An Evaluation of the Equilibrium Properties in Hexane and Ethanol Extractive Systems for Moringa oleifera Seeds and Fatty Acid Profiles of the Extracts
Abstract
:1. Introduction
2. Materials and Methods
2.1. Raw Material and Chemicals
2.2. Raw Material Characterization
2.2.1. Determining Water Content
2.2.2. Non-Extractable and Extractable Material
2.3. Kinetics Experiments
2.4. Determining the Equilibrium Parameters
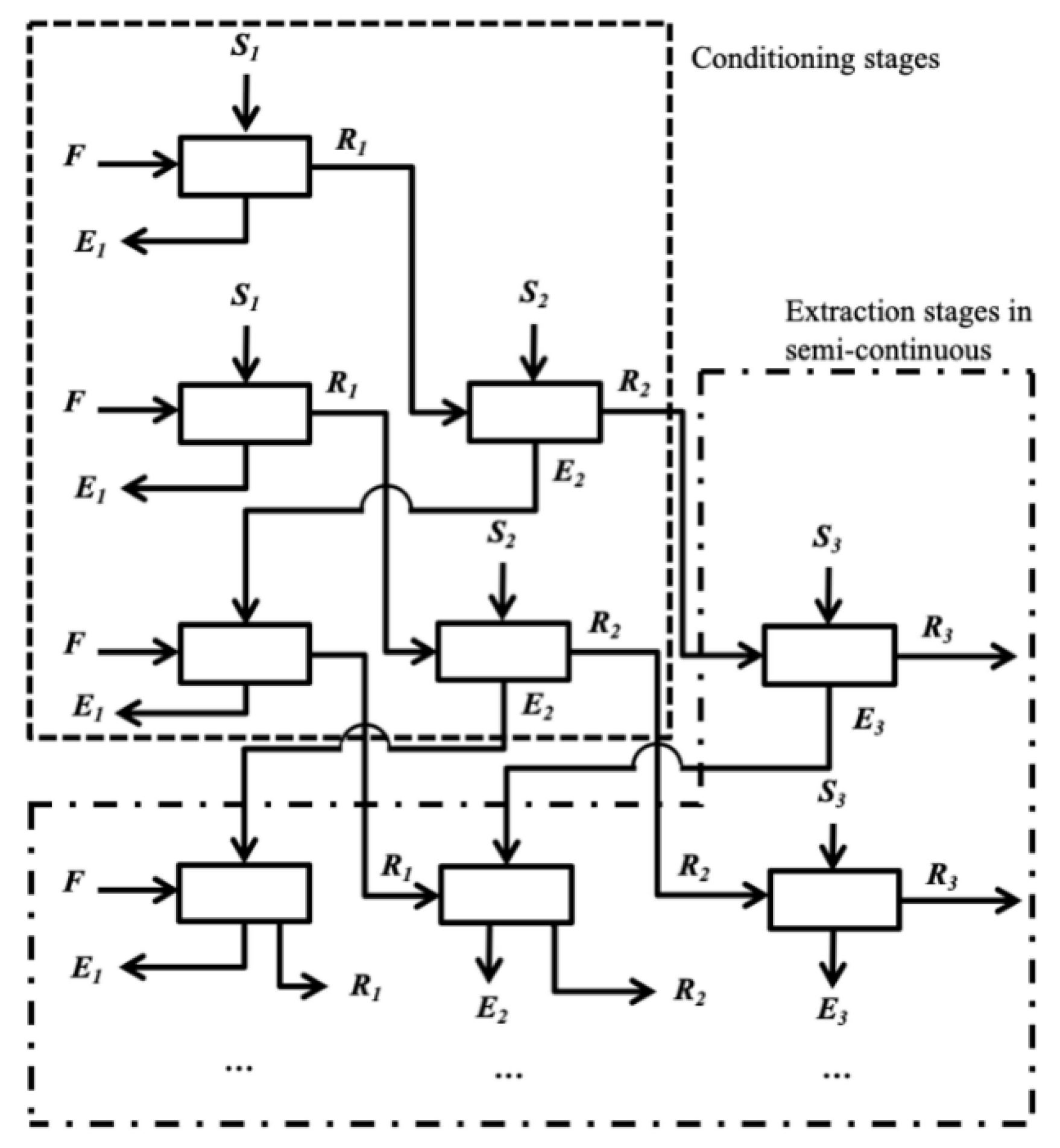
2.5. Modelling of the Counter-Current Multistage Extractions (CME)
2.6. Derivatization of the Fatty Acids
2.7. Fatty Acid Analysis
2.8. Statistical Analysis
3. Results and Discussion
3.1. Initial Composition of the Feed
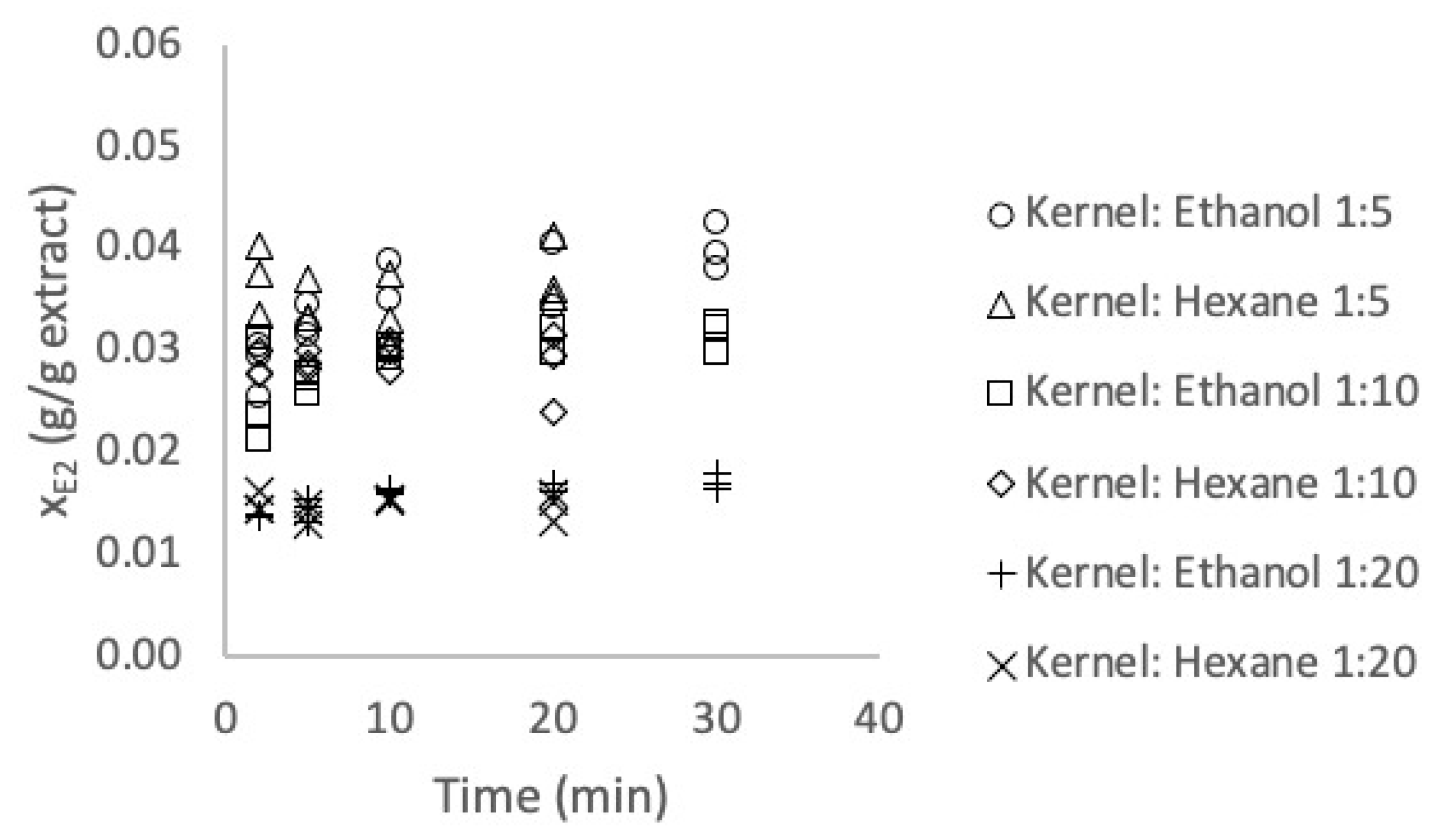
3.2. Extraction Kinetics
3.3. Experimental Evaluation of the Equilibrium Parameters
3.4. CME Simulation and Experimental Results
3.5. Determining Fatty Acid Content
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hamm, W.; Hamilton, R.J.; Calliauw, G. Edible Oil Processing, 2nd ed.; Wiley-Blackwell: West Sussex, UK, 2013; pp. 3–16. [Google Scholar]
- Leone, A.; Spada, A.; Battezati, A.; Schiraldi, A. Moringa oleifera Seeds and Oil: Characteristics and Uses for Human Health. Int. J. Mol. Sci. 2016, 17, 2141. [Google Scholar] [CrossRef] [Green Version]
- Lopez, H. Health effects of oleic acid and long chain omega-3 fatty acids (EPA and DHA) enriched milks. A review of intervention studies. Pharmacol. Res. 2010, 61, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Mat-Yusoff, M.; Gordon, M.H.; Ezeh, O.; Niranjan, K. High pressure pre-treatment of Moringa oleifera seed kernels prior to aqueous enzymatic oil extraction. IFSET 2016, 39, 129–136. [Google Scholar] [CrossRef]
- Abdulkarim, S.M.; Long, K.; Lai, O.M.; Muhammed, S.K.S.; Ghazali, H.M. Some physico-chemical properties of Moringa oleifera seed oil extracted using solvent and aqueous enzymatic methods. Food Chem. 2005, 93, 253–263. [Google Scholar] [CrossRef]
- Ruttarattanamongkol, K.; Siebenhandl-Ehn, S.; Schreiner, M.; Petrasch, A.M. Pilot-scale supercritical carbon dioxide extraction, physico-chemical properties and profile characterization of Moringa oleifera seed oil in comparison with conventional extraction methods. Ind. Crop. Prod. 2014, 58, 68–77. [Google Scholar] [CrossRef]
- Da Porto, C.; Decorti, D.; Natolino, A. Microwave pretreatment of Moringa oleifera seed: Effect on oil obtained by pilot-scale supercritical carbon dioxide extraction and Soxhlet apparatus. J. Supercrit. Fluids 2016, 107, 38–43. [Google Scholar] [CrossRef]
- Zhao, S.; Zhang, D. A parametric study of supercritical carbon dioxide extraction of oil from Moringa oleifera seeds using a response surface methodology. Sep. Purif. Technol. 2013, 113, 9–17. [Google Scholar] [CrossRef]
- Nguyen, H.N.; Gapillo, P.D.; Maridable, J.B.; Malaluan, R.N.; Hinode, H.; Salim, C.; Huynh, H.K.P. Extraction of oil from Moringa oleifera kernels using supercritical carbon dioxide with ethanol for pretreatment: Optimization of the extraction process. Chem. Eng. Process. 2011, 50, 1207–1213. [Google Scholar] [CrossRef]
- Abiola, O.; Atoo, E. Process optimization of mechanical oil expression from Moringa (Moringa oleifera) seeds. Ind. Crop. Prod. 2016, 90, 142–151. [Google Scholar] [CrossRef]
- Mani, S.; Jaya, S.; Vadivambal, R. Optimization of solvent extraction of moringa (Moringa oleifera) seed kernel oil using response surface methodology. Food Bioprod. Process. 2007, 85, 328–335. [Google Scholar] [CrossRef]
- Bhutada, P.R.; Jadhav, A.J.; Pinjari, D.V.; Nemade, P.R. Solvent assisted extraction of oil from Moringa oleifera Lam. seeds. Ind. Crop. Prod. 2017, 82, 74–80. [Google Scholar] [CrossRef]
- Zhong, J.; Wang, Y.; Yang, R.; Liu, X.; Yang, Q.; Qin, X. The application of ultrasound and microwave to increase oil extraction from Moringa oleifera seeds. Ind. Crop. Prod. 2018, 120, 1–10. [Google Scholar] [CrossRef]
- Akinoso, R.; Adeyanju, J.A. Optimization of edible oil extraction from ofada rice bran using response surface methodology. Food Bioproc. Technol. 2012, 5, 1372–1378. [Google Scholar] [CrossRef]
- Castillo-Santos, K.; Aguirre-Alonso, R.O.; Rodríguez-Jimenes, G.C.; Robles-Olvera, V.J.; Salgado-Cervantes, M.A.; García-Alvarado, M.A. An optimization based algorithm for solving design problems of counter-current multistage batch solid–liquid extractors for complex systems: Application to vanilla extract. Comput. Chem. Eng. 2016, 89, 53–61. [Google Scholar] [CrossRef]
- McCabe, W.; Smith, J.; Harriott, P. Unit Operations of Chemical Engineering, 7th ed.; McGraw-Hill: Singapore, 2004; ISBN 9780071127219. [Google Scholar]
- Vázquez-León, L.A.; Olguín-Rojas, J.A.; Páramo-Calderón, D.E.; Palma, M.; Barbero, G.F.; Robles-Olvera, V.J.; García-Alvarado, M.A.; Rodríguez-Jimenes, G.C. Modeling of counter-current multistage extractionof Moringa oleifera leaves using a mechanistic model. Food Bioprod. Process. 2019, 115, 165–174. [Google Scholar] [CrossRef]
- Seth, S.; Agrawal, Y.C.; Ghosh, P.K.; Jayas, D.S. Effect of moisture content on the quality of soybean oil and meal extracted by isopropyl alcohol and hexane. Food Bioproc. Technol. 2010, 3, 121. [Google Scholar] [CrossRef]
- Akemi, T.; Megumi, M.; Rodrigues, C.E.C. Kinetics of soybean oil extraction using ethanol as solvent: Experimental data and modeling. Food Bioprod. Process. 2016, 98, 1–10. [Google Scholar] [CrossRef]
- Baümler, E.R.; Carrín, M.E.; Carelli, A.A. Extraction of sunflower oil using ethanol as solvent. J. Food Eng. 2016, 178, 190–197. [Google Scholar] [CrossRef]
- Bessa, L.C.B.A.; Ferreira, M.C.; Rodrigues, C.E.C.; Batista, E.A.C.; Meirelles, A.J.A. Simulation and process design of continuous countercurrent ethanolic extraction of rice bran oil. J. Food Eng. 2017, 202, 99–113. [Google Scholar] [CrossRef]
- Colivet, J.; Oliveira, A.L.; Carvalho, R.A. Influence of the bed height on the kinetics of watermelon seed oil extraction with pressurized ethanol. Sep. Purif. Technol. 2016, 169, 187–195. [Google Scholar] [CrossRef]
- Oladipo, B.; Betiku, E. Process optimization of solvent extraction of seed oil from Moringa oleifera: An appraisal of quantitative and qualitative process variables on oil quality using D-optimal design. Biocatal. Agric. Biotechnol. 2019, 20, 1–12. [Google Scholar] [CrossRef]
- Turgut, N.; Zhang, M. Pressurized solvent extraction of wheat germ oil. Int. Food Res. J. 2003, 36, 905–909. [Google Scholar] [CrossRef]
- Navarro, S.L.B.; Capellini, M.C.; Aracava, K.K.; Rodrigues, C.E.C. Corn germ-bran oils extracted with alcoholic solvents: Extraction yield, oil composition and evaluation of protein solubility of defatted meal. Food Bioprod. Process. 2016, 100, 185–194. [Google Scholar] [CrossRef]
- Capellini, M.C.; Giacomini, V.; Cuevas, M.S.; Rodrigues, C.E.C. Rice bran oil extraction using alcoholic solvents: Physicochemical characterization of oil and protein fraction functionality. Ind. Crop. Prod. 2017, 104, 133–143. [Google Scholar] [CrossRef]
- Ferreyra, R.; Sellés, G.; Saavedra, J.; Ortíz, J.; Zúñiga, C.; Troncoso, C.; Rivera, S.A.; González, M.; Defilippi, B.G. Identification of pre-harvest factors that affect fatty acid profiles of avocado fruit (Persea americana Mill) cv. ‘Hass’ at harvest. S. Afr. J. Bot. 2016, 105, 15–20. [Google Scholar] [CrossRef]
- Barthet, V.J. (n-7) and (n-9) cis-monounsaturated fatty acid contents. Phytochemistry 2008, 69, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Vlahov, G.; Chepkwony, P.K.; Ndalut, P.K. 13C NMR Characterization of Triacylglycerols of Moringa oleifera Seed Oil: An “Oleic-Vaccenic Acid” Oil. J. Agric. Food Chem. 2002, 50, 970–975. [Google Scholar] [CrossRef]
- Sampaio, O.Z.; Caldas, E.A.; Almeida, A.J. The employment of ethanol as solvent to extract Brazil nut oil. J. Clean. Prod. 2018, 180, 866–875. [Google Scholar] [CrossRef]
- Baümler, E.R.; Carrín, M.E.; Careli, A.A. Diffusion of tocopherols, phospholipids and sugars during oil extraction from sunflower collets using ethanol as solvents. J. Food Eng. 2017, 194, 1–8. [Google Scholar] [CrossRef]

| Sample | Particle Size (mm) | Temperature (°C) | Total Extractable Material (g/g) | Water (g/g) | |
|---|---|---|---|---|---|
| Ethanol | Hexane | ||||
| Kernel | <0.85 | 40 | 0.424 ± 0.010 | 0.412 ± 0.014 | 0.039 ± 0.002 |
| <0.85 | 50 | 0.509 ± 0.019 | 0.424 ± 0.041 | 0.025 ± 0.001 | |
| Kernel | 1–4.5 | 50 | 0.270 ± 0.014 | 0.440 ± 0.013 | 0.035 ± 0.002 |
| Seed | <0.85 | 40 | 0.331 ± 0.011 | 0.281 ± 0.011 | 0.036 ± 0.001 |
| <0.85 | 50 | 0.410 ± 0.007 | 0.306 ± 0.046 | 0.027 ± 0.002 | |
| Temperature | Sample | Particle Size (mm) | Hexane | Ethanol | ||
|---|---|---|---|---|---|---|
| Keq | M | Keq | M | |||
| 40 °C | Kernel | <0.85 | 0.170 | 1.64 ± 0.25 | 0.110 | 2.58 ± 0.27 |
| Seed | <0.85 | 0.200 | 2.28 ± 0.22 | 0.200 | 3.72 ± 0.30 | |
| 50 °C | Kernel | <0.85 | 0.130 | 1.73 ± 0.19 | 0.100 | 3.15 ± 0.44 |
| Kernel | 1–4.5 | 0.206 | 0.96 ± 0.14 | 0.381 | 1.017 ± 0.06 | |
| Seed | <0.85 | 0.200 | 2.25 ± 0.18 | 0.120 | 3.90 ± 0.21 | |
| Solvent | Stages | Solvent (g) | ||||
|---|---|---|---|---|---|---|
| Predicted | Experimental | Simulated | Experimental | |||
| Ethanol | 1 | 50 | 45.39 | 44.64 ± 0.12 | 0.0441 | 0.0364 ± 0.0011 |
| 2 | 69.5 | 65.53 | 63.86 ± 0.30 | 0.0441 | 0.0389 ± 0.0004 | |
| 3 | 74.4 | 70.63 | 68.49 ± 0.93 | 0.0441 | 0.0413 ± 0.0003 | |
| Hexane | 1 | 50 | 47.26 | 44.90 ± 0.57 | 0.0497 | 0.0513 ± 0.0022 |
| 2 | 76.7 | 75.17 | 69.86 ± 1.33 | 0.0497 | 0.0431 ± 0.0031 | |
| 3 | 84.8 | 83.66 | 76.55 ± 0.26 | 0.0497 | 0.0475 ± 0.0028 | |
| Fatty Acid (%) | KE40 | KE50 | KH40 | KH50 | SE40 | SE50 | SH40 | SH50 |
|---|---|---|---|---|---|---|---|---|
| Caprilic | <0.1 b | <0.1 a | <0.1 b | <0.1 b | ND | <0.1 b | <0.1 b | ND |
| Myristic | 0.4 a | 0.3 a | 0.4 a | 0.4 a | 0.3 a | 0.4 a | 0.4 a | 0.4 a |
| Palmitic | 18.3 a | 19.2 a | 17.5 a | 18.0 a | 20.1 a | 17.9 a | 17.8 a | 17.9 a |
| Palmitoleic | 1.5 a | 1.0 a | 1.3 a | 1.4 a | 1.3 a | 1.3 a | 1.3 a | 1.3 a |
| Margaric | 0.2 a | 0.1 a | 0.3 a | 0.2 a | 0.2 a | 0.2 a | 0.3 a | 0.2 a |
| Estearic | 13.8 a | 14.1 a | 14.6 a | 14.6 a | 14.5 a | 13.9 a | 14.7 a | 14.6 a |
| Oleic | 58.9 a | 58.9 a | 59.4 a | 58.5 a | 58.3 a | 59.1 a | 58.7 a | 59.0 a |
| Not identified | 6.3 a | 5.9 a | 6.0 a | 6.4 a | 4.4 a | 6.5 a | 6.4 a | 6.2 a |
| Linoleic | 0.6 a | 0.5 a | 0.5 a | 0.6 a | 0.7 a | 0.6 a | 0.6 a | 0.6 a |
| ɣ-linolenic | <0.1 b | <0.1 a | ND | <0.1 c | <0.1 a | <0.1 bc | <0.1 bc | <0.1 a |
| Linolenic | <0.1 b | <0.1 a | ND | <0.1 c | <0.1 a | <0.1 bc | <0.1 bc | <0.1 a |
| Arachidic | ND | <0.1 a | <0.1 a | ND | <0.1 a | <0.1 a | <0.1 a | <0.1 a |
| Behenic | <0.1 a | ND | <0.1 a | <0.1 a | ND | <0.1 a | <0.1 a | <0.1 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arguelles-Peña, K.; Olguín-Rojas, J.A.; Acosta-Osorio, A.A.; Carrera, C.; Barbero, G.F.; García-Alvarado, M.Á.; Rodríguez-Jimenes, G.d.C. An Evaluation of the Equilibrium Properties in Hexane and Ethanol Extractive Systems for Moringa oleifera Seeds and Fatty Acid Profiles of the Extracts. Separations 2021, 8, 217. https://doi.org/10.3390/separations8110217
Arguelles-Peña K, Olguín-Rojas JA, Acosta-Osorio AA, Carrera C, Barbero GF, García-Alvarado MÁ, Rodríguez-Jimenes GdC. An Evaluation of the Equilibrium Properties in Hexane and Ethanol Extractive Systems for Moringa oleifera Seeds and Fatty Acid Profiles of the Extracts. Separations. 2021; 8(11):217. https://doi.org/10.3390/separations8110217
Chicago/Turabian StyleArguelles-Peña, Karina, José Arturo Olguín-Rojas, Andrés Antonio Acosta-Osorio, Ceferino Carrera, Gerardo F. Barbero, Miguel Ángel García-Alvarado, and Guadalupe del Carmen Rodríguez-Jimenes. 2021. "An Evaluation of the Equilibrium Properties in Hexane and Ethanol Extractive Systems for Moringa oleifera Seeds and Fatty Acid Profiles of the Extracts" Separations 8, no. 11: 217. https://doi.org/10.3390/separations8110217
APA StyleArguelles-Peña, K., Olguín-Rojas, J. A., Acosta-Osorio, A. A., Carrera, C., Barbero, G. F., García-Alvarado, M. Á., & Rodríguez-Jimenes, G. d. C. (2021). An Evaluation of the Equilibrium Properties in Hexane and Ethanol Extractive Systems for Moringa oleifera Seeds and Fatty Acid Profiles of the Extracts. Separations, 8(11), 217. https://doi.org/10.3390/separations8110217








