A Mini-Review on Functional Near-Infrared Spectroscopy (fNIRS): Where Do We Stand, and Where Should We Go?
Abstract
1. Introduction
2. Where Do We Stand
3. Where Should We Go?
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hutchinson, M.R.; Stoddart, P.R.; Mahadevan-Jansen, A. Challenges and opportunities in neurophotonics discussed at the International Conference on Biophotonics. Neurophotonics 2018, 5, 040402. [Google Scholar]
- Tanner, K.; D’Amico, E.; Kaczmarowski, A.; Kukreti, S.; Malpeli, J.; Mantulin, W.W.; Gratton, E. Spectrally resolved neurophotonics: A case report of hemodynamics and vascular components in the mammalian brain. J. Biomed. Opt. 2005, 10, 064009. [Google Scholar] [CrossRef][Green Version]
- Jöbsis, F.F. Noninvasive, infrared monitoring of cerebral and myocardial oxygen sufficiency and circulatory parameters. Science 1977, 198, 1264–1267. [Google Scholar] [CrossRef]
- Bigio, I.J.; Fantini, S. Quantitative Biomedical Optics Theory, Methods, and Applications, 1st ed.; Cambridge University Press: Cambridge, UK, 2016. [Google Scholar]
- Fantini, S.; Frederick, B.; Sassaroli, A. Perspective: Prospects of non-invasive sensing of the human brain with diffuse optical imaging. APL Photonics 2018, 3, 110901. [Google Scholar] [CrossRef]
- Wang, L.; Jacques, S.L.; Zheng, L. MCML—Monte Carlo modeling of light transport in multi-layered tissues. Comput. Methods Programs Biomed. 1995, 47, 131–146. [Google Scholar] [CrossRef]
- Sakakibara, Y.; Kurihara, K.; Okada, E. Evaluation of improvement of diffuse optical imaging of brain function by high-density probe arrangements and imaging algorithms. Opt. Rev. 2016, 23, 346–353. [Google Scholar] [CrossRef]
- Ferrari, M.; Norris, K.H.; Sowa, M.G. Medical near infrared spectroscopy 35 years after the discovery. J. Near Infrared Spectrosc. 2012, 20, vii–ix. [Google Scholar] [CrossRef]
- Ferrari, M.; Quaresima, V. Review: Near infrared brain and muscle oximetry: From the discovery to current applications. J. Near Infrared. Spectrosc. 2012, 20, 1–14. [Google Scholar] [CrossRef]
- Garvey, A.A.; Dempsey, E.M. Applications of near infrared spectroscopy in the neonate. Curr. Opin. Pediatr. 2018, 30, 209–215. [Google Scholar] [CrossRef]
- La Cour, A.; Greisen, G.; Hyttel-Sorensen, S. In vivo validation of cerebral near-infrared spectroscopy: A review. Neurophotonics 2018, 5, 040901. [Google Scholar] [CrossRef]
- Serraino, G.F.; Murphy, G.J. Effects of cerebral near-infrared spectroscopy on the outcome of patients undergoing cardiac surgery: A systematic review of randomised trials. BMJ Open. 2017, 7, e016613. [Google Scholar] [CrossRef]
- Yu, Y.; Zhang, K.; Zhang, L.; Zong, H.; Meng, L.; Han, R. Cerebral near-infrared spectroscopy (NIRS) for perioperative monitoring of brain oxygenation in children and adults. Cochrane Database Syst. Rev. 2018, 1, CD010947. [Google Scholar] [CrossRef]
- Lecrux, C.; Bourourou, M.; Hamel, E. How reliable is cerebral blood flow to map changes in neuronal activity? Auton. Neurosci. 2019, 217, 71–79. [Google Scholar] [CrossRef]
- Shetty, P.K.; Galeffi, F.; Turner, D.A. Cellular links between neuronal activity and energy homeostasis. Front. Pharmacol. 2012, 3, 43. [Google Scholar] [CrossRef]
- Tsao, A.; Galwaduge, P.T.; Kim, S.H.; Shaik, M.; Hillman, E.M.C. Measuring the thermodynamic effects of neurovascular coupling in the awake, behaving mouse brain. In Biomedical Optics 2016, OSA Technical Digest; Optical Society of America: Washington, DC, USA, 2016. [Google Scholar]
- Nakada, T.; Kwee, I.L. Fluid dynamics inside the brain barrier: Current concept of interstitial flow, glymphatic flow, and cerebrospinal fluid circulation in the brain. Neuroscientist 2019, 25, 155–166. [Google Scholar] [CrossRef]
- Scholkmann, F.; Kleiser, S.; Metz, A.J.; Zimmermann, R.; Pavia, J.M.; Wolf, U.; Wolf, M. A review on continuous wave functional near-infrared spectroscopy and imaging instrumentation and methodology. Neuroimage 2014, 85, 6–27. [Google Scholar] [CrossRef]
- Tachtsidis, I.; Scholkmann, F. False positives and false negatives in functional near-infrared spectroscopy: Issues, challenges, and the way forward. Neurophotonics 2016, 3, 031405. [Google Scholar] [CrossRef]
- Martelli, F. An ABC of near infrared photon migration in tissues: The diffusive regime of propagation. J. Near Infrared Spectrosc. 2012, 20, 29–42. [Google Scholar] [CrossRef]
- Ferrari, M.; Quaresima, V. A brief review on the history of human functional near-infrared spectroscopy (fNIRS) development and fields of application. Neuroimage 2012, 63, 921–935. [Google Scholar] [CrossRef]
- Yücel, M.A.; Selb, J.J.; Huppert, T.J.; Franceschini, M.A.; Boas, D.A. Functional near infrared spectroscopy: Enabling routine functional brain imaging. Curr. Opin. Biomed. Eng. 2017, 4, 78–86. [Google Scholar] [CrossRef]
- Nosrati, R.; Vesely, K.; Schweizer, T.A.; Toronov, V. Event-related changes of the prefrontal cortex oxygen delivery and metabolism during driving measured by hyperspectral fNIRS. Biomed. Opt. Express 2016, 7, 1323–1335. [Google Scholar] [CrossRef]
- Phan, P.; Highton, D.; Lai, J.; Smith, M.; Elwell, C.; Tachtsidis, I. Multi-channel multi-distance broadband near-infrared spectroscopy system to measure the spatial response of cellular oxygen metabolism and tissue oxygenation. Biomed. Opt. Express 2016, 7, 4424–4440. [Google Scholar] [CrossRef]
- Giannoni, L.; Lange, F.; Tachtsidis, I. Hyperspectral imaging solutions for brain tissue metabolic and hemodynamic monitoring: Past, current and future developments. J. Opt. 2018, 20, 044009. [Google Scholar] [CrossRef]
- Torricelli, A.; Contini, D.; Pifferi, A.; Caffini, M.; Re, R.; Zucchelli, L.; Spinelli, L. Time domain functional NIRS imaging for human brain mapping. Neuroimage 2014, 85, 28–50. [Google Scholar] [CrossRef]
- Yamada, Y.; Suzuki, H.; Yamashita, Y. Time-domain near-infrared spectroscopy and imaging: A review. Appl. Sci. 2019, 9, 1127. [Google Scholar] [CrossRef]
- Lange, F.; Tachtsidis, I. Clinical brain monitoring with time domain NIRS: A review and future perspectives. Appl. Sci. 2019, 9, 1612. [Google Scholar] [CrossRef]
- Hoshi, Y.; Yamada, Y. Overview of diffuse optical tomography and its clinical applications. J. Biomed. Opt. 2016, 21, 091312. [Google Scholar] [CrossRef]
- Lee, C.W.; Cooper, R.J.; Austin, T. Diffuse optical tomography to investigate the newborn brain. Pediatr. Res. 2017, 82, 376–386. [Google Scholar] [CrossRef]
- Zhao, H.; Cooper, R.J. Review of recent progress toward a fiberless, whole-scalp diffuse optical tomography system. Neurophotonics 2018, 5, 011012. [Google Scholar] [CrossRef]
- Strangman, G.E.; Ivkovic, V.; Zhang, Q. Wearable brain imaging with multimodal physiological monitoring. J. Appl. Physiol. 2018, 124, 564–572. [Google Scholar] [CrossRef]
- Pinti, P.; Aichelburg, C.; Gilbert, S.; Hamilton, A.; Hirsch, J.; Burgess, P.; Tachtsidis, I. A review on the use of wearable functional near-infrared spectroscopy in naturalistic environments. Jpn. Psychol. Res. 2018, 60, 347–373. [Google Scholar] [CrossRef]
- Orihuela-Espina, F.; Leff, D.R.; James, D.R.C.; Darzi, A.W.; Yang, G.Z. Quality control and assurance in functional near infrared spectroscopy (fNIRS) experimentation. Phys. Med. Biol. 2010, 55, 3701–3724. [Google Scholar] [CrossRef]
- Kassab, A.; Le Lan, J.; Vannasing, P.; Sawan, M. Functional near-infrared spectroscopy caps for brain activity monitoring: A review. Appl. Opt. 2015, 54, 576–586. [Google Scholar] [CrossRef]
- Brigadoi, S.; Cooper, R. How short is short? Optimum source–detector distance for short-separation channels in functional near-infrared spectroscopy. Neurophotonics 2015, 2, 025005. [Google Scholar] [CrossRef]
- Yücel, M.A.; Selb, J.; Aasted, C.M.; Lin, P.Y.; Borsook, D.; Becerra, L.; Boas, D.A. Mayer waves reduce the accuracy of estimated hemodynamic response functions in functional near-infrared spectroscopy. Biomed. Opt. Express 2016, 7, 3078–3088. [Google Scholar] [CrossRef]
- Pinti, P.; Scholkmann, F.; Hamilton, A.; Burgess, P.; Tachtsidis, I. Current status and issues regarding pre-processing of fNIRS neuroimaging data: An investigation of diverse signal filtering methods within a general linear model framework. Front. Hum. Neurosci. 2019, 12, 505. [Google Scholar] [CrossRef]
- Tsuzuki, D.; Dan, I. Spatial registration for functional near-infrared spectroscopy: From channel position on the scalp to cortical location in individual and group analyses. Neuroimage 2014, 85, 92–103. [Google Scholar] [CrossRef]
- Aasted, C.M.; Yücel, M.A.; Cooper, R.J.; Dubb, J.; Tsuzuki, D.; Becerra, L.; Petkov, M.P.; Borsook, D.; Dan, I.; Boas, D.A. Anatomical guidance for functional near-infrared spectroscopy: AtlasViewer tutorial. Neurophotonics 2015, 2, 020801. [Google Scholar] [CrossRef]
- Tak, S.; Ye, J.C. Statistical analysis of fNIRS data: A comprehensive review. Neuroimage 2014, 85, 72–91. [Google Scholar] [CrossRef]
- De Roever, I.; Bale, G.; Mitra, S.; Meek, J.; Robertson, N.J.; Tachtsidis, I. Investigation of the pattern of the hemodynamic response as measured by functional near-infrared spectroscopy (fNIRS) studies in newborns, less than a month old: A systematic review. Front. Hum. Neurosci. 2018, 12, 371. [Google Scholar] [CrossRef]
- Issard, C.; Gervain, J. Variability of the hemodynamic response in infants: Influence of experimental design and stimulus complexity. Dev. Cognit. Neurosci. 2018, 33, 182–193. [Google Scholar] [CrossRef]
- Chiarelli, A.M.; Zappasodi, F.; Di Pompeo, F.; Merla, A. Simultaneous functional near-infrared spectroscopy and electroencephalography for monitoring of human brain activity and oxygenation: A review. Neurophotonics 2017, 4, 041411. [Google Scholar] [CrossRef]
- Scarapicchia, V.; Brown, C.; Mayo, C.; Gawryluk, J.R. Functional magnetic resonance imaging and functional near-infrared spectroscopy: Insights from combined recording studies. Front. Hum. Neurosci. 2017, 11, 419. [Google Scholar] [CrossRef]
- Curtin, A.; Tong, S.; Sun, J.; Wang, J.; Onaral, B.; Ayaz, H. A systematic review of integrated functional near-infrared spectroscopy (fNIRS) and transcranial magnetic stimulation (TMS) studies. Front. Hum. Neurosci. 2019, 13, 84. [Google Scholar] [CrossRef]
- Pinti, P.; Tachtsidis, I.; Hamilton, A.; Hirsch, J.; Aichelburg, C.; Gilbert, S.; Burgess, P.W. The present and future use of functional near-infrared spectroscopy (fNIRS) for cognitive neuroscience. Ann. N. Y. Acad. Sci. 2018. [Google Scholar] [CrossRef]
- Quaresima, V.; Ferrari, M. Functional near-infrared spectroscopy (fNIRS) for assessing cerebral cortex function during human behavior in natural/social situations: A concise review. Organ. Res. Methods 2019, 22, 46–68. [Google Scholar] [CrossRef]
- Boas, D.A.; Elwell, C.E.; Ferrari, M.; Taga, G. Twenty years of functional near-infrared spectroscopy: Introduction for the special issue. Neuroimage 2014, 85, 1–5. [Google Scholar] [CrossRef]
- Val-Laillet, D.; Aarts, E.; Weber, B.; Ferrari, M.; Quaresima, V.; Stoeckel, L.E.; Alonso-Alonso, M.; Audette, M.; Malbert, C.H.; Stice, E. Neuroimaging and neuromodulation approaches to study eating behavior and prevent and treat eating disorders and obesity. Neuroimage Clin. 2015, 8, 1–31. [Google Scholar] [CrossRef]
- Aslin, R.N.; Shukla, M.; Emberson, L.L. Hemodynamic correlates of cognition in human infants. Annu. Rev. Psychol. 2015, 66, 349–379. [Google Scholar] [CrossRef]
- Vanderwert, R.E.; Nelson, C.A. The use of near-infrared spectroscopy in the study of typical and atypical development. Neuroimage 2014, 85, 264–271. [Google Scholar] [CrossRef]
- Wilcox, T.; Biondi, M. fNIRS in the developmental sciences. Wiley Interdiscip. Rev. Cognit. Sci. 2015, 6, 263–283. [Google Scholar] [CrossRef]
- Soltanlou, M.; Sitnikova, M.A.; Nuerk, H.C.; Dresler, T. Applications of functional near-infrared spectroscopy (fNIRS) in studying cognitive development: The case of mathematics and language. Front. Psychol. 2018, 9, 277. [Google Scholar] [CrossRef]
- Bendall, R.C.; Eachus, P.; Thompson, C. A brief review of research using near-infrared spectroscopy to measure activation of the prefrontal cortex during emotional processing: The importance of experimental design. Front. Hum. Neurosci. 2016, 10, 529. [Google Scholar] [CrossRef]
- Herold, F.; Wiegel, P.; Scholkmann, F.; Müller, N.G. Applications of functional near-infrared spectroscopy (fNIRS) neuroimaging in exercise–cognition science: A systematic, methodology-focused review. J. Clin. Med. 2018, 7, 466. [Google Scholar] [CrossRef]
- Homae, F. A brain of two halves: Insights into interhemispheric organization provided by near-infrared spectroscopy. Neuroimage 2014, 85, 354–362. [Google Scholar] [CrossRef]
- Cutini, S.; Basso Moro, S.; Bisconti, S. Functional near infrared optical imaging in cognitive neuroscience: An introductory review. J. Near Infrared Spectrosc. 2012, 20, 75–92. [Google Scholar] [CrossRef]
- McDonald, N.M.; Perdue, K.L. The infant brain in the social world: Moving toward interactive social neuroscience with functional near-infrared spectroscopy. Neurosci. Biobehav. Rev. 2018, 87, 38–49. [Google Scholar] [CrossRef]
- Kopton, I.M.; Kenning, P. Near-infrared spectroscopy (NIRS) as a new tool for neuroeconomic research. Front. Hum. Neurosci. 2014, 8, 549. [Google Scholar] [CrossRef]
- Quaresima, V.; Bisconti, S.; Ferrari, M. A brief review on the use of functional near-infrared spectroscopy (fNIRS) for language imaging studies in human newborns and adults. Brain Lang. 2012, 121, 79–89. [Google Scholar] [CrossRef]
- Rossi, S.; Telkemeyer, S.; Wartenburger, I.; Obrig, H. Shedding light on words and sentences: Near-infrared spectroscopy in language research. Brain Lang. 2012, 121, 152–163. [Google Scholar] [CrossRef]
- Curtin, A.; Ayaz, H. The age of neuroergonomics: Towards ubiquitous and continuous measurement of brain function with fNIRS. Jpn. Psychol. Res. 2018, 60, 374–386. [Google Scholar] [CrossRef]
- Zhu, Y.; Rodriguez-Paras, C.; Rhee, J.; Mehta, R.K. Methodological approaches and recommendations for functional near-infrared spectroscopy applications in HF/E research. Hum. Factors 2019. [Google Scholar] [CrossRef]
- Naseer, N.; Hong, K.S. fNIRS-based brain-computer interfaces: A review. Front. Hum. Neurosci. 2015, 9, 3. [Google Scholar] [CrossRef]
- Liu, T.; Pelowski, M.; Pang, C.; Zhou, Y.; Cai, J. Near-infrared spectroscopy as a tool for driving research. Ergonomics 2016, 59, 368–379. [Google Scholar] [CrossRef]
- Lohani, M.; Payne, B.R.; Strayer, D.L. A review of psychophysiological measures to assess cognitive states in real-world driving. Front. Hum. Neurosci. 2019, 13, 57. [Google Scholar] [CrossRef]
- Ahn, S.; Jun, S.C. Multi-modal integration of EEG-fNIRS for brain-computer interfaces—Current limitations and future directions. Front. Hum. Neurosci. 2017, 11, 503. [Google Scholar] [CrossRef]
- Hong, K.S.; Khan, M.J.; Hong, M.J. Feature extraction and classification methods for hybrid fNIRS-EEG brain-computer interfaces. Front. Hum. Neurosci. 2018, 12, 246. [Google Scholar] [CrossRef]
- Scholkmann, F.; Holper, L.; Wolf, U.; Wolf, M. A new methodical approach in neuroscience: Assessing inter-personal brain coupling using functional near-infrared imaging (fNIRI) hyperscanning. Front. Hum. Neurosci. 2013, 7, 813. [Google Scholar] [CrossRef]
- Minagawa, Y.; Xu, M.; Morimoto, S. Toward interactive social neuroscience: Neuroimaging real-world interactions in various populations. Jpn. Psychol. Res. 2018, 60, 374–386. [Google Scholar] [CrossRef]
- Wang, M.Y.; Luan, P.; Zhang, J.; Xiang, Y.T.; Niu, H.; Yuan, Z. Concurrent mapping of brain activation from multiple subjects during social interaction by hyperscanning: A mini-review. Quant. Imaging Med. Surg. 2018, 8, 819–837. [Google Scholar] [CrossRef]
- Herold, F.; Wiegel, P.; Scholkmann, F.; Thiers, A.; Hamacher, D.; Schega, L. Functional near-infrared spectroscopy in movement science: A systematic review on cortical activity in postural and walking tasks. Neurophotonics 2017, 4, 041403. [Google Scholar] [CrossRef]
- Niu, H.; He, Y. Resting-state functional brain connectivity: Lessons from functional near-infrared spectroscopy. Neuroscientist 2014, 20, 173–188. [Google Scholar] [CrossRef]
- Vitorio, R.; Stuart, S.; Rochester, L.; Alcock, L.; Pantall, A. Fnirs response during walking - Artefact or cortical activity? A systematic review. Neurosci. Biobehav. Rev. 2017, 83, 160–172. [Google Scholar] [CrossRef]
- Pelicioni, P.H.S.; Tijsma, M.; Lord, S.R.; Menant, J. Prefrontal cortical activation measured by fNIRS during walking: Effects of age, disease and secondary task. Peer J. 2019, 7, e6833. [Google Scholar] [CrossRef]
- Stuart, S.; Vitorio, R.; Morris, R.; Martini, D.N.; Fino, P.C.; Mancini, M. Cortical activity during walking and balance tasks in older adults and in people with Parkinson’s disease: A structured review. Maturitas 2018, 113, 53–72. [Google Scholar] [CrossRef]
- Mauri, M.; Nobile, M.; Bellina, M.; Crippa, A.; Brambilla, P. Light up ADHD: I. Cortical hemodynamic responses measured by functional near infrared spectroscopy (fNIRS). J. Affect. Disord. 2018, 234, 358–364. [Google Scholar] [CrossRef]
- Basura, G.J.; Hu, X.S.; Juan, J.S.; Tessier, A.M.; Kovelman, I. Human central auditory plasticity: A review of functional near-infrared spectroscopy (fNIRS) to measure cochlear implant performance and tinnitus perception. Laryngoscope Investig. Otolaryngol. 2018, 3, 463–472. [Google Scholar] [CrossRef]
- Liu, T.; Liu, X.; Yi, L.; Zhu, C.; Markey, P.S.; Pelowski, M. Assessing autism at its social and developmental roots: A review of autism spectrum disorder studies using functional near-infrared spectroscopy. Neuroimage 2019, 185, 955–967. [Google Scholar] [CrossRef]
- Zhang, F.; Roeyers, H. Exploring brain functions in autism spectrum disorder: A systematic review on functional near-infrared spectroscopy (fNIRS) studies. Int. J. Psychophysiol. 2019, 137, 41–53. [Google Scholar] [CrossRef]
- Agbangla, N.F.; Audiffren, M.; Albinet, C.T. Use of near-infrared spectroscopy in the investigation of brain activation during cognitive aging: A systematic review of an emerging area of research. Ageing Res. Rev. 2017, 38, 52–66. [Google Scholar] [CrossRef]
- Grazioli, S.; Mauri, M.; Crippa, A.; Maggioni, E.; Molteni, M.; Brambilla, P.; Nobile, M. Light up ADHD: II. Neuropharmacological effects measured by near infrared spectroscopy: Is there a biomarker? J. Affect. Disord. 2019, 244, 100–106. [Google Scholar] [CrossRef]
- Peng, K.; Pouliot, P.; Lesage, F.; Nguyen, D.K. Multichannel continuous electroencephalography-functional near-infrared spectroscopy recording of focal seizures and interictal epileptiform discharges in human epilepsy: A review. Neurophotonics 2016, 3, 031402. [Google Scholar] [CrossRef]
- Gramigna, V.; Pellegrino, G.; Cerasa, A.; Cutini, S.; Vasta, R.; Olivadese, G.; Martino, I.; Quattrone, A. Near-infrared spectroscopy in gait disorders: Is it time to begin? Neurorehabilit. Neural Repair 2017, 31, 402–412. [Google Scholar] [CrossRef]
- Beishon, L.; Haunton, V.J.; Panerai, R.B.; Robinson, T.G. Cerebral hemodynamics in mild cognitive impairment: A systematic review. J. Alzheimers Dis. 2017, 59, 369–385. [Google Scholar] [CrossRef]
- Ehlis, A.; Barth, B.; Hudak, J.; Storchak, H.; Weber, L.; Kimmig, A.S.; Kreifelts, B.; Dresler, T.; Fallgatter, A.J. Near-infrared spectroscopy as a new tool for neurofeedback training: Applications in psychiatry and methodological considerations. Jpn. Psychol. Res. 2018, 60, 225–241. [Google Scholar] [CrossRef]
- Benoit, B.; Martin-Misener, R.; Newman, A.; Latimer, M.; Campbell-Yeo, M. Neurophysiological assessment of acute pain in infants: A scoping review of research methods. Acta Paediatr. 2017, 106, 1053–1066. [Google Scholar] [CrossRef]
- Rupawala, M.; Dehghani, H.; Lucas, S.J.E.; Tino, P.; Cruse, D. Shining a light on awareness: A review of functional near-infrared spectroscopy for prolonged disorders of consciousness. Front. Neurol. 2018, 9, 350. [Google Scholar] [CrossRef]
- Ehlis, A.C.; Schneider, S.; Dresler, T.; Fallgatter, A.J. Application of functional near-infrared spectroscopy in psychiatry. Neuroimage 2014, 85, 478–488. [Google Scholar] [CrossRef]
- Berger, A.; Horst, F.; Müller, S.; Steinberg, F.; Doppelmayr, M. Current state and future prospects of EEG and fNIRS in robot-assisted gait rehabilitation: A brief review. Front. Hum. Neurosci. 2019, 13, 172. [Google Scholar] [CrossRef]
- Kumar, V.; Shivakumar, V.; Chhabra, H.; Bose, A.; Venkatasubramanian, G.; Gangadhar, B.N. Functional near infra-red spectroscopy (fNIRS) in schizophrenia: A review. Asian J. Psychiatr. 2017, 27, 18–31. [Google Scholar] [CrossRef]
- Yang, M.; Yang, Z.; Yuan, T.; Feng, W.; Wang, P. A systemic review of functional near-infrared spectroscopy for stroke: Current application and future directions. Front. Neurol. 2019, 10, 58. [Google Scholar] [CrossRef]
- Jasińska, K.K.; Guei, S. Neuroimaging field methods using functional near infrared spectroscopy (NIRS) neuroimaging to study global child development: Rural sub-saharan africa. J. Vis. Exp. 2018, 132, e57165. [Google Scholar] [CrossRef]
- Reindl, V.; Konrad, K.; Gerloff, C.; Kruppa, J.A.; Bell, L.; Scharke, W. Conducting hyperscanning experiments with functional near-infrared spectroscopy. J. Vis. Exp. 2019, 143, e58807. [Google Scholar] [CrossRef]
- Sukal-Moulton, T.; de Campos, A.C.; Stanley, C.J.; Damiano, D.L. Functional near infrared spectroscopy of the sensory and motor brain regions with simultaneous kinematic and EMG monitoring during motor tasks. J. Vis. Exp. 2014, 94, e52391. [Google Scholar] [CrossRef]
- Noah, J.A.; Ono, Y.; Nomoto, Y.; Shimada, S.; Tachibana, A.; Zhang, X.; Bronner, S.; Hirsch, J. Fmri validation of fNIRS measurements during a naturalistic task. J. Vis. Exp. 2015, 100, e52116. [Google Scholar] [CrossRef]
- Pinti, P.; Aichelburg, C.; Lind, F.; Power, S.; Swingler, E.; Merla, A.; Hamilton, A.; Gilbert, S.; Burgess, P.; Tachtsidis, I. Using fiberless, wearable fNIRS to monitor brain activity in real-world cognitive tasks. J. Vis. Exp. 2015, 106, e53336. [Google Scholar] [CrossRef]
- Villringer, A.; Chance, B. Non-invasive optical spectroscopy and imaging of human brain function. Trends Neurosci. 1997, 20, 435–442. [Google Scholar] [CrossRef]
- Villringer, A.; Planck, J.; Hock, C.; Schleinkofer, L.; Dirnagl, U. Near infrared spectroscopy (NIRS): A new tool to study hemodynamic changes during activation of brain function in human adults. Neurosci. Lett. 1993, 154, 101–104. [Google Scholar] [CrossRef]
- Strangman, G.; Culver, J.P.; Thompson, J.H.; Boas, D.A. A quantitative comparison of simultaneous BOLD fMRI and NIRS recordings during functional brain activation. Neuroimage 2002, 17, 719–731. [Google Scholar] [CrossRef]
- Obrig, H.; Villringer, A. Beyond the visible—Imaging the human brain with light. J. Cereb. Blood Flow Metab. 2003, 23, 1–18. [Google Scholar] [CrossRef]
- Maki, A.; Yamashita, Y.; Ito, Y.; Watanabe, E.; Mayanagi, Y.; Koizumi, H. Spatial and temporal analysis of human motor activity using noninvasive NIR topography. Med. Phys. 1995, 22, 1997–2005. [Google Scholar] [CrossRef]
- Strangman, G.; Boas, D.A.; Sutton, J.P. Non-invasive neuroimaging using near-infrared light. Biol. Psychiatry 2002, 52, 679–693. [Google Scholar] [CrossRef]
- Boas, D.A.; Dale, A.M.; Franceschini, M.A. Diffuse optical imaging of brain activation: Approaches to optimizing image sensitivity, resolution, and accuracy. Neuroimage 2004, 23, S275–S288. [Google Scholar] [CrossRef]
- Hoshi, Y.; Kobayashi, N.; Tamura, M. Interpretation of near-infrared spectroscopy signals: A study with a newly developed perfused rat brain model. J. Appl. Physiol. 2001, 90, 1657–1662. [Google Scholar] [CrossRef]
- Ward, J.; Pinti, P. Wearables and the brain. IEEE Pervasive Comput. 2019, 18, 94–100. [Google Scholar] [CrossRef]
- Buttafava, M.; Martinenghi, E.; Tamborini, D.; Contini, D.; Mora, A.D.; Renna, M.; Torricelli, A.; Pifferi, A.; Zappa, F.; Tosi, A. A compact two-wavelength time-domain NIRS system based on SiPM and pulsed diode lasers. IEEE Photonics J. 2017, 9, 7792611. [Google Scholar] [CrossRef]
- Di Sieno, L.; Dalla Mora, A.; Torricelli, A.; Spinelli, L.; Re, R.; Pifferi, A.; Contini, D. A versatile setup for time-resolved functional near infrared spectroscopy based on fast-gated single-photon avalanche diode and on four-wave mixing laser. Appl. Sci. 2019, 9, 2366. [Google Scholar] [CrossRef]
- Lange, F.; Dunne, F.; Hale, L.; Tachtsidis, I. Maestros: A multiwavelength time-domain NIRS system to monitor changes in oxygenation and oxidation state of cytochrome-c-oxidase. IEEE J. Sel. Top. Quant. Electron. 2019, 25, 1–12. [Google Scholar] [CrossRef]
- Bale, G.; Elwell, C.E.; Tachtsidis, I. From Jöbsis to the present day: A review of clinical near-infrared spectroscopy measurements of cerebral cytochrome-c-oxidase. J. Biomed. Opt. 2016, 21, 091307. [Google Scholar] [CrossRef]
- De Roever, I.; Bale, G.; Cooper, R.J.; Tachtsidis, I. Functional NIRS measurement of cytochrome-c-oxidase demonstrates a more brain-specific marker of frontal lobe activation compared to the haemoglobins. Adv. Exp. Med. Biol. 2017, 977, 141–147. [Google Scholar]
- Scholkmann, F.; Hafner, T.; Metz, A.J.; Wolf, M.; Wolf, U. Effect of short-term colored-light exposure on cerebral hemodynamics and oxygenation, and systemic physiological activity. Neurophotonics 2017, 4, 045005. [Google Scholar] [CrossRef]
- Beisteiner, R.; Pernet, C.; Stippich, C. Can we standardize clinical functional neuroimaging procedures? Front. Neurol. 2019, 9, 1153. [Google Scholar] [CrossRef]
- Woods, A.J.; Bikson, M.; Chelette, K.; Dmochowski, J.; Dutta, A.; Esmaeilpour, Z.; Gebodh, N.; Nitsche, M.A.; Stagg, C. Transcranial direct current stimulation integration with magnetic resonance imaging, magnetic resonance spectroscopy, near infrared spectroscopy imaging and electroencephalography. In Practical Guide to Transcranial Direct Current Stimulation, 1st ed.; Knotkova, H., Nitsche, M., Bikson, M., Woods, A., Eds.; Springer International Publisher: Cham, Switzerland, 2019; pp. 293–345. [Google Scholar]
- Wintermark, M.; Colen, R.; Whitlow, C.T.; Zaharchuk, G. The vast potential and bright future of neuroimaging. Br. J. Radiol. 2018, 91, 20170505. [Google Scholar] [CrossRef]
- Perrey, S.; Besson, P. Studying brain activity in sports performance: Contributions and issues. Prog. Brain Res. 2018, 240, 247–267. [Google Scholar]
- Durduran, T.; Yodh, A.G. Diffuse correlation spectroscopy for non-invasive, micro-vascular cerebral blood flow measurement. Neuroimage 2014, 15, 51–63. [Google Scholar] [CrossRef]
- Roche-Labarbe, N.; Carp, S.A.; Surova, A.; Patel, M.; Boas, D.A.; Grant, P.E.; Franceschini, M.A. Noninvasive optical measures of CBV, StO(2), CBF index, and rCMRO(2) in human premature neonates’ brains in the first six weeks of life. Hum. Brain Mapp. 2010, 31, 341–352. [Google Scholar] [CrossRef]
- Andresen, B.; De Carli, A.; Fumagalli, M.; Giovannella, M.; Durduran, T.; Michael Weigel, U.; Contini, D.; Spinelli, L.; Torricelli, A.; Greisen, G. Cerebral oxygenation and blood flow in normal term infants at rest measured by a hybrid near-infrared device (BabyLux). Pediatr. Res. 2019. [Google Scholar] [CrossRef]
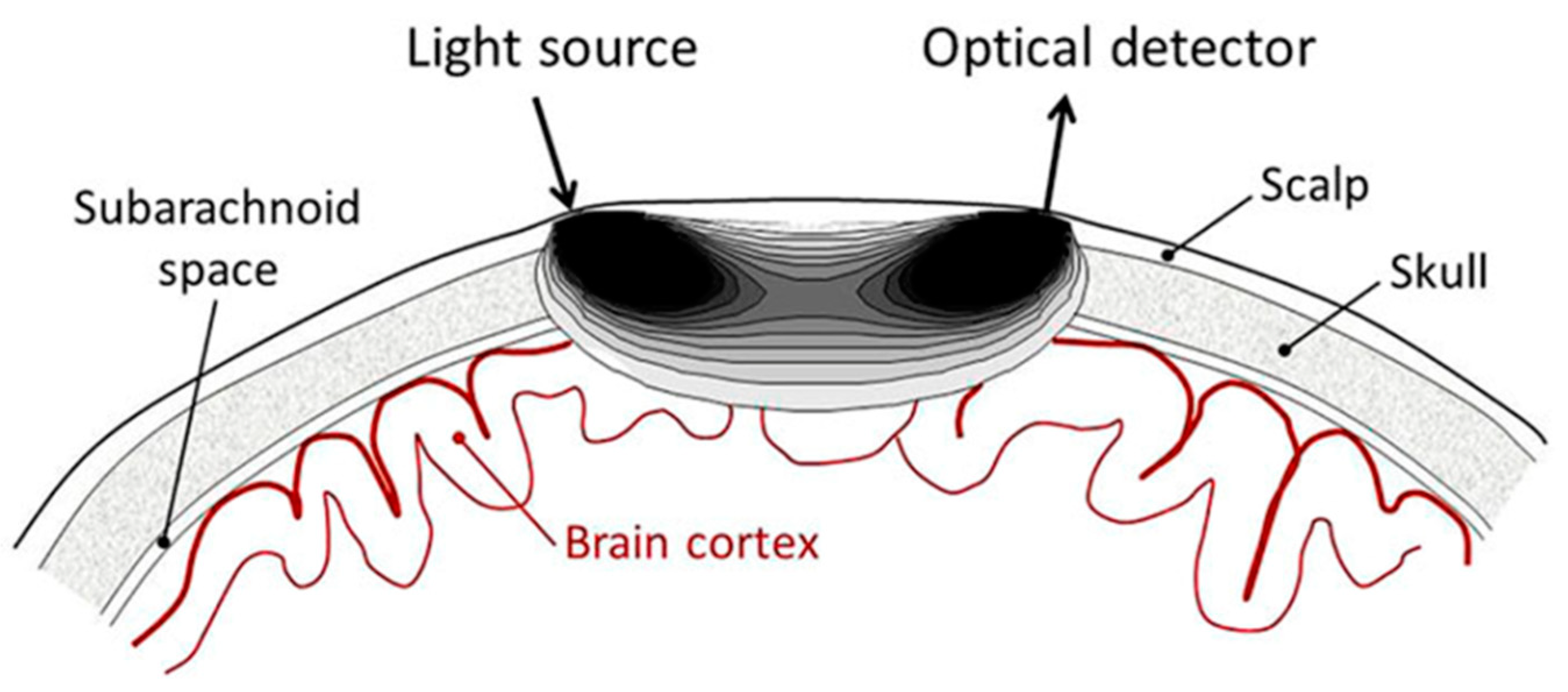
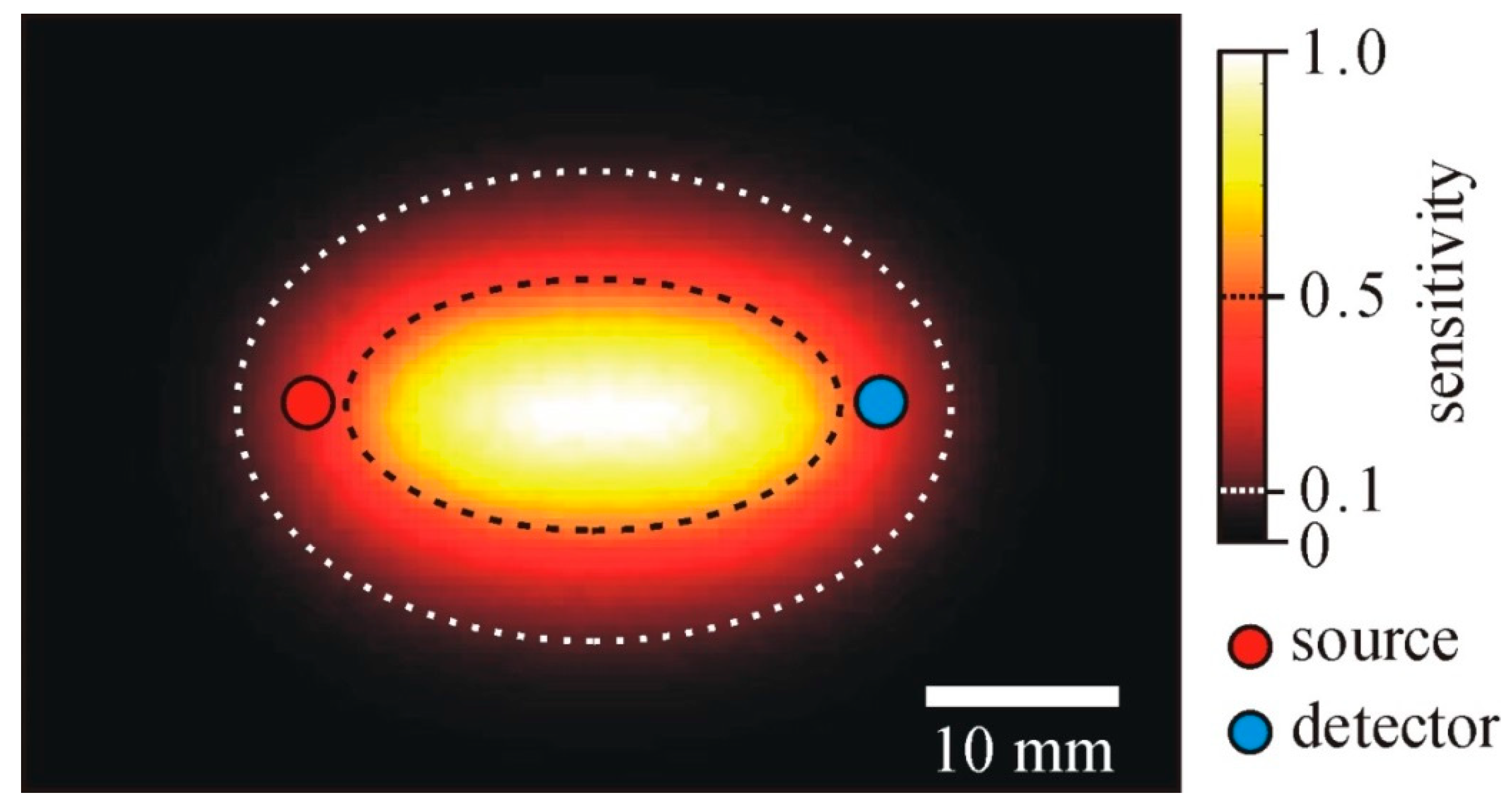

| Topic | Year | 1st Author [Ref] |
|---|---|---|
| Modeling near-infrared photon propagation in biological tissue | 2012 | Martelli [20] |
| 2016 | Bigio [4] | |
| 2018 | Fantini [5] | |
| History of fNIRS | 2012 | Ferrari [21] |
| State of the art of continuous-wave multispectral fNIRS instrumentation | 2014 | Scholkmann [18] |
| 2017 | Yücel [22] | |
| State of the art of continuous-wave hyperspectral fNIRS instrumentation | 2016 | Nsorati [23] |
| 2016 | Pham [24] | |
| 2018 | Giannoni [25] | |
| State of the art of time-domain fNIRS instrumentation | 2014 | Torricelli [26] |
| 2019 | Yamada [27] | |
| Clinical brain monitoring by time-domain fNIRS instrumentation | 2019 | Lange [28] |
| State of the art of diffuse optical imaging | 2016 | Hoshi [29] |
| 2017 | Lee [30] | |
| 2018 | Fantini [5] | |
| 2018 | Zhao [31] | |
| State of the art of wearable fNIRS | 2018 | Strangman [32] |
| 2018 | Pinti [33] | |
| State of the art of functional connectivity measurements | 2018 | Fantini [5] |
| Factors influencing fNIRS data and recommendations | 2010 | Orihuela-Espina [34] |
| Caps for long term fNIRS measurements | 2015 | Kassab [35] |
| Selection of the optimum source–detector distance | 2015 | Brigadoi [36] |
| Mayer waves interference | 2016 | Yücel [37] |
| Multiple components of the fNIRS signal | 2016 | Tachtsidis [19] |
| Signal pre-processing procedures | 2019 | Pinti [38] |
| Anatomical guidance for fNIRS | 2014 | Tsuzuki [39] |
| 2015 | Aasted [40] | |
| Statistical analysis of fNIRS data | 2014 | Tak [41] |
| Pattern of hemodynamic response in newborn < 1 month | 2018 | de Roever [42] |
| Pattern of hemodynamic response in infants | 2018 | Issard [43] |
Integration of fNIRS with:
| ||
| 2017 | Chiarelli [44] | |
| 2017 | Scarapicchia [45] | |
| 2019 | Curtin [46] | |
| Recent fNIRS general reviews including the advantages and limitations of fNIRS | 2018 | Fantini [5] |
| 2018 | Pinti [47] | |
| 2019 | Quaresima [48] |
| Field of Application | Topic | Year | N. | Subjects | 1st Author [Ref] |
|---|---|---|---|---|---|
| Psychology/education | Cognition and food | 2015 | 39 | A | Val-Laillet [50] |
| Cognition in infants | 2015 | 171 | C | Aslin [51] | |
| Development (typical and atypical) | 2014 | 29 | C | Vanderwert [52] | |
| 2015 | 149 | C | Wilcox [53] | ||
| Development of mathematics/language skills in children | 2018 | 7 | C | Soltanlou [54] | |
| Emotion | 2016 | 11 | A | Bendall [55] | |
| Influence of exercise on cognition | 2018 | 35 | A | Herold [56] | |
| Interhemispheric organization | 2014 | 32 | A | Homae [57] | |
| Psychology general review | 2012 | 106 | A | Cutini [58] | |
| Social development during infancy | 2018 | 29 | C | McDonald [59] | |
| Economics | Neuroeconomic research | 2014 | 15 | A | Kopton [60] |
| Linguistics | Language and its development | 2012 | 60 | A C | Quaresima [61] |
| Word and sentence processing | 2012 | 9 | C | Rossi [62] | |
| Neuroergonomics | Neuroergonomics and fNIRS | 2018 | 68 | A | Curtin [63] |
| 2019 | 37 | A | Zhu [64] | ||
| Functional Neuroimaging Basic Research | Brain computer interface | 2015 | 33 | A | Naseer [65] |
| Driving research | 2016 | 10 | A | Liu [66] | |
| 2019 | 13 | A | Lohani [67] | ||
| Hybrid fNIRS-EEG brain-computer interfaces | 2017 | 11 | A | Ahn [68] | |
| 2018 | 43 | A | Hong [69] | ||
| Hyperscanning with multi-subject measurements | 2013 | 7 | A | Scholkmann [70] | |
| 2018 | 15 | A | Minagawa [71] | ||
| 2018 | 18 | A | Wang [72] | ||
| Postural and walking tasks | 2017 | 57 | A | Herold [73] | |
| Resting-state functional brain connectivity | 2014 | 16 | A | Niu [74] | |
| Walking | 2017 | 31 | A | Vitorio [75] | |
| 2019 | 35 | A | Pelicioni [76] | ||
| Walking and balance tasks in older adults | 2018 | 24 | A | Stuart [77] | |
| Medicine | Attention deficit disorder | 2018 | 11 | C | Mauri [78] |
| Auditory cortex plasticity after cochlear implant | 2018 | 7 | A | Basura [79] | |
| Autism spectrum disorder | 2019 | 15 | C | Liu [80] | |
| 2019 | 30 | C | Zhang [81] | ||
| Cognitive aging | 2017 | 34 | A | Agbangla [82] | |
| Developmental age attention deficit/hyperactivity disorder | 2019 | 13 | C | Grazioli [83] | |
| Eating disorders | 2015 | 11 | A | Val-Laillet [50] | |
| Epilepsy | 2016 | 23 | A | Peng [84] | |
| Gait disorders | 2017 | 12 | A | Gramigna [85] | |
| Mild cognitive impairment | 2017 | 8 | A | Beishon [86] | |
| Neurofeedback training | 2018 | 127 | A | Ehlis [87] | |
| Pain assessment in infants | 2017 | 9 | C | Benoit [88] | |
| Parkinson’s disease and walking balance tasks | 2018 | 5 | A | Stuart [77] | |
| Prolonged disorder of consciousness | 2018 | 7 | A | Rupawala [89] | |
| Psychiatry | 2014 | 168 | A | Ehlis [90] | |
| Robot-assisted gait training | 2019 | 2 | A | Berger [91] | |
| Schizophrenic disorders | 2017 | 17 | A | Kumar [92] | |
| Stroke therapy/recovery/rehabilitation | 2019 | 66 | A | Yang [93] |
| Topic | Year | 1st Author [Ref] | Device, Company, Country | Number of Channels |
|---|---|---|---|---|
| Brain development. Language processing study (rhyme judgment task) on primary school aged children. | 2018 | Jasińska [94] | LightNIRS, Shimadzu, Japan | 47 |
| Hyper-scanning. Parent–child dyads for analyzing brain-to-brain synchrony during a cooperative and a competitive computer task. | 2019 | Reindl [95] | ETG-4000, Hitachi, Japan | 44 |
| Motor cortex activation during different motor tasks (cycling, walking) on adults. | 2014 | Sukal-Moulton [96] | CW6, TechEn, Milford, MA, USA | 24 |
| Temporal cortex activation during a dance video game task revealed by fNIRS and fMRI on adults. | 2015 | Noah [97] | LABNIRS, Shimadzu, Japan | 22 |
| Wearable fNIRS. Real-world ecological prospective memory tasks on adults. | 2015 | Pinti [98] | WOT-100, NeU Corporation, Japan | 16 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quaresima, V.; Ferrari, M. A Mini-Review on Functional Near-Infrared Spectroscopy (fNIRS): Where Do We Stand, and Where Should We Go? Photonics 2019, 6, 87. https://doi.org/10.3390/photonics6030087
Quaresima V, Ferrari M. A Mini-Review on Functional Near-Infrared Spectroscopy (fNIRS): Where Do We Stand, and Where Should We Go? Photonics. 2019; 6(3):87. https://doi.org/10.3390/photonics6030087
Chicago/Turabian StyleQuaresima, Valentina, and Marco Ferrari. 2019. "A Mini-Review on Functional Near-Infrared Spectroscopy (fNIRS): Where Do We Stand, and Where Should We Go?" Photonics 6, no. 3: 87. https://doi.org/10.3390/photonics6030087
APA StyleQuaresima, V., & Ferrari, M. (2019). A Mini-Review on Functional Near-Infrared Spectroscopy (fNIRS): Where Do We Stand, and Where Should We Go? Photonics, 6(3), 87. https://doi.org/10.3390/photonics6030087






