Synthesis, Crystal Structure Analyses, and Antibacterial Evaluation of the Cobalt(II) Complex with Sulfadiazine-Pyrazole Prodrug
Abstract
:1. Introduction
2. Results and Discussion
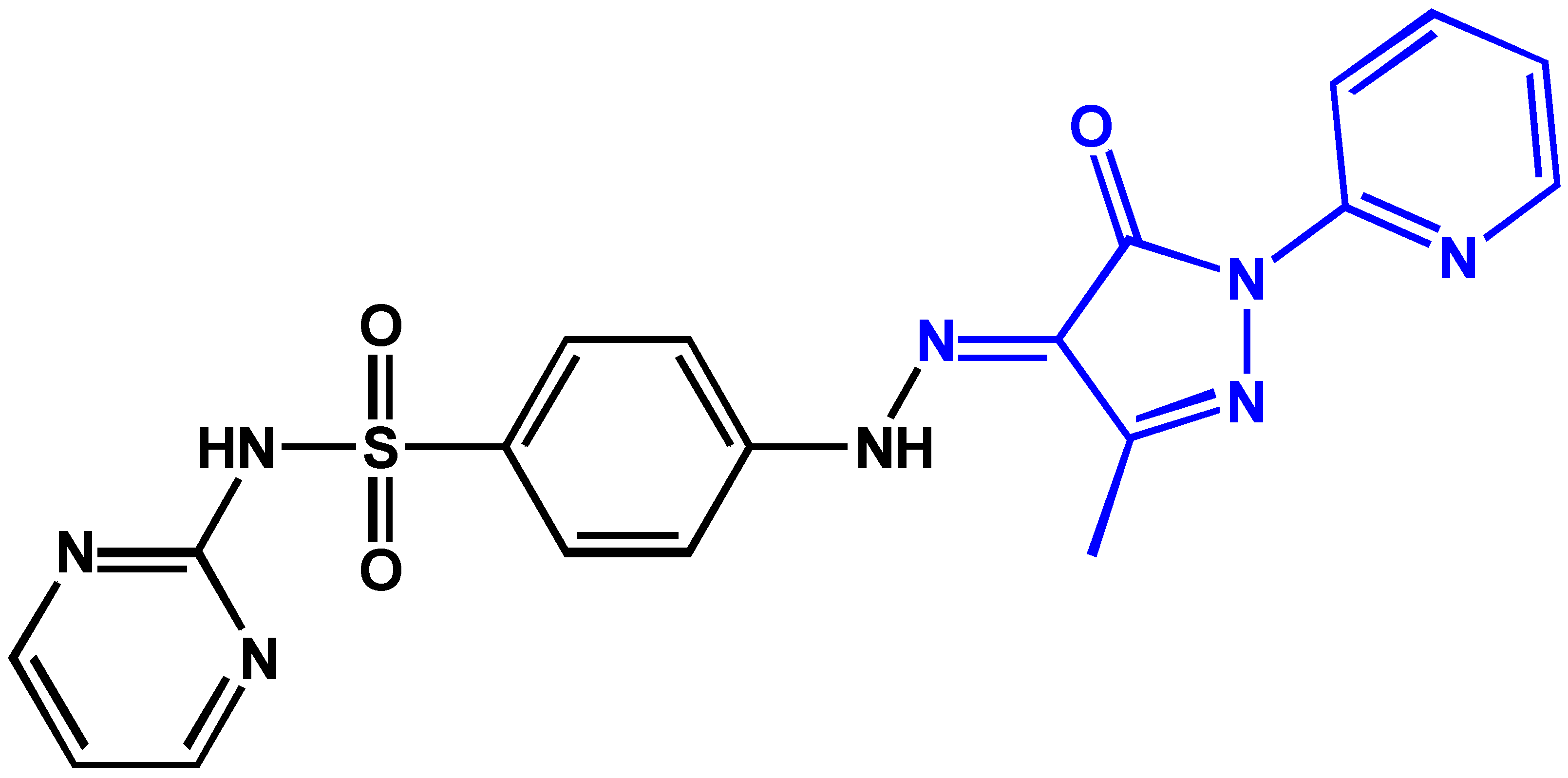
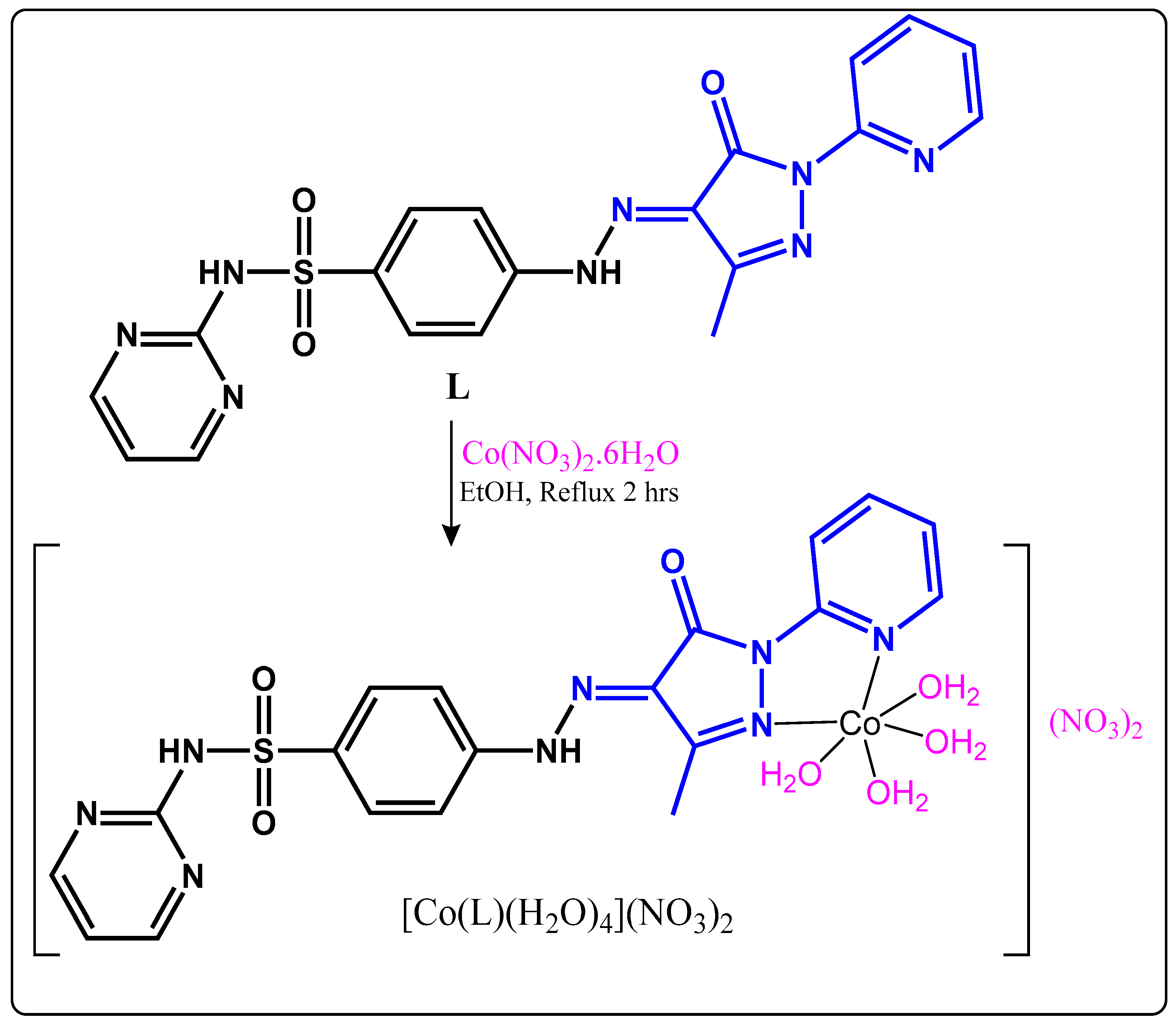
2.1. Synthesis and Characterization
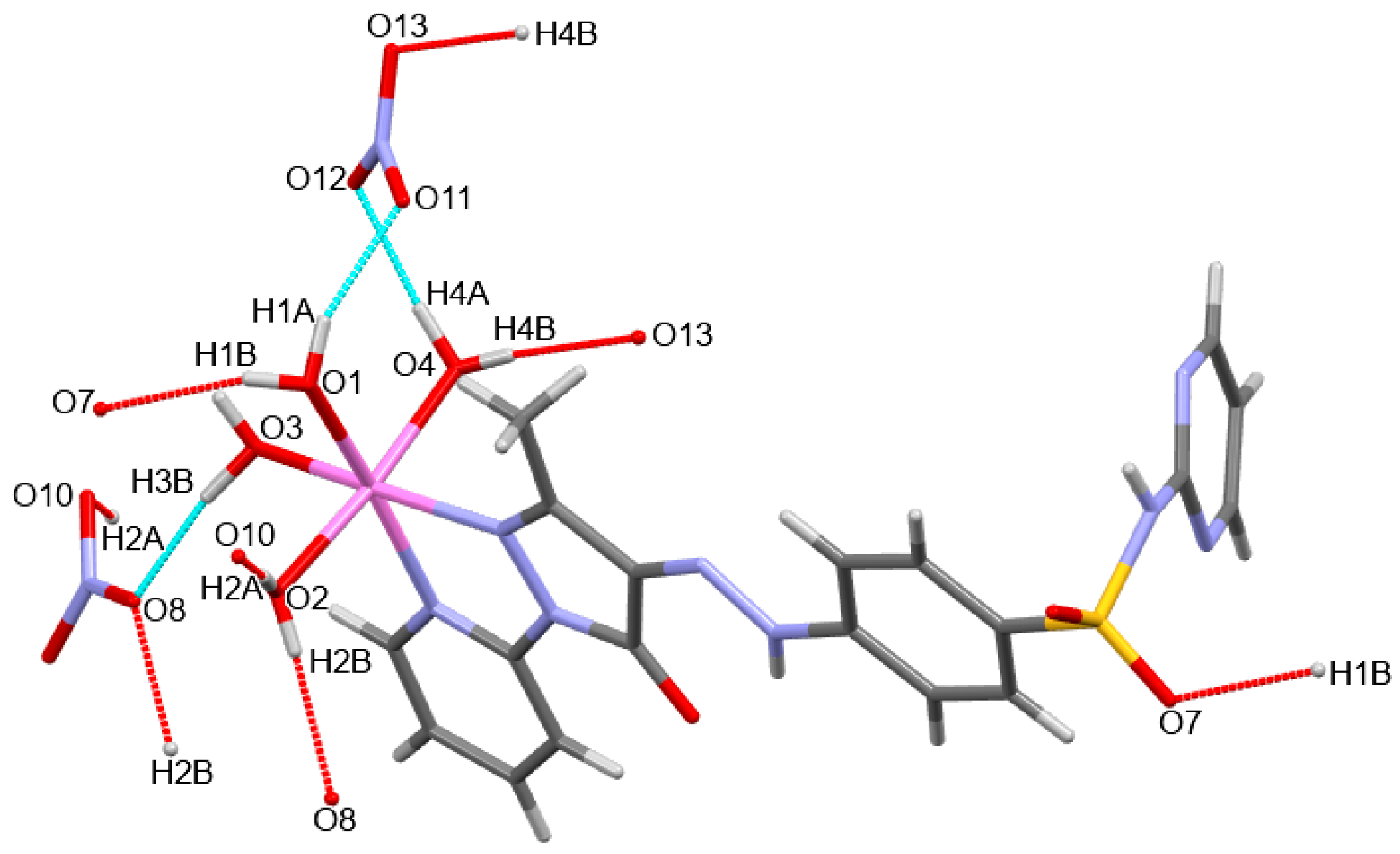
2.2. X-ray Structure Description
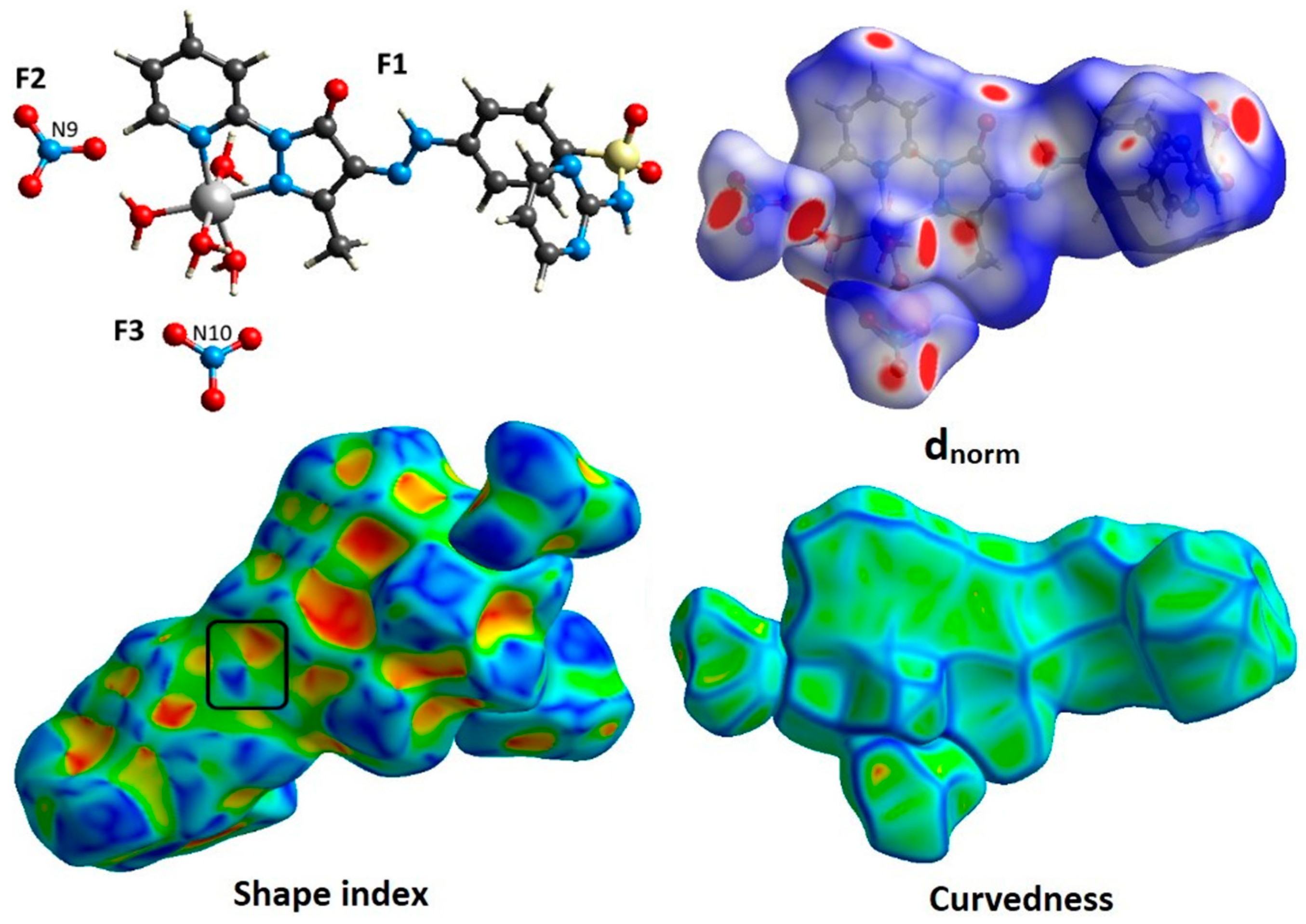
2.3. Molecular Packing Analysis
2.4. Antimicrobial Studies
3. Materials and Methods
3.1. Chemicals and Instrumentations
3.2. Syntheses
Synthesis of [Co(L)(H2O)4](NO3)2 Complex
3.3. Crystal Structure Determination
3.4. Hirshfeld Surface Analysis
3.5. Antimicrobial Studies
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Koshti, S.M.; Patil, P.A.; Patil, C.B.; Patil, A.S. Synthesis and characterization of prodrugs of sulfonamides as an azo derivatives of carvacrol. Pharma. Chem. 2018, 10, 1–15. [Google Scholar]
- Pradere, U.; Garnier-Amblard, E.C.; Coats, S.J.; Amblard, F.; Schinazi, R.F. Synthesis of nucleoside phosphate and phosphonate prodrugs. Chem. Rev. 2014, 114, 9154–9218. [Google Scholar] [CrossRef]
- Connor, E.E. Sulfonamide antibiotics. Prim. Care Update Ob/Gyns 1998, 5, 32–35. [Google Scholar] [CrossRef]
- Venkatesan, M.; Fruci, M.; Verellen, L.A.; Skarina, T.; Mesa, N.; Flick, R.; Pham, C.; Mahadevan, R.; Stogios, P.J.; Savchenko, A. Molecular mechanism of plasmid-borne resistance to sulfonamide antibiotics. Nat. Commun. 2023, 14, 4031. [Google Scholar] [CrossRef] [PubMed]
- Gil, D.M.; Pérez, H.; Echeverría, G.A.; Piro, O.E.; Frontera, A. Role of imidazole co–ligand in the supramolecular network of a Co(II) complex with sulfadiazine: Crystal structure, Hirshfeld surface analysis and energetic calculations. ChemistrySelect 2020, 5, 6331–6338. [Google Scholar] [CrossRef]
- Yang, X.-L.; Liu, J.; Yang, L.; Zhang, X.-Y. Synthesis, characterization, and susceptibility of bacteria of selenium dioxide complexes with sulfadrugs. Synth. React. Inorg. Met. 2005, 35, 761–766. [Google Scholar] [CrossRef]
- El-Baradie, K.; Gaber, M. Synthesis, spectral, thermal, and electrical conductivity studies of cobalt(II) and copper(II) sulfadiazine complexes. Chem. Pap. 2003, 57, 317–321. [Google Scholar]
- Shi, W.-B.; Cui, A.-L.; Kou, H.-Z. Sulfadiazine/dimethylsulfadiazine transition metal complexes: Synthesis, crystal structures and magnetic properties. Polyhedron 2015, 99, 252–259. [Google Scholar] [CrossRef]
- Schapira, A.; Beales, P.; Halloran, M. Malaria: Living with drug resistance. Parasitol. Today 1993, 9, 168–174. [Google Scholar] [CrossRef]
- Fernández-Villa, D.; Aguilar, M.R.; Rojo, L. Folic acid antagonists: Antimicrobial and immunomodulating mechanisms and applications. Int. J. Mol. Sci. 2019, 20, 4996. [Google Scholar] [CrossRef]
- Mani, F. Model systems containing pyrazole chelates and related groups: Recent developments and perspectives. Coord. Chem. Rev. 1992, 120, 325–359. [Google Scholar] [CrossRef]
- Trofimenko, S. Coordination chemistry of pyrazole-derived ligands. Chem. Rev. 1972, 72, 497–509. [Google Scholar] [CrossRef]
- Seth, S.K.; Saha, N.C.; Ghosh, S.; Kar, T. Structural elucidation and electronic properties of two pyrazole derivatives: A combined X-ray, Hirshfeld surface analyses and quantum mechanical study. Chem. Phys. Lett. 2011, 506, 309–314. [Google Scholar] [CrossRef]
- Shafiq, N.; Shahzad, N.; Rida, F.; Ahmad, Z.; Nazir, H.A.; Arshad, U.; Zareen, G.; Attiq, N.; Parveen, S.; Rashid, M. One-pot multicomponent synthesis of novel pyridine derivatives for antidiabetic and antiproliferative activities. Future Med. Chem. 2023, 15. [Google Scholar] [CrossRef]
- Altowyan, M.S.; Soliman, S.M.; Ismail, M.M.; Haukka, M.; Barakat, A.; Ayoup, M.S. New bioprecursor prodrugs of sulfadiazine: Synthesis, X-ray structure and Hirshfeld analysis. Crystals 2022, 12, 1016. [Google Scholar] [CrossRef]
- Wong, E.; Giandomenico, C.M. Current status of platinum-based antitumor drugs. Chem. Rev. 1999, 99, 2451–2466. [Google Scholar] [CrossRef]
- Ott, I.; Gust, R. Non platinum metal complexes as anti-cancer drugs. Arch. Pharm. Int. J. Pharm. Med. Chem. 2007, 340, 117–126. [Google Scholar] [CrossRef]
- Northcote-Smith, J.; Suntharalingam, K. Targeting chemotherapy-resistant tumour sub-populations using inorganic chemistry: Anti-cancer stem cell metal complexes. Curr. Opin. Chem. Biol. 2023, 72, 102237. [Google Scholar] [CrossRef]
- Nasiri Sovari, S.; Zobi, F. Recent studies on the antimicrobial activity of transition metal complexes of groups 6–12. Chemistry 2020, 2, 418–452. [Google Scholar] [CrossRef]
- Fahim, A.M.; Hasanin, M.; Habib, I.; El-Attar, R.O.; Dacrory, S. Synthesis, antimicrobial activity, theoretical investigation, and electrochemical studies of cellulosic metal complexes. J. Iran. Chem. Soc. 2023, 20, 1699–1718. [Google Scholar] [CrossRef]
- Yousri, A.; El-Faham, A.; Haukka, M.; Ayoup, M.S.; Ismail, M.M.; Menofy, N.G.E.; Soliman, S.M.; Öhrström, L.; Barakat, A.; Abu-Youssef, M.A. A novel Na(I) coordination complex with s-triazine pincer ligand: Synthesis, X-ray structure, Hirshfeld analysis, and antimicrobial activity. Crystals 2023, 13, 890. [Google Scholar] [CrossRef]
- Yousry, A.; Haukka, M.; Abu-Youssef, M.A.; Ayoup, M.S.; Ismail, M.M.; El Menofy, N.; Soliman, S.M.; Barakat, A.; Noa, F.M.A.; Öhrström, L.R. Synthesis, structure diversity, and antimicrobial studies of Ag(I) complexes with quinoline-type ligands. CrystEngComm 2023, 25, 3922–3930. [Google Scholar] [CrossRef]
- García-Raso, Á.; Fiol, J.J.; Rigo, S.; López-López, A.; Molins, E.; Espinosa, E.; Borrás, E.; Alzuet, G.; Borrás, J.N.; Castiñeiras, A. Coordination behaviour of sulfanilamide derivatives.: Crystal structures of [Hg(sulfamethoxypyridazinato)2], [Cd(sulfadimidinato)2(H2O)].2H2O and [Zn (sulfamethoxazolato)2-(pyridine)2(H2O)2]. Polyhedron 2000, 19, 991–1004. [Google Scholar] [CrossRef]
- Hossain, G.G.; Amoroso, A.; Banu, A.; Malik, K. Syntheses and characterisation of mercury complexes of sulfadiazine, sulfamerazine and sulfamethazine. Polyhedron 2007, 26, 967–974. [Google Scholar] [CrossRef]
- Villa-Pérez, C.; Oyarzabal, I.; Echeverría, G.A.; Valencia-Uribe, G.C.; Seco, J.M.; Soria, D.B. Single-ion magnets based on mononuclear cobalt(II) complexes with sulfadiazine. Eur. J. Inorg. Chem. 2016, 2016, 4835–4841. [Google Scholar] [CrossRef]
- Brown, C.; Cook, D.; Sengier, L. Bis[N1-(2-pyrimidinyl)sulphanilamido]zinc–ammonia (1/2), [Zn(C10H9N4O2S)2].2NH3. Acta Crystallogr. C Cryst. Struct. Commun. 1985, 41, 718–720. [Google Scholar] [CrossRef]
- Greenhalgh, D.G. Topical antimicrobial agents for burn wounds. Clin. Plast. Surg. 2009, 36, 597–606. [Google Scholar] [CrossRef]
- Fox, C.L. Silver sulfadiazine-a new topical therapy for pseudomonas in burns: Therapy of pseudomonas infection in burns. Arch. Surg. 1968, 96, 184–188. [Google Scholar] [CrossRef]
- Fox, C.L.; Modak, S.; Stanford, J.; Fox, P.L. Metal sulfonamides as antibacterial agents in topical therapy. Scand. J. Plast. Reconstr. Surg. 1979, 13, 89–94. [Google Scholar] [CrossRef]
- Ajibade, P.A.; Kolawole, G.A.; O’Brien, P.; Helliwell, M.; Raftery, J. Cobalt(II) complexes of the antibiotic sulfadiazine, the X-ray single crystal structure of [Co(C10H9N4O2S)2(CH3OH)2]. Inorg. Chim. Acta 2006, 359, 3111–3116. [Google Scholar] [CrossRef]
- Rocha, M.; Piro, O.E.; Echeverría, G.A.; Pastoriza, A.C.; Sgariglia, M.A.; Soberón, J.R.; Gil, D.M. Co(II), Ni(II) and Cu(II) ternary complexes with sulfadiazine and dimethylformamide: Synthesis, spectroscopic characterization, crystallographic study and antibacterial activity. J. Mol. Struct. 2019, 1176, 605–613. [Google Scholar] [CrossRef]
- Anacona, J.; Noriega, N.; Camus, J. Synthesis, characterization and antibacterial activity of a tridentate schiff base derived from cephalothin and sulfadiazine, and its transition metal complexes. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2015, 137, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Lutsenko, I.A.; Yambulatov, D.S.; Kiskin, M.A.; Nelyubina, Y.V.; Primakov, P.V.; Bekker, O.B.; Levitskiy, O.A.; Magdesieva, T.V.; Imshennik, V.K.; Maksimov, Y.V. Improved in vitro antimycobacterial activity of trinuclear complexes cobalt(II, III) and iron(III) with 2-furoic acid against mycolicibacterium smegmatis. ChemistrySelect 2020, 5, 11837–11842. [Google Scholar] [CrossRef]
- Chadghan, A.; Pons, J.; Caubet, A.; Casabó, J.; Ros, J.; Alvarez-Larena, A.; Piniella, J.F. Cobalt(II) complexes with pyrazole-derived ligands: Crystal structure of {bis [3-phenyl-5-(2-pyridyl) pyrazole] aquachlorocobalt(II)} chloride monohydrate. Polyhedron 2000, 19, 855–862. [Google Scholar] [CrossRef]
- Liu, C.-S.; Zhang, H.; Chen, R.; Shi, X.-S.; Bu, X.-H.; Yang, M. Two new Co(II) and Ni(II) complexes with 3-(2-pyridyl) pyrazole-based ligand: Synthesis, crystal structures, and bioactivities. Chem. Pharm. Bull. 2007, 55, 996–1001. [Google Scholar] [CrossRef]
- Jelsch, C.; Ejsmont, K.; Hudera, L. The enrichment ratio of atomic contacts in crystals, an indicator derived from the Hirshfeld surface analysis. IUCrJ 2014, 1, 119–128. [Google Scholar] [CrossRef]
- Rikagu Oxford Diffraction. CrysAlisPro; Rikagu Oxford Diffraction Inc.: Yarnton, UK, 2020. [Google Scholar]
- Sheldrick, G. Sadabs-Bruker Nonius Scaling and Absorption Correction; Bruker AXS Inc.: Madison, WI, USA, 2012. [Google Scholar]
- Sheldrick, G.M. Shelxt–integrated space-group and crystal-structure determination. Acta Cryst. 2015, A71, 3–8. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Cryst. 2015, C71, 3–8. [Google Scholar]
- Hübschle, C.B.; Sheldrick, G.M.; Dittrich, B. A Qt Graphical User Interface for SHELXL. J. Appl. Cryst. 2011, 44, 1281–1284. [Google Scholar] [CrossRef]
- Hirshfeld, F.L. Bonded-atom fragments for describing molecular charge densities. Theor. Chim. Acta 1977, 44, 129–138. [Google Scholar] [CrossRef]
- Mackenzie, C.F.; Spackman, P.R.; Jayatilaka, D.; Spackman, M.A. Crystalexplorer model energies and energy frameworks: Extension to metal coordination compounds, organic salts, solvates and open-shell systems. IUCrJ 2017, 4, 575–587. [Google Scholar] [CrossRef] [PubMed]
- Wayne, P. Clinical and Laboratory Standards Institute: Performance Standards for Antimicrobial Susceptibility Testing: 20th Informational Supplement. CLSI Document M100-S20. Kansenshogakuzasshi 2010, 85, 355–359. [Google Scholar] [CrossRef]




| Bond | Distance | Bond | Distance |
| Co(1)-O(2) | 2.0635(11) | Co(1)-O(1) | 2.1036(10) |
| Co(1)-O(4) | 2.0909(11) | Co(1)-N(1) | 2.1240(10) |
| Co(1)-O(3) | 2.0986(10) | Co(1)-N(3) | 2.1406(10) |
| Bonds | Angle | Bonds | Angle |
|---|---|---|---|
| O(2)-Co(1)-O(4) | 174.25(4) | O(3)-Co(1)-N(1) | 96.86(4) |
| O(2)-Co(1)-O(3) | 90.11(4) | O(1)-Co(1)-N(1) | 170.36(4) |
| O(4)-Co(1)-O(3) | 86.97(4) | O(2)-Co(1)-N(3) | 94.99(4) |
| O(2)-Co(1)-O(1) | 85.00(5) | O(4)-Co(1)-N(3) | 88.51(4) |
| O(4)-Co(1)-O(1) | 90.00(4) | O(3)-Co(1)-N(3) | 171.61(4) |
| O(3)-Co(1)-O(1) | 89.31(4) | O(1)-Co(1)-N(3) | 97.76(4) |
| O(2)-Co(1)-N(1) | 87.57(4) | N(1)-Co(1)-N(3) | 76.74(4) |
| O(4)-Co(1)-N(1) | 97.69(4) |
| D-H···A | d(D-H)/Å | d(H···A)/Å | d(D···A)/Å | <(DHA)/° | Symm. Code |
|---|---|---|---|---|---|
| O(1)-H(1B)···O(7) | 0.80(3) | 2.06(3) | 2.8154(14) | 159(2) | x − 1,y − 1, z |
| O(1)-H(1A)···O(11) | 0.84(2) | 1.92(3) | 2.7190(18) | 159(2) | |
| O(2)-H(2B)···O(8) | 0.81(3) | 2.03(3) | 2.8323(17) | 171(2) | −x, −y, −z |
| O(2)-H(2A)···O(10) | 0.84(2) | 2.02(2) | 2.8583(19) | 174(2) | x + 1, y, z |
| O(3)-H(3B)···O(8) | 0.96(3) | 1.78(3) | 2.7352(16) | 170(2) | |
| O(4)-H(4A)···O(12) | 0.92(3) | 1.90(3) | 2.8135(17) | 172(3) | |
| O(4)-H(4B)···O(13) | 0.75(3) | 2.05(3) | 2.7922(18) | 168(3) | −x, −y, −z + 1 |
| N(5)-H(5)···O(5) | 0.77(2) | 2.13(2) | 2.7657(16) | 139(2) |
| Contact | %Contact | ||
|---|---|---|---|
| [Co(L)(H2O)4]2+ (F1) | NO3− (F2) | NO3− (F3) | |
| O···O | 0.9 | 2.9 | 1.8 |
| O···N | 2.1 | 8.0 | 7.2 |
| O···C | 5.2 | 5.2 | 9.1 |
| O···H | 38.8 | 79.6 | 77.8 |
| N···N | 0.6 | 0 | 1.3 |
| N···C | 2.3 | 0.5 | 0.6 |
| N···H | 9.9 | 3.8 | 2.2 |
| C···C | 2.4 | 0 | 0 |
| C···H | 10 | 0 | 0 |
| H···H | 27.8 | 0 | 0 |
| X···X/X···Y | C | R | E | Atom | S |
|---|---|---|---|---|---|
| O···O | 0.9 | 5.74 | 0.16 | O | 23.95 |
| O···N | 2.1 | 3.71 | 0.57 | H | 57.15 |
| O···C | 5.2 | 5.34 | 0.97 | N | 7.75 |
| O···H | 38.8 | 27.37 | 1.42 | C | 11.15 |
| N···N | 0.6 | - | - | ||
| N···C | 2.3 | 1.73 | 1.33 | ||
| N···H | 9.9 | 8.86 | 1.12 | ||
| C···C | 2.4 | 1.24 | 1.93 | ||
| C···H | 10 | 12.74 | 0.78 | ||
| H···H | 27.8 | 32.66 | 0.85 |
| Tested Compound | L | [Co(L)(H2O)4](NO3)2 | Amoxicillin |
|---|---|---|---|
| Gram-positive bacteria | |||
| S. aureus (ATCC 25923) | 125 | >500 | ≤7.8 |
| MRSA (1) | 125 | 500 | >500 |
| Gram-negative bacteria | |||
| E. coli (ATCC 25922) | 125 | 250 | >500 |
| K. pneumonia (ATCC 700603) | 125 | >500 | >500 |
| P. mirabilis | 125 | >500 | 125 |
| A. baumannii (8) | 62.5 | 500 | >500 |
| CCDC | 2282609 |
|---|---|
| empirical formula | C19H24CoN10O13S |
| fw | 691.47 |
| temp (K) | 170(2) |
| λ (Å) | 0.71073 |
| cryst syst | Triclinic |
| space group | |
| a (Å) | 7.73330(10) |
| b (Å) | 12.5152(2) |
| c (Å) | 15.5216(2) |
| α (deg) | 76.2120(10) |
| β (deg) | 77.9620(10) |
| γ (deg) | 78.9690(10) |
| V (Å3) | 1410.92(4) |
| Z | 2 |
| ρcalc (Mg/m3) | 1.628 |
| μ(Mo Kα) (mm−1) | 0.765 |
| No. reflns. | 50692 |
| Unique reflns. | 14152 |
| Completeness to θ = 25.242° | 99.8% |
| GOOF (F2) | 1.028 |
| Rint | 0.0239 |
| R1 a (I ≥ 2σ) | 0.0416 |
| wR2 b (I ≥ 2σ) | 0.1172 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Altowyan, M.S.; Haukka, M.; Ayoup, M.S.; Ismail, M.M.F.; El Menofy, N.G.; Soliman, S.M.; Barakat, A.; Sharaf, M.M.; Abu-Youssef, M.A.M.; Yousri, A. Synthesis, Crystal Structure Analyses, and Antibacterial Evaluation of the Cobalt(II) Complex with Sulfadiazine-Pyrazole Prodrug. Inorganics 2023, 11, 382. https://doi.org/10.3390/inorganics11100382
Altowyan MS, Haukka M, Ayoup MS, Ismail MMF, El Menofy NG, Soliman SM, Barakat A, Sharaf MM, Abu-Youssef MAM, Yousri A. Synthesis, Crystal Structure Analyses, and Antibacterial Evaluation of the Cobalt(II) Complex with Sulfadiazine-Pyrazole Prodrug. Inorganics. 2023; 11(10):382. https://doi.org/10.3390/inorganics11100382
Chicago/Turabian StyleAltowyan, Mezna Saleh, Matti Haukka, Mohammed Salah Ayoup, Magda M. F. Ismail, Nagwan G. El Menofy, Saied M. Soliman, Assem Barakat, Mona M. Sharaf, Morsy A. M. Abu-Youssef, and Amal Yousri. 2023. "Synthesis, Crystal Structure Analyses, and Antibacterial Evaluation of the Cobalt(II) Complex with Sulfadiazine-Pyrazole Prodrug" Inorganics 11, no. 10: 382. https://doi.org/10.3390/inorganics11100382
APA StyleAltowyan, M. S., Haukka, M., Ayoup, M. S., Ismail, M. M. F., El Menofy, N. G., Soliman, S. M., Barakat, A., Sharaf, M. M., Abu-Youssef, M. A. M., & Yousri, A. (2023). Synthesis, Crystal Structure Analyses, and Antibacterial Evaluation of the Cobalt(II) Complex with Sulfadiazine-Pyrazole Prodrug. Inorganics, 11(10), 382. https://doi.org/10.3390/inorganics11100382










