholar] [CrossRef] [PubMed]
- Vohra, M.; Kour, A.; Kalia, N.P.; Kumar, M.; Sharma, S.; Jaglan, S.; Kamath, N.; Sharma, S. A comprehensive review of genomics, transcriptomics, proteomics, and metabolomic insights into the differentiation of Pseudomonas aeruginosa from the planktonic to biofilm state: A multi-omics approach. Int. J. Biol. Macromol. 2024, 257, 128563. [Google Scholar] [CrossRef] [PubMed]
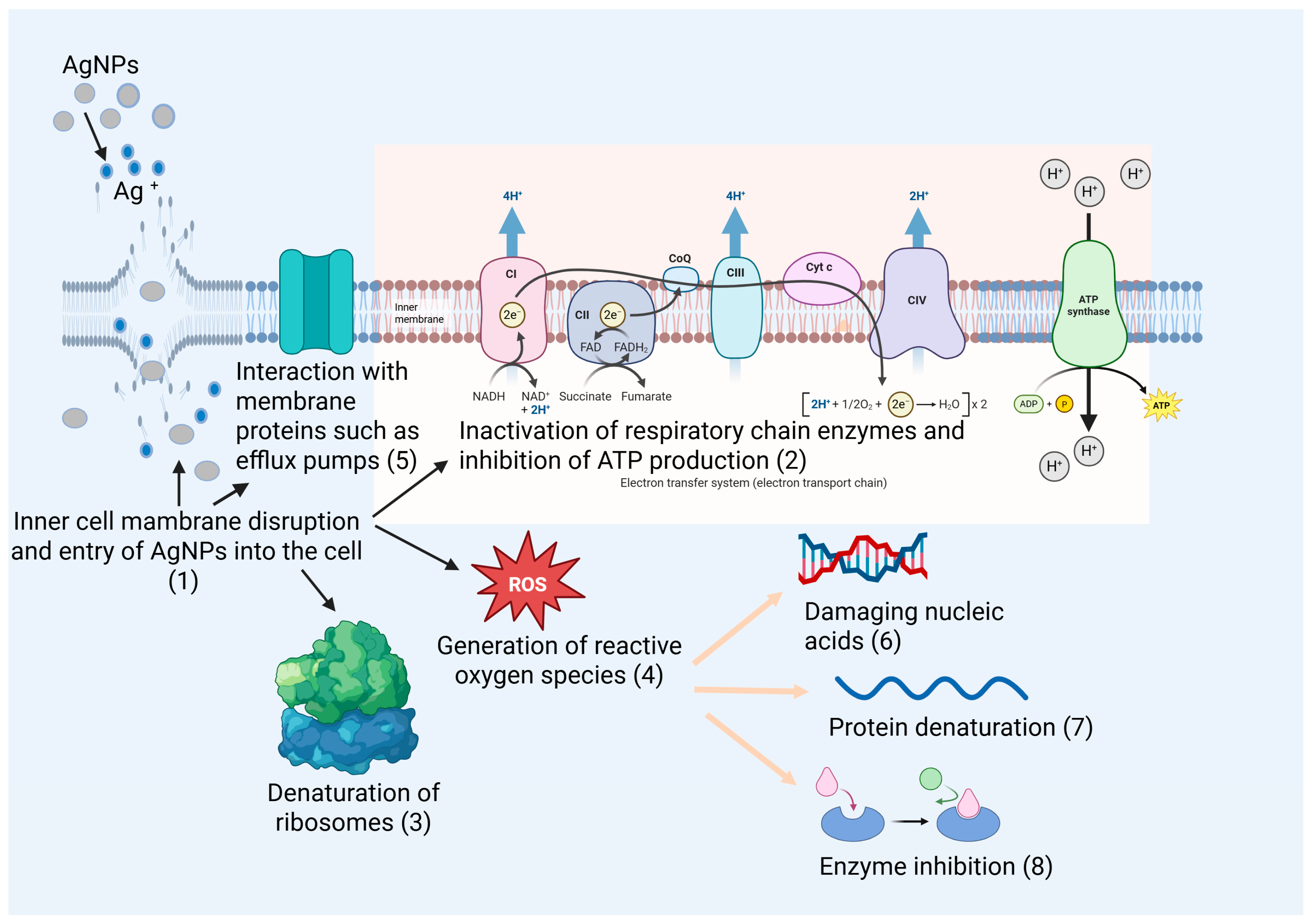
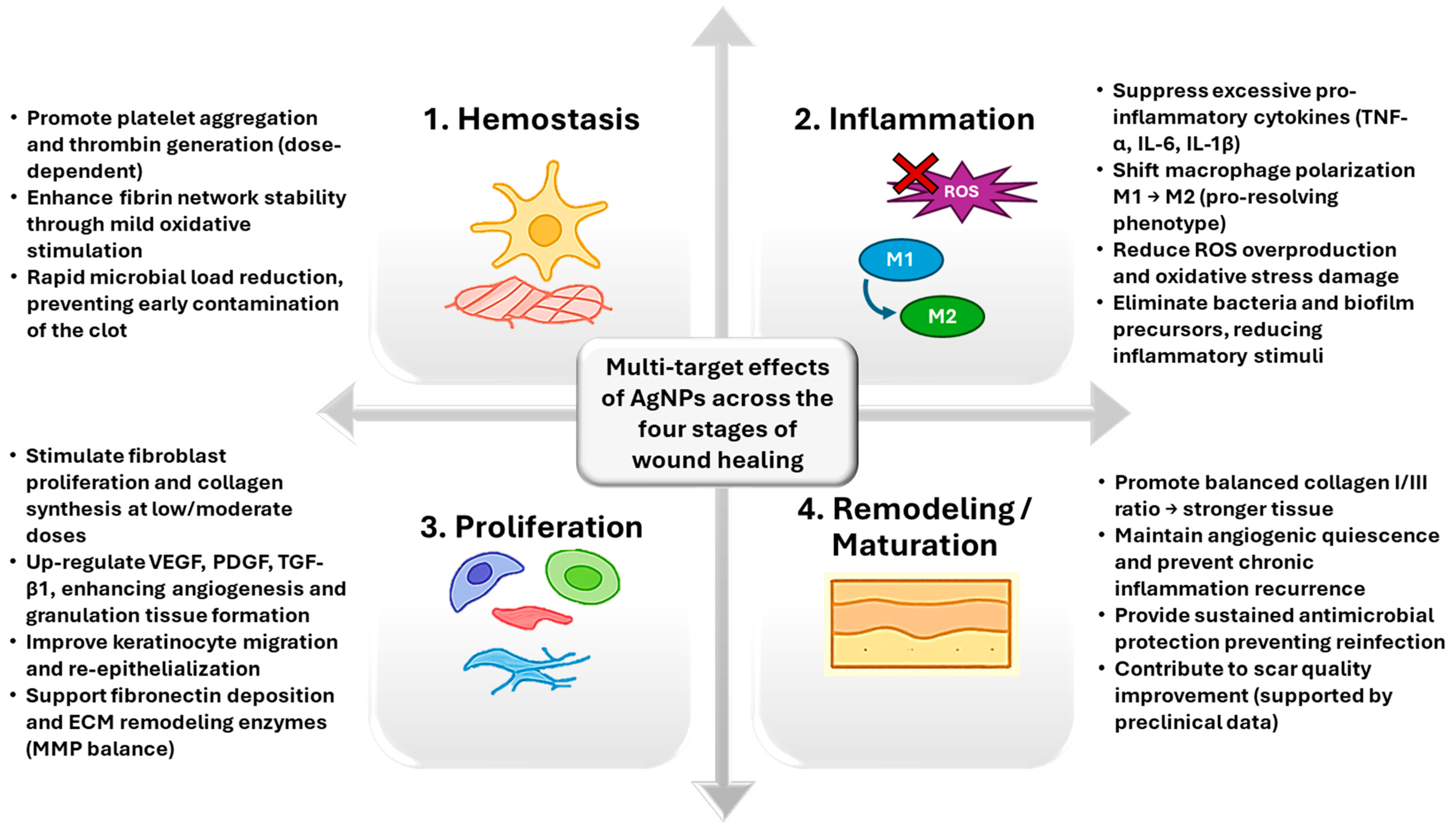

| Type of AgNPs | Method of Preparation | Size | Targeted Microorganisms | Magnitude of the Antibacterial Activity | Ref. |
|---|---|---|---|---|---|
| Citrate-capped AgNPs of different sizes depending on pH | Reduction of AgNO3 by NaBH4 in the presence of citrate as the capping agent | 2 nm 12 nm 32 nm | E. coli Staphylococcus aureus | The smaller the NPs, the stronger the activity | [40] |
| AgNPs of different sizes, depending on the amount of fresh leaf extract | Reduction of AgNO3 by an aqueous extract of Arbutus unedo | 40 nm 58 nm | E. coli Pseudomonas aeruginosa Bacillus subtilis Staphylococcus epidermidis | [41] | |
| AgNPs of different sizes, depending on the nature of the extraction solvent | Reduction of AgNO3 by the petals extract of Rosa Indica | 12 nm 18 nm 700 nm | E. coli S. aureus Observation: good biocompatibility assessed by haemolysis assay | [42] | |
| AgNPs of different sizes, depending on the degree of pectin esterification and specific functional groups | Reduction of AgNO3 by pectin in alkaline medium | 8–13 nm 28 ± 7 nm | Bacillus subtilis Bacillus pumilus E. coli | [43] | |
| Thermo-sensitive co-polymer-capped AgNPs | Reduction of AgNO3 by NaBH4 in the presence of a thermos-responsive copolymer as the capping agent | 1.59 nm 2.29 nm 3.91 nm | E. coli S. aureus Kanamycin-resistant E. coli Observation: No toxicity against mammalian cells | [44] |
| Type of AgNPs | Method of Preparation | Shape | Targeted Microorganisms | Magnitude of the Antibacterial Activity | Ref. |
|---|---|---|---|---|---|
| AgNPs of different shapes | Microwave-assisted reduction of AgNO3 by ethylene glycol in the presence of PVP and varying amounts of NaCl | Nanocubes Nanospheres Nanowires | E. coli | The antimicrobial activity decreased in the order Nanocubes > Nanospheres > Nanowires. | [49] |
| AgNPs with different edge morphologies | Solvothermal method involving the reduction of AgNO3 in dimethylformamide in the presence of PVP | Anisotropic plate-like NP with: - sharp edges - round edges | E. coli S. aureus Bacillus (MTCC 1789) Streptococcus pyogenes Vibrio cholerae | The AgNPs with sharp edges and corners showed greater antibacterial activity than those with rounded edges and corners. | [50] |
| Nanocrystals of supramolecular coordination polymers of different shapes, depending on the type of imidazole ligands | Mixing at room temperature an aqueous solution of AgNO3 with previously sonicated ethanolic solutions of three different imidazole ligands | Octahedral Hexagonal Nanosheet Nanoribbon | E. coli B. subtilis | The antibacterial activity decreased in the order: Ag-2Imid (octahedral nanocrystals) > Ag-Imid (hexagonal nanostructures > Ag-Benz (nanoribbons). | [51] |
| Type of AgNPs | Method of Preparation | Targeted Microorganisms | Magnitude of the Antibacterial Activity | Ref. |
|---|---|---|---|---|
| Polycationic silver nanoclusters | Reduction of AgNO3 in the presence of chitosan as both a reducing and capping agent | Fusobacterium Nucleatum Streptococcussanguinis | Positively charged AgNPs enhance penetration to the bacterial cell membrane compared to the corresponding negatively charged counterparts. | [45] |
| Negatively charged MSA-coated AgNPs | Reduction of AgNO3 by NaBH4 in the presence of mercaptosuccinic acid (MSA or thiomalic acid) | |||
| Neutrally PVP-capped AgNPs | Reduction of AgNO3 by glucose in the presence of PVP | - | The neutral PVP-capped AgNPs have a higher dissolution rate than citrate-coated AgNP. | [35,46] |
| Negatively charged citrate-capped AgNPs | Reduction of AgNO3 by sodium citrate |
| Antibiotic Classes | Antibiotics | Primary Targets | Genetic and Biochemical Processes That Are Disrupted |
|---|---|---|---|
| Fluoroquinolones (DNA synthesis inhibitors | Nalidixic acid, ciprofloxacin, levofloxacin, gemifloxacin | Topoisomerase II (DNA gyrase), topoisomerase IV | DNA replication, SOS response, cell division, ATP generation, TCA cycle, Fe–S cluster synthesis, ROS formation, redox-responsive two-component systems, and envelope |
| Trimetropim-sulfamethoxazole (DNA synthesis inhibitor) | Co-trimoxazole | Tetrahydrofolic acid synthesis inhibitors | Nucleotide biosynthesis and DNA replication |
| Rifamycins (RNA synthesis inhibitors) | Rifamycins, rifampin, rifapentine | DNA-dependent RNA polymerase | RNA transcription, DNA replication, SOS response |
| β-Lactams | Penicillins (penicillin, ampicillin, oxacillin), cephalosporins (cefazolin, cefoxitin, ceftriaxone, cefepime), carbapenems (imipenem) | Penicillin-binding proteins | Cell wall synthesis, cell division, autolysin activity (regulated by LytSR–VncRS two-component system), SOS response, TCA cycle, Fe–S cluster synthesis, ROS formation, envelope, redox-responsive two-component systems |
| Glycopeptides and glycolipopeptides (cell wall inhibitors) | Vancomycin, teicoplanin | Peptidoglycan units (terminal D-Ala-D-Ala dipeptide) | Cell wall synthesis, transglycosylation, transpeptidation, and autolysin activation (VncRS two-component system) |
| Lipopeptides (cell wall synthesis inhibitors) | Daptomycin, polymixin B | Cell membrane | Cell wall synthesis, envelope two-component systems |
| Aminoglycosides (protein synthesis inhibitors) | Gentamicin, tobramycin, streptomycin, kanamycin | 30S ribosome | Protein translation (mistranslation by tRNA mismatching), ETC, SOS response, TCA cycle, Fe–S cluster synthesis, ROS formation, envelope, redox-responsive two-component systems |
| Tetracyclines (protein synthesis inhibitors) | Tetracycline, doxycycline | 30S ribosome | Protein translation (through inhibition of aminoacyl tRNA binding to ribosome) |
| Macrolides (protein synthesis inhibitors) | Erythromycin, azythromycin | 50S ribosome | Protein translation (through inhibition of elongation and translocation steps) and free tRNA depletion |
| Streptogramins (protein synthesis inhibitors) | Pristinamycin, dalfopristin, quinupristin | 50S ribosome | Protein translation (through inhibition of initiation, elongation, and translocation steps) and free tRNA depletion |
| Phenicols (protein synthesis inhibitor) | Chloramphenicol | 50S ribosome | Protein translation (through inhibition of the elongation step) |
| AgNP System/Formulation | AgNP Size (nm) | Ag Concentration/Dose | Model (In Vitro/In Vivo) | Main Observed Biological Outcomes | Ref. |
|---|---|---|---|---|---|
| Dispersed AgNPs | 9 nm | 16, 32, or 100 µM AgNPs | in vitro: mouse embryonic fibroblasts | 16 µM AgNPs stimulated fibroblast proliferation; Concentration-dependent switch from pro-regenerative to toxic effects | [113] |
| CMC/OS/MFC sponge decorated with rhCol III and in situ TA-reduced AgNPs | n.r. | n.r. (AgNO3 reduced in situ) | in vitro scratch assay; in vivo diabetic rat full-thickness infected wounds | rhCol III/AgNP sponge: 77.3% scratch closure at 48 h vs. 43.4% for AgNP sponge without rhCol III; Complete wound closure by day 14 in infected diabetic rats vs. ~17% residual area in controls. | [115] |
| Carbopol nanogels with mycosynthesized AgNPs | n.r. | 0.1, 0.5, 1 mg g−1 AgNPs in nanogels | in vivo: Wistar rat incision, excision, and burn wound models | All three doses improved healing; 0.1 mg g−1 most effective in excision wounds, 1 mg g−1 best in burn wounds; histology showed good regeneration and no toxicity, highlighting dose-dependent optimization. | [116] |
| Dispersed AgNPs | 5–15 nm (mean 10 nm) | 1 mM AgNPs dispersion ≤100 µM non-toxic to fibroblasts (MTT); in vivo dose not numerically specified | in vivo: mouse surgical wounds; ex vivo keratinocytes and fibroblasts | AgNP treatment accelerated wound closure vs. 1% SSD; increased keratinocyte proliferation up to day 7; induced fibroblast differentiation to α-SMA+ myofibroblasts, enhancing wound contraction without cytotoxicity below 100 µM | [117] |
| Eggshell membrane (ESM) biomaterial loaded with commercial/green AgNPs | ~10 nm | 5 µg/mL AgNPs in ESM samples | in vitro: human dermal fibroblasts and BJ fibroblasts; ex ovo CAM assay | Strong pro-angiogenic response in CAM with increased vascular branching, supporting ECM remodeling and later repair | [119] |
| RSF/CMC-Na composite hydrogel (CoHy) with UV-photoreduced AgNPs | 50–200 nm (mean ~92 nm) | 1 or 2 mg AgNO3 per mL RSF solution (in situ AgNP formation) | in vitro: rat BMSCs; antimicrobial tests | Broad-spectrum antimicrobial activity; 1 mg/mL AgNO3 (mean 92 nm) gave best balance of cytocompatibility and antibacterial performance | [124] |
| CMC hydrogel with in situ green-synthesized AgNPs | 6.87–8.51 nm | MICs: 5.15 µg/mL (E. coli); 30 µg/mL (S. aureus); 27 µg/mL (P. aeruginosa); 100 µg/mL hydrogel in scratch assay | in vitro: 3T3 fibroblasts, scratch assay; ex vivo DFU-relevant assays | 88% fibroblast viability; Strong antibacterial activity at low µg/mL range; 100 µg/mL hydrogel gave ~75% scratch closure at 24 h; reduced MPO and collagenase activity, supporting ECM preservation and regeneration. | [127] |
| Injectable hydrogel from decellularized swine dermis loaded with GA-capped AgNPs (Ag@ADMH) | 8.763 ± 1.221 nm | 0.43% silver within Ag@ADMH | in vivo: MRSA-infected wound model | Sustained AgNP release from dermal matrix; Downregulated iNOS (M1 marker) and upregulated CD206 (M2 marker), indicating M1→M2 macrophage polarization; Enhanced fibroblast migration/proliferation and transition from inflammatory to proliferative phase. | [128] |
| Bilayer electrospun PCL/PVA patch co-loaded with vitamins B12/C and AgNPs | n.r. | n.r. | in vivo: rat full-thickness dorsal skin wounds | Showed stronger inhibition of S. aureus and E. coli than fibers without AgNPs Maintained > 93% cell viability, and led to faster wound closure Improved epithelialization and collagen deposition vs. dressings without AgNPs/vitamins | [132] |
| PHB/CS nanofibrous scaffold containing curcumin-derived biogenic AgNPs | Average 19.83 nm | 0.25–1%wt AgNPs in the matrix | in vitro: fibroblast compatibility and antibacterial assays | Enhanced antibacterial activity (notably vs. S. aureus), indicating suitability as an antibacterial, pro-healing scaffold | [133] |
| Composition | Stimuli | Properties and Effects | References |
|---|---|---|---|
| Dual-crosslinked, mussel-inspired hydrogels based on oxidized dextran (OD), dopamine (DA), and water-soluble quaternary ammonium chitosan (HTCC) co-loaded with deferoxamine (DFO) | pH | Antibacterial activity against S. aureus and E. coli; controls inflammation, promotes neovascularization | [159] |
| Methacrylic acid (MAA)—acrylamide (AAm), crosslinked with N,N′-methylenebisacrylamide (MBAm) copolymer hydrogel | pH | Antibacterial against Staphylococcus epidermidis and P. aeruginosa; no significant cytotoxicity to human skin fibroblasts | [163] |
| Polydopamine (PDA)@Ag plasmonic nanoparticles embedded in a cationic guar gum (CG) hydrogel matrix (CG/PDA@Ag) | NIR | Antibacterial activity against both Gram-negative E. coli (99.9%) and Gram-positive S. aureus (99.8%); excellent bactericidal efficacy in a rat wound infection model | [164] |
| Semi-interpenetrating polymeric membrane composed of crosslinked PNIPAM and linear thermoplastic polyurethane (TPU) elastomer | Temperature | Excellent tensile properties, high wound exudate absorption capacity, and easy re-movability from the wound surface; increased water vapor transmission rate (WVTR); good cytocompatibility with human dermal fibroblasts; in vitro antimicrobial activity against S. aureus, P. aeruginosa, and C. albicans | [165] |
| Polydopamine (PDA) nanoparticles surface-modified with AgNPs and curcumin (Cur) and then overcoated with the phase-change material (PCM), 1-tetradecanol (PDA@Ag@Cur@PCM) | NIR/temperature | Stable photothermal cycling over five heating–cooling rounds; QS inhibition; suppression of colony growth; broad-spectrum bactericidal activity; local hyperthermia | [166] |
| Core/shell (D@Ag/LR) silicasomes comprising a dendritic mesoporous silica nanoparticle (DMSN) core (D@Ag) surrounded by a cationic liposomal shell functionalized with the EGFR-targeting peptide GE11 and co-loaded with resveratrol (Res); silicasomes were dispersed in an enzyme-responsive gelatin-methacryloyl (GelMA) covalently crosslinked hydrogel | Enzymes (MMP-9) | Antibacterial, anti-inflammatory, haemostatic, and pro-regenerative; clearance rate of intracellular MRSA reached 83.75% in BJ fibroblasts and 88.14% in HaCaT keratinocytes | [167] |
| 3-aminophenylboronic-acid–modified hyaluronic acid (HB) crosslinked with black chokeberry (BCE) extract (HB—BCE/Ag) | pH/hyaluronidases (HAas) | Bactericidal rates of 99.12 ± 0.08% against MRSA and 99.32 ± 0.13% against S. aureus; antioxidant, adhesive, self-healing, and biocompatible properties; up-regulation of CDH1 (E-cadherin) and down-regulation of CXCL1/CXCL2; inflammation control, collagen deposition, and promotion of angiogenesis in a rat full-thickness MRSA-infected wound model | [169] |
| Carboxymethylated starch (cassava-waste)/PVA/glycerol hydrogels embedding tannic-acid (TA)–capped AgNPs (H-AgNPs) | NIR/pH | H-AgNPs-200 formulation achieved ~100% kill of E. coli and 98.2% of S. under NIR, while maintaining high mammalian-cell viability | [170] |
| Platelet-inspired biodegradable hydrogel based on GelMA, TA, polyphosphate (PolyP), and gallic acid functionalized AgNPs (Gel/PP-TA-Ag) | NIR | Better haemostatic effect than commercial gauze in the mice-bleeding model; 97.57% of methicillin-resistant MRSA and 95.99% of E. coli are eliminated in vitro; 91.76% of MRSA in wounds is removed in vivo; reduced inflammation; improved collagen deposition and angiogenesis | [173] |
| Biocompatible polysaccharide carrageenan (Carr) embedded with gallic acid functionalized AgNPs (GA-Ag NP hydrogel) | NIR | Sterilization property against E. coli and S. aureus; biosafety; accelerated wound healing in S. aureus-infected mice | [174] |
| Mesoporous-silica–modified Ag2S quantum-dots (QDs) hydrogel covalently embedded in a poly(N-isopropylacrylamide-co-acrylamide) network | NIR (PTT + PDT) | Produced ~99.7% kill of E. coli and 99.8% of MRSA within 4 min; accelerated wound closure and increased collagen deposition and angiogenesis in vivo in a BALB/c mouse full-thickness MRSA-infected wound model; low cytotoxicity towards Vero cells and NIH 3T3 mouse embryonic fibroblasts | [176] |
| Injectable, self-healing hydrogel built from phenylboronic-acid–modified hyaluronic acid (HA-PBA) and AgNPs-capped plant-derived TA | pH/ROS | Faster drug release at pH 7.2 (chronic-wound-like) than at pH 5.0 (healthy skin); the fastest cargo release rate was reached at pH 7.2 in the presence of H2O2; antioxidant activity, low haemolytic toxicity, cytocompatibility towards L929 cells | [177] |
| PNIPAM-polyacrylic acid (PAA) copolymer hydrogel embedded with ultrasmall AgNPs | pH/temperature | Over 95% of S. epidermidis and P. aeruginosa eradicated in vitro at pH 7.4 and pH 10; accelerated wound closure, enhanced re-epithelialization, and increased collagen deposition in an in vivo S. aureus-infected mouse wound model; cytocompatibility with human fibroblasts (HFFs) and keratinocytes (HaCaTs) | [180] |
| Dual crosslinked nanocomposite hydrogel (CAO/ATR) built up from carboxyethyl chitosan (CEC), oxidized sodium alginate (OSA), and TA-capped AgNPs functionalized with red-cabbage anthocyanin aqueous–ethanolic extract (ATR) | colorimetric pH-responsive | Bioadhesive, haemostatic properties, inhibition of the growth of E. coli and S. aureus in vitro; simple visual monitoring of wound status | [181,182] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Balaure, P.C.; Niculescu, A.-G.; Anghel, D.; Grumezescu, A.M.; Alberts, A. Role of Silver Nanoparticles in Wound Healing: Mechanisms, Efficacy, and Clinical Applications. Inorganics 2025, 13, 401. https://doi.org/10.3390/inorganics13120401
Balaure PC, Niculescu A-G, Anghel D, Grumezescu AM, Alberts A. Role of Silver Nanoparticles in Wound Healing: Mechanisms, Efficacy, and Clinical Applications. Inorganics. 2025; 13(12):401. https://doi.org/10.3390/inorganics13120401
Chicago/Turabian StyleBalaure, Paul Cătălin, Adelina-Gabriela Niculescu, Daniela Anghel, Alexandru Mihai Grumezescu, and Adina Alberts. 2025. "Role of Silver Nanoparticles in Wound Healing: Mechanisms, Efficacy, and Clinical Applications" Inorganics 13, no. 12: 401. https://doi.org/10.3390/inorganics13120401
APA StyleBalaure, P. C., Niculescu, A.-G., Anghel, D., Grumezescu, A. M., & Alberts, A. (2025). Role of Silver Nanoparticles in Wound Healing: Mechanisms, Efficacy, and Clinical Applications. Inorganics, 13(12), 401. https://doi.org/10.3390/inorganics13120401









