Residual Characteristics of Atrazine and Its Metabolites in the Liaoning Province of China
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Sample Collection and Preparation
2.3. Analytical Conditions
3. Results and Discussion
3.1. Development and Evaluation of the Analytical Method
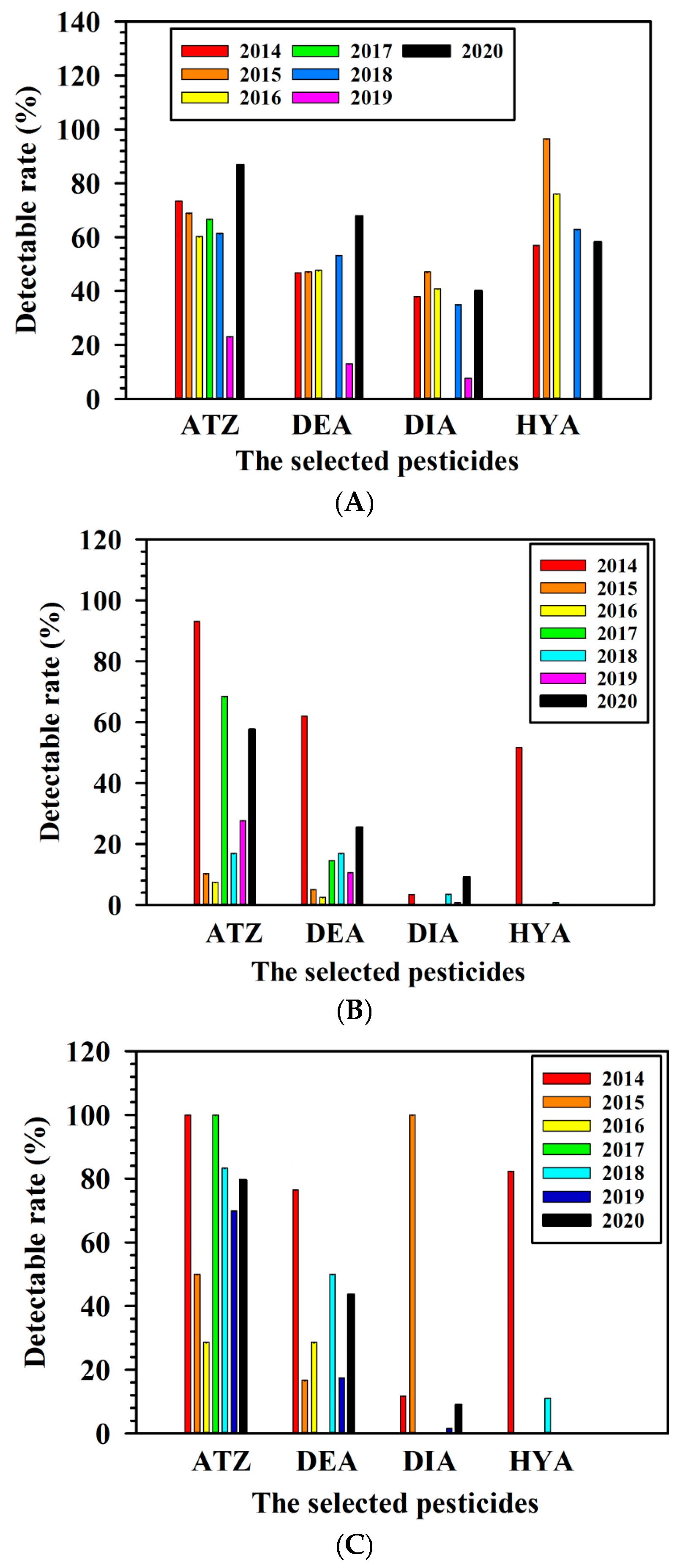
3.2. Detectable Rate of Pesticides in the Collected Samples
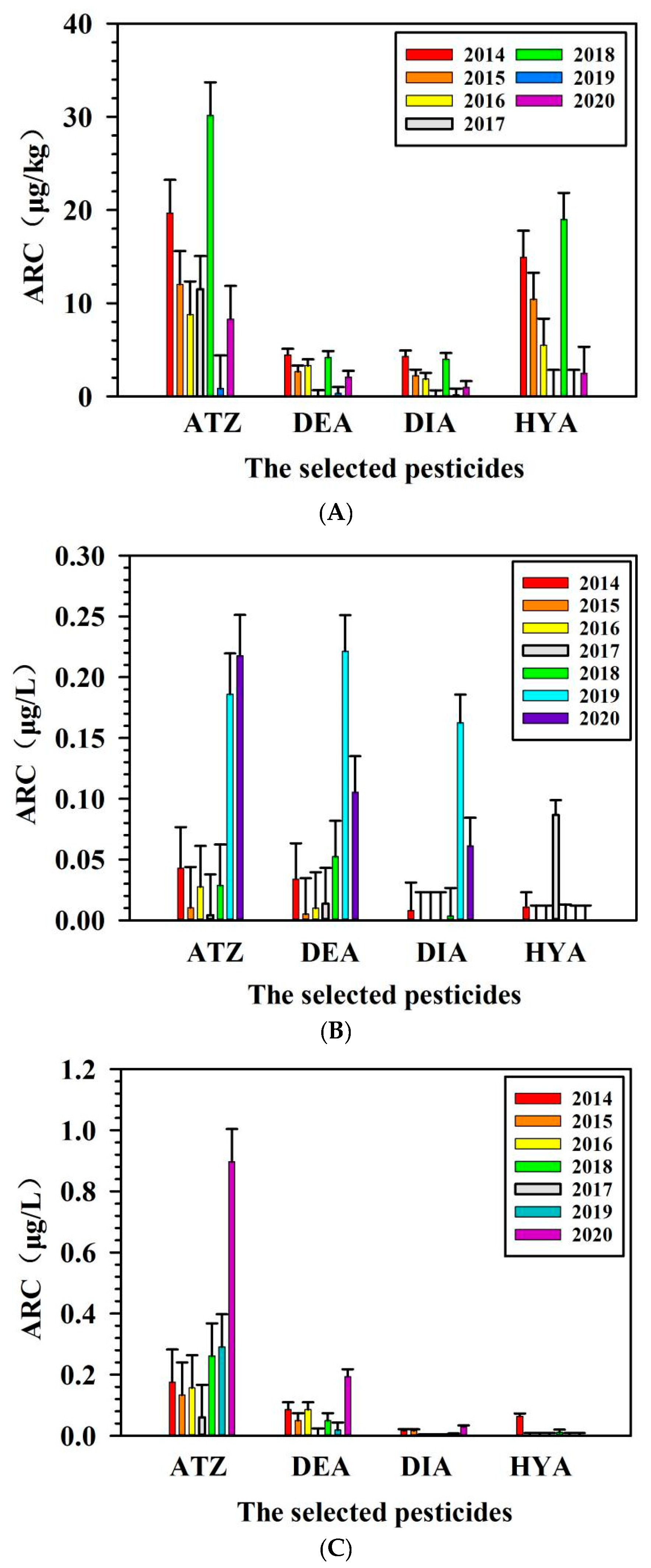
3.3. Detectable Level of Pesticides in the Collected Samples
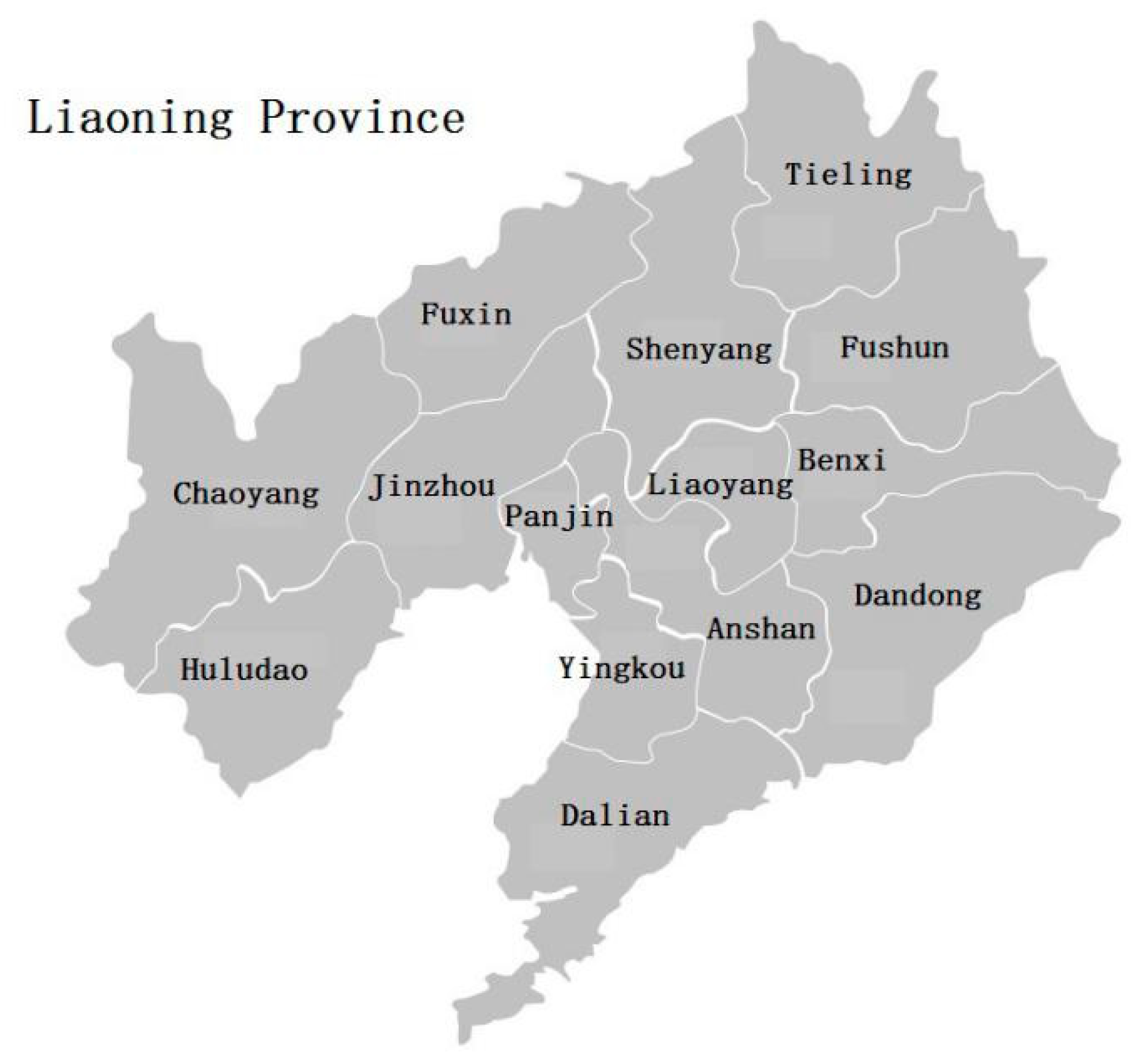
3.4. Residual Level of the Analytes in Different Regions of Liaoning in 2018
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kumar, V.; Vaid, K.; Bansal, S.A.; Kim, K.H. Nanomaterial-based immunosensors for ultrasensitive detection of pesticides/herbicides: Current status and perspectives. Biosens. Bioelectron. 2020, 165, 112382. [Google Scholar] [CrossRef] [PubMed]
- El-Nahhal, I.; El-Nahhal, Y. Pesticide residues in drinking water, their potential risk to human health and removal options. J. Environ. Manag. 2021, 299, 113611. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Hong, X.; Ma, L.; Liu, R.; Bu, Y. Non-targetedmetabolomic profiling of atrazine in Caenorhabditis elegans using UHPLC-QE Orbitrap/MS. Ecotoxicol. Environ. Saf. 2020, 206, 111170. [Google Scholar] [CrossRef] [PubMed]
- Mudhoo, A.; Garg, V.K. Sorption, Transport and Transformation of Atrazine in Soils, Minerals and Composts: A Review. Pedosphere 2011, 21, 11–25. [Google Scholar] [CrossRef]
- Qu, M.; Li, H.; Li, N.; Liu, G.; Zhao, J.; Hua, Y.; Zhu, D. Distribution of atrazine and its phytoremediation by submerged macrophytes in lake sediments. Chemosphere 2017, 168, 1515–1522. [Google Scholar] [CrossRef]
- Pérez, D.J.; Doucette, W.J.; Moore, M.T. Atrazine uptake, translocation, bioaccumulation and biodegradation in cattail (Typha latifolia) as a function of exposure time. Chemosphere 2022, 287, 132104. [Google Scholar] [CrossRef]
- Liu, Y.F.; Fan, X.X.; Zhang, T.; He, W.Y.; Song, F.Q. Effects of the long-term application of atrazine on soil enzyme activity and bacterial community structure in farmlands in China. Environ. Pollut. 2020, 262, 114264. [Google Scholar] [CrossRef]
- Jablonowski, N.D.; Koeppchen, S.; Hofmann, D.; Schaeffer, A.; Burauel, P. Persistence of 14C-labeled atrazine and its residues in a field lysimeter soil after 22 years. Environ. Pollut. 2009, 157, 2126–2131. [Google Scholar] [CrossRef]
- Salazar-Ledesma, M.; Prado, B.; Zamora, O.; Siebe, C. Mobility of atrazine in soils of a wastewater irrigated maize field. Agric. Ecosyst. Environ. 2018, 255, 73–83. [Google Scholar] [CrossRef]
- Dousset, S.; Mouvet, C.; Schiavon, M. Leaching of atrazine and some of its metabolites in undisturbed field lysimeters of three soil types. Chemosphere 1995, 30, 511–524. [Google Scholar] [CrossRef]
- Liu, Z.; Wang, Y.; Zhu, Z.; Yang, E.; Feng, X.; Fu, Z.; Jin, Y. Atrazine and its main metabolites alter the locomotor activity of larval zebrafish (Danio rerio). Chemosphere 2016, 148, 163–170. [Google Scholar] [CrossRef]
- Wang, A.; Hu, X.; Wan, Y.; Mahai, G.; Jiang, Y.; Huo, W.; Zhao, X.; Liang, G.; He, Z.; Xia, W.; et al. A nationwide study of the occurrence and distribution of atrazine and its degradates in tap water and groundwater in China: Assessment of human exposure potential. Chemosphere 2020, 252, 126533. [Google Scholar] [CrossRef]
- Bohn, T.; Cocco, E.; Gourdol, L.; Guignard, C.; Hoffmann, L. Determination of atrazine and degradation products in Luxembourgish drinking water: Origin and fate of potential endocrine-disrupting pesticides. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2011, 28, 1041–1054. [Google Scholar] [CrossRef]
- Montiel-León, M.J.; Duy, S.V.; Munoz, G.; Bouchard, M.F.; Amyot, M.; Sauvé, S. Quality survey and spatiotemporal variations of atrazine and desethylatrazine in drinking water in Quebec, Canada. Sci. Total Environ. 2019, 671, 578–585. [Google Scholar] [CrossRef]
- Jin, Y.; Wang, L.; Chen, G.; Lin, X.; Miao, W.; Fu, Z. Exposure of mice to atrazine and its metabolite diaminochlorotriazine elicits oxidative stress and endocrine disruption. Environ. Toxicol. Pharmacol. 2014, 37, 782–790. [Google Scholar] [CrossRef]
- Xue, Y.; Zhang, Z.M.; Zhang, R.R.; Li, Y.Q.; Sun, A.L.; Shi, X.Z.; Chen, J.; Song, S. Aquaculture-derived distribution, partitioning, migration, and transformation of atrazine and its metabolites in seawater, sediment, and organisms from a typical semi-closed mariculture bay. Environ. Pollut. 2021, 271, 116362. [Google Scholar] [CrossRef]
- Triassi, M.; Montuori, P.; Provvisiero, D.P.; Rosa, E.D.; Duca, F.D.; Sarnacchiaro, P.; Díez, S. Occurrence and spatial-temporal distribution of atrazine and its metabolites in the aquatic environment of the Volturno River estuary, southern Italy. Sci. Total Environ. 2022, 803, 149972. [Google Scholar] [CrossRef]
- Rostami, S.; Jafari, S.; Moeini, Z.; Jaskulak, M.; Keshtgar, L.; Badeenezhad, A.; Azhdarpoor, A.; Rostami, M.; Zorena, K.; Dehghani, M. Current methods and technologies for degradation of atrazine in contaminated soil and water: A review. Environ. Technol. Innov. 2021, 24, 102019. [Google Scholar] [CrossRef]
- Gabardo, R.P.; Toyama, N.P.; Amaral, B.D.; Boroski, M.; Toci, A.T.; Benassi, S.F.; Peralta-Zamora, P.G.; Cordeiro, G.A.; Liz, M.V.D. Determination of atrazine and main metabolites in natural waters based on a simple method of QuEChERS and liquid chromatography coupled to a diode-array detector. Microchem. J. 2021, 168, 106392. [Google Scholar] [CrossRef]
- Min, G.; Wang, S.; Zhu, H.; Fang, G.; Zhang, Y. Multi-walled carbon nanotubes as solid-phase extraction adsorbents for determination of atrazine and its principal metabolites in water and soil samples by gas chromatography-mass spectrometry. Sci. Total Environ. 2008, 396, 79–85. [Google Scholar] [CrossRef]
- Zumpano, R.; Manghisi, M.; Polli, F.; D’Agostino, C.; Ietto, F.; Favero, G.; Mazzei, F. Label-free magnetic nanoparticles-based electrochemical immunosensor for atrazine detection. Anal. Bioanal. Chem. 2022, 414, 2055–2064. [Google Scholar] [CrossRef] [PubMed]
- Stavra, E.; Petroua, P.S.; Koukouvinos, G.; Kiritsis, C.; Pirmettis, I.; Papadopoulos, M.; Goustouridis, D.; Economou, A.; Misiakos, K.; Raptis, I.; et al. Simultaneous determination of paraquat and atrazine in water samples with a white light reflectance spectroscopy biosensor. J. Hazard. Mater. 2018, 359, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Buarque, F.S.; Lima, N.S.; Soares, C.M.F.; Marques, M.N.; Cavalcanti, E.B.; Pereira, M.M.; Souza, R.L.; Lima, A.S. Preconcentration and chromatographic detection of atrazine in real water sample using aqueous two-phase system based on tetrahydrofuran and glycerol. Environ. Qual. Manag. 2021, 31, 39–48. [Google Scholar] [CrossRef]
- Na, Y.; Sheng, W.; Yuan, M.; Li, L.L.; Liu, B.; Zhang, Y.; Wang, S. Enzyme-linked immunosorbent assay and immunochromatographic strip for rapid detection of atrazine in water samples. Microchim Acta 2012, 177, 177–184. [Google Scholar] [CrossRef]
- Cao, D.; He, S.; Li, X.; Shi, L.; Wang, F.; Yu, S.; Xu, S.; Ju, C.; Fang, H.; Yu, Y. Characterization, genome functional analysis, and detoxification of atrazine by Arthrobacter sp. C2. Chemosphere 2021, 264, 128514. [Google Scholar] [CrossRef]
- Hernandes, P.T.; Franco, D.S.P.; Georgin, J.; Salau, N.P.G.; Dotto, G.L. Investigation of biochar from Cedrella fissilis applied to the adsorption of atrazine herbicide from an aqueous medium. J. Environ. Chem. Eng. 2022, 10, 107408. [Google Scholar] [CrossRef]
- Gfrerer, M.; Wenzl, T.; Quan, X.; Platzer, B.; Lankmayr, E. Occurrence of triazines in surface and drinking water of Liaoning province in eastern china. J. Biochem. Biophys. Methods 2002, 53, 217–228. [Google Scholar] [CrossRef]
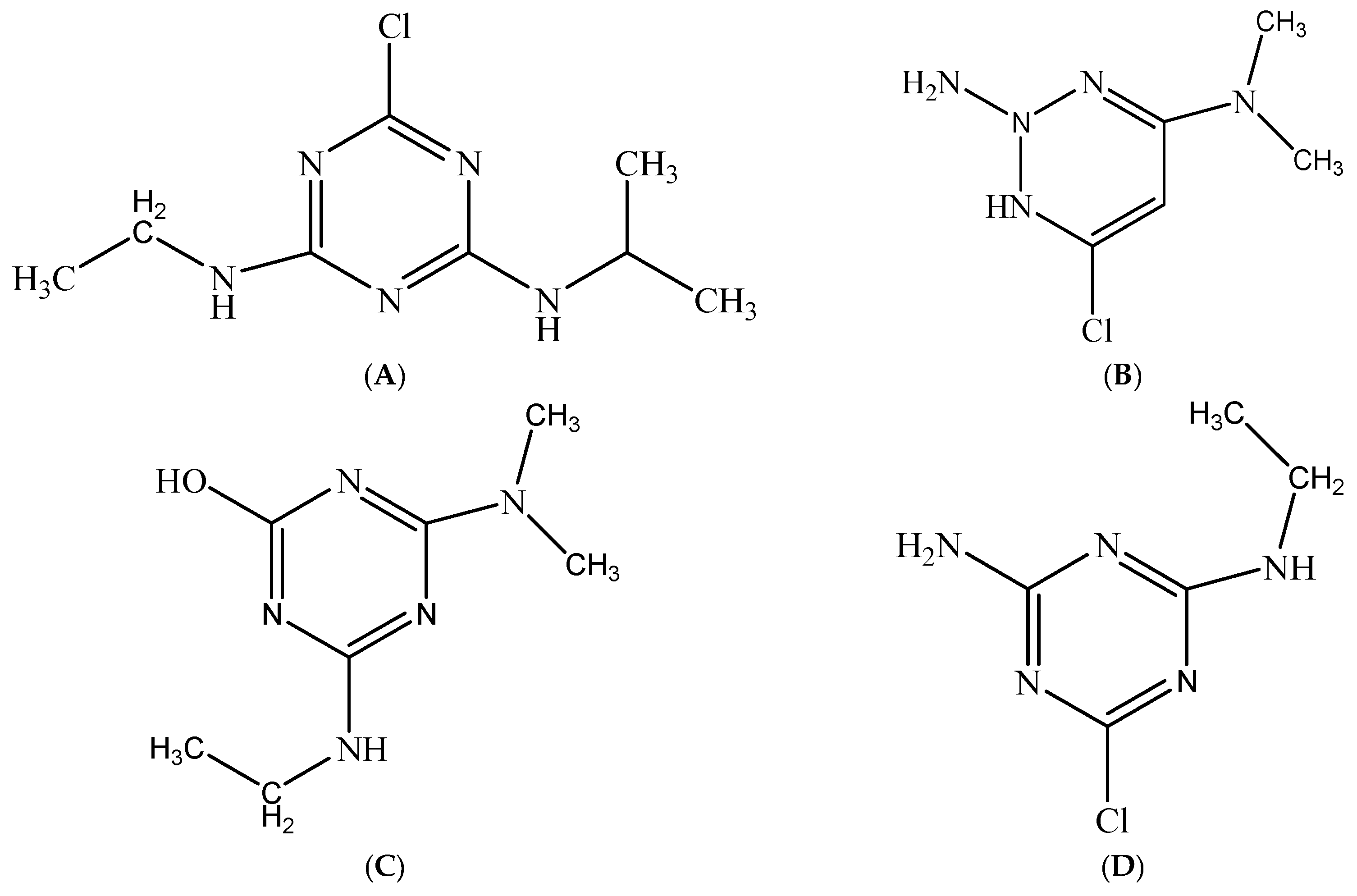

| Analyte | Retention Time min | Parent Ion m/z | Product Ion m/z | Dwell Time ms | Collision Energy eV |
|---|---|---|---|---|---|
| HYA | 2.46 | 198.1 | 156 a | 10 | 24 |
| 86 | 10 | 29 | |||
| DIA | 2.56 | 173.8 | 95.9 | 10 | 25 |
| 103.8 a | 10 | 29 | |||
| DEA | 3.19 | 187.9 | 146 a | 10 | 23 |
| 104 | 10 | 32 | |||
| ATZ | 4.05 | 216 | 174 a | 10 | 23 |
| 104.1 | 10 | 39 |
| Matrices | Analytes | Average Recoveries (%) a | Intra-Day Precision b (RSD, %) | Inter-Day Precision b (RSD, %) | ||
|---|---|---|---|---|---|---|
| 0.1 (μg/kg) | 0.5 (μg/kg) | 1.0 (μg/kg) | ||||
| Soil | ATZ | 75 (9.1) | 80 (8.0) | 83 (8.5) | 8.4 | 8.9 |
| DIA | 82 (8.2) | 90 (7.7) | 86 (9.5) | 8.6 | 9.1 | |
| DEA | 81 (7.3) | 87 (8.7) | 101 (9.2) | 8.9 | 10.0 | |
| HYA | 75 (9.6) | 80 (7.5) | 84 (8.9) | 9.7 | 10.5 | |
| Surface water | ATZ | 82 (7.9) | 78 (9.2) | 91 (7.9) | 9.3 | 10.4 |
| DIA | 90 (9.0) | 85 (8.6) | 98 (8.1) | 8.9 | 9.2 | |
| DEA | 90 (8.1) | 78 (7.6) | 89 (8.9) | 9.0 | 9.5 | |
| HYA | 94 (8.0) | 79 (9.4) | 106 (9.0) | 9.4 | 10.3 | |
| Groundwater | ATZ | 77 (9.0) | 85 (7.6) | 90 (8.3) | 8.7 | 9.2 |
| DIA | 80 (8.2) | 83 (8.0) | 87 (7.8) | 7.8 | 9.4 | |
| DEA | 81 (9.5) | 80 (8.6) | 84 (9.0) | 8.3 | 8.5 | |
| HYA | 82 (9.4) | 81 (7.6) | 90 (7.9) | 8.8 | 9.6 | |
| Method | Instrument | Analytes | Sample | LOD | Recovery (%) | Reference |
|---|---|---|---|---|---|---|
| QuEChERS | HPLC-DAD | ATZ, DEA, DIA | Natural water | 0.08 µg/L | 83~105 | [19] |
| SPE a | GC-MS | ATZ, DEA, DIA | Soil, water | 0.02 µg/kg (water); 1.0 µg/kg (soil) | 72.27~109.68 | [20] |
| SPE | Label-free electrochemical immunosensor | ATZ | Drinking water | 0.011 ng/mL | 95.7~108.4 | [21] |
| Without pretreatment | White light reflectance spectrocscopy | ATZ, paraquat | Water | 50 pg/mL (ATZ); 40 pg/mL (paraquat) | 90~110 | [22] |
| ABS | UFLC-MS | ATZ | Water | 50 ng/L | 70.9~96.5 | [23] |
| Without pretreatment | ELISA; ICG strip | ATZ | Water | 0.01 ng/mL (ELISA); 2 ng/mL (ICG) | 96.5 | [24] |
| LLE b | UPLC-TOF-MS/MS | ATZ | Soil, bacterial strain | 0.01 mg/kg | No report | [25] |
| LLE | LC-MS/MS | ATZ, DEA, DIA, HYA | Soil, water | 0.04 µg/kg | ≥75 | This work |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meng, W.; Wang, D.; Li, S.; Wang, Y.; Jiang, C.; Tian, H.; Ji, M. Residual Characteristics of Atrazine and Its Metabolites in the Liaoning Province of China. Separations 2022, 9, 397. https://doi.org/10.3390/separations9120397
Meng W, Wang D, Li S, Wang Y, Jiang C, Tian H, Ji M. Residual Characteristics of Atrazine and Its Metabolites in the Liaoning Province of China. Separations. 2022; 9(12):397. https://doi.org/10.3390/separations9120397
Chicago/Turabian StyleMeng, Wei, Daifeng Wang, Shizhong Li, Yuying Wang, Ce Jiang, Hongzhe Tian, and Mingshan Ji. 2022. "Residual Characteristics of Atrazine and Its Metabolites in the Liaoning Province of China" Separations 9, no. 12: 397. https://doi.org/10.3390/separations9120397
APA StyleMeng, W., Wang, D., Li, S., Wang, Y., Jiang, C., Tian, H., & Ji, M. (2022). Residual Characteristics of Atrazine and Its Metabolites in the Liaoning Province of China. Separations, 9(12), 397. https://doi.org/10.3390/separations9120397





