Hydrophobic Natural Eutectic Solvents for the Gas Chromatographic Determination of Suspected Allergens in Fragrances by Dispersive Liquid-Liquid Microextraction
Abstract
:1. Introduction
2. Materials and Methods
2.1. Samples and Chemicals
2.2. Instrument Set-Up
2.3. Eutectic Solvents Preparation
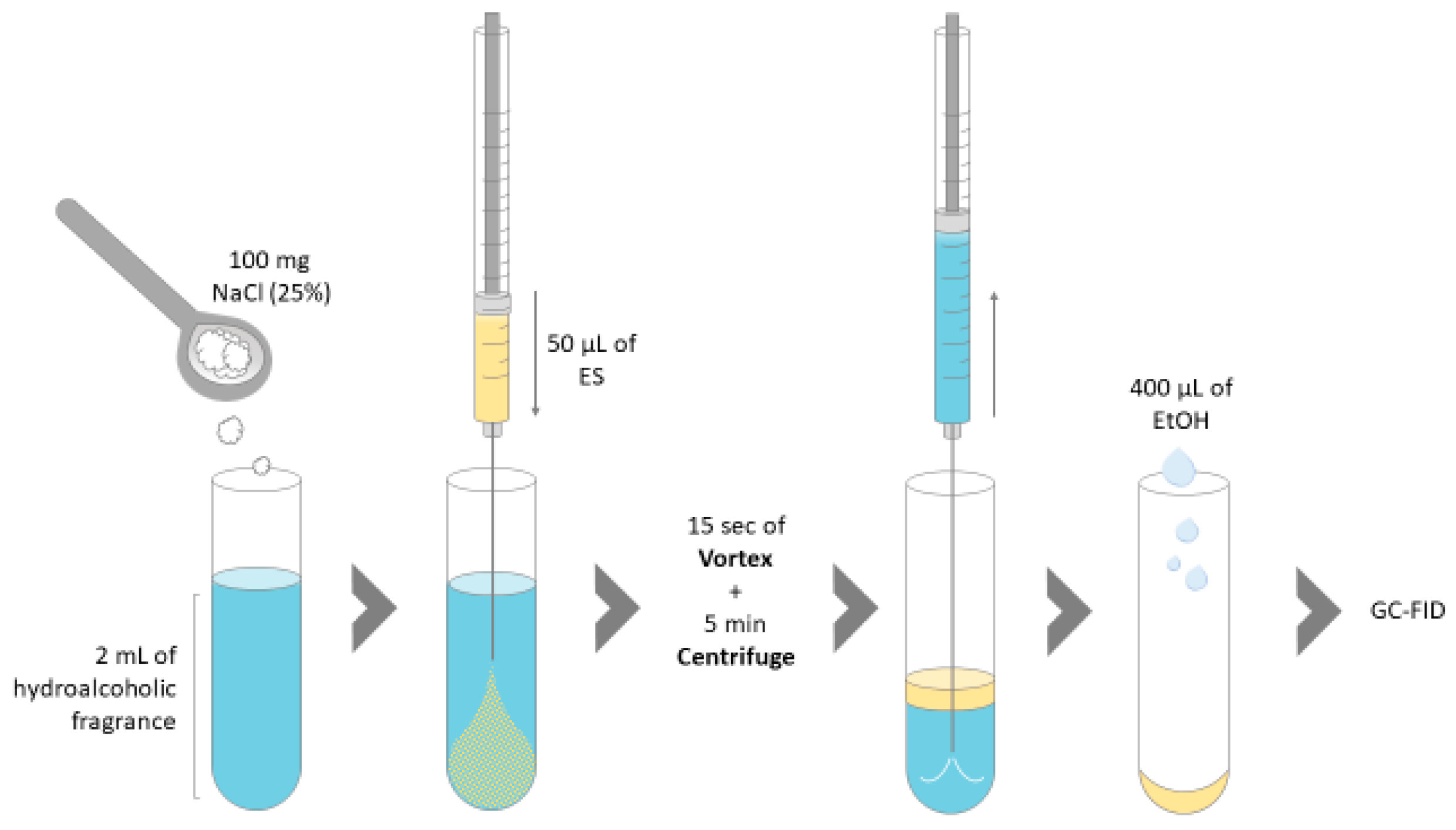
2.4. Dispersive Liquid-Liquid Microextraction with HES
2.5. Statistical Analysis
3. Results and Discussion
3.1. Screening of Different Hydrophobic Eutectic Solvents
3.2. Optimization of the Dispersive Liquid-Liquid Microextraction Method
3.2.1. Ultrasound Step
3.2.2. Ionic Strength
3.2.3. HES Volume Effect
3.2.4. Extraction Time Effect
3.3. Analytical Performance for the HES-Based DLLME Method
3.4. Analysis of Real Samples and Comparison with Direct Injection
3.5. Comparison with Previously Reported Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- International Fragrance Association (IFRA). Available online: https://ifrafragrance.org/priorities/ingredients/ifra-transparency-list (accessed on 14 September 2022).
- Bickers, D.R.; Calow, P.; Greim, H.A.; Hanifin, J.M.; Rogers, A.E.; Saurat, J.-H.; Glenn Sipes, I.; Smith, R.L.; Tagami, H. The safety assessment of fragrance materials. Regul. Toxicol. Pharmacol. 2003, 37, 218–273. [Google Scholar] [CrossRef]
- European Union, Directive 2003/15/EC of the European Parlaiment and of The Council of 27 February 2003. Available online: https://eur-lex.europa.eu/legal-content/IT/TXT/?uri=CELEX:32003L0015 (accessed on 14 September 2022).
- Sanchez-Prado, L.; Lamas, J.P.; Alvarez-Rivera, G.; Lores, M.; Garcia-Jares, C.; Llompart, M. Determination of suspected fragrance allergens in cosmetics by matrix solid-phase dispersion gas chromatography–mass spectrometry analysis. J. Chromatogr. A 2011, 1218, 5055–5062. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Begnaud, F.; Chaintreau, A.; Pawliszyn, J. Quantification of perfume compounds in shampoo using solid-phase microextraction. Flavour Fragr. J. 2006, 21, 822–832. [Google Scholar] [CrossRef]
- David, F.; Devos, C.; Joulain, D.; Chaintreau, A.; Sandra, P. Determination of suspected allergens in non-volatile matrices using PTV injection with automated liner exchange and GC-MS. J. Sep. Sci. 2006, 29, 1587–1594. [Google Scholar] [CrossRef] [PubMed]
- Desmedt, B.; Canfyn, M.; Pype, M.; Baudewyns, S.; Hanot, V.; Courselle, P.; De Beer, J.O.; Rogiers, V.; De Paepe, K.; Deconinck, E. HS–GC–MS method for the analysis of fragrance allergens in complex cosmetic matrices. Talanta 2015, 131, 444–451. [Google Scholar] [CrossRef]
- Cooper, J.; Jones, M.; Duban, S. Fast Analysis of Cosmetic Allergens Using UltraPerformance Convergence Chromatography (UPC2) with MS Detection- Application Note. Available online: https://www.waters.com/waters/library.htm?cid=511436&lid=134878656 (accessed on 14 September 2022).
- Devos, C.; Ochiai, N.; Sasamoto, K.; Sandra, P.; David, F. Full evaporation dynamic headspace in combination with selectable one-dimensional/two-dimensional gas chromatography–mass spectrometry for the determination of suspected fragrance allergens in cosmetic products. J. Chromatogr. A 2012, 1255, 207–215. [Google Scholar] [CrossRef]
- Liu, Q.; Zenobi, R. Rapid analysis of fragrance allergens by dielectric barrier discharge ionization mass spectrometry. Rapid Commun. Mass Spectrom. 2021, 35, e9021. [Google Scholar] [CrossRef]
- Celeiro, M.; Garcia-Jares, C.; Llompart, M.; Lores, M. Recent Advances in Sample Preparation for Cosmetics and Personal Care Products Analysis. Molecules 2021, 26, 4900. [Google Scholar] [CrossRef]
- Schettino, L.; Peris-Pastor, G.; Benedé, J.L.; Chisvert, A. A comprehensive review on the use of microextraction techniques in the analysis of cosmetic products. Adv. Sample Prep. 2022, 3, 100024. [Google Scholar] [CrossRef]
- Kuhn, E.R. Water injections in GC—How wet can you get? LC-GC N. Am. 2002, 20, 474–478. [Google Scholar]
- Mastellone, G.; Marengo, A.; Sgorbini, B.; Rubiolo, P.; Cagliero, C. New phases for analytical scale extraction from plants: Current and future trends. TrAC Trends Anal. Chem. 2021, 141, 116288. [Google Scholar] [CrossRef]
- Ivanović, M.; Islamčević Razboršek, M.; Kolar, M. Innovative Extraction Techniques Using Deep Eutectic Solvents and Analytical Methods for the Isolation and Characterization of Natural Bioactive Compounds from Plant Material. Plants 2020, 9, 1428. [Google Scholar] [CrossRef] [PubMed]
- Davoodi, R.; Nodehi, R.N.; Rastkari, N.; Zinatizadeh, A.A.; Mahvi, A.H.; Fattahi, N. Solid-phase extraction followed by deep eutectic solvent based dispersive liquid–liquid microextraction and GC-MS detection of the estrogenic compounds in wastewater samples. New J. Chem. 2020, 44, 9844–9851. [Google Scholar] [CrossRef]
- Nie, J.; Yu, G.; Song, Z.; Wang, X.; Li, Z.; She, Y.; Lee, M. Microwave-assisted deep eutectic solvent extraction coupled with headspace solid-phase microextraction followed by GC-MS for the analysis of volatile compounds from tobacco. Anal. Methods 2017, 9, 856–863. [Google Scholar] [CrossRef]
- Makoś, P.; Fernandes, A.; Przyjazny, A.; Boczkaj, G. Sample preparation procedure using extraction and derivatization of carboxylic acids from aqueous samples by means of deep eutectic solvents for gas chromatographic-mass spectrometric analysis. J. Chromatogr. A 2018, 1555, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Mastellone, G.; Marengo, A.; Sgorbini, B.; Rubiolo, P.; Cagliero, C. Development of a dispersive solid-liquid microextraction method using natural eutectic solvents for a greener extraction of phytochemicals from fiber-type Cannabis sp. Ind. Crops Prod. 2022, 187, 115476. [Google Scholar] [CrossRef]
- Boateng, I.D. A Critical Review of Emerging Hydrophobic Deep Eutectic Solvents’ Applications in Food Chemistry: Trends and Opportunities. J. Agric. Food Chem. 2022, 70, 11860–11879. [Google Scholar] [CrossRef] [PubMed]
- Makoś, P.; Przyjazny, A.; Boczkaj, G. Hydrophobic deep eutectic solvents as “green” extraction media for polycyclic aromatic hydrocarbons in aqueous samples. J. Chromatogr. A 2018, 1570, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Farooq, M.Q.; Abbasi, N.M.; Anderson, J.L. Deep eutectic solvents in separations: Methods of preparation, polarity, and applications in extractions and capillary electrochromatography. J. Chromatogr. A 2020, 1633, 461613. [Google Scholar] [CrossRef]
- European Pharmacopoeia, 10th ed. Available online: https://www.edqm.eu/en/web/edqm/european-pharmacopoeia-ph-eur-10th-edition- (accessed on 14 September 2022).
- López-Lorente, Á.I.; Pena-Pereira, F.; Pedersen-Bjergaard, S.; Zuin, V.G.; Ozkan, S.A.; Psillakis, E. The ten principles of green sample preparation. TrAC Trends Anal. Chem. 2022, 148, 116530. [Google Scholar] [CrossRef]
- Wojnowski, W.; Tobiszewski, M.; Pena-Pereira, F.; Psillakis, E. AGREEprep—Analytical greenness metric for sample preparation. TrAC Trends Anal. Chem. 2022, 149, 116553. [Google Scholar] [CrossRef]
- Pérez-Outeiral, J.; Millán, E.; Garcia-Arrona, R. Ultrasound-assisted emulsification microextraction coupled with high-performance liquid chromatography for the simultaneous determination of fragrance allergens in cosmetics and water. J. Sep. Sci. 2015, 38, 1561–1569. [Google Scholar] [CrossRef] [PubMed]


| HBA a | HBD b | Molar Ratio |
|---|---|---|
| Thymol | Carvacrol | 1:1 |
| Thymol | Eugenol | 1:1 |
| Terpinen-4-ol | Menthol | 1:1 |
| Anethole | Menthol | 1:1 |
| Thymol | Cumarine | 1:1 |
| Neral | Geranial | Citronellol | Geraniol | Hydroxycitronellal | Linalool | |
|---|---|---|---|---|---|---|
| HES-based DLLME method | ||||||
| Investigated linear range (mg/L) | 0.5–80 | 0.5–120 | 0.5–200 | 0.5–200 | 0.5–200 | 0.5–200 |
| Calibration equation | y = 4304.5x + 3980.8 | y = 4710.2x + 4577.4 | y = 5537.8x − 9534.2 | y = 5618.7x + 3673.4 | y = 3588.3x − 6788.9 | y = 6383.8x − 7117.3 |
| Linearity (R2) | 0.994 | 0.997 | 0.986 | 0.994 | 0.996 | 0.996 |
| LOD (mg/L) | 0.2 | 0.2 | 0.2 | 0.2 | 0.2 | 0.2 |
| LOQ (mg/L) | 0.5 | 0.5 | 0.5 | 0.5 | 0.5 | 0.5 |
| Repeatability 1 mg/L (% RSD), n = 3 | 7.5 | 4.6 | 16.4 | 7.2 | 2.5 | 3.6 |
| Enrichment factor a | 3.2 | 3.7 | 3.5 | 3.5 | 3.0 | 3.9 |
| Direct injection | ||||||
| Investigated linear range (mg/L) | 2–80 | 2–120 | 2–200 | 2–200 | 2–200 | 2–200 |
| Calibration equation | y = 1343x + 46.441 | y = 1284.1x + 881.02 | y = 1577.5x + 165.08 | y = 1597.9x + 475.17 | y = 1174.9x − 868.18 | y = 1633x + 453.01 |
| Linearity (R2) | 0.998 | 0.997 | 0.997 | 0.995 | 0.995 | 0.997 |
| LOD (mg/L) | 1 | 1 | 1 | 1 | 1 | 1 |
| LOQ (mg/L) | 2 | 2 | 2 | 2 | 2 | 2 |
| Compound | Concentration Measured by DLLME (mg/L) | Precision (% RSD), n = 3 | Relative Recovery (% RR) |
|---|---|---|---|
| Fragrance 1 | |||
| Linalool | 475 ± 5 | 1.1 | 85.2 |
| Citronellol | 390 ± 23 | 6.4 | 86.4 |
| Geraniol | 326 ± 6 | 1.7 | n.c. a |
| Hydroxycitronellal | 389 ± 10 | 3.1 | 84.1 |
| Fragrance 2 | |||
| Linalool | 1256 ± 94 b | 7.7 | 107.3 |
| Citronellol | 22 ± 2 | 7.5 | 100.6 |
| Sample | Extraction Method | Solvent/Sorbent | Volume of Solvent | Analytical Platform | LOD | LOQ a | Reference |
|---|---|---|---|---|---|---|---|
| Fragrances | - b | Methyl pivalate | Several hundreds of mL | GC-MS/FID | - | 2 mg/L | [1] |
| Cosmetic matrices | LLE | Hexane | 10 mL | HS c-GC-MS | - | 8 mg/L | [7] |
| Cosmetic matrices | MSPD d | Hexane/acetone 1:1 Florisil® e (2 g) | 5 mL | GC-MS | 0.02–1 mg/Kg | 0.05–2.5 mg/kg | [4] |
| Cosmetic matrices | - b | Dichloromethane | 10 mL | PTV f-GC-MS with liner packed with PDMS g foam | 5 mg/kg | [6] | |
| Cosmetic matrices | FEDHS h | - | - | GC-MS | - | 10 mg/L | [9] |
| Shampoo | HS-SPME i | PDMS, PA j | - | GC-MS | 0.001–3.0 mg/L | - | [5] |
| Cosmetics and water | USAEME k | 2-dodecanol | 50 µL | HPLC-DAD | 0.001–0.154 mg/L | 0.004–0.463 mg/L | [26] |
| Perfumes | - b | MeOH | - | DBDI l-MS | 0.0001–0.01 mg/L | 0.001–0.05 mg/L | [10] |
| Hydroalcoholic fragrances | DLLME | Natural HES | 50 µL | GC-FID | 0.1 mg/L | 0.5 mg/L | This work |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bechis, G.; Mastellone, G.; Marengo, A.; Sgorbini, B.; Cagliero, C.; Rubiolo, P. Hydrophobic Natural Eutectic Solvents for the Gas Chromatographic Determination of Suspected Allergens in Fragrances by Dispersive Liquid-Liquid Microextraction. Separations 2022, 9, 318. https://doi.org/10.3390/separations9100318
Bechis G, Mastellone G, Marengo A, Sgorbini B, Cagliero C, Rubiolo P. Hydrophobic Natural Eutectic Solvents for the Gas Chromatographic Determination of Suspected Allergens in Fragrances by Dispersive Liquid-Liquid Microextraction. Separations. 2022; 9(10):318. https://doi.org/10.3390/separations9100318
Chicago/Turabian StyleBechis, Gaia, Giulia Mastellone, Arianna Marengo, Barbara Sgorbini, Cecilia Cagliero, and Patrizia Rubiolo. 2022. "Hydrophobic Natural Eutectic Solvents for the Gas Chromatographic Determination of Suspected Allergens in Fragrances by Dispersive Liquid-Liquid Microextraction" Separations 9, no. 10: 318. https://doi.org/10.3390/separations9100318
APA StyleBechis, G., Mastellone, G., Marengo, A., Sgorbini, B., Cagliero, C., & Rubiolo, P. (2022). Hydrophobic Natural Eutectic Solvents for the Gas Chromatographic Determination of Suspected Allergens in Fragrances by Dispersive Liquid-Liquid Microextraction. Separations, 9(10), 318. https://doi.org/10.3390/separations9100318











